Abstract
Non-photochemical quenching (NPQ) of the excited state of chlorophyll a is a major photoprotective mechanism plants utilize to survive under high light. Here, we report the impact of long-term light quality treatment on photosynthetic properties, especially NPQ in rice. We used three LED-based light regimes, i.e., red (648–672 nm), blue (438–460 nm), and “warm” white light (529–624 nm), with the incident photon flux density of 300 µmol photons m−2 s−1, the difference in the absorbed photon flux densities by leaves grown under different light quality being less than 7%. Our results show that blue light, as compared to white light, induced a significant decrease in Fv/Fm, a decreased rate of reduction of P700+ after P700 was completely oxidized; furthermore, blue light also induced higher NPQ with an increased initial speed of NPQ induction, which corresponds to the qE component of NPQ, and a lower maximum quantum yield of PSII, i.e., Y(II). In contrast, rice grown under long-term red light showed decreased Y(II) and increased NPQ, but with no change in Fv/Fm. Furthermore, we found that rice grown under either blue or red light showed decreased transcript abundance of both catalase and ascorbate peroxidase, together with an increased H2O2 content, as compared to rice grown under white light. All these data suggest that even under a moderate incident light level, rice grown under blue or red light led to compromised antioxidant system, which contributed to decreased quantum yield of photosystem II and increased NPQ.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Light influences photosynthesis through multiple mechanisms. Most importantly, light absorbed by photosynthetic pigments is the source of energy for photosynthesis. The wavelength of light is a major factor influencing both the development of photosynthetic machinery and the efficiency of photosynthesis. As compared to plants grown under red light (660–670 nm), those grown under blue light (430–480 nm) exhibit higher chlorophyll (Chl) a/b ratio, higher amount of antenna Chl in their light-harvesting complex II (LHCII), higher electron transfer activity per Chl (Leong and Anderson 1984; Leong et al. 1985; Senger and Bauer 1987), and higher ribulose-1, 5-bisphosphate (RuBP) carboxylase/oxygenase (Rubisco) activity per unit leaf area (Bukhov et al. 1995). Further, Cucumis sativus (cucumber) plants, grown under red light alone, as compared to those grown under red + blue light, show a decrease in the maximum quantum yield of photosystem II (PSII), as measured by variable to maximum chlorophyll (Chl) fluorescence, Fv/Fm, a relatively low rate of CO2 fixation, and a slow response of stomata to light (Hogewoning et al. 2010).
Photon flux densities (i.e., light intensities) influence photosynthesis dramatically (see e.g., Norcini et al. 1991). When plants are grown under low light, the antenna size of the photosystems increases to better capture (and transfer) the excitation energy to the photosynthetic reaction centers (Green and Parson 2003). However, when plants receive too much light, the excess excitation energy is dissipated through non-photochemical quenching (NPQ) of the excited state of Chl a in the form of heat (Demmig-Adams et al. 2014; Ort 2001). Under such a condition, plants may experience damage to PSII by reactive oxygen species (ROS) (Asada 2006), either on the (electron) donor or the (electron) acceptor side of PSII (Pospíšil 2009, 2012). When the donor side is damaged, the oxygen evolving complex (OEC) cannot efficiently reduce the reaction center Chl of PSII (P680+), which can further oxidize other intermediates. In addition, the donor-side inhibition is often associated with the formation of hydrogen peroxide (H2O2), which can be oxidized to the superoxide radical (O2•−), or reduced to the hydroxyl radical (HO•). However, inhibition on the PSII acceptor side leads to reverse electron transfer from the reduced QA (QA•−) to pheophytin, Pheo, leading, ultimately, to the formation of excited triplet state of P680 (3P680*), which in turn may interact with the triplet ground state of molecular oxygen (3O2) forming damaging singlet oxygen (1O2) (Alboresi et al. 2011; Pospíšil and Prasad 2014; Triantaphylides and Havaux 2009).
To cope with the deleterious effects of high-light stress, plants have developed a number of photoprotection responses: (1) leaf movement at the whole plant level (Björkman and Powles 1987; Koller 1990); (2) chloroplast movement at the cellular level (Cazzaniga et al. 2013; Kagawa and Wada 2000; Sztatelman et al. 2010); and (3) non-radiative dissipation of energy as heat (through the NPQ process) at the molecular level (Derks et al. 2015; Müller et al. 2001). There are different components of NPQ that can be distinguished according to their induction and relaxation kinetics following light exposure: (1) a major and fast component, which is associated with the build-up of the trans-thylakoid ΔpH gradient (qE); (2) a slower component, which relaxes within seconds to minutes and is related to state transitions (qT); and (3) the slowest component, which relaxes from minutes to hours and is due to photoinhibition of photosynthesis (qI) (Leonelli et al. 2017; Papageorgiou and Govindjee 2014; Rochaix 2014; Ruban 2018).
NPQ has been considered to be a prime target to increase light use efficiency under fluctuating light and extensive research has been devoted to study the mechanism of NPQ (Demmig-Adams et al. 2014). Zhu et al. (2004) proposed that increase in the speed of recovery of NPQ from high light to low light may improve photosynthetic efficiency and increase biomass under dynamic light conditions. This hypothesis was tested by Kromdijk et al. (2016) when they observed that plants with accelerated recovery of NPQ showed increased biomass production in the field. Furthermore, the effect of different light intensity on NPQ has been thoroughly investigated (Belgiol et al. 2018; Miyake et al. 2005; Tian et al. 2017). In addition, Oguchi et al. (2011) have observed different extent of photoinhibition, represented by the decrease of the maximal quantum yield of PSII (Fv/Fm), in plants grown under different colors of light, but with the same incident photon flux density. However, the potential impact of light quality on NPQ has been relatively less studied. Understanding the impact of light spectrum on photosynthetic properties can not only help better understand growth of plants in the field, but also help design better lighting system for greenhouses to obtain optimal plant performance. On this aspect, earlier studies have shown that leaves under lower layers of a canopy experience more green light proportionally, as compared to those at the top layer of that canopy (Grant 1997). Many LED-based lighting systems are used in greenhouse industry, which also requires a thorough understanding of responses of plants grown under different light spectra to enable better lighting design. This work aims to investigate the impact of different wavelengths of light with the same incident PFD on photosynthetic performances. Specifically, we grew rice (Oryza sativa) under red (peak at 662 nm), blue (peak at 447 nm), or “warm” white (peak at 568 nm) LED light conditions and examined changes in light reaction-related parameters. We show that under an incident light level of 300 µmol photons m−2 s−1, which is a moderate light intensity for rice, there is a significant decrease of quantum yield of PSII and increase in NPQ when rice was grown under monochromatic red or blue light, as compared to plants grown under white light conditions. We also show that these observed effects are mainly due to the light quality rather than the very small (7%) difference in the PPFD absorbed by leaves in different treatments.
Materials and methods
Growth conditions
We used three different colors of light (one for each light-controlled chamber) using a custom design LED light source, i.e., blue light (peak at 447 nm), red light (peak at 662 nm), and “warm” white light (a broad band, with a peak at 568 nm) (Fig. 1a and Table S1). Light intensities were measured with a photosynthetic active radiation (PAR) meter, and the photosynthetic photon flux density (PPFD), at the top of plants, was maintained, for all the three light regimes, at ~ 300 µmol photons m−2 s−1. First, seeds were germinated in Petri dishes on wet filter paper at ~ 27 °C in the dark for 5 days. After that, the germinated seeds were kept for another 5 days in Petri dishes under a single LED light (blue, red or white), and then they were transplanted into plastic pots (6 L volume) containing commercial peat soil (Pindstrup Substrate no. 4). Rice (O. sativa, cv. XS134) was grown for ~ 50 days in three different light-controlled chambers for 14 h L/10 h D photoperiod and at ~ 27 °C. For the data shown in Fig. 4b, we first grew plants under either red or blue light during the germination stage, and then transferred plants to white light. The plants were grown for ~ 50 days with a 14 h L/10 h D photoperiod under this white light. A measurement representing a mean of data from six plants at each condition was taken for a randomized design of growth conditions.
Light spectrum and photon flux density (PFD) absorbed by leaves. a Spectral scans of the incident PFD used for plant growth: red (peak at 662 nm), blue (peak at 447 nm) and “warm” white (peak at 568 nm) LED light. Inset Estimated absorbed PFD for leaves grown under different light regimes. Bars represent average ± sd of five independent experiments. b Leaf % absorptance (absorbance) spectrum, each curve represents the average of five independent measurements
Lighting system
Three different LEDs were used to obtain blue (438–460 nm; peak at 447 nm), red (648–672 nm; peak 662 nm), and warm white light (a broad band 529–624 nm with a peak at 568 nm). The transmission spectra for all three LEDs were recorded using an Ocean Optics QE 65000 spectrometer (Ocean Optics Inc., Dunedin, FL) (Fig. 1a and Table S1). As noted above, the PFD at the top of the plant was maintained at ~ 300 µmol photons m−2 s−1; this was done by adjusting the distance between the LED and the plant canopy. To minimize the potential impact of pot location relative to the LED light on light environments experienced by plants, pots were systematically moved under light source after every 2 days.
Transmission and reflectance spectra of the leaves
The transmission and reflectance spectrum of each leaf was recorded from 200 to 1100 nm, using an Ocean Optics Spectrometer HR2000 (Ocean Optics Inc., Dunedin, FL) (Fig. S1). Absorbance (% absorptance) at each wavelength was calculated after measurement of reflectance and of transmittance using the following formula (Inada 1980; see Fig. 1b):
The absorbed PFD at each wavelength was calculated based on a total PFD between 400 and 700 nm, the spectra of incident light, and the absorption at each wavelength by the leaf. With the calculated PFD at each wavelength, we found that the total absorbed by leaves under blue light, red light, and white light was 275.9 µmol photons m−2 s−1, 268.2 µmol photons m−2 s−1, and 253.5 µmol photons m−2 s−1, respectively (see inset of Fig. 1a) (See details of calculation in Online Appendix—1).
Chlorophyll (Chl) and carotenoid content
The content of Chl and of the carotenoids was determined according to methods described earlier (Holm 1954; Porra et al. 1989). Leaf samples (0.05 g) were incubated overnight with 1 mL 80% acetone at 4 °C, the resulting suspension was centrifuged at 13,000×g for 5 min, and then Chl a, Chl b, and carotenoids were measured using a UV visible spectrophotometer (50 Bio Varian, Varian Inc., Walnut Creek, CA), at 663.6 nm, 645.6 nm, and 440.5 nm, respectively. Concentration (g/g) of pigments was determined from the following equations (Holm 1954; Porra et al. 1989):
Vm represents the volume of supernatant with a unit of mL, and Wt represents the sample weight with a unit of gram.
Maximum quantum yield of PSII (as inferred from F v/F m), slow Chl a fluorescence induction (PSMT) and transmission changes at 820 nm (i 820)
Variable (Fv = Fm − Fo; Fo is the “O” level) to maximum fluorescence (Fm, the “P” level), Fv/Fm, the fluorescence transient, the PSMT (P is for the peak; S is for a steady state, M is for a maximum, and T is for terminal steady state) phases of fluorescence, and the transmission change at 820 nm, I820, were measured using the Multifunctional Plant Efficiency Analyser (M-PEA) (Hansatech, King Lynn, Norfolk, UK). Plants were first kept overnight at 24 °C in darkness before measurements. Then, after a further 10-min dark adaptation, the detached uppermost fully expanded leaves were exposed for 0.5 s to saturating (5000 µmol photons m−2 s−1) orange–red (625 nm) actinic light for Fv/Fm measurements. Simultaneously, 820 nm modulated light was provided by a LED lamp for I820 measurements. During the measurements, upon light excitation, the P700 is first oxidized then gradually reduced due to the faster transfer of electrons from P700 than the slower arrival of electrons from PSII or cyclic electron flow around PSI. After a 10-min dark adaptation, the same leaves were exposed to 300 s of low intensity (100 µmol photons m−2 s−1) orange–red (625 nm) actinic light for the PSMT measurement. Measurements were repeated six times for each light condition. The maximum quantum yield of PSII was determined by
where Fv is variable fluorescence; Fm is fluorescence at the P level, and Fo is fluorescence at the O level.
Fluorescence quenching analysis
Chl a fluorescence quenching analysis was done using a Dual-PAM-100 instrument (Walz, Effeltrich, Germany). Plants were first kept overnight at 24 °C in darkness, and then after a 10-min additional dark adaptation period, the upper side of detached leaves was exposed to weak modulated measuring light, obtained from a 620 nm LED. This provided the Fo (the O level) values, the initial minimum value for fluorescence. A saturating light pulse of 500 ms duration and 20,000 µmol photons m−2 s−1 light from 620 nm LED arrays was given to the sample to obtain Fm, the maximum fluorescence value (equivalent to the P level). After the first saturating pulse, a blue actinic light of 815 µmol photons m−2 s−1 from 460 nm LED arrays was switched on for 10 min followed by a 10-min dark period. During this time, a saturating pulse of light was applied every 20 s to measure Fm′. In order to obtain Fo′, blue actinic light was switched off and far red light (720 nm) was given. The effective PSII quantum yield, Y(II), was calculated as described previously (Genty et al. 1989) by the formula:
Furthermore, the quantum yield of regulated energy dissipation Y(NPQ) and quantum yield of non-regulated energy dissipation in PSII Y(NO) were calculated as described previously (Kramer et al. 2004):
where NPQ is non-photochemical quenching and qL is the coefficient of photochemical quenching (Klughammer and Schreiber 2008; Pfündel et al. 2008).
Detection of hydrogen peroxide (H2O2)
Mature plants grown under sunlight were acclimated to red, blue or white LED light at around 800 µmol photons m−2 s−1 for 7 days. Then, the detached leaves, from each light condition, were infiltrated with 5 mM of 3, 3′diaminobenzidine (DAB) at pH 3.8, and incubated in dark for 2 h. After that, the upper side of the detached leaves was exposed to high intensity (1400 µmol photons m−2 s−1) white light for 60 min. Finally, Chl was extracted from the leaves prior to imaging by boiling them in DAB for 12 min and incubating in 99.5% alcohol. The observed brown spots reflected the interaction between DAB and H2O2 in the presence of peroxidase. This measurement was repeated three times using three different leaves from each light condition.
RNA isolation and real-time RT-PCR analysis
Total RNA was extracted from leaves of the same plants used for H2O2 detection, as previously described (Hamdani et al. 2015). The real-time RT-PCR was carried out with LightCycle480 System (Roche Applied Science, Indianapolis, USA). The primers for CAT (CAT-A: LOC_Os02g02400) and APX (OsAPx8: LOC_Os02g34810.1) were designed using information from the website of IDT (integrated DNA technologies, USA), while the primers for the housekeeping gene, Ubiquitin 5 (UBQ5, AK061988), were designed as described previously (Jain et al. 2006). To avoid the mismatch during PCR amplification of APX gene, we have used the 3′UTR region as template. The primer sequences were as follows:
Primers for CAT-A
-
Forward: 5′-CCACACCTTCTTCTTCCTCTTC-3′
-
Reverse: 5′-CAGTGGAACTTGACGTACCTG-3′
Primers for OsAPx8
-
Forward: 5′-ACCTCCTCTGACGAGTGTTT-3′
-
Reverse: 5′-CCATTCACATGCCCATCCTT-3′
Primers for UBQ5
-
Forward: 5′-ACCACTTCGACCGCCACTACT-3′
-
Reverse: 5′-ACGCCTAAGCCTGCTGGTT-3′
PCR reactions were performed in 96-well white plates with a volume of 20 µL containing 1 µL of cDNA template (~ 100 ng), 1 µM for each primer, and 10 µL of 10× SYBR Green I PCR Master Mix (Roche Applied Science, Indianapolis, USA). The above reactions were subjected to a heating step at 95 °C for 5 min, followed by 35 cycles of denaturation at 95 °C for 10 s, annealing at 58 °C for 20 s and elongation at 72 °C for 20 s. Transcript levels were normalized to that of UBQ5 (internal control gene). Each PCR reaction was performed in triplicate and the calculated error bars represent the standard deviation from three to four PCR results. Statistical analyses involved the \({{\text{2}}^ - }^{{\Delta \Delta {C_{\text{T}}}}}\) method (Livak and Schmittgen 2001).
Statistical analysis
We analyzed the different set of data using a student t test analysis and show the P values in the Figures. We used the Microsoft Excel software (Microsoft Inc.) to perform the t test analysis.
Results
Effect of light quality on pigment content
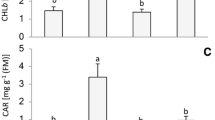
In our experiment, although the same incident light was used, plants grown under red or blue light exhibit slightly higher leaf absorption as compared to those grown under white light (Inset of Fig. 1a). However, this difference in PFD absorbed by leaves between different light spectra is less than 7% (Inset of Fig. 1a). In this study, we first examined the effect of these light regimes on the pigment content. We measured the concentration of total Chls (Chl a + Chlb) and carotenoids in plants grown under red, blue, or white light. Figure 2a shows that red light (peak at 662 nm) induced a decrease in the content of Chl (a + b) by about 45% as compared to blue (peak at 447 nm) and white (peak at 568 nm) light. These changes in Chls are much higher than the slight differences in absorbed intensities. Thus, rice grown under red light, as compared to the others, clearly had less Chl. In addition, the carotenoid content, in rice grown under red light, was also lower by about 32% than rice under blue and white light. These results are consistent with the visual observation of these leaves (during the germination stage) grown under red, blue, and white light (Fig. S2). This result suggests a down-regulation in the biosynthesis of photosynthetic pigments induced by red light, especially during the germination stage of rice. Furthermore, we observe that plants grown under blue light showed an approximately 7% higher Chl a/b ratio as compared to those grown under white light. However, no significant change in the Chl a/b ratio was observed in plants grown under red light compared to those grown under either blue or white light (Fig. 2b). This ratio is generally thought to reflect changes in the photosystem stoichiometry and the antenna size of each photosystem (see e.g., Melis et al. 1987). Since most of Chl b is associated with LHCII (Evans 1989; Green and Durnford 1996), this result may suggest a slightly smaller PSII antenna size and a lower PSII/PSI ratio in plants grown under blue light as compared to white light.
Chlorophyll (Chl) and carotenoid contents. a Chl and carotenoid contents in leaves of rice grown for 50 days under different LED lights: blue light (peak at 447 nm), red light (peak at 662 nm), and “warm” white light (peak at 568 nm); the light intensity at the top of plants was ~ 300 µmol photons m−2 s−1 for all three regimes. b Chl a/b ratio in rice grown under different LED lights. Bars in a represent average ± sd of 3–4 independent measurements; bars in b represent average ± sd of six independent measurements. Different lower-case letters indicate significant difference at P ≤ 0.05
Effect of light quality on the maximum quantum yield of PSII, as estimated by measurements of F v/F m, and on the electron transfer activity
Fv/Fm in leaves of rice plants grown under blue light showed a decrease of approximately 7% as compared to those grown in both red and white light. However, no difference was observed between red and white light (less than 1%) (Fig. 3a). Measurement of the transmittance at 820 nm, I820, shows that rice acclimated to blue light exhibited a 20% slower reduction rate of P700+. In addition, blue light induced a 32% higher accumulation of P700+ as compared to those grown under white light (Fig. 3b and inset). Such behavior suggests a slight delay in the arrival of electrons from PSII, induced by a transient block in the electron transfer from PSII in blue light, which is consistent with the observed decrease in Fv/Fm. Rice under red light only induced a 5% increase in the P700+ accumulation than rice grown under white light (Fig. 3b and inset).
Chl a fluorescence and I820 measurements. a The maximum quantum yield of PSII, represented by Fv/Fm, in rice grown under different wavelengths of LED light (see the legend of Fig. 2 for details). Rice leaves were first kept in dark for 10 min, and then excited with orange–red (625 nm) actinic light (5000 µmol photons m−2 s−1) for 0.5 s. F0, basal (initial, minimal) level of Chl a fluorescence; Fm, maximal level of Chl a fluorescence; Fv, variable Chl a fluorescence (Fm − Fo). b Transmission changes at 820 nm (I820), reflecting mostly changes in the reaction center of PSI (P700). Data are shown, on a log time scale, for leaves of rice grown under different wavelengths of LED light (see the legend of Fig. 2 for details). Each curve represents the average of 4–6 independent measurements. The vertical solid line shows the approximate time for the end of the P700 oxidation and the start of the P700+ reduction phase. Inset: A plot of the P700+ reduction rate calculated from the initial slope of the P700+ reduction phase. Bars represent average ± sd of 4–6 independent measurements. Difference in lower-case letters indicates significant differences at P ≤ 0.05
Effect of light quality on the slow chl a fluorescence induction (PSMT)
The slow (1–300 s) PSMT fluorescence phases can provide information on the dynamic changes of NPQ (Papageorgiou and Govindjee 2011). Our results show that rice acclimated to either blue or red light had a faster P–S decline with the slopes of the PS phases being − 63.9 and − 53.2 for the rice grown under blue light and red light, respectively. We also observed a faster S–M rise with the slopes of the SM phases for rice grown under blue and red light being 8.2 and 8.4, respectively, as compared to those grown under white light for which the slope for the PS Phase was − 31.3 and the slope for SM phase was 1.3. For the MT decline phase, the slope for rice grown under blue light was − 8.9, which was faster than in rice grown under white light, which has a slope of − 12.8. The MT phase started sooner at 7.5 s under blue light, but was more pronounced under white light with a starting time at 15.6 s (Fig. 4a; Table 1).
Chlorophyll a fluorescence induction curve measurements. a Chl a fluorescence induction curves (PSMT) measured in leaves of rice grown under different LED lights (see the legend of Fig. 2 for details). Leaves were first kept in dark for 10 min, and then excited with orange–red (625 nm) actinic light (100 µmol photons m−2 s−1) for 300 s. Each curve represents average of 4–6 independent measurements. Inset: carotenoids/Chl (a + b) ratio in rice grown under different LED lights. Bars represent average ± sd of three independent measurements. Difference in lower-case letters indicates significant differences at P ≤ 0.05. b Slow fluorescence (PSMT) curves in response to a new light regime: plants were first grown under either red or blue light during the germination stage, and then exposed to white light. Inset: Carotenoids/Chl (a + b) ratio in rice lines grown under different LED light regimes. White: rice grown under white light; Blue + White: rice first grown under blue, and then switched to white light; Red + White: rice first grown under red light and then switched to white light. Bars represent average ± sd of three independent measurements. Difference in lower-case letters indicates significant differences at P ≤ 0.05
Interestingly, the carotenoids/Chl (a + b) ratio was similar under both red and blue light, but both being approximately 18% higher than under white light (Inset of Fig. 4a). To test the relationship between the carotenoids/Chl (a + b) ratio and the PSMT phase, rice plants grown under both red and blue light during the germination stage were switched to white light (Fig. 4b and Inset). The transition from red to white light induced a significant modification, i.e., a 32% decrease in carotenoids/Chl (a + b) ratio as compared to the ratio under white light, in parallel with a considerable change in the shape of the PSMT fluorescence transient (Fig. 4b and Inset). In contrast, the transition from blue to white light did not induce a significant modification in the carotenoids/Chl (a + b) ratio (Fig. 4b).
Light quality and photoprotection
To further investigate the effect of light quality on plant photoprotection, we measured the photochemical and non-photochemical Chl a fluorescence quenching, using a pulse amplitude modulation (PAM) fluorometer with the saturating light pulse method (Schreiber 2004). Figure 5a shows that plants acclimated to blue light exhibited a faster quenching of chlorophyll fluorescence with a slope of − 0.0043 (slope calculated during the first 130 s of actinic light) and a 15% lower steady-state Fm′ during light exposure as compared to those grown under white light with a slope of around − 0.0040. In contrast, red light induced: (1) a slightly slower quenching; (2) a 5% higher steady-state Fm′ during light exposure; however, this difference is not statistically significant; and (3) a significantly slower recovery than that in white light grown plants, with a slope of 0.001 for red and 0.002 for white (recovery slope was calculated during the first 100 s of dark relaxation). Figures 5b and S3 show that the steady-state NPQ was 22.5% and 6% higher in plants grown under blue and red light, respectively, as compared to plants grown under white light. In addition to this, we observed a faster NPQ induction rate in plants grown under red and blue light as compared to white light, with the slope of NPQ induction being 0.018, 0.015, and 0.013 for rice grown under blue, red, and white light, respectively. However, relaxation of NPQ in darkness was slightly slower in rice grown under red light than those under blue and white light (see Table 2). In rice treated with blue or red light, the steady-state quantum yield of PSII, i.e., Y(II), was lower (by 24% and 36%, respectively) and the quantum yield of regulated energy dissipation in PSII [Y(NPQ)] was higher (by 11% and 6% respectively) than in rice grown under white light (Fig. 6a). In rice treated with red light, quantum yield of non-regulated energy dissipation in PSII [Y(NO)] was higher by 19%.
Fluorescence quenching analysis. a Kinetics of maximum Chl a fluorescence under light (Fm′) normalized to Fm in rice grown under different LED lights (see the legend of Fig. 2). After a 10-min dark period, leaves were excited by blue actinic light (815 µmol photons m−2 s−1) for 10 min followed by a 10-min dark. During this time, a saturating (20,000 µmol photons m−2 s−1) light pulse of 500 ms duration was applied every 20 s to measure Fm′. b Dynamic changes of non-photochemical quenching, NPQ, in rice grown under different LED lights (see the legend of Fig. 2 for details). Each curve represents average ± sd of 4–6 independent measurements. Arrows indicate switching on (down arrow) and off (up arrow) of the blue actinic light
Measurement of the quantum yield of PSII under steady state and dynamic light conditions. a Steady state of quantum yields of regulated energy dissipation Y(NPQ), non-regulated energy dissipation in PSII, Y(NO), and effective quantum yield of PSII, Y(II), in rice grown under different wavelengths of LED light (see the legend of Fig. 2 for details). Stacked bars represent the average (%) of 4–6 independent measurements. Difference in lower-case letters indicates significant differences at P ≤ 0.05. b Y(II) in rice grown under red, blue, or white LED light (see the legend of Fig. 2 for details). Each curve represents average ± sd of 4–6 independent measurements. Arrows indicate switching on (down arrow) and off (up arrow) of the blue actinic light (815 µmol photons m−2 s−1)
Light quality and the antioxidant system
Rice acclimated to either red or blue light, as compared to white light, showed a higher H2O2 accumulation in the area of leaf exposed to high light (Fig. 7a), as indicated by staining with 3, 3′diaminobenzidine (DAB) (Fryer et al. 2002). We further measured changes in transcript levels of both CAT (isozyme CAT-A: LOC_Os02g02400) and APX (OsAPx8: LOC_Os02g34810.1), which specifically target H2O2, using the real-time RT-PCR method. Figure 7b shows a significant decrease in both CAT and APX transcript levels under both blue and red light as compared to white light (around 1.5–3.5 fold, respectively, for CAT, and around 1.15–2.5 fold, respectively, for APX), suggesting that the acclimation under both blue and red light, especially under red light, down-regulated key enzymes involved against ROS and hence potentially weakened the antioxidant system. Our results are consistent with those of Bian et al. (2018) on lettuce, where the APX and CAT activities were found to decrease under continuous “Red + Blue” light treatment for 24 and 48 h as compared to that under “Red + Blue + Green” light with the same time frame treatment.
Detection of hydrogen peroxide (H2O2). a Image of three rice leaves infiltrated with 3, 3′diaminobenzidine (DAB). Mature plants grown under sunlight were acclimated to red, blue, or white LED light at around 800 µmol photons m−2 s−1 for 7 days, then the left side of the leaf (indicated by a yellow horizontal bar) was excited by strong white light (1400 µmol photons m−2 s−1) for 60 min. The brown staining reflects the interaction between DAB and H2O2. b Transcript levels of CAT (CAT-A: LOC_Os02g02400) and APX (OsAPx8: LOC_Os02g34810.1) measured by real-time RT-PCR in rice acclimated to different wavelengths of LED light (see the legend of Fig. 2 for details) for 7 days. The transcript levels of both CAT and APX were normalized to that of UBQ gene
Discussion
In this study, we grew plants under the same incident PFD but with different light quality, i.e., red, blue, or white light. Plants grown under different light quality absorbed different amount of light; however, the difference of absorbed PFD under different light quality is less than 7%. Therefore, the observed differences in photosynthetic properties and photoprotective properties observed in this study are mainly due to light quality. Our results show that rice plants grown under red or blue light, in contrast to white light, experience greater photo-damage despite their high NPQ capacity. Furthermore, we suggest, based on measured expression levels of catalase and ascorbate peroxidase in these plants, that deficient antioxidant capacities might have contributed to these observed photo-damage under monochromatic red or blue light (Fig. 8). Now, we shall discuss the data supporting this statement.
A schematic representation of the electron transfer process and photoprotection in rice grown under white (a) blue (b) or red light (c). Results from this study suggest that blue light, as compared to the white light, induces: (1) a partial inactivation of the oxygen evolving complex, increasing reactive oxygen species (ROS) production; (2) a decreased rate of electron transfer process; (3) an increase in heat dissipation through non-photochemical quenching (NPQ) of the excited Chl a; and (4) an inefficient antioxidant system through down-regulation of key enzymes involved against reactive oxygen species (ROS). On the other hand, red light induces: (1) an increase in the NPQ process; and (2) an inefficient antioxidant system through down-regulation of key enzymes involved against ROS
Rice plants grown under long-term red or blue light, as compared to white light, show decreased PSII quantum efficiency and increased NPQ
As compared to white light, there was a significant increase in the Chl a/b ratio (Fig. 2b) and lower Fv/Fm in blue light treated samples (Fig. 3a). One possibility of a lower Fv/Fm under blue light is blue-light induced limitation on the (electron) donor side of PSII since blue light has been suggested to damage the Mn cluster in the OEC of Synechococcus elongatus (Ohnishi et al. 2005), and thus the deterioration of OEC activity may have led to a decrease in Fv/Fm (Beauchemin et al. 2007; Hamdani and Carpentier 2009; Hasni et al. 2013). In addition, we found a delay in the reduction of P700+ (Fig. 3b and inset) under blue light, which is again consistent with a donor-side limitation. Compared to white light, red light was shown to induce a decrease in both carotenoid and chlorophyll concentrations (Fig. 2a), but there were insignificant changes in Chl a/b ratio, Fv/Fm and P700+ accumulation (Figs. 2b, 3a, b). The decreased pigment concentration can be related to the regulation of expression of genes related to biosynthesis or degradation of these pigments under different lights, as shown in a recent study in strawberry (Zhang et al. 2018).
The PSMT phase of Chl a fluorescence transient provides information on the NPQ process (Fratanico et al. 2016). Our results showed that the PS phase, when rice was grown under red or blue light, decreased much faster as compared to that in white light (Fig. 4a; Table 1). This probably reflects the ability of rice acclimated to either red or blue light to more efficiently activate the qE component. Furthermore, rice grown under red or blue monochromatic light showed increased magnitude of SM phase (Fig. 4a), suggesting an increased state transition (state 2 to state 1) in these plants, as compared to rice grown under white light (Papageorgiou and Govindjee 2011; Kodru et al. 2015). In the blue light- or red light-treated plants, the carotenoids/Chl(a + b) ratio was higher (Inset of Fig. 4a), suggesting that an increased Zx concentration might be one possible reason for this increased NPQ response (Rubin and Horton 1999; Demmig-Adams et al. 2014; Sacharz et al. 2017).
The changes in NPQ under blue light were also seen in the changes in the Fm′ under light. As compared to rice grown under red or white light, we observed a faster and larger decrease in Fm′ in rice grown under blue light when exposed to a 10-min blue actinic light (815 µmol photons m−2 s−1) (Fig. 5a). The faster decrease in Fm′ can be attributed to a rapid induction of NPQ as discussed above. In addition, the decrease of Fm′ might also be related to changes in the structure of the LHCII in leaves grown under blue light, since in these leaves, the Chl a/b ratio increased significantly (Fig. 2b), suggesting a decrease in the relative abundance of LHCII compared to other photosynthetic pigment-protein complexes. This is because Chl b is mainly associated with LHCII and LHCII usually has a much lower Chl a/b ratio compared to other major photosynthetic pigment-protein complexes (Evans 1989; Green and Durnford 1996).
The faster NPQ formation in the blue-light grown plants is also observed in the measured kinetics of NPQ and Y(II) in leaves grown under blue, red, or white light. Upon exposure to actinic light, the NPQ curves showed three distinct phases: fast, intermediate and slow. The fast phase (0–100 s), which is thought to be regulated by ΔpH formation, but without de-epoxidation of Vx–Zx (Johnson et al. 2008), was faster (NPQ Induction slope for rice grown under blue, red, and white light being 0.018, 0.015, and 0.013, respectively). These observed changes in the fast phase are in full agreement with the PSMT measurements, which showed a faster and a pronounced PS phase, induced by both blue and red light as compared to that by white light (Fig. 4a). The intermediate phase of NPQ (100–200 s) was higher in rice grown under blue and red light than that in rice grown in white light. This phase is probably related to Zx accumulation and its binding to LHCII (Dall’Osto et al. 2005; Nilkens et al. 2010). There was an increase in the amplitude of the slowest phase (200–600 s) of about 22.5% and 6% in blue and red light (with lower level) grown rice, respectively, as compared to that in rice grown in white light (Figs. 5b and S3, Table 2), which was probably related to the decreased Y(II) during this period (Fig. 6b).
Growing rice under either blue or red light regime decreased Y(II), as compared to rice grown under white light (Fig. 6a). During the entire period of Y(II) kinetics measurement, white light grown plants showed higher Y(II) as compared to those grown under red or blue light (Fig. 6b; Table 3). Furthermore, during the transition from dark to light, the speed of increase of Y(II) was much higher; during the transition from light to dark, the speed of recovery of Y(II) was also much higher (Fig. 6b; Table 3). The decreased steady-state Y(II) under light in rice grown under either red or blue light regime was due to increased Y(NPQ) and Y(NO) (See Fig. 6a; Table 3). Here, our data also show that rice plants grown under either red or blue light also have decreased capacity in restoring the maximum efficiency during dark.
Decreased capacity of antioxidant system in rice grown under long-term red or blue light
Under high light, plants use different mechanisms to avoid photodamage caused by excessive light energy, including NPQ, movement of leaf, movement of chloroplasts, and up-regulation of the antioxidant system (Cazzaniga et al. 2013; Derks et al. 2015; Gill and Tuteja 2010; Koller 1990; Niyogi 1999; Ort 2001). In this study, we used an incident light level of 300 µmol photons m−2 s−1, which is considered to be a moderate light level for rice growth. However, growing rice under this moderate light level with monochromatic red or blue light resulted in substantial decrease of Y(II) in rice as compared to rice plants grown under white light (Fig. 6a). Data from this study suggest that a compromised antioxidant system in these plants might have contributed to this observed decrease in Y(II). This is because, though the incident light level was not too high, under monochromatic light conditions, the light absorption by PSI and PSII may have been less balanced, leading to increased production of reactive oxygen species which demands for a robust antioxidant system (Asada 2006; Huang et al. 2018). Considering that rice leaves treated with monochromatic light have increased Y(NPQ) and Y(NO), we speculate that these plants may have compromised antioxidant capacity as compared to plants grown under white light, resulting in increased quantity of reactive oxygen species. To test this possibility, we had conducted an experiment where we first grew rice plants under sunlight, then treated the mature rice plants by red, blue, or white light for 7 days. Then, we used the DAB staining to detect one representative reactive oxygen species, hydrogen peroxide (H2O2), in these leaves (Daudi and O’Brien 2012). Our results show that there was indeed higher concentration of H2O2 in rice plants treated by red or blue light, as compared to white light (Fig. 7a).
Since light quality can dramatically influence gene expression (Glick et al. 1986; Gyula et al. 2003), we examined the expression levels of two genes coding two antioxidant enzymes i.e., CAT and APX (Asada 2006). Results showed that a significant down-regulation of both CAT and APX transcripts was observed in rice treated with both red and blue light as compared to those treated with white light. Interestingly, Bian et al. (2018) recently showed that green light supplementation enhanced CAT and APX enzyme activities. Hence, the weakened antioxidant system and correspondingly the accumulated reactive oxygen species, as represented by H2O2 here, in rice plants grown under blue or red light regimes might have played a role in the observed changes in Y(II), Y(NO), Y(NPQ) and even in Fv/Fm (Figs. 3a, 6a).
Conclusion
In the present work, we show that monochromatic blue or red light give substantial stress to rice. Though there is difference in the absorbed light in leaves grown under different light spectrums with the same incident PFD, the difference in absorbed light between these three light regimes was less than 7% (Inset of Fig. 1a). Therefore, our observed difference in Y(II), Y(NPQ), and Y(NO) can be mainly attributed to the differences in the light spectrum, though other factors might also play a role in these changes. Recently, Bian et al. (2018) showed that green light plays a key role in maintaining better PSII efficiency and efficient photoprotection. In this study, although plants acclimated under both blue and red light led to higher NPQ (22.5% and 6%), as compared to rice grown under white light, they still did not have sufficient protection to photosystem, as shown by the decreased Y(II), which might be due to inefficient antioxidant activity (Figs. 5, 6), as shown by these elevated H2O2 contents along with decreased transcription abundance of CAT and APX (Fig. 7). Here we do recognize that the potential role of photoreceptors (Möglich et al. 2010) in generating the observed phenotype reported was not studied in this study; it is likely that they also have contributed to the observed changes in pigment composition and NPQ dynamics. It is worth noting here that several previous reports have shown a beneficial effect of monochromatic light (blue or red and others) on photosynthesis activities (Eskins et al. 1991; Hogewoning et al. 2010; Jung et al. 2013; Leong et al. 1985). This is because most of these earlier experiments were done under either much lower photon flux densities or under much shorter treatment periods compared to this study (Table S1). Taken all these together, we show that long-term monochromatic light treatment can compromise antioxidant system in rice, leading to decreased quantum yield of PSII and increased NPQ. In other words, even under moderate ambient light levels, plants require a robust antioxidant system to remove reactive oxygen species to maintain high quantum yield of PSII.
References
Alboresi A, DallOsto L, Aprile A, Carillo P, Roncaglia E, Cattivelli L, Bassi R (2011) Reactive oxygen species and transcript analysis upon excess light treatment in wild-type Arabidopsis thaliana vs a photosensitive mutant lacking zeaxanthin and lutein. BMC Plant Biol 11:62–84
Asada K (2006) Production and scavenging of reactive oxygen species in chloroplasts and their function. Plant Physiol 141:391–396
Beauchemin R, Gauthier A, Harnois J, Boisvert S, Govindachary S, Carpentier R (2007) Spermine and spermidine inhibition of photosystem II: disassembly of the oxygen evolving complex and consequent perturbation in electron donation from TyrZ to P680 + and the quinone acceptors QA – to QB. Biochim Biophys Acta 1767:905–912
Belgiol E, Trsková E, Kotabová E, Ewe D, Prášil O, Kaňa R (2018) High light acclimation of Chromera velia points to photoprotective NPQ. Photosynth Res 135:263–274
Bian Z, Yang Q, Lib T, Cheng R, Barnetta Y, Lua C (2018) Study of the beneficial effects of green light on lettuce grown under short-term continuous red and blue light-emitting diodes. Physiol Plant. http://irep.ntu.ac.uk/id/eprint/33129
Björkman O, Powles SB (1987) Leaf movement in the shade species Oxalis oregana L I. Response to light level and light quality. Carnegie Inst Wash Year Book 80:59–62
Bukhov NG, Drozdova IS, Bondar VV (1995) Light response curves of photosynthesis in leaves of sun-type and shade-type plants grown in blue or red light. J Photochem Photobiol B 30:39–41
Cazzaniga S, Osto LD, Kong SG, Wada M, Bassi R (2013) Interaction between avoidance of photon absorption, excess energy dissipation and zeaxanthin synthesis against photooxidative stress in Arabidopsis. Plant J 76:568–579
Dall’Osto L, Caffarri S, Bassi R (2005) A mechanism of nonphotochemical energy dissipation, independent from Psbs, revealed by a conformational change in the antenna protein CP26. Plant Cell 17:1217–1232
Daudi A, O’Brien JA (2012) Detection of hydrogen peroxide by DAB staining in Arabidopsis leaves. Bio Protoc 2(18):1–4
Demmig-Adams B, Garab G, Adams WW, Govindjee (eds) (2014) Non-photochemical quenching and energy dissipation in plants, algae and cyanobacteria. Springer, Dordrecht
Derks A, Schaven K, Bruce D (2015) Diverse mechanisms for photoprotection in photosynthesis. Dynamic regulation of photosystem II excitation in response to rapid environmental change. Biochim Biophys Acta 1847:468–485
Eskins K, Jiang CZ, Shibles R (1991) Light-quality and irradiance effects on pigments, light-harvesting proteins and Rubisco activity in a chlorophyll- and light-harvesting-deficient soybean mutant. Physiol Plant 83:47–53
Evans JR (1989) Partitioning of nitrogen between and within leaves grown under different irradiances. Aust J Plant Physiol 16:533–548
Fratanico A, Tocquin P, Franck F (2016) The chlorophyll a fluorescence induction curve in the green microalga Haematococcus pluvialis: further insight into the nature of the P-S-M fluctuation and its relationship with the “low wave” phenomenon sat steady state. Photosynth Res 128:271–285
Fryer MJ, Oxborough K, Mullineaux PM, Baker NR (2002) Imaging of photo-oxidative stress responses in leaves. J Exp Bot 53:1249–1254
Genty B, Briantais JM, Baker NR (1989) The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochim Biophys Acta 990:87–92
Gill SS, Tuteja N (2010) Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem 48:909–930
Glick RE, McCauley SW, Gruissem W, Melis A (1986) Light quality regulates expression of choloroplast genes and assembly of photosynthetic membrane complexes. Proc Natl Acad Sci USA 83:4287–4291
Grant RH (1997) Partitioning of biologically active radiation in plant canopies. Int J Biometeorol 40:26–40
Green BR, Durnford DG (1996) The chlorophyll-carotenoid proteins of oxygenic photosynthesis. Ann Rev Plant Physiol Plant Mol Biol 47:685–714
Green BR, Parson WW (eds) (2003) Light-harvesting antennas in photosynthesis. Springer, Dordrecht
Gyula P, Schäfer E, Nagy F (2003) Light perception and signalling in higher plants. Curr Opin Plant Biol 6:446–452
Hamdani S, Carpentier R (2009) Interaction of methylamine with extrinsic and intrinsic subunits of photosystem II. Biochim Biophys Acta 1787:1223–1229
Hamdani S, Qu M, Xin CP, Li M, Chu C, Govindjee Zhu XG (2015) Variations between the photosynthetic properties of elite and landrace Chinese rice cultivars revealed by simultaneous measurements of 820 nm transmission signal and chlorophyll a fluorescence induction. J Plant Physiol 177:128–138
Hasni I, Hamdani S, Carpentier R (2013) Destabilization of the oxygen evolving complex of photosystem II by Al3+. Photochem Photobiol 89:1135–1142
Hogewoning SW, Trouwborst G, Maljaars H, Poorter H, Ieperen WV, Harbinson J (2010) Blue light dose–responses of leaf photosynthesis, morphology, and chemical composition of Cucumis sativus grown under different combinations of red and blue light. J Exp Bot 61:3107–3117
Holm G (1954) Chlorophyll mutations in barley. Acta Agric Sci 4:457–461
Huang W, Tikkanen M, Zhang SB (2018) Photoinhibition of photosystem I in Nephrolepis falciformis depends on reactive oxygen species generated in the chloroplast stroma. Photosynth Res 137:129–140
Inada K (1980) Spectral absorption property of pigments in living leaves and its contribution to photosynthesis. Japan Jour Crop Sci 49:286–294
Jain M, Nijhawan A, Tyagi AK, Khurana JP (2006) Validation of housekeeping genes as internal control for studying gene expression in rice by quantitative real-time PCR. Biochem Biophys Res Commun 345:646–651
Johnson MP, Davison PA, Ruban AV, Horton P (2008) The xanthophyll cycle pool size controls the kinetics of non-photochemical quenching in Arabidopsis thaliana. FEBS Lett 582:262–266
Jung ES, Lee S, Lim SH, Ha SH, Liu KH, Lee CH (2013) Metabolite profiling of the short-term responses of rice leaves (Oryza sativa cv. Ilmi) cultivated under different LED lights and its correlations with antioxidant activities. Plant Sci 210:61–69
Kagawa T, Wada M (2000) Blue light-induced chloroplast relocation in Arabidopsis thaliana as analyzed by microbeam irradiation. Plant Cell Physiol 41:84–93
Klughammer C, Schreiber U (2008) Complementary PSII quantum yields calculated from simple fluorescence parameters measured by PAM fluorometry and the Saturation Pulse method. PAM Appl Notes 1:27–35
Kodru S, Malavath T, Devadasu E, Nellaepalli S, Stirbet A, Subramanyam R, Govindjee (2015) The slow S to M rise of chlorophyll a fluorescence reflects transitions from state 2 to state 1 in the green alga Chlamydomonas reinhardtii. Photosynth Res 125:219–231
Koller D (1990) Light-driven leaf movements. Plant Cell Environ 13:615–632
Kramer DM, Johnson G, Kiirats O, Edwards GE (2004) New flux parameters for the determination of QA redox state and excitation fluxes. Photosynth Res 79:209–218
Kromdijk J, Glowacka K, Leonelli L, Gabilly ST, Iwai M, Niyogi KK, Long SP (2016) Improving Photosynthesis and crop productivity by accelerating recovery from photoprotection. Science 354:857–861
Leonelli L, Brooks MD, Niyogi KK (2017) Engineering the lutein epoxide cycle into Arabidopsis thaliana. Proc Natl Acad Sci USA 114:7002–7008
Leong TY, Anderson JM (1984) Effect of light quality on the composition and function of thylakoid membranes in Atriplex triangularis. Biochim Biophys Acta 766:533–541
Leong TY, Goodchild DJ, Anderson JM (1985) Effect of light quality on the composition, function, and structure of photosynthetic thylakoid membranes of Asplenium australasicum (Sm.). Hook Plant Physiol 78:561–567
Livak K, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−∆∆CT method. Methods 25:402–408
Melis A, Spangfort M, Andersson B (1987) Light-absorption and electron-transport balance between photosystem II and photosystem I in spinach chloroplasts. Photochem Photobiol 45:129–136
Miyake C, Horiguchi S, Makino A, Shizaki Y, Yamamoto H, Tomizawa KI (2005) Effects of light intensity on cyclic electron flow around PSI and its relationship to non-photochemical quenching of Chl fluorescence in tobacco leaves. Plant Cell Physiol 46:1819–1830
Möglich A, Yang X, Ayers RA, Moffat K (2010) Structure and function of plant photoreceptors. Ann Review Plant Biol 61:21–47
Müller P, Li XP, Niyogi KK (2001) Non-photochemical quenching. A response to excess light energy. Plant Physiol 125:1558–1566
Murchie EH, Lawson T (2013) Chlorophyll fluorescence analysis: a guide to good practice and understanding some new applications. J Exp Bot 64:3983–3998
Nilkens M, Kress E, Lambrev P, Milosalvina Y, Müller M, Holzwarth AR, Jahns P (2010) Identification of a slowly inducible zeaxanthin-dependent component of non-photochemical quenching of chlorophyll fluorescence generated under steady-state conditions in Arabidopsis. Biochim Biophys Acta 1797:466–475
Niyogi KK (1999) Photoprotection revisited: genetic and molecular approaches. Ann Rev Plant Physiol Plant Mol Biol 50:333–359
Norcini JG, Andersen PC, Knox GW (1991) Light intensity influences leaf physiology and plant growth characteristics of Photinia × fraseri. J Am Hort Sci 116:1046–1051
Oguchi R, Douwstara P, Fujita T, Chow WS, Terashima I (2011) Intra-leaf gradients of photoinhibition induced by different color lights: implications for the dual mechanisms of photoinhibition and for the application of conventional chlorophyll fluorometers. New Phytol 191:146–159
Ohnishi N, Allakhverdiev SI, Takahashi S, Higashi S, Watanabe M, Nishiyama N, Murata N (2005) Two-step mechanism of photodamage to photosystem II: step 1 occurs at the oxygen-evolving complex and step 2 occurs at the photochemical reaction center. Biochemistry 44:8494–8499
Ort DR (2001) When there is too much light. Plant Physiol 125:29–32
Papageorgiou GC, Govindjee (2011) Photosystem II fluorescence: slow changes—scaling from the past. J Photochem Photbiol B 104:258–270
Papageorgiou GC, Govindjee (2014) The non-photochemical quenching of the electronically excited state of chlorophyll a in plants: definitions, timeline, viewpoints. Open questions. In: Demmig B, Adams G, Garab WW, Adams III, Govindjee (eds) Non-photochemical quenching and energy dissipation in plants, algae and cyanobacteria. advances in photosynthesis and respiration including bioenergy and related processes. Springer, Dordrecht
Pfündel E, Klughammer C, Schreiber U (2008) Monitoring the effects of reduced PSII antenna size on quantum yields of photosystems I and II using the Dual-PAM-100 measuring system. PAM Appl Notes 1:21–24
Porra RJ, Thompson WA, Kriedelman PE (1989) Determination of accurate extraction and simultaneously equation for assaying chlorophyll a and b extracted with different solvents: verification of the concentration of chlorophyll standards by atomic absorption spectroscopy. Biochim Biophys Acta 975:384–394
Pospíšil P (2009) Production of reactive oxygen species by photosystem II. Biochim Biophys Acta -Bioenerg 1787:1151–1160
Pospíšil P (2012) Molecular mechanisms of production and scavenging of reactive oxygen species by photosystem II. Biochim Biophys Acta 1817:218–231
Pospíšil P, Prasad A (2014) Formation of singlet oxygen and protection against its oxidative damage in photosystem II under abiotic stress. J Photochem Photobiol B 137:39–48
Rochaix J (2014) Regulation and dynamics of the light-harvesting system. Annu Rev Plant Biol 65:287–309
Ruban AV (2018) Light harvesting control in plants. FEBS Lett. https://doi.org/10.1002/1873-3468.13111
Rubin AV, Horton P (1999) The xanthophyll cycle modulates the kinetics of nonphotchemical energy dissipation in isolated light harvesting complexes, intact chloroplasts and leaves of spinach. Plant Physiol 119:531–542
Sacharz J, Giovagnetti V, Ungerer P, Mastroianni G, Ruban A (2017) The xanthophyll cycle affects reversible PsbS-LHCII interactions to control non-photochemical quenching. Nat Plants 3:16225–16234
Schreiber U (2004) Pulse amplitude modulation fluorometry and saturation pulse method: an overview. In: Papageorgiou GC, Govindjee (eds) Chlorophyll a fluorescence: a signature of photosynthesis. advances in photosynthesis and respiration including bioenergy and related processes. Springer, Dordrecht
Senger H, Bauer B (1987) The influence of light quality on adaptation and function of the photosynthetic apparatus. Photochem Photobiol 45:939–946
Sztatelman O, Waloszek A, Banas AK, Gabrys H (2010) Photoprotective function of chloroplast avoidance movement: in vivo chlorophyll fluorescence study. J Exp Bot 167:709–716
Tian Y, Sacharz J, Ware MA, Zhang H, Ruban AV (2017) Effects of periodic photoinhibitory light exposure on physiology and productivity of Arabidopsis plants grown under low light. J Exp Bot 68:4249–4262
Triantaphylides C, Havaux M (2009) Singlet oxygen in plants: production, detoxification and signaling. Trends Plant Sci 14:219–228
Zhang Y, Jiang L, Li Y, Chen Q, Ye Y, Zhang Y, Luo Y, Sun B, Wang X, Tang H (2018) Effect of red and blue light on anthocyanin accumulation and differential gene expression in strawberry (Fragaria x anaassa). Molecules 234:820. https://doi.org/10.3390/molecules23040820
Zhu XG, Ort DR, Whitmarsh J, Long SP (2004) The slow reversibility of photosystem II thermal energy dissipation on transfer from high to low light may cause large losses in carbon gain by crop canopies: a theoretical analysis. J Exp Bot 55:1167–1175
Acknowledgements
This work was supported by Strategic Priority Research Program of the Chinese Academy of Sciences, Grant No. XDB27020105, CAS-TWAS President’s Fellowship Program, the Chinese Academy of Sciences (CAS) strategic leading project on agriculture XDA08020301 and National Basic Research and Development Program of the Ministry of Science and Technology of China 2015CB150104, 2014AA101601, National Science Foundation # C020401. Govindjee thanks the office of Information Technology, Life Sciences, the Department of Plant Biology and the Department of Biochemistry for support.
Author information
Authors and Affiliations
Contributions
SH and XZ designed and conducted the research. SH, NK, SP, MQ, and JJ performed research. SH, NK, G, and XZ wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial interests.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hamdani, S., Khan, N., Perveen, S. et al. Changes in the photosynthesis properties and photoprotection capacity in rice (Oryza sativa) grown under red, blue, or white light. Photosynth Res 139, 107–121 (2019). https://doi.org/10.1007/s11120-018-0589-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-018-0589-6