Abstract
Breast cancer is a leading cause of mortality and the most prevalent form of malignant tumor among women worldwide. Breast cancer cells exhibit an elevated glycolysis and altered glucose metabolism. Moreover, these cells display abnormal glycosylation patterns, influencing invasion, proliferation, metastasis, and drug resistance. Consequently, targeting glycolysis and mitigating abnormal glycosylation represent key therapeutic strategies for breast cancer. This review underscores the importance of protein glycosylation and glucose metabolism alterations in breast cancer. The current research efforts in developing effective interventions targeting glycolysis and glycosylation are further discussed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In 2020, the global incidence of cancer reached 19.3 million new cases, resulting in nearly 10 million deaths. Among these cases, breast cancer is the most prevalent type of malignant tumor [1, 2] and remains a prominent cause of mortality among women worldwide, posing significant threats to their well-being [3]. Physiologically, normal breast ducts consist of basal membrane, luminal epithelial cells, and myoepithelial cells. However, the onset of breast cancer is marked by myoepithelial cell shrinkage, stromal cell proliferation, and basement membrane degradation [4,5,6]. As the basement membrane and myoepithelial cells diminish, cancer cells can invade neighboring tissues. The mortality in most breast cancer cases is attributed to metastasis to critical organs such as the brain, liver, lungs, and bones, leading to compromised organ function and eventual demise [7]. Breast cancer is characterized by heterogeneity, comprising various subtypes with distinct morphological and clinical characteristics. Among these, ductal carcinoma arising from breast epithelial cells is the predominant subtype [8, 9]. Clinically, breast cancer cells are classified into four subtypes according to their expression levels of estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor-2 (HER2), commonly known as Luminal A, Luminal B, HER2-positive, and triple-negative breast cancer (TNBC) [10]. Among these subtypes, Luminal A breast cancer patients constitute the largest number with the most favorable prognosis, whereas TNBC patients exhibit the worst prognosis [11].
Carbohydrates provide nutrients and calories for the body. When carbohydrates are digested, they are converted to glucose. Previous studies have found altered glucose metabolism and abnormal glycosylation in breast cancer cells [12, 13]. On the one hand, the modified glucose metabolism in these cells primarily occurs via the glycolytic pathway, which provides cells with sufficient energy and promotes their proliferative capacity, thus contributing to the development of breast cancer. On the other hand, abnormal glucose metabolism or aerobic glycolysis may also alter the glycosylation profile of cancer cells, which in turn affects tumor growth, invasion, and metastasis [14, 15]. This review elucidates the impact of glucose metabolism and glycosylation in breast cancer cells and discuss potential therapeutic interventions targeting these alterations.
Glucose metabolism in breast cancer cells - Warburg effect
Glucose serves as a vital nutrient essential for cell growth. In normal breast cells, glucose metabolism predominantly involves the tricarboxylic acid (TCA) cycle in mitochondria, the glycolysis pathway, and the pentose phosphate pathway (PPP) in the cytoplasm. A critical step in glucose metabolism is the phosphorylation of glucose to glucose-6-phosphate by hexokinase (HK). The resulting glucose-6-phosphate not only participates in glycolysis but also intersects with the PPP, glycogen synthesis, and hexosamine biosynthesis pathway (HBP) [16]. In addition, glycolytic intermediates may participate in other metabolic pathways, such as the serine pathway, to provide the energy and substances necessary for cell proliferation [17].
Cancer development and progression represent intricate processes [18]. The alteration of the genome in a cell disrupts the normal processes of apoptosis and aging [19]. Cells proliferate indefinitely and autonomously change various metabolic pathways to satisfy their own and biosynthetic demands for large amounts of nutrients and energy [20]. The onset, progression, and metastasis of breast cancer are linked to metabolic reprogramming in tumor cells [21]. The energy metabolism of normal breast cells is mainly generated by oxidative phosphorylation, with only a small percentage coming from glycolysis [22].
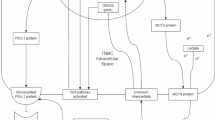
Breast cancer cells exhibit heightened glucose uptake. Glucose metabolism is mainly dependent on glycolysis and produces lactate (Fig. 1). This phenomenon is commonly referred to as the “Warburg effect” [23]. Worth noting, although glycolysis is an extremely important process in tumor cell metabolism, the metabolic reprogramming induced by increased glycolysis is uncommon in all cancers types, including various subtypes of breast cancer. For instance, analysis of gene expression signatures revealed significant variations in glycolysis scores among different cancer types. Some cancers exhibit elevated glycolysis levels, such as kidney renal clear cell carcinoma, head and neck squamous cell carcinoma, lung squamous cell carcinoma, and colon adenocarcinoma, while others, such as the thyroid carcinoma, prostate adenocarcinoma, stomach adenocarcinoma, and thymoma, demonstrate lower glycolysis levels [24].
Glycolysis
Glycolysis is an oxygen-independent metabolic pathway converting glucose to pyruvate, which occurs in the cytoplasm of most organisms [22, 25]. After being transported into the cytoplasm by glucose transporters, glucose is subsequently converted into fructose-1,6-bisphosphate by HK, glucose-6-phosphate isomerase, and phosphofructokinase. Aldolase then breaks down the 6-carbon skeleton of fructose-1,6-diphosphate to yield dihydroxyacetone phosphate and glyceraldehyde-3-phosphate. In addition, these two molecules can be transformed by triphosphate isomerase. After a series of reactions catalyzed by glyceraldehyde-3-propionate dehydrogenase, phosphoglycerate kinase, phosphoglycerate mutase, enolase, and pyruvate kinase, the glyceraldehyde-3-phosphate is metabolized into pyruvate and produces ATP.
In normal breast cells with adequate oxygen and functional mitochondria, pyruvate generated from glycolysis undergoes oxidation to form acetyl-CoA catalyzed by pyruvate dehydrogenase. Acetyl-coenzymes then enter the tricarboxylic acid (TCA) cycle, participating in oxidative phosphorylation (OXPHOS) to yield carbon dioxide and ATP. Conversely, OXPHOS is impeded under hypoxic conditions, leading to the conversion of pyruvate to lactate by lactate dehydrogenase (LDH). In addition, when mitochondria are defective, OXPHOS is reduced, and lactate production is increased.
For breast cancer cells, pyruvate generated from glycolysis is often converted to lactate even in the presence of oxygen, instead of entering the TCA cycle, which is referred to as “aerobic glycolysis” [26,27,28]. The lactate produced is extruded from the cell by the monocarboxylate transporter (MCT) to maintain intracellular pH stability [29].
Other glucose metabolic pathways
Pentose phosphate pathway (PPP)
The PPP is a metabolic route that occurs in conjunction with glycolysis [30], providing pentose phosphate for nucleic acid synthesis and NADPH for fatty acid synthesis and cell survival. Therefore, it plays a key role in cell growth [31]. It comprises two branches, an oxidative branch and a non-oxidative branch. The oxidative branch involves three irreversible reactions of glucose-6-phosphate, leading to the production of NADPH and ribulose 5-phosphate. Enzymes in this branch include 6-phosphogluconate dehydrogenase and glucose 6-phosphate dehydrogenase. The non-oxidizing branch produces pentose phosphate after a series of reactions, which is used in the synthesis of ribonucleotides. Additionally, this branch yields glycolytic intermediates like fructose 6-phosphate and glyceraldehyde 3-phosphate, crucial for amino acid synthesis. Enzymes in the non-oxidative branch include ribulose-5-phosphate epimerase, ribose 5-phosphate isomerase, transketolase, and transaldolase [32].
Hexosamine biosynthesis pathway (HBP)
HBP converts glucose into uridine diphosphate N-acetylglucosamine (UDP-GlcNAc) through a six-step procedure. The final product UDP-GlcNAc can participate in protein modification [33]. Initially, glucose-6-phosphate from glycolysis is converted to fructose-6-phosphate by glucose-6-phosphate isomerase (GPI). Then, via fructose-6-phosphate amidotransferase (GFAT), glucosamine-6-phosphate N-acetyltransferase 1 (GNA1), phosphoglucomutase 3 (PGM3) and UDP-N-acetylhexosamine pyrophosphorylase (UAP), fructose-6-phosphate is converted to UDP-GlcNAc. UDP-GlcNAc is important substrate for not only N-glycan synthesis but also the formation of other monosaccharides and eventually glycans. At the same time, UDP-GlcNAc plays a crucial role in promoting glycosylation as a key regulatory metabolite.
Research progress of glycolysis inhibitors in the treatment of breast cancer
Compared to normal breast cells, the energy metabolism of breast cancer cells increases. However, cells produce less ATP per glucose molecule, and cancer cells need to take up more glucose to meet their basic metabolic needs. Glycolysis serves as the main pathway of sugar metabolism in tumor cells, and the use of this process to develop anticancer drugs has been recognized as a viable approach [27]. Therefore, targeting glucose uptake characteristics of tumor cells to impede glucose utilization and consequently disrupt energy supply represents a therapeutic strategy for breast cancer. At present, research has identified several anti-glycolysis drugs for breast cancer treatment, targeting glucose transporters (Table 1) or enzymes involved in glycolysis (Table 2).
Inhibitors targeting glucose transporters (SGLTs and GLUTs)
The transportation of glucose into cancer cells requires glucose transporters. Two major glucose transporters have been identified including glucose transporters (GLUTs) and sodium-glucose cotransporters (SGLTs) [34]. Among them, GLUTs transport glucose along the concentration gradient, while SGLTs use the electrochemical gradient of Na+ to actively transport glucose. It is noteworthy that the magnitude of extracellular glucose concentration is independent of glucose transport by SGLTs [35].
SGLT1 and SGLT2 are expressed in breast cancer cells compared to normal breast cells. For SGLT1, in vitro experiments have shown that down-regulating of SGLT1 levels inhibits cell growth in breast cancer cell lines of TNBC [36] or HER2-positive [37]. It has been reported that KGA-2727 [38] and mizagliflozin [39] are two specific SGLT1 inhibitors. Tsunokake et al. evaluated the impact of mizagliflozin and KGA-2727 on breast cancer MCF-7 and MDA-MB-468 cells, respectively, and found an inhibition of cell proliferation by these two inhibitors. In addition, mizagliflozin also exhibited inhibitory effects on MCF-7 cell proliferation under low glucose conditions [35]. For SGLT2, canagliflozin [40], dapagliflozin [40] and ipragliflozin [41] have been reported to serve as specific inhibitors of SGLT2, which have been applied to clinical treatment of type 2 diabetes. All these SGLT2 inhibitors can effectively suppress the growth of MCF-7 breast cancer cells.
The GLUT family consists of 14 members, including GLUT1-GLUT12, GLUT14 and the H+/myo-inositol transporter [42]. Some of these members, such as GLUT1, GLUT2 and GLUT4, are overexpressed in breast cancer cells. Therefore, GLUTs are promising target for breast cancer treatment. By developing GLUT inhibitors to block the GLUT-mediated transportation of glucose into cells, the growth of cancer cells is inhibited or apoptosis is activated, so as to achieve therapeutic effect.
It is important to note that GLUT inhibitors function in diverse ways. For example, 2-deoxy-D-glucose (2-DG) is a glucose analog and indirect inhibitor that interferes with GLUTs by competing with glucose. 2-DG serves as a glycolysis inhibitor in glucose metabolism [43]. It inhibits glucose uptake by cells through competitive binding with GLUTs, thereby reducing glucose transport efficiency [44]. Although 2-DG exerts anti-proliferative activity in breast cancer cells (MCF-7, MDA-MB-231), the inhibitory effect of 2-DG alone was not significant. Studies have shown that the use of 2-DG in combination therapy can enhance the chemical sensitivity of breast cancer cells [45]. Andrad-vieira et al. inhibited the mechanistic target of rapamycin (mTOR) pathway and glycolytic metabolism after combined administration of AZD8055 and 2-DG, thereby reducing the growth of breast cancer cells [46]. The addition of 2-DG makes tumor cells more sensitive to the inhibition of mTOR signaling pathway. In addition, although 2-DG is overall safe, previous study has shown that high doses of 2-DG can reduce blood pressure and respiratory rate in animals [47].
There are inhibitors and drugs that inhibit GLUTs expression. Silibinin is a GLUT inhibitor and a natural flavonoid with diverse biological activities [48]. Studies have shown that Silibinin inhibits GLUT1 expression through up-regulation of Bak, P53 and p21 and down-regulation of Bcl-xl, affects the proliferation of Luminal A (MCF7) breast cancer cells, induces apoptosis, and halts the cell cycle in the G0/ G1 phase [49]. However, due to the poor bioavailability of Silibinin, high doses are required to exert significant inhibitory effects, halting it in the phase I clinical trial stage [50]. Quercetin is another natural polyphenolic compound with GLUT inhibiting effect. Studies have found that Quercetin can reduce the level of GLUT in MCF-7 and MDA-MB-231 cells and reduce glucose uptake [51]. Compared with other glucose transport proteins, BAY-876, an inhibitor with high selectivity for GLUT1, is currently in the preclinical research stage [53]. Wu et al. found that there was a RB1 protein in TNBC cell line, and the expression level of the protein was related to GLUT1. The experiment showed that BAY-876 could block the expression level of RB1 protein [54]. In addition, Phloretin [55], Kaempferol [56], Indinavir [57], Sorafenib [58], and STF-31 [59] have also been found to inhibit GLUTs.
In addition to the inhibitors described above, there are some drugs that bind directly to GLUTs, thereby blocking GLUTs from functioning. WZB117 is a specific GLUT1 inhibitor, which can effectively block the transportation of glucose and reduce the level of intracellular ATP, thus achieving anti-proliferation effect [60]. However, WZB117 alone was not effective in inducing apoptosis. De et al. adopted the combined treatment method, and the prepared polymer (WZB117-OCMC-MET) could double target GLUT1 and mammalian target of rapamycin pathway to change the metabolism of breast cancer cells and improve the therapeutic effect [61].
Drugs targeting the glycolysis process
When glucose enters cancer cells, glycolysis and other pathways occur. The initial enzyme involved in the glycolysis process is HK, which catalyzes the rate-limiting phosphorylation of glucose to produce glucose 6-phosphate, which activates and carries out subsequent glycolysis or PPP. 2-DG is not only a GLUT inhibitor, but also a representative HK inhibitor. It interferes with glucose phosphorylation by competing with glucose-6-phosphate after entering the cell via GLUTs. In addition, 2-DG phosphorylated by HK, the product 2-deoxyglucose-6-phosphate, cannot be further metabolized by cells and gradually accumulates in cells, reducing the production of ATP [63]. Lonidamine (LN), an indole derivative and HK inhibitor, shows promise in breast cancer treatment. Studies have shown that LN can inhibit HK, induce endoplasmic reticulum stress response, down-regulate apoptosis inhibitory proteins, and reduce ATP levels [64]. LN was found in early clinical trials to have significant toxicities on the hepatic and pancreatic [65]. This prevents it from being formally used in the clinic. Therefore, in order to reduce the damage of LN to the liver, the research focus has been on the modification and local targeted delivery of LN. 3-Bromopyruvate (3-BrPA) can selectively inhibit HK and DAPDH, thereby inhibiting the glycolysis process, leading to ATP consumption and ultimately apoptosis [66]. Genistein-27 (GEN-27), an HK inhibitor, has been found to down-regulate HK2 expression in MDA-MB-231 breast cancer cells, inhibit glycolysis and promote apoptosis [67].
LDH includes two subtypes, LDH-A and LDH-B. LDH-A catalyzes the conversion of pyruvate to lactate during glycolysis. Cancer cells undergo glycolysis to produce excess lactate, which is associated with the upregulation of hypoxia-inducible factor-1α and c-Myc. This up-regulation subsequently increases LDH-A and MCT expression [68, 69]. Gossypol, an inhibitor of LDH-A, is a complex polyphenolic compound that occurs naturally in various parts of cotton plants, particularly in the seeds [70, 71]. There is evidence that gossypol can induce autophagy of MCF-7 cells and promote apoptosis [72].
The task of transporting lactate molecules outside the cell during glycolysis is performed by MCTs. Studies have found that human breast cancer generally has high MCT1 expression [74, 75]. AZD3965 is a promising selective inhibitor of MCT1 and is currently in clinical trials. Previously, it has been shown that AZD3965 has a strong anti-cancer effect in breast cancer models [74]. Moreover, Benyahia et al. used representative Luminal A breast cancer cells (T-47D and MCF-7) and non-malignant cells (MCF-10 A and human BJ fibroblasts) and several behavioral tests in mice to determine the anti-cancer effects and safety of AZD3965 [76]. Phenotypic characterization of mice focusing on metabolism, muscle and brain physiology found partial and transient memory retention defect as the only side effect of AZD3965. At present, AZD3965 is still in the clinical trial stage and certain side effects have been reported. However, it is believed that the clinical safety of AZD3965 in inhibiting MCT1 can be established through continuous trials.
Although some of these drugs are still in the clinical trial stage, and some have been terminated due to adverse reactions. Nonetheless, these drugs serve as valuable references for the future discovery and development of effective and selective anti-breast cancer agents.
Abnormal glycosylation in breast cancer
Glycans and glycosylation
Glycans are important constituent of cells, mainly composed of monosaccharides, oligosaccharides or polysaccharides, and are key components of glycoproteins, glycosaminoglycans, proteoglycans and glycolipids on cell surfaces [77]. The synthesis of glycans in cells is promoted by hundreds of enzymes, including glycosyltransferase, glycosidase, and enzymes involved in sugar modification, metabolism, and transportation [78]. These enzymes attach specific sugar residues to new glycans, so the synthesis of glycans is not templated, which gives them a specific structure [79]. During protein synthesis, the presence of glycans ensures the stability of endoplasmic reticulum folding and participates in the distribution of newly synthesized proteins [80]. Characteristics of different cancers can be associated with aberrant glycan structure, including tumor cell growth, survival, migration, immune evasion, and drug resistance [81,82,83]. In addition, as tumors develop and progress, certain glycans covering the surface of cells will also change. So, we can monitor tumor progression based on changes in glycans.
Alterations in glycosylation represent a well-established hallmark of cancer [84]. Glycosylation not only influences cell migration, proliferation, adhesion and apoptosis but also plays an important role in cell matrix interactions, protease activities, and immune recognition [85]. Glycosylation refers to the enzymatic process of attaching glycans to proteins, lipids or other sugars to produce glycoproteins, glycolipids, and proteoglycans. Glycosylation occurs in the endoplasmic reticulum and Golgi apparatus. When abnormal glycosylation occurs in the body, it induces a variety of pathological changes. In particular, aberrant glycosylation occurs in association with pathological changes occurring in epithelial-mesenchymal transition in tumor diseases [86]. Glycosylation is among the most prevalent forms of post-translational modifications of proteins [84]. According to the nature of glycosidic bond formation, protein glycosylation can be classified into O-glycosylation, N-glycosylation and GPI [87]. Among them, O-glycosylation and N-glycosylation are the two main types. O-glycosylation is also called mucin glycosylation [88]. O-glycosylation in which the attachment of glycans to the side chains of serine and threonine, and N-glycosylation refers to the attachment of polysaccharides to the side chain of asparagine [89]. The presence of glycosyltransferases and glycosidases plays a regulatory role in the glycosylation process.
Proteins undergo glycosylation to form glycoproteins. Therefore, glycosylation plays an important role in modifying protein and regulating protein function. On one hand, glycosylation does not affect the spatial structure of the protein. Studies have shown that protein glycosylation has little effect on the secondary structure of proteins [90, 91]. For example, Kajihara et al. used a semi-synthetic approach to obtain erythropoietin (EPO). The CD spectral analysis showed that glycoylation has no significant impact on the secondary structure of EPO [92]. In addition, experimental results showed that glycosylation does not cause major conformation changes of Fractalkine at room temperature [93]. On the other hand, glycosylation promotes the correct folding of protein, makes it more stable, and also impact its thermal stability and aggregation. Raines et al. recently evaluated the effect of N-glycosylation on ribonuclease 1 (RNase 1) and found that N-glycosylation of RNase 1 at any location can enhance the thermal stability of the protein and inhibit its hydrolysis [94].
Role of glycosylation in breast cancer
Glycosylation affects not only the proliferation but also the metastasis and survival of cancer cells. Firstly, abnormal glycosylation regulates glucose metabolism in breast cancer cells and accelerates the growth of tumor cells. Unlike normal cells, whose proliferation is regulated by precise signals, cancer cells can multiply indefinitely. The reason is that signal regulation in cancer cells is disrupted, which allows cancer cells to autonomously change metabolic pathways to obtain the energy needed for their own proliferation. For example, The final product of HBP pathway UDP-GlcNAc can participate in O-linked/N-linked glycosylation and can also participate in O-GlcNAcylation as a substrate of O-GlcNAc transferase (OGT) [95]. A study has shown that the enhanced binding of O-GlcNAc acylated DNA topoisomerase IIα (TOP2A) to DNA enhances the interaction between TOP2A and cycle regulators such as CDK1 and UPF1, affecting the cell cycle process and ultimately promoting the proliferation of breast cancer cells [96]. In addition, OGT expression levels catalyzing O-GlcNAc acylation were elevated in breast cancer cells [97]. There is an ER-independent link between OGT and the transcription factor FOXA1, which promotes oglcn acylation at three FOXA1 sites (Thr432, Ser441, and Ser443), thereby promoting breast cancer metastasis [98]. Therefore, the growth of tumor cells can be inhibited by reducing the level of OGT. In addition, by comparing the effects of OGT inhibition on the proliferation and survival of different subtypes of breast cancer cells, it was found that TNBC cell lines were more dependent on OGT [99].
Secondly, the glycosylation modifications affect cell-cell adhesion and mediates cell-matrix interactions. Tumor cells disrupt intercellular adhesion and invade surrounding tissues to promote breast cancer progression [100]. The transmembrane glycoprotein mucin 1 (MUC1) is the most easily identifiable transmembrane protein in the mucin family with a highly glycosylated extracellular domain. Normally, MUC1 forms a tight mesh structure and covers the surface of epithelial cells to protect the cells from extreme conditions [101,102,103]. However, in cancer cells, MUC1 expression is upregulated, leading to altered protein structure, glycosylation patterns, and spatial distribution. These changes in MUC1 are pivotal in the onset and progression of breast cancer and other malignancies. Abnormal glycosylation in the MUC1 makes the O-chain truncation, increased expression of Tn and STn, formation of new side chains, induction of carcinogenic characteristics of cells, and promotion of cell growth and invasion by disrupting intercellular adhesion contact [104]. Extracellular matrix (ECM) is a complex network composed of glycoproteins, glycosaminoglycans, collagen and proteoglycans, which influence tumor development and cancer progression [105]. Notably, integrins, pivotal cell surface adhesion glycoproteins, play a crucial role in ECM interactions and metastasis [106]. Evidence suggests that alterations in integrin glycosylation can modulate the adhesive properties of cancer cells [107]. It can be concluded that the alteration of integrin glycosylation may affect cell adhesion to ECM, and thus play a role in cell migration. Singh et al. examined the differential expression of integrins avb3 and avb6 in breast cancer cells and untransformed breast cells at two different disease stages. The results suggest the necessity of N-glycosylation on integrins for cell-ECM interactions [108].
Thirdly, glycosylation plays a role in the metastasis of breast cancer cells. More and more evidence has revealed the important role of abnormal glycosylation in malignant transformation and metastasis of tumors. Abnormal N-glycosylation can alter the three-dimensional structure of MUC1 and promote cancer cell migration [109]. However, there are also some glycoproteins that possess inhibitory effects on breast cancer metastasis. SGK196 is a transmembrane protein that is commonly expressed in skeletal muscle, brain, heart and kidney tissues. Studies have shown that SGK196 is mainly modified by N-glycosylation in breast cancer cells, and is a glycosylated specific kinase, which plays a role in inhibiting cell metastasis and invasion in breast cancer. Li et al. found that N-glycosylated SGK196 can inhibit the metastasis of BLBC cells. Through mechanism studies, it was found that N-glycosylated SGK196 plays a regulatory role through the PI3K/AKT/GSK3β signaling pathway [110]. This finding provides new insights into the function of SGK196 in breast cancer.
Drugs for treating breast cancer by inhibiting abnormal glycosylation
At present, breast cancer treatment mainly includes conventional therapy, immunotherapy and small-molecule inhibitors [111]. Conventional therapies refer to radiation therapy, chemotherapy and surgery, which have certain side effects and lack of specificity. Immunotherapy has clinical significance for the treatment of triple-negative breast cancer and HER2 positive breast cancer. In immunotherapy, the clinical application of therapeutic antibodies, including PD-L1 [112], CAR-T cell therapy [113], trastuzumab [114], and other clinical applications, has improved the survival rate of breast cancer patients. However, immunotherapy also has many disadvantages, such as high heterogeneity, laborious production and treatment resistance. Meanwhile, the use of small-molecule inhibitors also has the above disadvantages. It is considered that altered glycans play a role in the occurrence, progression, and metastasis of breast cancer. Therefore, targeted glycosylation has become one of the options to develop therapeutic agents for breast cancer.
Targeted glycosylation therapy for breast cancer can be divided into the following three categories, as shown in Table 3. The first category is synthetic monosaccharide analogs. Because cell membranes and secretory proteins are glycoproteins, small changes in the glycan chains may affect the function of the glycoconjugate, affecting cell adhesion, immune recognition, and the growth and metastasis of tumor cells [115]. Monosaccharide analogs with active groups are glycosylated after uptake by cells, and can then act as inhibitors to interfere with the formation of oligosaccharide chains [116, 117]. Currently, synthesized monosaccharide analogs include glucose analog 2-DG [118], GalNAc analog 2-KetoGal [119], and focusing analog 6-Azidofucose [120]. Among them, 2-DG is a monosaccharide analog that not only inhibits glycolysis, but also interferes with glycosylation [121]. In terms of glycosylation, 2-DG exerts anticancer effects in Luminal A (MCF7) and TNBC (MDA-MB-231) breast cancer cell lines by blocking glucose-6-phosphate isomerase. The second category involves glycosyltransferase/glycoprotein inhibitors. Because glycosylation refers to the process of attaching glycans to protein under the catalysis of enzymes. Therefore, enzymes or protein-related inhibitors involved in the glycosylation can compete with glycosyltransferase to participate in protein glycosylation, block the abnormal glycosylation process, and thus inhibit tumor cell metastasis. PGM3 is an enzyme involved in the HBP pathway which can be targeted by inhibitors. As a competitive inhibitor of PGM3, FR054 induces a decrease in intracellular UDP-GlcNAc, resulting in a decrease in N-glycans. Treatment of breast cancer cells with FR054 can induce apoptosis and reduce TNBC (MDA-MB-231 cells) adhesion and migration [122]. The third category is antibodies against glycosylation. Tunicamycin is an antibiotic. HER2 positive (SKBR-3 cells) can prevent the transfer of GLNAC-1-p to dolichol-P under tunicamycin. This is an important step in the production of N-glycans. Therefore, tunicamycin reduces cell responsiveness to growth factors by inhibiting N-glycosylation, thereby inhibiting cancer cell growth [124]. Erbitux is a lgG1 monoclonal antibody targeting EGFR. In Luminal breast cancer cell lines, EGFR was competitively blocked from binding to other ligands, EGFR glycosylation was inhibited, ERK1/2 signaling pathway was inhibited, and FUT4 expression was significantly down-regulated [125]. In summary, Erbitux plays an anticancer role by inhibiting fucosylation. Epigallocatechin gallate (EGCG) is the monomer with the highest content and biological activity in green tea [126]. In breast cancer cells, MUC1 expression is upregulated and protein glycosylation is altered. EGCG can interfere with the production of MUC1 and inhibit O-glycosylation. Studies have found that EGCG inhibits the expression of miR-25 in Luminal A (MCF-7 cells), induces cell cycle arrest in G2/ M phase, and thus plays a role in apoptosis and inhibition of cell proliferation [127]. In addition, EGCG can inhibit the growth of TNBC (MDA-MB-231) cells and can be used as a potential therapeutic agent for TNBC patients [128]. Due to its low bioavailability and lack of targeting, the use of EGCG in clinical practice has been limited. However, EGCG can be used as a chemotherapy adjuvant to enhance the sensitivity of tumors to drugs [129]. Ginsenoside Rg3 [130] and N-acetylcysteine (NAC) [131] inhibited fucosylation in Luminal A (MCF7) and TNBC (MDA-MB-231) cell lines.
Links between glucose metabolism and glycosylation in breast cancer
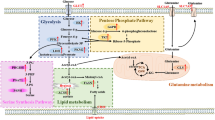
The specific changes and effects of glucose metabolism and glycosylation in breast cancer have been described in the previous chapters. This also brings to light a question worth pondering: Is there a link between altered glucose metabolism, or aerobic glycolysis, and abnormal glycosylation in cancer cells? There is evidence that these two processes can influence each other (Fig. 2). The HBP links glucose metabolism with abnormal glycosylation. An intermediate product of the glycolysis pathway (fructose-6-phosphate) is a critical reactant of HBP and a substrate for UDP-GlcNAc. The activation of GlcNAc forms UDP-GlcNAc, and UDP-GlcNAc is a metabolic precursor of UDP-GalNAc and CMP-sialic acid [132], which is used for the synthesis of N- and O-linked protein glycosylation and modification of O-GlcNAcylation [84, 133, 134]. Therefore, the UDP-GlcNAc concentration is critical for the glycosylation process. In addition, O-GlcNAC modifies PFK activity, thereby blocking glycolysis in cancer cells. This suggests that important metabolic reprogramming in cancer cells is carried out via O-GlcNAC [135]. It can be concluded that metabolism and glycosylation are interrelated and abnormal changes in cancer cells, providing the characteristic microenvironment of proliferation and metastasis for cancer cells.
In normal cells, HBP can strictly maintain UDP-GlcNAc levels in response to material fluctuations in cells, which establishing cell homeostasis [136]. In cancer cells, altered HBP fluxes lead to changes in glucose metabolism and abnormal glycosylation [137]. The level of enzymes involved in the HBP are often elevated in cancer cells [138]. GFAT is the main rate-limiting enzyme involved in HBP. When GFAT expression is up-regulated, HBP increases. Therefore, GFAT activity can regulate HBP flow [139]. Chanmee et al. found that GFAT expression is upregulated in breast cancer patients and is associated with overexpression of a cancer-related hyaluronan (HA) [140]. Chokchaitaweesuk et al. further demonstrated through animal experiments that HA overexpressing breast cancer cells rapidly progress to invasive breast cancer [139]. However, it is worth noting that GFAT inhibitors can also inhibit the synthesis of amino acids and so are not specific. In addition, some substances that affect metabolism can also have an impact on the HBP pathway. For example, the glycolysis inhibitor 2-DG can also inhibit the phosphorylation of GFAT and reduce the generation of UDP-GlcNAc, thus affecting the glycosylation pathway [141]. How to design and use metabolic inhibitors to regulate HBP and play a therapeutic role needs to be further studied.
The HBP pathway links glycolysis and glycosylation. Created with PowerPoint. GLUT: Glucose transporter; HK: hexokinase; GPI: glucose-6-phosphate isomerase; GFAT: ructose-6-phosphate amidotransferase; GNA1: glucosamine-6-phosphate N-acetyltransferase 1; PGM3: phosphoglucomutase 3; UAP: UDP-N-acetylhexosamine pyrophosphorylase
Conclusion and future perspectives
At present, breast cancer is still the main disease threatening women’s life and health, affecting their quality of life. Glucose serves as the primary energy substrate for cancer cells, and a series of changes in the cell are worth studying. Existing studies have shown that alterations in glucose metabolic pathways (especially increased glycolysis) and abnormal glycosylation are associated with breast cancer progression. In addition, the HBP pathway links glucose metabolism to the process of glycosylation. Therefore, the inhibition of HBP pathway can also be considered in the future discovery and development of breast cancer drugs from the perspective of glucose changes. Moreover, although there are limited studies on the development of therapeutic agents for breast cancer by targeting glycolysis and inhibiting glycosylation, the existing studies still provide implications for the future development of novel agents against breast cancer. Much more importantly, considering the genetically heterogeneous nature of breast cancer involving complex heritable and somatic factors, future studies should focus on exploring different signaling networks associated with various breast cancer subtypes. This effort aims to clarify sensitivity to therapeutic agents and provide personalized benefits against breast cancer.
Data availability
No datasets were generated or analysed during the current study.
References
Sung H, Ferlay J, Siegel RL et al (2020) Global Cancer statistics 2020: GLOBOCAN estimates of incidence and Mortality Worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71:209–249
Chhikara BS, Parang K (2023) Global Cancer statistics 2022: the trends Projection Analysis. Chem Biol Lett 10:451
Chen W, Zheng R, Baade PD et al (2016) Cancer statistics in China, 2015. CA Cancer J Clin 66:115–132
Polyak K (2007) Breast cancer: origins and evolution. J Clin Invest 117:3155–3163
Kleibl Z, Kristensen VN (2016) Women at high risk of breast cancer: molecular characteristics, clinical presentation and management. Breast 20:136–144
Scott DA, Drake RR (2019) Glycosylation and its implications in breast cancer. Expert Rev 16:665–680
Ibrahim E, Al-Gahmi AM, Zeenelin AA et al (2009) Luminal A breast cancer subtypes: a matched case-control study using estrogen receptor, progesterone receptor, and HER-2 as surrogate markers. Med Oncol 26:372–378
Apostolou P, Fostira F (2013) Hereditary breast cancer: the era of new susceptibility genes. Biomed Res Int 2013: 747318
Sotiriou C, Pusztai L (2009) Gene-expression signatures in breast cancer. N Engl J Med 360:790
Fragomeni SM, Sciallis A, Jeruss JS (2018) Molecular subtypes and local-regional control of breast cancer. Surg Oncol Clin N Am 27:95–120
Szymiczek A, Lone A, Akbari MR (2021) Molecular intrinsic versus clinical subtyping in breast cancer: a comprehensive review. Clin Genet 99:613–637
Seyfried TN, Arismendi-Morillo G, Mukherjee P, Chinopoulos C (2020) On the origin of ATP synthesis in cancer. iScience 23:101761
Reily C, Stewart TJ, Renfrow MB, Novak J (2019) Glycosylation in health and disease. Nat Rev Nephrol 15:346–366
Wells L, Vosseller K, Hart GW (2001) Glycosylation of nucleocytoplasmic proteins: signal transduction and O-GlcNAc. Science 291:2376–2378
Slawson C, Copeland RJ, Hart GW (2010) O-GlcNAc signaling: a metabolic link between diabetes and cancer? Trends Biochem Sci 35:547–555
Hay N (2016) Reprogramming glucose metabolism in cancer: can it be exploited for cancer therapy? Nat Rev Cancer 16:635–649
Li Z, Zhang H (2016) Reprogramming of glucose, fatty acid and amino acid metabolism for cancer progression. Cell Mol Life Sci 73:377–392
Zhang X, Xiang J (2019) Remodeling the Microenvironment before occurrence and metastasis of Cancer. Int J Biol Sci 15:105–113
Martínez-Reyes I, Chanel NS (2021) Cancer metabolism: looking forward. Nat Rev Cancer 21:669–680
Cairns RA, HARRIS IS, Mak TW (2011) Regulation of cancer cell metabolism. Nat Rev Cancer 11:85–95
Dias AS, Almeida CR, Helguero LA, Duarte IF (2019) Metabolic crosstalk in the breast cancer microenvironment. Eur J Cancer 121:154–171
Gill KS, Fernandes P, O’Donovan TR et al (2016) Glycolysis inhibition as a cancer treatment and its role in an anti-tumour immune response. Biochim Biophys Acta 1866:87–105
Warburg O (1956) On the origin of cancer cells. Science 3191:309–314
Wei J, Huang K, Chen Z et al (2020) Characterization of Glycolysis-Associated molecules in the Tumor Microenvironment revealed by Pan-cancer tissues and Lung Cancer single cell data. Cancers (Basel) 12:1788
Warburg O (1928) The chemical constitution of respiratory ferment. Science 68:437–443
Warburg O (1956) On respiratory impairment in cancer cells. Science 124:269–270
Ganapathy-Kanniappan S, Geschwind JF (2013) Tumor glycolysis as a target for cancer therapy: progress and prospects. Mol Cancer 12:152
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144:646–674
Bononi G, Masoni S, Di Bussolo V et al (2022) Historical perspective of tumor glycolysis: a century with Otto Warburg. Semin Cancer Biol 86:325–333
Ramos-Martinez JI (2017) The regulation of the pentose phosphate pathway:remember Krebs. Arch Biochem Biophys 614:50–52
Patra KC, Hay N (2014) The pentose phosphate pathway and cancer. Trends Biochem Sci 39:347–354
Shin E, Koo JS (2021) Glucose metabolism and glucose transporters in breast Cancer. Front Cell Dev Biol 9:728759
Wellen KE, Lu C, Mancuso A et al (2010) The hexosamine biosynthetic pathway couples growth factor-induced glutamine uptake to glucose metabolism. Genes Dev 24:2784–2799
Wright EM, Loo DD, Hirayama BA (2011) Biology of human sodium glucose transporters. Physiol Rev 91(2):733–794
Tsunokake S, Iwabuchi E, Miki Y et al (2023) SGLT1 as an adverse prognostic factor in invasive ductal carcinoma of the breast. Breast Cancer Res Treat 201:499–513
Liu H, Ertay A, Peng P et al (2019) SGLT1 is required for the survival of triple-negative breast cancer cells via potentiation of EGFR activity. Mol Oncol 13:1874–1886
Wang J, Ji H, Niu X et al (2020) Sodium-Dependent Glucose Transporter 1 (SGLT1) Stabled by HER2 Promotes Breast Cancer Cell Proliferation by Activation of the PI3K/Akt/mTOR Signaling Pathway in HER2 + Breast Cancer. Dis Markers 2020: 6103542
Shibazaki T, Tomae M, Ishikawa-Takemura Y et al (2012) KGA-2727, a novel selective inhibitor of a high-affinity sodium glucose cotransporter (SGLT1), exhibits antidiabetic efficacy in rodent models. J Pharmacol Exp Ther 342:288–296
Fukudo S, Endo Y, Hongo M et al (2018) Safety and efficacy of the sodium-glucose cotransporter 1 inhibitor mizagliflozin for functional constipation: a randomised, placebo-controlled, double-blind phase 2 trial. Lancet Gastroenterol Hepatol 3:603–613
Zhou J, Zhu J, Yu SJ et al (2020) Sodium-glucose co-transporter-2 (SGLT-2) inhibition reduces glucose uptake to induce breast cancer cell growth arrest through AMPK/mTOR pathway. Biomed Pharmacother 132:110821
Komatsu S, Nomiyama T, Numata T et al (2020) SGLT2 inhibitor ipragliflozin attenuates breast cancer cell proliferation. Endocr J 67:99–106
Mueckler M, Thorens B (2013) The SLC2 (GLUT) family of membrane transporters. Mol Aspects Med 34:121–138
Wang Z, Zhang L, Zhang D et al (2015) Glycolysis inhibitor 2-deoxy-dglucose suppresses carcinogen-induced rat hepatocarcinogenesis by restricting cancer cell metabolism. Mol Med Rep 11:1917–1924
Kuntz S, Mazerbourg S, Boisbrun M et al (2014) Energy restriction mimetic agents to target cancer cells: comparison between 2-deoxyglucose and thiazolidinediones. Biochem Pharmacol 92:102–111
Tagg SL, Foster PA, Leese MP et al (2008) 2-Methoxyoestradiol-3,17-O,O-bis-sulphamate and 2-deoxy-D-glucose in combination: a potential treatment for breast and prostate cancer. Br J Cancer 99:1842–1848
Andrade-Vieira R, Goguen D, Bentley HA et al (2014) Pre-clinical study of drug combinations that reduce breast cancer burden due to aberrant mTOR and metabolism promoted by LKB1 loss. Oncotarget 5:12738–12752
Vijayaraghavan R, Kumar D, Dube SN et al (2006) Acute toxicity and cardio-respiratory effects of 2-deoxy-D-glucose: a promising radio sensitiser. Biomed Environ Sci 19:96–103
Zhu XX, Ding YH, Wu Y et al (2016) Silibinin: a potential old drug for cancer therapy. Expert Rev Clin Pharmacol 9(10):1323–1330
Pirouzpanah MB, Sabzichi M, Pirouzpanah S et al (2015) Silibilin-induces apoptosis in breast cancer cells by modulating p53, p21, Bak and Bcl-XL pathways. Asian Pac J Cancer Prev 16:2087–2092
Flaig TW, Gustafson DL, Su LJ et al (2007) A phase I and pharmacokinetic study of silybin-phytosome in prostate cancer patients. Invest New Drugs 25:139–146
Jia L, Huang S, Yin X et al (2018) Quercetin suppresses the mobility of breast cancer by suppressing glycolysis through Akt-mTOR pathway mediated autophagy induction. Life Sci 208:123–130
Kasiri N, Rahmati M, Ahmadi L et al (2020) Therapeutic potential of quercetin on human breast cancer in different dimensions. Inflammopharmacology 28:39–62
Siebeneicher H, Cleve A, Rehwinkel H et al (2016) Identification and optimization of the First highly selective GLUT1 inhibitor BAY-876. ChemMedChem 11:2261–2271
Wu Q, Ba-Alawi W, Deblois G et al (2020) GLUT1 inhibition blocks growth of RB1-positive triple negative breast cancer. Nat Commun 11:4205
Chen M, Gowd V, Wang M et al (2021) The apple dihydrochalcone phloretin suppresses growth and improves chemosensitivity of breast cancer cells via inhibition of cytoprotective autophagy. Food Funct 12:177–190
Azevedo C, Correia-Branco A, Araújo JR, Guimarães JT, Keating E, Martel F et al (2015) The chemopreventive effect of the dietary compound kaempferol on the MCF-7 human breast cancer cell line is dependent on inhibition of glucose cellular uptake. Nutr Cancer 67: 504–513
Toschi E, Sgadari C, Malavasi L et al (2011) Human immunodeficiency virus protease inhibitors reduce the growth of human tumors via a proteasome-independent block of angiogenesis and matrix metalloproteinases. Int J Cancer 128:82–93
Fumarola C, Caffarra C, la Monica S et al (2013) Effects of Sorafenib on energy metabolism in breast cancer cells: role of AMPK-mTORC1 signaling. Breast Cancer Res Treat 141:67–78
Xintaropoulou C, Ward C, Wise A et al (2015) A comparative analysis of inhibitors of the glycolysis pathway in breast and ovarian cancer cell line models. Oncotarget 6:25677–25695
Qian Y, Wang X, Liu Y et al (2014) Extracellular ATP is internalized by macropinocytosis and induces intracellular ATP increase and drug resistance in cancer cells. Cancer Lett 351:242–251
De A, Wadhwani A, Sauraj et al (2023) WZB117 decorated metformin-carboxymethyl Chitosan nanoparticles for targeting breast Cancer metabolism. Polym (Basel) 15:976
Chen Q, Meng YQ, Xu XF, Gu J (2017) Blockade of GLUT1 by WZB117 resensitizes breast cancer cells to adriamycin. Anticancer Drugs 28:880–887
Dwarakanath B, Jain V (2009) Targeting glucose metabolism with 2-deoxy-D-glucose for improving cancer therapy. Future Oncol 5:581–585
Di Cosimo S, Ferretti G, Papaldo P et al (2003) Lonidamine: efficacy and safety in clinical trials for the treatment of solid tumors. Drugs Today 39:157
Price GS, Page RL, Riviere JE et al (1996) Pharmacokinetics and toxicity of oral and intravenous lonidamine in dogs. Cancer Chemother Pharmacol 38:129–135
Liu Z, Zhang YY, Zhang QW et al (2014) 3-Bromopyruvate induces apoptosis in breast cancer cells by downregulating Mcl-1 through the PI3K/Akt signaling pathway. Anticancer Drugs 25:447–455
Tao L, Wei L, Liu Y et al (2017) Gen-27, a newly synthesized flavonoid, inhibits glycolysis and induces cell apoptosis via suppression of hexokinase II in human breast cancer cells. Biochem Pharmacol 125:12–25
Masoud GN, Li W (2015) HIF-1α pathway: role, regulation and intervention for cancer therapy. Acta Pharm Sin B 5:378–389
Wang ZH, Peng WB, Zhang P et al (2021) Lactate in the tumour microenvironment: from immune modulation to therapy. EBioMedicine 73:103627
Gadelha ICN, Fonseca NBS, Oloris SCS et al (2014) Gossypol toxicity from cottonseed products. Sci World J 2014: 231635
Kenar JA (2006) Reaction chemistry of gossypol and its derivatives. J Am Oil Chem Soc 83:269–302
Gao P, Bauvy C, Souquère S et al (2010) The Bcl-2 homology domain 3 mimetic gossypol induces both Beclin 1-dependent and Beclin 1-independent cytoprotective autophagy in cancer cells. The Journal of biological chemistry. 2010, 285, 25570–25581
Zhou M, Zhao Y, Ding Y et al (2010) Warburg effect in chemosensitivity: targeting lactate dehydrogenase-A re-sensitizes taxol-resistant cancer cells to taxol. Mol Cancer 9:33
Hong CS, Graham NA, Gu W et al (2016) MCT1 modulates cancer cell pyruvate export and growth of tumors that co-express MCT1 and MCT4. Cell Rep 14:1590–1601
Pinheiro C, Albergaria A, Paredes J et al (2010) Monocarboxylate transporter 1 is up-regulated in basal-like breast carcinoma. Histopathology 56:860–867
Benyahia Z, Blackman MCNM, Hamelin L et al (2021) In Vitro and in vivo characterization of MCT1 inhibitor AZD3965 confirms Preclinical Safety compatible with breast Cancer Treatment. Cancers 13:569
Chang X, Obianwuna UE, Wang J et al (2023) Glycosylated proteins with abnormal glycosylation changes are potential biomarkers for early diagnosis of breast cancer. Int J Biol Macromol 236:123855
Neelamegham S, Mahal LK (2016) Multi-level regulation of cellular glycosylation: from genes to transcript to enzyme to structure. Curr Opin Struct Biol 40:145–152
Moremen KW, Tiemeyer M, Nairn AV (2012) Vertebrate protein glycosylation: diversity, synthesis and function. Nat Rev Mol Cell Biol 13:448–462
Helenius A, Markus A (2001) Intracellular functions of N-linked glycans. Science 291:2364–2369
Cornelissen LA, Van Vliet SJ (2016) A bitter sweet symphony: immune responses to altered O-glycan epitopes in cancer. Biomolecules 6:26
Nardy AFFR, Freire-de-Lima L, Freire-de-Lima GG, Morrot A (2016) The sweet side of immune evasion: role of glycans in the mechanisms of cancer progression. Front Oncol 6:54
Li X, Wang X, Tan Z et al (2016) Role of glycans in cancer cells undergoing epithelial-mesenchymal transition. Front Oncol 6:33
Pinho SS, Reis CA (2015) Glycosylation in cancer: mechanisms and clinical implications. Nat Rev Cancer 15:540–555
Varki A (2017) Biological roles of glycans. Glycobiology 27:3–49
Tan FY, Tang CM, Exley RM (2015) Sugar coating: bacterial protein glycosylation and host-microbe interactions. Trends Biochem Sci 40:342–350
Corfield AP, Berry M (2015) Glycan variation and evolution in the eukaryotes. Trends Biochem Sci 40:351–359
Duarte HO, Freitas D, Gomes C et al (2016) Mucin-type O-Glycosylation in gastric carcinogenesis. Biomolecules 6:33–42
Spiro RG (2002) Protein glycosylation: nature, distribution, enzymatic formation, and disease implications of glycopeptide bonds, vol 12. Glycobiology, pp 43R–56R
Lee HS, Qi Y, Im W (2015) Effects of N-glycosylation on protein conformation and dynamics: protein data Bank analysis and molecular dynamics simulation study. Sci Rep 5:8926
Solá RJ, Griebenow K (2010) Glycosylation of therapeutic proteins. BioDrugs 24:9–21
Murakami M, Kiuchi T, Nishihara M et al (2016) Chemical synthesis of erythropoietin glycoforms for insights into the relationship between glycosylation pattern and bioactivity. Sci Adv 2:e1500678
Minh Hien N, Izumi M, Sato H et al (2017) Chemical synthesis of glycoproteins with the specific installation of gradient-enriched 15 N-labeled amino acids for getting insights into glycoprotein behavior. Chem Eur J 23:6579–6585
Ressler VT, Raines RT (2019) Consequences of the endogenous N-glycosylation of human ribonuclease 1. Biochemistry 58:987–996
Itano N, Iwamoto S (2023) Dysregulation of hexosamine biosynthetic pathway wiring metabolic signaling circuits in cancer. Biochim Biophys Acta Gen Subj 1867:130250
Liu Y, Yu K, Zhang K et al (2023) O-GlcNAcylation promotes topoisomerase IIα catalytic activity in breast cancer chemoresistance. EMBO Rep 24(7), e56458
Ma Z, Vosseller K (2014) Cancer metabolism and elevated O-GlcNAc in oncogenic signaling. J Biol Chem 289:34457–34465
Liu Y, Yu K, Kong X et al (2023) FOXA1 O-GlcNAcylation-mediated transcriptional switch governs metastasis capacity in breast cancer. Sci Adv 9(33):eadg7112
Barkovskaya A, Seip K, Hilmarsdottir B et al (2019) O-GlcNAc transferase inhibition differentially affects breast Cancer subtypes. Sci Rep 9:5670
Inman JL, Robertson C, Mott JD, Bissell MJ (2015) Mammary gland development: cell fate specification. stem Cells Microenvironment Dev 142:1028–1042
Nath S, Mukherjee P (2014) MUC1: a multifaceted oncoprotein with a key role in cancer progression. Trends Mol Med 20:332–342
Bose M, Mukherjee P (2020) Microbe-MUC1 crosstalk in Cancer-Associated infections. Trends Mol Med 26:324–336
Carson DD (2008) The cytoplasmic tail of MUC1: a very busy place. Sci Signal 1:pe35
Chen W, Zhang Z, Zhang S et al (2021) MUC1: structure, function, and clinic application in epithelial cancers. Int J Mol Sci 22:12
Kim SH, Turnbull J, Guimond S (2011) Extracellular matrix and cell signalling: the dynamic cooperation of integrin, proteoglycan and growth factor receptor. J Endocrinol 209:139–151
Schwartz MA, Ginsberg MH (2002) Networks and crosstalk: integrin signalling spreads. Nat Cell Biol 4:E65–E68
Janik ME, Przybylo M, Pochec E et al (2010) Effect of alpha3beta1 and alphavbeta3 integrin glycosylation on interaction of melanoma cells with vitronectin. Acta Biochim Pol 57:55–61
Singh C, Shyanti RK, Singh V et al (2018) Integrin expression and glycosylation patterns regulate cell-matrix adhesion and alter with breast cancer progression. Biochem Biophys Res Commun 499:374–380
Huanna T, Tao Z, Xiangfei W et al (2015) GALNT14 mediates tumor invasion and migration in breast cancer cell MCF-7. Mol Carcinog 54:1159–1171
Xu C, Zhang M, Bian L et al (2020) N-glycosylated SGK196 suppresses the metastasis of basal-like breast cancer cells. Oncogenesis 9:4
Costa AF, Campos D, Reis CA, Gomes C (2020) Targeting glycosylation: a New Road for Cancer Drug Discovery. Trends Cancer 6:757–766
Bertucci F, Gonçalves A (2017) Immunotherapy in breast Cancer: the emerging role of PD-1 and PD-L1. Curr Oncol Rep 19:64
Yang YH, Liu JW, Lu C, Wei JF (2022) CAR-T cell therapy for breast Cancer: from Basic Research to Clinical Application. Int J Biol Sci 18:2609–2626
Yang T, Kang L, Li D, Song Y (2023) Immunotherapy for HER-2 positive breast cancer. Front Oncol 13:1097983
Liu H, Ma L, Lin J et al (2020) Advances in molecular mechanisms of drugs affecting abnormal glycosylation and metastasis of breast cancer. Pharmacol Res 155:104738
Landuyt LV, Lonigro C, Meuris L, Callewaert N (2019) Customized protein glycosylation to improve biopharmaceutical function and targeting. Curr Opin Biotechnol 60:17–28
Chen F, Huang G (2019) Application of glycosylation in targeted drug delivery. Eur J Med Chem 182:111612
Ricciardiello F, Bergamaschi L, De Vitto H et al (2021) Suppression of the HBP function increases pancreatic cancer cell sensitivity to a pan-RAS inhibitor. Cells 10:431
Hang HC, Bertozzi CR (2001) Ketone isosteres of 2-N-acetamidosugars as substrates for metabolic cell surface engineering. J Am Chem Soc 123:1242–1243
Rabuka D, Hubbard SC, Laughlin ST et al (2006) A chemical reporter strategy to probe glycoprotein fucosylation. J Am Chem Soc 128:12078–12079
Berthe A, Zafno M, Muller C et al (2018) Protein N-glycosylation alteration and glycolysis inhibition both contribute to the antiproliferative action of 2-deoxyglucose in breast cancer cells. Breast Cancer Res Treat 171:581–591
Ricciardiello F, Votta G, Palorini R et al (2018) Inhibition of the hexosamine biosynthetic pathway by targeting PGM3 causes breast cancer growth arrest and apoptosis. Cell Death Dis 9:1–17
Zada S, Hwang JS, Ahmed M et al (2019) Protein kinase A activation by βLapachone is associated with apoptotic cell death in NQO1overexpressing breast cancer cells. Oncol Rep 42:1621–1630
Peiris D, Spector AF, Lomax-Browne H et al (2017) Cellular glycosylation affects herceptin binding and sensitivity of breast cancer cells to doxorubicin and growth factors. Sci Rep 7:43006
Crozier Jennifer A, LaPlant B, Timothy H et al (2016) A phase II trial of irinotecan with cetuximab in patients with metastatic breast Cancer previously exposed to anthracycline and/or taxane-containing therapy. Clin Breast Cancer 16:23–30
Zhang Y, Yang ND, Zhou F et al (2012) (-)-Epigallocatechin-3-Gallate induces non-apoptotic cell death in human cancer cells via ROS-mediated lysosomal membrane permeabilization. PLoS ONE 7:e46749
Zan L, Chen Q, Zhang L, Li X (2019) Epigallocatechin gallate (EGCG) suppresses growth and tumorigenicity in breast cancer cells by downregulation of miR-25. Bioengineered 10:374–382
Hong OY, Noh EM, Jang HY et al (2017) Epigallocatechin gallate inhibits the growth of MDA-MB-231 breast cancer cells via inactivation of the β-catenin signaling pathway. Oncol Lett 4:441–446
Wang L, Li P, Feng K (2023) EGCG adjuvant chemotherapy: current status and future perspectives. Eur J Med Chem 250:115197
Kim BM, Kim DH, Park JH (2013) Ginsenoside Rg3 induces apoptosis of human breast Cancer (MDA-MB-231) cells. J Cancer Prev 18(2):177–185
Agarwal A, Klueh U, Shih SC et al (2004) N-acetyl-cysteine promotes angiostatin production and vascular collapse in an orthotopic model of breast cancer. Am J Pathol 164:1683–1696
Almaraz RT, Tian Y, Bhattarcharya R et al (2012) Metabolic flux increases glycoprotein sialylation: implications for cell adhesion and cancer metastasis. Mol Cell Prot 11:M112
Nagel AK, Ball LE (2015) Intracellular protein O-GlcNAc modification integrates nutrient status with transcriptional and metabolic regulation. Adv Cancer Res 126:137–166
Lee JB, Pyo KH, Kim HR (2021) Role and function of O-GlcNAcylation in cancer. Cancers 13:5365
Yi W, Clark PM, Mason DE et al (2012) Phosphofructokinase 1 glycosylation regulates cell growth and metabolism. Science 337:975e980
Paneque A, Fortus H, Zheng J et al (2023) The Hexosamine Biosynthesis Pathway: regulation and function. Genes (Basel) 14:933
de Queiroz RM, Oliveira IA, Piva B et al (2019) Hexosamine Biosynthetic Pathway and Glycosylation Regulate Cell Migration in Melanoma cells. Front Oncol 9:116
Jia C, Li H, Fu D, Lan Y (2020) GFAT1/HBP/O-GlcNAcylation Axis Regulates β-Catenin Activity to Promote Pancreatic Cancer Aggressiveness. BioMed Res Int 2020: 1921609
Chokchaitaweesuk C, Kobayashi T, Izumikawa T, Itano N (2019) Enhanced hexosamine metabolism drives metabolic and signaling networks involving hyaluronan production and O-GlcNAcylation to exacerbate breast cancer. Cell Death Dis 10:803
Chanmee T, Ontong P, Izumikawa T et al (2016) Hyaluronan production regulates metabolic and cancer stem-like properties of breast cancer cells via hexosamine biosynthetic pathway-coupled HIF-1 signaling. J Biol Chem 291:24105–24120
Moloughney JG, Vega-Cotto NM, Liu S et al (2018) mTORC2 modulates the amplitude and duration of GFAT1 Ser-243 phosphorylation to maintain flux through the hexosamine pathway during starvation. J Biol Chem 293:16464–16478
Acknowledgements
We would like to thank the discussion and language editing by Prof. Song Li from Dalian Medical University.
Funding
This work was supported by the National Natural Science Foundation of China (NSFC 32170499 and 32070440).
Author information
Authors and Affiliations
Contributions
ZJ and SH drafted the main manuscript text; ZJ prepared figures; ZJ and WC edited and finalized the manuscript; WC and SD designed and supervised the whole project. All authors reviewed the manuscript and approved the submission.
Corresponding authors
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhao, J., Sun, H., Wang, C. et al. Breast cancer therapy: from the perspective of glucose metabolism and glycosylation. Mol Biol Rep 51, 546 (2024). https://doi.org/10.1007/s11033-024-09466-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11033-024-09466-w