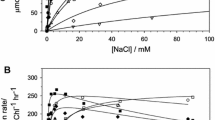
Abstract
In this study, we presented a new approach for quantification of bicarbonate (HCO3−) molecules bound to PSII. Our method, which is based on a combination of membrane-inlet mass spectrometry (MIMS) and 18O-labelling, excludes the possibility of “non-accounted” HCO3− by avoiding (1) the employment of formate for removal of HCO3− from PSII, and (2) the extremely low concentrations of HCO3−/CO2 during online MIMS measurements. By equilibration of PSII sample to ambient CO2 concentration of dissolved CO2/HCO3−, the method ensures that all physiological binding sites are saturated before analysis. With this approach, we determined that in spinach PSII membrane fragments 1.1 ± 0.1 HCO3− are bound per PSII reaction center, while none was bound to isolated PsbO protein. Our present results confirmed that PSII binds one HCO3− molecule as ligand to the non-heme iron of PSII, while unbound HCO3− optimizes the water-splitting reactions by acting as a mobile proton shuttle.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- Chl:
-
chlorophyll
- MIMS:
-
membrane-inlet mass spectrometry
- OEC:
-
oxygen-evolving complex
- PQ:
-
plastoquinone
- PQH2 :
-
plastoquinol
- RC:
-
reaction center
- NHI:
-
non-heme iron
References
Ago H., Adachi H., Umena Y. et al.: Novel features of eukaryotic photosystem II revealed by its crystal structure analysis from a red alga.–J. Biol. Chem. 291: 5676–5687, 2016.
Ananyev G., Nguyen T., Putnam-Evans C., Dismukes G.C.: Mutagenesis of CP43-arginine-357 to serine reveals new evidence for (bi)carbonate functioning in the water oxidizing complex of photosystem II.–Photochem. Photobiol. Sci. 4: 991–998, 2005.
Aoyama C., Suzuki H., Sugiura M., Noguchi T.: Flash-induced FTIR difference spectroscopy shows no evidence for the structural coupling of bicarbonate to the oxygen-evolving Mn cluster in photosystem II.–Biochemistry 47: 2760–2765, 2008.
Baranov S.V., Tyryshkin A.M., Katz D. et al.: Bicarbonate is a native cofactor for assembly of the manganese cluster of the photosynthetic water oxidizing complex. Kinetics of reconstitution of O2 evolution by photoactivation.–Biochemistry 43: 2070–2079, 2004.
Berthold D.A., Babcock G.T., Yocum C.F.: A highly resolved, oxygen-evolving photosystem II preparation from spinach thylakoid membranes.–FEBS Lett. 134: 231–234, 1981.
Blubaugh D.J., Govindjee: The molecular mechanism of the bicarbonate effect at the plastoquinone reductase site of photosynthesis.–In: {ieGovindjee (ed.): Molecular Biology of Photosynthesis. Pp. 441–484. Springer, Dordrecht 1988.
Brinkert K., De Causmaecker S., Krieger-Liszkay A. et al.: Bicarbonate-induced redox tuning in photosystem II for regulation and protection.–P. Natl. Acad. Sci. USA 113: 12144–12149, 2016.
Dasgupta J., Tyryshkin A.M., Dismukes G.C.: ESEEM spectroscopy reveals carbonate and an N-donor protein-ligand binding to Mn2+ in the photoassembly reaction of the Mn4Ca cluster in photosystem II.–Angew. Chem. Int. Ed. 46: 8028–8031, 2007.
Diamond W.D., Akinfiev N.N.: Solubility of CO2 in water from–1.5 to 100°C and from 0.1 to 100 MPa: evaluation of literature data and thermodynamic modelling.–Fluid Phase Equilibr. 208: 265–290, 2003.
Enami I., Kamino K., Shen J.-R. et al.: Isolation and characterization of photosystem II complexes which lack lightharvesting chlorophyll a/b proteins but retain three extrinsic proteins related to oxygen evolution from spinach.–BBABioenergetics 977: 33–39, 1989.
Ford R.C., Evans M.C.W.: Isolation of a photosystem 2 preparation from higher plants with highly enriched oxygen evolution activity.–FEBS Lett. 160: 159–164, 1983.
Govindjee, Weger H.G., Turpin D.H. et al.: Formate releases carbon dioxide/bicarbonate from thylakoid membranes - measurements by mass spectroscopy and infrared gas analyzer.–Naturwissenschaften 78: 168–170, 1991.
Govindjee, Xu C., van Rensen J.J.S.: On the requirement of bound bicarbonate for photosystem II activity.–Z. Naturforsch. 52: 24–32, 1997.
Guskov A., Gabdulkhakov A., Broser M. et al.: Recent progress in the crystallographic studies of photosystem II.–ChemPhysChem 11: 1160–1171, 2010.
Klimov V.V., Allakhverdiev S.I., Feyziev Y.M., Baranov S.V.: Bicarbonate requirement for the donor side of photosystem II.–FEBS Lett. 363: 251–255, 1995.
Klimov V.V., Allakhverdiev S.I., Nishiyama Y. et al.: Stabilization of the oxygen-evolving complex of photosystem II by bicarbonate and glycinebetaine in thylakoid and subthylakoid preparations.–Funct. Plant Biol. 30: 797–803, 2003.
Klimov V.V., Baranov S.V.: Bicarbonate requirement for the water-oxidizing complex of photosystem II.–BBABioenergetics 1503: 187–196, 2001.
Klimov V.V., Baranov S.V., Allakhverdiev S.I.: Bicarbonate protects the donor side of photosystem II against photoinhibition and thermoinactivation.–FEBS Lett. 418: 243–246, 1997.
Koroidov S., Shevela D., Shutova T. et al.: Mobile hydrogen carbonate acts as proton acceptor in photosynthetic water oxidation.–P. Natl. Acad. Sci. USA 111: 6299–6304, 2014.
Kozlov Y.N., Tikhonov K.G., Zastrizhnaya O.M., Klimov V.V.: pH dependence of the composition and stability of MnIIIbicarbonate complexes and its implication for redox interaction of MnII with photosystem II.–J. Photoch. Photobio. B 101: 362–366, 2010.
McConnell I.L., Eaton-Rye J.J., van Rensen J.J.S.: Regulation of photosystem II electron transport by bicarbonate.–In: Eaton-Rye J.J., Tripathy B.C., Sharkey T.D. (ed.): Photosynthesis: Plastid Biology, Energy Conversion and Carbon Assimilation. Pp. 475–500. Springer, Dordrecht 2012.
Messinger J., Badger M.R., Wydrzynski T.: Detection of one slowly exchanging substrate water molecule in the S3 state of photosystem II.–P. Natl. Acad. Sci. USA 92: 3209–3213, 1995.
Müh F., Glöckner C., Hellmich J., Zouni A.: Light-induced quinone reduction in photosystem II.–Biochim. Biophys. Acta 1817: 44–65, 2012.
Pobeguts O.V., Smolova T.N., Timoshevsky D.S., Klimov V.V.: Interaction of bicarbonate with the manganese-stabilizing protein of photosystem II.–J. Photoch. Photobio. B 100: 30–37, 2010.
Pobeguts O.V., Smolova T.N., Zastrizhnaya O.M., Klimov V.V.: Protective effect of bicarbonate against extraction of the extrinsic proteins of the water-oxidizing complex from photosystem II membrane fragments.–Biochim. Biophys. Acta 1767: 624–632, 2007.
Rappaport F., Diner B.A.: Primary photochemistry and energetics leading to the oxidation of the (Mn)4Ca cluster and to the evolution of molecular oxygen in Photosystem II.–Coord. Chem. Rev. 252: 259–272, 2008.
Shen J.-R., Katoh S.: Inactivation and calcium-dependent reactivation of oxygen evolution in photosystem II preparations treated at pH 3.0 or with high concentrations of NaCl.–Plant Cell Physiol. 32: 439–446, 1991.
Shen J.-R.: The structure of photosystem II and the mechanism of water oxidation in photosynthesis.–Annu. Rev. Plant Biol. 66: 23–48, 2015.
Shevela D., Eaton-Rye J.J., Shen J.-R., Govindjee.: Photosystem II and the unique role of bicarbonate: A historical perspective.–Biochim. Biophys. Acta 1817: 1134–1151, 2012.
Shevela D., Messinger J.: Studying the oxidation of water to molecular oxygen in photosynthetic and artificial systems by time-resolved membrane-inlet mass spectrometry.–Front. Plant Sci. 4: 473, 2013.
Shevela D., Nöring B., Koroidov S. et al.: Efficiency of photosynthetic water oxidation at ambient and depleted levels of inorganic carbon.–Photosynth. Res. 117: 401–412, 2013.
Shevela D., Su J.H., Klimov V., Messinger J.: Hydrogencarbonate is not a tightly bound constituent of the wateroxidizing complex in photosystem II.–Biochim. Biophys. Acta 1777: 532–539, 2008.
Shutova T., Kenneweg H., Buchta J. et al.: The photosystem IIassociated Cah3 in Chlamydomonas enhances the O2 evolution rate by proton removal.–EMBO J. 27: 782–791, 2008.
Stemler A., Babcock G.T., Govindjee.: Effect of bicarbonate on photosynthetic oxygen evolution in flashing light in chloroplast fragments.–P. Natl. Acad. Sci. USA 71: 4679–4683, 1974.
Stemler A., Govindjee.: Bicarbonate ion as a critical factor in photosynthetic oxygen evolution.–Plant Physiol. 52: 119–123, 1973.
Stemler A.J.: The bicarbonate effect, oxygen evolution, and the shadow of Otto Warburg.–Photosynth. Res. 73: 177–183, 2002.
Ulas G., Olack G., Brudvig G.W.: Evidence against bicarbonate bound in the O2-evolving complex of photosystem II.–Biochemistry 47: 3073–3075, 2008.
Umena Y., Kawakami K., Shen J.-R., Kamiya N.: Crystal structure of oxygen-evolving photosystem II at a resolution of 1.9 Å.–Nature 473: 55–60, 2011.
van Rensen J.J.S.: Role of bicarbonate at the acceptor side of photosystem II.–Photosynth. Res. 73: 185–192, 2002.
van Rensen J.J.S., Klimov V.V.: Bicarbonate interactions.–In: Wydrzynski T., Satoh K. (ed.): Photosystem II. The Light-Driven Water:Plastoquinone Oxidoreductase. Pp. 329–346. Springer, Dordrecht 2005.
van Rensen J.J.S., Xu C., Govindjee.: Role of bicarbonate in photosystem II, the water-plastoquinone oxido-reductase of plant photosynthesis.–Physiol. Plantarum 105: 585–592, 1999.
Villarejo A., Shutova T., Moskvin O. et al: A photosystem IIassociated carbonic anhydrase regulates the efficiency of photosynthetic oxygen evolution.–EMBO J. 21: 1930–1938, 2002.
Vinyard D.J., Ananyev G.M., Dismukes G.C.: Photosystem II: The reaction center of oxygenic photosynthesis.–Annu. Rev. Biochem. 82: 577–606, 2013.
Wei X., Su X., Cao P. et al.: Structure of spinach photosystem II–LHCII supercomplex at 3.2 Å resolution.–Nature 534: 69–74, 2016.
Wydrzynski T., Govindjee.: New site of bicarbonate effect in photosystem II of photosynthesis–Evidence from chlorophyll fluorescence transients in spinach-chloroplasts.–Biochim. Biophys. Acta 387: 403–408, 1975.
Author information
Authors and Affiliations
Corresponding author
Additional information
This paper is dedicated to the memory of Prof. Vyacheslav V. Klimov (12 January 1945 – 9 May 2017), our dear colleague and co-author, a world leader in the field of photosynthesis research for almost five decades. Among many scientific achievements, Vyacheslav Klimov (known to many as “Slava”) was one of the discoverers of pheophytin as the primary electron acceptor in PSII, and rediscoverer of “bicarbonate effect” on the electron donor side of PSII.
Acknowledgements: The authors thank Govindjee and A. Stemler for fruitful and stimulating discussions on “bicarbonate effects” over the years, the reviewers and the editor for their valuable comments and suggestions, and T.N. Smolova for isolation of PsbO protein. This work was supported by the Russian Foundation for basic research (grant No. 17-04-01011), by the Knut and Wallenberg Foundation, and by the Swedish Science Foundation (VR, grant No. 2016-05183).
Rights and permissions
About this article
Cite this article
Tikhonov, K., Shevela, D., Klimov, V.V. et al. Quantification of bound bicarbonate in photosystem II. Photosynthetica 56, 210–216 (2018). https://doi.org/10.1007/s11099-017-0758-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11099-017-0758-4