Abstract
2-Naphthol or β-naphthol is an important starting material that has drawn great attention in various organic transformations because of its attributes, such as low cost, easy to handle and eco-friendliness. The electron-rich aromatic framework of 2-naphthol with multiple reactive sites allows it to be utilized in several kinds of organic reactions eventuated to several organic molecules with potent biological properties. Multicomponent reaction approach has been tremendously utilized to explore the synthetic utility of 2-naphthol for the construction of diverse N/O-containing heterocyclic framework. In this review, we summarize recent data pertaining to multicomponent reactions, wherein heterocyclic compounds are synthesized utilizing 2-naphthol as one of the starting materials. It is anticipated that this review will stimulate the researchers to design new multicomponent strategies complying with the Green Chemistry principles for the further exploitation of 2-naphthol for the rapid synthesis of versatile biologically relevant heterocycles.
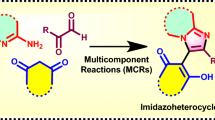
Graphic abstract
This review provides a concise overview of the different 2-naphthol based multicomponent reactions utilized for the construction of diverse bioactive heterocyclic scaffold.

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
2-Naphthol also known as β-naphthol, 2-hydroxynaphthalene with molecular formula C10H8O and melting point 122 °C, is a naphthalene homologue of phenol, bearing hydroxyl group at 2-position. 2-Naphthol has attracted considerable attention as valuable precursors for the synthesis of diverse heterocyclic compounds in organic synthesis owing to the presence of three available nucleophilic site, i.e., C-1 position, phenolic oxygen and C-3 position (to a lesser extent). This unique reactivity of 2-naphthol along with its easy accessibility and handling, moisture stability and low cost makes it fascinating candidate for organic chemists. 2-Naphthol has been used in the design and synthesis of privileged scaffolds like xanthenes, chromenes, oxazines, furans, naphthopyrans, etc. Heterocyclic compounds hold a prominent position in medicinal chemistry owing to their wide spectrum of biological activities such as antimalarial [1], antimicrobial [2], antitumor [3], anticancer [4], antidepressant [5], antiviral [6], antidiabetic [7], anti-inflammatory [8] and anti-HIV [9]. Moreover, they also contribute in the field of material science [10], dyes and pigment science [11] as well as agrochemistry [12]. Therefore, there is considerable thrust for the development of efficient synthetic strategies for producing these compounds. MCRs open diverse avenues to create novel concatenations in one-pot fashion leading to diverse biologically potent heterocyclic scaffolds [13, 14]. Having a cascade of reactions occurring in one pot is highly beneficial in the context of modern trends for organic synthesis, where sustainability is as relevant as efficiency and selectivity. Multicomponent reactions being atom economic, efficient and extremely convergent in nature offer a number of advantages over stepwise sequential approaches [15,16,17]. Ring-forming multicomponent reactions involving 2-naphthol promise an enhancement of structural complexity and functional diversity. This review provides an account of synthesis of a variety of heterocyclic compounds via one-pot multicomponent reactions of 2-naphthol.
Synthesis of heterocyclic compounds via multicomponent reactions of 2-naphthol
Xanthene
Xanthenes and benzoxanthenes are important oxygen-containing heterocyclic scaffolds that are found in natural products as well as in pharmaceutically active agents. The xanthene nucleus also referred as 9H-xanthene corresponds to dibenzo[b,e]pyran (Fig. 1). Furthermore, based on their orientation of annulation, benzoxanthenes and their derivatives essentially exist as three plausible isomers, benzo[a]xanthene 2, benzo[b]xanthene 3 and benzo[c]xanthene 4 (Fig. 1). They exhibit an array of biological activities like anti-inflammatory [18], antibacterial [19], antiviral [20], antioxidant [21] and antiplasmodial [22]. Moreover, they also find applications as dyes [23], fluorescent materials for the visualization of biomolecules [24] and in laser technology [25]. A large number of methods have been reported in the literature for the preparation and scaffold manipulation of these compounds.
The synthesis of 14-aryl-14H-dibenzo[a,j]xanthenes 3 by condensation of 2-naphthol 1 with aldehydes 2 has been reported utilizing various Brønsted acid catalysts (Scheme 1, Table 1).
Development of efficient synthetic methodologies has become imperative in the field of organic synthesis. In this context, the principles of ‘Green Chemistry’ such as atom economy, waste reduction and efficiency are given utmost importance. Compared with homogeneous catalysts, heterogeneous catalysts have received much attention owing to their merits like high activity, ease of separation and recycling. Several methodologies involving use of heterogeneous Brønsted acid catalysts under solvent-free conditions have been reported for the synthesis of 14-aryl-14H-dibenzo[a,j]xanthenes 3 like silica-supported perchloric acid (HClO4–SiO2) [26], silica sulfuric acid [27], cellulose sulfuric acid [28], aluminum hydrogen sulfate (Al(HSO4)3) [29], preyssler-type heteropolyacid, H14[NaP5W30O110] [30], poly(4-vinylpyridinium) hydrogen sulfate (P(4-VPH)HSO4) [31], silica-supported fluoroboric acid (HBF4–SiO2) [32], Fe3O4@SiO2-imid-H-3PMo12O40 nanoparticles [33], PEG-SO3H [34], sulfonated diatomite (diatomite-SO3H) [35], cucurbit[6]uril-OSO3H (CB[6]-OSO3H) [36], amberlyst-15 [37], phosphosulfonic acid (PSA) [38], silicotungstic acid (H4[SiW12O40]) [39], silica-supported ammonium dihydrogen phosphate (NH4·H2PO4·SiO2) [40], tungstophosphoric acid/zirconia composites (ZrTPA60BT100) [41], magnetite–sulfuric acid (Fe3O4·SO3H) magnetic nanoparticles [42], sulfonated single walled carbon nanotube (SWCNT-SO3H) [43], sulfonic acid-functionalized mesoporous SBA-15 (SBA-15/SO3H) [44], sodium hydrogen sulfate (NaHSO4·H2O) [45], p-sulfonic acid calix[4]arenes [46], sulfamic acid [47], DOWEX -50 W [48], D-camphorsulfonic acid (CSA) [49], Indion-130 [50] and NaHSO4–SiO2 [51]. These solvent-free protocols have emerged as a powerful tool in the light of current paradigm shift to “Green Chemistry” due to avoidance of harmful organic solvents, decrease energy consumption, short reaction time, simple work-up, ease of isolation, environmental benign nature. Furthermore, the synergy of solvent-free reactions with non-conventional energy source like microwave irradiation used by authors [45, 47, 49] illustrates another facet of sustainable chemistry.
Furthermore, replacement of hazardous solvents with environmentally benign reaction media like water, polyethylene glycol and ionic liquids is also one of the major focus areas of green chemistry. The above condensation has also been reported utilizing acid-functionalized hybrid mesoporous organosilica, AFS-1 [52] and polyvinylsulfonic acid (PVSA) [53] in aqueous media. The high polarity, hydrogen bonding capability and hydrophobic effect of water are also known to enhance the rate of reaction. Mahdavinia and co-workers [54] have developed an ultrasound-assisted protocol for the synthesis of 3 in aqueous media using silica-supported ammonium dihydrogen phosphate (NH4·H2PO4·SiO2) as catalyst. Significant reduction in reaction time and improvement in yield of the product were observed by authors under ultrasonic irradiation as compared to conventional heating method.
Solid Brønsted acid catalysts like magnesium hydrogen sulfate (Mg(HSO4)2) [55], boric acid (H3BO3) [56], p-toluene sulfonic acid (pTSA) [57], wet-2,4,6-trichloro[1,3,5]triazine (TCT) [58] were also employed for the synthesis of 3.
The probable mechanism for the synthesis of 14-aryl-14H-dibenzo[a,j]xanthenes is shown in Scheme 2.
Several Lewis acidic catalysts have been reported to catalyze the condensation of 2-naphthol and aldehydes for the synthesis of 14-aryl-14H-dibenzo[a,j]xanthenes 3 such as LiBr [59], ferric chloride (FeCl3) [60], ytterbium triflate (Yb(OTf)3) [61], ceric ammonium nitrate (CAN) [62], iron triflate (Fe(OTf)3) [63], bismuth chloride [64], tungsten(VI) chloride (WCl6) [65], tantalum chloride (TaCl5) [66], zirconium(IV) oxide chloride (ZrOCl2.8H2O) [67], ceric sulfate (Ce(SO4)2) [68], tetra-n-butylammonium bromide (TBAB) [69], P2O5/Al2O3 [70], dipyridine copper chloride (CuPy2Cl2) [71], dipyridine cobalt chloride (CoPy2Cl2) [72], iodine [73, 74], silica-supported boron trifluoride (BF3·SiO2) [75] and SelectfluorTM [76]. Nanocatalysts like CuS quantum dots (CuS QDs) [77], nano-CuO [78], nano-ZnO [79], Fe2+ supported on hydroxyapatite-core–shell-γ-Fe2O3 nanoparticles (γ-Fe2O3-HAp-Fe2+ Nps) [80], poly(4-vinylpyridine)-supported copper iodide nanoparticles (P4VPy-CuI Nps) [81] and ruthenium anchored on multi-walled carbon nanotubes (Ru@SH-MWCNT) [82] have also been utilized for the above condensation (Scheme 1) (Table 2). The use of nanocatalysts offers unique properties such as high surface area, enhanced catalytic sites, chemical inertness, durability and insolubility in most solvents. Moreover, their high surface allows higher loads of the catalytic sites.
Catalyst-free condensation of 2-naphthol 1 with aldehydes 2 has been reported in various acidic ionic liquids like 1,10-disulfo-[2,20-bipyridine]-1,10-diium chloride, [BiPy](SO3H)2Cl [83], DSIMHS [84], 1-methyl-3-propane sulfonic-imidazolium hydrosulfate ([MIMPS]HSO4) [85], di-n-propylammonium hydrogen sulfate ([(n-propyl)2NH2]HSO4) [86], 1,3-disulfonic acid imidazolium carboxylate ionic liquids (i.e., [DISM]CCl3COO & [DISM]CF3COO) [87]) as well as basic ionic liquid like bis-2,3,4,6,7,8,9,10-octahydropyrimido[1,2-a]azepinnium-ethyl disulfate [DBU]2EDS [88]. The task-specific ionic liquids serve the dual role of catalyst as well as solvent and bears interesting properties like excellent chemical and thermal stability, non-volatility, good solvating ability, wide liquid range as well as ease of recyclability. Several catalysts were also employed in ionic liquid as reaction medium such as trifluoroacetic acid in TMGT [89], Mg(BF4)2 doped in [Bmim]BF4 [90] and 2-1′-methylimidazolium-3-yl-1-ethyl sulfate in [Bmim]BF4 [91] (Scheme 1, Table 3). The integrity of the TMGT [89] and [Bmim]BF4 [90] remains reasonably unchanged after separation from the reaction mixture and was reportedly recycled several times without any loss of activity.
The synthesis of 12-aryl-8,9,10,12-tetrahydrobenzo[a]xanthen-11-one derivatives 6 via one-pot condensation reaction of 2-naphthol 1 with aldehydes 2 and dimedone 4/cyclohexa-1,3-dione 5 has been reported by Brønsted acids both liquids like H2SO4 [92], methanesulfonic acid [93] and solid such as dodecatungstophosphoric acid [94], p-dodecylbenzenesulfonic acid (DBSA) [95], pTSA [92, 96], HY zeolite [97], 1,3,5-triazo-2,4,6-triphosphorine-2,2,4,4,6,6-hexachloride supported on boehmite nanoparticles (BNPs-TAPC) [98], nanoparticle silica-supported sulfuric acid (NPs SiO2–H2SO4) [99], polymeric catalyst [poly(AMPS-co-AA)] [100], boronsulfonic acid (B(HSO4)3) [101], caro’s acid–silica [102], N-sulfonic acid modified poly(styrene-maleic anhydride) (SMI-SO3H) [103], polyvinylpolypyrrolidone-supported triflic acid (PVPP.OTf) [104], nanocrystalline TiO2–HClO4 [105] and silica-supported catalysts (HBF4/SiO2) [106] (Scheme 3, Table 4). The use of homogeneous catalysts like sulfuric acid and methanesulfonic acid has some disadvantages like corrosive nature of catalyst and more laborious work-up. The application of natural catalysts like lignosulfonic acid [107], citric acid [108], cellulose-SO3H [109], glucose sulfonic acid [110], D-xylonic acid [111], being efficient, cost-effective and biodegradable contributes to the green credentials of these protocols.
The mechanism for the formation of 12-aryl-8,9,10,12-tetrahydrobenzo[a]xanthen-11-one derivatives 6 in the presence of Brønsted acid (pTSA) is depicted in Scheme 4.
Solid catalysts showing Lewis acidic nature were reportedly used to bring the condensation of 2-naphthol 1 with cyclohexane-1,3-dione derivatives (4/5) and aldehydes 2 like CAN [62], ruthenium anchored on multi-walled carbon nanotubes [82], iodine [112, 113], ammonium chloride (NH4Cl) [114], cerium(III) chloride (CeCl3) [115], InCl3,/P2O5 [116], strontium triflate (Sr(OTf)2) [117], alum [118], ruthenium chloride hydrate [119], nano-TiCl4/SiO2 [120], rice husk [121], Ce(SO4)2·4H2O [122], Cu/SiO2 [123], manganese perchlorate [124], tetrabutylammonium fluoride (TBAF) [125], cerium-impregnated-MCM-41 [126], chitosan synergistically enhanced by successive Fe3O4 and silver nanoparticles [127] and trityl chloride [128] (Scheme 3, Table 5). Use of microwave or ultrasonication techniques by authors [82, 113, 124] has shown to significantly enhance the rate of the reactions, improve the yields as well as decrease the reaction time.
Tetrahydrobenzo[a]xanthene-11-ones 6 were also reported to be synthesized via one-pot three-component condensation of 2-naphthol 1 with cyclohexane-1,3-dione derivatives 4/5 and aldehydes 2 (Scheme 4, Table 6) using task-specific acidic ionic liquids like [BiPy](SO3H)2Cl2 [83], DSIMHS [84], [DBU]2EDS [88], [bmim]PF6 [129], [DDPA]HSO4 [130], 1-butane sulfonic acid-3-methylimidazolium tosylate ([BSMIM]Ts [131], [Dsim]Cl/[Msim]PF6/[Msim]BF4) [132] and [NMP]H2PO4 [133] under solvent-free conditions.
Basic organocatalysts like imidazole and isoquinoline are also employed as catalyst for carrying out the above condensation [134] (Scheme 5, Table 7). Herein, initially imidazole or isoquinoline catalyzed Knoevenagel condensation between aldehyde and dimedone takes place, followed by reaction of 2-naphthol with above formed intermediate to give desired xanthene 6. This three-component condensation reaction went well with a variety of aldehydes bearing both electron-withdrawing and electron-releasing substituents.
The condensation of dimedone 4/cyclohexane-1,3-dione 5 and aromatic aldehydes 2 with 2,6-naphthalenediol 1a for the synthesis of 3-hydroxy-12-arylbenzo[a]xanthen-11-ones 6a has been described in the presence of ionic liquids, viz. [NMP]H2PO4 [133] and [Bmim]BF4 [135] (Scheme 6). Moreover, the synthesis of 2-hydroxy-12-aryl-8,9,10,12-tetrahydrobenzo[a]xanthene-11-one derivatives 6b by condensation of 2,7-naphthalenediol 1b with dimedone 4/cyclohexane-1,3-dione 5 and aldehydes 2 has also been accomplished in ammonium chloride [114], [NMP]H2PO4 [133], pTSA in ethanol under reflux as well as in [Bmim]BF4 [136] (Scheme 6, Table 8).
3-Bromo-12-aryl-8,9,10,12-tetrahydrobenzo[a]xanthene-11-ones 6c were reportedly synthesized via reactions of dimedone 4 and aldehydes 2 with 6-bromo-2-naphthol 1d in the presence of ammonium chloride by Foroughifar et al. [114] (Scheme 7).
Bis-tetrahydrobenzo[a]xanthen-11-ones or 5,12-diarylxantheno[2,1-a]xanthene-4,12-diones 7 were synthesized by reaction of 2,6-naphthalenediol 1b with 5,5-dimethylcyclohexane-1,3-dione 4 and aromatic aldehydes 2 in [Bmim]BF4 [135] (Scheme 8). It was noticed that the reactions with aldehydes bearing electron-deficient substituent gave the corresponding bis-products when the molar concentrations of aldehyde and dimedone were doubled, whereas electron-rich aldehydes required higher concentrations (1:3:3) to afford the desired products 7.
The synthesis of 14-aryl-14H-dibenzo[a,i]xanthene-8,13-diones 9 by condensation of 2-hydroxy-1,4-naphthoquinone 8, aromatic aldehydes 2 and substituted 2-naphthols 1/1a/1b (Scheme 9, Table 9) has been reported in the presence of sulfuric acid [137], [bmim]HSO4 [137], under solvent-free condition by using Lewis acid like silica chloride [138], polystyrene-supported GaCl3 [139], as well as Brønsted acids such as acetic acid [140], pTSA [141], heterogeneous nanocatalysts like Fe3O4@SiO2–SO3H nanoparticles [142] and nano-Fe3O4/PEG/succinic anhydride [143].
One-pot three-component reaction of 2-naphthol 1, 2-hydroxy-1,4-naphthoquinone 8 and isatins 10 in the presence of silicotungstic acid (H4SiW12O40) [144] as well as [hmim]HSO4 [145] afforded spiro[dibenzo[a,i]-xanthene-14,30-indoline]-20,8,13-triones 11 (Scheme 10). In both cases, electronic effect of the substituents on indole ring had no significant effect on the product yield as well as reaction time. Moreover, the reusability of the recycled catalyst has also been demonstrated in the above protocols.
A possible mechanism for the above three-component reaction is outlined in Scheme 11. 2-Naphthol is believed to initially react with isatin to afford condensation product, followed by addition of 2-hydroxy-1,4- naphthoquinone. Subsequently, cyclization takes place to afford the desired product.
The one-pot three-component condensation of 2-naphthol 1, isatins 10 and cyclic 1,3-dicarbonyl compounds like dimedone 4/cyclohexa-1,3-dione 5 in the presence of mesoporous magnetite nanoparticles (Fe3O4@MCM-41-SO3H@[HMIm]HSO4) as catalyst was successfully established for the synthesis of spiro[benzoxanthene-indoline]diones 12 [146] (Scheme 12). The above protocol offers several advantages like reusability of magnetite nanoparticle, high yield, short reaction time, solvent-free conditions and ease of isolation of product.
Pyrans
Polyfunctionalized pyrans and chromenes belong to interesting class of heterocycles due to their vast biological and pharmacological importance. Pyrans commonly classified on the basis in the presence of the 2H- or 4H-pyran scaffold (Fig. 2) have been reported to possess biological properties such as antitumor [147], anti-proliferative [148], antiviral [149], antibacterial [150] and antifungal [151]. They also find application as insect pheromones [152, 153] and photoactive materials [154, 155]. Moreover, benzopyrans or chromenes being crucial components of a variety of natural compounds like alkaloids, flavonoids and tocopherols hold a position of prominence attributed to their biological activities which include antibacterial [156], antimicrobial [157], antioxidant [158], anti-hyperglycemic and anti-dyslipidemic [159]. Benzopyrans, a bicyclic organic compound that results from the fusion of a benzene ring to a pyran ring, include commonly structural skeletons such as 4H-chromene (4H-1benzopyran) and 2H-chromene (2H-1-benzopyran). Based on fusion of benzene or naphthalene ring with 2H-chromene, they are called as benzo[c]chromene, benzo[f]chromene, benzo[g]chromene and benzo[h]chromene (Fig. 2).
The synthesis of 3-amino-1H-benzo[f]chromenes 16, via MCRs of 2-naphthol 1, aldehydes 2 and malononitrile 13/ethyl cyanoacetate 14/cyanoacetamide 15 has been described under various conditions (Scheme 13).
Acid catalysts like CAN [62], TBAC [160], ferric hydrogen sulfate [161], disodium hydrogen phosphate (Na2HPO4) [162], molecular sieves 5Ao [163], SBA-15@methenamine-HPA [164], nano-sawdust–BF3 [165] and titanium tetrachloride (TiCl4) [166] were used for the synthesis of 16.
Various basic catalysts [167,168,169,170,171,172,173,174,175,176,177,178,179,180,181,182] were employed for the condensation of 2-naphthol with aldehydes and active methylene compounds. (Scheme 13, Table 10). The clean, transition-metal-free and environmentally friendly approaches for the synthesis of 16 have been realized using organocatalysts like diazabicyclo[2.2.2]octane (DABCO) [167], morpholine [168], piperazine [169], potassium phthalimide (POPI) [170], tetraethylammonium 2-(carbamoyl)benzoate (TEACB) [171] and triazine-based porous organic polymer TPOP-2 [172]. These procedures offer many advantages including short reaction times, low cost and straightforward work-up.
Nanocatalysts like tetragonal ZrO2 nanoparticles (t-ZrO2 NPs) [173], nano-mixed CuO–ZnO [174], nano-silica-bonded aminoethylpiperazine (SB-APP) [175], 1,5,7-triazabicyclo-[4,4,0]-dec-1-ene (TBD) a cyclic guanidine base anchored by mesoporous silica nanoparticles (MSN) [176] and nano-polypropylenimine dendrimer (DAB-PPI-G1) [177], were utilized for the above condensation. Biocatalysts like “Amano” lipase AS [178] and bael fruit extract (BFE) [179] were also used as catalyst for the synthesis of 16 by Jiang et al. and Shinde and co-workers, respectively. The exploitation of low cost, biodegradable and highly efficient catalyst, i.e., BFE, obtained from the rind of Aegle marmelos (bael) fruit offers benefits like elimination of toxic catalysts/reagents, reuse of catalyst and excellent yield of product in a very short reaction time.
Catalytic activity of other basic catalysts such as sodium carbonate [180, 181], high surface MgO [182], KF-Al2O3 [183] and poly(4-vinyl pyridine) [184] was also demonstrated by various researchers. Recyclable basic ionic liquids such as 3-hydroxypropanaminium acetate [185] and [Et2NH(CH2)2CO2H]AcO [186] were also employed as catalyst for above condensation.
The possible reaction pathway for this three-component reaction catalyzed by base proceeds via the Knoevenagel condensation of aldehyde and active methylene compound to afford Knoevenagel adduct. Thereafter, nucleophilic addition of the OH group of 2-naphthol to the CN moiety of adduct occurs through Michael addition. This is followed by intramolecular cyclization to form 3-amino-1H-benzo[f]chromenes 16 (Scheme 14).
The synthesis of 1,2-bis(4-nitrophenyl)-1-benzo[f]chromen-3-amine derivative 18 has been described by one-pot three-component reaction of 2-naphthol 1, aromatic aldehydes 2, 4-nitrophenyl acetonitrile 17 by using pTSA as the catalyst in ethanol reflux (Scheme 15) [187].
The synthesis of benzo[f]chromenes 19–20 via condensation of aromatic aldehydes 2 and malononitrile 13 with 2,3-dihydroxynaphthalene 1c or 2,7-dihydroxynaphthalene 1b using guanidine hydrochloride as the catalyst under solvent-free conditions [188] (Scheme 16) and CuO–ZnO nanocatalyst in water under reflux conditions [177] (Scheme 17), respectively, has been reported. CuO–ZnO nanocatalyst used by Albadi and co-workers was reportedly recycled up 6 times without any significant loss of its catalytic activity.
Fused 1H-benzo[f]chromen-indoles 22 were synthesized regioselectively by triethylamine-catalyzed condensation of 2-naphthol 1 with aromatic aldehydes 2 and 3-cyanoacetylindoles 21 in methanol under both ultrasonic irradiations and conventional conditions (Scheme 18) [189]. The reaction promoted by ultrasound offered better yields and much lower reaction times than the conventional heating. Notably, aldehydes with electron-donating substituents on phenyl ring furnished lower yields of furans than those with electron-withdrawing substituents.
Condensation of 2-naphthol 1 and aldehydes 2 was carried out with a variety of substrates under different conditions to afford chromenes. One-pot three-component condensation reactions of 2-naphthol 1 and aldehydes 2 with cyclopentane-1,3-dione 23 with strontium triflate afforded 8,9-dihydrobenzo-[f]cyclopenta[b]chromen-10(11H)-ones 26 [117], with Meldrum’s acid 24 in the presence of cerium(III) chloride [115] as well as TBAF yielded benzo[f]chromen-3-ones 27 [125], with indane-1,3-dione 25 gave benzo[f]indeno[1,2-b]chromen-12(13H)-one 28 in the presence of cerium-impregnated-MCM-41 [126], respectively (Scheme 19).
The synthesis of naphtho[10,20:5,6]pyrano-[3,2-c]chromen-6-ones 30 was accomplished by three-component condensation reaction of 2-naphthol 1, aromatic aldehydes 2 and 4-hydroxycoumarin 29 catalyzed by reusable catalysts like 1-methyl-3-(2-(sulfoxy)ethyl)-1H-imidazol-3-ium chloride [190], melamine trisulfonic acid (MTSA) [191], potassium 2-oxoimidazolidine-1,3-diide (POImD) [192], respectively (Scheme 20). The electronic and steric effects of substituents in aromatic aldehydes had no significant effect on the yields of the product in all three above-mentioned protocols [190,191,192].
Triethylamine-catalyzed one-pot three-component condensation reaction between naphthols like 2-naphthol 1/6-bromonaphthol 1e, formaldehyde 31 and trans-β-nitrostyrene 32 for the formation of benzo[f]chromene derivatives 33 was carried out by Bhattacharjee and co-workers [193] (Scheme 21). Reactions attempted using aliphatic nitroalkene in place of trans-β-nitrostyrene in the above condensation were unfruitful.
Boron trifluoride etherate has been successfully employed as catalyst for the synthesis of chromenes 36a–c via condensation of aldehydes 2 and acetonylacetone 34/ethylacetoacetate 35 with 2-naphthol 1 as well as its derivatives like 2,3-dihydroxynaphthalene 1c and 2,7-dihydroxynaphthalene 1b and by Mashraqui and co-workers [194] (Scheme 22).
One-pot multicomponent condensation of 2-naphthol 1 with aromatic aldehydes 2 and β-oxobenzenepropane (dithioates) 37 has been described using catalytic amount of BF3·OEt2 for the regioselective synthesis of several 1H-naphtho[2,1-b]pyrans 38 under solvent-free conditions (Scheme 23) [195]. The reactions attempted using phenol or 1-naphthol instead of 2-naphthol were not successful due to their lower reactivity.
Benzo[5,6]chromeno[2,3-d]pyrimidine-9,11(10H)-dione derivatives 40a–c were reportedly synthesized by one-pot multicomponent condensation reaction of 2-naphthols 1/1a/1b with aromatic aldehyde 2 and 1,3-dimethyl barbituric acid 39 in the presence of indium trichloride [116], phosphorus pentoxide [116], iodine [196], iodine in acetic acid [113], ZrOCl2/nano-TiO2 [197] and alum (KAl(SO4)2.12H2O) [198] (Scheme 24) (Table 11).
Moreover, Nandi and co-workers have reported the synthesis of benzo[5,6]chromeno[2,3-d]pyrimidine-9,11(10H)-diones 40 by reacting 2-naphthol 1 with 6-amino-1,3-dimethyl uracil 41 and aromatic aldehydes 2 using indium trichloride under solvent-free conditions [199] (Scheme 25). The reaction attempted using aliphatic aldehydes did not give desired product.
Cimarelli and co-workers [200] have reported the stereoselective synthesis of 2,3-dihydro-1H-benzo[f]chromen-3-amine derivatives 44 under catalyst and solvent-free conditions (Scheme 26) via three-component one-pot reaction between 2-naphthol 1, α, β-unsaturated aldehydes 42 and chiral phenylethylamine 43.
3H-Benzo[f]chromene-2-carboxamides 47 were synthesized from three-component cyclocondensation reaction of 2-naphthol 1, propargyl alcohols 45 and cyclohexylisocyanide 46 in the presence of ZnI2 and FeCl3 (Scheme 27) [201]. The mechanism for the formation of 47 is depicted in Scheme 28. Reactions attempted using either 2-methyl-3-butyn-2-ol or other isocyanides such as t-butylisocyanide and isopropylisocyanide gave a mixture of unidentified product.
Yadav et al. [202] have described the synthesis of 1,3-diaryl-3H-benzo[f]chromenes 49 by reaction of 2-naphthol 1, aldehydes 2 and phenyl acetylene 48 using catalytic amount of gallium chloride in toluene as solvent under reflux conditions (Scheme 29). The reaction is believed to proceed via arylation of alkyne to afford vinylnaphthalene-2-ol which subsequently undergoes cyclization with aldehyde to give the desired chromenes 49 (Scheme 30).
Spironaphthopyrano[2,3-d]pyrimidine-5,3′-indolines 51 were reportedly synthesized by one-pot condensation of 2-naphthol 1, barbituric acids 39/thiobarbituric acid 50 with isatins 10 and under solvent-free and catalyst-free conditions [203] and also in the presence of [Hmim]HSO4 under solvent-free conditions [145], sodium dodecyl sulfate (SDS) in water [204] and sulfonic acid-functionalized SBA-15 (SBA-Pr-SO3H) [205] (Scheme 31, Table 12).
The mechanistic pathway for the formation of spironaphthopyrano[2,3-d]pyrimidine-5,3′-indolines 51 catalyzed by SBA-Pr-SO3H is depicted in Scheme 32. Initially, acid-catalyzed condensation of 2-naphthol with isatin takes place to furnish intermediate A. Then, a subsequent addition of barbituric acid to the intermediate A, followed by a cyclization and dehydration provides the desired product 51 (Scheme 32).
Asadi and co-workers [203] have efficiently synthesized a series of spironaphthopyrano[1,2-b]indeno-7,3-indolines 52 by multicomponent reactions of 2-naphthol 1, indane-1,3-dione 25 and isatins 10 under solvent-free conditions without any catalyst (Scheme 33). Electron-withdrawing substituents on isatin were found to reduce the reaction time.
The synthesis of spiro-oxindoles with fused chromenes 53 (Scheme 34), through the three-component reaction of isatin derivatives 10, malononitrile 13 or cyanoacetic ester 14 and 2-naphthol 1 compounds using l-proline [206], indium trichloride [207] and cellulose-SO3H [138] (Table 13).
2-Aminospiro[benzo[g]chromene-4,11′-indeno[1,2-b]quinoxaline]-3-carbonitriles 56 were reportedly synthesized by condensation of malononitrile 13 or ethyl 2-cyanoacetate 14, ninhydrin 54, 1,2-phenylenediamine 55, 2-naphthol 1 under refluxing or ultrasound irradiation at room temperature in good yields (Scheme 35) [208] using green and recyclable trifluoroethanol as catalyst. The reaction time under ultrasonic irradiation was significantly reduced as compared to conventional heating.
The synthesis of 10-methyl-8H-spiro[benzo[5,6]chromeno[2,3-c]pyrazole-11,3′-indol]-2′(1′H)-ones 58 by four-component reactions of phenylhydrazine/hydrazine hydrate 57, isatins 10, ethylacetoacetate 35 and 2-naphthol 1 using nano-Co3S4 under microwave irradiation (Scheme 36) [209]. The method offers several advantages including utilization of microwave irradiation as clean procedure, high atom economy, high yields, shorter reaction times, low catalyst loading and reusability of the catalyst.
1,3-Oxazines
Oxazines are six-membered heterocyclic compounds which contain one nitrogen and one oxygen atom. Depending on the relative position of the heteroatoms, i.e., oxygen and nitrogen atom, they are known to exist in 3 isomeric forms, viz. 1,2-oxazine, 1,3-oxazine and 1,4-oxazine. 1,3-Oxazines are privileged heterocyclic scaffolds with interesting biological activities such as antimicrobial [210], non-nucleoside reverse transcriptase inhibitor [211], nonsteroidal progesterone receptor agonists [212], and antitumor [213].
Azizian and co-workers [214] investigated the microwave-assisted one-pot condensation reaction of N,N,N’,N’-tetramethylguanidine (TMG) 59, aryl-/heteroaryl-aldehydes 2 and 2-naphthol 1 using acetic acid as catalyst for the synthesis of 1-aryl-N,N-dimethyl-1H-naphtho[1,2-e] [1, 3] -oxazine-3-amine derivatives 60 (Scheme 37). Scheme 38 represents the mechanism for the formation of 60. The first step involves the formation of intermediate by reaction of the aldehyde with TMG. Subsequently, addition of 2-naphthol to the intermediate occurs, followed by cyclization to affords the corresponding products 60.
Several 1,2-dihydro-1-arylnaphtho[1,2-e] [1, 3] oxazine-3-ones 62 were synthesized by three-component condensation of 2-naphthol 1, aldehydes 2 and urea 61 in the presence of nano-silica-supported ferric chloride [215], potassium carbonate and copper nanoparticles [216], ZnO nanoparticles [217], AgI nanoparticles [218], sulfuric acid-functionalized silica-coated magnetic nanoparticles (MgFe2O4@SiO2–SO3H) [219], magnetite (Fe3O4) nanoparticles [220] as nanocatalysts (Table 14) (Scheme 39).
Several other catalysts such as pTSA [221], iodine [222], montmorillonite K10 clay [223], zinc triflate [224], phosphomolybdic acid [225], Amberlite IRA-400 Cl resin [226], graphene oxide [227], TMSCl/NaI [228], N-propane sulfonic acid pyridinium hydrogen sulfate ([PSPy]HSO4) [229], RuCl2(PPh3)3 [230], cellulose sulfuric acid/SDS [231] and thiamine hydrochloride (VB1) [232] were also found to effect the above synthesis. The condensation of 2-naphthol 1 and aldehydes 2 with thiourea 63 instead of urea 61 leads to the formation of 1,2-dihydro-3H-naphtho[1,2-e] [1, 3] oxazine-3-thione 64 [219, 220, 230, 231] (Scheme 40, Table 15).
An efficient stereoselective synthesis of diverse trans-naphtho[1,2-e] [1, 3] oxazines 66 via one-pot condensation reaction of 2-naphthol 1, 3-amino-5-methylisoxazole 65 and arylaldehydes 2 catalyzed by bismuth(III) trifluoromethane sulfonate has been described by Shafiee et al. [233] (Scheme 41). Aldehydes with both electron-donating and electron-withdrawing substituents afforded the desired product in high stereoselectivity.
Catalyst-free synthesis of 1,3-disubstituted-2,3-dihydro-1H-naphth[1,2-e] [1, 3] oxazines 68 was achieved by Turgut and co-workers [234] via condensation of 2-naphthol 1 with two equivalents of aryl-/heteroaryl-aldehydes 2 in the presence of ammonia 67 at room temperature (Scheme 42).
The synthesis of 2,3-dihydro-1,2,3-trisubstituted-1H-naphth[1,2-e] [1, 3] oxazines 70 has been described by condensation reaction of 2-naphthol 1 with aldehydes 2 and primary amines 69 using Brønsted acids like pTSA [235], sulfamic acid [235], 1,3-disulfoimidazolium trifluoroacetate ([DSIM]CF3COO) [235], as well as triphenyl sulfophosphonium chlorometallates [236] with both Brønsted acid and Lewis acid property (Scheme 43, Table 16).
Various catalysts like sodium hydrogen sulfate [237], [Bmim]HSO4 [237], TBAB [237], iron(III) trifluroacetate ([Fe(CF3CO2)3]) [238], Fe3O4@nano-cellulose/TiCl [239], silica-supported boron trifluoride, BF3–SiO2 [240], thiamine hydrochloride (VB1) [241], glycerol [242], Fe3O4@MAP Nps [243], alum [244] were explored for the one-pot multicomponent condensation reaction of 2-naphthol 1, formaldehyde 31 and primary amines 69 for the synthesis of 2,3-dihydro-1H-naphtho-[1,2-e] [1, 3] oxazine derivatives 71 (Scheme 44, Table 17).
1,3-Oxazine-4-thione derivatives 74 were synthesized via one-pot two-step domino protocol from ammonium thiocyanate 72 and acid chlorides 73 and 2-naphthol 1 in the presence of an effective recyclable bifunctional organocatalyst, i.e., l-proline as in water [245] (Scheme 45). It was further noticed that benzoyl chlorides with electron-withdrawing substituents increased the rate of reaction and gave higher yields than those with electron-releasing groups.
Furan
Furans, an aromatic five-membered aromatic ring with oxygen as heteroatom (Fig. 3), constitute core entities in many natural products are very imperative among heterocyclic structures owing to their remarkable biological properties like anticancer, antidepressant, antianxiolytic, anti-inflammatory, muscle relaxant, antihypertensive, antidiuretic, anti-ulcer, antihistaminic, antiarrhythmic and analgesic. [246].
An efficient one-pot synthesis of benzamidobenzo[b]furans 77 has been developed via reaction of arylglyoxals 75, benzamide 76 and 2-naphthol 1 using yttrium nitrate hexahydrate or tungstate sulfuric acid (TSA) as a catalyst under solvent-free conditions [247, 248] (Scheme 46). TSA employed as catalyst by Vahabinia and co-workers can be recycled over three times without significant loss of activity.
Several naphthofuran-2(3H)-one analogues 80 were efficiently synthesized by three-component condensation reaction using 2-naphthols 1/1d, acetaldehyde 78 and carbon monoxide 79 in the presence of a palladium catalyst (Scheme 47) [249].
Miscellaneous
The reaction of dimethyl acetylenedicarboxylate (DMAD) 41 with 2-naphthols 1 in the presence of trimethyl or triphenylphosphite 81 leads to stable dimethyl oxa-2λ5-phosphaphenanthrene derivatives 82 [250] (Scheme 48).
One-pot, solvent-free microwave-assisted synthesis of 1,3,2-aryldioxaborines 84 in the presence of acidic alumina by reaction of 2-naphthol 1, phenylboronic acid 83 and aldehydes 2 (Scheme 49) has been reported by Reza Naimi-Jamal and co-workers [251]. The above method is tolerant to different aromatic and aliphatic aldehydes as well as naphthols.
Conclusion
A plethora of heterocyclic compounds like xanthenes, furans, pyrans and oxazines have been reportedly synthesized from 2-naphthol analogues. This review exemplifies the multicomponent reactions of 2-naphthol as building block for the synthesis of a variety of heterocyclic compounds. The potential of 2-naphthol in multicomponent reactions is still being discovered; thus, this review might trigger new ideas to use 2-naphthol as a building block for future research in heterocyclic chemistry.
References
Kalaria PN, Karad SC, Raval DK (2018) A review on diverse heterocyclic compounds as the privileged scaffolds in antimalarial drug discovery. Eur J Med Chem 158:917–936
Desai N, Trivedi A, Pandit U, Dodiya A, Rao VK, Desai P (2016) Hybrid bioactive heterocycles as potential antimicrobial agents: a review. Mini Rev Med Chem 16:1500–1526
Fouad MM, El-Bendary ER, Suddek GM, Shehata IA, El-Kerdawy MM (2018) Synthesis and in vitro antitumor evaluation of some new thiophenes and thieno[2,3-d]pyrimidine derivatives. Bioorg Chem 81:587–598
Martins P, Jesus J, Santos S, Raposo LR, Roma-Rodrigues C, Baptista PV, Fernandes AR (2015) Heterocyclic anticancer compounds: recent advances and the paradigm shift towards the use of nanomedicine’s tool box. Molecules 20:16852–16891
Siddiqui N, Andalip Bawa S, Ali R, Afzal O, Akhtar MJ, Azad B, Kumar R (2011) Antidepressant potential of nitrogen-containing heterocyclic moieties: an updated review. J Pharm Bioallied Sci 3:194–212
Sokolova AS, Yarovaya OI, Bormotov NI, Shishkina LN, Salakhutdinov NF (2018) Synthesis and antiviral activity of camphor-based 1,3-thiazolidin 4-one and thiazole derivatives as Orthopoxvirus-reproduction inhibitors. MedChemComm 9:1746–1753
Goel A, Agarwal N, Singh FV, Sharon A, Tiwari P, Dixit M, Pratap R, Srivastava AK, Maulik PR, Ram VJ (2004) Antihyperglycemic activity of 2-methyl-3,4,5-triaryl-1H-pyrroles in SLM and STZ models. Bioorg Med Chem Lett 14:1089–1092
Amir M, Javed SA, Kumar H (2007) Pyrimidine as anti-inflammatory agent: a review. Indian J Pharm Sci 69:337–343
Li W, Zhao SJ, Gao F, Lv ZS, Tu JY, Xu Z (2018) Synthesis and in vitro anti-tumor, anti-mycobacterial and anti-HIV activities of diethylene-glycol-tethered bis-isatin derivatives. ChemistrySelect 3:10250–10254
Zhao X, Chaudhry ST, Mei J (2017) Heterocyclic building blocks for organic semiconductors. Heterocyclic chemistry in the 21st Century—a Tribute to Alan Katritzky 121:133–171
Khattab TA, Rehan MA (2018) Review on synthesis of nitrogen-containing heterocyclic dyes for textile fibers—part 2: fused heterocycles. Egypt J Chem 61:989–1018
Lamberth C, Dinges J (2012) Bioactive heterocyclic compound classes: agrochemicals. Wiley-VCH Verlag GmbH & Co, KGaA
Zhi S, Ma X, Zhang W (2019) Consecutive multicomponent reactions for the synthesis of complex molecules. Org Biomol Chem 17:7632–7650
Ibarra IA, Islas-Jácome A, González-Zamora E (2018) Synthesis of polyheterocycles via multicomponent reactions. Org Biomol Chem 16:1402–1418
Tietze LF, Bsasche C, Gericke KM (2006) Domino reactions in organic synthesis. Wiley-VCH, Weinheim
Weber L, Illgen M, Almstetter M (1999) Discovery of new multi component reactions with combinatorial methods. Synlett 3:366–374
Herrera RP, Marqués-López E (2015) Multicomponent reactions: concepts and applications for design and synthesis. Wiley, Hoboken
Naya A, Sagara Y, Ohwaki K, Saeki T, Ichikawa D, Iwasawa Y, Noguchi K, Ohtake N (2001) Design, synthesis, and discovery of a novel CCR1 antagonist. J Med Chem 44:1429–1435
Limsuwan S, Trip EN, Kouwen TR, Piersma S, Hiranrat A, Mahabusarakam W, Voravuthikunchai SP, Dijl JMV, Kayser O (2009) Rhodomyrtone: a new candidate as natural antibacterial drug from Rhodomyrtus tomentosa. Phytomedicine 1:645–651
Carr AA, Grunwell JF, Sill AD, Meyer DR, Sweet FW, Scheve BJ, Grisar JM, Fleming RW, Mayer GD (1976) Bis-basic-substituted polycyclic aromatic compounds. A new class of antiviral agents. 7. Bisalkamine esters of 9-oxoxanthene-2,7-dicarboxylic acid, 3,6-bis-basic ethers of xanthen-9-one, and 2,7-bis(aminoacyl)xanthen-9-ones, -xanthenes, and –thioxanthenes. J Med Chem 19:1142–1148
Nishiyama T, Sakita K, Fuchigami T, Tsutomu Fukui T (1998) Antioxidant activities of fused heterocyclic compounds, xanthene-2,7-diols with BHT or catechol skeleton. Polym Degrad Stab 62:529–534
Zelefack F, Guilet D, Fabre N, Bayet C, Chevalle S, Ngouela S, Lenta BN, Valentin A, Tsamo E, Dijoux-Franca MG (2009) Cytotoxic and antiplasmodial xanthones from pentadesma butyracea. J Nat Prod 72:954–957
Banerjee A, Mukherjee AK (1981) Chemical aspects of santalin as a histological stain. Stain Technol 56:83–85
Knight CG, Stephens T (1989) Xanthene-dye-labelled phosphatidylethanolamines as probes of interfacial pH. Studies in phospholipid vesicles. Biochem J 258:683–687
Ahmad M, King TA, Do-Kyeong K, Cha BH, Jongmin L (2002) Performance and photostability of xanthene and pyrromethene laser dyes in sol–gel phases. J Phys D Appl Phys 35:1473–1476
Bigdeli MA, Heravi MM, Mahdavinia GH (2007) Silica supported perchloric acid (HClO4-SiO2): a mild, reusable and highly efficient heterogeneous catalyst for the synthesis of 14-aryl or alkyl-14-Hdibenzo[a, j]xanthenes. J Mol Catal A Chem 275:25–29
Shaterian HR, Ghashang M, Hassankhani A (2008) One-pot synthesis of aryl 14H-dibenzo[a, j]xanthene leuco-dye derivatives. Dyes Pigments 76:564–568
Madhav JV, Reddy YT, Reddy PN, Reddy MN, Kuarma S, Crooks A, Rajitha B (2009) Cellulose sulfuric acid. An efficient biodegradable and recyclable solid acid catalyst for the one-pot synthesis of aryl 14H-dibenzo[a.j]xanthenes under solvent-free conditions. J Mol Catal A Chem 304:85–87
Shaterian HR, Ghashang M, Mir N (2007) Aluminium hydrogensulfate as an efficient and heterogeneous catalyst for preparation of aryl 14H-dibenzo[a, j]xanthene derivatives under thermal and solvent-free condition. ARKIVOC 15:1–10
Heravi MM, Bakhtiari K, Daroogheha Z, Bamoharram FF (2007) Facile heteropolyacid-promoted synthesis of 14-substituted-14-H-dibenzo[a, j]xanthene derivatives under solvent-free conditions. J Mol Catal A Chem 272:99–101
Khaligh NG (2012) Poly(4-vinylpyridinium) hydrogen sulfate: an efficient catalyst for the synthesis of xanthene derivatives under solvent-free conditions. Catal Sci Technol 2:2211–2215
Liu YH, Tao XY, Lei LQ, Zhang ZH (2009) Fluoroboric acid adsorbed on silica-gel catalyzed synthesis of 14-aryl-14Hdibenzo[a, j]xanthene derivatives. Synth Commun 39:580–589
Esmaeilpour M, Javidi J, Dehghani F, Dodeji FN (2014) Fe3O4@SiO2-imid-PMAn magnetic porous nanospheres as recyclable catalysts for the one-pot synthesis of 14-aryl- or alkyl-14Hdibenzo[a, j]xanthenes and 1,8-dioxooctahydroxanthene derivatives under various conditions. New J Chem 38:5453–5461
Prasad D, Nath M (2012) PEG-SO3H catalyzed, environmentally benign synthesis of 14-aryl 14Hdibenzo[a, j]xanthenes under solvent-free conditions. Catal Sci Technol 2:93–96
Naeimi H, Nazifi ZS (2014) Sulfonated diatomite as heterogeneous acidic nanoporous catalyst for synthesis of 14-aryl-14-H-dibenzo[a, j]xanthenes under green conditions. Appl Catal A Gen 477:132–140
Saghanezhad SJ, Nazari Y, Davod F (2016) Cucurbit[6]uril-OSO3H: a novel acidic nanocatalyst for the one-pot preparation of 14-aryl-14H-dibenzo[a, j]xanthenes and 1,8-dioxo-octahydro-xanthenes. RSC Adv 6:25525–25530
Ko S, Yao CF (2006) Heterogeneous catalyst: amberlyst-15 catalyzes the synthesis of 14-substituted-14H-dibenzo[a, j]xanthenes under solvent-free conditions. Tetrahedron Lett 47:8827–8829
Rezayati S, Erfani Z, Hajinasiri R (2015) Phospho sulfonic acid as efficient heterogeneous Brønsted acidic catalyst for one-pot synthesis of 14H-dibenzo[a, j]xanthenes and 1,8-dioxo-octahydro-xanthenes. Chem Pap 69:536–543
Allameh S, Davoodni A, Khojastehnezhad A (2012) An efficient and eco-friendly synthesis of 14-aryl-14H-dibenzo[a, j]xanthenes using H4[SiW12O40] as a heterogeneous and reusable catalyst under solvent-free conditions. Chin Chem Lett 23:17–20
Rostamizadeh S, Amani AM, Mahdavinia GH, Shadjou N (2009) Silica supported ammonium dihydrogen phosphate (NH4H2PO4/SiO2): a mild, reusable and highly efficient heterogeneous catalyst for the synthesis of 14-aryl-14-Hdibenzo[a, j]xanthenes. Chin Chem Lett 20:779–783
Rivera TS, Blanco MN, Pizzio LR, Romanelli GP (2012) Green catalytic synthesis of 14-aryl-14H-dibenzo[a, j]xanthenes using recyclable mesoporous zirconia modified with tungstophosphoric acid. Green Chem Lett Rev 5:433–437
Karimi AR, Zeinab DK, Marzie K (2014) Magnetite-sulfuric acid magnetic nanoparticles: preparation and application in synthesis of mono-, bis-, and tris-14Hdibenzo[a, j]xanthen-14-yl-arenes under solvent-free conditions. Synthesis 46:917–922
Fareghi-Alamdari R, Golestanzadeh M, Agend F, Zekri N (2013) Application of highly sulfonated single-walled carbon nanotubes. An efficient heterogeneous catalyst for the one-pot synthesis of 14-aryl-14H-dibenzo[a, j]xanthenes under solvent-free conditions. CR Chim 16:878–887
Naik MA, Sachdev D, Dubey A (2010) Sulfonic acid functionalized mesoporous SBA-15 for one-pot synthesis of substituted aryl-14H-dibenzoxanthenes and bis(indolyl)methanes. Catal Commun 11:1148–1153
Shaterian HR, Doostmohammadi R, Ghashang M (2008) Sodium hydrogen sulfate as effective and reusable heterogeneous catalyst for the one-pot preparation of 14H-[(Un)substituted phenyl]-dibenzo[a, j]xanthene leuco-dye derivatives. Chin J Chem 26:338–342
Baghbanian SM, Khanzad G, Vahdat SM, Tashakkorian H (2015) p-Sulfonic acid calix[4]arene as an efficient and reusable catalyst for the synthesis of acridinediones and xanthenes. Res Chem Intermed 41:9951–9966
Rajitha B, Sunil Kumar B, Thirupathi Reddy Y, Narsimha Reddy P, Sreenivasulu N (2005) Sulfamic acid: a novel and efficient catalyst for the synthesis of aryl-14H-dibenzo[a.j]xanthenes under conventional heating and microwave irradiation. Tetrahedron Lett 46:8691–8693
Shakibaei GI, Mirzaei P, Bazgir A (2007) Dowex-50 W promoted synthesis of 14-aryl-14H-dibenzo[a, j]xanthene and 1,8-dioxo-octahydroxanthene derivatives under solvent-free conditions. Appl Catal A Gen 325:188–192
Kundu K, Nayak S (2014) Camphor-10-sulfonic acid catalyzed condensation of 2-naphthol with aromatic/aliphatic aldehydes to 14-aryl/alkyl-14H-dibenzo[a, j]xanthenes. J Ser Chem Soc 79:1051–1058
Patil SB, Bhat RP, Samant SD (2006) Cation-exchange resins: efficient heterogeneous catalysts for facile synthesis of dibenzoxanthene from β-naphthol and aldehydes. Synth Commun 36:2163–2168
Nagarapu L, Baseeruddin M, Kumari NV, Kantevari S, Rudradas AP (2007) Efficient synthesis of aryl-14H-dibenzo[a.j]xanthenes using NaHSO4–SiO2 or 5%WO3/ZrO2 as heterogeneous catalysts under conventional heating in a solvent-free media. Synth Commun 37:2519–2525
Nandi M, Mondal J, Sarkar K, Yamauchi Y, Bhaumik A (2011) Highly ordered acid functionalized SBA-15: a novel organocatalyst for the preparation of xanthenes. Chem Commun 47:6677–6679
Rahmatpour A (2011) An efficient, high yielding, and eco-friendly method for the synthesis of 14-aryl- or 14-alkyl-14H-dibenzo[a, j]xanthenes using polyvinylsulfonic acid as a recyclable Brønsted acid catalyst. Monatsh Chem 142:1259–1263
Mahdavinia GH, Rostamizadeh S, Amani AM, Emdadi Z (2008) Ultrasound-promoted greener synthesis of aryl-14-H-dibenzo[a, j]xanthenes catalyzed by NH4H2PO4/SiO2 in water. Ultrason Sonochem 16:7–10
Mirjalili BBF, Bamoniri A, Akbari A (2011) Synthesis of 14-aryl or alkyl-14Hdibenzo[a, j]xanthenes promoted by Mg(HSO4)2. Chin Chem Lett 22:45–48
Karimi-Jaberi Z, Keshavarzi M (2010) Efficient one-pot synthesis of 14-substituted-14H-dibenzo[a, j]xanthenes using boric acid under solvent-free conditions. Chin Chem Lett 21:547–549
Khosropour AR, Khodaei MM, Moghannian H (2005) A facile, simple and convenient method for the synthesis of 14-alkyl or aryl-14-H-dibenzo[a, j]xanthenes catalyzed by pTSA in solution and solvent-free conditions. Synlett 6:955–958
Bigdeli MA, Heravi MM, Mahdavinia GH (2007) Wet cyanuric chloride catalyzed simple and efficient synthesis of 14-aryl- or -alkyl-14H-dibenzo[a, j]xanthenes. Catal Commun 8:1595–1598
Saini A, Kumar S, Sandhu JS (2006) A new LiBr-catalyzed, facile and efficient method for the synthesis of 14-alkyl or aryl-14H-dibenzo[a, j]xanthenes and tetrahydrobenzo[b]pyrans under solvent-free conventional and microwave heating. Synlett 12:1928–1932
Wang B, Li P, Zhang Y, Wang L (2010) FeCl3-catalyzed condensation of 2-naphthol and aldehydes under solvent-free reaction conditions. An efficient and green alternative for the synthesis of 14-aryl(alkyl)-14-H dibenzo[a, j]xanthenes. Chin J Chem 28:2463–2468
Su W, Yang D, Jin C (2008) Yb(OTf)3 catalyzed condensation reaction of b-naphthol and aldehyde in ionic liquids: a green synthesis of aryl-14H-dibenzo[a, j]xanthenes. Tetrahedron Lett 49:3391–3394
Kumar A, Sharma S, Maury RA, Sarkar J (2010) Diversity oriented synthesis of benzoxanthene and benzochromene libraries via one-pot, three-component reactions and their anti-proliferative activity. ACS Comb Sci 12:20–24
Cao Y, Yao C, Qin B, Zhang H (2013) Solvent-free synthesis of 14-aryl-14H-dibenzo[a, j]xanthenes catalyzed by recyclable and reusable iron(III) triflate. Res Chem Intermed 39:3055–3062
Soleimani E, Khodaei MM, Koshvandi ATK (2011) The efficient synthesis of 14-alkyl or aryl 14H-dibenzo[a, j]xanthenes catalyzed by bismuth(III) chloride under solvent-free conditions. Chin Chem Lett 22:927–930
Zolfigol MA, Moosavi-Zare AR, Arghavani-Hadi P, Zare A, Khakyzadeh V, Darvishi G (2012) WCl6 as an efficient, heterogeneous and reusable catalyst for the preparation of 14-aryl-14H-dibenzo[a,j]xanthenes with high TOF. RSC Adv 2:3618–3620
Bhattacharya AK, Rana KC, Mujahid M, Sehar IS, Saxena AK (2009) Synthesis and in vitro study of 14-aryl-14Hdibenzo[a.j]xanthenes as cytotoxic agents. Bioorg Med Chem Lett 19:5590–5593
Mosaddegh E, Islami MR (2008) Synthesis of aryl 14H-dibenzo[a, j]xanthenes using zirconium(IV) oxide chloride as a catalyst. Org Prep Proc Int 40:586–589
Selvam NP, Shanthi G, Perumal PT (2007) Ceric-sulfate-catalyzed synthesis of 14-aryl- or 14-alkyl-14H-dibenzo[aj]xanthene under conventional heating and microwave irradiation. Can J Chem 85:989–995
Kantevari S, Chary MV, Das APR, Vuppalapati SVN, Lingaiah N (2008) Catalysis by an ionic liquid: highly efficient solvent-free synthesis of aryl-14-H-dibenzo[a.j]xanthenes by molten tetrabutylammonium bromide under conventional and microwave heating. Catal Commun 9:1575–1578
Zarei A, Hajipour AR, Khazdooz L (2010) The one-pot synthesis of 14-aryl or alkyl-14Hdibenzo[a,j]xanthenes catalyzed by P2O5/Al2O3 under microwave irradiation. Dyes Pigments 85:133–138
Madhav JV, Kumar VN, Someshwar P, Rajitha B (2008) A simple and convenient method for the synthesis of Aryl-14 H -dibenzo[a, j]xanthenes by using dipyridine copper chloride as Lewis acid catalyst. J Heterocycl Chem 45:119–121
Madhav JV, Kuarm BS, Rajitha B (2008) Dipyridine cobalt chloride: a novel and efficient catalyst for the synthesis of 14-aryl 14H-dibenzo[a, j]xanthenes under solvent-free conditions. ARKIVOC 2:204–209
Ding F-Q, An LT, Zou JP (2007) Iodine catalyzed microwave-assisted synthesis of 14-Aryl(Alkyl)-14H-dibenzo[a,j]xanthenes. Chin J Chem 25:645–648
Das B, Ravikanth B, Ramu R, Laxminarayana K, Rao BV (2006) Iodine catalyzed simple and efficient synthesis of 14-aryl or alkyl-14-H-dibenzo[a, j]xanthenes. J Mol Catal A Chem 255:74–77
Mirjalili BBF, Bamoniri A, Akbari A (2008) BF3·SiO2: an efficient alternative for the synthesis of 14-aryl or alkyl-14H-dibenzo[a, j]xanthenes. Tetrahedron Lett 49:6454–6456
Kumar PS, Kumar BS, Rajitha B, Reddy PN, Sreenivasulu N, Reddy YT (2006) A novel one pot synthesis of 14-aryl-14H-dibenzo[a, j]xanthenes catalyzed by SelectfluorTM under solvent free conditions. ARKIVOC 12:46–50
Bansal P, Chaudhary GR, Kaur N, Mehta SK (2015) An efficient and green synthesis of xanthene derivatives using CuS quantum dots as a heterogeneous and reusable catalyst under solvent free conditions. RSC Adv 5:8205–8209
Chaudhary GR, Bansal P, Kaur N, Mehta SK (2014) Recyclable CuO nanoparticles as heterogeneous catalysts for the synthesis of xanthenes under solvent free conditions. RSC Adv 4:49462–49470
Safaei-Ghomi J, Ghasemzadeh MA (2012) Zinc oxide nanoparticles, a highly efficient and readily recyclable catalyst for the synthesis of xanthenes. Chin Chem Lett 23:1225–1229
Haeri HH, Rezayati S, Nezhad ER, Darvishi H (2016) Fe2+ supported on hydroxyapatite-core-shell-γ-Fe2O3 nanoparticles: efficient and recyclable green catalyst for the synthesis of 14-aryl-14H-dibenzo[a,j]xanthene derivatives. Res Chem Intermed 42:4773–4784
Albadi A, Iravani N, Khoshakhlagh M (2012) A new, green and recyclable poly(4-vinylpyridine)-supported copper iodide nanoparticles catalyst for the synthesis of aryl-14H-dibenzo[a, j]xanthenes. Iran J Catal 2:85–89
Tabatabaeian K, Zanjanchi MA, Mamaghani M, Dadashi A (2015) Ruthenium anchored on multi-walled carbon nanotubes: an efficient and reusable catalyst for the synthesis of xanthenes. Res Chem Intermed 42:5049–5067
Shirini F, Abedini M, Seddighi M, Jolodar OG, Safarpoor M, Langroodi N, Zamani S (2014) Introduction of a new bi-SO3H ionic liquid based on 2,2-bipyridine as a novel catalyst forthe synthesis of various xanthene derivatives. RSC Adv 4:63526–63532
Shirini F, Yahyazadeh A, Mohammadi K (2014) One-pot synthesis of various xanthene derivatives using ionic liquid 1,3-disulfonic acid imidazolium hydrogen sulfate as an efficient and reusable catalyst under solvent free conditions. Chin Chem Lett 25:341–347
Gong K, Fang D, Wang HL, Zhou XL, Liu ZL (2008) The one-pot synthesis of 14-alkyl- or aryl-14H-dibenzo[a, j]xanthenes catalyzed by task-specific ionic liquid. Dyes Pigments 80:30–33
Das PJ, Das J (2015) Secondary amine based ionic liquid: an efficient catalyst for solvent free one pot synthesis of xanthenes and benzoxanthenes. RSC Adv 5:11745–11752
Dutta AK, Gogoi P, Borah R (2012) Synthesis of dibenzoxanthene and acridine derivatives catalyzed by 1,3-disulphonic acid imidazolium carboxylate ionic liquids. RSC Adv 4:41287–41291
Maleki B, Akbarzadeh E, Babaee S (2015) New basic ionic liquid from ethan-1,2-diylbis(hydrogen sulfate) and DBU (1,8- diazobicyclo[5.4.0]undec-7-ene) as an efficient catalyst for one-pot synthesis of xanthene derivatives. Dyes Pigments 123:222–234
Rahmati A (2010) A rapid and efficient method for the synthesis of 14H-dibenzo[a,j]xanthenes, aryl-5Hdibenzo[b.i]xanthene-5,7,12,14-(13H)-tetraones, and 1,8-dioxo-octahydroxanthenes by acidic ionic liquid. Chin Chem Lett 21:761–764
Rad-Moghadam K, Azimi SC (2012) Mg(BF4)2 doped in [BMIm][BF4]. A homogeneous ionic liquid-catalyst for efficient synthesis of 1,8-dioxo-octahydroxanthenes, decahydroacridines and 14-aryl-14-H-dibenzo[a, j]xanthenes. J Mol Catal A Chem 363–364:465–469
Wu H, Chen XM, Wan Y, Xin HQ, Xu HH, Yue CH, Ma R (2009) Synthesis and luminescence of 14-Aryl- or Alkyl-14H-dibenzo[a, j]xanthenes Catalyzed by 2–1′-Methylimidazolium-3-yl-1-ethyl Sulfate. Synth Commun 39:3762–3771
Khurana JM, Lumb A, Pandey A, Magoo D (2012) Green approaches for the synthesis of 12-aryl-8,9,10,12-tetrahydrobenzo[a]xanthen-11-ones in aqueous media and under microwave irradiation in solventless conditions. Synth Commun 42:1796–1803
Liu YH, Li L (2012) Methanesulfonic acid-catalyzed one-pot synthesis of 12-aryl- or 12-alkyl-8,9,10,12-tetrahydrobenzo[a]xanthen-11-one derivatives. J Heterocycl Chem 49:861–864
Wang HJ, Ren XQ, Zhang YY, Zhang ZH (2009) Synthesis of 12-aryl or 12-alkyl-8,9,10,12-tetrahydrobenzo[a]xanthen-11-one derivatives catalyzed by dodecatungstophosphoric acid. J Braz Chem Soc 20:1939–1943
Li JT, Li YW, Song YL (2012) Efficient synthesis of 12-aryl-8,9,10,12-tetrahydrobenzo[a]xanthen-11-one derivatives catalyzed by p-dodecylbenzenesulfonic acid in aqueous media under ultrasound irradiation. Synth Commun 42:2161–2170
Khurana JM, Magoo D (2009) pTSA-catalyzed one-pot synthesis of 12-aryl-8,9,10,12-tetrahydrobenzo[a]xanthen-11-ones in ionic liquid and neat conditions. Tetrahedron Lett 50:4777–4780
Rama V, Kanagaraj K, Pitchumani K (2012) A multicomponent, solvent-free, one-pot synthesis of benzoxanthenones catalyzed by HY zeolite: their anti-microbial and cell imaging studies. Tetrahedron Lett 53:1018–1024
Bahrami K, Khodaei MM, Roostaei M (2014) The preparation and characterization of boehmite nanoparticles-TAPC: a tailored and reusable nanocatalyst for the synthesis of 12-aryl-8,9,10,12-tetrahydrobenzo[a]xanthen-11-ones. New J Chem 38:5515–5520
Sadeghi B, Hassanabadi A, Taghvatalab E (2011) Nanoparticle silica supported sulfuric acid(NPs SiO2-H2SO4): a solid phase acidic catalyst for the one-pot synthesis of benzo[a]xanthen-11-one derivatives. J Chem Res 35:707–708
Maleki B, Barzegar S, Sepehr Z, Kermanian M, Tayebee R (2012) A novel polymeric catalyst for the one-pot synthesis of xanthene derivatives under solvent-free conditions. J Iran Chem Soc 9:757–765
Moghanian H, Mobinikhaledi A, Deinavizadeh M (2015) Efficient, one-pot synthesis of xanthene derivatives using boron sulphonic acid as a solid heterogeneous catalyst under solvent free conditions. Res Chem Intermed 41:4387–4394
Karimi N, Oskooie HA, Heravi MM, Tahershamsi L (2011) Caro’s acid-silica gel-catalyzed one-pot synthesis of 12-aryl-8,9,10,12- tetrahydrobenzo[a]xanthen-11-ones. Synth Commun 41:307–312
Heravi MM, Hashemi E, Beheshtiha YS, Kamjou K, Toolabi M, Hosseintash N (2014) Solvent-free multicomponent reactions using the novel N-sulfonic acid modified poly(styrene-maleic anhydride) as a solid acid catalyst. J Mol Catal A Chem 392:173–180
Vahdat S, Khaksar S (2015) Polyvinylpolypyrrolidone supported triflic acid (PVPP.OTf); an efficient and recyclable heterogeneous catalyst for one-pot condensation of b-naphthol, aldehydes, and cyclic 1,3-dicarbonyl compounds. Res Chem Intermed 41:4177–4186
Shaterian HR, Mohammadnia M (2013) Nanocrystalline TiO2-HClO4 catalyzed three-component preparation of derivatives of 1-amidoalkyl-2-naphthol, 1-carbamato-alkyl-2-naphthol, 1-(a-aminoalkyl)-2-naphthol, and 12-aryl-8,9,10,12-tetrahydrobenzo[a]-xanthen-11-one. Res Chem Intermed 39:4221–4237
Zhang ZH, Wang HJ, Ren XQ, Zhang YY (2009) A facile and efficient method for synthesis of xanthone derivatives catalyzed by HBF4/SiO2 under solvent-free conditions. Monatsh Chem 140:1481–1483
Chen W, Peng XW, Zhong LX, Li Y, Sun RC (2015) Lignosulfonic acid: a renewable and effective biomass-based catalyst for multicomponent reactions. ACS Sustain Chem Eng 3:1366–1373
Pawar PB, Jadhav SD, Deshmukh MB, Patil S (2014) Citric acid as a mild and inexpensive organocatalyst for synthesis of tetrahydrobenzo[a]xanthen-11-ones and dibenzo[a, j]xanthenes under solvent-free condition. Ind J Chem 14:1185–1193
Shaterian HR, Rigi F (2014) New applications of cellulose-SO3H as a bio-supported and biodegradable catalyst for the one-pot synthesis of some three-component reactions. Res Chem Intermed 40:2983–2999
Wan Y, Zhang XX, Wang C, Zhao LL, Chen LF, Liu GX (2013) The first example of glucose-containing Brønsted acid synthesis and catalysis: efficient synthesis of tetrahydrobenzo[a]xanthens and -tetrahydrobenzo[a]acridines in water. Tetrahedron 69:3947–3950
Ma J, Zhong L, Peng X, Sun R (2016) D-Xylonic acid: a solvent and an effective biocatalyst for a three-component reaction. Green Chem 18:1738–1750
Wang RZ, Zhang LF, Cui ZS (2009) Iodine-catalyzed synthesis of 12-aryl-8,9,10,12-tetrahydro-benzo[a]xanthen-11-one derivatives via multicomponent reaction. Synth Commun 39:2101–2107
Sun XJ, Zhou JF, Zhao PS (2011) Molecular iodine-catalyzed one-pot synthesis of tetrahydrobenzo[a]xanthene-11-one and diazabenzo[a]anthracene-9,11-dione derivatives under microwave irradiation. J Heterocycl Chem 48:1347–1350
Foroughifar N, Mobinikhaledi A, Moghanian H, Mozafari R, Esfahani HRM (2011) Ammonium chloride-catalyzed one-pot synthesis of tetrahydrobenzo[a]xanthen-11-one derivatives under solvent-free conditions. Synth Commun 41:2663–2673
Kidwai M, Mishra A, Jahan NK (2012) A novel method for the synthesis of tetrahydrobenzo[a]xanthen-11-one derivatives using cerium(III) chloride as a highly efficient catalyst. C R Chim 15:324–330
Nandi GC, Samai S, Kumar R, Singh MS (2009) An efficient one-pot synthesis of tetrahydrobenzo[a]xanthene-11-one and diazabenzo[a]anthracene-9,11-dione derivatives under solvent free condition. Tetrahedron 65:7129–7134
Li J, Tang W, Lu L, Su W (2008) Strontium triflate catalyzed one-pot condensation of b-naphthol, aldehydes and cyclic 1,3-dicarbonyl compounds. Tetrahedron Lett 49:7117–7120
Sharma RK, Khajuria R, Kapoor KK (2014) Alum-catalyzed domino synthesis of 12-substituted-8,9,10,12-tetrahydrobenzoxanthen-11-ones under Solvent-free conditions. Synth Commun 44:3538–3551
Tabatabaeian K, Khorshidi A, Mamaghani M, Dadashi A, Jalali MK (2011) One-pot synthesis of tetrahydrobenzo[a]xanthen-11-one derivatives catalyzed by ruthenium chloride hydrate as a homogeneous catalyst. Can J Chem 89:623–627
Mirjalili FBB, Bamoniri A, Zamani L (2012) Nano-TiCl4/SiO2: an efficient and reusable catalyst for the synthesis of tetrahydrobenzo[a]xanthen-11-ones. Lett Org Chem 9:338–343
Farhad Shirini F, Akbari-Dadamahaleh S, Mohammad-Khah A, Aliakbar AR (2013) Rice husk: a mild, efficient, green and recyclable catalyst for the synthesis of 12-Aryl-8, 9, 10, 12-tetrahydro[a]xanthene-11-ones and quinoxaline derivatives. C R Chim 16:207–216
Taghavi-Khorasani F, Davoodnia A (2015) A fast and green method for synthesis of tetrahydrobenzo[a]xanthene-11-ones using Ce(SO4)2·4H2O as a novel, reusable, heterogeneous catalyst. Res Chem Intermed 41:2415–2425
Oskooie HA, Heravi MM, Karimi N, Kohansal G (2011) Cu/SiO2-catalyzed one-pot synthesis of 12-aryl-8,9,10,12-tetrahydrobenzo[a]xanthen-11-ones under solvent-free conditions. Synth Commun 41:2763–2768
Kaur B, Parmar A, Kumar H (2012) Manganese Perchlorate-catalyzed greener synthesis of 12-aryl or 12-alkyl-8,9,10,12-tetrahydrobenzo[a]xanthen-11-one derivatives under ultrasonication. Synth Commun 42:447–453
Gao S, Tsai CH, Yao CF (2009) A simple and green approach for the synthesis of tetrahydrobenzo[a]-xanthen-11-one derivatives using tetrabutylammonium fluoride in water. Synlett 6:949–954
Akondi AM, Kantam ML, Trivedi R, Sreedhar B, Buddana SK, Prakasham RS, Bhargava S (2014) Formation of benzoxanthenones and benzochromenones via cerium-impregnated-MCM-41 catalyzed, solvent-free, three component reaction and their biological evaluation as anti-microbial agents. J Mol Catal A Chem 386:49–60
Mohammadi R, Eidi E, Ghavami M, Kassaee MZ (2014) Chitosan synergistically enhanced by successive Fe3O4 and silver nanoparticles as a novel green catalyst in one-pot, three component synthesis of tetrahydrobenzo[a]xanthene-11-ones. J Mol Catal A Chem 393:309–316
Khazaei A, Zolfigol MA, Moosavi-Zare AR, Zare A, Khojasteh M, Asgari Z, Khakyzadeh V, Khalafi-Nezhad A (2012) Organocatalyst trityl chloride efficiently promoted the solvent-free synthesis of 12-aryl-8,9,10,12-tetrahydrobenzo[a]-xanthen-11-ones by in situ formation of carbocationic system in neutral media. Catal Commun 20:54–57
Iniyavan P, Sarveswari S, Vijayakumar V (2015) Microwave-assisted clean synthesis of xanthenes and chromenes in [bmim][PF6] and their antioxidant studies. Res Chem Intermed 41:7413–7426
Fang D, Yang JM, Cao YF (2013) Synthesis of 12-aryl-8,9,10,12-tetrahydrobenzo[a]xanthene-11-ones catalyzed by biodegradable ionic liquid. Res Chem Intermed 39:1745–1751
Kundu D, Majee A, Hajra A (2011) Task-specific ionic liquid-catalyzed efficient microwave-assisted synthesis of 12-alkyl or aryl-8,9,10,12-tetrahydrobenzo [a]xanthen-11-ones ones under solvent-free conditions. Green Chem Lett Rev 4:205–209
Zolfigol MA, Khakyzadeh V, Moosavi-Zare AR, Zare A, Azimi SB, Asgari Z, Hasaninejad A (2012) Preparation of various xanthene derivatives over sulfonic acid-functionalized imidazolium salts (SAFIS) as novel, highly efficient and reusable catalysts. C R Chim 15:719–736
Singh H, Kumari S, Khurana JM (2014) A new green approach for the synthesis of 12-aryl-8,9,10,12-tetrahydrobenzo[a]xanthene-11-one derivatives using task specific acidic ionic liquid [NMP]H2PO4. Chin Chem Lett 25:1336–1340
Heydari R, Shahrekipour F (2015) One-pot synthesis of 12-aryl-8,9,10,12-tetrahydrobenzo[a]xanthen-11-ones by using of neutral and efficient organocatalysts under solvent-free conditions. Res Chem Intermed 41:4581–4586
Yadav S, Khurana JM (2013) An efficient synthesis of novel 3-hydroxy-12-arylbenzo[a]xanthen-11-ones and 5,12-diarylxantheno[2,1-a]xanthene-4,12-diones using pTSA in [bmim]BF4. Can J Chem 91:698–703
Khurana JM, Nand B, Sneha (2011) An efficient and convenient approach for the synthesis of novel 2-hydroxy-12-aryl-8,9,10,12-tetrahydrobenzo[a]xanthene-11-ones using p-toluenesulfonic acid in ethanol and ionic liquid. J Heterocycl Chem 48:1388–1392
Khurana JM, Chaudhary A, Lumb A, Nand B (2012) Efficient one-pot syntheses of dibenzo[a, i]xanthene-diones and evaluation of their antioxidant activity. Can J Chem 90:739–746
Wu L, Zhang J, Fang L, Yang C, Yan F (2010) Silica chloride catalyzed synthesis of 14-aryl-14H-dibenzo[a, i]xanthene-8,13-diones. Dyes Pigments 86:93–96
Rahmatpour A (2013) Polystyrene-supported GaCl3 as a highly efficient and reusable heterogeneous Lewis acid catalyst for the three-component synthesis of benzoxanthene derivatives. Monatsh Chem 144:1205–1212
Srinivas V, Rao VR (2012) Facile, one-pot, three-component synthesis of benzo[a]naphthacene-8,13-diones. Synth Commun 42:388–393
Liu D, Xu D, Gao J, Zhou S (2014) p-Toluenesulfonic acid catalyzed synthesis of 14-aryl-14H-dibenzo[a, i]xanthene-8,13-dioness. Chem Sci Trans 3:455–459
Ghasemzadeh MA, Azimi-Nasrabad M (2016) Nano-Fe3O4-encapsulated silica particles bearing sulfonic acid groups as a magnetically separable catalyst for the green and efficient synthesis of 14-aryl-14H-dibenzo[a, i]xanthene-8,13-dione derivatives. Res Chem Intermed 42:1057–1069
Safaei-Ghomi J, Eshteghal F (2017) Nano-Fe3O4/PEG/succinic anhydride: a novel and efficient catalyst for the synthesis of benzoxanthenes under ultrasonic irradiation. Ultrason Sonochem 38:488–495
Yang LM, Yin ZK, Wu LQ (2012) H4SiW12O40: an efficient catalyst for the synthesis of new spiro[dibenzo[a, i]xanthene-14,30-indoline]-20,8,13-triones. Chin Chem Lett 23:265–268
Yang X, Yang L, Wu L (2012) [Hmim][HSO4]: an efficient and reusable catalyst for the synthesis of spiro[dibenzo[a, i] xanthene-14,3′-indoline]-2′,8,13-triones and spironaphthopyran[2,3-d]pyrimidine-5,3′-indolines. Bull Korean Chem Soc 33:714–716
Kefayati H, Bazargard SJ, Vejdansefat P, Shariati S, Kohankar AM (2016) Fe3O4@MCM-41-SO3H@[HMIm][HSO4]: an effective magnetically separable nanocatalyst for the synthesis of novel spiro[benzoxanthene-indoline]diones. Dyes Pigm 125:309–315
Mohr SJ, Chirigos MA, Fuhrman FS, Pryor JW (1975) Pyran copolymer as an effective adjuvant to chemotherapy against a murine leukemia and solid tumor. Cancer Res 35:3750–3754
Dell C, Smith C Antiproliferative derivatives of 4H-naphtho [1, 2-b] Pyran and process for their preparation. EP537949. 1993:21
Smith PW, Sollis SL, Howes PD, Cherry PC, Starkey ID, Cobley KN, Weston H, Scicinski J, Merritt A, Whittington A (1998) Dihydropyrancarboxamides related to zanamivir: a new series of inhibitors of influenza virus sialidases. J Med Chem 41:787–789
Kumar D, Reddy VB, Sharad S, Dube U, Kapur S (2009) A facile one-pot green synthesis and antibacterial activity of 2-amino-4H-Pyrans and 2-amino-5-oxo-5,6,7,8-tetrahydro-4H-chromenes. Eur J Med Chem 44:3805–3809
Chattapadhyay TK, Dureja PJ (2006) Antifungal activity of 4-methyl-6-alkyl-2H-pyran-2-ones. J Agric Food Chem 54:2129–2133
Mineeva IV (2015) Cyclopropanol methodology in the synthesis of (4R)- and (4S)-4-methyltetrahydro-2H-pyran-2-ones. Application in the synthesis of insect pheromones with methyl-branched carbon skeleton. Russ J Org Chem 51:341–351
Bianchi G, Tava A (1987) Synthesis of (2R)-(+)-2,3-Dihydro-2,6-dimethyl-4H-Pyran-4-one, a homologue of pheromones of a species in the Hepialidae family. Agric Biol Chem 51:2001–2002
Armesto D, Horspool WM, Martin N, Ramos A, Seoane C (1989) Synthesis of cyclobutenes by the novel photochemical ring contraction of 4-substituted 2-amino-3,5-dicyano-6-phenyl-4H-Pyrans. J Org Chem 54:3069–3072
Awuah SG, Polreis J, Prakash J, Qiao Q, You Y (2011) New pyran dyes for dye-sensitized solar cells. J Photochem Photobio A Chem 224:116–122
Ahmed MM, El-Saghier MB, Naili BK, Rammash NA, Saleh Khaled MK (2007) Synthesis and antibacterial activity of some new fused chromenes. ARKIVOC 16:83–91
Tilak R, Richa KB, Rakesh KS, Vivek G, Deepak S, Mohan PSI (2009) Mechanism of unusual formation of 3-(5-phenyl-3H-[1,2,4]dithiazol-3yl)chromen-4-ones and 4-oxo-4H-chromene-3-carbothioic acid N-phenylamides and their antimicrobial evaluation. Eur J Med Chem 44:3209–3216
Okram MS, Nepram SD, Dhanaraj ST, Gurumayum JS (2010) Novel 3alkanoyl/aroyl/heteroaroyl-2H-chromene-2-thiones: synthesis and evaluation of their antioxidant activities. Eur J Med Chem 45:2250–2257
Kumar A, Maurya RA, Sharma S, Ahmad P, Singh AB, Bhatia G, Srivastava AK (2009) Pyranocoumarins: a new class of anti-hyperglycemic and anti-dyslipidemic agents. Bioorg Med Chem Lett 19:6447–6451
Mehrabi H, Kamali N (2012) Efficient and eco-friendly synthesis of 2-amino-4H-chromene derivatives using catalytic amount of tetrabutylammonium chloride (TBAC) in water and solvent-free conditions. J Iran Chem Soc 9:599–605
Eshghi H, Damavandi S, Zohuri GH (2011) Efficient one-pot synthesis of 2-amino-4H-chromenes catalyzed by ferric hydrogen sulfate and Zr-Based Catalysts of FI. Synth React Inorg Met Org Nano-Met Chem 41:1067–1073
Meng XY, Wang HJ, Wang CP, Zhang ZH (2011) Disodium hydrogen phosphate as an efficient and cheap catalyst for the synthesis of 2-aminochromenes. Synth Commun 41:3477–3484
Shekhar AC, Kumar AR, Sathaiah G, Raju K, Rao PS, Sridhar M, Sridhar B (2012) An efficient one-pot synthesis of substituted 1h-naphtho[2,1-b]pyrans and 4H-1-Benzopyrans (Chromenes) under solvent-free microwave-irradiation conditions. Helv Chim Acta 95:502–508
Sadjadi S, Heravi MM, Zadsirjan V, Ebrahimizadeh M (2017) SBA-15@methenamine-HPA: a novel, simple, and efficient catalyst for one-pot three-component synthesis of 2-amino-4H-chromene derivatives in aqueous medium. Res Chem Intermed 43:5467–5483
Sadeghi B, Zarepour I (2015) Nano-sawdust–BF3 as a new, cheap, and effective nano catalyst for one-pot synthesis of 2-amino benzo[h]chromene derivatives. J Nanostruct Chem 5:305–311
Sunil Kumar B, Srinivasulu N, Udupi RH, Rajitha B, Thirupathi Reddy Y, Narsimha Reddy P, Kumar PS (2006) Efficient synthesis of benzo[g]-and benzo[h]chromene derivatives by one-pot three-component condensation of aromatic aldehydes with active methylene compounds and naphthols. Russ J Org Chem 42:1813–1815
Balalaie S, Ramezanpour S, Bararjanian M, Gross JH (2008) DABCO-catalyzed efficient synthesis of naphthopyran derivatives via one-pot three-component condensation reaction at room temperature. Synth Commun 38:1078–1089
Heravi MM, Hosseinnejad T, Faghihi Z, Shiri M, Vazinfard M (2017) Synthesis of 2-amino-3-cyano 4-H-chromenes containing quinoline in water: computational study on substituent effects. J Iran Chem Soc 14:823–832
Mobinikhaledi A, Moghanian H, Sasani F (2011) Synthesis and reactivity in inorganic, microwave-assisted one-pot synthesis of 2-amino-2-chromenes using piperazine as a catalyst under solvent-free conditions. Synth React Inorg Met Org Nano-Met Chem 41:262–265
Dekamin MG, Eslami M (2014) Highly efficient organocatalytic synthesis of diverse and densely functionalized 2-amino-3-cyano-4H-pyrans under mechanochemical ball milling. Green Chem 16:4914–4921
Dekamin MG, Alikhani M, Javanshir S (2016) Organocatalytic clean synthesis of densely functionalized 4H-pyrans by bifunctional tetraethylammonium 2-(carbamoyl)benzoate using ball milling technique under mild conditions. Green Chem Lett Rev 9:96105
Kundu SK, Bhaumik A (2015) A triazine-based porous organic polymer: a novel heterogeneous basic organocatalyst for facile one-pot synthesis of 2-amino-4H-chromenes. RSC Adv 5:32730–32739
Saha A, Payra S, Banerjee S (2015) On water synthesis of pyran–chromenes via a multicomponent reactions catalyzed by fluorescent t-ZrO2 nanoparticles. RSC Adv 5:101664–101671
Albadi J, Alihoseinzadeh A, Mansournezhad A, Kaveiani L (2015) Novel metal oxide of CuO–ZnO nanocatalyst efficiently catalyzed the synthesis of 2-amino-4H-chromenes in water. Synth Commun 45:485–493
Tajbakhsh M, Kariminasab M, Alinezhad H, Hosseinzadeh R, Rezaee P, Tajbakhsh M, Gazvini HJ, Azizi Amiri M (2012) Nano silicabonded aminoethylpiperazine: a highly efficient and reusable heterogeneous catalyst for the synthesis of 4Hchromene and 12Hchromeno[2,3d]pyrimidine derivatives. J Iran Chem Soc 12:1405–1414
Karmakar B, Nandi R (2016) A green route towards substituted 2-amino-4H-chromenes catalyzed by an organobase (TBD) functionalized mesoporous silica nanoparticle without heating. Res Chem Intermed. https://doi.org/10.1007/s11164-016-2755-9
Maleki B, Sheikh S (2015) Nano polypropylenimine dendrimer (DAB-PPI-G1): as a novel nano basic-polymer catalyst for one-pot synthesis of 2-amino-2-chromene derivatives. RSC Adv 5:42997–43005
Jiang L, Yu H (2014) Enzymatic promiscuity: “Amano” lipase AS-catalysed synthesis of naphthopyran derivatives in anhydrous media. Chem Res Chin 30:396–399
Shinde S, Damate S, Morbale S, Patil M, Patil SS (2017) Aegle marmelos in heterocyclization: greener, highly efficient, one-pot three-component protocol for the synthesis of highly functionalized 4H-benzochromenes and 4H-chromenes. RSC Adv 7:7315–7328
Qareaghaj OH, Mashkouri S, Naimi-Jamal MR, Kaupp G (2014) Ball milling for the quantitative and specific solvent-free Knoevenagel condensation + Michael addition cascade in the synthesis of various 2-amino-4-aryl-3-cyano-4H-chromenes without heating. RSC Adv 4:48191–48201
Naimi-Jamal MR, Mashkouri S, Sharifi A (2010) An efficient, multicomponent approach for solvent-free synthesis of 2-amino-4H-chromene scaffold. Mol Div 14:473–477
Sheibani H, Saidi K, Abbasnejad M, Derakhshani A, Mohammadzadeh I (2016) A convenient one-pot synthesis and anxietic activity of 3-cyano-2(1H)iminopyridines and halogen derivatives of benzo[h]chromenes. Arabian J Chem 9:S901–S906
Wang X-S, Shi DQ, Yu HZ, Wang GF, Tu SG (2004) Synthesis of 2aminochromene derivatives catalyzed by KF/Al2O3. Synth Commun 34:509–514
Albadi J, Mansournezhad A, Darvishi-Paduk M (2013) Poly(4-vinylpyridine): as a green, efficient and commercial available basic catalyst for the synthesis of chromene derivatives. Chin Chem Lett 24:208–210
Shaterian HR, Mohammadnia M (2013) Mild preparation of 2-amino-3-cyano-4-aryl-4H-benzo[h]chromenes and 2-amino-3-cyano-1-aryl-1H-benzo[f]chromenes, under solvent-free conditions, catalyzed by recyclable basic ionic liquids. Res Chem Intermed 41:1301–1313
Shaikh MA, Farooqui M, Abed S (2019) Novel task-specific ionic liquid [Et2NH(CH2)2CO2H][AcO] as a robust catalyst for the efficient synthesis of some pyran-annulated scaffolds under solvent-free conditions. Res Chem Intermed 45:1595–1617
Warekar PP, Patil PT, Patil KT, Jamale DK, Kolekar GB, Anbhule PV (2017) PTSA-catalyzed straightforward novel approach for the synthesis of 1,2-bis(4-nitrophenyl)-1H-benzo[f]chromen-3-amine and the evaluation of their antituberculosis activity. Res Chem Intermed 43:4115–4127
Olyaei A, Shahsavari MS, Sadeghpour M (2017) Organocatalytic approach toward the green one-pot synthesis of novel benzo[f]chromenes and 12H-benzo[5,6]chromeno[2,3-b]pyridines. Res Chem Intermed 44:943–956
Hosseinnia R, Mamaghani M, Tabatabaeian K, Shirini F, Rassa M (2012) An expeditious regioselective synthesis of novel bioactive indole-substituted chromene derivatives via one-pot three-component reaction. Bioorg Med Chem Lett 22:5956–5960
Wan Y, Wang C, Wang H, Zhao L, Zhang X, Shi J, Wu H (2014) Efficient One-Pot Syntheses of 7-Alkyl-6H,7H-naphtho[1,2:5,6]pyrano-[3,2-c]chromen-6-ones by 1-Methyl-3-(2-(sulfooxy)ethyl)-1H-imidazol-3-ium Chloride. J Heterocycl Chem 51:1293–1297
Ma W, Wang X, Yan F, Wu L, Wang Y (2010) Reusable melamine trisulfonic acid-catalyzed three-component synthesis of 7-alkyl-6H,7H-naphtho[1′,2′:5,6]pyrano[3,2-c]chromen-6-ones. Monatsh Chem 142:163–167
Nikpassand M, Leila ZF, Zahra G, Jafarian Z (2018) Potassium 2-oxoimidazolidine-1,3-diide as a novel catalyst for grind synthesis of pyrano[4,3-b]chromenone. J Chil Chem Soc 63:41965–44199
Bhattacharjee S, Gattu R, Khan AT (2018) Triethylamine-mediated one-pot synthesis of benzo[f]chromene derivatives. ChemistrySelect 3:4760–4763
Mashraqui SH, Patil MB, Mistry HD, Ghadigaonkar S, Meetsma A (2004) A three-component reaction of phenol, aldehyde, and active methylene substrate under lewis acid catalysis: successful trapping ofo-quinone methide to afford benzopyran systems. Chem Lett 33:1058–1059
Devi KM, Chanu LG, Chanu IS, Singh OM (2014) One-pot synthesis of 1H-naphtho[2,1-b]pyran derivatives under solvent- free conditions. Lett Org Chem 11:743–747
Kumar KP, Satyanarayana S, Reddy PL, Narasimhulu G, Ravirala N, Reddy BVS (2012) Iodine-catalyzed three-component one-pot synthesis of naphthopyranopyrimidines under solvent-free conditions. Tetrahedron Lett 53:1738–1741
Mohaqeq M, Safaei-Ghomi J, Shahbazi-Alavi H (2015) ZrOCl2/nano TiO2 as an efficient catalyst for the one pot synthesis of naphthopyranopyrimidines under solvent-free conditions. Acta Chim Slov 62:967–972
Khurana JM, Lumb A, Chaudhary A, Nand B (2013) Synthesis and in vitro evaluation of antioxidant activity of diverse naphthopyranopyrimidines, diazaanthra[2,3-d][1,3]dioxole-7,9-dione and tetrahydrobenzo[a]xanthen-11-ones. RSC Adv 3:1844–1854
Singh M, Nandi G, Samai S (2010) First InCl3-catalyzed, three-component coupling of aldehydes, β-naphthol, and 6-amino-1,3-dimethyluracil to functionalized naphthopyranopyrimidines. Synlett 07:1133–1137
Cimarellin C, Fratoni D, Palmier G (2011) Novel stereoselective synthesis of 2,3-dihydro-1H-benzo[f]chromen-3-amine derivatives through a one-pot three component reaction. Tetrahedron Asymmetry 22:1542–1547
Nizami TA, Hua R (2018) Synthesis of 3 H -naphtho[2.1- b]pyran-2-carboxamides from cyclocoupling of β-naphthol, propargyl alcohols and isocyanide in the presence of Lewis acids. Tetrahedron 74:3776–3780
Yadav JS, Subba Reddy BV, Biswas SK, Sengupta S (2009) Gallium(III) chloride-catalyzed three-component coupling of naphthol, alkyne and aldehyde: a novel synthesis of 1,3-disubstituted-3H-benzo[f] chromenes. Tetrahedron Lett 50:5798–5801
Asadi S, Mohammadi Ziarani G, Rahimifard M, Abolhassani Soorki A (2014) A green one-pot synthesis of spiron/aphthopyrano[1,2-b]indeno-7,3′-indolines. Res Chem Intermed 41:6219–6227
Kong D, Lu G, Wu M, Shi Z, Li Q (2017) One-pot, catalyst-free synthesis of spiro[dihydroquinoline-naphthofuranone] compounds from isatins in water triggered by hydrogen bonding effects. ACS Sustain Chem Eng 5:3465–3470
Mohammadi Ziarani G, Lashgari N, Faramarzi S, Badiei A (2014) Efficient synthesis of spironaphthopyrano [2,3-d]pyrimidine-5,3′-indolines under solvent-free conditions catalyzed by SBA-Pr-SO3H as a nanoporous acid catalyst. Acta Chim Slov 61:574–579
Heravi M, Zakeri M, Moharami A (2012) Versatile three-component procedure for combinatorial synthesis of spiro-oxindoles with fused chromenes catalysed by l-proline. J Chem Sci 124:865–869
Shanthi G, Subbulakshmi G, Perumal PT (2007) A new InCl3-catalyzed, facile and efficient method for the synthesis of spirooxindoles under conventional and solvent-free microwave conditions. Tetrahedron 63:2057–2063
Heravi MR, Norouzy F (2017) One-pot, four-component synthesis of novel biologically important indeno[2,3-b]quinoxaline]-3-carbonitriles in fluoro-alcohols under ultrasound irradiation. Res Chem Intermed 43:4265–4282
Khojasteh-Khosro S, Shahbazi-Alavi H (2019) Preparation of spirooxindoles catalyzed by nano-Co3S4 under microwave irradiations. J Chem Res 43:107–111
Mathew BP, Kumar A, Sharma S, Shukla PK, Nath M (2010) An eco-friendly synthesis and antimicrobial activities of dihydro-2H-benzo- and naphtho-1,3-oxazine derivatives. Eur J Med Chem 45:1502–1507
Zanatta N, Squizani AMC, Fantinel L, Nachtigall FM, Borchhardt DM, Bonacorso HG, Martins MAP (2005) Synthesis of N-substituted 6-Trifluoromethyl-1,3-oxazinanes. J Braz Chem Soc 16:1255–1261
Zhang P, Terefenko EA, Fensome A, Wrobel J, Winneker R, Zhang Z (2003) Novel 6-Aryl-1,4-dihydrobenzo[d][1,3]oxazine-2-thiones as potent, selective, and orally active nonsteroidal progesterone receptor agonists. Bioorg Med Chem Lett 13:1313–1316
Kuehne ME, Konopka A (1962) Dihydro-1,3-oxazines as antitumor agents. J Med Chem 5:257–280
Azizian J, Yadollahzadeh K, Delbari AS, Ghanbari MM (2012) An efficient Biginelli one-pot synthesis of new naphthalene-condensed oxazine derivatives under microwave-assisted conditions. Monatsh Chem 143:1417–1420
Ghomi JS, Zahedi S, Ghasemzadeh MA (2012) An efficient green route for the preparation of naphthoxazinones, applying a three-component one-pot condensation reaction under solvent-free conditions. Iran J Catal 2(1):27–30
Kumar A, Saxena A, Dewan M, De A, Mozumdar S (2011) Recyclable nanoparticulate copper mediated synthesis of naphthoxazinones in PEG-400: a green approach. Tetrahedron Lett 52(38):4835–4839
Dharma Rao GB, Kaushik MP, Halve AK (2012) An efficient synthesis of naphtha[1,2-e]oxazinone and 14-substituted-14H-dibenzo[a, j]xanthene derivatives promoted by zinc oxide nanoparticle under thermal and solvent-free conditions. Tetrahedron Lett 53:2741–2744
Safaei Ghomi J, Zahedi S, Ghasemzadeh MA (2014) AgI nanoparticles as a remarkable catalyst in the synthesis of (amidoalkyl)naphthol and oxazine derivatives: an eco-friendly approach. Monatsh Chem 145:1191–1199
Salunkhe NG, Ladole CA, Thakare NV, Aswar AS (2017) MgFe2O4@SiO2–SO3H: an efficient, reusable catalyst for the microwave-assisted synthesis of benzoxazinone and benzthioxazinone via multicomponent reaction under solvent free condition. Res Chem Intermed 44:355–372
Basavegowda N, Somai Magar KB, Mishra K, Lee YR (2014) Green fabrication of ferromagnetic Fe3O4nanoparticles and their novel catalytic applications for the synthesis of biologically interesting benzoxazinone and benzthioxazinone derivatives. New J Chem 38:5415–5420
Bazgir A, Dabiri M, Delbari A (2007) A novel three-component, one-pot synthesis of 1,2-dihydro-1-aryl-naphtho[1,2-e][1,3]oxazine-3-one derivatives under microwave-assisted and thermal solvent-free conditions. Synlett 5:0821–0823
Sharma M, Manohar S, Rawat DS (2012) Lewis acid catalyzed synthesis of 1-aryl-1,2-dihydro-naphtho[1,2-e][1,3]oxazin-3-ones under solvent free conditions: a mechanistic approach. J Heterocycl Chem 49:589–595
Kantevari S, Vuppalapati SVN, Bantu R, Nagarapu L (2010) An efficient one-pot three component synthesis of 1,2-dihydro-1-arylnaphtho[1,2-e][1,3]oxazine-3-ones using montmorillonite k10 under solvent free conditions. J Heterocycl Chem 47:313–317
Hajra A, Kundu D, Majee A (2009) An efficient one-pot synthesis of naphthoxazinones by a three-component coupling of naphthol, aldehydes, and urea catalyzed by zinc triflate. J Heterocycl Chem 46:1019–1022
Chaskar A, Vyavhare V, Padalkar V, Phatangare K, Deokar H (2011) An environmentally benign one-pot synthesis of 1,2-dihydro-1-arylnaphtho[1,2-e][1,3]oxazine-3-one derivatives catalysed by phosphomolybdic acid. J Ser Chem Soc 76:21–26
Harichandran G, Parameswari P, Shanmugam P (2018) A one-pot multicomponent synthesis of naphthoxazin-3-one derivatives using Amberlite IRA-400 Cl resin as green catalyst. Lett Org Chem 15:600–605
Gupta A, Kour D, Gupta VK, Kapoor KK (2016) Graphene oxide mediated solvent-free three component reaction for the synthesis of 1-amidoalkyl-2-naphthols and 1,2-dihydro-1-arylnaphth[1,2- e][1,3]oxazin-3-ones. Tetrahedron Lett 57:4869–4872
Sabitha G, Arundhathi K, Sudhakar K, Sastry BS, Yadav JS (2010) A novel three-component one-pot reaction involving β-naphthol, aldehydes, and urea promoted by TMSCl/NaI. J Heterocycl Chem 47:272–275
Dong F, Yang Li-fang, Jin-ming Y (2012) Synthesis of 1,2-dihydro-1-arylnaphtho[1,2-e][1,3]oxazine-3-one catalyzed by pyridinium-based ionic liquid. Res Chem Intermed 39:2505–2512
Zhu X, Lee YR (2012) RuCl2(PPh3)3-catalyzed facile one-pot synthesis of 1,2-dihydro-1-arylnaphtho[1,2-e][1,3]oxazine-3-ones and 1,2-dihydro-1-arylnaphtho[1,2-e][1,3]oxazine-3-thiones. Bull Korean Chem Soc 33:3831–3834
Kumar A, Gupta MK, Kumar M (2012) Micelle promoted supramolecular carbohydrate scaffold-catalyzed multicomponent synthesis of 1,2-dihydro-1-aryl-3H-naphth[1,2-e][1,3]oxazin-3-one and amidoalkyl naphthols derivatives in aqueous medium. RSC Adv 2:7371
Lei M, Ma L, Hu L (2011) Highly chemoselective condensation of β-naphthol, aldehyde, and urea catalyzed by thiamine hydrochloride. Synth Commun 41:3424–3432
Shafiee M, Khosropour AR, Mohammadpoor-Baltork I, Moghadam M, Tangestaninejad S, Mirkhani V, Khavasi HR (2012) Synthesis of trans-1,3-diaryl-2-(5-methylisoxazol-3-yl)-2,3-dihydro-1H-naphtho[1,2-e][1,3]oxazines via bismuth(III)-catalyzed one-pot pseudo-four component reaction. Mol Div 16:727–735
Turgut Z, Pelit E, Köycü A (2007) Synthesis of new 1,3-disubstituted-2,3-dihydro-1H-naphth[1,2-e][1,3]oxazines. Molecules 12:345–352
Dutta AK, Gogoi P, Saikia MP, Borah R (2016) Development of environmentally benign methods towards the synthesis of anti-2,3-dihydro-1,2,3-trisubstituted-1H-naphth[1,2-e][1,3]oxazines using brønsted acidic catalysts. Catal Lett 146:902–908
Dutta AK, Gogoi P, Borah R (2017) Triphenylsulfophosphonium chlorometallates as efficient heterogeneous catalysts for the three-component synthesis of 2,3-dihydro-1,2,3-trisubstituted-1H-naphth[1,2-e][1,3]oxazines. Polyhedron 123:184–191
Sapkal SB, Shelke KF, Shingate BB, Shingare MS (2010) An efficient one-pot strategies for the synthesis of [1,3] oxazine derivatives. J Kor Chem Soc 54:437–442
Lohar T, Mane A, Kamat S, Salunkhe R (2018) A versatile water-stable fluorine containing organometallic lewis acid used in green synthesis of 1,3-oxazine scaffolds at room temperature. Polycyclc Aromat Comp. https://doi.org/10.1080/10406638.2018.1538057
Azad S, Mirjalili BBF (2018) One-pot solvent-free synthesis of 2,3-dihydro-2-substituted-1H-naphtho[1,2-e][1,3]oxazine derivatives using Fe3O4@nano-cellulose/TiCl as a bio-based and recyclable magnetic nano-catalyst. Mol Diver 23:413–420
Reddy MV, Lim KT, Kim JT, Jeong YT (2012) Ultrasound-assisted one-pot synthesis of 1,3-oxazine derivatives catalysed by BF3–SiO2 under neat conditions. J Chem Res 36:398–401
Dhakane VD, Gholap SS, Deshmukh UP, Chavan HV, Bandgar BP (2014) An efficient and green method for the synthesis of [1,3]oxazine derivatives catalyzed by thiamine hydrochloride (VB1) in water. C R Chim 17:431–436
Ganesan S, Rajendran N, Sundarakumar S, Ganesan A, Pemiah B (2013) β-Naphthol in glycerol: a versatile pair for efficient and convenient synthesis of aminonaphthols, naphtho-1,3-oxazines, and benzoxanthenes. Synthesis 45:1564–1568
Nongrum R, Kharkongor M, Nongthombam GS, Rani JWS, Rahman N, Kharmawlong GK, Nongkhlaw R (2019) [1,3]oxazines: green synthesis by sonication using a magnetically-separable basic nano-catalyst and investigation of its activity against the toxic effect of a pesticide on the morphology of blood cells. Environ Chem Lett 17:1325–1331
Sadaphal SA, Sonar SS, Shingate BB, Shingare MS (2010) Water mediated synthesis of various [1,3]oxazine compounds using alum as a catalyst. Green Chem Lett Rev. 3:213–216
Mohebat R, Mojahedi A, Yazdani-Elah-Abadi A (2018) Synthesis of 1,3-Oxazine-4-thione derivatives through an efficient, rapid and green method catalyzed by l-proline in aqueous medium. Org Prep Proced Int 50:424–431
Lukevits E, Demicheva L (1993) Biological activity of furan derivatives. Chem Heterocycl Compd 29:243–267
Vahabinia HR, Karami B, Khodabakhshi S (2013) One-pot synthesis of benzamidonaphtho[2,1-b]furans and benzamidobenzo[b]furans as novel polycyclic heteroaromatic compounds. J Chin Chem Soc 60:1323–1327
Karami B, Khodabakhshi S, Hashemi F (2003) Synthesis of a novel class of benzofurans via a three-component, regiospecific intramolecular heterocylization reaction. Tetrahedron Lett 54:358–3583
Satoh T, Tsuda T, Kushino Y, Miura M, Nomura M (1996) Synthesis of naphthofuran-2(3H)-one derivatives by palladium-catalyzed three-component coupling using naphthols, aldehydes, and carbon monoxide. J Org Chem 61:6476–6477
Yavari I, Anary-Abbasinejad M, Hossaini Z (2003) Reaction between naphthols and dimethyl acetylenedicarboxylate in the presence of phosphites. Synthesis of stable oxa-2λ5-phosphaphenanthrenes, and benzochromene derivatives. Org Biomol Chem 1:560–564
Reza Naimi-Jamal M, Mirzaei M, Bolourtchian M, Sharifi A (2006) One-pot solvent-free preparation of 2-phenyl-1,3,2-aryldioxaborins on acidic alumina. Synth Commun 36:2711–2717
Acknowledgements
Declared none.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chaudhary, A. Recent development in the synthesis of heterocycles by 2-naphthol-based multicomponent reactions. Mol Divers 25, 1211–1245 (2021). https://doi.org/10.1007/s11030-020-10076-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-020-10076-4