Abstract
Importance
High-grade gliomas (HGG) are the most aggressive and common malignant brain tumors in adults. They have a dismally fatal prognosis. Even if gross total resection of the enhancing tumor is achieved, inevitably, invading tumor cells that are indistinguishable to the un-aided eye are left behind, which eventually leads to tumor recurrence. 5-aminolevulinic acid (5-ALA) is an increasingly utilized intraoperative fluorescent imaging agent for patients with HGG. It enhances visualization of HGG tissue. Despite early promising randomized clinical trial data suggesting a survival benefit for 5-ALA-guided surgery, the growing body of literature must be analyzed to confirm efficacy on patient outcomes.
Objective
To perform a systematic review of the literature to evaluate whether there is a beneficial effect upon survival and extent of resection due to the utilization of 5-ALA in HGG surgery.
Evidence review
Literature regarding 5-ALA usage in HGG surgery was reviewed according to the PRISMA guidelines. Two databases, PubMed and SCOPUS, were searched for assorted combinations of the keywords “5-ALA,” “high-grade glioma,” “5-aminolevulinic acid,” and “resection” in July 2020 for case reports and retrospective, prospective, and randomized clinical trials assessing and analyzing 5-ALA intraoperative use in patients with HGG. Entailed studies on PubMed and SCOPUS were found for screening using a snowball search technique upon the initially searched papers. Systematic reviews and meta-analyses were excluded from our PRISMA table.
Findings
3756 previously published studies were screened, 536 of which were further evaluated, and ultimately 45 were included in our systematic review. There were no date restrictions on the screened publications. Our literature search was finalized on July 16, 2020. We found an observed increase in the overall survival (OS) and progression-free survival (PFS) of the 5-ALA group compared to the white light group, as well as an observed increase in the OS and PFS of complete resections compared to incomplete resections. Of the studies that directly compared the use of 5-ALA to white light (13 of the total analyzed 45, or 28.9%), 5-ALA lead to a better PFS and OS in 88.4 and 67.5% of patients, respectively.
When the studies that reported postoperative neurologic outcomes of surgeries using 5-ALA vs. white light were analyzed, 42.2% of subjects demonstrated 5-ALA use was associated with less post-op neurological deficits, whereas 34.5% demonstrated no difference between 5-ALA and without. 23.3% of studies showed that intraoperative 5-ALA guided surgeries lead to more post-op neurological deficits.
Conclusions and relevance
Utilization of 5-ALA was found to be associated with a greater extent of resection in HGG surgeries, as well as longer OS and PFS. Postop neurologic deficit rates were mixed and inconclusive when comparing 5-ALA groups to white light groups. 5-ALA is a useful surgical adjunct for resection of HGG when patient safety is preserved.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
High grade gliomas (HGG) are tumors of the glial cells in the central nervous system. These gliomas are referred to as high grade because they are rapidly growing and categorized as WHO grade III or IV tumors. HGG are the most aggressive and common types of brain tumors in adults. They include gliosarcomas, anaplastic astrocytomas, oligoastrocytomas, diffuse brainstem gliomas/diffuse pontine gliomas, pleiomorphic xanthoastrocytomas, and glioblastoma multiforme (GBM). Despite surgical resection, radiation therapy, and chemotherapy, microscopic residual tumor is inevitable due to the highly invasive nature of HGG, which leads to tumor recurrence.
One emerging tool in the neurosurgical arsenal is using intraoperative 5-aminolevulinic acid (5-ALA) for fluorescence-guided surgery to maximize tumor resection and minimize residual tumor. In 2017, the U.S. Food and Drug Administration (FDA) approved 5-ALA for use as an intraoperative fluorescent imaging agent in patients with HGG [1]. 5-ALA enhances visualization of malignant brain tumor tissue to potentially result in a more complete resection of the tumor. Approximately a decade prior to its approval in the United States, it was first approved in Europe by the European Medicine’s Agency (EMA) in 2007 [1]. The European approval followed Dr. Walter Stummer’s 2006 randomized controlled multicenter phase III trial which substantiated that 5-ALA can lead to more complete HGG resection with better outcomes and greater 6 month progression free survival than white light visualization alone [2]. Stummer’s work was a landmark paper describing a survival benefit from the use of 5-ALA. High-grade glioma patients fare better in terms of survival with gross-total resection [3]. The greater the extent of resection, the longer the overall survival of the patient [2]. Intraoperative 5-ALA can potentially help maximize tumor resection.
Currently, this solution is inadequately investigated, as it has been 14 years since Stummer’s paper was written, and there has not been much generalized conclusion regarding the survival benefit from the use of 5-ALA in GBM resection. However, during the past 14 years, a copious amount of papers have been written about individual 5-ALA experiences. It is imperative to analyze these papers to see if they confirm the benefit observed in the Stummer 2006 study. Additionally, it appears that although 5-ALA is associated with a greater extent of resection, its use may be associated with a potential increase in post-op complication/neurological deficit. This postulation must be further explored as well.
Methods
Search strategy
Two databases, PubMed and SCOPUS, were searched in July 2020 for case reports, retrospective, prospective, and randomized clinical trials assessing and analyzing 5-ALA intraoperative use in patients with HGG. Entailed studies on PubMed and SCOPUS were found for screening using a snowball search technique upon the initially searched papers. Systematic reviews and meta-analyses were excluded from our PRISMA table. Assorted combinations of the keywords “5-ALA,” “high-grade glioma,” “5-aminolevulinic acid,” and “resection” were used in the search. Our systematic review was reported in accordance to the preferred reporting items for systematic reviews and meta-analyses (PRISMA) guidelines. [4] We performed searches in PubMed and SCOPUS from inception to July 16, 2020. Case reports, retrospective, prospective, and randomized clinical trials evaluating the application, characteristics, and effects of 5-ALA use in HGG resection were included. There was no limit by year of publication. Editorials were excluded. The selection of sources was agreed upon by consensus. The first author, year, study design, country, number of participants, figures, content, diagnosis, pathology, prognosis, and timeline were all included data taken into consideration.
Study selection
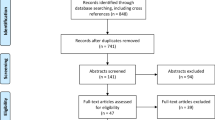
The included studies had to definitively use 5-ALA as an intraoperative imaging agent upon HGG neurosurgery. 3756 full-text studies were assessed for eligibility and inclusion in this systematic review (Fig. 1). 2029 studies were initially excluded due to complete irrelevance and duplication. Upon screening the remaining 1727 abstracts, a further 1191 studies were excluded for substantial irrelevance. Of the 536 remaining full texts that were read, 491 were excluded for the following reasons: limited relevance, low-grade gliomas (WHO grade I and II), focus upon 5-ALA sensitivity, specificity, PPV, and NPV, and failure to include statistics/results regarding survival, extent of resection, and percent gross-total resection. Thus, 45 studies were included (Table 1).
Results
Study characteristics
The reviewed studies included 21 prospective, 20 retrospective, 3 randomized phase trials, and 1 parallel, randomized, balanced, group-sequential, two-armed, controlled multicenter phase III study. A total of 3756 titles and abstracts were identified by the aforementioned search tactics, of which 45 met our inclusion criteria and were included in the qualitative synthesis. The total number of patients across all of the 45 included studies were at least 4599 (Table 1). One study by Stepp et al. [5] was carried out on patients at 18 clinics in Germany, however, the specific amount of patients was not specified in the text. The 45 studies all used 5-ALA to remove HGG tumors and often focused on 5-ALA vs. white light, 5-ALA only, and 5-ALA combined with technologies, such as intraoperative MRI (iMRI), contrast-enhanced ultrasound (CEUS), intraoperative ultrasound (ioUS), and intraoperative CT (i-CT).
Of the 45 analyzed studies, 13 of these studies (28.9%) compared HGG resection guided by 5-ALA vs. white light. These 13 studies totaled 1984 patients. Of the 1984 patients, a total of 1525 patients were analyzed for progression-free survival (PFS), 1616 analyzed for overall survival (OS), and 1077 explored post-op neurological deficit. Regarding PFS, 5-ALA was better in 1348 patients (88.4%), and there was no difference shown between 5-ALA and lack of 5-ALA in 177 patients (11.6%). As for OS, 5-ALA was better in 1090 (67.5%) and there was no difference between 5-ALA and lack thereof in 526 (32.5%). Regarding post-op neurological deficits, 5-ALA was better that white light in 454 patients (42.2%), worse in 251 (23.3%), and showed no difference between 5-ALA and white light in 372 patients (34.5%).
Findings
Fluorescence-guided resection (FGR) with 5-ALA for the removal of HGG tumors was associated with a higher extent of resection (EOR) [6,7,8] and a higher percentage of gross total resections (GTR) [2, 5, 6, 9] than white light. This corresponds to an ultimately longer PFS [2, 5,6,7, 9, 10] and OS [2, 6,7,8,9, 11]. These findings coincide with the findings of Eljamel et al. and Eyüpoglu et al. that showed 5-ALA usage as compared to its absence correlates with a higher OS [12, 13]. Complete resection as opposed to incomplete resection correlates with a longer PFS [14,15,16] as well as OS [14,15,16,17,18,19]. Panciani et al.’s study corroborates the conclusion that 5-ALA and neuronavigation leads to an increase in the obtained EOR than with conventional surgical strategy [20].
The rates of GTR when using FGR technique with 5-ALA greatly vary across studies. Multiple groups report rates of complete resection of 80% and greater when using 5-ALA [21], which can be considered a decent amount [14, 22,23,24,25,26,27,28]. Conversely, very low complete resection rates (< 50%) have been reported with the use of 5-ALA [29, 30]. Mediocre rates have also been reported, between 50 and 80% [16, 19, 31,32,33,34,35,36,37,38]. These varying rates of resection and the cause of such disparity should be further inspected.
The combination of 5-ALA with different intraoperative technological modalities yielded mixed results. Barbagallo et al. [39] did not find a statistically significant difference between the PFS and OS of intraoperative 5-ALA usage only versus 5-ALA + i-CT. A study by Coburger et al. [40] demonstrated that the combination of iMRI + 5-ALA versus iMRI without 5-ALA led to a greater amount of GTR and a greater OS. Similar studies by Eyüpoglu et al. [41] and Nickel et al. [42] both demonstrate that iMRI + 5-ALA led to a higher EOR as opposed to 5-ALA alone. Conversely, Tsugu et al. [43] concluded that 5-ALA alone resulted in a higher percentage of complete resection, but a lower EOR than iMRI + 5-ALA. Roder et al. [44] showed that surgery solely with iMRI led to a higher percentage of GTR, greater EOR, and a higher PFS than surgery solely with 5-ALA. Surgery guided by iMRI versus non-iMRI in a study by Schatlo et al. [45] indicated that iMRI usage led to a higher overall survival. Yamada et al. [46] reported an impressive amount of 95% complete resections executed by 5-ALA + iMRI. Additionally, Della Pepa et al. [47] studied the combined use of intraoperative 5-ALA + CEUS and found that the combination resulted in the highest EOR as compared with 5-ALA alone, CEUS alone, and conventional surgical technique. Neidert et al. [48] found that 5-ALA used in conjunction with ioUS resulted in a higher OS than 5-ALA alone.
Overall, OS and PFS were longer in the 5-ALA groups when compared to white light groups in studies individually, as well as complete resection groups to incomplete resection groups. The most common post-operative neurological deficits were motor, language, and visual in nature. They consisted more specifically of aphasia, hemiparesis, and hemianopsia. Post-op seizures and hemorrhages were also observed.
There could be an assumption that higher neurological complication rate of HGG resection using 5-ALA as compared to white light would occur due to its more aggressive surgical resection. Yet, the included studies were rather inconclusive and disparate regarding if 5-ALA led to more complications. Across the studies that included complication rates, 5-ALA was better that white light in 454 patients (42.2%), worse in 251 (23.3%), and showed no statistical difference between 5-ALA and white light in 372 patients (34.5%). In multiple studies, there were surgeries that were halted, leaving behind residual 5-ALA-fluorescing tissue in order to avoid causing neurological deficit. For example, in the study by Chan et al., minimal residual 5-ALA fluorescence was left in three patients to avoid post-operative deficit [31]. Della Puppa et al. stopped surgery in 26% of cases to avoid neurological deficits [22]; Feigl et al. stopped 24% of surgeries, leaving residual 5-ALA fluorescence to avoid deficits [35]. Jacquesson et al. noted leaving 5-ALA fluorescence to avoid post-operative neurological deficits as well in 31.8% of their cases [16].
0% of cases reported an allergy to 5-ALA reported. If a patient is known to have an allergy to 5-ALA or is an extremely atopic individual, proper prophylaxis is necessary to avoid allergenic complications. Or rather, as seen in studies such as Chan et al., 5-ALA was contraindicated in patients with an allergy to it [31]. Allergies to 5-ALA are rare, and anaphylaxis is incredibly rare. Supposedly, the first, and seemingly only, case of severe allergic reaction (anaphylaxis) to intravesical instillation of hexaminolevulinate hydrochloride in literature was reported by Colapaoli et al. [49]. However, 5-ALA in this case was not used for HGG resection, but rather for fluorescence cystoscopy. Generally, neurosurgeons utilizing 5-ALA to guide HGG resections should be very conscientious of fluorescing functional, healthy tumor. It is important to note the need to respect the boundaries of the actual HGG tissue and proceed with proper caution.
Discussion
Our analysis has demonstrated that 5-ALA is beneficial compared to white light alone. Additionally, our review has shown that gross total resection compared to incomplete resection favors longer PFS and OS across individual studies. These results are consistent with published findings in the literature showing that gross total, but not incomplete, resection of GBM prolongs survival in conjunction with radiochemotherapy [50]. Overall, OS and PFS of the 5-ALA group tended to be longer compared to the white light group, as well as longer after complete resection compared to incomplete resection. Of the studies that directly compared the use of 5-ALA to the lack thereof (13 of the total analyzed 45, or 28.9%), 5-ALA led to a better PFS and OS in 88.4% and 67.5% of patients, respectively (Table 2).
The results from analyzing the included literature support the utilization of intraoperative 5-ALA FGR in the removal of HGG tumor. 5-ALA use beneficially corresponds with a longer PFS and OS, which is what was found when pooling direct cohort studies only. This concept is consistent with the large body of literature demonstrating that increasing extent of resection correlates with longer overall survival [3, 51]. Illuminating cancerous tissue and its borders so that the surgeon can visualize what he/she must remove would sensibly lead to a more effective resection.
Combining intraoperative technological modalities with intraoperative 5-ALA utilization correlates with higher survival than does 5-ALA, iMRI, i-CT, ioUS, or CEUS each alone. This finding substantiates the need to further investigate the combination of fluorescent and technical modalities to potentially reach maximal survival rates and EOR. Such a combination may be the most optimized modality for HGG removal known to date.
There are some potential drawbacks to the use of intraoperative 5-ALA that should be considered. It appears that 5-ALA can help achieve a more complete resection, but with this comes a slight increase in post-op complication/neurological deficit. The reasoning behind this may be that the greater the amount resected, the higher the risk of excising healthy, functional tissue. There exists research positing that 5-ALA may enable extension of tumor resection beyond radiologically evident tumor. This extension may potentially put intact, functional adjacent tissue at risk of destruction. Therefore, 5-ALA may lead surgeons to resect too far beyond the HGG tumor’s borders and damage healthy, eloquent brain matter [52]. This risk may explain the discrepancy in numbers of post-op neurological deficit reported. It is important to note that due to the increased vigilance and more conservative resection executed in these studies, we believe that the neurological complication rate due to the use of 5-ALA in HGG resection was far lower than it would have been if the neurosurgeons had not preventively acted with the responsible foresight with which they did. Multiple studies documented the halting of surgeries as to not aggressively resect tissue beyond the borders of the HGG.
The mixed findings regarding the effect of 5-ALA use on post-operative neurological deficit complicate interpretation of these studies. It is possible that differences in tumor location, surgeon skill, and patient medical comorbidities may confound neurologic deficit data. Thus, no clear conclusion regarding the risk of 5-ALA florescence guided surgery on neurologic deficit can be reached. We recommend that surgeons use vigilance when operating near eloquent brain areas, particularly when relying on 5-ALA fluorescence-guided surgery, to optimize patient safety. However, more data is needed to properly decipher whether or not 5-ALA-guided HGG resection causes a statistically significant increase in neurological deficits. The extent of deficit is partially reliant on the skill of surgeon and the eloquence of the areas in which the tumor sits. Postoperative deficits could both be caused by surgeons resecting beyond the boundaries of fluorescent tumor and by 5-ALA highlighting functional brain tissue. Neurological deficits when using 5-ALA can be minimized by using imaging techniques, such as intraoperative neurophysiological monitoring, ultrasound, MRI, and iCT.
Additionally, 5-ALA used intraoperatively has been rarely found to be below fluorescing threshold. In this instance, 5-ALA succeeds in tissue uptake but fails in properly and effectively fluorescing. This failure could inhibit the surgeon from seeing the borders of the tumor, eliminating the intended benefits of using intraoperative 5-ALA in the first place. Thus, intimate knowledge of neuroanatomy and frequent anatomic re-orientation must be utilized in conjunction with 5-ALA to avoid new neurologic deficits by injuring eloquent brain or white matter tracts.
Limitations
Limitations of our study exist. The included studies are associated with different sample sizes and methods of analyses. Due to the overall small sample sizes because of the novelty of 5-ALA intraoperative usage in HGG resection, especially in the United States, the ability to draw similarities between cases is somewhat limited. There is a high degree of heterogeneity between studies with respect to their quantitative outcomes and as such, pooling and quantitative analysis of the meta-data is inappropriate statistically. Until more studies are available that can help overcome the risk of small study and publication biases, meta-analysis of the data should be deferred for now and only the systematic trends between studies highlighted. Finally, the studies tend to be performed by high volume cranial surgeons at expert centers. Generalizability of these results to surgeons who perform few cranial surgeries each year must be exercised with caution.
Conclusion
Despite potential drawbacks, overall, utilization of 5-aminolevulinic acid (5-ALA) may ultimately lead to better outcomes and longer survival rates for patients who undergo 5-ALA FGR. An implication of this finding is that 5-ALA FGR may be a better alternative to conventional surgical resection of HGG. Combining 5-ALA with an additional technical modality, such as iMRI, i-CT, ioUS, or CEUS may be even more beneficial than 5-ALA alone. This possibility warrants further research.
The results regarding postoperative neurologic deficits of surgeries using 5-ALA compared to those using white light do not coincide enough with one another to arrive at a clear conclusion. The reasoning behind this may be that the greater the amount resected, the higher the risk of excising healthy, functional tissue. The extent of deficit appears to be partially reliant on the skill of surgeon and the eloquence of the areas in which the tumor sits. When using 5-ALA, neurological deficits can be minimized by using imaging techniques, such as intraoperative neurophysiological monitoring, ultrasound, MRI, and iCT.
Data availability
The data that support the findings of this study are openly available at https://pubmed.ncbi.nlm.nih.gov and https://www.scopus.com/home.uri.
Abbreviations
- HGG:
-
High grade glioma
- 5-ALA:
-
δ-Aminolevulinic acid
- OS:
-
Overall survival
- PFS:
-
Progression-free survival
- FDA:
-
Food and drug administration
- GBM:
-
Glioblastoma multiforme
- EMA:
-
European medicine’s agency
- PRISMA:
-
Preferred reporting items for systematic reviews and meta-analyses
- WHO:
-
World Health Organization
- MD:
-
Mean difference
- CI:
-
Confidence interval
- iMRI:
-
Intraoperative magnetic resonance imaging
- CEUS:
-
Contrast-enhanced ultrasound
- ioUS:
-
Intraoperative ultrasound
- iCT:
-
Intraoperative computed tomography scan
- EOR:
-
Extent of resection
- GTR:
-
Gross total resection
References
Hadjipanayis CG, Stummer W (2019) 5-ALA and FDA approval for glioma surgery. J Neurooncol 141(3):479–486
Stummer W et al (2006) Fluorescence-guided surgery with 5-aminolevulinic acid for resection of malignant glioma: a randomised controlled multicentre phase III trial. Lancet Oncol 7(5):392–401
Lacroix M et al (2001) A multivariate analysis of 416 patients with glioblastoma multiforme: prognosis, extent of resection, and survival. J Neurosurg 95(2):190–198
Beller EM et al (2013) PRISMA for Abstracts: reporting systematic reviews in journal and conference abstracts. PLoS Med 10(4):e1001419
Stepp H et al (2007) ALA and malignant glioma: fluorescence-guided resection and photodynamic treatment. J Environ Pathol Toxicol Oncol 26(2):157–164
Kim SK et al (2014) Impact of fluorescence-guided surgery on the improvement of clinical outcomes in glioblastoma patients. Neuro-Oncol Pract 1(3):81–85
Picart T et al (2017) Is fluorescence-guided surgery with 5-ala in eloquent areas for malignant gliomas a reasonable and useful technique? Neurochirurgie 63(3):189–196
Slotty PJ et al (2013) The impact of improved treatment strategies on overall survival in glioblastoma patients. Acta Neurochir 155(6):959–963
Stummer W et al (2011) Counterbalancing risks and gains from extended resections in malignant glioma surgery: a supplemental analysis from the randomized 5-aminolevulinic acid glioma resection study: clinical article. J Neurosurg 114(3):613–623
Díez Valle R et al (2014) Observational, retrospective study of the effectiveness of 5-aminolevulinic acid in malignant glioma surgery in Spain (The VISIONA study). Neurologia 29(3):131–138
Ng WP et al (2017) Fluorescence-guided versus conventional surgical resection of high grade glioma: a single-centre, 7-year, comparative effectiveness study. Malays J Med Sci 24(2):78–86
Eljamel MS, Goodman C, Moseley H (2008) ALA and photofrin fluorescence-guided resection and repetitive PDT in glioblastoma multiforme: a single centre phase III randomised controlled trial. Lasers Med Sci 23(4):361–367
Eyüpoglu IY et al (2016) Supra-complete surgery via dual intraoperative visualization approach (DiVA) prolongs patient survival in glioblastoma. Oncotarget 7(18):25755
Hauser SB et al (2016) Combining 5-aminolevulinic acid fluorescence and intraoperative magnetic resonance imaging in glioblastoma surgery: a histology-based evaluation. Neurosurgery 78(4):475–483
Hickmann A-K, Nadji-Ohl M, Hopf NJ (2015) Feasibility of fluorescence-guided resection of recurrent gliomas using five-aminolevulinic acid: retrospective analysis of surgical and neurological outcome in 58 patients. J Neurooncol 122(1):151–160
Jacquesson T et al (2013) Surgery of high-grade gliomas guided by fluorescence: a retrospective study of 22 patients. Neurochirurgie 59(1):9–16
Aldave G et al (2013) Prognostic value of residual fluorescent tissue in glioblastoma patients after gross total resection in 5-aminolevulinic acid-guided surgery. Neurosurgery 72(6):915–920
Pichlmeier U et al (2008) Resection and survival in glioblastoma multiforme: an RTOG recursive partitioning analysis of ALA study patients. Neuro Oncol 10(6):1025–1034
Stummer W et al (2000) Fluorescence-guided resection of glioblastoma multiforme by using 5-aminolevulinic acid-induced porphyrins: a prospective study in 52 consecutive patients. J Neurosurg 93(6):1003–1013
Panciani PP et al (2012) Fluorescence and image guided resection in high grade glioma. Clin Neurol Neurosurg 114(1):37–41
Díez Valle R, Tejada Solis S (2015) To what extent will 5-aminolevulinic acid change the face of malignant glioma surgery? CNS Oncol 4(4):265–272
Della Puppa A et al (2014) 5-Aminolevulinic acid fluorescence in high grade glioma surgery: surgical outcome, intraoperative findings, and fluorescence patterns. Biomed Res Int 2014:232561
Della Puppa A et al (2013) 5-aminolevulinic acid (5-ALA) fluorescence guided surgery of high-grade gliomas in eloquent areas assisted by functional mapping. Our experience and review of the literature. Acta Neurochir 155(6):965–972
Díez Valle R et al (2011) Surgery guided by 5-aminolevulinic fluorescence in glioblastoma: volumetric analysis of extent of resection in single-center experience. J Neurooncol 102(1):105–113
Idoate MA et al (2011) Pathological characterization of the glioblastoma border as shown during surgery using 5-aminolevulinic acid-induced fluorescence. Neuropathology 31(6):575–582
Hefti M et al (2008) 5-Aminolaevulinic acid-induced protoporphyrin IX fluorescence in high-grade glioma surgery. Swiss Med Wkly 138(11–12):180–185
Schucht P et al (2012) Gross total resection rates in contemporary glioblastoma surgery: results of an institutional protocol combining 5-aminolevulinic acid intraoperative fluorescence imaging and brain mapping. Neurosurgery 71(5):927–935
Teixidor P et al (2016) Safety and efficacy of 5-aminolevulinic acid for high grade glioma in usual clinical practice: a prospective cohort study. PLoS ONE 11(2):e0149244
Eriksson M et al (2019) Improved treatment of glioblastoma—changes in survival over two decades at a single regional centre. Acta Oncol 58(3):334–341
Nabavi A et al (2009) Five-aminolevulinic acid for fluorescence-guided resection of recurrent malignant gliomas: a phase ii study. Neurosurgery 65(6):1070–1076
Chan DTM, Yi-Pin Sonia H, Poon WS (2018) 5-Aminolevulinic acid fluorescence guided resection of malignant glioma: Hong Kong experience. Asian J Surg 41(5):467–472
Cordova JS et al (2016) Semi-automated volumetric and morphological assessment of glioblastoma resection with fluorescence-guided surgery. Mol Imaging Biol 18(3):454–462
Cortnum S, Laursen RJ (2012) Fluorescence-guided resection of gliomas. Dan Med J 59(8):A4460
Pastor J et al (2013) Role of intraoperative neurophysiological monitoring during fluorescence-guided resection surgery. Acta Neurochir 155(12):2201–2213
Feigl GC et al (2010) Resection of malignant brain tumors in eloquent cortical areas: A new multimodal approach combining 5-aminolevulinic acid and intraoperative monitoring. J Neurosurg 113(2):352–357
Piquer J et al (2014) Fluorescence-guided surgery and biopsy in gliomas with an exoscope system. BioMed Res Int 2014:207974
Stummer W et al (2008) Extent of resection and survival in glioblastoma multiforme: identification of and adjustment for bias. Neurosurgery 62(3):564–576
Tykocki T et al (2012) Fluorescence-guided resection of primary and recurrent malignant gliomas with 5-aminolevulinic acid. Preliminary results. Neurol Neurochir Pol 46(1):47–51
Barbagallo GMV et al (2016) Portable intraoperative computed tomography scan in image-guided surgery for brain high-grade gliomas: analysis of technical feasibility and impact on extent of tumor resection. Oper Neurosurg 12(1):19–30
Coburger J et al (2015) Surgery for glioblastoma: impact of the combined use of 5-aminolevulinic acid and intraoperative MRI on extent of resection and survival. PLoS ONE 10(6):e0131872
Eyüpoglu IY et al (2012) Improving the extent of malignant glioma resection by dual intraoperative visualization approach. PLoS ONE 7(9):e44885
Nickel K et al (2018) The patients’ view: impact of the extent of resection, intraoperative imaging, and awake surgery on health-related quality of life in high-grade glioma patients—results of a multicenter cross-sectional study. Neurosurg Rev 41(1):207–219
Tsugu A et al (2011) Impact of the combination of 5-aminolevulinic acid-induced fluorescence with intraoperative magnetic resonance imaging-guided surgery for glioma. World Neurosurgery 76(1):120–127
Roder C et al (2014) Maximizing the extent of resection and survival benefit of patients in glioblastoma surgery: high-field iMRI versus conventional and 5-ALA-assisted surgery. Eur J Surg Oncol 40(3):297–304
Schatlo B et al (2015) Outcomes after combined use of intraoperative MRI and 5-aminolevulinic acid in high-grade glioma surgery. Neuro Oncol 17(12):1560–1567
Yamada S et al (2015) Role of neurochemical navigation with 5-aminolevulinic acid during intraoperative MRI-guided resection of intracranial malignant gliomas. Clin Neurol Neurosurg 130:134–139
Della Pepa GM et al (2020) 5-Aminolevulinic acid and contrast-enhanced ultrasound: the combination of the two techniques to optimize the extent of resection in glioblastoma surgery. Neurosurgery 86(6):E529–E540
Neidert MC et al (2016) The influence of intraoperative resection control modalities on survival following gross total resection of glioblastoma. Neurosurg Rev 39(3):401–409
Colapaoli L et al (2006) A case of anaphylactic shock possibly caused by intravesical hexvix. Acta Anaesthesiol Scand 50(9):1165–1167
Kreth FW et al (2013) Gross total but not incomplete resection of glioblastoma prolongs survival in the era of radiochemotherapy. Ann Oncol 24(12):3117–3123
Nader S et al (2011) An extent of resection threshold for newly diagnosed glioblastomas. J Neurosurg 115(1):3–8
Schucht P et al (2014) 5-ALA complete resections go beyond MR contrast enhancement: shift corrected volumetric analysis of the extent of resection in surgery for glioblastoma. Acta Neurochir 156(2):305–312
Funding
This systematic review received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material, preparation, data collection, and analysis were performed by TE, DE, and VML. The first draft of the manuscript was written by TE; DGE and MEI commented/edited and VML executed statistical/data analysis. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The following authors have no financial disclosures or personal conflicts of interest: TAE, DE, VL, LD, RJK, MEI.
Ethical approval
Ethical approval was not applicable for this study as only publicly accessible data was utilized.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Eatz, T.A., Eichberg, D.G., Lu, V.M. et al. Intraoperative 5-ALA fluorescence-guided resection of high-grade glioma leads to greater extent of resection with better outcomes: a systematic review. J Neurooncol 156, 233–256 (2022). https://doi.org/10.1007/s11060-021-03901-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-021-03901-9