Abstract
Liver cancer is the sixth common cancer and forth cause of cancer-related death worldwide. Based on usually advanced stages of hepatocellular carcinoma (HCC) at the time of diagnosis, therapeutic options are limited and, in many cases, not effective, and typically result in the tumor recurrence with a poor prognosis. Radioimmunotherapy (RIT) offers a selective internal radiation therapy approach using beta or alpha emitting radionuclides conjugated with tumor-specific monoclonal antibodies (mAbs), or specific selective peptides. When compared to chemotherapy or radiotherapy, radiolabeled mAbs against cancer-associated antigens could provide a high therapeutic and exclusive radiation dose for cancerous cells while decreasing the exposure-induced side effects to healthy tissues. The recent advances in cancer immunotherapy, such as blockade of immune-checkpoint inhibitors (ICIs), has changed the landscape of cancer therapy, and the efficacy of different classes of immunotherapy has been tested in many clinical trials. Taking into account the use of ICIs in the liver tumor microenvironment, combined therapies with different approaches may enhance the outcome in the future clinical studies. With the development of novel immunotherapy treatment options in the recent years, there has been a great deal of information about combining the diverse treatment modalities to boost the effectiveness of immunomodulatory drugs. In this opinion review, we will discuss the recent advancements in RIT. The current status of immunotherapy and internal radiotherapy will be updated, and we will propose novel approaches for the combination of both techniques.
Graphical abstract
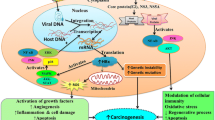
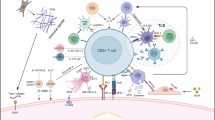
Potential target antigens for radioimmunotherapy in Hepatocellular carcinoma (HCC). HCC radioimmunotherapy target antigens are the most specific and commonly accessible antigens on the surface of HCC cells. CTLA-4 ligand and receptor, TAMs, PD-1/PD-L, TIM-3, specific IEXs/TEXs, ROBO1, and cluster of differentiation antigens CD105, CD147 could all be used in HCC radioimmunotherapy. Abbreviations: TAMs, tumor-associated macrophages; CTLA-4, cytotoxic T-lymphocyte associated antigen-4; PD-1, Programmed cell death protein 1; PD-L, programmed death-ligand1; TIM-3, T-cell immunoglobulin (Ig) and mucin-domain containing protein-3; IEXs, immune cell-derived exosomes; TEXs, tumor-derived exosomes.

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Radioimmunotherapy is an effective and selective internal radiation therapy. To avoid the off-target effects, radionuclides are attached to tumor-directed monoclonal antibodies
Hepatocellular carcinoma (HCC) is the sixth common cancer and the forth cause of cancer-related death worldwide [1]. Since the end of the last century, radioimmunotherapy (RIT) using a combination of intra-tumoral/intravenous radionuclide therapy and specific antigen targeting molecules has attracted a great interest [2, 3]. Nowadays, thanks to the RIT advancement, new methods for the adjustment of the absorbed radiation dose have been developed, both for cancerous and healthy tissues. They can be considered a basis for a personalized treatment of cancer and reduction of the toxicity of the extra-radiation exposure [4]. The timeline for the development of targeted therapeutics in HCC is depicted in Fig. 1.
Timeline of hepatocellular carcinoma targeted therapies. The first RIT application occurred about 1980, after the development of radiotherapy in 1941. In 2006, a breakthrough in the field of medical oncology was made with the creation of targeted radioimmunotherapy for the treatment of HCC. The results of the clinical trial, which indicated sorafenib's efficacy in HCC were presented in 2007. Seventeen therapies have been authorized since 2011. The US Food and Drug Administration (FDA) approved a combination of nivolumab 1 mg/kg and ipilimumab 3 mg/kg to treat patients with HCC who have previously been treated with sorafenib on March 10, 2020. Several medications that were approved as monotherapies would eventually be combined with radionuclides, resulting in improved clinical outcomes
Antigen specific radiolabeled monoclonal antibodies (mAbs) can release a high therapeutic radiation dose to cancerous cells while minimizing the exposure-associated side effects to healthy cells by their selective tropism to cancer-associated antigens on tumor cells. The striking and durable clinical responses in some patients [4, 5] resulted in a mainstream attention to this approach. RIT offers significant benefits including a favorable safety profile, rapid and durable response, and selective effects [6].
The development of immune-checkpoint inhibitors (ICIs) was a clinical breakthrough which substantially modified the paradigm of the cancer treatment. Interestingly, the recent application of targeted therapies against oncogenic drivers significantly enhanced the survival of non-small cell lung cancer (NSCLC) patients with a favorable toxicity profile [7]. Likewise, a meta-analysis confirmed the superiority of ICIs over docetaxel in pretreated NSCLC patients and indicated a slight benefit from anti-PD-1 than from anti-PD-L1 inhibitors [8]. The T-cell exhaustion is a state of T-cell failure caused by the current treatment strategies that restrict the T-cell activation. Numerous studies have shown that immune tolerance plays a role in the development of HCC, implying that ICIs inhibition could be a useful therapy approach. Collectively, the recent evidence has shown that immunotherapies improve the survival and are safe, but their effects are limited. Recently, the novel approach of ICIs combining with other medications is introduced to counteract the tumor-induced immunosuppression. Given that there is a lack of evidence regarding the various RIT combinations in HCC, in the current review we have discussed the recent advancements in RIT, which are being explored, and their outcomes are awaited [9].
Radioimmunotherapy approaches in hepatocellular carcinoma
Whereas the previously described approaches offer novel opportunities for the treatment of HCC, single-agent immunotherapy does not provide efficient patient outcomes. Since immunosuppressive components and factors such as Tregs, TAMs, and IL-35 are up-regulated in the liver, it has a higher physiological immune tolerance to antigens than other organs do. Another rationale is that various pathways involved in the growth of HCC produce an immunologically tolerogenic microenvironment for the mutant hepatocytes’ proliferation, leading to the resistance to single-agent immunotherapies. Furthermore, most immunotherapies have severe dosage-dependent adverse effects, including rash, diarrhea, pulmonary edema, and a cytokine storm, which become apparent when the dose is increased [10]. Thus, integrating different immunotherapies with classic therapeutic techniques is an important avenue of the anti-HCC therapy [11].
The remarkable therapeutic advantages of RIT are based on the specific targeting of tumor cells rather than noncancerous tissues. It is expected that radiation focuses only on the tumor, however this ideal can never be achieved practically due to the bystander effect [4]. Trans-arterial radioembolization is a catheter-directed internal radiation approach that delivers radionuclides directly into the tumor [12]. In addition, RIT is administered over a matter of minutes and delivers the radiation payload over a timescale of days, during which the patient does not need to return for additional injections [4]. Moreover, the interplay between the radiation effects and the immune system have been investigated and many studies have shown the synergistic effects on local and distant tumors when radiation therapy is combined with immunotherapy. The growing clinical enthusiasm for this approach is strengthened by many ongoing trials combining immunotherapy with definitive and palliative radiation.
The liver is a radiosensitive organ, which makes the liver toxicity a major concern in combined treatments [3]. To minimize the liver toxicity associated with a combined treatment, precise radiation dose adjustment for both treatments is crucial [13].
Radionuclides
In RIT, radionuclides are the cytotoxic agents which are conjugated to specific antibodies as a guide to selectively destroy tumor cells with their ionizing emissions. Radionuclides are usually selected according to their radio-physical properties such as the type of emission, energy level and half-life as well as labeling chemistry. The tumor response depends on multiple characteristics of radionuclides such as the dose rate, cumulative radiation dose, and the radio-sensitivity of the tumor cells. For an efficient tumor treatment, radioisotopes with short path lengths such as alpha (< 100 μm) or beta (1–10 mm) are required rather than gamma-emitting radioisotopes. Beta radiation is suitable for targeting tumors larger than 0.5 cm [14, 15]. Shorter-range beta emitters may be used to treat small tumor cell clusters [16], whereas more powerful and longer-range beta emitters can be used for larger tumors [16].
Currently, most RIT protocols use beta‐emitting radionuclides such as iodine‐131 (131I), yttrium‐90 (90Y), and lutetium‐177 (177Lu). 131I- or 90Y-labeled conjugated mAbs provide selective deposition of radionuclides in the tumor [17]. The application of alpha radiation in RIT has also been developed recently [18]. Alpha emitters transfer their energy to the surrounding molecules within a narrow range. Bismuth-213 (213Bi), astatine-211 (211At), and actinium-225 (225Ac) are well-studied alpha emitting atoms [19,20,21,22]. Radionuclides used in HCC-RIT are listed in Table 1.
Targeting tumor antigens, immune ligands and receptor candidates for the treatment of hepatocellular carcinoma
The liver has a major function to maintain the immune hemostasis. The immune system detects and responds to external antigens or mutant cells. Liver sinusoidal endothelial cells (LSECs), hepatocytes, Kupffer cells (KCs), hepatic stellate cells (HCSs), and dendritic cells (DCs) make together the internal hepatic immune network. Also, immunoglobulins and complements as a part of the humoral immune system are involved in the liver immune surveillance [19,20,21,22]. The initiation and progression of an HCC tumor are usually accompanied by chronic inflammation in the liver due to the presence of various cytokines such as IL-10, IL-35, and TGF-β and many immune cells, e.g., regulatory T-cells (Tregs), Th2 macrophages, and M2 macrophages. These components synergistically prepare a hypoxic immunosuppressive microenvironment (HISM) for cancerous cells. HCC-induced immunosuppression has not been fully studied, but many publications indicate that immunotherapy by targeting the anti-tumor immunity can lead to a change in the number of immune cells or their function, attenuation of cytotoxic T-lymphocyte responses, expression of immune receptors and ligands, and increased cytokine levels [11, 23]. Tracing a targetable antigen plays a crucial role in the success of immunotherapy-based clinical practice. The most intensively expressed surface antigen which is specific to a cancerous cell is an ideal target for RIT.
Programmed cell death protein 1 (PD-1) and programmed death-ligand1 (PD-L1, also known as B7-H1) are surface proteins expressed on a broad range of immune cells [24, 25]. PD-L1 expression in the tumor tissue occurs at an early stage and may represent an important contribution to the immune evasion during the HCC progression [25]. Due to the expression of PD-L1 and PD-L2 on KCs and LSECs in both normal liver tissues and HCC cancer cells, liver-infiltrating T-cells frequently encounter negative signals and become “exhausted” [25]. The PD-L1 expression level significantly correlates with the tumor size, recurrence rate, and PIVKA-II (prothrombin induced by vitamin K absence-II) levels. PD-L2 levels are also associated with the histological differences of tumors [26]. HCC patients with PD-L1 expression are at a significantly higher risk of the cancer recurrence [27]. Currently, anti-PD-1 antibodies (nivolumab and pembrolizumab) and anti-PD-L1 antibodies (atezolizumab and avelumab) are approved agents for the treatment of different types of cancers [28]. In September, 2017, nivolumab was approved for the treatment of patients with advanced HCC who were sorafenib-refractory/intolerant [29]. The results suggest that, in the treatment of patients with advanced HCC, nivolumab offers better opportunities as compared to the other conventional therapies [30]. Although FDA-approved pembrolizumab as a second-line treatment for patients with HCC, it failed to meet the primary endpoints for both the overall survival and progression-free survival [31].
Tumor-infiltrating lymphocytes (TILs) and tumor-associated macrophages (TAMs) play a major role in the progression and prognosis of HCC [32]. T-cell immunoglobulin (Ig) and mucin-domain containing protein-3 (Tim-3) have been well recognized as crucial negative regulators of the T-cell-mediated responses [32]. Tim-3 is expressed on TILs in HCC and impairs their function through the Tim-3/galectin-9 signaling pathway [33, 34]. Tim-3 and PD-1 are two critical suppressor molecules and their expression on TILs is associated with a poor prognosis [33]. In patients with adaptive resistance to the anti-PD-1 treatment, the Tim-3 expression was significantly up-regulated. A blockade of both PD-1 and Tim-3 showed a significant increase in the survival rate in a mouse model of lung cancer [35]. It has been demonstrated that Tim-3 is a negative regulator of IFN-secreting CD4+ Th1 and CD8+ T-cells, as well as a key participant in T-cell exhaustion in the tumor microenvironment [36]. Tim-3 overexpression in hepatocytes promotes tumor cell proliferation, while anti-Tim-3 antibodies or RNAi decrease the tumor growth in malignant hepatocytes, both in vitro and in Tim-3 mutant mice [37].
Another important checkpoint inhibitor molecule, cytotoxic T-lymphocyte associated antigen-4 (CTLA-4), is found on the surface of cytotoxic T and Treg cells and plays a multifaceted role. CTLA-4 molecules bind to CD80 and CD86 with a higher affinity than CD28 [38]. CTLA-4 is actively competing for binding to the co-stimulatory CD28 molecule, and after the attachment to its receptor, it stimulates the increased secretion of the immunoregulatory cytokine, IL-10. It also serves as a key mediator which inhibits the T-cell activation and muted immune response [39]. Tremelimumab is a fully humanized mAb targeting CTLA-4 and is well-tolerated when administered as a single agent to patients with HCC. It works by enhancing the T-cell activation and proliferation [40] through various mechanisms which are still under study. Reinforcement of the anti-tumor activity of cytotoxic T-cells has also been reported following the blockade of CTLA-4 in HCC patients [40]. Many studies applied Ipilimumab, an anti-CTLA-4, in combination with nivolumab (NCT01658878) and could show an acceptable safety profile with an objective response rate that was two times higher than that of the nivolumab monotherapy (31% VS. 14%) [41]. Furthermore, combining Tremelimumab with interventional radiologic procedures has been applied in a few studies and resulted in promising clinical outcomes with objective durable responses [42]. Sobhani et al., suggested that antibodies or small molecules that inhibit CTLA-4, but do not alter CTLA-4 levels in Treg cells, could be innovative and eventually more effective in eliminating cancer cells. It seems that such medications would not degrade CTLA-4 and so would not interfere with the function of Treg cells in preventing autoimmunity. As a result, CTLA-4 inhibition could be accomplished without CTLA-4 degradation or toxicity-related side effects. Testing their efficacy in combination with other ICIs, such as anti-PD1 and anti-PD-L1 could enhance therapeutic efficacy [38].
CD147 (also named EMMPRIN or HAb18G/CD147) is a member of the Ig superfamily of adhesion molecules and is always associated with the invasiveness in HCC [43]. The previous studies have demonstrated that CD147 is involved in the epithelial-mesenchymal transition in hepatocytes and also inhibits the Rho signaling pathways and the amoeboid movement by inhibiting annexin II phosphorylation. Moreover, the oncogenetic transmembrane CD147 protein activates the membrane localization of WAVE2 and Rac1 through the integrin-FAK-PI3K/PIP3 pathway, promotes the formation of lamellipodia and enhances the mesenchymal movement [44]. Furthermore, the main three functions of CD147 have been revealed in the regulation of glucose metabolism in HCC. CD147 acts as an important regulator of the Warburg effect in HCC cells by promoting glycolysis through inhibiting the mitochondrial biogenesis and oxidative phosphorylation. CD147 facilitates the expression of monocarboxylate transporter 1 (MCT1) and export of lactate, which leads to the activation of the PI3K/Akt/MDM2 pathway and consecutively induction of p53 degradation. Finally, the down-regulation of glucose metabolism by blocking CD147 suppresses proliferation of HCC cells, suggesting the metabolic impact of CD147 on the tumor growth in HCC [45]. The lipid metabolism in cancer cells is also regulated by CD147 [46,47,48]. The described findings indicate that the oncoprotein CD147 remarkably promotes the de novo fatty acid synthesis via up-regulating lipogenic enzymes ACC1 and FASN via the Akt/mTOR/SREBP1c signaling pathway, and coordinately inhibits fatty acid β-oxidation through the down-regulation of fatty acid oxidative enzymes CPT1A and ACOX1 via the p38 MAPK/PPARα signaling pathway [46].
These crucial roles make CD147 an attractive target for the therapeutic intervention in HCC. In HCC patients, the increased CD147 expression in tumor tissues, but not in the serum, is frequently linked to a poor prognosis [49]. Iodine-metuximab (Licartin) is an 131I-labeled murine mAb HAb18 F(ab’)2 fragment against the HAb18G/CD147 antigen and its specificity and efficient affinity have been proved in the preliminary studies [93, 94]. 131I metuximab has also been successfully applied to prevent the tumor recurrence following the liver transplantation or radiofrequency ablation (RFA) in patients with advanced HCC [50, 51]. The anti-CD147 treatment inhibits migration and invasion of HCC by down-regulation of the metal-matrix protease (MMP) production and rearrangement of the actin cytoskeleton [51].
A transmembrane glycoprotein Endoglin (CD105) is one of the co-receptors of the transforming growth factor β (TGF-β) [52, 53] and is implicated in multiple signaling pathways including proliferation, migration and adhesion of endothelial cells (ECs). CD105 is also expressed in the cytoplasm of non-ECs of normal and malignant tissues [54]. Undifferentiated and differentiated adult-derived human liver stem/progenitor cells [55], human adipose-derived stem cells [56], and hepatic perivascular mesenchymal stem cells (MSCs) [57] are also characterized by different expression levels of CD105.
The observations and collected data in terms of CD105 expression and its prognostic role in HCC are not consistent [58]. Limited evidence has shown that CD105 is not an appropriate target for the angiogenesis therapy, since the protein is expressed not only in neo-vessels of tumors but also in LSECs in non-tumoral tissues [59]. Several types of therapeutic approaches based on anti-CD105 mAbs have shown beneficial anti-angiogenic and anti-tumor impacts including radio-labelled antibodies or immunotoxin-conjugated antibodies [60]. Moreover, the use of CD105-targeting liposomes could be considered a novel strategic tool for the future application of CD105-directed neoplastic and anti-angiogenic therapies [61]. Furthermore, the clinical studies showed the effectiveness of chimeric IgG1 anti-CD105 mAb (TRC105) in terms of angiogenesis and tumor growth inhibition and apoptosis induction [53]. However, the use of TRC105 in those HCC patients who were refractory to the sorafenib treatment resulted in unsatisfactory results. Based on the observations, a combination of TRC105 with novel promising agents in patients with HCC could result in better results.
The human homologue of the Drosophila Roundabout gene, ROBO1, encodes a receptor that is considered a novel subfamily of the Ig superfamily. ROBO1 is significantly up-regulated in 85% of HCC patients and its role have been demonstrated in the cancer angiogenesis and metastasis [62]. The pro-angiogenic effect of ROBO1 in ECs is mediated by the modulation of the Rho family of GTPases and cytoskeleton in HCC. The Rho family of GTPases, including Rho, Rac, and Cdc42, has been implicated in many cellular processes, including actin and microtubules’ organization. They are also essential in the cell polarity, microtubule dynamics, membrane transport pathways, and transcription factor activity [63]. Inhibition of the RhoC/Rac GTPase activation resulted in decreased angiogenesis in HCC [63, 64]. Application of Mab against ROBO1 has shown anti-cancer activity in the HCC animal model [63]. Using 90Y-anti-ROBO1 Mab on a HCC xenograft tumor in nude mice, significant anti-tumor growth effects were found indicated by tumor cells degeneration and increased number of apoptotic cells without necrosis or fibrosis [65]. These findings supported radiolabeled anti-ROBO1 IgG as a potential candidate for RIT in HCC. However, immunotherapy of HCC by targeting ROBO-1 is still under study and requires further investigations to develop more effective therapeutic approaches [66].
Targeting tumor angiogenesis is considered an attractive therapeutic modality for the cancer treatment. HCC is a hyper vascular solid tumor, and inhibition of angiogenesis is an efficient intervention to treat HCC [67]. Among antibodies with the anti-angiogenetic activity which have been tested in patients with HCC, only bevacizumab, humanized anti-VEGF mAb, has received an approval for clinical indications [68, 69]. A novel combination therapy with bevacizumab and atezolizumab is under investigation for the treatment of advanced HCC (NCT03434379). Accordingly, the FDA has granted a breakthrough therapy designation for co-administration of atezolizumab and bevacizumab to be a first-line therapy for advanced or metastatic HCC [70].
Extra-vesicles and their therapeutic potential
The recent studies have revealed the possible role of biomarkers based on extra-vesicles (EVs) in the HCC progression. EVs are now recognized as important derivatives of both immune and tumor cells [71] which transfer bioactive components from the host cells to the recipient cells. It has been shown that immune cell-derived exosomes (IEXs) and tumor-derived exosomes (TEXs) can activate the humoral and cell-mediated immune system by transferring antigens to the antigen-presenting cells (APCs) [72]. Thereby, IEXs can initiate and promote the anti-tumoral responses and inhibit the tumor progression [73]. In contrast to IEXs, TEXs contain immunosuppressive factors affecting the anti-tumor activity of immune cells. A large number of studies have demonstrated that TEXs can suppress the activation of NK cells, interfere with the maturation of DCs, promote the development of myeloid-derived suppressor cells, and transform macrophages into the tumor-promoting phenotype [74, 75]. A recent study demonstrated that DCs treated with TEX can definitely elicit the tumor suppression by improving the tumor-specific immunity. Importantly, an intravenous injection of HCC-derived TEX-treated DCs increased infiltrated T-cells’ and interferon (IFN)-γ levels as well as decreased IL-10 and TGF-β at the tumor sites [76]. On the other hand, TEXs could undermine the function of T-cells and NK cells, and could increase the immuno-prohibitive M2 macrophages and N2 neutrophils in HCC [71].
EVs from HCC cells could play a significant role as a carrier of antigens in a wide range [77]. Recently, the role of HCC-derived EVs has been explored in immunotherapy. DCs were activated by pulsed HCC cells-derived TEXs which carried HCC antigens. TEX-pulsed DCs significantly activated the T-cell-dependent anti-tumor immunity in host HCC cells and improved the tumor microenvironment in host HCC cells [76].
PD-L1 expressing EVs may be produced by tumor cells, immune cells, MSCs [78] or other cells in the tumor microenvironment [79]. The circulating level of PD-L1 does not correlate with the intra-tumoral expression of this molecule [80]. HCC-derived EVs promoted the expression of PD-L1 in macrophages to prohibit the T-cell activation through miR-23a/PTEN/AKT [81]. Moreover, EVs derived from melatonin-treated HCC cells could decrease the expression of PD-L1 and suppress the secretion of cytokines (IL-6, IL-1β, IL-10, and TNF-α) in macrophages [82]. Combining DC-TEX vaccination with sorafenib and a PD-1 antibody could promote the immune responses in an orthotopic HCC-vaccinated mice model. Moreover, a combination of DC-TEX and sorafenib could significantly reduce the number of Treg cells and increase the number of CD8+ T-cells [76]. The data have shown that both the circulating levels and intra-tumoral expression of PD-L1 independently contribute to the HCC prognosis [83]. It has been demonstrated that TEXs can carry PD-L1 on their surface as well and play an important role in the tumor development [73]. However, the application of PD-L1 on EVs as a possible predictor for the anti-PD-1 therapy approach remains controversial [80] and a PD-1/PD-L1 blockade in these patients resulted in considerable individual differences. The resistance to immunotherapy against PD-L1 from EVs may be due to its low content relative to the surface PD-L1. Therefore, targeting PD-L1 on EVs with current immune-checkpoint therapies and applying more effective small molecules seems to be a crucial strategy. Alternatively, up-regulation of PD-L1 on EVs makes it more prone to the action of the delivered antibody. Targeting HCC-derived TEXs expressing PD-L1 by antibodies and application of combination components may help to improve the treatment efficacy in liver cancer. Moreover, HCC-derived TEXs can provide a source of multiple antigens to amplify the DCs-mediated immunotherapeutic effects in an HCC mouse model and human HCC cells. Importantly, HCC-derived TEXs not only produce tumor-specific cytolysis in DCs but also provide cross-protection for other types of HCC cells [77].
α-fetoprotein (AFP)-expressing DC-derived EVs (DEXAFP) have been studied to show the association between the tumor-immune niche and HCC inhibition. The study demonstrated that DEXAFP could induce an effective antigen-specific immune response in an ectopic or an orthotopic HCC murine model. Because of the boosted response of CD8+ T-cells, the expression levels of IFN-γ and IL-2 were up-regulated, whereas CD25+Foxp3+ Treg cells, IL-10, and TGF-β levels were declined at the tumor sites. DC-derived EVs can obviously impede the HCC proliferation and increase the survival rate in a murine model [84].
Studies have shown that EVs derived from hepatoma cells can promote the migration and invasion of recipient cells, down-regulate the E-cadherin expression, increase the vimentin expression, and promote the epithelial-mesenchymal transition (EMT) in cells. Moreover, those effects are more remarkable in highly invasive hepatoma-cells-derived EVs than in low-invasive hepatoma and normal liver cells EVs [85]. HCC-derived EVs could redirect the metastasis of tumor cells which lack the capacity to metastasize to a specific organ via generating a pre-metastatic niche [86]. EVs can facilitate the metastasis by transporting bioactive molecules. Studies have found that EVs can act as regulators of the tumor microenvironment to influence the tumor cell invasion and EMT [87]. These findings indicate the importance of EVs in the pathogenesis of HCC, both as a target for the treatment and a vehicle for drug delivery [86].
A substantial body of evidence highlighted the role of EVs as delivery vehicles [88, 89]. The efficacy and indication of drug delivery by EVs depend on the type of a donor cell, therapeutic agent, use of targeting peptides, loading method, and routes of administration [90]. The microRNAs (miRNAs) within TEXs have a significant role in regulating the cancer progression, possibly through facilitating the tumor development, metastasis, and angiogenesis, so they are attractive targets for the therapy [89]. It has been demonstrated that co-culture of stellate cells with HCC cells down-regulates miR-335-5p in both cells, which inhibits the HCC cell proliferation by delivering EVs. However, up-regulating the expression of miR-335-5p in stellate cell-derived EVs can inhibit the growth and invasion of HCC cells and lead to the tumor shrinkage in a murine model [91]. Another study showed that adipose tissue-derived-MSC-EVs, which were effectively transfected with miR-122 plasmids and transferred to HCC cells, significantly rendered HCC cells sensitive to chemotherapy agents (e.g.,5-FU and sorafenib) [92]. Similarly, adipose tissue-derived MSCs were used to deliver miRNA-199a (miR-199a) for improving the HCC chemosensitivity [93]. Experimental studies showed that TEX-miR-199a significantly sensitized HCC cells to doxorubicin by targeting and inhibiting the mTOR pathway [93].
HepG2-EVs have tetraspanin cell membrane proteins such as CD9, CD63, and CD81 on their surface, allowing cognate antibodies to bind [94]. Tetraspanin-8 (TSPAN8) has been described as a significant contributor in many tumors of the gastrointestinal system such as colorectal, hepatic, esophageal, and pancreatic cancer, and is associated with a poor prognosis [95, 96]. TSPAN8 also elevates the growth and invasion potential of tumors by stimulating angiogenesis and cell migration [95, 97]. In HCC, TSPAN8 is up-regulated and plays a role as an independent prognostic factor for the recurrence-free survival (RFS) and overall survival [95]. Targeting TSPAN8 with radiolabeled antibodies appears an effective anti-tumoral therapy. Two radiolabeled anti-TSPAN8 mAbs (Ts29.1 and Ts29.2) with Indium-111 (111In) have been investigated so far. High specificity of 111In-Ts29.2 in TSPAN8-expressing pancreatic tumors has been demonstrated in a murine model of both SW480-Co29/SW480 tumors [94]. Additionally, TSPAN8 is expressed on the surface of circulating EVs [97], and targeting them will be of interest as these vesicles facilitate the metastasis.
More investigations on exosomes’ biology will be the key to progress in the application of exosomes as a novel therapeutic target and drug delivery platform for RIT.
The future prospective and limitations of extra-vesicles as drug delivery tools for radioimmunotherapy in hepatocellular carcinoma
During recent years, different disciplines for efficient drug delivery approaches emerged. However, we are far from an ideal approach regarding efficiency, tolerability, and targeted drug delivery tools. EVs as intercellular communication system, have shown promising results in practical, non-immunogenic, and efficient drug delivery platform. A number of useful features qualified them as an alternative strategy for drug delivery such as their wonderful biocompatibility, ability to cross physical barriers, exploit natural intracellular cross-talk, and their inherent targeting features [98].
Bioengineering paternal cells can provide enough conjugating targeting moieties for EVs in RIT applications. However, more basic and preclinical studies should be made in order to translate this technique in the clinic. Scale-up production of EVs and making their production cost–benefit are major challenges in their clinical translation. Batch-to-batch variation in the industrial production of EVs is the other obstacle in broad application of EVs in approved therapeutic protocols [99]. EVs could be used in RIT as well in order to boost therapeutic efficacy of medical settings. However, learning the molecular machinery which controls cellular production and characteristics of EVs in terms of physiology and pathophysiology of biological processing is critical in translational studies [100].
Clinical studies
Sorafenib is an oral multi-kinase inhibitor which acts through inhibiting the activity of Raf kinase, the VEGF-Receptor, and the Platelet-Derived Growth Factor Receptor (PDGF-R) [101]. Although sorafenib has remained the recommended choice for the first-line systemic therapy in advanced HCC, the therapeutic results were not satisfactory [102].
On the other hand, despite the promising effects of immunotherapy in some HCC patients, a significant number of patients did not attain a clinically significant and durable improvement [103]. Recently, a phase III clinical trial reported that the overall survival rate did not significantly differ between the HCC patients who underwent selective internal radiotherapy (SIRT) with 90Y-resin microspheres and those who received sorafenib (NCT01482442) [102]. Moreover, in a phase lll clinical trial, the application of sorafenib in the adjuvant setting following the surgical resection did not present an effective treatment in HCC patients [104]. This unsatisfactory response to the sorafenib monotherapy in the conditions of a heterogeneous tumor microenvironment suggests the use of combinational strategies. Recently, more research and clinical studies have been designed to assess the synergistic effects of ICIs and radiotherapy in various cancers such as melanoma, head and neck, colorectal, sarcoma, and renal cancer [105]. The therapeutic potential of RIT in HCC is still under investigation in several clinical trials. The registered trials at www.ClinicalTrials.gov are listed in Table 2.
PD-L1/PD-1 and CTLA-4 are the main immune checkpoints extensively inhibited with targeted therapies that leads to remarkable clinical improvements in HCC patients [70]. Nivolumab, an anti-PD-1 antibody, was reported to be well-tolerated by patients who were treated for other malignancies [106, 107]. Nivolumab also led to a durable response in patients irrespectively of their hepatitis B or C viral status, compared to sorafenib as a primary treatment [108]. Currently, a phase I trial is recruiting patients with advanced HCC for a combination therapy using nivolumab and 90Y with the intent for resection (NCT03812562). Another clinical trial has also been designed to evaluate the efficacy and tolerability of simultaneous application of nivolumab and 90Y (NCT02837029) [109]. Despite the disappointing results, pembrolizumab is still under investigation in HCC patients as an adjunctive treatment for trans-catheter arterial chemoembolization (TACE) in a phase II/III study [28]. Interestingly, more clinical trials are currently underway combining a regional therapy of 90Y radioembolization with pembrolizumab (phase I, NCT03099564) [110] or nivolumab (phase I, NCT03812562) [110]. Moreover, a phase II trial evaluating 90Y radioembolization with nivolumab has been initiated (NCT03033446). Currently, the combination of a novel anti-PD-1 antibody, SHR-1210 and apatinib, a tyrosine kinase inhibitor selectively acting on VEGF receptor 2, is under investigation (NCT03463876). In 2018, the phase 1 trial of this combination was completed and showed acceptable tolerability and the response rate of 38.9%, with a median progression-free survival of 7.2 months for 18 patients with HCC. Overall, adverse events were relatively tolerable. A phase II trial of SHR-1210 combined with apatinib is currently ongoing in the USA, comparing this combination to systemic chemotherapy in advanced HCC (NCT02942329) [111].
According to the preclinical and early phase I/II clinical trials, a combination of durvalumab and tremelimumab, anti-PD-L1 and anti-CTLA-4, respectively, enhanced the anti-tumor activity compared to the monotherapy and proposed acceptable safety and a durable objective response rate (NCT02519348) [112]. Thus, a phase III trial is currently underway (NCT03298451) [28]. The initial results of a clinical trial on administration of tremelimumab with subtotal TACE or radiofrequency ablation (RFA) in patients with advanced HCC showed that the treatment was safe and feasible, as well (NCT01853618) [113] and a phase I/II trial is currently underway in the USA [42]. In addition, clinical trials are also conducted on the use of external beam photon stereotactic body radiotherapy (SBRT) in combination with nivolumab (Phase II, NCT03316872) or ipilimumab (Phase I, NCT03203304). In metastatic breast cancer, tremelimumab appears to be safe when combined with palliative radiotherapy [114]. Despite a large number of antibodies against CTLA-4 in HCC patients, the efficiency of combining ICIs and radiotherapy needs more investigations. Therefore, clinical trials are expected to closely monitor the safety profile of RIT by targeting CTLA-4. The administration of 131I-metuximab has also been studied as an adjuvant therapy after the liver resection in HCC patients and resulted in improvement of the 5-year RFS [115]. Promising anti-recurrence efficacy of treatment with 131I-metuximab has also been reported after the liver transplantation and early HCC ablation [50]. Following the injection of 27.75 MBq/kg in 106 patients, the half-life of 131I-metuximab in the blood was 90.56–63.93 h and the survival rate was 44.54%. Additionally, in a phase II trial (NCT00819650) the role of 131I-metuximab as an adjuvant treatment has been studied after hepatectomy for HCC patients. Accordingly, the patients who underwent one trans-arterial injection of 131I-metuximab within 4–6 weeks after hepatectomy exhibited an improved 5-year RFS [116]. Similarly, in 2007, 131I metuximab was reported as a promising drug for preventing the tumor recurrence in advanced HCC after the liver transplantation. The combination of a trans-hepatic arterial injection of 131I-metuximab and TACE has also showed promising results and is well-tolerated in patients with advanced and unresectable HCC [117]. Thus, these findings have demonstrated that 131I-metuximab has effective targeting properties and allows one to achieve the maximum protection with significantly reduced side effects.
The therapeutic efficacy of 131I-anti-CD105 RIT has been investigated in a mouse model of HCC, and remarkable inhibition of the tumor growth has been reported [68]. Moreover, a combination of 131I-anti-CD105 RIT and 5-FU was effective in the HCC mouse model [52]. Although CD105 is a promising target of anti-angiogenic therapy in solid tumors, the number of clinical trials using this potential target in advanced HCC is limited [53]. In some clinical trials, the safety and efficacy of CD105 monotherapy (TRC105) has been assessed [118]. Moreover, an anti-CD105 antibody has been applied in combination with different drugs including bevacizumab [119] or sorafenib [120]. A combination of TRC105 and sorafenib showed encouraging results (25% partial response) in HCC patients (NCT01806064) [121]. Owing to the insufficient efficacy of TRC105 monotherapy in phase I/II clinical trials [53], further studies are required to assess the effectiveness of RIT using TRC105 in HCC patients.
The role of hepatitis infection in the treatment of HCC with immunotherapy
Regarding the role of HBV and HCV infection in the development of HCC, various clinical studies looked at how these viral agents reacted to immunotherapeutic therapies [122]. Throughout the CheckMate 040 trial, the objective response rate (ORRs) with nivolumab in patients infected with HCV and HBV were 20% and 14%, respectively, but ORRs in patients with no viral infection were 22% [30]. In addition, the 6- and 9-month OS rates in HBV and HCV-positive patients were 84%, 70%, 85%, and 81%, respectively, compared to 74% in the whole population. ORR indicated no statistically significant difference between HCC cases with HBV+, HCV+, and uninfected patients in the KEYNOTE-224 trial, which employed pembrolizumab (13% vs 13% vs 20%, respectively) [123]. A previous clinical trial (NCT02702401) has shown that tumor size reductions ranged from 50% in all individuals to 57% in HBV+ and 39% in HCV+ patients. Moreover, pembrolizumab was shown to be 18% effective in the KEYNOTE-240 trial, compared to 4% effective in the placebo group. Interestingly, a substantial benefit for HBV+ patients compared to the placebo group has been identified in terms of overall survival [HR 0.57 (CI 0.35–0.94)], but no meaningful benefit was detected for HCV+ patients. The combination of atezolizumab and bevacizumab resulted in a longer median PFS in HCC patients with HBV infection as compared to sorafenib therapy, but not in non-viral HCC patients (median PFS, HBV+ HCC: 6.7 vs. 2.8 months; non-viral HCC: 7.1 vs. 5.6 + months). In both conditions, a meta-analysis evaluating the effectiveness of PD-1/PD-L1 inhibitors as monotherapy or in combination with other treatment drugs found no significant difference in ORR between the two groups [124]. In both arms, the authors found no improvement in ORR and disease control rates (DCRs) in HCV patients. Additionally, ICIs may be less effective for HCC caused by nonalcoholic fatty liver disease (NAFLD) than for HCC caused by viruses [125].
Future prospective
While the surgery is considered the treatment of choice for advanced HCC, no effective systemic therapy has been approved so far. Sorafenib is the only standard FDA-approved medicine for advanced HCC, and the disease progression is common in patients [126].
In HCC, the heterogeneous tumor microenvironment interacts with various immune cells to sustain the growth. A comprehensive understanding of tumor-immune interactions led to the development of ICIs as a new therapeutic strategy. However, targeting the immune-checkpoint molecules reinvigorates the anti-tumor immunity by restoring exhausted T-cells, and many immunosuppressive mechanisms can limit the efficacy of ICIs monotherapy in HCC [127, 128].
On the other hand, radiotherapy is an essential therapeutic modality for HCC and has gained an extensive attention as promising in combination with ICIs. RIT may enhance the endogenous anti-tumor responses compared to the current monotherapies. The efficacy of RIT has been well-documented in numerous preclinical and clinical studies on various types of cancers, but the literature related to HCC in this context is very limited [6].
Regarding the therapeutic potential of RIT in HCC, many challenges remain to be addressed. First, application of ICIs should not be considered the best associate treatment with radiotherapy in HCC. Other immunotherapeutic strategies such as cytokine-induced killer cells or gene therapy using adenoviral vectors have already been assessed in HCC patients. Moreover, the optimized radiotherapy technique in combination with ICIs has not been established yet [129].
Furthermore, the evaluation of HCC patients’ suitability for RIT by next-generation sequencing-based profiling of the tumor mutation burden, immune gene expression signatures, T-cell receptor repertoire, and T-cell-inflamed gene expression could be helpful. Further efforts are needed to identify novel biomarkers to guide the selection of appropriate HCC patients for RIT [130, 131].
One of the most important RIT challenges is that most HCC patients suffer liver cirrhosis and the combination of medications may increase the liver toxicity resulting in adverse consequences [132]. In the new clinical setting, it is crucial to ensure a safe liver toxicity profile when using combinatory approaches. Moreover, in patients with chronic hepatitis B, hepatitis C, human immunodeficiency virus or tuberculosis, the T-cell or NKC-mediated antiviral or antibacterial immune response is attenuated [133]. Due to the risk of viral reactivation, it is important to monitor the differential responses to RIT in HCC patients with HBV or HCV. A further experimental research and clinical studies are necessary on the interplay of HCC and viral hepatitis in patients receiving RIT [126]. RIT is also valuable in targeting both primary and secondary tumors, besides the residual tumor margins after resection [18].
Furthermore, an anti-angiogenic therapy has been developed based on the rationale that reduction in the tumoral vascular network can result in a hypoxic microenvironment [134]. The recent advances include combining ICIs with anti-angiogenic agents to treat advanced HCC [135]. Besides sorafenib, several anti-angiogenic drugs including lenvatinib, regorafenib, cabozantinib, and ramucirumab have been investigated in the first-line clinical trials with the aim of developing molecular targeted agents showing a better efficacy than sorafenib [136,137,138,139,140]. However, hypoxia as a potential side effect induced by anti-angiogenic drugs may increase the invasiveness of tumor cells and accelerate the metastasis [53]. One way to overcome these limitations is developing new types of anti-angiogenic therapy of HCC using different approaches, such as a combination of numerous anti-angiogenic compounds with other treatment regimens [141].
Conclusion
The combinatorial approaches and optimized targeting a heterogeneous, immunosuppressive tumor are expected to be the main avenue of the HCC treatment in the future. RIT is highly targeted and destructive to tumor tissues and causes as little injury as possible to normal tissues compared to chemotherapy or radiotherapy. These features enable one to increase the survival and decrease the recurrence rate. Since RIT provides a high binding affinity, it promotes a selective uptake of energy in tumoral tissues. There are few relevant clinical studies on the efficacy of RIT in the literature which mostly involve a small number of HCC patients. Until now 131I-metuximab has been the only confirmed RIT approach in the clinical trials which have the efficacy and acceptable safety in the HCC treatment. The efficacy and safety of RIT as applied to HCC still requires high-quality, evidence-based randomized controlled studies.
Data availability
Not applicable.
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: Cancer J Clin 68(6):394–424
Shokoohian B, Negahdari B, Aboulkheyr Es H, Abedi-Valugerdi M, Baghaei K, Agarwal T et al (2021) Advanced therapeutic modalities in hepatocellular carcinoma: novel insights. J Cell Mol Med 25(18):8602–8614
Kang-Da L, Zhao-You T, Yan-Ming B, Ji-Zhen L, Feng Q, Ai-Na Y et al (1989) Radioimmunotherapy for hepatocellular carcinoma (HCC) using 131I-anti HCC isoferritin IgG: preliminary results of experimental and clinical studies. Int J Radiat Oncol* Biol* Phys 16(2):319–323
Larson SM, Carrasquillo JA (2015) Cheung N-KV, Press OWJNRC. Radioimmunother Hum Tumours 15(6):347
Sahlin M, Bauden MP, Andersson R, Ansari DJTB (2015) Radioimmunotherapy—a potential novel tool for pancreatic cancer therapy? Tumor Biol 36(6):4053–4062
Wu L, Yang Y-F, Ge N-J, Shen S-Q, Liang J, Wang Y et al (2010) Hepatic arterial Iodine-131–labeled metuximab injection combined with chemoembolization for unresectable hepatocellular carcinoma: interim safety and survival data from 110 patients. Cancer Biother Radiopharm 25(6):657–663
Tartarone A, Lapadula V, Di Micco C, Rossi G, Ottanelli C, Marini A et al (2021) Beyond conventional: the new horizon of targeted therapy for the treatment of advanced non small cell lung cancer. Front Oncol 11:632256
Tartarone A, Roviello G, Lerose R, Roudi R, Aieta M, Zoppoli P (2019) Anti-PD-1 versus anti-PD-L1 therapy in patients with pretreated advanced non-small-cell lung cancer: a meta-analysis. Future Oncol 15(20):2423–2433
Lee HW, Cho KJ, Park JY (2020) Current status and future direction of immunotherapy in hepatocellular carcinoma: what do the data suggest? Immune Netw 20(1):e11-e
Bourke JM, O’Sullivan M, Khattak MA (2016) Management of adverse events related to new cancer immunotherapy (immune checkpoint inhibitors). Med J Aust 205(9):418–424
Zongyi Y, Xiaowu L (2020) Immunotherapy for hepatocellular carcinoma. Cancer Lett 470:8–17
Llovet JM, Real MI, Montaña X, Planas R, Coll S, Aponte J et al (2002) Arterial embolisation or chemoembolisation versus symptomatic treatment in patients with unresectable hepatocellular carcinoma: a randomised controlled trial. The Lancet 359(9319):1734–1739
Wang T-H, Huang P-I, Hu Y-W, Lin K-H, Liu C-S, Lin Y-Y et al (2018) Combined Yttrium-90 microsphere selective internal radiation therapy and external beam radiotherapy in patients with hepatocellular carcinoma: from clinical aspects to dosimetry. PLoS ONE 13(1):e0190098
Asadian S, Piryaei A, Gheibi N, Aziz Kalantari B, Reza Davarpanah M, Azad M et al (2022) Rhenium Perrhenate ((188)ReO(4)) induced apoptosis and reduced cancerous phenotype in liver cancer cells. Cells 11(2):305
Sigmund P (2014) Particle penetration and radiation effects volume 2. Springer Series in Solid-State Sciences, vol 179. Springer, Berlin
Kim Y-S, Brechbiel MW (2012) An overview of targeted alpha therapy. Tumor Biol 33(3):573–590
Asadian S, Mirzaei H, Kalantari BA, Davarpanah MR, Mohamadi M, Shpichka A et al (2020) β-radiating radionuclides in cancer treatment, novel insight into promising approach. Pharmacol Res 160:105070
Kawashima H (2014) Radioimmunotherapy: a specific treatment protocol for cancer by cytotoxic radioisotopes conjugated to antibodies. Sci World J. https://doi.org/10.1155/2014/492061
Heymann F, Tacke F (2016) Immunology in the liver—from homeostasis to disease. Nat Rev Gastroenterol Hepatol 13(2):88
Trivedi PJ, Adams DH (2016) Gut–liver immunity. J Hepatol 64(5):1187–1189
Horst AK, Neumann K, Diehl L, Tiegs G (2016) Modulation of liver tolerance by conventional and nonconventional antigen-presenting cells and regulatory immune cells. Cell Mol Immunol 13(3):277–292
Jenne CN, Kubes P (2013) Immune surveillance by the liver. Nat Immunol 14(10):996–1006
Chen D-P, Ning W-R, Jiang Z-Z, Peng Z-P, Zhu L-Y, Zhuang S-M et al (2019) Glycolytic activation of peritumoral monocytes fosters immune privilege via the PFKFB3-PD-L1 axis in human hepatocellular carcinoma. J Hepatol 71(2):333–343
Nguyen LT, Ohashi PS (2015) Clinical blockade of PD1 and LAG3—potential mechanisms of action. Nat Rev Immunol 15(1):45
Wang B-J, Bao J-J, Wang J-Z, Wang Y, Jiang M, Xing M-Y et al (2011) Immunostaining of PD-1/PD-Ls in liver tissues of patients with hepatitis and hepatocellular carcinoma. WJG 17(28):3322
Jung HI, Jeong D, Ji S, Ahn TS, Bae SH, Chin S et al (2017) Overexpression of PD-L1 and PD-L2 is associated with poor prognosis in patients with hepatocellular carcinoma. Cancer Res treat 49(1):246
Gao Q, Wang X-Y, Qiu S-J, Yamato I, Sho M, Nakajima Y et al (2009) Overexpression of PD-L1 significantly associates with tumor aggressiveness and postoperative recurrence in human hepatocellular carcinoma. Clin Cancer Res 15(3):971–979
Okusaka T, Ikeda M (2018) Immunotherapy for hepatocellular carcinoma: current status and future perspectives. ESMO open 3(Suppl 1):e000455
Adcock CS, Puneky LV, Campbell GS (2019) Favorable response of metastatic hepatocellular carcinoma to treatment with trans-arterial radioembolization followed by Sorafenib and Nivolumab. Cureus. https://doi.org/10.7759/cureus.4083
El-Khoueiry AB, Sangro B, Yau T, Crocenzi TS, Kudo M, Hsu C et al (2017) Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): an open-label, non-comparative, phase 1/2 dose escalation and expansion trial. The Lancet 389(10088):2492–2502
Merck (2019) Merck provides update on KEYNOTE‐240, a phase 3 study of KEYTRUDA®(pembrolizumab) in previously treated patients with advanced hepatocellular carcinoma [press release]
Yan W, Liu X, Ma H, Zhang H, Song X, Gao L et al (2015) Tim-3 fosters HCC development by enhancing TGF-β-mediated alternative activation of macrophages. Gut 64(10):1593–1604
Liu F, Zeng G, Zhou S, He X, Sun N, Zhu X et al (2018) Blocking Tim-3 or/and PD-1 reverses dysfunction of tumor-infiltrating lymphocytes in HBV-related hepatocellular carcinoma. Bull Cancer 105(5):493–501
Farzaneh Z, Vosough M, Agarwal T, Farzaneh M (2021) Critical signaling pathways governing hepatocellular carcinoma behavior; small molecule-based approaches. Cancer Cell Int 21(1):1–15
Koyama S, Akbay EA, Li YY, Herter-Sprie GS, Buczkowski KA, Richards WG et al (2016) Adaptive resistance to therapeutic PD-1 blockade is associated with upregulation of alternative immune checkpoints. Nat Commun 7(1):1–9
Wolf Y, Anderson AC, Kuchroo VK (2020) TIM3 comes of age as an inhibitory receptor. Nat Rev Immunol 20(3):173–185
Zhang H, Song Y, Yang H, Liu Z, Gao L, Liang X et al (2018) Tumor cell-intrinsic Tim-3 promotes liver cancer via NF-κB/IL-6/STAT3 axis. Oncogene 37(18):2456–2468
Sobhani N, Tardiel-Cyril DR, Davtyan A, Generali D, Roudi R, Li Y (2021) CTLA-4 in regulatory T cells for cancer immunotherapy. Cancers 13(6):1440
Han Y, Chen Z, Yang Y, Jiang Z, Gu Y, Liu Y et al (2014) Human CD14+ CTLA-4+ regulatory dendritic cells suppress T-cell response by cytotoxic T-lymphocyte antigen-4-dependent IL-10 and indoleamine-2, 3-dioxygenase production in hepatocellular carcinoma. Hepatology 59(2):567–579
Sangro B, Gomez-Martin C, de la Mata M, Iñarrairaegui M, Garralda E, Barrera P et al (2013) A clinical trial of CTLA-4 blockade with tremelimumab in patients with hepatocellular carcinoma and chronic hepatitis C. J Hepatol 59(1):81–88
Yau T, Kang Y-K, Kim T-Y, El-Khoueiry AB, Santoro A, Sangro B et al (2019) Nivolumab (NIVO)+ ipilimumab (IPI) combination therapy in patients (pts) with advanced hepatocellular carcinoma (aHCC): results from CheckMate 040. Am Soc Clin Oncol. https://doi.org/10.1200/JCO.2019.37.15_suppl.4012
Duffy AG, Ulahannan SV, Makorova-Rusher O, Rahma O, Wedemeyer H, Pratt D et al (2017) Tremelimumab in combination with ablation in patients with advanced hepatocellular carcinoma. J Hepatol 66(3):545–551
Lu M, Wu J, Hao ZW, Shang YK, Xu J, Nan G et al (2018) Basolateral CD147 induces hepatocyte polarity loss by E-cadherin ubiquitination and degradation in hepatocellular carcinoma progress. Hepatology 68(1):317–332
Wu J, Ru N, Zhang Y, Li Y, Wei D, Ren Z et al (2011) HAb18G/CD147 promotes epithelial–mesenchymal transition through TGF-β signaling and is transcriptionally regulated by Slug. Oncogene 30(43):4410
Huang Q, Li J, Xing J, Li W, Li H, Ke X et al (2014) CD147 promotes reprogramming of glucose metabolism and cell proliferation in HCC cells by inhibiting the p53-dependent signaling pathway. J Hepatol 61(4):859–866
Li J, Huang Q, Long X, Zhang J, Huang X, Aa J et al (2015) CD147 reprograms fatty acid metabolism in hepatocellular carcinoma cells through Akt/mTOR/SREBP1c and P38/PPARα pathways. J Hepatol 63(6):1378–1389
Felmlee DJ, Baumert TF (2016) CD147 handles lipid: a new role for anti-cancer target. Transl Cancer Res 5(3):238–240
Ju H-L, Ro SW (2016) Making cancer fat: reprogramming of lipid metabolism by CD147 in hepatocellular carcinoma. Chin J Cancer Res 28(3):380
Xiao W, Zhao S, Shen F, Liang J, Chen J (2017) Overexpression of CD147 is associated with poor prognosis, tumor cell migration and ERK signaling pathway activation in hepatocellular carcinoma. Exp Ther Med 14(3):2637–2642
Bian H, Zheng J-S, Nan G, Li R, Chen C, Hu C-X et al (2014) Randomized trial of [131I] metuximab in treatment of hepatocellular carcinoma after percutaneous radiofrequency ablation. J Natl Cancer Inst 106(9):dju239
Wang Y, Yuan L, Yang X-M, Wei D, Wang B, Sun X-X et al (2015) A chimeric antibody targeting CD147 inhibits hepatocellular carcinoma cell motility via FAK-PI3K-Akt-Girdin signaling pathway. Clin Exp Metastasis 32(1):39–53
Duan C, Li Y, Yang W, Zhang C (2018) Combination of 131I-anti-endoglin monoclonal antibody and 5-fluorouracil may be a promising combined-modality radioimmunotherapy strategy for the treatment of hepatocellular carcinoma. Biotechnol Biotechnol Equip 32(4):974–981
Kasprzak A, Adamek A (2018) Role of Endoglin (CD105) in the progression of hepatocellular carcinoma and anti-angiogenic therapy. Int J Mol Sci 9(12):3887
Liu Q, Zhang R-Z, Li D, Cheng S, Yang Y-H, Tian T et al (2016) Muse cells, a new type of pluripotent stem cell derived from human fibroblasts. Cell Reprogram 18(2):67–77
El-Kehdy H, Sargiacomo C, Fayyad-Kazan M, Fayyad-Kazan H, Lombard C, Lagneaux L et al (2017) Immunoprofiling of adult-derived human liver stem/progenitor cells: impact of hepatogenic differentiation and inflammation. Stem Cells Int. https://doi.org/10.1155/2017/2679518
Fu Y, Deng J, Jiang Q, Wang Y, Zhang Y, Yao Y et al (2016) Rapid generation of functional hepatocyte-like cells from human adipose-derived stem cells. Stem cell Res Ther 7(1):1–12
Shenoy PS, Bose B (2018) Hepatic perivascular mesenchymal stem cells with myogenic properties. J Tissue Eng Regener Med 12(3):e1297–e1310
Li Y, Zhai Z, Liu D, Zhong X, Meng X, Yang Q et al (2015) CD105 promotes hepatocarcinoma cell invasion and metastasis through VEGF. Tumor Biol 36(2):737–745
Lin Y, Huang Y, Wu M, Wu S, Chi H, Liao C et al (2013) Thyroid hormone suppresses cell proliferation through endoglin-mediated promotion of p21 stability. Oncogene 32(33):3904–3914
Muñoz R, Arias Y, Ferreras JM, Jiménez P, Langa C, Rojo MA et al (2013) In vitro and in vivo effects of an anti-mouse endoglin (CD105)–immunotoxin on the early stages of mouse B16MEL4A5 melanoma tumours. Cancer Immunol Immunother 62(3):541–551
Tansi FL, Rüger R, Kollmeier AM, Rabenhold M, Steiniger F, Kontermann RE et al (2018) Endoglin based in vivo near-infrared fluorescence imaging of tumor models in mice using activatable liposomes. Biochim Biophys Acta 1862(6):1389–1400
Mehlen P, Delloye-Bourgeois C, Chédotal A (2011) Novel roles for Slits and netrins: axon guidance cues as anticancer targets? Nat Rev Cancer 11(3):188–197
Ao J-Y, Chai Z-T, Zhang Y-Y, Zhu X-D, Kong L-Q, Zhang N et al (2015) ROBO1 promotes angiogenesis in hepatocellular carcinoma through the Rho family of guanosine triphosphatases’ signaling pathway. Tumor Biol 36(11):8413–8424
Zheng F, Liao Y-J, Cai M-Y, Liu T-H, Chen S-P, Wu P-H et al (2015) Systemic delivery of microRNA-101 potently inhibits hepatocellular carcinoma in vivo by repressing multiple targets. PLoS Genet 11(2):e1004873
Fujiwara K, Koyama K, Suga K, Ikemura M, Saito Y, Hino A et al (2014) A 90 Y-labelled anti-ROBO1 monoclonal antibody exhibits antitumour activity against hepatocellular carcinoma xenografts during ROBO1-targeted radioimmunotherapy. EJNMMI Res 4(1):1–10
Rai V, Abdo J, Alsuwaidan AN, Agrawal S, Sharma P, Agrawal DK et al (2018) Cellular and molecular targets for the immunotherapy of hepatocellular carcinoma. Mol Cell Biochem 437(1–2):13–36
Zhou J, Hu P, Si Z, Tan H, Qiu L, Zhang H et al (2019) Treatment of hepatocellular carcinoma by intratumoral injection of 125I-AA98 mAb and its efficacy assessments by molecular imaging. Front Bioeng Biotechnol 7:319
Duan C-L, Hou G-H, Liu Y-P, Liang T, Song J, Han J-K et al (2014) Tumor vascular homing endgolin-targeted radioimmunotherapy in hepatocellular carcinoma. Tumor Biol 35(12):12205–12215
Sitohy B, Nagy JA, Dvorak HF (2012) Anti-VEGF/VEGFR therapy for cancer: reassessing the target. Cancer Res 72(8):1909–1914
Ghavimi S, Apfel T, Azimi H, Persaud A, Pyrsopoulos NT (2020) Management and treatment of hepatocellular carcinoma with immunotherapy: a review of current and future options. J Clin Transl Hepatol 8(2):168
Han Q, Zhao H, Jiang Y, Yin C, Zhang J (2019) HCC-derived exosomes: critical player and target for cancer immune escape. Cells 8(6):558
Dai W, Wang Y, Yang T, Wang J, Wu W, Gu J et al (2019) Downregulation of exosomal CLEC3B in hepatocellular carcinoma promotes metastasis and angiogenesis via AMPK and VEGF signals. Cell Commun Signal 17(1):113
Xie F, Xu M, Lu J, Mao L, Wang S (2019) The role of exosomal PD-L1 in tumor progression and immunotherapy. Mol Cancer 18(1):146
Chen W, Jiang J, Xia W, Huang J (2017) Tumor-related exosomes contribute to tumor-promoting microenvironment: an immunological perspective. J Immunol Res. https://doi.org/10.1155/2017/1073947
Olejarz W, Dominiak A, Żołnierzak A, Kubiak-Tomaszewska G, Lorenc T (2020) Tumor-derived exosomes in immunosuppression and immunotherapy. J Immunol Res. https://doi.org/10.1155/2020/6272498
Shi S, Rao Q, Zhang C, Zhang X, Qin Y, Niu Z (2018) Dendritic cells pulsed with exosomes in combination with PD-1 antibody increase the efficacy of Sorafenib in hepatocellular carcinoma model. Transl Oncol 11(2):250–258
Rao Q, Zuo B, Lu Z, Gao X, You A, Wu C et al (2016) Tumor-derived exosomes elicit tumor suppression in murine hepatocellular carcinoma models and humans in vitro. Hepatology 64(2):456–472
Liu H, Gao W, Yuan J, Wu C, Yao K, Zhang L et al (2016) Exosomes derived from dendritic cells improve cardiac function via activation of CD4+ T lymphocytes after myocardial infarction. J Mol Cell Cardiol 91:123–133
Herbst RS, Soria J-C, Kowanetz M, Fine GD, Hamid O, Gordon MS et al (2014) Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature 515(7528):563–567
Sasaki R, Kanda T, Yokosuka O, Kato N, Matsuoka S, Moriyama M (2019) Exosomes and hepatocellular carcinoma: from bench to bedside. Int J Mol Sci 20(6):1406
Liu J, Fan L, Yu H, Zhang J, He Y, Feng D et al (2019) Endoplasmic reticulum stress causes liver cancer cells to release exosomal miR-23a-3p and up-regulate programmed death ligand 1 expression in macrophages. Hepatology 70(1):241–258
Cheng L, Liu J, Liu Q, Liu Y, Fan L, Wang F et al (2017) Exosomes from melatonin treated hepatocellularcarcinoma cells alter the immunosupression status through STAT3 pathway in macrophages. Int J Biol Sci 13(6):723–734
Sideras K, Robert A, Harrington SM, Polak WG, Zhou G, Schutz HM et al (2019) Circulating levels of PD-L1 and Galectin-9 are associated with patient survival in surgically treated Hepatocellular Carcinoma independent of their intra-tumoral expression levels. Sci Rep 9(1):1–10
Lu Z, Zuo B, Jing R, Gao X, Rao Q, Liu Z et al (2017) Dendritic cell-derived exosomes elicit tumor regression in autochthonous hepatocellular carcinoma mouse models. J Hepatol 67(4):739–748
Qu Z, Feng J, Pan H, Jiang Y, Duan Y, Fa ZJO et al (2019) Exosomes derived from HCC cells with different invasion characteristics mediated EMT through TGF-β/Smad signaling pathway. OncoTargets Ther 12:6897
Ge Y, Mu W, Ba Q, Li J, Jiang Y, Xia Q et al (2020) Hepatocellular carcinoma-derived exosomes in organotropic metastasis, recurrence and early diagnosis application. Cancer Lett 477:41–48
Vella L (2014) The emerging role of exosomes in epithelial–mesenchymal-transition in cancer. Front Oncol 4:361
Yang T, Martin P, Fogarty B, Brown A, Schurman K, Phipps R et al (2015) Exosome delivered anticancer drugs across the blood-brain barrier for brain cancer therapy in Danio rerio. Pharm Res 32(6):2003–2014
Ha D, Yang N, Nadithe V (2016) Exosomes as therapeutic drug carriers and delivery vehicles across biological membranes: current perspectives and future challenges. Acta Pharm Sin 6(4):287–296
Johnsen KB, Gudbergsson JM, Skov MN, Pilgaard L, Moos T, Duroux M (2014) A comprehensive overview of exosomes as drug delivery vehicles—endogenous nanocarriers for targeted cancer therapy. Biochim Biophys Acta 1846(1):75–87
Wang F, Li L, Piontek K, Sakaguchi M, Selaru FM (2018) Exosome miR-335 as a novel therapeutic strategy in hepatocellular carcinoma. Hepatology 67(3):940–954
Lou G, Song X, Yang F, Wu S, Wang J, Chen Z et al (2015) Exosomes derived from miR-122-modified adipose tissue-derived MSCs increase chemosensitivity of hepatocellular carcinoma. J Hematol Oncol 8(1):1–11
Lou G, Chen L, Xia C, Wang W, Qi J, Li A et al (2020) MiR-199a-modified exosomes from adipose tissue-derived mesenchymal stem cells improve hepatocellular carcinoma chemosensitivity through mTOR pathway. J Exp Clin Cancer Res 39(1):4
Bonnet M, Maisonial-Besset A, Zhu Y, Witkowski T, Roche G, Boucheix C et al (2019) Targeting the tetraspanins with monoclonal antibodies in oncology: focus on Tspan8/Co-029. Cancers 11(2):179
Fang T, Lin J, Wang Y, Chen G, Huang J, Chen J et al (2016) Tetraspanin-8 promotes hepatocellular carcinoma metastasis by increasing ADAM12m expression. Oncotarget 7(26):40630
Huang M-Y, Wang H-M, Chang H-J, Hsiao C-P, Wang J-Y, Lin S-RJD et al (2012) Overexpression of S100B, TM4SF4, and OLFM4 genes is correlated with liver metastasis in Taiwanese colorectal cancer patients. DNA Cell Biol 31(1):43–49
Yue S, Mu W, Erb U, Zöller M (2015) The tetraspanins CD151 and Tspan8 are essential exosome components for the crosstalk between cancer initiating cells and their surrounding. Oncotarget 6(4):2366
Elsharkasy OM, Nordin JZ, Hagey DW, de Jong OG, Schiffelers RM, Andaloussi SE et al (2020) Extracellular vesicles as drug delivery systems: why and how? Adv Drug Deliv Rev 159:332–343
Herrmann IK, Wood MJA, Fuhrmann G (2021) Extracellular vesicles as a next-generation drug delivery platform. Nat Nanotechnol 16(7):748–759
Mir B, Goettsch C (2020) Extracellular vesicles as delivery vehicles of specific cellular cargo. Cells 9(7):1601
Forner A, Reig M, Bruix J (2018) Hepatocellular carcinoma 391(10127):1301–1314
Vilgrain V, Pereira H, Assenat E, Guiu B, Ilonca AD, Pageaux G-P et al (2017) Efficacy and safety of selective internal radiotherapy with yttrium-90 resin microspheres compared with sorafenib in locally advanced and inoperable hepatocellular carcinoma (SARAH): an open-label randomised controlled phase 3 trial. Lancet Oncol 18(12):1624–1636
Nishida N, Kudo M (2018) Immune checkpoint blockade for the treatment of human hepatocellular carcinoma. Hepatol Res 48(8):622–634
Bruix J, Takayama T, Mazzaferro V, Chau G-Y, Yang J, Kudo M et al (2015) Adjuvant sorafenib for hepatocellular carcinoma after resection or ablation (STORM): a phase 3, randomised, double-blind, placebo-controlled trial. Lancet Oncol 16(13):1344–1354
Chen G, Huang AC, Zhang W, Zhang G, Wu M, Xu W et al (2018) Exosomal PD-L1 contributes to immunosuppression and is associated with anti-PD-1 response. Nature 560(7718):382–386
Lesokhin AM, Ansell SM, Armand P, Scott EC, Halwani A, Gutierrez M et al (2016) Nivolumab in patients with relapsed or refractory hematologic malignancy: preliminary results of a phase Ib study. J Clin Oncol 34(23):2698
Morris VK, Salem ME, Nimeiri H, Iqbal S, Singh P, Ciombor K et al (2017) Nivolumab for previously treated unresectable metastatic anal cancer (NCI9673): a multicentre, single-arm, phase 2 study. Lancet Oncol 18(4):446–453
Kudo M (2017) Immune checkpoint blockade in hepatocellular carcinoma: 2017 update. Liver Cancer 6(1):1–12
Singh P, Toom S, Avula A, Kumar V, Rahma OE (2020) The immune modulation effect of locoregional therapies and its potential synergy with immunotherapy in hepatocellular carcinoma. J Hepatocell Carcinoma 7:11
Longo V, Brunetti O, Gnoni A, Licchetta A, Delcuratolo S, Memeo R et al (2019) Emerging role of immune checkpoint inhibitors in hepatocellular carcinoma. Medicina 55(10):698
Xu J, Zhang Y, Jia R, Yue C, Chang L, Liu R et al (2019) Anti-PD-1 antibody SHR-1210 combined with apatinib for advanced hepatocellular carcinoma, gastric, or esophagogastric junction cancer: an open-label, dose escalation and expansion study. Clin Cancer Res 25(2):515–523
Kudo M (2018) Combination cancer immunotherapy in hepatocellular carcinoma. Liver Cancer 7(1):20
Duffy AG, Ulahannan SV, Fioravanti S, Walker M, Figg WD, Compton K et al (2014) A pilot study of tremelimumab, a monoclonal antibody against CTLA-4, in combination with either transcatheter arterial chemoembolization (TACE) or radiofrequency ablation (RFA) in patients with hepatocellular carcinoma (HCC). Am Soc Clin Oncol
Di Maria Jiang AF, Nguyen LT, Neel BG, Sacher A, Rottapel R, Wang BX et al (2019) Phase I study of local radiation and tremelimumab in patients with inoperable locally recurrent or metastatic breast cancer. Oncotarget 10(31):2947
Li J, Xing J, Yang Y, Liu J, Wang W, Xia Y et al (2018) Adjuvant 131 I-Metuximab for hepatocellular carcinoma after liver resection: an open-label, randomised, Phase 2 Trial
Li J, Xing J, Yang Y, Liu J, Wang W, Xia Y et al (2020) Adjuvant 131I-metuximab for hepatocellular carcinoma after liver resection: a randomised, controlled, multicentre, open-label, phase 2 trial. Lancet Gastroenterol Hepatol 5(6):548–560
Zhang J-C, Tian Y-Y, Yang C, Ma J-P, Jiang F-Q, Yang J et al (2018) Effect of licartin combined with pirarubicin TACE on apoptosis and epithelial-mesenchymal transition in unresectable liver cancer tissue. J Hainan Med Univ 24(2):111–114
Karzai FH, Apolo AB, Cao L, Madan RA, Adelberg DE, Parnes H et al (2015) A phase I study of TRC 105 anti-endoglin (CD 105) antibody in metastatic castration-resistant prostate cancer. BJU Int 116(4):546–555
Gordon MS, Robert F, Matei D, Mendelson DS, Goldman JW, Chiorean EG et al (2014) An open-label phase Ib dose-escalation study of TRC105 (anti-endoglin antibody) with bevacizumab in patients with advanced cancer. Clin Cancer Res 20(23):5918–5926
Duffy A, Ulahannan S, Cao L, Rahma O, Makarova-Rusher O, Kleiner D et al (2015) A phase II study of TRC105 in patients with hepatocellular carcinoma who have progressed on sorafenib. U Eur Gastroenterol J 3(5):453–461
Raghav KPS, Mody K, Greten TF, Paluri RK, Lee RT, Simpson BE et al (2018) An open label phase 1b/2 trial of TRC105 and sorafenib in patient with advanced/metastatic hepatocellular carcinoma (HCC)(NCT01806064). Am Soc Clin Oncol
Roudi R, D’Angelo A, Sirico M, Sobhani N (2021) Immunotherapeutic treatments in hepatocellular carcinoma; achievements, challenges and future prospects. Int Immunopharmacol 101:108322
Zhu AX, Finn RS, Edeline J, Cattan S, Ogasawara S, Palmer D et al (2018) Pembrolizumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib (KEYNOTE-224): a non-randomised, open-label phase 2 trial. Lancet Oncol 19(7):940–952
Li B, Yan C, Zhu J, Chen X, Fu Q, Zhang H et al (2020) Anti–PD-1/PD-L1 blockade immunotherapy employed in treating hepatitis B virus infection–related advanced hepatocellular carcinoma: a literature review. Front Immunol 11:1037
Pfister D, Núñez NG, Pinyol R, Govaere O, Pinter M, Szydlowska M et al (2021) NASH limits anti-tumour surveillance in immunotherapy-treated HCC. Nature 592(7854):450–456
Lee S, Loecher M, Iyer R (2018) Immunomodulation in hepatocellular cancer. J Gastrointest Oncol 9(1):208
Lu C, Rong D, Zhang B, Zheng W, Wang X, Chen Z et al (2019) Current perspectives on the immunosuppressive tumor microenvironment in hepatocellular carcinoma: challenges and opportunities. Mol Cancer 18(1):1–12
Choi C, Yoo GS, Cho WK, Park HC (2019) Optimizing radiotherapy with immune checkpoint blockade in hepatocellular carcinoma. World J Gastroenterol 25(20):2416
Kon E, Benhar I (2019) Immune checkpoint inhibitor combinations: current efforts and important aspects for success. Drug Resist Updates 45:13–29
Chan TA, Yarchoan M, Jaffee E, Swanton C, Quezada SA, Stenzinger A et al (2019) Development of tumor mutation burden as an immunotherapy biomarker: utility for the oncology clinic. Ann Oncol 30(1):44–56
Bernicker E (2016) Next-generation sequencing and immunotherapy biomarkers: a medical oncology perspective. Arch Pathol Lab Med 140(3):245–248
Pinter M, Trauner M, Peck-Radosavljevic M, Sieghart W (2016) Cancer and liver cirrhosis: implications on prognosis and management. ESMO Open 1(2):e000042-e
Schuch A, Hoh A, Thimme R (2014) The role of natural killer cells and CD8+ T cells in hepatitis B virus infection. Front Immunol 5:258
Moawad AW, Szklaruk J, Lall C, Blair KJ, Kaseb AO, Kamath A et al (2020) Angiogenesis in hepatocellular carcinoma; pathophysiology, targeted therapy, and role of imaging. J Hepatocell Carcinoma 7:77
Liu X, Qin S (2019) Immune checkpoint inhibitors in hepatocellular carcinoma: opportunities and challenges. Oncologist 24(Suppl 1):S3
Kudo M (2017) Molecular targeted agents for hepatocellular carcinoma: current status and future perspectives. Liver Cancer 6(2):101–112
Kudo M, Finn RS, Qin S, Han K-H, Ikeda K, Piscaglia F et al (2018) Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: a randomised phase 3 non-inferiority trial. The Lancet 391(10126):1163–1173
Cheng A-L, Finn RS, Qin S, Han K-H, Ikeda K, Piscaglia F et al (2017) Phase III trial of lenvatinib (LEN) vs sorafenib (SOR) in first-line treatment of patients (pts) with unresectable hepatocellular carcinoma (uHCC). Am Soc Clin Oncol
Personeni N, Pressiani T, Santoro A, Rimassa L (2018) Regorafenib in hepatocellular carcinoma: latest evidence and clinical implications. Drugs Context 7:212533
Zhu AX, Kang Y-K, Yen C-J, Finn RS, Galle PR, Llovet JM et al (2019) Ramucirumab after sorafenib in patients with advanced hepatocellular carcinoma and increased α-fetoprotein concentrations (REACH-2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol 20(2):282–296
Medavaram S, Zhang Y (2018) Emerging therapies in advanced hepatocellular carcinoma. Exp Hematol Oncol 7(1):17
Acknowledgements
Authors would like to express their gratitude to their colleagues in Royan institute for their support.
Funding
The authors would like to thank the Royan Institute, Tehran, Iran, for supporting this study. The study was partly supported by “National Cancer Control Charity Foundation”, to MV, registration number 41476, Tehran, IRAN. AS and PT were supported by the Ministry of Science and Higher Education of the Russian Federation within the framework of state support for the creation and development of World-Class Research Centers “Digital biodesign and personalized healthcare” (No 075-15-2020-926).
Author information
Authors and Affiliations
Contributions
MP, SA, and MV outlined the manuscript. SA and MP drafted the manuscript with primary editing and revision support from HM, PM, AM, ES, AS, HAE, PT, MH, and MV. PT, MH, and MV critically revised the manuscript for all content and coordinated the manuscript preparation and submission. All authors read and approved the final version.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest regarding the publication of this paper.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pourhamzeh, M., Asadian, S., Mirzaei, H. et al. Novel antigens for targeted radioimmunotherapy in hepatocellular carcinoma. Mol Cell Biochem 478, 23–37 (2023). https://doi.org/10.1007/s11010-022-04483-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-022-04483-4