Abstract
The challenge of antimicrobial resistance requires new solutions, and Rhodiola sp. is a promising candidate due to its rich medicinal history. This review focuses on Rhodiola sp., especially R. rosea, highlighting its phytochemical constituents, such as salidroside, rosavins, and other phenolics, and their antimicrobial potential. We investigate the efficacy of Rhodiola sp. against various pathogenic bacteria, suggesting their value in combating antimicrobial resistance. Additionally, Rhodiola compounds are being investigated both as direct antimicrobials and as resistance modifiers that increase the efficacy of antibiotics. Rhodiola extracts and salidroside modulate gut microbiota, thereby affecting health and physiological and metabolic disorders. This illustrates the comprehensive therapeutic potential of Rhodiola sp. We emphasize the importance of understanding the complex interactions between Rhodiola phytochemicals, their antimicrobial effects, and their effects on gut microbiota as well as the need for in-depth research. Further studies will be crucial for the development of holistic treatment approaches. Finally, Rhodiola sp. is highlighted as an important natural resource in the fight against microbial pathogens and antibiotic resistance. Research on Rhodiola sp. should continue to further unravel pharmacological and health-promoting potential and thus address an important public health challenge.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
During the last decade, Rhodiola plants have gained much attention, illustrated by around 150 publications every year (CAS SciFindern 2023). Species of the genus Rhodiola L. (Crassulaceae) are characterized as perennial herbs with rhizomes and usually fleshy persistent leaves (WFO 2023a). Their natural habitat spreads over the subarctic and subalpine northern hemisphere (PWO 2023). The number of reported species varies between 53 (WFO 2023a) and 72 (IPNI 2023), and this variability is due to numerous species names being mentioned in the literature that are not accepted species names anymore. Several Rhodiola species have been reported for their traditional medicinal uses, including R. rosea L. (roseroot, golden rod), R. kirilowii (Regel) Maxim., R. algida (Ledeb.) Fisch. & C.A.Mey., R. wallichiana (Hook.) Fu, and R. crenulata (Hook.f. & Thomson) H.Ohba (Tao et al. 2019). The frequently mentioned species R. sachalinensis Boriss. and R. imbricata Edgew. are regarded as synonyms for R. rosea L. (WFO 2023b) and are thus not mentioned separately in this review. Similarly, R. sacra (Raym.-Hamet) Fu is a synonym for R. chrysanthemifolia subsp. sacra (Raym.-Hamet) H.Ohba, and thus the currently accepted name is used (WFO 2023c). The most well-known is roseroot, R. rosea; its rhizomes and roots are used as traditional herbal medicinal products to relieve symptoms of stress, such as fatigue and exhaustion (EMA 2023). Aside from these traditional uses, the chemistry (see “The phytochemistry of Rhodiola sp.” section) of Rhodiola compounds implies potential antibacterial effects.
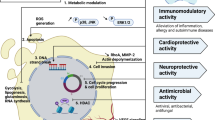
The worldwide spread of antibiotic resistance represents an alarming situation that calls for immediate action (WHO 2023). Natural plant products, including those discovered in Rhodiola, are increasingly recognized as promising agents to fight antibacterial resistance (Klančnik et al. 2021; Waditzer und Bucar 2021; Abdallah et al. 2023; Goncalves et al. 2023; Liu et al. 2023a). The antimicrobial activities of Rhodiola sp. should also be discussed in light of their impact on the complex microbial ecosystem in the digestive tract, i.e., gut microbiota (GM). The crucial role of GM in various physiological and immunological functions is being increasingly recognized, and thus the reported activities of Rhodiola sp. in this complex relationship between GM, health, and disease are covered by this review. Overall, the present review provides an overview of the current knowledge on Rhodiola extracts and compounds as antibacterials, research gaps, and perspectives for future developments towards Rhodiola antibacterial preparations.
The phytochemistry of Rhodiola sp.
The chemistry of Rhodiola species has been extensively studied in the last years (Chiang et al. 2015; Dong et al. 2023; Marchev et al. 2016, 2020; Michels et al. 2020; Langeder und Grienke 2021; Kunčič et al. 2022; Kosakowska et al. 2018; Tao et al. 2019; Olennikov et al. 2020). Quality control of Rhodiola roots and herbal medicinal products is still based on phenylethanoids (mainly salidroside) and phenylpropanoids (rosavins) (Marchev et al. 2020; Dimpfel et al. 2018). However, attempts are being made to expand the range of marker compounds by including flavonoids (Zomborszki et al. 2019). Additionally, the monoterpene alcohol glycoside rosiridin was shown to be present in significant amounts in Rhodiola plant material and medicinal products on the market (Langeder und Grienke 2021). Tang et al. (2023) revealed that rosiridin naturally occurs as two C-4 epimers based on one enantiomeric aglycone. The spectrum of activity of relevant Rhodiola phytochemicals is even wider, illustrated by recent studies on the memory-enhancing effect of long-chain feruloyl fatty alcohol esters such as ferulic acid eicosyl ester (FAE-20) (Michels et al. 2020, 2018). Kunčič et al. (2022) revealed that proanthocyanidins (including catechins such as epigallocatechin gallate (EGCG)) and flavonoid glycosides obtained from R. rosea crude ethanolic extracts exert inhibitory effects on Campylobacter intercellular signaling. This clearly indicates that several Rhodiola sp. phytochemicals are of relevance for quality control of Rhodiola plant material and products, and that current strategies focusing on only salidroside and rosavins should be re-evaluated. A list of the most frequently reported compound classes is provided in Table 1, and characteristic compounds of each class are presented in Fig. 1.
Medicinal uses of Rhodiola sp.
Among Rhodiola species, R. rosea has been most extensively studied in the context of traditional medicine. Detailed outlines of the long history of R. rosea in European and Asian materia medica have been presented by Anheyer et al. (2021) and Pannossian et al. (2010). The first written reports of medicinal uses of R. rosea even date back to ancient Greece. Rhodiolae radix (“Rodia riza”) was mentioned by Dioscurides as an externally applied remedy against headache, an indication which is later frequently mentioned in herbal books of the Middle Ages and early modern age (Anheyer et al. 2021). Further traditional indications include tumors, mental conditions (“hysteria”), liver disease, swellings, edema, hernias, discharges, and skin disorders, and the use of R. rosea as a stimulant and astringent (Panossian et al. 2010; Anheyer et al. 2021; Chiang et al. 2015). In the European Union, herbal medicinal products containing a dry extract (DER 1.5–5:1) of R. rosea rhizomes and roots (extracted by 67–70% ethanol (v/v)), are used to relieve symptoms of stress (e.g., fatigue and exhaustion) (EMA 2023). This indicates adaptogenic and stress-protective effects.
Recently, the pharmacological activities and mechanisms of action of R. rosea in chronic diseases have been reviewed (Bernatoniene et al. 2023). Furthermore, a critical review of the effectivity of Rhodiola preparations in enhancing exercise performance was published (Tinsley et al. 2024). It concluded that they exhibit potential to enhance performance and performance-related outcomes for several types of exercise but that the findings were inconsistent, possibly due to variable preparations, doses, durations of administration, and patient cohorts (Tinsley et al. 2024). The dose–response action of Rhodiola extracts and compounds was demonstrated to exhibit hormesis, i.e., a relationship in which low doses are beneficial, whereas high doses can even be toxic (Calabrese et al. 2023).
Key compounds for activities
Salidroside is considered a key Rhodiola compound that acts on the central nervous system (affecting Parkinson’s disease and Alzheimer’s disease) and heart (affecting atrial fibrosis and coronary heart disease) and attenuates hepatotoxic effects and oxidative stress (Jin et al. 2022; Hai et al. 2023; Gao et al. 2023; Li and Yao 2023; Liang et al. 2023a; Zhang et al. 2023; Tao et al. 2023). Additionally, rosavin (a cinnamyl alcohol glycoside) is regarded as a quality-determining compound, and its activities have been discussed by Wang et al. (2023). Another study identified FAE-20 and β-sitosterol glucoside as important R. rosea compounds for learning in Drosophila melanogaster larvae (Michels et al. 2020). The exhibited learning effect does not require known dopaminergic reward neurons and was also shown for synthetic FAE-20, not only in Drosophila but also in mouse models (Michels et al. 2018).
Aside from R. rosea, R. crenulata (used in traditional Tibetan medicine) has gained recent attention because of its pharmacological and medicinal effects. A 70% ethanolic extract of R. crenulata dried roots exhibited better regulating effects on senescence, which was correlated with antioxidant activities, in assays of senescence-associated β-galactosidase staining and lifespan in LO2 cells (Liang et al. 2023b). The strongest antibacterial effects were found for an ethyl acetate extract of R. crenulata roots; the ethyl acetate and crude ethanolic extract exhibited the highest antioxidant activities (Zhong et al. 2020).
Antibacterial activity of Rhodiola sp.
A variety of high-quality herbal medicines from Rhodiola, such as capsules, tablets, and oral liquids, are commercially available, reflecting the worldwide popularity of Rhodiola products (Furmanowa et al. 2002). Furthermore, Rhodiola root extracts are widely used in food additives and other commercial pharmaceutical preparations. This use indicates various potential applications of the plant, ranging from traditional medicine to modern pharmacology and the food industry (Nikoo et al. 2018). Numerous studies have investigated Rhodiola sp. bioactivity, each describing different potential beneficial effects, such as controlling microorganisms or influencing microbiota. The widespread use of Rhodiola sp. has led to the discovery of its numerous bioactive compounds. For example, Rattan et al. (2020) investigated the cultivation of Rhodiola sp. (e.g., R. rosea) in bioreactors to obtain higher metabolite yields and thus enable industrial applications. However, wild samples generally contain higher concentrations of bioactive compounds (Rattan et al. 2020).
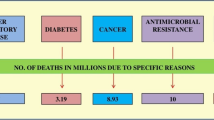
Increasing antimicrobial resistance, fueled by the (over)use of antibiotics and the slow development of new antimicrobials, is emerging as a major global threat to healthcare, agriculture, food production, and environment. The World Health Organization (WHO) predicts that antibiotic resistance will be the leading cause of death worldwide by 2050, especially as the (over)use of antibiotics in human and veterinary medicine contributes to the spread of resistant pathogens (WHO 2023). Therefore, the urgency to find innovative alternative strategies has led to the exploration of natural alternatives, such as extracts and secondary metabolites from various plants, including Rhodiola sp., as potential antimicrobial agents (Zhong et al. 2020).
Rhodiola sp. are known for their diverse bioactive compounds, including phenols, phenylpropanoids, and flavonoids, as described in the previous section. The most studied compounds include salidroside, rosavin, and tyrosol, which are predominantly found in roots and rhizomes (Furmanowa et al. 2002; Peschel et al. 2013; Nikoo et al. 2018). Due to their unique bioactive profiles, Rhodiola sp. are promising candidates for developing novel antimicrobials and thereby addressing the challenges of antimicrobial resistance. Additionally, their bioactive compounds act as resistance-modifying agents that increase the efficacy of existing antibiotics. In the context of food safety, the antioxidant and antimicrobial properties of bioactive Rhodiola compounds may represent natural preservatives in the food industry, which is in line with the growing consumer preference for natural additives (Nikoo et al. 2018). Studies such as that by Kosakowska et al. (2018) have highlighted the antioxidant and antibacterial activities of R. rosea ethanolic extracts, which are rich in phenolic compounds such as salidroside, tyrosol, and rosavin derivatives.
This review highlights the diverse research on antibacterial effects of Rhodiola sp., emphasizing the different species, plant parts, solvents, extracts, bioactive constituents, methods for determining antimicrobial activity, and target microorganisms (Table 2). The use of different parts of the Rhodiola plant, such as roots, rhizomes, and stems, has been a focus of research. For example, Furmanowa et al. (2002) investigated ethanolic extracts from R. rosea roots and rhizomes, whereas Ming et al. (2005) from dried stems.
Various solvents are used for extraction, including ethanol, water, n-hexane, ethyl acetate, and n-butanol. The choice of solvent influences the efficiency of the extraction and the resulting bioactivity of the compounds (Ming et al. 2005). Research has investigated both total extracts and specific fractions of Rhodiola. For example, fractions from ethanolic extracts, which are rich in distinctive bioactive compounds, have been investigated for their antibacterial properties (Kunčič et al. 2022). Various bioactive components in Rhodiola extracts, such as salidroside, rosavin, tyrosol, gossypetin-7-O-L-rhamnopyranoside, and rhodioflavonoside, have been identified and exhibit different levels of activity against microorganisms (Zaushintsena et al. 2020; Ming et al. 2005).
Methods such as liquid–liquid partitioning, disk diffusion tests, and minimum inhibitory concentration measurements, have been used to determine antimicrobial activity. These methods have provided insights into the efficacy of Rhodiola extracts against various microorganisms (Ming et al. 2005; Tsvetov et al. 2023), including the bacteria Staphylococcus aureus and Acinetobacter baumannii and fungi (Zaushintsena et al. 2020; Tsvetov et al. 2023). R. rosea extracts have been found to inhibit biofilm formation and extracellular polysaccharide synthesis in Streptococcus mutans and affect intercellular signaling in Campylobacter jejuni (Zhang et al. 2020; Šimunović et al. 2020). NADES-based R. rosea extracts showed bactericidal activity against cultures of Micrococcus luteus, Pseudomonas fluorescens, and Bacillus subtilis, highlighting the broad-spectrum antimicrobial potential of Rhodiola (Tsvetov et al. 2023). Rhodiola bioactive compounds have shown not only direct antimicrobial effects but also properties that alter resistance and increase the efficacy of existing antibiotics. Additionally, their antioxidant properties have potential applications in food safety as natural preservatives (Nikoo et al. 2018).
Mechanism of antibacterial action
Research on bioactive compounds from natural sources has uncovered a variety of substances with antimicrobial properties and has thus enabled decisive advances in combating microbes. However, these compounds exhibit diverse mechanisms of action, ranging from disruption of bacterial cell walls and microbial metabolism to interactions with microbial cell membranes and intracellular processes. This diversity in their modes of action underlines their potential for developing new approaches against microbial resistance and infection.
For example, certain Rhodiola compounds can disrupt bacterial cell walls and alter microbial metabolism, and thereby contributing to their antimicrobial efficacy. These compounds include salidroside and rosavin from R. rosea and R. crenulata, which showed antibacterial activity against Propionibacterium acnes and improved the survival rate of D. melanogaster by affecting the expression of antimicrobial peptide genes and decreasing the levels of reactive oxygen species (Ioset et al. 2011; Chiang et al. 2015). Other research has identified compounds with a broad spectrum of pharmacological activities, such as anti-inflammatory, antioxidant, and immunostimulatory effects, which are particularly effective against pathogens such as Aeromonas hydrophila by destroying bacterial cell walls and reducing virulence (Chiang et al. 2015; Chung et al. 2017; Zhang et al. 2021; Zhao et al. 2022a). In addition, some Rhodiola compounds have been shown to interfere with microbial cell processes and have improved antibacterial properties compared to conventional agents such as ferulic acid. This leads to the disruption of cell walls and biofilms, resulting in membrane hyperpolarization and leakage of cell contents (Chiang et al. 2015; Song et al. 2023). Their efficacy against multidrug-resistant strains is remarkable, particularly due to their ability to interact with microbial cell membranes, trigger cell lysis, and disrupt cytoplasmic membranes, thereby altering membrane permeability and stability (Chiang et al. 2015; Tagousop et al. 2018; Strugała et al. 2017).
Proanthocyanidins, which belong to the class of condensed tannins, have a sophisticated mode of action against microorganisms, primarily due to their structure as oligomers or polymers of monomeric flavan-3-ols, products of the flavonoid biosynthesis pathway. These phytochemicals, known for their potent antimicrobial properties, also provide a wide range of health benefits that include antioxidant, anticancer, antidiabetic, and neuroprotective effects, as detailed by Rauf et al. (2019).
Similarly, EGCG, a prominent component of R. rosea, is known for its versatile bioactive properties, which are particularly effective in combating bacterial growth and resistance. Its importance extends beyond microbial control to crucial aspects of food safety and preservation. EGCG shares its beneficial properties with other phenolic compounds such as epigallocatechin, gallic acid, and catechin (Zhang et al. 2022; Nikoo et al. 2018). These properties make EGCG and similar compounds valuable agents for addressing current public health and food safety challenges. The antibacterial effects of EGCG include binding to the peptidoglycan layers of Gram-positive bacteria and inducing oxidative stress in Gram-negative bacteria. It inhibits microbial efflux pumps, such as CmeABC and CmeDEF, in Campylobacter, which can restore the efficacy of macrolides in resistant strains (Nikoo et al. 2018; Smole Možina et al. 2011; Kurinčič et al. 2012). This aspect is particularly important as C. jejuni has shown resistance in food processing plants, leading to bacterial gastroenteritis and posing a challenge due to antibiotic resistance (Smole Možina et al. 2018; Taha-Abdelaziz et al. 2023). The potential of EGCG as a resistance-modifying agent, particularly in combination with novel antibiotics against Campylobacter strains, indicates new possibilities for its application in vitro and in vivo. This multi-faceted approach, ranging from direct interactions with bacteria to inhibition of efflux pumps, underlines the value of EGCG as an alternative to conventional antibiotics and requires further research to explore its full potential (Taha-Abdelaziz et al. 2023).
Recent advances in the fight against pathogens emphasize the effectiveness of natural antimicrobial formulations, particularly those combining organic acids and plant extracts. Studies such as those by Balta et al. (2021) and Kunčič et al. (2022) have highlighted the significant effect of such extracts on inhibiting quorum sensing in bacterial pathogens, particularly C. jejuni. The ethanolic extract of R. rosea effectively disrupts autoinducer-2-mediated intercellular signaling in C. jejuni, a crucial pathway for bacterial communication and pathogenicity. This finding indicates the potential of R. rosea extracts as antimicrobial agents that can attenuate bacterial virulence. The study by Šimunović et al. (2020) further supports this idea, demonstrating that natural (including R. rosea) extracts reduce quorum sensing in C. jejuni, leading to reduced surface adhesion and invasion of host cells. A luxS-deficient C. jejuni mutant exhibited reduced biofilm formation, motility, and virulence, underscoring the critical role of quorum sensing, particularly autoinducer-2 signaling, in the pathogenicity of the bacterium. In addition, EGCG has been recognized as an effective modulator of antibiotic resistance (Klančnik et al. 2012) and inhibitor of quorum sensing. EGCG also inhibits biofilm formation and thus further enhances the antimicrobial potential of these natural extracts.
Overall, these studies suggest a comprehensive approach to combating microbial threats, utilizing compounds such as EGCG and the inhibitory effects of R. rosea extracts on quorum sensing. Despite promising results, further extensive research is needed to fully understand the therapeutic potential and specific antimicrobial actions of these compounds (Elgamoudi and Korolik 2021; Castillo et al. 2015; Klančnik et al. 2021). This line of research provides promising opportunities for developing innovative antimicrobial strategies using natural extracts.
Rhodiola sp. and the gut microbiota (GM)
The complex ecosystem of microbes in the digestive tract, known as GM, plays a crucial role in various physiological and immunological functions, and GM imbalance is associated with numerous diseases. This complex relationship between GM and health is reflected in the effects of natural compounds such as Rhodiola extracts and salidroside. Recent studies (summarized in Table 3) show that these compounds can significantly affect the composition and metabolism of GM, with implications for health and disease management.
For example, R. crenulata and R. rosea have been observed to alter GM in mice and D. melanogaster, leading to more favorable bacterial profiles and potential anti-ageing benefits. The effects of salidroside on GM in various disease models have been associated with improved gut health and decreased inflammation. These changes in GM composition correlate with improved physical performance and alleviated memory impairment in certain experimental models. Beyond the specific changes in GM, the broader pharmacological effects of Rhodiola extracts and salidroside include roles in metabolic, cardiovascular, and neurological disorders, which are often mediated by GM. The evidence for these interactions and their effects are briefly outlined in Table 3, which provides an overview of the current state of knowledge on how Rhodiola and salidroside affect GM and contribute to health and disease management.
The roles of GM in both health and disease have been extensively studied in recent decades. GM is instrumental in the digestion and absorption of nutrients, especially indigestible carbohydrates, the synthesis of short-chain fatty acids, vitamins, amino acids, and secondary bile acids; and many other physiological and immune functions. It is an essential component of the host’s immune system and prevents pathogen colonization and allergy development. GM organisms can also significantly alter certain medications once ingested (Bull and Plummer 2014; Lynch and Pedersen 2016).
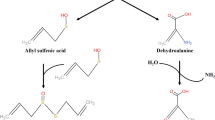
Aside from various phenolic compounds, Rhodiola sp. also contains cyanogenic glycosides, like lotaustralin (see Table 1). The mode of action of cyanogenic glycosides on microorganisms is closely linked to their enzymatic degradation, a process that mainly occurs in the human colon, an environment rich in GM. In this part of the digestive system, bacterial hydrolysis plays a crucial role in the degradation of cyanogenic glycosides and leads to the formation of hydrocyanic acid, which is known for its potential toxicity. This degradation involves two important steps: initial hydrolysis of cyanogenic glycosides to cyanohydrin and subsequent degradation to cyanide. Several factors influence the rate of cyanide production, including the presence of specific enzymes such as β-glucosidase, the sugar content in the glycoside molecule, and the stability of the cyanohydrin intermediate. Some cyanogenic glycosides (e.g., amygdalin, linustatin, and neolinustatin) are first hydrolyzed to simpler sugars (e.g., glucose), which then form compounds such as prunasin, linamarin, and lotaustralin. This process of hydrolysis and subsequent cyanide production is inevitably associated with interactions with GM. Although the formation of hydrocyanic acid suggests high toxicity, the actual effects of cyanogenic glycosides on GM are generally less severe than theoretically predicted. This is due to the complex interplay of metabolic factors within the colon, highlighting the nuanced relationship between these glycosides and the GM environment (Cressey and Reeve 2019).
Moreover, imbalanced GM has been associated with pathological gastrointestinal conditions (e.g., inflammatory bowel disease and irritable bowel syndrome), colorectal cancer, systemic disease manifestations (e.g., type 2 diabetes and obesity), and chronic diseases (e.g., gastrointestinal, respiratory, liver, and cardiovascular diseases) (Fan and Pedersen 2021). Owing to complex bidirectional communication along the microbiota–gut–brain axis, GM is also crucial for brain function. In addition to physiological disorders, GM dysbiosis can also cause mental illness. Studies suggest that abnormal microbiota and microbiota–gut–brain dysfunction may directly cause mental disorders, as treating GM can alleviate anxiety/depression (Liang et al. 2018). The research gap is highlighted in recent reviews on the interactions between natural products and GM microorganisms, particularly in relation to mood disorders (Korczak et al. 2023; Pferschy-Wenzig et al. 2022). Future studies evaluating the efficacy of medicinal plants (known as adaptogens, e.g., R. rosea), their isolated compounds, or the postbiotic metabolites formed from them in the treatment of disease symptoms should consider the bidirectional interaction between phytoconstituents and GM microorganisms. This well-established bidirectional interaction can influence the efficacy of natural products in the treatment of various diseases (Wilson and Nicholson 2017). The proven examples of R. rosea and R. crenulata extracts and salidroside modulating host GM are listed in Table 3. These studies suggest that GM may mediate the therapeutic activity of Rhodiola preparations in alleviating disease symptoms as diverse as intestinal inflammation, metabolic syndromes, and neurodegenerative diseases and in exerting anti-fat effects.
Potential applications and future perspectives
The increasing resistance of bacteria to conventional antibiotics has drawn scientific attention to the potential of Rhodiola sp. extracts and their bioactive components as antibacterial agents. This line of research, underscored by studies by Altantsetseg et al. (2007), Kurinčič et al. (2012), Klančnik et al. (2021), and Zhang et al. (2022), is becoming increasingly important in addressing the challenges posed by antimicrobial resistance. A major advantage of using Rhodiola sp. or their derivatives lies in their natural origin, which represents a more organic and potentially safer alternative to synthetic additives, especially in the context of food safety and preservation. These natural antimicrobials improve the efficacy of existing antibiotics and provide a crucial solution to the problem of bacterial multidrug resistance. In addition, the use of Rhodiola sp. as additives in animal feed, disinfectants in packaging, and inhibitors of bacterial growth is becoming a promising strategy. As Elgamoudi and Korolik (2021) suggest, this approach not only ensures safer food production “from farm to fork” but can also improve the effectiveness of antibiotics in treating foodborne infections. However, despite these promising developments, more extensive research is still needed to fully understand the antibacterial mechanisms of Rhodiola sp. extracts and their active compounds, as emphasized by Zhang et al. (2022). Such further research is essential for maximizing the potential of Rhodiola sp. to combat bacterial resistance and improve food safety.
Recent research has investigated the use of plant-based nanoparticles as an innovative alternative to conventional antibiotics, particularly in the treatment of multi-drug-resistant bacterial infections. These nanoparticles, especially when derived from plants, have the advantage of being less toxic and more effective against bacterial pathogens. The development of antimicrobial nanotherapeutics using plant-derived nanoparticles has been characterized by their lower toxicity and environmental impact (Anand et al. 2022). A notable study by Singh et al. (2018) investigated the use of R. rosea extracts for the synthesis of gold and silver nanoparticles. These nanoparticles could inhibit the formation of biofilms by bacteria such as Pseudomonas aeruginosa and Escherichia coli. This research not only presents an environmentally friendly method of synthesizing nanoparticles but also highlights the stability and antimicrobial efficacy of these nanoparticles. The presence of bioactive R. rosea compounds on nanoparticle surfaces significantly enhances antimicrobial functionality and shows much promise for medical applications (Singh et al. 2018; Anand et al. 2022).
The further development of in vitro systems and bioreactor cultures for Rhodiola sp., as demonstrated in the work of Rattan et al. (2020) and Marchev et al. (2016), represents a sustainable and feasible method for improving the production of bioactive substances. This approach particularly focuses on R. rosea and the production of secondary metabolites with pharmaceutical value. Supported by an improved understanding of R. rosea biosynthetic pathways and enzymes involved in metabolite production, these biotechnological methods are very promising for the sustainable production of secondary metabolites. Furthermore, Chiang et al. (2015) underscore the anti-ageing and antioxidant properties of Rhodiola sp. and emphasize its potential for developing treatments or supplements that promote longevity and combat age-related diseases. This is consistent with research into in vitro R. rosea cultivation as a means of providing sustainable sources of these valuable secondary metabolites. The prospect of creating green cell factories for Rhodiola compounds not only promotes pharmaceutical development but is also in line with environmentally friendly practices in the cultivation and use of medicinal plants.
Furthermore, ongoing research highlights the need to further explore the antibacterial mechanisms of Rhodiola extracts and their active compounds, while emphasizing the need for safe and environmentally friendly methods for their use (Anand et al. 2022). Given that Rhodiola sp. often grow slowly and are endangered in their natural habitats, there is a growing need for alternative sustainable sources of important phytochemicals. This need can be met through bioengineering and sustainable production techniques, as proposed by Kasprzyk et al. (2022). For example, a study by Nikoo et al. (2018) investigated a modified EGCG molecule with improved solubility and antioxidant activity achieved by methods such as nano-encapsulation and spray drying.
Conducting toxicity studies in vitro and in animal models is essential to demonstrate the safety of Rhodiola products, especially when considering their use in dietary supplements, food ingredients, and pharmaceuticals. As Nikoo et al. (2018) and Kasprzyk et al. (2022) emphasize, this research is crucial for ensuring the safe use of these compounds in human nutrition and therapy. These extracts are promising for the development of new drugs and dietary supplements, especially those with antitumor, antimicrobial, and antioxidant properties, thus expanding the scope of Rhodiola in health and medicine. The growing interest in Rhodiola sp. and promising results underline the need for continued research and development in this area. Such sustained efforts are crucial to fully exploit the potential of Rhodiola sp. in medicine and biotechnology.
Conclusions
Our research on Rhodiola sp. shows a unique integration of traditional herbal knowledge and advanced pharmacological research. Species such as R. rosea and R. imbricata are rich sources of bioactive compounds, particularly salidrosides, rosavins, and phenolics, which have significant antimicrobial potential. These phytochemicals are effective against a broad spectrum of pathogenic microorganisms and thus importantly contribute to global efforts to combat antibiotic resistance. Additionally, these compounds are not only potent antimicrobials but also serve as resistance-modifying agents that can increase the efficacy of existing antibiotics. This dual role of Rhodiola compounds offers promising opportunities for developing innovative antimicrobial strategies and therapies. In addition to their antimicrobial properties, a key aspect of the therapeutic potential of Rhodiola sp. lies in their effects on GM. The modulation of GM by Rhodiola extracts and their active constituents (e.g., salidroside) affects health in general. Changes in GM composition correlate with improved physiological and metabolic conditions, underlining the comprehensive health benefits of Rhodiola sp.
This review has revealed a clear need for more in-depth research. Future studies should explore the complex interactions between the antimicrobial effects and effects on GM of Rhodiola phytochemical constituents. This research is crucial for the development of holistic treatment approaches that fully utilize the phytochemical spectrum of Rhodiola sp. Overall, Rhodiola sp. represents a significant natural resource in the ongoing fight against microbial pathogens and antimicrobial resistance, offering a versatile approach to health and well-being. Its rich phytochemical profile combined with its effect on GM makes it a valuable candidate for future pharmacological applications and health management strategies.
References
Abdallah EM, Alhatlani BY, de Paula Menezes R, Martins CHG (2023) Back to nature: medicinal plants as promising sources for antibacterial drugs in the post-antibiotic era. Plants (basel) 12(17):3077. https://doi.org/10.3390/plants12173077
Altantsetseg K, Przybyl JL, Weglarz Z, Geszprych A (2007) Content of biologically active compounds in roseroot (Rhodiola sp.) raw material of different derivation. Herba Pol 53(4):20–26
Anand U, Carpena M, Kowalska-Góralska M, Garcia-Perez P, Sunita K, Bontempi E et al (2022) Safer plant-based nanoparticles for combating antibiotic resistance in bacteria: a comprehensive review on its potential applications, recent advances, and future perspective. Sci Total Environ 821:153472. https://doi.org/10.1016/j.scitotenv.2022.153472
Anheyer D, Schröter M, Dobos G, Cramer H (2021) Traditionelle nutzung von Arzneimitteln aus Rhodiola rosea (Rosenwurz). Z Phytother 42(06):280–291. https://doi.org/10.1055/a-1540-9820
Bai X-L, Deng X-L, Wu G-J, Li W-J, Jin S (2019) Rhodiola and salidroside in the treatment of metabolic disorders. Mini Rev Med Chem 19(19):1611–1626. https://doi.org/10.2174/1389557519666190903115424
Balta I, Linton M, Pinkerton L, Kelly C, Stef L, Pet I et al (2021) (2021) The effect of natural antimicrobials against Campylobacter spp. and its similarities to Salmonella spp., Listeria spp., Escherichia coli, Vibrio spp., Clostridium spp. and Staphylococcus spp. Food Control 121:107745. https://doi.org/10.1016/j.foodcont.2020.107745
Bernatoniene J, Jakstas V, Kopustinskiene DM (2023) Phenolic compounds of Rhodiola rosea L. as the potential alternative therapy in the treatment of chronic diseases. Int J Mol Sci 24(15):12293. https://doi.org/10.3390/ijms241512293
Bull MJ, Plummer NT (2014) Part 1: the human gut microbiome in health and disease. Integr Med (encinitas) 13(6):17–22
Calabrese EJ, Dhawan G, Kapoor R, Agathokleous E, Calabrese V (2023) Rhodiola rosea and salidroside commonly induce hormesis, with particular focus on longevity and neuroprotection. Chem Biol Interact 380:110540. https://doi.org/10.1016/j.cbi.2023.110540
Castillo S, Heredia N, García S (2015) 2(5H)-Furanone, epigallocatechin gallate, and a citric-based disinfectant disturb quorum-sensing activity and reduce motility and biofilm formation of Campylobacter jejuni. Folia Microbiol (praha) 60(1):89–95. https://doi.org/10.1007/s12223-014-0344-0
Chiang H-M, Chen H-C, Wu C-S, Wu P-Y, Wen K-C (2015) Rhodiola plants: chemistry and biological activity. J Food Drug Anal 23(3):359–369. https://doi.org/10.1016/j.jfda.2015.04.007
Chung D, Kim SY, Ahn J-H (2017) Production of three phenylethanoids, tyrosol, hydroxytyrosol, and salidroside, using plant genes expressing in Escherichia coli. Sci Rep 7(1):2578. https://doi.org/10.1038/s41598-017-02042-2
Cressey P, Reeve J (2019) Metabolism of cyanogenic glycosides: a review. Food Chem Toxicol 125:225–232. https://doi.org/10.1016/j.fct.2019.01.002
Dimpfel W, Schombert L, Panossian AG (2018) Assessing the quality and potential efficacy of commercial extracts of Rhodiola rosea L. by analyzing the salidroside and rosavin content and the electrophysiological activity in hippocampal long-term potentiation, a synaptic model of memory. Front Pharmacol 9:425. https://doi.org/10.3389/fphar.2018.00425
Dong T, Liu H, Sha Y, Sun L (2023) A comparative study of phytochemical metabolites and antioxidant properties of Rhodiola. Arab J Chem 16(1):104420. https://doi.org/10.1016/j.arabjc.2022.104420
Elgamoudi BA, Korolik V (2021) Campylobacter biofilms: potential of natural compounds to disrupt Campylobacter jejuni transmission. Int J Mol Sci 22(22):12159. https://doi.org/10.3390/ijms222212159
EMA (2023) European Union herbal monograph on Rhodiola rosea L., rhizoma et radix. https://www.ema.europa.eu/en/medicines/herbal/rhodiolae-roseae-rhizoma-et-radix
Evstatieva L, Todorova M, Antonova D, Staneva J (2010) Chemical composition of the essential oils of Rhodiola rosea L. of three different origins. Pharmacogn Mag 6(24):256–258. https://doi.org/10.4103/0973-1296.71782
Fan Y, Pedersen O (2021) Gut microbiota in human metabolic health and disease. Nat Rev Microbiol 19(1):55–71. https://doi.org/10.1038/s41579-020-0433-9
Furmanowa M, Starosciak B, Lutomski J, Kozlowski J, Urbanska N, Krajewska-Patan A et al (2002) Antimicrobial effect of Rhodiola rosea L. roots and callus extracts on some strains of Staphylococcus aureus. Herba Pol 48(1):23–31
Galambosi B, Galambosi Z, Hethelyi E, Szoke E, Volodin V, Poletaeva I, Iljina I (2010) Importance and quality of roseroot (Rhodiola rosea L.) growing in the European North. Z Arznei- Gewuerzpflanzen 15(4):160–169
Gao Z, Zhan H, Zong W, Sun M, Linghu L, Wang G et al (2023) Salidroside alleviates acetaminophen-induced hepatotoxicity via Sirt1-mediated activation of Akt/Nrf2 pathway and suppression of NF-κB/NLRP3 inflammasome axis. Life Sci 327:121793. https://doi.org/10.1016/j.lfs.2023.121793
Gonçalves ASC, Leitão MM, Simões M, Borges A (2023) The action of phytochemicals in biofilm control. Nat Prod Rep 40(3):595–627. https://doi.org/10.1039/D2NP00053A
Hai Z, Wu Y, Ning Z (2023) Salidroside attenuates atrial fibrosis and atrial fibrillation vulnerability induced by angiotensin-II through inhibition of LOXL2-TGF-β1-Smad2/3 pathway. Heliyon 9(11):e21220. https://doi.org/10.1016/j.heliyon.2023.e21220
Ioset KN, Nyberg NT, van Diermen D, Malnoe P, Hostettmann K, Shikov AN, Jaroszewski JW (2011) Metabolic profiling of Rhodiola rosea rhizomes by 1H NMR spectroscopy. Phytochem Anal 22(2):158–165. https://doi.org/10.1002/pca.1262
IPNI (2023) The international plant names index and world checklist of vascular plants. http://www.ipni.org and https://powo.science.kew.org/
Jafari M, Juanson Arabit JG, Courville R, Kiani D, Chaston JM, Nguyen CD et al (2022) The impact of Rhodiola rosea on biomarkers of diabetes, inflammation, and microbiota in a leptin receptor-knockout mouse model. Sci Rep 12(1):10581. https://doi.org/10.1038/s41598-022-14241-7
Jiao Y, Zhao Z, Li X, Li L, Xiao D, Wan S et al (2023) Salidroside ameliorates memory impairment following long-term ethanol intake in rats by modulating the altered intestinal microbiota content and hippocampal gene expression. Front Microbiol 14:1172936. https://doi.org/10.3389/fmicb.2023.1172936
Jin Y, Park DW, Li X, Li D, Row KH (2010) Primary study of volatiles composition of Rhodiola sachalinensis by using gas chromatography and mass spectrometry (GC/MS). Korean J Chem Eng 27(4):1262–1268. https://doi.org/10.1007/s11814-010-0170-2
Jin M, Wang C, Xu Y, Zhang Z, Wu X, Ye R et al (2022) Pharmacological effects of salidroside on central nervous system diseases. Biomed Pharmacother 156:113746. https://doi.org/10.1016/j.biopha.2022.113746
Kasprzyk PG, Vickery C, Ye M, Sewastianik M, Gong W, Ding S et al (2022) Safety of a sustainably produced, bioengineered, nature-identical salidroside compound. Nutrients 14(11):2330. https://doi.org/10.3390/nu14112330
Klančnik A, Možina SS, Zhang Q (2012) Anti-Campylobacter activities and resistance mechanisms of natural phenolic compounds in Campylobacter. PLoS ONE 7(12):e51800. https://doi.org/10.1371/journal.pone.0051800
Klančnik A, Šimunović K, Sterniša M, Ramić D, Smole Možina S, Bucar F (2021) Anti-adhesion activity of phytochemicals to prevent Campylobacter jejuni biofilm formation on abiotic surfaces. Phytochem Rev 20(1):55–84. https://doi.org/10.1007/s11101-020-09669-6
Korczak M, Pilecki M, Granica S, Gorczynska A, Pawłowska KA, Piwowarski JP (2023) Phytotherapy of mood disorders in the light of microbiota-gut-brain axis. Phytomedicine 111:154642. https://doi.org/10.1016/j.phymed.2023.154642
Kosakowska O, Bączek K, Przybył JL, Pióro-Jabrucka E, Czupa W, Synowiec A et al (2018) Antioxidant and antibacterial activity of roseroot (Rhodiola rosea L.) dry extracts. Molecules 23(7):1767. https://doi.org/10.3390/molecules23071767
Kunčič A, Bucar F, Smole Možina S (2022) Rhodiola rosea reduces intercellular signaling in Campylobacter jejuni. Antibiotics (basel) 11(9):1220. https://doi.org/10.3390/antibiotics11091220
Kurinčič M, Klančnik A, Smole Možina S (2012) Epigallocatechin gallate as a modulator of Campylobacter resistance to macrolide antibiotics. Int J Antimicrob Agents 40(5):467–471. https://doi.org/10.1016/j.ijantimicag.2012.07.015
Labachyan KE, Kiani D, Sevrioukov EA, Schriner SE, Jafari M (2018) The impact of Rhodiola rosea on the gut microbial community of Drosophila melanogaster. Gut Pathog 10:12. https://doi.org/10.1186/s13099-018-0239-8
Langeder J, Grienke U (2021) A supercritical fluid workflow for the quality assessment of herbal drugs and commercial preparations from Rhodiola rosea. Phytochem Anal 32(6):982–991. https://doi.org/10.1002/pca.3040
Lei Y, Nan P, Tsering T, Bai Z, Tian C, Zhong Y (2003) Chemical composition of the essential oils of two Rhodiola species from Tibet. Z Naturforsch C J Biosci 58(3–4):161–164. https://doi.org/10.1515/znc-2003-3-402
Lei Y, Peng N, Tsering T, Wang L, Liu S, Zhong Y (2004) Interpopulation variability of rhizome essential oils in Rhodiola crenulata from Tibet and Yunnan. China Biochem Syst Ecol 32(6):611–614. https://doi.org/10.1016/j.bse.2003.10.007
Li L, Yao W (2023) The therapeutic potential of salidroside for Parkinson’s disease. Planta Med 89(4):353–363. https://doi.org/10.1055/a-1948-3179
Li Y, Pham V, Bui M, Di Song L, Wu C, Walia A et al (2017) Rhodiola rosea L: an herb with anti-stress, anti-aging, and immunostimulating properties for cancer chemoprevention. Curr Pharmacol Rep 3(6):384–395. https://doi.org/10.1007/s40495-017-0106-1
Li H, Xi Y, Xin X, Tian H, Hu Y (2020) Salidroside improves high-fat diet-induced non-alcoholic steatohepatitis by regulating the gut microbiota-bile acid-farnesoid X receptor axis. Biomed Pharmacother 124:109915. https://doi.org/10.1016/j.biopha.2020.109915
Liang S, Wu X, Hu X, Wang T, Jin F (2018) Recognizing depression from the microbiota-gut-brain axis. Int J Mol Sci 19(6):1592. https://doi.org/10.3390/ijms19061592
Liang Q, Guo R, Tsao J-R, He Y, Wang C, Jiang J et al (2023a) Salidroside alleviates oxidative stress in dry eye disease by activating autophagy through AMPK-Sirt1 pathway. Int Immunopharmacol 121:110397. https://doi.org/10.1016/j.intimp.2023.110397
Liang T, Zhou J, Jing P, He Z, Jiao S, Zhao W et al (2023b) Anti-senescence effects of Rhodiola crenulata extracts on LO2 cells and bioactive compounds. J Ethnopharmacol 306:116179. https://doi.org/10.1016/j.jep.2023.116179
Liu C, Dong S, Wang X, Xu H, Yang X, Wu S et al (2023a) Research progress of polyphenols in nanoformulations for antibacterial application. Mater Today Bio 21:100729. https://doi.org/10.1016/j.mtbio.2023.100729
Liu J, Cai J, Fan P, Dong X, Zhang N, Tai J, Cao Y (2023b) Salidroside alleviates dextran sulfate sodium-induced colitis in mice by modulating the gut microbiota. Food Funct 14(16):7506–7519. https://doi.org/10.1039/d3fo01929b
Liu J, Cai J, Fan P, Dong X, Zhang N, Tai J, Cao Y (2023c) Salidroside protects mice from high-fat diet-induced obesity by modulating the gut microbiota. Int Immunopharmacol 120:110278. https://doi.org/10.1016/j.intimp.2023.110278
Liu X, Zhou M, Dai Z, Luo S, Shi Y, He Z, Chen Y (2023d) Salidroside alleviates ulcerative colitis via inhibiting macrophage pyroptosis and repairing the dysbacteriosis-associated Th17/Treg imbalance. Phytother Res 37(2):367–382. https://doi.org/10.1002/ptr.7636
Lynch SV, Pedersen O (2016) The human intestinal microbiome in health and disease. N Engl J Med 375(24):2369–2379. https://doi.org/10.1056/NEJMra1600266
Ma G, Li W, Dou D, Chang X, Bai H, Satou T et al (2006) Rhodiolosides A-E, monoterpene glycosides from Rhodiola rosea. Chem Pharm Bull (tokyo) 54(8):1229–1233. https://doi.org/10.1248/cpb.54.1229
Ma L, Jiang X, Huang Q, Chen W, Zhang H, Pei H et al (2023) Traditional Chinese medicine for the treatment of Alzheimer’s disease: a focus on the microbiota–gut–brain axis. Biomed Pharmacother 165:115244. https://doi.org/10.1016/j.biopha.2023.115244
Marchev AS, Dinkova-Kostova AT, György Z, Mirmazloum I, Aneva IY, Georgiev MI (2016) Rhodiola rosea L.: from golden root to green cell factories. Phytochem Rev 15(4):515–536. https://doi.org/10.1007/s11101-016-9453-5
Marchev AS, Koycheva IK, Aneva IY, Georgiev MI (2020) Authenticity and quality evaluation of different Rhodiola species and commercial products based on NMR-spectroscopy and HPLC. Phytochem Anal 31(6):756–769. https://doi.org/10.1002/pca.2940
Michels B, Zwaka H, Bartels R, Lushchak O, Franke K, Endres T et al (2018) Memory enhancement by ferulic acid ester across species. Sci Adv 4(10):eaat6994. https://doi.org/10.1126/sciadv.aat6994
Michels B, Franke K, Weiglein A, Sultani H, Gerber B, Wessjohann LA (2020) Rewarding compounds identified from the medicinal plant Rhodiola rosea. J Exp Biol 223(16):jeb223982. https://doi.org/10.1242/jeb.223982
Ming DS, Hillhouse BJ, Guns ES, Eberding A, Xie S, Vimalanathan S, Towers GHN (2005) Bioactive compounds from Rhodiola rosea (Crassulaceae). Phytother Res 19(9):740–743. https://doi.org/10.1002/ptr.1597
Nikoo M, Regenstein JM, Ahmadi Gavlighi H (2018) Antioxidant and antimicrobial activities of (-)-epigallocatechin-3-gallate (EGCG) and its potential to preserve the quality and safety of foods. Compr Rev Food Sci Food Saf 17(3):732–753. https://doi.org/10.1111/1541-4337.12346
Olennikov DN (2023) New metabolites of Rhodiola rosea. II. Hibiscetin glycosides. Chem Nat Compd 59(2):254–258. https://doi.org/10.1007/s10600-023-03969-x
Olennikov DN, Chirikova NK, Vasilieva AG, Fedorov IA (2020) LC-MS profile, gastrointestinal and gut microbiota stability and antioxidant activity of Rhodiola rosea herb metabolites: a comparative study with subterranean organs. Antioxidants (basel) 9(6):526. https://doi.org/10.3390/antiox9060526
Panossian A, Wikman G, Sarris J (2010) Rosenroot (Rhodiola rosea): traditional use, chemical composition, pharmacology and clinical efficacy. Phytomedicine 17(7):481–493. https://doi.org/10.1016/j.phymed.2010.02.002
Peschel W, Prieto JM, Karkour C, Williamson EM (2013) Effect of provenance, plant part and processing on extract profiles from cultivated European Rhodiola rosea L. for medicinal use. Phytochemistry 86:92–102. https://doi.org/10.1016/j.phytochem.2012.10.005
Pferschy-Wenzig E-M, Pausan MR, Ardjomand-Woelkart K, Röck S, Ammar RM, Kelber O et al (2022) Medicinal plants and their impact on the gut microbiome in mental health: a systematic review. Nutrients 14(10):2111. https://doi.org/10.3390/nu14102111
PWO (2023) Plants of the World online. https://powo.science.kew.org/taxon/urn:lsid:ipni.org:names:30061153-2
Rattan S, Sood A, Kumar P, Kumar A, Kumar D, Warghat AR (2020) Phenylethanoids, phenylpropanoids, and phenolic acids quantification vis-à-vis gene expression profiling in leaf and root derived callus lines of Rhodiola imbricata (Edgew.). Ind Crops Prod 154:112708. https://doi.org/10.1016/j.indcrop.2020.112708
Rauf A, Imran M, Abu-Izneid T, Iahtisham-Ul-Haq PS, Pan X et al (2019) Proanthocyanidins: a comprehensive review. Biomed Pharmacother 116:108999. https://doi.org/10.1016/j.biopha.2019.108999
Rohloff J (2002) Volatiles from rhizomes of Rhodiola rosea L. Phytochemistry 59(6):655–661. https://doi.org/10.1016/s0031-9422(02)00004-3
CAS SciFindern (2023) CAS, American Chemical Society. https://scifinder-n.cas.org/
Shi J, Zhao Q, Hao DD, Miao HX, Wan S, Zhou CH et al (2022) Gut microbiota profiling revealed the regulating effects of salidroside on iron metabolism in diabetic mice. Front Endocrinol (lausanne) 13:1014577. https://doi.org/10.3389/fendo.2022.1014577
Šimunović K, Ramić D, Xu C, Smole Možina S (2020) Modulation of Campylobacter jejuni motility, adhesion to polystyrene surfaces, and invasion of INT407 cells by quorum-sensing inhibition. Microorganisms 8(1):104. https://doi.org/10.3390/microorganisms8010104
Singh P, Pandit S, Beshay M, Mokkapati VRSS, Garnaes J, Olsson ME et al (2018) Anti-biofilm effects of gold and silver nanoparticles synthesized by the Rhodiola rosea rhizome extracts. Artif Cells Nanomed Biotechnol 46(sup3):S886–S899. https://doi.org/10.1080/21691401.2018.1518909
Smole Možina S, Kurinčič M, Klančnik A, Mavri A (2011) Campylobacter and its multi-resistance in the food chain. Trends Food Sci Technol 22(2):91–98. https://doi.org/10.1016/j.tifs.2010.09.003
Smole Možina S, Klančnik A, Kovac J, Jeršek B, Bucar F (2018) Antimicrobial natural products against Campylobacter. In: Mérillon J-M, Riviere C (eds) Natural antimicrobial agents. Springer, Cham, pp 3–30. https://doi.org/10.1007/978-3-319-67045-4_1
Song W, Xin J, Yu C, Xia C, Pan Y (2023) Alkyl ferulic acid esters: evaluating their structure and antibacterial properties. Front Microbiol 14:1135308. https://doi.org/10.3389/fmicb.2023.1135308
Strugała P, Tronina T, Huszcza E, Gabrielska J (2017) Bioactivity in vitro of quercetin glycoside obtained in Beauveria bassiana culture and its interaction with liposome membranes. Molecules 22(9):1520. https://doi.org/10.3390/molecules22091520
Sun T, Yang J, Lu L (2022) Salidroside improves antibiotic-induced gut microbiota disturbance and low levels of short-chain fatty acids in mice. Foods 11(19):3073. https://doi.org/10.3390/foods11193073
Tagousop CN, Tamokou J-D, Ekom SE, Ngnokam D, Voutquenne-Nazabadioko L (2018) Antimicrobial activities of flavonoid glycosides from Graptophyllum grandulosum and their mechanism of antibacterial action. BMC Complement Altern Med 18(1):252. https://doi.org/10.1186/s12906-018-2321-7
Taha-Abdelaziz K, Singh M, Sharif S, Sharma S, Kulkarni RR, Alizadeh M et al (2023) Intervention strategies to control Campylobacter at different stages of the food chain. Microorganisms 11(1):113. https://doi.org/10.3390/microorganisms11010113
Tang Y, Friesen JB, Lankin DC, McAlpine JB, Nikolić D, Chen S-N, Pauli GF (2023) Geraniol-derived monoterpenoid glucosides from Rhodiola rosea: resolving structures by QM-HifSA methodology. J Nat Prod 86(2):256–263. https://doi.org/10.1021/acs.jnatprod.2c00836
Tao H, Wu X, Cao J, Peng Y, Wang A, Pei J et al (2019) Rhodiola species: a comprehensive review of traditional use, phytochemistry, pharmacology, toxicity, and clinical study. Med Res Rev 39(5):1779–1850. https://doi.org/10.1002/med.21564
Tao L, Liang Z-F, Miao L, Guo Y-J, Li Y, Liu Y-L et al (2023) Mechanism of salidroside against coronary artery disease by network pharmacology analysis. BMC Complement Med Ther 23(1):194. https://doi.org/10.1186/s12906-023-04027-3
Tinsley GM, Jagim AR, Potter GDM, Garner D, Galpin AJ (2024) Rhodiola rosea as an adaptogen to enhance exercise performance: a review of the literature. Br J Nutr 131(3):461–473. https://doi.org/10.1017/S0007114523001988
Tsvetov N, Paukshta O, Fokina N, Volodina N, Samarov A (2023) Application of natural deep eutectic solvents for extraction of bioactive components from Rhodiola rosea (L.). Molecules 28(2):912. https://doi.org/10.3390/molecules28020912
Waditzer M, Bucar F (2021) Flavonoids as inhibitors of bacterial efflux pumps. Molecules 26(22):6904. https://doi.org/10.3390/molecules26226904
Wang Y, Tao H, Huang H, Xiao Y, Wu X, Li M et al (2021) The dietary supplement Rhodiola crenulata extract alleviates dextran sulfate sodium-induced colitis in mice through anti-inflammation, mediating gut barrier integrity and reshaping the gut microbiome. Food Funct 12(7):3142–3158. https://doi.org/10.1039/d0fo03061a
Wang S, Feng Y, Zheng L, He P, Tan J, Cai J et al (2023) Rosavin: research advances in extraction and synthesis, pharmacological activities and therapeutic effects on diseases of the characteristic active ingredients of Rhodiola rosea L. Molecules 28(21):7412. https://doi.org/10.3390/molecules28217412
WFO (2023a) World Flora Online, Rhodiola L. http://www.worldfloraonline.org/taxon/wfo-4000033013
WFO (2023b) World Flora Online, Rhodiola sachalinensis Boriss. http://www.worldfloraonline.org/taxon/wfo-0000399409
WFO (2023c) World Flora Online, Rhodiola sacra (Raym.-Hamet) Fu. http://www.worldfloraonline.org/taxon/wfo-0001300649
WHO (2023) World Health Organization, Antimicrobial resistance. https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance
Wilson ID, Nicholson JK (2017) Gut microbiome interactions with drug metabolism, efficacy, and toxicity. Transl Res 179:204–222. https://doi.org/10.1016/j.trsl.2016.08.002
Xie Z, Lu H, Yang S, Zeng Y, Li W, Wang L et al (2020) Salidroside attenuates cognitive dysfunction in senescence-accelerated mouse prone 8 (SAMP8) mice and modulates inflammation of the gut-brain axis. Front Pharmacol 11:568423. https://doi.org/10.3389/fphar.2020.568423
Yang Y, Liu Z, Feng Z, Jiang J, Zhang P (2012) Lignans from the root of Rhodiola crenulata. J Agric Food Chem 60(4):964–972. https://doi.org/10.1021/jf204660c
Yang Z, Zeng G, Peng L, Zhang M, Cheng J, Zhan R (2015) Preliminary study on effect of Rhodiolae crenulatae radix et rhizoma cell wall-broken decoction pieces on intestinal flora of mice. Zhongguo Zhong Yao Za Zhi 40(15):3053–3058
Yousef GG, Grace MH, Cheng DM, Belolipov IV, Raskin I, Lila MA (2006) Comparative phytochemical characterization of three Rhodiola species. Phytochemistry 67(21):2380–2391. https://doi.org/10.1016/j.phytochem.2006.07.026
Yuan Y, Wu X, Zhang X, Hong Y, Yan H (2019) Ameliorative effect of salidroside from Rhodiola rosea L. on the gut microbiota subject to furan-induced liver injury in a mouse model. Food Chem Toxicol 125:333–340. https://doi.org/10.1016/j.fct.2019.01.007
Zapesochnaya GG, Kurkin VA (1983) The flavonoids of the rhizomes of Rhodiola rosea II. A. flavonolignan and glycosides of herbacetin. Chem Nat Compd 19(1):21–29. https://doi.org/10.1007/BF00579955
Zaushintsena A, Babich O, Bryukhachev E, Milenteva I, Starcev A (2020) Opportunities for using biologically active substances Rhodiola rosea L. in the production of functional food with consideration for antimicrobial activity. E3S Web Conf 176:1011. https://doi.org/10.1051/e3sconf/202017601011
Zhang Z, Liu Y, Lu M, Lyu X, Gong T, Tang B et al (2020) Rhodiola rosea extract inhibits the biofilm formation and the expression of virulence genes of cariogenic oral pathogen Streptococcus mutans. Arch Oral Biol 116:104762. https://doi.org/10.1016/j.archoralbio.2020.104762
Zhang X, Xie L, Long J, Xie Q, Zheng Y, Liu K, Li X (2021) Salidroside: a review of its recent advances in synthetic pathways and pharmacological properties. Chem Biol Interact 339:109268. https://doi.org/10.1016/j.cbi.2020.109268
Zhang S, Jiang S, Deng N, Zheng B, Li T, Liu RH (2022) Phytochemical profiles, antioxidant activity and antiproliferative mechanism of Rhodiola rosea L. phenolic extract. Nutrients 14(17):3602. https://doi.org/10.3390/nu14173602
Zhang N, Nao J, Dong X (2023) Neuroprotective mechanisms of salidroside in Alzheimer′s disease: a systematic review and meta-analysis of preclinical studies. J Agric Food Chem 71(46):17597–17614. https://doi.org/10.1021/acs.jafc.3c06672
Zhao C-C, Wu X-Y, Yi H, Chen R, Fan G (2021) The therapeutic effects and mechanisms of salidroside on cardiovascular and metabolic diseases: an updated review. Chem Biodivers 18(7):e2100033. https://doi.org/10.1002/cbdv.202100033
Zhao L, Jin X, Xiong Z, Tang H, Guo H, Ye G et al (2022a) The antivirulence activity of umbelliferone and its protective effect against A. hydrophila-infected grass carp. Int J Mol Sci 23(19):11119. https://doi.org/10.3390/ijms231911119
Zhao Q, Shi J, Chen S, Hao D, Wan S, Niu H, Zhang Y (2022b) Salidroside affects gut microbiota structure in db/db mice by affecting insulin, blood glucose and body weight. Diabetes Metab Syndr Obes 15:2619–2631. https://doi.org/10.2147/DMSO.S372192
Zhong L, Peng L, Fu J, Zou L, Zhao G, Zhao J (2020) Phytochemical, antibacterial and antioxidant activity evaluation of Rhodiola crenulata. Molecules 25(16):3664. https://doi.org/10.3390/molecules25163664
Zhu H, Shen F, Wang X, Cheng Y, Guo Y, Qian H, Liu Y (2023) Reshaped gut microbial composition and functions associated with the antifatigue effect of salidroside in exercise mice. Mol Nutr Food Res 67(12):e2300015. https://doi.org/10.1002/mnfr.202300015
Zomborszki PZ, Kúsz N, Csupor D, Peschel W (2019) Rhodiosin and herbacetin in Rhodiola rosea preparations: additional markers for quality control? Pharm Biol 57(1):295–305. https://doi.org/10.1080/13880209.2019.1577460
Acknowledgements
We thank dr. Eva Lasic for reviewing a draft of this manuscript. The authors would like to thank the Slovenian Agency for Research and Innovation for funding research projects no. P4-0116, J4-4548, J4-3088 and J2-50064. The authors acknowledge the financial support by the University of Graz.
Funding
Open access funding provided by University of Graz.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Klančnik, A., Kunčič, A., Smole Možina, S. et al. The antibacterial potential and effects of Rhodiola sp. on gut microbiota. Phytochem Rev (2024). https://doi.org/10.1007/s11101-024-09965-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11101-024-09965-5