Abstract
Background Medication reconciliation prevents medication errors at care transition points. This process improves communication with general practitioners regarding the reasons for therapeutic changes, allowing those changes to be maintained after hospital discharge. Objective To investigate the impact of medication reconciliation in geriatrics on the sustainability of therapeutic optimization after hospital discharge. Setting This study was conducted in a geriatric unit in a University Hospital Centre in France. Method This was a retrospective study. For 6 months, all patients over 65 years who underwent the process of medication reconciliation performed by a clinical hospital pharmacist and a physician at admission and discharge, were included. A comparison between drug prescriptions at hospital discharge and the first prescription made outside the hospital was made to identify any differences. Main outcome measure The main outcome measures were the provision of the results of the medication reconciliation performed in the hospital to the relevant general practitioner, the subsequent acceptance of that information, the type of medication discrepancies one month after discharge and the therapeutic classes affected by the modifications. Results Among the 112 patients, medication reconciliation allowed us to identify and correct 87 unintentional discrepancies at admission (88% corrected) and 54 at discharge (92% corrected). Patients were discharged to homes or nursing homes (61%), geriatric rehabilitation units (38%) or psychiatric clinics (1%). A general practitioner wrote the first prescription renewal a mean of 36 ± 23 days after discharge, having been made aware of the medication reconciliation in only 24% of the cases (received and taken into account). The impact was a decrease in the number of patients with at least one discrepancy. Twenty-five percent of general practitioners who were aware about the medication reconciliation process accepted all therapeutic changes, while only 7% of those who were not informed did so (p = 0.02). The number of medication discrepancies observed was correlated with the number of medications for which prescriptions were renewed (p < 0.01). Conclusion Medication reconciliation involving therapeutic optimization and the justification of changes is essential to ensure the safety of the prescriptions written for patients. However, its impact after discharge is hampered by the fact that the results are often not received or taken into account by general practitioners. Taking medication reconciliation into account was associated with a significant increase in prescriptions that maintained therapeutic changes made in the hospital, confirming the positive impact of communication between care providers on therapeutic optimization.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Impact of findings on practice statements
-
Polypharmacy is associated with a higher risk of discrepancies at care transition points in patients > 65 years of age.
-
Taking medication reconciliation into account is associated with a continuation of the therapeutic changes made during hospitalization.
-
Medication reconciliation seems to make physicians aware of the reasons for the removal or modification of medicines and promotes the maintenance of such changes at discharge.
-
Communication between hospitals and community health care providers must be optimized to increase the involvement of general practitioners in sustaining changes made during hospitalization.
Introduction
The ageing of the population leads to an increase in the number of patients with multiple chronic diseases, and caring for such patients poses a major challenge to health systems [1]. The presence of multiple chronic diseases is often associated with polypharmacy, which constitutes a serious risk to health, especially for older patients [2, 3]. Other factors promote iatrogenesis in this population, including pharmacodynamic and pharmacokinetic changes, inappropriate drug use, multiple prescribers, and the presence of cognitive impairment. In France, 1.3% of admissions are the result of preventable serious adverse drug events [4]. In other countries, the situation is similar, with 6% to 12% of hospitalizations of older patients related to iatrogenesis [5]. Despite recent guidelines on improving the quality and safety of prescriptions, transitions to different levels of health care remain critical points for the patient with regard to treatment continuity. Preventable medication errors can appear because of incomplete or poorly communicated information at the community-hospital interface [6]. It has been reported that 19–75% of the discrepancies at admission were unintentional [7]. Similarly, when patients are discharged from the hospital, unintentional discrepancies impact up to 56% of patients [8]. These discrepancies are associated with a risk of adverse events, which can have serious consequences in frail older patients, leading to the aggravation of the patient’s clinical condition, the prolongation of hospitalization, readmission and increased consumption of medical resources [9]. In this context, medication reconciliation (MR), which is recommended by the French National Authority for Health [6], is an approach to reducing the incidence of medication errors (inappropriate interruption of medication, the absence of continued treatment on discharge…) at care transition points by promoting the complete and accurate transmission of information regarding a patient’s medications between health professionals [6, 10]. It has been shown that medication discrepancies with more serious clinical impacts were easily identified during pharmacy-led interventions [11]. MR at hospital admission and discharge has been shown to decrease medication errors [12, 13]. However, MR also highlights intentional discrepancies, which are voluntary therapeutic changes made to optimize treatment [14]. These intentional therapeutic modifications are not always continued after discharge [15]. Viktil et al. showed that 4 months after hospitalization, 90/105 patients had a different prescription [16]. Indeed, in the absence of explanations of these changes, patients and general practitioners (GPs) can be confused about the reasons for starting or stopping a treatment and can interpret these changes as errors. This is a problem in many health systems. There is a lack of communication between health care professionals in the community and at the hospital, with GPs receiving little information about therapeutic modifications made [17, 18]. To ensure the maintenance of therapeutic changes after hospital discharge, more complete and accurate information needs to be provided to GPs about the reasons therapeutic changes were made.
Aim of the study
This study was performed to evaluate the post-hospitalization prescriptions of patients > 65 years of age who underwent the process of MR. The main outcomes were the transfer of the information from the MR process to care providers outside of the hospital and the maintenance of therapeutic changes made during hospitalization, as reflected in the first prescription written after discharge. We also investigated the types of medication discrepancies and the therapeutic classes affected by the modifications.
Ethics approval
This study was conducted in accordance with good clinical practices and the principles of the Declaration of Helsinki. The study was authorized by The French Health Agency (number 79U69Y) and approved by the National Commission for Information Technology and Civil Liberties (French CNIL). In accordance with French legislation, formal approval from an ethics committee was not required for this type of retrospective non-interventional study based on the reuse of already recorded data.
Methods
Type of study
This single-centre, retrospective, descriptive study was conducted in the geriatric unit (21 beds with patients from the emergency unit) of Timone Hospital, Marseille, France. For 6 months, all patients older than 65 years who underwent the process of MR at admission and at discharge were included.
Medication reconciliation
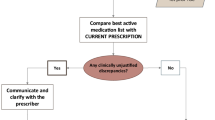
MR was performed by clinical hospital pharmacists (3 pharmacists) specially trained in geriatrics and the information was discussed with the senior physician of the unit. Inspired by the documents proposed by the HAS, we created a standardized form to group and analyse drug data. The initial MR was conducted retroactively during the first 72 h after admission, and at least 3 sources of information (including, if possible, patient communication) were considered. For each unintentional discrepancy, the physician who prescribed the medication decided on the necessary correction. The final MR was performed the day before discharge and involved a comparison of the initial medication list, the prescriptions written during hospitalization and the prescriptions included in the discharge information. Discrepancies were reported in a table, and all changes were justified. On the day of discharge, the MR information and care plan were given to the patient and transferred to the appropriate post-discharge location (such as the inpatient geriatric rehabilitation unit (GRU) or a nursing home), the patient’s community pharmacy (or the hospital pharmacy for those in the GRU) and to the patient’s physician(s), along with the hospitalization report.
Data collection
For each patient, age, sex, number of comorbidities, number of drugs at hospital admission (prescribed medications and over-the-counter medications) and discharge, community pharmacy, GP, nurse and discharge destination (home, nursing home, GRU) were collected. Subsequently, after discharge, the first prescription filled by the community pharmacy (other than discharge prescriptions written while the patient was still hospitalized), namely, the “post-hospitalization prescription” (PHP), was recorded. To obtain information about the PHP, the community pharmacy was contacted first, followed by the GP or the nursing home and, as a last resort, the patient or the patient’s family. The PHP was not necessarily written by the patient's GP, particularly in the case of patients in the GRU or nursing homes (local physicians).
The PHP and the prescriptions at discharge were compared to detect discrepancies with consideration of the following factors:
-
The awareness on the part of the GP of the MR
-
Patient outcomes, which were collected over the phone from the patient, the patient’s family or the patient’s GP. If the patient had been hospitalized in the last 30 days, the reason for hospitalization was also recorded.
Statistical analysis
The quantitative variables are expressed as averages with standard deviations, and the qualitative variables are expressed as numbers with percentages. The qualitative parameters were compared using the chi-square test or Fisher’s exact test, as appropriate. The Mann–Whitney U test and Student’s t-test, as appropriate, were applied for quantitative data. Finally, potential correlations between quantitative variables was established by Pearson’s correlation. Reported p values were two-sided, and p < 0.05 was considered statistically significant.
Results
Population description
Among the 112 included patients, 68 patients (61%) returned to their homes or nursing homes after discharge, 43 (38%) went to the GRU, and 1 was discharged to a psychiatric clinic. The baseline characteristics of the study population are summarized in Table 1. The mean age of the patients was 84.5 ± 7.6 years (median 86 years).
Five patients died soon after hospital discharge, without any evident iatrogenic causes (2 were hospitalized for palliative care). Eight patients (including 5 patients in the GRU) had been rehospitalized within 30 days: 2 were rehospitalized due to acute pulmonary oedema (for 1 patient, treatment with a diuretic was stopped during the initial hospitalization), 2 due to sepsis, 1 due to a fall (receiving lormetazepam at discharge), 1 due to confusion that turned out to be a seizure crisis that was a stroke sequela, 1 due to chest pain (patient with coronary heart disease whose cardiovascular treatment was not modified), and 1 for an unknown reason.
Medication reconciliation at admission and discharge
At admission, the patients had 4.1 ± 1.9 comorbidities and took 7.7 ± 3.5 drugs. At discharge, the patients took significantly fewer medications (6.3 ± 2.8, p < 0.01). We found 87 unintentional discrepancies and 832 intentional discrepancies at admission (0.77 and 7.4 per patient, respectively). At hospital discharge, we noted 54 unintentional and 336 intentional discrepancies (0.48 and 3 per patient, respectively). In 88% and 92% of the patients at admission and discharge, respectively, the unintentional discrepancies were corrected.
Continuity of treatment after discharge
After discharge (mean 36 ± 23 days), we retrieved 99 PHPs (88% of the 112 patients) (Table 2). Of the 99 prescriptions retrieved, 33 were obtained from the GRU (which had received the MR information), and 66 had been written by the patients’ GPs, including those for 7 patients in nursing homes. Of the 66 GPs, only 20 confirmed that they had received the hospitalization report and discharge MR information. Eighteen out of 20 GPs and 6 out of 33 GRU physicians reported that they considered the MR information when writing the new prescription and found it useful. Thus, only 24 prescriptions out of the 99 (24%) were written by physicians who were aware of the therapeutic changes made during hospitalization. In these cases, a decrease in the number of patients with at least one discrepancy was observed. In total, 25% of GPs who were aware about the MR information fully accepted the therapeutic changes, while only 7% who had not received the MR information did so (p = 0.02). The analysis of the PHPs showed an average of 6.4 ± 3.1 drugs, with no differences from the prescriptions at discharge (6.3 ± 2.7; p = 0.84). The most heavily modified list of prescriptions, which had 11 discrepancies, came from a physician who had no knowledge of the discharge MR information. Three prescriptions issued by GPs, including one who had read the discharge MR information, were for the same medications that the patients had taken before hospitalization. No significant discrepancies were found between the patients’ outcomes and the awareness of the discharge MR information on the part of the physician (p = 0.14).
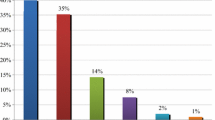
Among the 88 PHPs with at least one discrepancy, we observed 321 treatment modifications: 138 (43%) were added drugs, 130 (40%) were removed drugs, 34 (11%) were dose modifications, 17 (5%) were changes in the Anatomical Therapeutic Chemical classification, and only 2 (1%) were dosage adjustments (Fig. 1).
No link was found between the number of comorbidities and the number of discrepancies observed (correlation coefficient 0.07, p = 0.50). However, the more medicines taken by the patient before admission and at discharge, the more discrepancies there were in the PHPs (correlation coefficients were 0.27 (p = 0.01) and 0.45 (p < 0.01), respectively). This suggests that polypharmacy is associated with a higher risk of discrepancies. The number of medicines on the PHP was 5.5 ± 2.5 when the physician considered the MR information and 6.7 ± 3.2 when the MR information was not received or considered (p = 0.06). Consideration of the MR information was associated with a higher rate of unmodified prescriptions and a significant decrease in the number of patients with at least one discrepancy in the PHPs (p = 0.02). The average number of discrepancies was 2.6 ± 2.2 when the physician considered the MR information and 3.5 ± 2.6 when he/she did not consider the information (p = 0.12). Twelve therapeutic subclasses accounted for 75% of the discrepancies (Table 3).
The 3 therapeutic classes most commonly missing from the PHPs were vitamin supplements, analgesics (with paracetamol) and dietary supplements, and the 3 most commonly added to the PHPs were vitamin supplements, benzodiazepines and laxatives (Fig. 2). Before hospitalization, these treatments were prescribed to 16.7%, 42.9% and 30.8% of the patients, respectively.
Benzodiazepines (stopped during hospitalization in 5 out of the 14 patients) were reintroduced by physicians who did not consider the MR information. We also noted the addition of 18 vitamin supplements. In 3 cases, these were reintroduced by physicians who did not consider the MR information. With regard to the addition of 7 drugs for acid-related disorders, 3 were reintroduced, 2 of which were reintroduced by physicians who did not consider the MR information. The 34 discrepancies linked to dose modifications were equally distributed between dose increases and decreases. The three drug classes with the most changes in doses were antihypertensive drugs (42.9% increase), benzodiazepines (66.7% increase) and drugs for acid-related disorders (33.3% increase).
Discussion
This study evaluated the impact of MR on the maintenance of therapeutic changes after hospital discharge.
In the literature, older age and polypharmacy are the main predictors of unintentional discrepancies at transition points [19]. Krause et al. showed that in the absence of adequate communication and justification of the therapeutic changes made during hospitalization, GPs often adjust discharge medications prescribed to geriatric patients (79%); such adjustments are generally unnecessary due to changes in the health of the patients [20]. Better communication can limit unplanned consultations or readmissions with iatrogenic causes. In this context, MR promotes the complete and accurate communication of information about a patient’s medications between health professionals.
At admission, we found, on average, 0.7 unintentional discrepancies per patient between the initial medication list and the admission prescriptions recorded by the clinical pharmacist. The physicians agreed to and implemented 88% of the changes recommended by the clinical pharmacists. These results are consistent with the findings in the literature [21].
At discharge, we found 0.5 discrepancies per patient between the discharge prescriptions and the initial medication list. The physicians implemented 92% of the changes. After hospital discharge, 11% of prescriptions remained unchanged, which is a lower proportion than that reported by Houlind et al. [21]. In their study, 83% of the changes made to prescriptions remained unchanged 30 days after discharge. However, they had only collected data at one follow-up point that was 30 days after discharge, regardless of whether patients had seen their GPs.
The main strength of our study is that we gathered information from the first prescription filled by the community pharmacy that had been written after the patient had been discharged from the hospital (36 ± 23 days after discharge).
In our study, only 24% of the physicians reported receiving and considering the MR information, while 73.7% reported doing so in the study by Moulis et al. [22]. In addition, we showed that 25% of the GPs who were aware of the MR information fully accepted the changes made to prescriptions during hospitalization, while only 7% of GPs who were not aware of that information maintained all the therapeutic changes. The proportion of patients with discrepancies in our study was comparable to that reported by Viktil et al. (89% of the patients had at least one discrepancy on the PHP versus 86% [16]). Moulis et al. observed that in more than half of the 30 patients older than 65 years, there was a return to the initial prescription [22]. In our study, only 3 PHPs were identical to the pre-hospitalization prescriptions, and 11% of the PHPs showed no discrepancies from the prescriptions at discharge, which is a comparable proportion to that reported in the study by Mansur et al. (16%) [23]. As previously reported [24], we found that polypharmacy, which is defined as the simultaneous use of more than 5 drugs, was associated with an increased risk of discrepancies after discharge from the hospital.
The consideration of the MR information was associated with a significant increase in the number of prescriptions that maintained the therapeutic changes made during hospitalization, confirming the positive impact of communication between care providers on the continuity of treatment, as shown by Moulis et al. [22]. Among the changes observed after discharge, the removal of medications accounted for 40% of the discrepancies; these removed medications were mainly vitamin supplements, paracetamol and dietary supplements. Healing and improvements in the acute pathology for which the patient was hospitalized can lead to the removal of paracetamol that was prescribed for analgesic or antipyretic purposes and the prescription of dietary supplements to avoid weight loss related to hypercatabolism. Vitamin supplements are often prescribed for a limited duration. The initiation of medications accounted for 43% of the discrepancies; and most newly prescribed medications were vitamins, benzodiazepines and laxatives. Vitamin supplements and laxatives were also among the 3 most common types of medications that were newly prescribed after hospital discharge in the study by Moulis et al. [22]. In approximately one-third of the cases, benzodiazepines prescribed by a health care professional in the community were reintroduced by physicians who had not received the MR information. The reintroduction of these drugs, which were gradually ceased, cannot be explained by withdrawal syndrome. Drugs prescribed for acid-related disorders, which have limited validated indications and are overprescribed due to their good initial tolerance, were also reintroduced in a quarter of the cases by physicians who did not consider the MR information [25]. Despite the limitations of our study, MR seems essential to informing GPs of the reasons treatments were modified during hospitalization and promoting the maintenance of those changes after discharge.
The goal of MR is to reduce medication errors and avoid iatrogenic and unplanned readmissions [26]. In our study, we observed that 7% of the patients were readmitted within 30 days, and less than 2% were admitted for a reason that could be related to a change in treatment made during hospitalization. Khoehler et al. showed that the performance of MR, education of patients and their families about the changes, collaborative sharing of the information from the MR with the community GPs and follow-up of geriatric patients significantly decreased the rate of unplanned readmission in frail older patients [27]. Only the implementation of several coordinated interventions significantly reduced the readmission rate [28].
In our study, we found that only 30% of the GPs reported receiving the information. This result is consistent with that reported in the study by Kripalani et al. [28], who found that hospitalization reports were received by GPs in 12 to 34% of the cases. This finding raises questions about how this essential communication can be improved. The development of secure professional messaging systems could improve communication between health professionals, as could the shared use of personal medical files and shared pharmaceutical files. One of the limitations of this work is the limited number of physicians who responded that they had received and considered the discharge MR information. We have not studied the relevance of justification for the therapeutic changes observed on the PHPs.
In the future, we could investigate the potential clinical severity of the consequences of the observed discrepancies to further classify the medication errors that MR could prevent and the effect of MR on patient safety.
Conclusion
Medication reconciliation should be taken into account by community GPs to optimize the management of the drug treatment of older patients after hospital discharge. Within our sample of patients, the consideration of the MR by GP was associated with a significant increase in the number of prescriptions that maintained the therapeutic changes made during hospitalization. This result confirm the positive impact of communication between care providers on the continuity of treatment. However, due to the lack of the awareness of MR information on the part of community GPs in our study, it is essential to continue with this study for longer in order to obtain a sufficiently representative sample of patients in order to confirm these preliminary results.
References
Ministère des Affaires sociales et de la Santé. L’état de santé de la population en France. 2017 [cited 2020 Aug 04]. French. Available from: https://drees.solidarites-sante.gouv.fr/.
Lazarou J, Pomeranz BH, Corey PN. Incidence of adverse drug reactions in hospitalized patients: a meta-analysis of prospective studies. JAMA. 1998;279(15):1200–5.
Alhawassi TM, Krass I, Bajorek BV, Pont LG. A systematic review of the prevalence and risk factors for adverse drug reactions in the elderly in the acute care setting. Clin Interv Aging. 2014;9:2079–86.
Direction de la recherche, des études, de l’évaluation et des statistiques. Enquête nationale sur l’ensemble des événements indésirables graves associés aux soins. 2009 [cited 2020 Aug 04]. French. Available from: https://drees.solidarites-sante.gouv.fr/.
Parameswaran Nair N, Chalmers L, Peterson GM, Bereznicki BJ, Castelino RL, Bereznicki LR. Hospitalization in older patients due to adverse drug reactions -the need for a prediction tool. Clin Interv Aging. 2016;11:497–505.
Haute Autorité de Santé. Mettre en œuvre la conciliation des traitements médicamenteux en établissement de santé. 2008 [cited 2020 Aug 04]. French. Available from: https://www.has-sante.fr/.
Tam VC, Knowles SR, Cornish PL, Fine N, Marchesano R, Etchells EE. Frequency, type and clinical importance of medication history errors at admission to hospital: a systematic review. CMAJ. 2005;173(5):510–5.
Moro Agud M, Menéndez Colino R, Mauleón Ladrero Mdel C, Ruano Encinar M, Díez Sebastián J, Villamañán Bueno E et al. Analysis of an electronic medication reconciliation and information at discharge programme for frail elderly patients. Int J Clin Pharm. 2016;38(4):996–1001.
Bates DW, Spell N, Cullen DJ, Burdick E, Laird N, Petersen LA, et al. The costs of adverse drug events in hospitalized patients. JAMA. 1997;277(4):307–11.
Haute Autorité de Santé. Conciliation des traitements médicamenteux—Prévenir les erreurs. 2018 [cited 2020 Aug 04]. French. Available from: https://www.has-sante.fr/.
Mekonnen AB, McLachlan AJ, Brien J-AE. Pharmacy-led medication reconciliation programmes at hospital transitions: a systematic review and meta-analysis. J Clin Pharm Ther. 2016;41(2):128–44.
Andreoli L, Alexandra J-F, Tesmoingt C, Eerdekens C, Macrez A, Papo T, et al. Medication reconciliation: a prospective study in an internal medicine unit. Drugs Aging. 2014;31(5):387–93.
Monfort AS, Curatolo N, Begue T, Rieutord A, Roy S. Medication at discharge in an orthopaedic surgical ward: quality of information transmission and implementation of a medication reconciliation form. Int J Clin Pharm. 2016;38(4):838–47.
Pérennes M, Carde A, Nicolas X, Dolz M, Bihannic R, Grimont P, et al. Medication reconciliation: an innovative experience in an internal medicine unit to decrease errors due to inacurrate medication histories. La Presse Médicale. 2012;41(3):e77–86. https://doi.org/10.1016/j.lpm.2011.09.016.French.
Cochrane RA, Mandal AR, Ledger-Scott M, Walker R. Changes in drug treatment after discharge from hospital in geriatric patients. BMJ. 1992;305(6855):694–6.
Viktil KK, Blix HS, Eek AK, Davies MN, Moger TA, Reikvam A. How are drug regimen changes during hospitalisation handled after discharge: a cohort study. BMJ Open. 2012;2:e001461. https://doi.org/10.1136/bmjopen-2012-001461.
Pichat C. Évolution de l’ordonnance des patients a un mois de la sortie d’un service de médecine polyvalente: étude prospective sur trois mois [thesis]. Université de Montpellier I; 2013. French.
Kripalani S, LeFevre F, Phillips CO, Williams MV, Basaviah P, Baker DW. Deficits in communication and information transfer between hospital-based and primary care physicians: implications for patient safety and continuity of care. JAMA. 2007;297(8):831–41.
Hias J, Van der Linden L, Spriet I, Vanbrabant P, Willems L, Tournoy J, et al. Predictors for unintentional medication reconciliation divergences in preadmission medication: a systematic review. Eur J Clin Pharmacol. 2017;73(11):1355–77.
Krause O, Glaubitz S, Hager K, Schleef T, Wiese B, Junius-Walker U. Post-discharge adjustment of medication in geriatric patients: a prospective cohort study. Z Gerontol Geriatr. 2019. https://doi.org/10.1007/s00391-019-01601-8. PubMed PMID: 31440831.
Houlind MB, Andersen AL, Treldal C, Jørgensen LM, Kannegaard PN, Castillo LS, et al. A collaborative medication review including deprescribing for older patients in an emergency department: a longitudinal feasibility study. J Clin Med. 2020;9(2):348.
Moulis M, Gibert P, Payen M, Grevy A, Gavazzi G, Calop J. La conciliation médicamenteuse en gériatrie: un moyen de pérenniser l’optimisation thérapeutique après la sortie d’hospitalisation? Le Pharmacien Hospitalier et Clinicien. 2014;49(2):e126. https://doi.org/10.1016/j.phclin.2014.04.261. (French).
Mansur N, Weiss A, Hoffman A, Gruenewald T, Beloosesky Y. Continuity and adherence to long-term drug treatment by geriatric patients after hospital discharge: a prospective cohort study. Drugs Aging. 2008;25(10):861–70.
Coleman EA, Smith JD, Raha D, Min SJ. Posthospital medication discrepancies: prevalence and contributing factors. Arch Intern Med. 2005;165(16):1842–7.
Haute Autorité de santé. Les inhibiteurs de la pompe à protons chez l’adulte. 2009 [cited 2020 Aug 04]. French. Available from: https://www.has-sante.fr/.
Boockvar KS, Carlson LaCorte H, Giambanco V, Fridman B, Siu A. Medication reconciliation for reducing drug-divergence adverse events. Am J Geriatr Pharmacother. 2006;4(3):236–43.
Koehler BE, Richter KM, Youngblood L, Cohen BA, Prengler ID, Cheng D, et al. Reduction of 30-day postdischarge hospital readmission or emergency department (ED) visit rates in high-risk elderly medical patients through delivery of a targeted care bundle. J Hosp Med. 2009;4(4):211–8.
Kripalani S, Theobald CN, Anctil B, Vasilevskis EE. Reducing hospital readmission: current strategies and future directions. Annu Rev Med. 2014;65:471–85.
Acknowledgements
The authors would like to thank Dr Clémence Tabele, Dr Alexandre Bacci, Dr Marjorie Legrand, Dr Nirvina Gobin and Dr Magali Pellerey for their assistance with the routine medication reconciliation process.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Montaleytang, M., Correard, F., Spiteri, C. et al. Medication reconciliation in the geriatric unit: impact on the maintenance of post-hospitalization prescriptions. Int J Clin Pharm 43, 1183–1190 (2021). https://doi.org/10.1007/s11096-021-01229-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11096-021-01229-y