Abstract
Large-scale ecological networks (ENs) are an important mitigation measure in agriculturally transformed landscapes. However, understanding the multitude of pressures influencing the presence/absence of species, and subsequent degree of species spatial heterogeneity, is important when planning effective ENs. We aim here to measure these pressures and determine species heterogeneity in ENs against natural reference sites. We use arthropods, as they are effective bioindicators for measuring these pressures and heterogeneity, as many are habitat sensitive. Here we use many arthropod taxa to determine how a suite of variables influences the spatially sensitive grassland interior species of both EN corridors and protected areas (PAs). At each of 48 selected sites, nine stations were sampled for arthropods, with six stations in plantation block (i.e. transformed grassland) or natural indigenous forest, as well as associated edge zone and three stations in EN corridor or PA interior. Eleven variables were measured and classed into environmental, design, and current and historical management variables. Data were split into: overall data, recording all species found in interior zones, and datasets containing only species that had >50 or >75 % of their abundance sampled in the interior zone. These datasets were split into total species and singleton-removed datasets. Overall, the richness of non-singleton species, i.e. those frequently sampled in grassland interiors, were most responsive to natural background environmental variables, while design and management variables were most important for datasets with singletons retained. This means that when planning ENs, we first need to conserve the natural range of environmental heterogeneity to conserve a range of interior specialists. This natural spatial heterogeneity then needs to be incorporated into design and management planning to conserve the full range of biodiversity in ENs, as if in PAs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Habitat loss and fragmentation from anthropogenic pressures is a serious threat to biodiversity (Ewers and Didham 2006; Filgueiras et al. 2011). There is still much conversion of the natural landscape for agroforestry (Cubbage et al. 2010). This can lead to isolation of populations, which in turn, leads to the breakdown of metapopulations (Hanski 1998). Corridors are one solution to fragmentation, and have been shown to work empirically for focal taxa (Haddad 1999; Haddad and Baum 1999; Levey et al. 2005). Recently, there has been a focus on ecological networks (ENs) as a mitigation measure against biodiversity loss in highly fragmented landscapes (Jongman 1995; Beier and Noss 1998; Jongman 2004; Yu et al. 2006; Hepcan et al. 2009; Gurrutxaga et al. 2010). ENs are defined as “systems of nature reserves and their interconnections that make a fragmented natural system coherent to support more biological diversity than in its non-connected form” (Jongman 2004). Focus on ENs takes the emphasis away from single species requirements in corridors and places it more on favourable conditions across the landscape to accommodate as many species as possible. As sampling entire communities is impractical, we need to research the effectiveness of ENs using selected multiple taxa. While ENs are being designed for biodiversity maintenance across extensive landscapes, few studies have actually measured the efficacy of ENs (Boitani et al. 2007), although this is now being done for ENs associated with the South African timber industry (Pryke and Samways 2012a, b). However, there is still a dearth of information on the dispersion patterns of species across the landscape, in other words, how assemblage composition varies and whether this is the same in ENs as in reference natural areas.
These South African ENs consist mainly of remnant grasslands (ca. 80 %), with indigenous forest and waterways making up the other 20 %, totalling >500,000 ha of ENs nationally among commercial timber (Samways et al. 2010). Local regulations and laws already protect the wetlands and natural forests in the area and it is the grasslands that are under the most pressure (Kirkman and Pott 2002). Although naturally the most dominant vegetation type, grasslands are now the most threatened ecosystem in this landscape, as they have mostly been converted to plantation forestry (Mucina and Rutherford 2006). These ENs are essential, as plantation forestry using alien trees is a major threat to local biodiversity, and blocks of exotic trees contain little indigenous biodiversity (Samways and Moore 1991; Pryke and Samways 2009; Bremer and Farley 2010). In response, ENs were established to mitigate the adverse effects of plantation forestry blocks by improving connectivity between, and improving extensiveness of, natural habitats (Samways et al. 2010). However, creation of these ENs leads to more edge than would be the case naturally (Pryke and Samways 2012a). Although these grassland patches are set aside for conservation reasons, they are fire managed systems, thus frequently burned, and domestic stock grazing is permitted. These plantations have now replaced natural grasslands and as a result, we expect decline of some grassland species through either the immediate footprint of the plantation trees, or from edge effects from these alien trees. This means that a conservation priority in these transformed landscapes is focus on the species and interactions which require the interior of grassland patches (i.e. species that spend the majority of their time in the grassland interior of the ENs), as these are under the greatest spatial pressure.
The presence or absence of a species in space and time can be seen as a result of many factors, both historical and contemporary (Forman 1995). If we want to maximise the conservation value of ENs then we need to understand how biodiversity can be maintained over time inside ENs. Yet inside ENs there are many pressures that influence the abundance of the species (Samways et al. 2010; Bazelet and Samways 2011). Factors such as natural environmental gradients, EN design variables, current and historical management effects can all influence the distribution and local abundance of species (Stevens 1992; Romero-Alcaraz and Avila 2000; Lomolino 2001; Pryke and Samways 2010). How biodiversity responds to these variables will then determine where management should be directed to maintain local biodiversity.
Arthropods make a suite of effective focal taxa as they are good representatives of biodiversity. They are relatively small, hyperdiverse, can be sampled in large numbers, and some are sensitive to environmental variability at point localities (Weaver 1995; McGeogh 1998; McGeoch et al. 2011; Gerlach et al. 2013). Because they rely almost entirely on point resources, it makes them important in conservation assessment (Stork and Eggleton 1992). However, caution is required when using arthropods in biodiverse habitats such as sub-tropical grasslands. In such cases, rare species, which are often sampled as singletons or doubletons (only one or two individuals sampled in the whole dataset), can strongly influence species richness and compositional measures (Fizgerald and Carlson 2006; Barlow et al. 2010; Straatsma and Egli 2012). Also, as each arthropod species responds differently to the environmental and ecological pressures (Gerlach et al. 2013), there is a need to take a multi-taxon approach, as it is important as to record all ranges of arthropod responses (Kotze and Samways 1999; Tropek et al. 2008; Pryke and Samways 2010).
Although ENs are able to conserve arthropod diversity within the timber production landscape (Pryke and Samways 2012b), to ensure the long term success of ENs, we also need to understand how the interior and most sensitive species respond to a variety of variables. This is our particular aim here, because ultimately, such variables can be used to design ENs that best represent local biodiversity. Here we take a multi-taxon approach to specifically test how environmental, design, quality, and management variables variously influence species richness of interior assemblages. We do this by including and removing singleton species, as well as species that are tolerant of the timber plantations, namely species that are found either in the plantations or on their edges. Using conventional wisdom, we would expect the natural heterogeneity to have an influence on specific taxa (Crous et al. 2013; Pryke et al. 2013), although this would ultimately be influenced by management (Bazelet and Samways 2011) and to a lesser extent by design (Pryke and Samways 2012a). By ascertaining which are the most influential variables affecting species heterogeneity and influencing the interior’s sensitive and rare species, we can then determine which variables require focus for maximizing on biodiversity conservation within ENs.
Methods
Study area and design
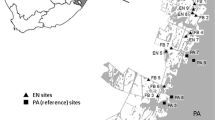
The study sites were established on three different commercial plantations across the KwaZulu-Natal Midlands, namely: Gilboa (29°16S; 30°18E), Good Hope (29°40S; 29°58E) and Maybole (29°44S; 30°15E) (Fig. 1). This area is dominated by threatened Midlands Mistbelt Grassland and Drakensberg Foothill Moist Grassland, and is generally hilly and has clay soils with rocky outcrops. These sites were burned annually. Gilboa lies ±50 km from both Good Hope and Maybole, which were ±30 km from each other. All these plantations had adjacent protected areas (PAs), here defined as large patches of natural vegetation managed by the local conservation authority. These PAs also acted as reference sites. These three plantations were also part of the local EN and had a network of corridors and nodes running through them. These sections of ENs are here described as ‘corridors’ and are managed by a commercial forestry company. All sites were 1,000–1,700 m asl.
Location map showing the three areas and the 48 transects used in this study in the KwaZulu-Natal Midlands, South Africa. Dark grey areas show the extent of the commercial plantation area, light grey areas show the extent of the natural area managed by the timber company. NF = natural forest, MaP = mature pine, Eu = mature eucalypt, MeP = medium-aged pine, YP = young pine, PA = protected area
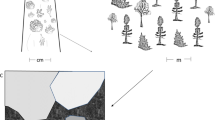
At each site, transects were set across the landscape mosaic: from plantation block or indigenous forest into a grassland corridor or into the adjoining PA. Each transect began inside the wooded area and ran across a grassland corridor or PA (Fig. 2). Nine sampling stations were placed along each transect, set out on a log2 scale, with three in the plantation block (32, 16 and 8 m from the edge), three in the edge zone (0, 8 and 16 m from the plantation block edge) and three in the interior zone (32, 63 and 128 m from the plantation block edge) (Pryke and Samways 2012a; Fig. 2). Eight replicated transects were laid out for each of the five adjacent patches (landscape feature where the transect originated): mature pine, medium aged pine, young pine, mature euclaypt, or the natural reference of indigenous forest patches running into a grassland corridor, as well as eight transects from mature pine blocks into the PA (Pryke and Samways 2012a). This gave a total of 48 transects, 144 zones and 432 sampling stations. All field work was intensive to avoid seasonal effects until >10,000 individuals were sampled and the focal species accumulation curve began to flatten out. Field work was done by three workers, with two working at any one time, during late summer between February and April 2009.
Conceptual diagram showing the placement of sampling stations along the sampling transects. The stations were grouped into two zones: wooded and edge zone; and interior zone. This interior zone distance is based on the 32 m established by (Pryke and Samways 2012a). Four sampling techniques were used at each station on the transect, namely: a 200-m diurnal search, a 100-m nocturnal search, two pitfall traps and 100 sweeps of a sweep net
Results of a Generalised Linear Mixed Model (with Poisson distribution) for the differences in species richness in response to the adjacent patches, for all species and for species which were found more than 50 or 75 % within the interior zones respectively, for all species and with singletons and doubletons removed. Ind For = indigenous forest, Mat Pine = mature pine, Med Pine = medium aged pine, PA = protected area, Y Pine = young pine. Different letters above bars represent pairwise differences, thus those without letters show no significant differences to other adjacent patches
At each station, 11 variables were measured. These were the natural environmental variables of grassland type and elevation, the EN design variables of corridor width, adjacent patches (vegetation structure of the nearest other patch), and whether sites were in a corridor or in a PA, the current management variables of vegetation height and vegetation cover, and finally, the historic management variables represented by the presence of alien woody plant species, alien grass species, increaser three grasses and decreaser grasses (Table 1). Here we regard increaser three grasses that rangeland ecologists have identified as indicators of land which has been historically overgrazed by large herbivores, while decreaser grasses are those grasses which indicate good grazing conditions and are only found in grasslands that are neither over- nor under-grazed (Rensburg and Bosch 1990; O’Connor et al. 2010).
Invertebrate sampling
At all nine stations along the 48 transects, we sampled arthropods using four sampling techniques: 200 m diurnal searches, 100 m nocturnal searches, two pitfall traps and 100 sweeps of a sweep net. No taxa were specifically chosen, although the sampling techniques favour the collection of Formicidae, Orthoptera, Araneae, Lepidoptera, Odonata, Scarabaeidae, Carabidae, Mantodea, Neuroptera, Phasmatodea and Opiliones.
Diurnal searches at each station targeted flying arthropods, between 10h00 and 15h00, on sunny, windless days. Nocturnal searches were done with search lights after 20h00, only on clear, windless nights. Both diurnal and nocturnal searches were conducted by one observer (JSP) to reduce observer bias. The observer walked 200 m for diurnal, and 100 m for nocturnal sampling, parallel to the plantation edge, recording all focal arthropods. If a specimen was not identifiable in the field, it was captured and preserved for later identification.
At each station, two pitfall traps were placed 1 m apart and each trap was 70 mm in diameter, which captures many rare species of ants (Abensperg-Traun and Steven 1995) and spiders (Brennan et al. 2005). No barriers or bait were used to ensure that all captured individuals were intercepted at the point locality. Traps were half-filled with a 50 % ethylene glycol solution (Woodcock 2005) and left open for three days at a time. Arthropods on vegetation at all stations were sampled using 100 sweeps of a 40 cm sweep net. One sweep was a single back and forth movement through the grass, rotated between three field workers. The sweep netting was also conducted parallel to the forest edge at each station along the transect. All individuals sampled were identified to species or morphospecies, and all spider vouchers are housed in the South African National Collection of Arachnida, ARC, Pretoria, South Africa, while the other vouchers are housed in the Stellenbosch University Entomological Collection, South Africa.
Data analyses
As the purpose of this study was to determine the influence of various variables on specialist interior species, only data from the interior zones of the corridors were analysed, although the wooded and edge zones were used to identify generalist species. The data were split into three sub-sets: the overall data, recording all species in the interior zones; “50 % species”, a dataset containing only species that had >50 % of their abundance sampled in the interior zone; “75 % species”, a dataset containing only species that had >75 % of their abundance sampled in the interior zone. Singletons and doubletons (henceforth just called singletons) can represent a chance sampling event that can have major effects on results, with some studies suggesting their removal from biodiversity datasets (Barlow et al. 2010). Singletons may also be non-randomly distributed (Fitzgerald and Carlson 2006), and so they would add a critical layer to the species richness and community compositional results. Here we have chosen to analyse data with all taxa (i.e. with singletons) and with taxa excluded when their abundances totalled one (singletons) or two (doubletons) (i.e. without ‘singletons’) (Wan et al. 2010). Thus, six datasets were analysed, three with all the singletons still remaining in the analyses (overall, 50 and 75 % datasets) and three with singletons removed (overall, 50 and 75 % datasets).
The relative proportion of species richness per taxon was calculated to assess how they change proportionally when only interior specialists were recorded. Changes in the relative proportion of each taxon between the overall dataset and the specialist datasets were analysed using a Likelihood Ratio Chi squared test (Legendre and Legendre 1998). The study variables were placed into the categories in Table 1, and these were then tested for covariance using Spearman’s rank order correlations (supplementary Table 1).
To test the influence that each of the factors had on species richness Generalized Linear Mixed Models (GLMM) were calculated for each of the six datasets. These GLMMs were performed in R (R Development Core Team 2007) using the lme4 package (Bates and Sarkar 2007). The overall model incorporated all the explanatory variables listed in Table 1 as fixed effects, with the three commercial plantations where sampling was conducted (i.e. Gilboa, Good Hope or Maybole) added as a random effect to the model (McCulloch et al. 2008). Furthermore, GLMM models were built to analyse the influence variable category (i.e. environmental, design, current and historical management variables; Table 1) had on species richness. These data were non-normal, although fitted a Possion curve when a Likelihood Ratio Test was performed, thus a GLMM fit by a Laplace approximation and with a Poisson distribution was used (Bolker et al. 2009). As these analyses showed no overdispersion of variances compared to the models (Pearson residuals = 1.17), a χ2 statistic and p–value were calculated (Bolker et al. 2009). Post-hoc analyses were performed on significant factors using a Tukey post hoc test in the R package multcomp (Hothorn et al. 2008).
Results
Overall, 22,588 individuals were sampled from 469 species. From the interior of corridors or PAs, 9,104 individuals were sampled from 336 species (40.3, 71.6 % of all abundance and species richness respectively). The 50 % dataset (containing only species that were sampled 50 % or more times in the corridor interiors) had 2,241 individuals from 145 species (representing 24.6 % of the abundance and 43.2 % of the species richness of all interior species). The 75 % dataset (containing only species that were sampled 75 % or more times in the corridor interiors) had 562 individuals from 71 species (representing 6.2 % of the abundance and 21.2 % of the species richness of all interior species). When singletons were removed, the overall interior dataset had 8 976 individuals from 262 species (98.6 and 78.0 % of all interior samples respectively), the 50 % dataset had 2 136 individuals from 92 species (23.5 and 27.4 % of all interior samples respectively) and the 75 % dataset had 470 individuals from 24 species (5.2 and 7.1 % of all interior samples respectively). There were proportionally more grasshoppers, dragonflies, mantids, lacewings and stick-insects in the 75 % dataset compared to the overall interior dataset (Table 2).
There were significant changes in species richness for all the datasets (overall, 50 % and 75 %), with singletons included due to the adjacent patch, vegetation cover and the presence of increaser three grasses (Table 3). Elevation gave significant differences only in species richness for the overall and 75 % datasets, while corridor width was the only significant variable for the overall dataset (Table 3). The presence of woody alien species and decreaser grass species significantly influenced species richness in the 50 % dataset only (Table 3). Although the adjacent patch had significant pairwise comparisons for all three datasets, this was a varied response, with only the indigenous forests and PAs always being significantly higher than some of the other matrices (Fig. 3). When the singletons were removed, only elevation significantly influenced all datasets (Table 3). Vegetation height influenced both the overall and 50 % dataset, while corridor width only influenced the overall dataset (Table 3); (Fig. 3) .
When the environmental variables were grouped into categories, the overall dataset with the inclusion of singletons only showed significant changes in species richness to the historical management variables, while the 50 and 75 % datasets were significantly influenced by the design, current and historical management variables (Table 4). When singletons were removed, these data were only significant for environmental variables in the 50 and 75 % datasets (Table 4). Environmental variables also significantly influenced the assemblage composition in all the datasets (including those with and without singletons) (Table 4). The overall dataset with singletons also showed compositional variation due to design variables (Table 4).
Discussion
Overall, species richness (for all species except singletons-included datasets) and were highly responsive to the natural background environmental variables of grassland type and elevation. This implies that in the case of these two variables, a range of natural landscape heterogeneity, as manifested by the character and composition of point abiotic/biotic factors (i.e. mesofilters) across the landscape need to be in place when conserving the whole range of species, common and rare. However, for datasets with arthropod singletons included, design and management variables were the most important factors. These design and management variables are particularly important for retaining the rare species, within the system of well managed interior zones. Importantly, the results showed significant changes due to elevation, grassland type and adjacent patch in both arthropod species richness. Thus, these were the three most important variables to consider in these commercially designed landscapes. As reported by Pryke and Samways (2012b), there was no significant difference between interiors of EN corridors and PAs.
Bazelet and Samways (2011) showed that for grasshoppers, management was more important than design, but here using multi-taxon datasets, we found no evidence for that. Design and management variables (which equated to maximal heterogeneity) both were important for maintaining overall biodiversity in the ENs, particularly the rare species. This underscores the importance of using a multi-taxon approach for determining the overall importance of design vis-à-vis management variables. Of the design variables, the adjacent patch was the most important, emphasizing the importance of context. The indigenous forest adjacent to grassland was important here, as it added new species to these grasslands. This underscores the importance of maintaining these natural landscape elements in association with one another (i.e. grassland and forest).
Vegetation cover had an effect on species richness, which is likely due to recent burning and grazing regimes (management variables), and so emphasises the dynamic nature of these grassland systems for arthropods. The presence of increaser three or decreaser grasses also had more effect on the presence of arthropods than did the presence of woody alien plant species which suggests that the structural changes by long term fire and grazing regimes are more important than the presence of alien plants. However, these alien plants have only recently established in these areas, and their long-term effects remain unknown.
Similar to Fitzgerald and Carlson (2006), the presence of singletons does not appear to be random, and they responded to specific environmental variables. Thus the exploration of data with and without singletons (i.e. including and excluding rare species respectively) is an appropriate way to determine how the overall assemblage changes due to specific ecological factors. The inclusion of singletons, or not, still remains embedded in the ecological question that is being asked (Wan et al. 2010). The use of singletons is appropriate for assessing subtle changes in assemblages to changing environments, whereas their removal is important when monitoring general responses of the assemblage to the environment. These singleton removed datasets suggests that these responses were driven by the overall environment, with the common species responding to similar variables. Here we determined the grassland specialist species by determining which species were more often found in the interiors of corridors of ENs and PAs. This worked well at highlighting the most important variables to consider in landscape planning.
Spatial heterogeneity has been shown to be important for many arthropods (Zulka et al. 2014), for example bees (Lentini et al. 2012), butterflies (Crous et al. 2014; Slancarova et al. 2014), dragonflies (Grant and Samways 2011) and dung beetles (Pryke et al. 2013). Yet the most important landscape planning tools are size, management, structure, etc. (Forman 1995). This disjunction between how the arthropods respond to the landscapes and how landscapes are planned is most likely due to landscape planning being based on anthropocentric concepts, whereas, the arthropods are responding to specific mesofilters/microhabitats (Hunter 2005; Crous et al. 2013). Here we did not aim to examine the role of transformed landscape heterogeneity (i.e. spatial configuration, relative size, shape, etc. of patches). Rather we focused on the degree of natural heterogeneity and the importance of incorporating it into the ecological network model, especially for conservation planning.
This natural heterogeneity is also not just restricted to the spatial aspects, as reported here, but most likely also influenced by temporal heterogeneity, especially as species use different microhabitats for different behaviours at different times of the year (Sobek et al. 2009; Dennis 2010). Furthermore, the dynamic between the spatial and the temporal requirements for species is an important consideration for ENs, as the landscape needs to provide specific resources for populations at specific times of the year for certain species to persist.
When planning the ENs within future production landscapes, we first need to consider the natural variation of the landscape (Benton et al. 2003). For this, it is critical to include a range of natural heterogeneity, particularly for the rare, dependant (on plants or other mesofilter manifestations) species in the landscape (Hunter 2005; Crous et al. 2013). Thus, the cornerstone to implementing ENs or any transformation of pristine landscapes into production landscapes is the understanding of the spatial composition of species (and their interactions), as well as the natural heterogeneity within and across the landscape. Once the natural, reference level of spatial heterogeneity is incorporated into the EN design, then other design variables, especially corridor width and adjacent habitats (Driscoll et al. 2013), as well as management procedures for these landscapes, can be implemented so as to maintain the natural level of biodiversity across the transformed landscape. This can only be determined when this heterogeneity is referenced against sites of good quality, i.e. nearby PAs. Thus, to effectively manage and design ENs, we need to maintain spatial and also, most likely, temporal heterogeneity. By integrating the dynamic of both spatial and temporal elements, there is the possibility of maintaining habitat heterogeneity over time. Such maintenance of spatial heterogeneity may be viewed as a temporal shifting mosaic of habitat patches of similar seral stages. It can be achieved by using appropriate intervention management (such as burning) where like seral stages are maintained across the landscape but not necessarily in the same locations over time (Swengel 1998).
References
Abensperg-Traun M, Steven D (1995) The effects of pitfall trap diameter on ant species richness (Hymenoptera: formicidae) and species composition of the catch in a semi-arid eucalypt woodland. Aust J Ecol 20:282–287
Barlow J, Gardner TA, Louzada J, Peres CA (2010) Measuring the conservation value of tropical primary forests: the effect of occasional species on estimates of biodiversity uniqueness. PLoS ONE 5:e9609
Bates DM, Sarkar D (2007) Lme4: linear mixed-effects models using S4 classes. R package version 0.99875-6
Bazelet CS, Samways MJ (2011) Relative importance of management vs. Design for implementation of large-scale ecological networks. Landscape Ecol 26:341–353
Beier P, Noss RF (1998) Do habitat corridors provide connectivity? Conserv Biol 12:1241–1252
Benton TG, Vickery JA, Wilson JD (2003) Farmland biodiversity: is habitat heterogeneity the key? Trends Ecol Evol 18:182–188
Boitani L, Falcucci A, Maiorano L, Rondinini C (2007) Ecological networks as conceptual frameworks or operational tools in conservation. Conserv Biol 21:1414–1422
Bolker BM, Brooks ME, Clark CJ, Geange SW, Poulsen JR, Stevens MHH, White JSS (2009) Generalized linear mixed models: a practical guide for ecology and evolution. Trends Ecol Evol 24:127–135
Bremer LL, Farley KA (2010) Does plantation forestry restore biodiversity or create green deserts? A synthesis of the effects of land-use transitions on plant species richness. Biodivers Conserv 19:3893–3915
Brennan KEC, Majer JD, Moir ML (2005) Refining sampling protocols for inventorying invertebrate biodiversity: influence of drift-fence length and pitfall trap diameter on spiders. J Arachnol 33:681–702
Crous CJ, Samways MJ, Pryke JS (2013) Exploring the mesofilter as a novel operational scale in conservation planning. J Appl Ecol 50:205–214
Crous CJ, Samways MJ, Pryke JS (2014) Rockiness determines meso-scale conservation of butterflies in Afro-montane grassland. J Insect Conserv 18:77–83
Cubbage F, Koesbandana S, Mac Donagh P, Rubilar R, Balmell G, Olmos VM, De La Torre R, Murara M, Hoeflich VA, Kotze H, Gonzalez R, Carrero O, Frey G, Adams T, Turner J, Lord R, Huang J, MacIntyre C, McGinley K, Abt R, Phillips R (2010) Global timber investments, wood costs, regulation, and risk. Biomass Bioenerg 34:1667–1678
Dennis RHL (2010) A resource-based habitat view for conservation. Wiley-Blackwell, Oxford
Driscoll DA, Banks SC, Barton PS, Lindenmeyer DB, Smith AL (2013) Conceptual domain of the matrix in fragmented landscapes. Trends Ecol Evol 28:605–623
Ewers RM, Didham RK (2006) Confounding factors in the detection of species responses to habitat fragmentation. Biol Rev 81:117–142
Filgueiras BKC, Iannuzzi L, Leal IR (2011) Habitat fragmentation alters the structure of dung beetle communities in the Atlantic forest. Biol Conserv 144:362–369
Fitzgerald P, Carlson S (2006) Examining the latitudinal diversity gradient in Paleozoic terebratulide brachiopods: should singleton data be removed? Palebiology 32:367–386
Forman RTT (1995) Land mosaics: the ecology of landscapes and regions. Cambridge University Press, Cambridge
Gerlach J, Samways MJ, Pryke JS (2013) Terrestrial invertebrates as bioindicators: an overview of available taxonomic groups. J Insect Conserv 17:831–850
Grant PBC, Samways MJ (2011) Micro-hotspot determination and buffer zone value for Odonata in a globally significant reserve. Biol Conserv 144:772–781
Gurrutxaga M, Lozano PJ, Del Barrio G (2010) GIS-based approach for incorporating the connectivity of ecological networks into regional planning. J Nature Conserv 18:318–326
Haddad NM (1999) Corridor and distance effects on interpatch movements: a landscape experiment with butterflies. Ecol Appl 9:612–622
Haddad NM, Baum KA (1999) An experimental test of corridor effects on butterfly densities. Ecol Appl 9:623–633
Hanski I (1998) Metapopulation dynamics. Nature 396:41–50
Hepcan S, Hepcan CC, Bouwma IM, Jongman RHG, Ozkan MB (2009) Ecological networks as a new approach for nature conservation in Turkey: a case study of Izmir Province. Landsc Urban Plann 90:143–154
Hothorn T, Bretz F, Westfall P (2008) Simultaneous inference in general parametric models. Biom J 50:346–363
Hunter ML (2005) A mesofilter conservation strategy to complement fine and coarse filters. Conserv Biol 19:1025–1029
Janse van Rensburg FP, Bosch OJH (1990) Influence of habitat differences on the ecological grouping of grass species on a grazing gradient. J Grassl Soc Southern Afr 7:11–15
Jongman RHG (1995) Nature conservation planning in Europe: developing ecological networks. Landsc Urban Plann 32:169–183
Jongman RHG (2004) European ecological networks and greenways. Landsc Urban Plann 68:305–319
Kirkman KE, Pott RM (2002) Biodiversity conservation in plantation forestry. In: Pierce SM, Cowling RM, Sandwith T, MacKinnon K (eds) Mainstreaming biodiversity in development—case studies from South Africa. The World Bank Environmental Department, Washington DC, pp 33–42
Kotze DJ, Samways MJ (1999) Support for the multi-taxa approach in biodiversity assessment, as shown by epigaeic invertebrates in an Afromontane forest archipelago. J Insect Conserv 3:125–143
Legendre P, Legendre L (1998) Numerical ecology, 2nd edn. Elsevier, Amsterdam
Lentini PE, Martin TG, Gibbons P, Fischer J, Cunningham SA (2012) Supporting wild pollinators in a temperate agricultural landscape: maintaining mosaics of natural features and production. Biol Conserv 149:84–92
Levey DJ, Bolker BM, Tewksbury JJ, Sargent S, Haddad NM (2005) Effects of landscape corridors on seed dispersal by birds. Science 309:146–148
Lomolino MV (2001) Elevation gradients of species-density: historical and prospective views. Global Ecol Biogeog 10:3–13
McCulloch CE, Searle SR, Neuhaus JM (2008) Generalized. Linear, and Mixed Models, 2nd edn. John Wiley and Sons, Inc, New Jersey
McGeoch MA, Sithole H, Samways MJ, Simaika JP, Pryke JS, Picker M, Uys C, Armstrong AJ, Dippenaar-Schoeman AS, Engelbrecht IA, Braschler B, Hamer M (2011) Conservation and monitoring of invertebrates in terrestrial protected areas. Koedoe 53:137–149
McGeogh MA (1998) The selection, testing and application of terrestrial insects as bioindicators. Biol Rev 73:181–201
Mucina L, Rutherford MC (2006) The vegetation of South Africa, Lesotho and Swaziland. Strelitzia 19. South African National Biodiversity Institute, Pretoria
O’Connor TG, Kuyler P, Kirkman KP, Corcoran B (2010) Which grazing management practices are most appropriate for maintaining biodiversity in South African grassland? Afri J Range Forage Sci 27:67–76
Pryke JS, Samways MJ (2009) Recovery of invertebrate diversity in a rehabilitated city landscape mosaic in the heart of a biodiversity hotspot. Landsc Urban Plann 93:54–62
Pryke JS, Samways MJ (2010) Significant variables for the conservation of mountain invertebrates. J Insect Conserv 14:247–256
Pryke JS, Samways MJ (2012a) Conservation management of complex natural forest and plantation edge effects. Landscape Ecology 27:73–85
Pryke JS, Samways MJ (2012b) Ecological networks act as extensions of protected areas for arthropod biodiversity conservation. J Appl Ecol 49:591–600
Pryke JS, Roets F, Samways MJ (2013) Importance of habitat heterogeneity in remnant patches for conserving dung beetles. Biodivers Conserv 22:2857–2873
Romero-Alcaraz E, Avila J (2000) Landscape heterogeneity in relation to variations in epigaeic beetle diversity of a Mediterranean ecosystem implications for conservation. Biodivers Conserv 9:985–1005
Samways MJ, Moore SD (1991) Influence of exotic conifer patches on grasshopper (Orthoptera) assemblages in a grassland matrix at a recreational resort, Natal, South-Africa. Biol Conserv 57:117–137
Samways MJ, Bazelet CS, Pryke JS (2010) Provision of ecosystem services by large scale corridors and ecological networks. Biodivers Conserv 19:2949–2962
Slancarova J, Benes J, Kristynek M, Kepka P, Konvicka M (2014) Does the surrounding landscape heterogeneity affect the butterflies of insular grassland reserves? A contrast between composition and configuration. J Insect Conserv 18:1–12
Sobek S, Steffan-Dewenter I, Scherber C, Tscharntke T (2009) Spatiotemporal changes of beetle communities across a tree diversity gradient. Divers Distrib 15:660–670
Stevens GC (1992) The elevational gradient in altitudinal range - an extension of Rapoport latitudinal rule to altitude. Am Nat 140:893–911
Stork NE, Eggleton P (1992) Invertebrates as determinants and indicators of soil quality. Am J Altern Agri 7:38–47
Straatsma G, Egli S (2012) Rarity in large data sets: singletons, modal values and the location of the species abundance distribution. Basic Appl Ecol 13:380–389
Swengel AB (1998) Effects of management on butterfly abundance in tallgrass prairie and pine barrens. Biol Conserv 83:77–89
Tropek R, Spitzer L, Konvicka M (2008) Two groups of epigeic arthropods differ in colonising of piedmont quarries: the necessity of multi-taxa and life-history traits approaches in the monitoring studies. Commun Ecol 9:177–184
Wan H, Chizinski CJ, Dolph CL, Vondracek B, Wilson BN (2010) The impact of rare taxa on a fish index of biotic integrity. Ecol Indicator 10:781–788
Weaver JC (1995) Indicator species and scale of observation. Conserv Biol 9:939–942
Woodcock BA (2005) Pitfall trapping in ecological studies. In: Leather S (ed) Insect Sampling in Forest Ecosystems. Blackwell Science, Oxford, pp 37–57
Yu K, Li D, Li N (2006) The evolution of greenways in China. Landsc Urban Plann 76:223–239
Zulka KP, Abensperg-Traun M, Milasowszky N, Bieringer G, Gereben-Krenn B-A, Holzinger W, Hölzler G, Rabitsch W, Reischütz A, Querner P, Sauberer N, Schmitzberger I, Willner W, Thomas Wrbka T, Zechmeister H (2014) Species richness in dry grassland patches of eastern Austria: a multi-taxon study on the role of local, landscape and habitat quality variables. Agri Ecosys Environ 182:25–36
Acknowledgments
We thank Ezemvelo KZN Wildlife and Mondi South Africa for permitting sampling on their holdings. Funding was from the Mondi Ecological Network Programme (MENP) and co-funding from the National Research Foundation of South Africa (NRF).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pryke, J.S., Samways, M.J. Conserving natural heterogeneity is crucial for designing effective ecological networks. Landscape Ecol 30, 595–607 (2015). https://doi.org/10.1007/s10980-014-0096-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-014-0096-x