Abstract
The recurrence of glioblastoma (rGBM) is inevitable and often short-term. Therefore, information on the prognosis and effectiveness of tumor-specific versus purely palliative approaches should be more in-depth than a mere list of available treatment options for patients in this situation. However, follow-up data on the course of the disease in unselected patient populations after completion of primary treatment are scarce. This single-center analysis investigated the rate and number of glioblastoma recurrences after initial radiotherapy in 189 consecutive GM patients, focusing on the incidence of early death and the frequency of tumor-specific treatment (TST) versus best-supportive care (BSC) as well as the outcomes for the different approaches. In 61 % of initial population first recurrence (rGBM) could be determined by histology or imaging. 47 % received TST. 58 % of the patients with rGBM and TST were diagnosed with a second recurrence. Up to five recurrences were treated. 35–45 % of patients died before undergoing imaging studies to confirm the next recurrence. Multivariate analysis identified male sex and KPS score as independent factors (p < 0.01) for the choice of TST over BSC. Median overall survival from the diagnosis of first recurrence was 267 days in the TST group versus 65 days in patients receiving BSC (p < 0.0001). Nearly half of all rGBM patients received second-line TST, but a remarkably high proportion died early. Gender and KPS played a role in the choice of TST over BSC for recurrence treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
WHO grade IV glioblastoma (GBM), the most common primary brain malignancy in adults, has a poor prognosis—survival at 24 month still only 14–26 % [1, 2]. The current standard of care for patients with newly diagnosed glioblastoma is surgery followed by radiotherapy with concurrent and adjuvant temozolomide (TMZ) [1, 3, 4]. Nevertheless, intracranial glioblastoma recurrences develop in almost all of these patients. Surgery, radiotherapy and chemotherapy are discussed as treatment options for recurrent glioblastoma (rGBM) in the literature [5–10]. The published data on the prospects of success of these therapeutic options are limited to highly selected patient populations. The Karnofsky performance status (KPS), MGMT promoter status, and patient age are possible prognostic factors for the overall survival after glioblastoma treatment [11–13].
However, there is little information about how many rGBM patients are actually suitable candidates for tumor-specific treatment, how often it is performed, and whether it achieves better results than BSC alone. Therefore, the aim of this retrospective single-center study was to determine how often TST is performed in rGBM patients, what selection criteria are applied, and which results are achieved with BSC versus TST in different prognostic groups.
Materials and methods
Patients
All 189 adults treated for newly diagnosed GBM at our radiotherapy department from January 1, 2006 to December 31, 2011 were included in the study. The patients generally received surgical treatment followed by radiotherapy plus concomitant and adjuvant TMZ. Patients in reduced general condition received radiotherapy alone. After completion of treatment, the patients were given the option to return to the Department of Neurosurgery and Radiotherapy at quarterly intervals for follow-up. Follow-up consisted of a clinical examination and magnetic resonance imaging (MRI) every 3 months. If the neurological status deteriorated, MRI follow-up was performed immediately, i.e., earlier than 3 months. GBM cases with an MRI suggestive of disease progression were jointly discussed at the interdisciplinary tumor board. The diagnosis of recurrence was made by consensus between neurosurgeons, neuropathologists, neuroradiologists, neurologists and radiation oncologists. Regarding the differential diagnosis, if pseudoprogression was suspected, another MRI study (in some cases FET-PET/CT) was performed 1 month later. Glioblastoma recurrence and progression was defined as evidence of a new contrast-enhancing lesion or an increase in size of a known contrast-enhancing lesion on MRI, analogous to the radiological criteria of MacDonald and the Response Assessment in Neuro-Oncology (RANO) classification [14, 15]. If pseudoprogression was suspected, steroid treatment was administered and a new MRI study was performed in 4–6 weeks for differential diagnosis. SUVmax > 2 was the criterion used for the diagnosis of recurrence by FET-PET/CT.
Tumor resectability was assessed in every case of recurrence. If the results were equivocal, even after a second MRI, a specimen was obtained on at least one occasion. If there were no viable surgical treatment options, the feasibility of second irradiation was examined. For second radiation, a dose of up to 45 Gy was delivered to the already irradiated area, and rGBM located outside the former irradiation volume was irradiated with 60 Gy. The cumulative doses of the entire irradiation series did not exceed 59 Gy in the brainstem and 54 Gy in the optical system. The feasibility of cancer drug treatment was also evaluated. The re-induction of temozolomide at conventional doses or in a dose-dense temozolomide regimen was the first chemotherapy option considered for treatment of first recurrences. The second chemotherapy option considered was bevacizumab, which was administered with or without CPT-11, depending on insurance coverage. Procarbazine/CCNU was the third-line chemotherapy.
Statistical analysis
The endpoint of the study was median overall survival (mOS). The zero point was the date of primary diagnosis or recurrence diagnosis, depending on the analytical objective. The cutoff date for the statistical analysis was September 2, 2013. Frequency analysis was performed using the Chi square test, Fischer’s exact test and logistic regression. OS was calculated using the Kaplan–Meier method and compared with the log rank test. Multivariate analysis was performed to find independent factors for survival by introducing parameters that showed significant differences in survival in the univariate analysis (p < 0.05). The statistical analyses were performed with SPSS version 21.
Results
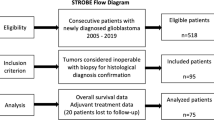
Frequency of recurrence treatment (Fig. 1)
On the cut-off date, eight of the 189 patients studied had tumor-free survival and three were lost to follow-up. Of the remaining 178 patients (investigated population), 109 (61 %) had first recurrences confirmed by histology or imaging, diagnosed at a median of 304 days after primary diagnosis. 69/178 patients (39 %) died before the diagnosis of recurrence could be confirmed by imaging or other methods, a median 150 days after primary diagnosis. 35 of these patients died during primary treatment or within 6 weeks of primary treatment, and another 34 did not return to our hospital for follow-up, nor to their primary care physicians or attending oncologists, who were asked about patients lost to follow-up. Therefore, it must be assumed that the general condition of these 69 patients had deteriorated so severely that neither diagnostics nor treatment was possible.
TST was performed for 84/178 (47 %) first recurrences, achieving a median OS of 267 days after first-recurrence diagnosis and 575 days after primary diagnosis. 25 patients had a confirmed diagnosis of recurrence, but the interdisciplinary tumor board decided not to treat the first recurrence due to the poor general condition of the patient. This BSC subgroup achieved a median OS of 65 days after first-recurrence diagnosis and 290 days after primary diagnosis.
In summary: 94 (53 %) of 178 patients did not receive second-line TST (25 pts with “untreated” first recurrences and 69 pts before diagnosis of recurrence), and 47 % of all patients who received primary treatment received second-line TST at first recurrence. Median OS after primary diagnosis was significantly different for the 84 TST patients (575 days) versus the 94 patients (189 days, p = 0.000).
Of the 84 patients receiving TST for a first recurrence, two were still second recurrence-free at the cut-off date, 49/84 (58 %) had a confirmed second recurrence, and the other 33/84 (39 %) were in such poor general condition after second line TST that no further diagnostic studies were possible. 32 (65 %) of the 49 patients with a confirmed second recurrence (18 % of the initial population) received TST (third-line treatment).
By the cut-off date, a third recurrence was diagnosed in 17 patients (53 % of those treated for a second recurrence, 9.5 % of the initial population), a fourth recurrence was diagnosed in three patients, and a fifth recurrence in one patient.
In summary, recurrent glioblastoma was diagnosed in 61–53 % of our previously treated GBM patients, and treated in 65–82 % of patients with one to three confirmed recurrences. This percentage was remarkably constant. In the other patients with non-confirmed recurrences, the cancer was obviously so aggressive that further diagnostic studies were not possible.
Treatments performed by recurrence number
In our cohort, we observed one to five recurrences per patient, which were treated as shown in Table 1. Both the rate of surgical resection and the probability of multimodal treatment decreased with the individual number of recurrences.
Intervals between recurrence treatments and survival times after recurrence diagnosis and treatment
First recurrences (n = 109) were diagnosed a median of 304 days after primary diagnosis, second recurrences (n = 49) a median of 151 days after first recurrence, third recurrences (n = 17) a median of 184 days after second recurrence, fourth recurrences (n = 6) a median 61 days after third recurrence, and the fifth recurrence (n = 1) a median 215 days after the fourth recurrence.
Patients (n = 109) survived a median of 214 days after diagnosis of the first recurrence as determined by imaging, resection or biopsy, 199 days after diagnosis of the second recurrence (n = 49), 168 days after diagnosis of the third recurrence (n = 17), and 85 days after diagnosis of fourth recurrence (n = 6). The one patient with a fifth recurrence survived 136 days after diagnosis of fifth recurrence (Table 2).
First-recurrence patients who received TST (group A, n = 84) survived 267 days after first recurrence diagnosis, while those who received BSC (group B, n = 25) survived 65 days. Second-recurrence patients survived 262 days after recurrence diagnosis with TST (group A, n = 32), and 106 days with BSC (group B, n = 17). Third-recurrence patients receiving TST (n = 14) survived 214 days after recurrence diagnosis, while those receiving BSC (n = 3) survived 18 days. Fourth-recurrence patients survived 137 days after recurrence diagnosis with TST (n = 3) compared to 50 days with BSC (n = 3). The one patient with a 5th recurrence survived 136 days with TST. There is a significant difference in OS in favor of patients receiving TST versus BSC at first recurrence, and although significance is not reached for 2nd, 3rd and 4th recurrence there was a trend in favor of TST (Table 2).
Criteria for selection of TST versus BSC for first recurrence
Key decision variables for selecting tumor-specific treatment versus “no treatment” (BSC) for first recurrences were gender, KPS score and time until the recurrence diagnosis (Table 3). TST of first recurrences was administered in 88 % of men compared to only 65 % of women (p = 0.008, logistic regression). Patients with a KPS score ≥ 80 % received TST more often than those with a KPS score < 80 % (97 % vs. 66 %, p = 0.005, multivariate).
The indication for recurrence treatment was not influenced by age (p = 0.052), size (p = 0.71), tumor growth pattern (unifocal vs. multifocal; p = 0.174), or resection status at initial treatment (biopsy/incomplete resection vs. complete resection; p = 0.055).
Comparison of the periods 2006–2008 versus 2009–2011 showed a slight increase in the rate of diagnosis and treatment of first, second and third recurrences over time (Table 4). TST of first recurrences was performed in 43 % of patients in the first cohort (2006–2008) compared to 51 % of those in the second cohort (2009–2011). From one period to the next, the rates rose from 17 to 19 % for the second recurrence, from 5 to 11 % for the third recurrence. Likewise, the rate of diagnosis rose from 56 to 66 % for first recurrence, from 26 to 29 % for second recurrence and from 8 to 11 % for third recurrence between the two time periods.
Parameters for survival after first-recurrence diagnosis
After first-recurrence diagnosis (n = 109), univariate analysis showed differences in survival for the following parameters: age (≥65 vs. <65 years, p = 0.005), KPS score (≥80 % vs. <80 %, p = 0.000), recurrent tumor size (>30 cm3 vs. ≤30 cm3, p = 0.02), recurrence interval (≥365 days vs. <365 days, p = 0.004) and unifocal versus multifocal recurrence (p = 0.048). No statistically significant differences in survival were detected for sex. In the 84 patients, survival did not correlate with the type of recurrence treatment: with and without resection (p = 0.12), with and without radiotherapy (p = 0.19), with and without chemotherapy (p = 0.98), and monotherapy versus combination therapy (p = 0.20). Multivariate analysis identified KPS score, age and recurrence interval as independent parameters for survival.
Based on the hazard ratio (HR), patients with a KPS score of <80 % were 3.41 times more likely to die than those with a KPS score of ≥80 %. Patients aged 65 years and older were 1.84 times more likely to die than those under 65. Patients with an initial recurrence interval of <365 days were 1.65 times more likely to die than those with an interval of 365 days or longer. Therefore, adverse prognostic factors were older age, impaired KPS score and short initial recurrence interval.
After dividing the patients into two groups according to the three adverse prognostic factors (only one or none adverse prognostic factor vs. two or three)—median OS for the first group with one or less adverse prognostic was found to be 478 days with TST (n = 43) versus 135 days with BSC (n = 5, p = 0.016). In the second group, median OS was 206 days with TST (n = 41) and 57 days with BSC (n = 20, p = 0.000). The benefit of TST decreased with increasingly poor prognosis. The difference in OS between TST and BSC was 343 days in the first group and 149 days in the latter.
Discussion
This study is the first to provide data on the frequency and effectiveness of treatment of the first to fourth glioblastoma recurrences. Such detailed follow-up data describing the course of the disease after completion of primary treatment were not available until now. We showed that rGBM could be treated in 30–50 % of cases, and that treatment very likely had a positive effect on their patient’s prognosis (median OS after recurrence therapy: 130–260 days). This is clinically relevant because the proportion of patients with longer survival is increasing due to the availability of more effective initial treatments, such as temozolomide, navigation-guided surgery, and intensity modulated radiation therapy [16–18]. The success of primary treatment has raised expectations with regard to recurrence treatment; GBM patients and their family members are at least asking about the therapeutic success of recurrence treatment. When counseling glioblastoma patients and their family members, it is important to provide an estimate of the patient’s remaining lifetime. Our data shed some light on this subject.
There have been previous reports in the lay press about long-term GBM survivors with multiple glioblastoma recurrences and treatments, but it was unclear whether these were only isolated cases. Our data show that a third or fourth recurrence can actually be diagnosed and treated in approximately 20–40 % of patients. This finding was not limited to isolated cases but was observed in a well-defined population of glioblastoma patients. Until now, epidemiological data supporting these claims were lacking. Since all patients with a diagnosis of primary GBM who were treated at our hospital during the observation period were included in the analysis, the findings of this study are certainly representative of a large hospital.
More specifically, the relative percentage of patients receiving TST for rGBM remained approximately constant from one recurrence to the next: the observed percentages were 47 % for the first recurrence, 38 % for the second recurrence, and 44 % for later recurrences. This could be due to the effectiveness of recurrence treatment, but may also be related to tumor biology. We were unable to examine molecular markers for long-term survival due to the retrospective nature of the study. In the Stupp trial, follow-up was more intensive: 96.7 % of patients were diagnosed with rGBM, and 89.5 % were treated for recurrences [19]. However, the authors only provided data on the first recurrence and based their diagnosis of rGBM on the MacDonald criteria which, unlike our diagnostic criteria, are not solely based on MRI findings, but also take deterioration of the neurological status into account. Discrepancies in evaluating the presence of recurrence makes it difficult to compare the follow-up data and frequencies of treatment between our unselected patient population and other study cohorts.
It is even more important to identify parameters associated with the initiation of tumor-specific treatment of rGBM since reliable comparative data from non-study populations are also lacking on this subject. As was already known, younger GBM patients are more likely to receive TST than older GBM patients. The same is true for patients with long versus short treatment-free intervals and a good versus low KPS [11, 19, 20]. The latter was also be duplicated in our study. Biological factors were not studied here because they were not used in therapeutic decision-making, and because of the lack of reproducibility of MGMT hypermethylation determinations [10]. Surprisingly, we detected recurrence treatment bias in favor of men, independent of all other factors. For example, 51/58 men received TST for rGBM compared to only 33/51 women. The sex ratio (M:F) was only 1.14:1 at the time of primary treatment, and remained relatively constant (1.13:1) until the date of first-recurrence diagnosis. After the onset of first recurrence treatment, the sex ratio rose to 1.54:1, where it remained relatively stable. Likewise, the sex ratio was 1.45:1 and 1.46:1 at the time of second-recurrence diagnosis and treatment. Whether this recurrence treatment bias in favor of male patients results in better survival is debatable since long-term survivors are often young women [21]. Speculatively, we attributed the recurrence treatment bias in favor of males to the fact that married men are better cared for by their wives [22] or that family members place more pressure on men to undergo treatment than women.
Tumor-specific treatment extends the lives of rGBM patients by several months. As shown in the present study, patients receiving specific treatment for recurrences survived 267 days after the first recurrence, 262 days after the second recurrence, and 214 days after the third recurrence. This is consistent with data from single-center monotherapy trials on first recurrence treatment [5–7, 23, 24]. Even though the difference in OS in patients treated by TST versus BSC is only significant for the first recurrence, there is a trend, without statistical significance towards longer survival for TST in favor of BSC in the 2nd, 3rd and 4th recurrence.
One indication of the effectiveness of treatment is the observation that, in the individual prognosis groups divided by differences in KPS score, patient age and initial recurrence interval, patients receiving TST outlived those receiving BSC alone. Even elderly first-recurrence patients with a low KPS score survived 178 days with TST compared to only 53 days with BSC. Early professional palliative care can help to improve survival, as is well demonstrated in lung cancer patients [25]. Nonetheless, it is important to remember that even recurrent glioblastoma patients who are elderly or have poorer KPS scores can benefit from additional tumor-specific treatment.
Until now, little was known about how often GBM patients use multidisciplinary follow-up services, how often this results in an offer to provide treatment, and how often the offer to provide tumor-specific or purely symptomatic treatment is accepted. In particular, 40–60 % of our GBM patients did not return for follow-up and died within a short time. This patient subgroup is considered to be representative of the typical course of the disease. The cause of death is often uncritically classified as disease progression, and the fact that these patients have concomitant diseases, and that complications can arise due to treatments, concomitant treatments and malignant diseases is often ignored. Complications of steroid therapy, such as hyperglycemia, arterial hypertension or other thromboembolic events, and symptomatic epilepsy are a case in point [26–28]. For example, the incidence of thromboembolic events in glioblastoma patients is estimated to be 20–30 % [27, 29]. The importance of opportunistic infections is also unclear. Goerig et al. [30] suggested that the association of CMV infection with neurological deterioration may be misinterpreted as progression. In the future, special emphasis should be placed on the treatment of complications of various treatments and the prevention of opportunistic infections as this alone could possibly contribute to the survival of GBM patients.
In summary, our data show the importance of providing GBM patients adequate follow-up services and TST, which can contribute to improving the prognosis of glioblastoma.
References
Stupp R, Mason WP, van den Bent MJ et al (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996
Dolecek TA, Propp JM, Stroup NE, Kruchko C (2012) CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2005–2009. Neuro Oncol 14(Suppl 5):1–49
Stummer W, Pichlmeier U, Meinel T et al (2006) Fluorescence-guided surgery with 5-aminolevulinic acid for resection of malignant glioma: a randomised controlled multicentre phase III trial. Lancet Oncol 7:392–401
Walker MD, Strike TA, Sheline GE (1979) An analysis of dose-effect relationship in the radiotherapy of malignant gliomas. Int J Radiat Oncol Biol Phys 5:1725–1731
Weller M, Tabatabai G, Kastner B et al (2015) MGMT promoter methylation is a strong prognostic biomarker for benefit from dose-intensified temozolomide rechallenge in progressive glioblastoma: the DIRECTOR trial. Clin Cancer Res 21:2057–2064
Combs SE, Edler L, Rausch R et al (2013) Generation and validation of a prognostic score to predict outcome after re-irradiation of recurrent glioma. Acta Oncol 52:147–152
Park JK, Hodges T, Arko L et al (2010) Scale to predict survival after surgery for recurrent glioblastoma multiforme. J Clin Oncol 28:3838–3843
Taal W, Oosterkamp HM, Walenkamp AM et al (2014) Single-agent bevacizumab or lomustine versus a combination of bevacizumab plus lomustine in patients with recurrent glioblastoma (BELOB trial): a randomised controlled phase 2 trial. Lancet Oncol 15:943–953
Fokas E, Wacker U, Gross MW et al (2009) Hypofractionated stereotactic reirradiation of recurrent glioblastomas: a beneficial treatment option after high-dose radiotherapy? Strahlenther Onkol 185:235–240
Levin VA, Mendelssohn ND, Chan J et al (2015) Impact of bevacizumab administered dose on overall survival of patients with progressive glioblastoma. J Neurooncol 122:145–150
Fietkau R, Putz F, Lahmer G et al (2013) Can MGMT promoter methylation status be used as a prognostic and predictive marker for glioblastoma multiforme at the present time? A word of caution. Strahlenther Onkol 189:993–995
Gorlia T, Stupp R, Brandes AA et al (2012) New prognostic factors and calculators for outcome prediction in patients with recurrent glioblastoma: a pooled analysis of EORTC Brain Tumour Group phase I and II clinical trials. Eur J Cancer 48:1176–1184
Balducci M, Diletto B, Chiesa S et al (2014) Low-dose fractionated radiotherapy and concomitant chemotherapy for recurrent or progressive glioblastoma: final report of a pilot study. Strahlenther Onkol 190:370–376
Macdonald DR, Cascino TL, Schold SC Jr, Cairncross JG (1990) Response criteria for phase II studies of supratentorial malignant glioma. J Clin Oncol 8:1277–1280
Wen PY, Macdonald DR, Reardon DA et al (2010) Updated response assessment criteria for high-grade gliomas: response assessment in neuro-oncology working group. J Clin Oncol 28:1963–1972
Brodbelt A, Greenberg D, Winters T, Williams M, Vernon S, Collins VP, National Cancer Information Network Brain Tumour G (2015) Glioblastoma in England: 2007–2011. Eur J Cancer 51:533–542
Ho VK, Reijneveld JC, Enting RH, Bienfait HP, Robe P, Baumert BG, Visser O, Dutch Society for N-O (2014) Changing incidence and improved survival of gliomas. Eur J Cancer 50:2309–2318
Barone DG, Lawrie TA, Hart MG (2014) Image guided surgery for the resection of brain tumours. Cochrane Database Syst Rev 28:1
Stupp R, Hegi ME, Mason WP et al (2009) Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol 10:459–466
Kim HR, Kim KH, Kong DS, Seol HJ, Nam DH, Lim DH, Lee JI (2015) Outcome of salvage treatment for recurrent glioblastoma. J Clin Neurosci 22:468–473
Krex D, Klink B, Hartmann C et al (2007) Long-term survival with glioblastoma multiforme. Brain 130:2596–2606
Su D, Stimpson JP, Wilson FA (2015) Racial disparities in mortality among middle-aged and older men: does marriage matter? Am J Mens Health 9:289–300
Ening G, Huynh MT, Schmieder K, Brenke C (2015) Repeat-surgery at glioblastoma recurrence, when and why to operate? Clin Neurol Neurosurg 136:89–94
Hasan S, Chen E, Lanciano R et al (2015) Salvage fractionated stereotactic radiotherapy with or without chemotherapy and immunotherapy for recurrent glioblastoma multiforme: a single institution experience. Front Oncol 15:106
Bakitas MA, Tosteson TD, Li Z et al (2015) Early versus delayed initiation of concurrent palliative oncology care: patient outcomes in the ENABLE III randomized controlled trial. J Clin Oncol 33:1438–1445
Tieu MT, Lovblom LE, McNamara MG et al (2015) Impact of glycemia on survival of glioblastoma patients treated with radiation and temozolomide. J Neurooncol 124:119–126
Silvani A, Gaviani P, Lamperti E et al (2011) Metabolic, electrolytes disorders and tromboembolic risk in malignant glioma patients. Neurol Sci 32(Suppl 2):229–231
Mayer A, Vaupel P, Struss HG et al (2014) Strong adverse prognostic impact of hyperglycemic episodes during adjuvant chemoradiotherapy of glioblastoma multiforme. Strahlenther Onkol 190:933–938
Pan E, Tsai JS, Mitchell SB (2009) Retrospective study of venous thromboembolic and intracerebral hemorrhagic events in glioblastoma patients. Anticancer Res 29:4309–4313
Goerig NL, Gaipel U, Frey B et al (2015) Cytomegalovirus-Encephalitis als Ursache neurologischer Verschlechterung während der Radiotherapie und/oder Chemotherapie des Cerebrums. Strahlenther Onkol 191(Suppl 1):39
Acknowledgments
The present work was performed in fulfillment of the doctoral requirements of Friedrich-Alexander University of Erlangen-Nuremberg for Rieke Steffens obtaining the degree title: “Dr. med”.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human and Animal Rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Rieke Steffens and Sabine Semrau share joint first authorship.
Rights and permissions
About this article
Cite this article
Steffens, R., Semrau, S., Lahmer, G. et al. Recurrent glioblastoma: who receives tumor specific treatment and how often?. J Neurooncol 128, 85–92 (2016). https://doi.org/10.1007/s11060-016-2079-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-016-2079-z