Abstract
A combined chromatographic columns including LN resin and P204 resin were fabricated to separate Th4+, La3+ and Ba2+ ions with a series of parallel experiments at different acidity and initial concentrations. The results show that LNS resin can selectively adsorb Th4+ ions but not La3+ and Ba2+ ions in 1 mol/L HNO3, and the separation factor of Th/La and Th/Ba can reach up to 8.260 × 104 and 8.224 × 105, respectively. The La3+ and Ba2+ ions can be separated by P204 resin at pH = 3 with a separation factor of 2.191 × 104. The successful separation and enrichment of La in this separation system provides a possibility for the separation of Ac, which is of great significance to TAT.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Nuclear medicine has great application value in both diagnosis and treatment [1,2,3]. Radionuclides used for diagnosis or treatment are typically attached to selective biomolecules (peptides, antibody fragments, or intact antibodies) to deliver imaging or therapeutic doses to targets in vivo (tumors or other tissues). Targeted alpha therapy (TAT) is a rapidly developing field that alpha radionuclides are used selectively to deliver radiation doses capable of killing cells to tumor sites [4]. Low penetration range (50–90 μm) and high linear energy transfer of α-particles (tens to hundreds of keV/μm) are able to destroy cancer cells to the maximum with minimal damage to surrounding healthy tissue. 225Ac (t1/2 = 9.92 d) has significant potential as a TAT nuclide because its favorable half-life and decay cascade including multiple short-lived alpha nuclides [5, 6]. For example, 225Ac can not only be directly used for therapy by binding with biological targeting vectors, but also the nuclide 213Bi (t1/2 = 45.6 min) produced by 225Ac decay can be used for targeted therapy through chelation or complexation in vivo. Currently, the main source of 225Ac is the decay of the parent body 229Th (t1/2 = 7340 a), which was obtained early from a 233U reactor as fuel [7]. However, the global supply of 225Ac created by decay does not exceed 63 GBq (1.7 Ci) per year, which cannot meet the current demand for 225Ac [8]. Therefore, it is urgent to explore a new method of mass production of 225Ac.
Different groups, including Los Alamos National Laboratory (LANL), have investigated alternative methods for producing 225Ac [9,10,11]. One of the most promising pathways is thought to be the 232Th(p, x) nuclear reaction via the activation of 232Th by a medium-energy (> 70 MeV) proton. Data obtained by LANL indicate that irradiation of 232Th with a proton of 250 μA, 100 MeV energy for 10 days yields 225Ac of 73.2 GBq (1.98 Ci) [9]. This amount exceeds the accumulation of 225Ac prepared by 229Th by research departments around the world. Although significant quantities of 225Ac can be produced from the 232Th(p, x) nuclear reaction, separating actinium from irradiated thorium targets is a challenge. The system of 232Th(p, x) nuclear reaction mainly contains a large amount of Th, minor amounts of Ac and Ra, and trace amounts of other radioactive elements [9]. On the one hand, the concentration difference of thorium and actinium is several orders of magnitude, because thorium and actinium are the relationship between the decay parent and the second generation of the child. On the other hand, thorium, also an actinide element, has similar chemical properties to actinium. Therefore, it is necessary to seek an efficient and rapid method for the chemical separation and purification of Th, Ac and Ra, which not only provides a basis for the research and development of TAT, but also has great significance for the development of radionuclide drugs. The methods for separating Ac contain precipitation method [12], liquid–liquid extraction method [13, 14], extraction column [11, 15], ion exchange method [7], adsorption method [16] and so on. Compared to other separation techniques, chromatographic column separation has the high efficiency and speed of column separation [17]. Here, a combined chromatographic column was constructed to achieve Th separation and Ac enrichment by a two-step separation method [18]. Since actinium and radium are highly radioactive and difficult to obtain, Ac and Ra can be replaced by La and Ba with similar properties in the reported literature [19]. V. Ostapenko et al. found that commercial series LN resin had excellent separation effect on rare earth elements and actinides [20]. In addition, P204 resin can achieve the separation of Sr and Y, so it is speculated that it also has the separation effect on La and Ba of the same main group [21, 22].
This paper describes the separation of Th4+, La3+ and Ba2+ ions using a combine chromatographic column with LN resin and P204 resin. The advantages of this separation are that the effect of concentration ratio on separation is investigated, which has not been seen in previous articles, meanwhile the three-ion separation can be accomplished in a simple process. The novelty of this paper mainly lies in the establishment of a set of separation system by which Ac can be conveniently and quickly separated and enriched from Th and Ra.
Experiment
Materials
The thorium standard solution was obtained from Beijing North Weiye Metrology Technology Research Institute. The single-element standard solution of lanthanum and barium was purchased from the National Nonferrous Metals and Electronic Materials Analysis Center. LN series resin was provided by TrisKem International. P204 resin was supplied by Lanzhou University. Other reagents were purchased from Tianjin Damao Chemical Reagent Factory. All reagents are analytical reagent grade and can be used directly without further purification. All tests were performed using deionized water unless otherwise noted.
Setting the chromatographic column
The resin is immersed in deionized water to remove impurities, ensuring that the volume of the deionized water is twice the volume of the resin. The pre-treated resin was prepared into a suspended water solution and transferred to the glass dropper with a length of 0.15 m and a diameter of 6 mm (Fig. 1). A microporous sheet of teflon is placed at the bottom of the glass dropper to avoid resin loss. The column was filled with wet packing method. Finally, another microporous sheet of teflon is placed on top of the packed resin, which prevents the resin from being dispersed. The feed rate should be controlled to ensure stable column pressure during the addition of the stock solution, which facilitates full contact between the solution and the resin.
Separation of Th4+ ions with LN resin
The solution with different concentration ratio (Th: La: Ba) was configured under the condition of 1 mol/L HNO3. Firstly, 20 mL of 1 mol/L HNO3 was used to flush the loaded LN column, then a mixture solution of Th4+, La3+ and Ba2+ ions was poured into the separation funnel and passed through the LN column at a flow rate of 1 mL/min, meanwhile, the outflow was collected sequentially in 5 mL containers. The ion concentration in the outflow was measured by inductively coupled plasma emission spectrometer (ICP-OES, PQ9000, Germany). The distribution coefficient (Kd) and the separation factor (SF) were calculated according to the volume of outflow liquid and the ion concentration of outflow liquid to explore the separation performance of resin. The Kd and the SF were calculated according to the following equation:
where C0, Ce represent the concentration of metal ions at initial and equilibrium conditions (mg/L), V emblems the volume of the solution (L), and m is the mass of adsorbent (g), respectively.
Separation of La3+ ions with P204 resin
The column containing P204 resin was rinsed with 20 mL of nitric acid solution with the same acidity as the feed solution. The mixed solution containing La3+ and Ba2+ ions breaks through the column, and the outflow was collected at intervals with the volume of 5 mL. ICP-OES was performed to test the ion concentration in the effluent. Kd and SF were calculated to optimize the optimal separation conditions. To further separate and enrich La3+ ions, 1 mol/L HNO3 was used to elute La3+ ions adsorbed in the P204 column, still 5 mL fraction collected and La3+ ions concentration measured by ICP-OES.
The overall adsorption flow chart is shown in Fig. 2.
Results and discussion
Optimization of resin for separation of Th4+, La3+ and Ba2+ ions
Figure 3 displays the separation effect of Th, La, and Ba on an ion chromatographic column containing LNA resin and P204 resin, respectively. As shown in Fig. 3a, LNA resin only adsorbed Th4+ ions in 1 mol/L HNO3, while La3+ and Ba2+ ions were adsorbed hardly. The calculated Kd and SF values of LNA resin and P204 resin for Th, La and Ba are listed in Table 1. Among them, SFTh/La and SFTh/Ba reach up to 6.423 × 104 and 1.711 × 105, respectively, which is superior to some previous reports (Table 2) [23, 24]. As displayed in Fig. 3b, P204 resin not only can adsorb obviously Th4+ and La3+ ions, but also can trap a small amount of Ba2+ ions, implying that P204 cannot realize the separation of the three elements. P204 is a kind of acidic phospholipid extraction agent, and usually present in the form of dimer (H2L2) in non-polar solvent[25] which can form complex though cation exchange, chelation with high valence states ions is similar, so it is difficult to separate Th4+ and La3+ ions. For LN resin, which is used for actinide lanthanide separation has a similar mechanism The adsorption mechanism of P204 and LN resin in the acid environment is generally described as Eqs. (3) and (4) [20, 26], respectively. The difference of the adsorption effect of the two resins on metal ions can be attributed to the different complexing ability of different cations and ligands under different acidity conditions.
Therefore, LN resin was selected to separate Th4+ ions from mixed solution, and subsequent experiments were performed in 1 mol/L HNO3 to explore the separation effect of LN resin on Th4+, La3+, and Ba2+ under different initial concentration ratios.
Optimization of concentration on LN resin for separation of Th4+, La3+ and Ba2+ ions
In the 232Th(p, x) nuclear reaction, only a trace amount of Ac can be obtained after irradiation, leading to a difference of several orders of magnitude in the content of Th and Ac. In order to enrich and separate trace amounts of Ac and Ra in a large amount of Th, it is necessary to explore the influence of the initial concentration ratio of Th: La: Ba on the separation of three metal ions.
In the actual separation system, the content of thorium is very different from that of actinide, so it is particularly important to study the separation of thorium and actinide under different initial concentration ratios. LNA resin and LNS resin of LN series were selected to investigate the separation performance for Th4+, La3+ and Ba2+ ions by adjusting the initial concentration ratio (Fig. 4). The particle sizes of LNA resin and LNS resin are 100–150 μm and 50–100 μm, respectively. When the initial concentration ratio of Th4+: La3+: Ba2+ is 28.50 ppm: 1.44 ppm: 1.05 ppm, LNA resin and LNS resin only adsorb Th4+ ions, but not La3+ and Ba2+ ions (Fig. 4a, b). The calculated SFTh/La and SFTh/Ba values of LNA resin and LNS resin are higher than 104, suggesting that Th(IV) separation can be successfully achieved (Table 3). It is worth noting that the SF values calculated by LNS resin separation are slightly higher than those of LNA resin, which means that LNS resin possesses better separation performance for Th4+, La3+ and Ba2+ ions. With the increase of Th4+ proportion (Th: La: Ba = 100 ppm: 0.897 ppm: 0.931 ppm), the SF values by LNA resin and LNS resin were all over 104, indicating that Th(IV) separation could still be achieved well (Fig. 4c, d and Table 3). As displayed in Fig. 4e, when concentration ratio of Th: La: Ba is 500 ppm: 1.00 ppm: 1.10 ppm, When the initial solution of Th, La and Ba with high concentration difference still flowed at the original flow rate, the LNA resin with large particle size had certain adsorption effect on Th(IV) but could not completely adsorb it. In addition, it still had no adsorption effect on La(III) and Ba(II). Whereas, the LNS resin was still able to completely adsorb Th4+, but not La3+ and Ba2+ ions at all (Fig. 4f). The SFTh/La and SFTh/Ba values by LNS resin can reach up to 8.260 × 104 and 8.224 × 105, respectively, which be superior to the SFTh/La (13.29) and SFTh/Ba (16.25) values by LNA resin. The above results indicate that the resin particle size has obvious effect, the smaller the resin particle size, the better the separation performance of Th4+, La3+ and Ba2+ ions.
Effect of different concentrations on LN resin for separation of Th4+, La3+ and Ba2+ ions. a LNA resin, b LNS resin (Th: La: Ba = 28.50 ppm: 1.44 ppm: 1.05 ppm,acidity: 1 mol/L HNO3, mLN = 573 mg); c LNA resin, d LNS resin (Th: La: Ba = 100 ppm: 0.897 ppm: 0.931 ppm,acidity: 1 mol/L HNO3, mLN = 573 mg); e LNA resin, f LNS resin (Th: La: Ba = 500 ppm: 1.00 ppm: 1.10 ppm, acidity: 1,mol/L HNO3, mLN = 573 mg)
Optimization of acidity on P204 resin for separation of La3+ and Ba2+ ions
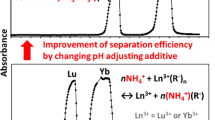
P204 was selected as the resin for the separation of La3+ and Ba2+ ions due to its strong adsorption capacity for La3+ ions in previous experiments. Meanwhile, in order to better reflect the adsorption performance of P204 resin on La3+ ions, the initial concentration of La3+ and Ba2+ ions were increased to 10 ppm under the condition that the ratio of La3+ and Ba2+ ions was kept at 1:1. Figure 5 shows the separation performance of P204 resin for La3+ and Ba2+ ions under different acidity. The separation factors calculated by P204 resin disposing of La3+ and Ba2+ ions at different acidity are listed in Table 4. It is clearly observed that P204 resin has a comparatively higher SFLa/Ba at pH = 3–5, which is attributed to the high pH promoting ion exchange, suggesting that P204 resin can successfully achieve the separation of La3+ and Ba2+ ions.
Effect of Al3+ on the adsorption of La3+ by P204 resin
It is worth noting that a small amount of aluminum is produced by fission during the 232Th(p, x) nuclear reaction. Al will interfere with the separation of Ac due to the same valence, while the interference with Th and Ra can be ignored [29].Therefore, it is necessary to separately explore the adsorption properties of P204 on La3+ ions in the presence of Al3+ ions. As shown in Fig. 6a, the P204 resin can still completely adsorb La3+ ions in the presence of Al3+ ions, but unfortunately some Al3+ ions are also retained in P204 resin, which is not conducive to the enrichment and purification of La3+ ions. This is precisely because of the adsorption mechanism of P204 resin which has a certain degree of adsorption of trivalent elements under this acidity condition. Nonetheless, La3+ ions can be quickly eluted and Al3+ ions are still adsorbed on the resin using 1 mol/L HNO3 as the eluent. In conclusion, La3+ ions can be successfully enriched with LN resin and P204 resin by step separation method.
Conclusions
Combined columns were assembled using LN resin and P204 resin for Th(IV) separation and La(III) enrichment, respectively. The results show that LNS resin can selectively adsorb Th4+ ions at the initial concentration ratio of Th: La: Ba = 500 ppm: 1.00 ppm: 1.10 ppm with high separation ratio of SFTh/La and SFTh/Ba of 8.260 × 104 and 8.224 × 105, respectively. In addition, P204 resin could only absorb La3+ ions at pH = 3, leading to SFLa/Ba up to 2.191 × 104. Even with the interference of Al3+ ion, P204 resin can still enrich La3+ ion by elution. Although both La3+ and Al3+ ions were simultaneously retained on the P204 resin, La3+ ions were subsequently eluted with 1 mol/L HNO3 to achieve La(III) separation and enrichment. La3+ ions obtained by combined LNS/P204 resin chromatographic column have high radionuclide purity, which provides a possibility for the separation of high purity radiopharmaceutical nuclide Ac from the activation of 232Th by a medium-energy (> 70 MeV) proton.
References
Bhattacharyya S, Dixit M (2011) Metallic radionuclides in the development of diagnostic and therapeutic radiopharmaceuticals. Dalton Trans 40:6112–6128
Cutler CS, Hennkens HM, Sisay N, Huclier-Markai S, Jurisson SS (2013) Radiometals for combined imaging and therapy. Chem Rev 113:858–883
Rosch F, Baum RP (2011) Generator-based PET radiopharmaceuticals for molecular imaging of tumours: on the way to THERANOSTICS. Dalton Trans 40:6104–6111
Maalouf BN (2012) Targeted use of alpha particles: current status in cancer therapeutics. J Nucl Med 3(4):1000136
Morgenstern A, Bruchertseifer F, Apostolidis C (2012) Bismuth-213 and actinium- 225–generator performance and evolving therapeutic applications of two generator-derived alpha-emitting radioisotopes. Curr Radiopharm 5(3):221–227
Scheinberg DA, McDevitt MR (2011) Actinium-225 in targeted alpha-particle therapeutic applications. Curr Radiopharm 4(4):306–320
Boll RA, Malkemus D, Mirzadeh S (2005) Production of actinium-225 for alpha particle mediated radioimmunotherapy. Appl Radiat Isot 62:667–679
Chérel M, Barbet J (2013) Alpha emitting radionuclides and radiopharmaceuticals for therapy
Engle JW, Mashnik SG, Weidner JW, Wolfsberg LE, Fassbender ME, Jackman K, Couture A, Bitteker LJ, Ullmann JL, Gulley MS, Pillai C, John KD, Birnbaum ER, Nortier FM (2013) Cross sections from proton irradiation of thorium at 800 MeV. Phys Rev C 88:014604
Weidner JW, Mashnik SG, John KD, Ballard B, Birnbaum ER, Bitteker LJ, Couture A, Fassbender ME, Goff GS, Gritzo R, Hemez FM (2012) 225Ac and 223Ra production via 800 MeV proton irradiation of natural thorium targets. Appl Radiat Isot 70(11):2590–2595
Zhuikov BL, Kalmykov SN, Ermolaev SV, Aliev RA, Kokhanyuk VM, Matushko VL, Tananaev IG, Myasoedov BF (2011) Production of 225Ac and 223Ra by irradiation of Th with accelerated protons. Radiochemistry 53:73–80
Sani AR (1970) Carrier-free separation of 228Ac from aged thorium nitrate. J Radioanal Nuc Chem 4:127–129
Hyde EK (1960) The radiochemistry of thorium, National technical information service, US. Department of commerce, Springfield, VA, pp. 22161
Tsoupko-Sitnikov V, Norseev Y, Khalkin V (1996) Generator of 225Ac. J Rad Nucl Chem 205:75–83
Hu Y, Giret S, Meinusch R, Han J, Fontaine F-G, Kleitz F, Larivière D (2019) Selective separation and preconcentration of Th(IV) using organo-functionalized, hierarchically porous silica monoliths. J Mater 7:289–302
Brown MA (2020) Metal oxide sorbents for the separation of radium and actinium. Ind Eng Chem Res 59:20472–20477
Coskun O (2016) Separation techniques: chromatography. North Clin Istanb 3:156–160
Radchenko V, Engle JW, Wilson JJ, Maassen JR, Nortier FM, Taylor WA, Birnbaum ER, Hudston LA, John KD, Fassbender ME (2015) Application of ion exchange and extraction chromatography to the separation of actinium from proton-irradiated thorium metal for analytical purposes. J Chromatogr A 1380:55–63
Fitzsimmons J, Abraham A, Catalano D, Younes A, Cutler CS, Medvedev D (2019) Evaluation of inorganic ion exchange materials for purification of 225Ac from thorium and radium radioisotopes. JMIRS 50(1):11
Ostapenko V, Vasiliev A, Lapshina E, Ermolaev S, Aliev R, Totskiy Y, Zhuikov B, Kalmykov S (2015) Extraction chromatographic behavior of actinium and REE on DGA, Ln and TRU resins in nitric acid solutions. J Radioanal Nucl Chem 306:707–711
Kandwal P, Ansari SA, Mohapatra PK, Manchanda VK (2011) Separation of carrier free 90Y from 90Sr by hollow fiber supported liquid membrane containing Bis(2-ethylhexyl) phosphonic acid. Sep Sci Technol 46:904–911
Naik PW, Jagasia P, Dhami PS, Achuthan PV, Tripathi SC, Munshi SK, Dey PK, Venkatesh M (2010) Separation of carrier-free 90Y from 90Sr by SLM technique using D2EHPA in N-Dodecane as carrier. Sep Sci Technol 45:554–561
Eskandari NM, Milani SA, Sam A (2011) Extractive separation of Th(IV), U(VI), Ti(IV), La(III) and Fe(III) from Zarigan ore. J Radioanal Nucl Chem 288:677–683
Xiong Y, Gao Y, Guo X, Wang Y, Su X, Sun X (2018) Water-stable metal-organic framework material with uncoordinated terpyridine site for selective Th(IV)/Ln(III) separation. ACS Sustain Chem Eng 7:3120–3126
Principe F, Demopoulos GP (2004) Comparative study of iron(III) separation from zinc sulphate–sulphuric acid solutions using the organophosphorus extractants, OPAP and D2EHPA. Hydrometallurgy 74:93–102
Ye Q, Li G, Deng B, Luo J, Rao M, Peng Z, Zhang Y, Jiang T (2019) Solvent extraction behavior of metal ions and selective separation Sc3+ in phosphoric acid medium using P204. Sep Purif 209:175–181
Rizk HE, El-Din AMS, Afifi EME, Attallah MF (2021) Potential separation of zirconium and some lanthanides of the nuclear and industrial interest from zircon mineral using cation exchanger resin. J Dispers Sci Technol 43:1642–1651
Dakroury GA, Allan KF, Attallah MF, El Afifi EM (2020) Sorption and separation performance of certain natural radionuclides of environmental interest using silica/olive pomace nanocomposites. J Radioanal Nucl Chem 325:625–639
Moshin Q, Syed AN (1971) Synthesis and ion-exchange properties of thorium tungstate: separation of La3+ from Ba2+, Sr2+, Ca2+, nd Y3+ and of VO2+ from Fe3+ and Mn2+. J Chem Soc A: Inorg Phys Theor 139–143
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lin, Y., Wang, J., Shao, K. et al. Two-step separation of Th, La and Ba using combined chromatographic columns. J Radioanal Nucl Chem 332, 1245–1252 (2023). https://doi.org/10.1007/s10967-023-08761-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-023-08761-3