Abstract
Context
Forest management and disturbances cause habitat fragmentation for saproxylic species living on old-growth attributes. The degree of habitat spatiotemporal continuity required by these species is a key question for designing biodiversity-friendly forestry, and it strongly depends on species’ dispersal. The “stability–dispersal” model predicts that species using stable habitats should have lower dispersal abilities than species associated with ephemeral habitat and thus respond to habitat availability at smaller scales.
Objectives
We aimed at testing the stability–dispersal model by comparing the spatial scales at which saproxylic beetle guilds using substrates with contrasted stability (from stable to ephemeral: cavicolous, fungicolous, saproxylophagous and xylophagous guilds) are affected by landscape structure (i.e. habitat amount and aggregation).
Methods
We sampled saproxylic beetles using a spatially nested design (plots within landscape windows). We quantified habitat availability (tree cavities, polypores and deadwood) in 1-ha plots, 26-ha buffers around plots and 506-ha windows, and analyzed their effect on the abundance and diversity of associated guilds.
Results
The habitat amount within plots and buffers positively affected the abundance of the cavicolous and the fungicolous guilds whereas saproxylophagous and xylophagous did not respond at these scales. The habitat aggregation within windows only positively affected the saproxylophagous species richness within plots and also on the similarity in species composition among plots.
Conclusions
Beetle guilds specialized on more stable habitat were affected by landscape structure at smaller spatial scales, which corroborated the stability–dispersal model. In managed forests, the spatial grain of conservation efforts should therefore be adapted to the target habitat lifetime.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Anthropogenic fragmentation, induced by the expansion of human activities, leads to a modification of the landscape structure (i.e. amount and spatial arrangement of habitats) which is currently considered a major threat to biodiversity (Haddad et al. 2015). It has long been recognized that the scale at which analyses are conducted can influence the detection of environmental effects on biodiversity (Wiens 1989; Levin 1992). However, the scales at which landscape variables have the strongest effect on biodiversity (i.e. “scale of effect”) are not always clear and their determinants are currently poorly known (Miguet et al. 2016).
Among several factors, dispersal ability is considered to be one of the most important mechanisms explaining response to landscape structure at species (Hanski 1999) and community levels (Cadotte 2006). Indeed, highly-dispersive organisms should be able to move more freely across the landscape, hence colonize habitats over larger distances than poorly-dispersive species. Thus, contrasted species dispersal ability, underlying different landscape perception, should lead to response at various spatial scales (D’Eon et al. 2002). In that sense, a theoretical study showed that dispersal distance strongly and positively influences the scale of effect of habitat amount on population abundance (Jackson and Fahrig 2012). More particular, by using an individual based simulation model, these authors suggested that landscape structure should be measured at a radius equal to 4–9 times the median dispersal distance of the target species. Nevertheless, few studies have empirically tested the relationship between dispersal abilities and scale of effect—especially for invertebrates (Jackson and Fahrig 2015). Besides, the relationship between landscape structure and dispersal ability may be mediated by habitat stability, including habitat-related characteristics such as year-to-year resource persistence and habitat predictability, through the “stability–dispersal” conceptual model (Southwood 1977; Travis and Dytham 1999). According to this model, species associated with ephemeral habitat (e.g. early-successional habitats) are expected to harbor better dispersal abilities and therefore should respond to landscape structure at larger spatial scale than species living on more persistent substrates (about insects, see e.g. Harrison 1980; Barbosa et al. 1989).
Spatial patterns of old-growth attributes in managed forest provide an ideal semi-natural case study for testing the stability–dispersal conceptual model. In managed forests, the distribution of old-growth attributes such as deadwood (DW) and tree-related microhabitats (TreMs) is altered due to intensive harvesting. Compared with subnatural forest ecosystems—where old-growth attributes are continuously created at fine grain through natural disturbances (Larrieu et al. 2014b), ensuring a spatiotemporal continuity of habitats for associated saproxylic species (McMullin and Wiersma 2019)—managed forest areas consist in a mosaic of simplified stands, relatively uniform in size and shape (Boutin and Hebert 2002), and characterized by reduced DW and TreM density and diversity (Lombardi et al. 2008; Larrieu et al. 2012, 2014a; Bouget et al. 2014).
Little information is currently available concerning the dispersal ability of many saproxylic taxa (e.g. flying beetles), mainly due to methodological challenges combined with the high cost to obtain dispersal estimates at the species level (review in Feldhaar and Schauer 2018; Komonen and Müller 2018). Indirect cues about dispersal are thus crucial for overcoming this empirical limitation. Studies about processes driving the response of saproxylic species to fragmentation have frequently used body size (e.g. Brin et al. 2016; Janssen et al. 2017) or wing size (e.g. Gibb et al. 2006a; Bouget et al. 2015) as proxies for flight performance and dispersal. However these phenotypic surrogates could lead to misinterpretation (see Davies et al. 2000; Ewers and Didham 2006 for body size). Using the habitat-stability conceptual framework to ordinate dispersal abilities may be a relevant alternative, especially when it comes to saproxylic species, which live on a range of habitats with contrasted stability in time. For example, hollow trees are generally considered to be stable habitats, with a persistence way above the typical harvesting rotation length of managed forests (Ranius et al. 2009), while the fruiting bodies of wood-decaying fungi are more short-lived habitats with a lifetime estimated at only a few decades or less for perennial species such as Fomes spp. (Stokland et al. 2012).
To date, only partial evidence of the relationship between saproxylic habitat stability and the scale of effect of landscape structure has been provided by previous studies. Jacobsen et al. (2015) highlighted a smaller scale of response for generalist beetles using deadwood stems from many tree species occurring throughout the forest cycle than for specialist beetles associated to aspen deadwood, which occurs only during the pioneer forest stages and thus considered as a more ephemeral habitat. Conversely, in a multi-scale study about the effect of forest amount on 31 longhorned beetles, Holland et al. (2005) did not find any support that species using more stable habitat (i.e. fresh deadwood) responded at smaller scales than species developing in more ephemeral conditions (i.e. decayed deadwood). In addition, these studies only focused on few species within taxonomical or ecological groups based on host tree preferences, restricting the generalization of results to the whole of saproxylic beetles community.
In light of the stability–dispersal conceptual framework, our objective was to compare the spatial scales at which the composition of distinct saproxylic beetle guilds using substrates with contrasted stability (from stable to ephemeral: cavicolous, fungicolous, saproxylophagous and xylophagous guilds) is affected by the availability of their habitat within a managed temperate forest in France. Using a spatially nested sampling design, we addressed the following two questions. Firstly, at what scale does the amount of surrounding habitat have an effect on the abundance and species richness of each saproxylic beetle guild within plots? We hypothesized that saproxylic beetles developing in stable habitats (e.g. the cavicolous guild) would respond to the habitat amount at smaller spatial scales than those using more ephemeral habitats (e.g. the xylophagous guild). Secondly, how does habitat aggregation within landscape windows affect the γ-diversity (species richness within a given window) and the β-diversity (dissimilarity of species composition among plots within a given windows) of each guild? We hypothesized that, for some beetle guilds (whose dispersal abilities is limited at window scale), habitat aggregation would positively affect the γ-diversity because it should generally provide species with an easier access to the totality of the habitat within windows. We expected a negative effect of habitat aggregation upon β-diversity (i.e. a greater homogenization of species composition among plots within windows where habitats are well-aggregated), because species can move more easily or “percolate” across the entire landscape and therefore occupy more plots (i.e. increase of the plot occupancy rate within windows).
Materials and methods
General description of the studied forest
The study was carried out in the French lowland temperate forest of Compiegne, 14,382 ha in area dominated by deciduous tree species (91%), particularly Fagus sylvatica (41%), Quercus robur (20%), and Q. petraea (7%) often mixed with Carpinus betulus. Most of the forest is managed under an even-aged silvicultural regime, with “mature” (i.e. 50 ≤ DBH < 70 cm) and “overmature” (i.e. DBH ≥ 70 cm) stands representing respectively 30% and 5% of its area. The spatial heterogeneity of saproxylic habitat has been increased by conservation schemes (set-aside some forest stands) and recent catastrophic events (storms, drought-induced decline), making the Compiegne forest a relevant area for study the effects of landscape structure on saproxylic beetle diversity.
Spatial distribution of saproxylic beetle habitats at forest scale
We built a distribution map of the habitats used by saproxylic beetles (i.e. TreMs and DW) over the entire forest, at the highest spatial resolution possible. For this purpose, we computed a statistical model (GLM) calibrated on field surveys (687 plots of 0.28 ha) to predict the availability of nine elementary saproxylic habitat within 30 m-pixels. The pixel represents an arbitrary spatial unit of discretization to describe and predict the distribution of habitats within forest. The pixel size (0.09 ha) was chosen to preserve the complex shape and boundaries of stands. Eight forest stand variables (five silvicultural and three ecological factors), known to affect the occurrence of saproxylic habitats, were used as predictors. We restricted the mapping to stands dominated by deciduous species. Therefore, we did not model coniferous stands where the availability of habitats was considered null. Finally, we obtained raster distribution maps over the entire forest of predicted occurrence probability per pixel for each elementary saproxylic habitat (see online supplementary material, Appendix 1 for an extensive description of saproxylic habitats, predictors, selected models, and distribution maps).
Because analyzing each elementary habitat independently would have been too complex both in terms of sampling design and in terms of data analysis, we grouped them into two broad categories: (i) cavities and (ii) deadwood plus polypores (see online supplementary material, Appendix 2 for further justification of these categories). Within a category, all the distribution maps were summed to generate a single map where pixel scores (a) correspond to the expected number of distinct elementary habitats in the pixel, which may be interpreted as a “potential habitat” value (i.e. reflects the ability of each pixel to provide saproxylic habitats). For example, for cavities, we summed the occurrence probability of trunk-base rot-hole, trunk rot-hole and woodpecker breeding cavities (see online Appendix 2 for details).
Windows selection procedure
We selected windows contrasted in terms of habitat aggregation but homogeneous in terms of habitat amount, which was fixed at an intermediate level within the existing range in the Compiegne forest. We controlled the habitat amount at window scale to intermediate values because we expected this setting to maximize the effects of habitat aggregation (Villard and Metzger 2014), which are otherwise hard to evidence. We considered windows of 506 ha (2.25 × 2.25 km). The habitat amount within windows (AMOUNT) was calculated by summing the score (a) of each pixel included in the window. The AMOUNT index for a category of habitats therefore quantified the sum of potential habitat areas over each elementary habitat included in the category. Second, an index of habitat aggregation within windows (AGGREG) was calculated based on the connectivity probability index (PC, Saura and Pascual-Hortal 2007) derived from graph theory:
where pij is the probability that an individual in pixel i can successfully disperse to pixel j and is modelled using a decreasing function of distance including a scale parameter α:
AGGREG corresponds to the probability that two elementary habitats randomly drawn from the window get connected by a dispersal process with typical distance α.
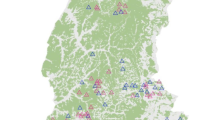
Overall, 16 windows were selected—eight for the cavity category and eight for the deadwood-polypore category (Fig. 1). We ensured that the selected windows within each category were distributed over the entire forest. Since analyses for the two habitat categories were conducted independently, window overlap between them was allowed (see online Appendix 2 for a detailed description of the selected windows).
Location of the Compiegne forest in France and description of the nested study design. Eight 506-ha landscape windows representing a habitat aggregation gradient, independently of amount, have been selected for each saproxylic habitat category—cavity (red squares) and deadwood-polypore (blue squares). Each window included six 1-ha sampling plots with a 26-ha buffer centered on plots. Grey polygons correspond to forest stands and the blue color gradient represents the potential habitat value for each pixel (maximum values are reached when the pixel is likely to have a high occurrence probability for all elementary saproxylic habitats included in the category). (Color figure online)
Saproxylic beetle sampling design
Inside the 1.75 × 1.75 km (306 ha) central area of the 16 selected windows, six sampling plots were set up in oak or beech mature high forest stands (i.e. dominant trees with DBH ≥ 50 cm). Within each plot, two unbaited transparent flight-interception traps (Polytrap™, E.I.P, Toulouse, France) were installed, one with and one without the central part colored in black (Bouget et al. 2008a). The traps were set approximately 1.5 m above the ground and 20–40 m from each other. Four monthly samples were collected from April to August 2016, and all the samples from each plot were pooled before analysis. Thus, 48 sampling plots were set up in each habitat category; however, since some plots were used for both habitat categories due to window overlap, the sampling design comprised 160 traps on 80 plots in total.
Identifying saproxylic beetles and defining ecological groups
Except for a few hard-to-identify families, which were excluded from the dataset, flying saproxylic beetles were identified to the species or genus level (see online supplementary material, Appendix 3 for a complete taxonomic list). Based on ecological traits available from the FRISBEE database (Bouget et al. 2008b), we first identified three groups according to “habitat preference”—i.e. cavicolous, fungicolous and lignicolous species, associated with cavities, wood-decaying fungi and deadwood, respectively. Then, we used “trophic regime” to distinguished lignicolous saproxylophagous species developing in decayed deadwood from xylophagous species developing in fresh deadwood. Finally, we defined four guilds along a continuum from stable to ephemeral habitat: (i) cavicolous species dwelling in stable and slowly maturing tree cavities, which we considered as “perennial stayer species” sensu Nordén and Appelqvist (2001) because we believe that such species are typically sedentary, (ii) fungicolous species associated with fruiting bodies of wood-decaying fungi (polypores) and (iii) lignicolous saproxylophagous species associated with decayed deadwood, both considered as “shuttle species” sensu Nordén and Appelqvist (2001), and (iv) lignicolous xylophagous species associated with ephemeral fresh deadwood, considered as “tracking-colonist species”.
Landscape structure metrics for biodiversity analysis at three spatial scales
At the window scale, we recorded the aggregation index (AGGREG; Eq. 1) with a scale parameter α = 5 (typical dispersal distance equal to 5 × 30 m = 150 m). Within windows, we defined an intermediate spatial scale materialized by 26-ha square buffers (510 m × 510 m) centered on each sampling plots. We computed a habitat amount index within each buffer (AMOUNT.BUF) by summing the potential habitat value of each pixel included in the buffer. At the 1-ha plot scale, we used a density index (DENSITY) directly obtained from field record of saproxylic habitats, following the same protocol as for the calibration of the predictive model of elementary saproxylic habitats distribution at the forest scale.
Statistical analyses of saproxylic beetle diversity at plot and window scale
First, we ran generalized linear mixed models (GLMMs; lme4 R-package; Bates et al. 2017) to test the response of saproxylic beetle diversity within plots to landscape structure. The dependent variables were the abundance of individuals (AB) and the species richness (SR) for each stability–dispersal guild—i.e. cavicolous, fungicolous, saproxylophagous, and xylophagous. The independent variables were AGRREG index measured at 506-ha windows scale, AMOUNT.BUF index measured at 26-ha buffer scale and DENSITY index measured at 1-ha plot scale. These three landscape structure metrics were computed for each habitat associated with beetle guilds—i.e. cavities, polypores and deadwood (Table 1). All the independent variables were standardized to a mean of 0 and a standard deviation of 1. Species richness was fitted with a Poisson distribution and abundance with a negative binomial distribution to control for over-dispersion. In the models, we have considered “windows” as spatially implicit random effects on the intercept. We analyzed the contribution of the independent variables through model averaging (“dredge” function in the MuMIn R-package; Barton 2018). Only models with ΔAIC < 2 compared to the best model were included in the estimation of coefficients. We calculated the relative importance of each independent variable by summing the Akaike weights of all plausible models where the target variable was included (“importance” function in the MuMIn R-package; Barton 2018).
Second, for each of four stability–dispersal guilds, we evaluated the effect of habitat aggregation at the window scale on the β-diversity—i.e. species composition dissimilarity between the six sampled plots within windows. A dissimilarity index was computed for each window (based on the Sorensen index) with the “beta.multi” function in the betapart R-package (Baselga et al. 2013). As a complementary analysis, we also evaluated the effect of habitat aggregation at the window scale on the average plot occupancy rate—i.e. proportion of plots in the window occupied by each species, averaged across all the species of the focal guild (high values meant that all species tended to occupy all the plots in the window, while low values meant that species tended to occur in a small number of plots in the window). At last, we evaluated whether habitat aggregation at the window scale affected the γ-diversity—i.e. species number pooled from the six sampled plots within each window. For these three analyses (plot occupancy rate, β- and γ-diversity), the significance of habitat aggregation at the window scale on the response variables was assessed with Spearman correlation tests. All statistical analyses were performed with the R software (version 3.4.1).
Results
Beetle data overview
Overall, 308 saproxylic beetle species associated to deciduous tree species were trapped, corresponding to 13,007 individuals. The numbers of species and individuals trapped in windows set for cavity habitat category were similar to those trapped in windows set for deadwood-polypore habitat category, with 272 species (8225 individuals) and 265 species (7527 individuals), respectively. In cavity category windows, we analyzed the response of the 13 cavicolous beetle species (159 individuals), whereas in deadwood-polypore category windows, we included the 44 fungicolous beetle species (1105 individuals), the 54 saproxylophagous beetle species (1928 individuals) and the 66 xylophagous beetle species (2095 individuals) (see online supplementary material, Appendix 3 for taxonomic list).
Multi-scale effects of landscape structure on α-diversity
The four stability–dispersal guilds of saproxylic beetles did not respond to the same landscape structure metrics at the same spatial scales (Table 2). Cavicolous guild abundance showed a significant positive response to the amount of cavities only at small spatial scales—i.e. 1-ha plot (β = 0.28, p = 0.03) and 26-ha buffer (β = 0.36, p = 0.02). Similarly, fungicolous guild abundance increased with the amount of polypores only at the 26-ha buffer scale (β = 0.21, p = 0.003). The saproxylophagous guild was rather sensitive to habitat aggregation at window scale. The saproxylophagous species richness in plots responded positively and significantly to an increase in deadwood aggregation within windows (β = 0.10, p = 0.03), and no significant effect was demonstrated at smaller spatial scales. Finally, regarding xylophagous beetles, no significant effect of landscape structure metrics at any spatial scale was found, either in terms of abundance or species richness.
Effect of habitat aggregation within windows on β-diversity and occupancy rate
For saproxylophagous beetles, the β-diversity analysis (i.e. species composition dissimilarity between the six sampled plots within a given windows) revealed a significant negative correlation (Spearman rank correlation coefficient rSXY = − 0.83, p = 0.02; Fig. 2) with habitat aggregation measured within each landscape window. No significant effect was detected for the other three guilds. Multi-site β-diversity appeared to be higher for cavicolous beetles, despite a considerable variation (meanCAV = 0.80, sdCAV = 0.14), than for fungicolous, saproxylophagous and xylophagous beetles (respectively, meanFUN = 0.69, sdFUN = 0.05; meanSXY = 0.67, sdSXY = 0.04; meanXYL = 0.70, sdXYL = 0.05; Fig. 2).
Effects of habitat aggregation at window scale on the β-diversity for each of the four stability–dispersal guilds. Each point represents the value of species dissimilarity index between the six sampled plots within given window. Significance was tested with a Spearman correlation test. The solid horizontal red line represents the mean and the dashed horizontal red line the standard deviation of β-diversity over the eight landscape windows. a Cavicolous, b Fungicolous, c Saproxylophagous, d Xylophagous beetles. (Color figure online)
The analysis of the average plot occupancy rate for each guild also only showed a significant effect of habitat aggregation on saproxylophagous beetles—i.e. the average number of occupied plots within windows was higher for this guild as deadwood aggregation increased (rSXY = 0.83, p = 0.02; Fig. 3). Contrary to β-diversity, the average plot occupancy rate was lower for cavicolous beetles than for the other three guilds, independently of habitat aggregation (meanCAV = 0.30, sdCAV = 0.11; meanFUN = 0.40, sdFUN = 0.05; meanSXY = 0.41, sdSXY = 0.04; meanXYL = 0.38, sdXYL = 0.05; Fig. 3).
Effects of habitat aggregation at window scale on the average plot occupancy rate for each of the four stability–dispersal guilds. Each point represents the average number (± standard error) of plots occupied by all species present in the window. Significance was tested with a Spearman correlation test. The solid horizontal red line represents the mean and the horizontal dashed red line the standard deviation of the average plot occupancy rate over the eight landscape windows. a Cavicolous, b Fungicolous, c Saproxylophagous, d Xylophagous beetles. (Color figure online)
Effect of habitat aggregation within windows on γ-diversity
We did not observe any significant effects of habitat aggregation on γ-diversity (species richness) at the window scale for the four stability–dispersal guilds (Spearman rCAV = − 0.14, p = 0.73; rFUN = 0.02, p = 0.95; rSXY = 0.34, p = 0.41; rXYL = 0.52, p = 0.19; Fig. 4). This is probably partly due to the low number of windows available to test correlations (n = 8 windows).
Discussion
Saproxylic beetles respond to landscape structure at different spatial scales due to contrasting habitat stability
One of our most important findings is that the scale of response of the four saproxylic beetle guilds (i.e. cavicolous, fungicolous, saproxylophagous and xylophagous) increases with decreased stability of their habitat. This results is consistent with the stability–dispersal conceptual framework which predict that species using ephemeral habitat should have higher dispersal abilities than species developing in more stable habitat (Southwood 1977). Particularly, with our multi-scale nested design, we showed that the cavicolous guild, which we considered as “perennial stayer species” according to the classification proposed by Nordén and Appelqvist (2001), was the only group positively affected (in terms of abundance) by habitat density within 1-ha plots. At the 26-ha buffer scale, both cavicolous and fungicolous beetles responded to habitat amount. By contrast, at window scale, only the saproxylophagous species richness showed a positive significant response to habitat aggregation whereas the others stability–dispersal guilds did not respond. Finally, we did not find any significant response of xylophagous species considered as “tracking-colonist species” to landscape structure at the three spatial scales (Table 2).
Furthermore, we found that an increase in habitat aggregation at the window scale significantly increased the similarity among plots (i.e. lower β-diversity and higher species occupancy rate) only for the saproxylophagous guild. This means that increased aggregation of deadwood facilitates the colonization of more plots by these species and contributes to the homogenization of species communities within windows. The fact that changes in habitat aggregation at the window scale affects the saproxylophagous guild species distribution and that the habitat amount at buffer scale does not affect abundance or species richness in plots tends to suggest that dispersal limitation of this group occurs somewhere between 250 m (the half of buffer side length), which seems to be an distance easy to overcome, and 2.25 km (the side length of the window) where limited dispersal seems to happen. This range is congruent with the distance of 1 km already highlighted for saproxylophagous beetles associated with oak deadwood in temperate (Franc et al. 2007) and boreal forest (Gibb et al. 2006b). By contrast, habitat aggregation did not have a significant effect on the β-diversity of the three others guilds. We suggest distinct explanations for the cavicolous guild on the one hand, and fungicolous and xylophagous guilds on the other hand. The average plot occupancy rate across cavicolous species remained at a relatively low level compared to fungicolous and xylophagous species. This suggests that increasing cavities aggregation within a 506-ha landscape did not allow cavicolous species to occupy more plots. Therefore, cavicolous species tended to stay near their habitat plots and did not seem to percolate in the window, even when habitat are well-aggregated, suggesting strong dispersal limitation. This result is congruent with previously published data about the low dispersal ability of some cavicolous beetles (Ranius 2006; Hedin et al. 2008). Conversely, for fungicolous and xylophagous species, the number of occupied plots (resp. β-diversity) remained at high level (resp. low level) even when habitat aggregation was low within windows, which suggests that they can spread in windows without distance limitation. For xylophagous beetles, this finding is consistent with the fact that we found no effect of habitat amount at smaller scales, and it globally suggests that these species are able to move quite freely within windows. These beetles are therefore likely to be sensitive to landscape structure at larger scales (> 500 ha) due to their high average dispersal ability, which clocks in at several tens of kilometers for some species (e.g. Jactel and Gaillard 1991). It is more surprising to find no effect of habitat aggregation at windows scale for fungicolous guild which was affected by habitat amount at buffer scale (in terms of abundance) but not at plot scale. Windows could thus a priori seem in the ideal range of scale of effect (sensu Jackson and Fahrig 2012) to show some sensitivity to habitat aggregation. Of course, we cannot discard the explanation that aggregation effects are studied only on eight windows, resulting in low power, and that the effect could have remained undetected. In addition, the scales of effect need not be the same for community composition (species richness, occupancy rates, Sorensen β-diversity indices), which rely on presence-absence of species, and community abundance, which rely on species abundance (Miguet et al. 2016).
Overall, our results are consistent with the predictions of the stability–dispersal conceptual model: (i) cavicolous has sufficiently low dispersal abilities to show sensitivity to the plot scale (in abundance), while other groups associated to less stable habitat do not; (ii) cavicolous and fungicolous species have sufficiently low dispersal ability to show sensitivity to the buffer scale (in terms of abundance), while other groups associated to less stable habitats do not; (iii) only saproxylophagous guild show sensitivity to habitat aggregation at window scale (in terms of species richness) while cavicolous and fungicolous guilds associated with more stable habitat do not because changes occur at too large a scale for their dispersal abilities, and the xylophagous species do not either because any configuration at window scale is equivalent for them given their strong dispersal abilities. Importantly, we emphasize that our results revealed no inconsistency such as a “gap” in the guild responses along the stability gradient that is guilds associated to very stable and very ephemeral habitats responding to some scale but guilds associated to habitat with intermediate stability not doing so.
Even if the stability–dispersal conceptual model represents an interesting theoretical framework to compare scales of response between different species guilds, it may not be systematically successful in any context and should be applied cautiously. In particular the homogeneity of species within guilds in terms of dispersal ability can be questioned. The variation in scale of response had already been observed for fungicolous beetles (Komonen 2008), even among species with similar habitat requirements (Jonsson 2003; Jonsson et al. 2003). This finding was also raised for others beetle guilds—i.e. for cavicolous beetles (from 192 to 2760 m; Ranius et al. 2011a); for saproxylophagous longhorn beetles (from 800 to 2000 m; Saint-Germain and Drapeau 2011) and for xylophagous species associated with fresh aspen deadwood (scale ranging from 10 to 1000 m; Ranius et al. 2011b).
Implications for forest management: towards a spatialized conservation strategy
Understanding species response mechanisms to landscape structure and identifying the scales of effect in relation to species dispersal ability are critical for implementing efficient biodiversity conservation strategies. In our study, we found that habitat aggregation in a logged forest matrix, with controlled intermediate habitat amount, had a positive influence on the saproxylophagous species by facilitating access to habitats. Consequently, although the amount of habitat in the landscape could be the dominant driver for saproxylic beetles (e.g. Seibold et al. 2017), our results suggests that habitat spatial arrangement should also be considered, which is congruent with a recent study on saproxylic beetles inhabiting hollow oaks (Mestre et al. 2018). As already suggested by Kouki et al. (2001) in Fennoscandian fragmented forests, aggregation of saproxylic habitat (DW, TreMs) could be improved by simultaneously setting aside forest areas (i.e. forest reserves; Bouget et al. 2014; Paillet et al. 2015; Larrieu et al. 2017) and increasing saproxylic habitat availability in the managed forest matrix (Franklin and Lindenmayer 2009).
One of the greatest challenges for taking spatial arrangement into account in conservation strategies comes from the fact that species respond to habitat aggregation at different spatial scales. According to our results, for stable habitat species (with probably low dispersal abilities), such as cavicolous beetles, it seems more relevant to concentrate protection efforts within sites where the species are already present than to scatter sites across the landscape—i.e. increase the amount of habitat in the surrounding area (Ranius et al. 2011a). By contrast, ephemeral habitat species (with probably higher dispersal abilities), such as (sapro-) xylophagous beetles, could benefit from a greater spread of stand with high quality habitat at much larger scales (Ranius and Kindvall 2006).
A second challenge for managers stems from the high species turnover among landscape windows: different landscapes harbor different species (see partitioning diversity in online supplementary material, Appendix 4). This is probably due to unmeasured environmental gradients (e.g. a sub-canopy temperature gradient in the Compiegne forest; Lenoir et al. 2017), which are the main drivers of community composition at the forest scale (i.e. inter-window scale) through a “species sorting” process (Leibold et al. 2004). As a result, landscape features present at the window scale, such as habitat aggregation, had no predictable effect on γ-diversity for our studied guilds, even for saproxylophagous species for which the window scale seemed an appropriate spatial extent (Fig. 4). Environmental gradients commonly gain in importance as the scale of analysis increases in studies of species diversity patterns (e.g. Thuiller et al. 2015). Our findings regarding high species turnover are consistent with other studies in forest contexts, which found significant β-diversity even at relatively small scales—e.g. between stands within a forest area of more than 500 ha (Müller and Goßner 2010), or between sites within a landscape of ca. 10 km2 (Rubene et al. 2015). In terms of conservation, fine-scale species sorting indicates that forest biodiversity could benefit from dispersing conservation sites spatially over the entire forest.
Conclusion
Our study highlighted that four guilds of saproxylic beetles respond to landscape structure (i.e. habitat amount and aggregation) at different spatial scales in compliance with a gradient of habitat lifetime. Therefore, the stability–dispersal model seems to be an adequate framework to analyze saproxylic beetles spatial distribution within managed forests. From an applied conservation perspective, the stability of habitats may contribute to determine the spatial scale at which conservation efforts should be aggregated for targeted guilds. For each saproxylic guild, our study suggests that an ideal network of habitat should be aggregated at the appropriate scale and cover the various environmental conditions present in the forest. Such a strategy would then achieve two objectives simultaneously: (i) improving species persistence at the fine scale by facilitating the movement of individuals among habitat units, and (ii) maximizing species diversity at larger scale (i.e. over the whole forested area). The next challenging question is now to determine whether it is possible to meet the requirements of habitat continuity for several guilds and taxa in a single conservation design. In that respect, fractal theory (Mandelbrot 1983; With 1997) offers stimulating perspectives towards creating networks of areas with high densities old-growth attributes that efficiently conciliate contrasted scales within a single design.
References
Barbosa P, Krischik V, Lance D (1989) Life-history traits of forest-inhabiting flightless Lepidoptera. Am Midl Nat 122(2):262–274
Barton Maintainer K (2018) Package “MuMIn”: multi-model inference. R-package version 1.40.4. https://cran.r-project.org/web/packages/MuMIn/
Baselga A, Orme D, Villeger S (2013) Package “betapart”: partitioning beta diversity into turnover and nestedness components. R-package version 1.2. https://cran.r-project.org/web/packages/betapart/
Bates D, Maechler M, Bolker B, Walker S, Christensen RHB, Singmann H, Dai B, Scheipl F, Grothendieck G, Green P (2017) Package “lme4”: linear mixed-effects models using “Eigen” and S4. R-package version 1.1-15. https://cran.r-project.org/web/packages/lme4/
Bouget C, Brin A, Tellez D, Archaux F (2015) Intraspecific variations in dispersal ability of saproxylic beetles in fragmented forest patches. Oecologia 177(3):911–920.
Bouget C, Brustel H, Brin A, Noblecourt T (2008a) Sampling saproxylic beetles with window flight traps: methodological insights. Rev d’Ecologie (La Terre la Vie) 63:21–32
Bouget C, Brustel H, Zagatti P (2008b) The FRench Information system on Saproxylic BEetle Ecology (FRISBEE): an ecological and taxonomical database to help with the assessment of forest conservation status. Rev d’Ecologie (La Terre la Vie) suppl. 10: 3–36. hal-00454436f
Bouget C, Parmain G, Gilg O, Noblecourt T, Nusillard B, Paillet Y, Pernot C, Larrieu L, Gosselin F (2014) Does a set-aside conservation strategy help the restoration of old-growth forest attributes and recolonization by saproxylic beetles? Anim Conserv 17(4):342–353.
Boutin S, Hebert D (2002) Landscape ecology and forest management: developing an effective partnership. Ecol Appl 12(2):390–397
Brin A, Valladares L, Ladet S, Bouget C (2016) Effects of forest continuity on flying saproxylic beetle assemblages in small woodlots embedded in agricultural landscapes. Biodivers Conserv 25(3):587–602.
Cadotte MW (2006) Dispersal and species diversity: a meta-analysis. Am Nat 167(6):913–924.
D’Eon RGD, Glenn SM, Parfitt I, Fortin M (2002) Landscape connectivity as a function of scale and organism vagility in a real forested landscape. Conserv Ecol 6(2):10
Davies KF, Margules CR, Lawrence JF (2000) Which traits of species predict population declines in experimental forest fragments? Ecology 81(5):1450–1461
Ewers RM, Didham RK (2006) Confounding factors in the detection of species responses to habitat fragmentation. Biol Rev Camb Philos Soc 81:117–142.
Feldhaar H, Schauer B (2018) Dispersal of Saproxylic Insects. In: Ulyshen MD (ed) Saproxylic insects. Zoological monographs, vol 1. Springer, Cham, pp 515–546
Franc N, Götmark F, Økland B, Nordén B, Paltto H (2007) Factors and scales potentially important for saproxylic beetles in temperate mixed oak forest. Biol Conserv 135:86–98.
Franklin JF, Lindenmayer DB (2009) Importance of matrix habitats in maintaining biological diversity. Proc Natl Acad Sci 106(2):349–350.
Gibb H, Hjältén J, Ball JP, Pettersson RB, Landin J, Alvini O, Danell K (2006a) Wing loading and habitat selection in forest beetles: Are red-listed species poorer dispersers or more habitat-specific than common congenerics? Biol Conserv 132(2):250–260.
Gibb H, Hjältén J, Ball JP, Atlegrim O, Pettersson RB, Hilszczański J, Johansson T, Danell K (2006b) Effects of landscape composition and substrate availability on saproxylic beetles in boreal forests: a study using experimental logs for monitoring assemblages. Ecography 29(2):191–204.
Haddad NM, Brudvig LA, Clobert J, Davies KF, Gonzalez A, Holt RD, Lovejoy TE, Sexton JO, Austin MP, Collins CD, Cook WM (2015) Habitat fragmentation and its lasting impact on Earth’s ecosystems. Sci Adv 1(2):1–9.
Hanski I (1999) Habitat connectivity, habitat continuity, and metapopulations in dynamic landscapes. Oikos 87(2):209–219
Harrison RG (1980) Dispersal polymorphisms in insects. Annu Rev Ecol Syst 11(1):95–118.
Hedin J, Ranius T, Nilsson SG, Smith HG (2008) Restricted dispersal in a flying beetle assessed by telemetry. Biodivers Conserv 17(3):675–684.
Holland JD, Fahrig L, Cappuccino N (2005) Body size affects the spatial scale of habitat-beetle interactions. Oikos 110(1):101–108.
Jackson HB, Fahrig L (2012) What size is a biologically relevant landscape? Landsc Ecol 27(7):929–941.
Jackson HB, Fahrig L (2015) Are ecologists conducting research at the optimal scale? Glob Ecol Biogeogr 24(1):52–63.
Jacobsen RM, Sverdrup-Thygeson A, Birkemoe T (2015) Scale-specific responses of saproxylic beetles: combining dead wood surveys with data from satellite imagery. J Insect Conserv 19(6):1053–1062.
Jactel H, Gaillard J (1991) A preliminary study of the dispersal potential of Ips sexdentatus (Boern) (Col., Scolytidae) with an automatically recording flight mill. J Appl Entomol 112:138–145.
Janssen P, Fuhr M, Cateau E, Nusillard B, Bouget C (2017) Forest continuity acts congruently with stand maturity in structuring the functional composition of saproxylic beetles. Biol Conserv 205:1–10.
Jonsson M (2003) Colonisation ability of the threatened tenebrionid beetle Oplocephala haemorrhoidalis and its common relative Bolitophagus reticulatus. Ecol Entomol 28(2):159–167.
Jonsson M, Johannesen J, Seitz A (2003) Comparative genetic structure of the threatened tenebrionid beetle Oplocephala haemorrhoidalis and its common relative Bolitophagus reticulatus. J Insect Conserv 7(2):111–124.
Komonen A (2008) Colonization experiment of fungivorous beetles (Ciidae) in a lake-island system. Entomol Tidskr 129:141–145
Komonen A, Müller J (2018) Dispersal ecology of deadwood organisms and connectivity conservation. Conserv Biol 32(3):535–545.
Kouki J, Löfman S, Martikainen P, Rouvinen S, Uotila A (2001) Forest fragmentation in Fennoscandia: linking habitat requirements of wood-associated threatened species to landscape and habitat changes. Scand J For Res suppl. 3:27–37.
Larrieu L, Cabanettes A, Delarue A (2012) Impact of silviculture on dead wood and on the distribution and frequency of tree microhabitats in montane beech-fir forests of the Pyrenees. Eur J For Res 131(3):773–786.
Larrieu L, Cabanettes A, Brin A, Bouget C, Deconchat M (2014a) Tree microhabitats at the stand scale in montane beech–fir forests: practical information for taxa conservation in forestry. Eur J For Res 133(2):355–367.
Larrieu L, Cabanettes A, Gonin P, Lachat T, Paillet Y, Winter S, Bouget C, Deconchat M (2014b) Deadwood and tree microhabitat dynamics in unharvested temperate mountain mixed forests: a life-cycle approach to biodiversity monitoring. For Ecol Manage 334:163–173.
Larrieu L, Cabanettes A, Gouix N, Burnel L, Bouget C, Deconchat M (2017) Development over time of the tree-related microhabitat profile: the case of lowland beech–oak coppice-with-standards set-aside stands in France. Eur J For Res 136(1):37–49.
Leibold MA, Holyoak M, Mouquet N, Amarasekare P, Chase JM, Hoopes MF, Holt RD, Shurin JB, Law R, Tilman D, Loreau M (2004) The metacommunity concept: a framework for multi-scale community ecology. Ecol Lett 7(7):601–613.
Lenoir J, Hattab T, Pierre G (2017) Climatic microrefugia under anthropogenic climate change: implications for species redistribution. Ecography 40(2):253–266.
Levin SA (1992) The problem of pattern and scale in ecology. Ecology 73(6):1943–1967
Lombardi F, Lasserre B, Tognetti R, Marchetti M (2008) Deadwood in relation to stand management and forest type in Central Apennines (Molise, Italy). Ecosystems 11(6):882–894.
Mandelbrot B (1983) Fractals and the geometry of nature, vol 173. WH Freeman, New-York
McMullin RT, Wiersma YF (2019) Out with OLD growth, in with ecological continNEWity: new perspectives on forest conservation. Front Ecol Environ 17(3):176–181.
Mestre L, Jansson N, Ranius T (2018) Saproxylic biodiversity and decomposition rate decrease with small-scale isolation of tree hollows. Biol Conserv 227:226–232.
Miguet P, Jackson HB, Jackson ND, Martin AE, Fahrig L (2016) What determines the spatial extent of landscape effects on species? Landsc Ecol 31(6):1177–1194.
Müller J, Goßner MM (2010) Three-dimensional partitioning of diversity informs state-wide strategies for the conservation of saproxylic beetles. Biol Conserv 143(3):625–633.
Nordén B, Appelqvist T (2001) Conceptual problems of ecological continuity and its bioindicators. Biodivers Conserv 10(5):779–791.
Paillet Y, Pernot C, Boulanger V, Debaive N, Fuhr M, Gilg O, Gosselin F (2015) Quantifying the recovery of old-growth attributes in forest reserves: a first reference for France. For Ecol Manage 346:51–64.
Ranius T (2006) Measuring the dispersal of saproxylic insects: a key characteristic for their conservation. Popul Ecol 48(3):177–188.
Ranius T, Kindvall O (2006) Extinction risk of wood-living model species in forest landscapes as related to forest history and conservation strategy. Landsc Ecol 21(5):687–698.
Ranius T, Niklasson M, Berg N (2009) Development of tree hollows in pedunculate oak (Quercus robur). For Ecol Manage 257(1):303–310.
Ranius T, Johansson V, Fahrig L (2011a) Predicting spatial occurrence of beetles and pseudoscorpions in hollow oaks in southeastern Sweden. Biodivers Conserv 20(9):2027–2040.
Ranius T, Martikainen P, Kouki J (2011b) Colonisation of ephemeral forest habitats by specialised species: beetles and bugs associated with recently dead aspen wood. Biodivers Conserv 20(13):2903–2915.
Rubene D, Schroeder M, Ranius T (2015) Diversity patterns of wild bees and wasps in managed boreal forests: effects of spatial structure, local habitat and surrounding landscape. Biol Conserv 184:201–208.
Saint-Germain M, Drapeau P (2011) Response of saprophagous wood-boring beetles (Coleoptera: Cerambycidae) to severe habitat loss due to logging in an aspen-dominated boreal landscape. Landsc Ecol 26(4):573–586.
Saura S, Pascual-Hortal L (2007) A new habitat availability index to integrate connectivity in landscape conservation planning: comparison with existing indices and application to a case study. Landsc Urban Plan 83(2):91–103.
Seibold S, Bässler C, Brandl R, Fahrig L, Förster B, Heurich M, Hothorn T, Scheipl F, Thorn S, Müller J (2017) An experimental test of the habitat-amount hypothesis for saproxylic beetles in a forested region. Ecology 98(6):1613–1622.
Southwood TRE (1977) Habitat, the templet for ecological strategies? J Anim Ecol 46(2):336–365
Stokland JN, Siitonen J, Jonsson BG (2012) Biodiversity in dead wood. Cambridge University Press, Cambridge
Thuiller W, Pollock LJ, Gueguen M, Münkemüller T (2015) From species distributions to meta-communities. Ecol Lett 18(12):1321–1328.
Travis JMJ, Dytham C (1999) Habitat persistence, habitat availability and the evolution of dispersal. Proc R Soc B Biol Sci 266(1420):723–728.
Villard MA, Metzger JP (2014) Beyond the fragmentation debate: a conceptual model to predict when habitat configuration really matters. J Appl Ecol 51(2):309–318.
Wiens JA (1989) Spatial scaling in ecology. Funct Ecol 3(4):385–397
With KA (1997) The application of neutral models in conservation biology. Conserv Biol 11(5):1069–1080
Acknowledgements
We are grateful to Carl Moliard and Benoit Nusillard for their considerable field assistance. Our grateful thanks are also extended to Thierry Noblecourt, Fabien Soldati, Thomas Barnouin and Guilhem Parmain, as well as Oliver Rose (Ciidae), Yves Gomy (Histeridae) and Benoit Nusillard (Latriidae, Curculionidae), for their valuable contribution to insect identification. We are also grateful to the agents from the National Forest Office involved in this project, particularly Vincent Boulanger, Michel Leblanc and Marguerite Delaval for their assistance in the project logistics and the provision of forest management data, as well as Nicolas Caillé and Nicolas Hilt for insect trap collection. We thank Laurent Larrieu for his helpful advice in field sampling of tree-related microhabitats, Vicki Moore for checking the English language and three anonymous reviewers for their constructive comments on previous versions of the manuscript. This work was funded by the National Research Institute of Science and Technology for Environment and Agriculture (IRSTEA) and the National Forest Office (ONF).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Percel, G., Laroche, F. & Bouget, C. The scale of saproxylic beetles response to landscape structure depends on their habitat stability. Landscape Ecol 34, 1905–1918 (2019). https://doi.org/10.1007/s10980-019-00857-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-019-00857-0