Abstract
Cladocerans are valuable indicators of environmental change in lakes. Their fossils provide information on past changes in lake environments. However, few studies have quantitatively examined the relationships between contemporary and sub-fossil cladoceran assemblages and no investigations are available from Mediterranean lakes where salinity, eutrophication and top-down control of large-bodied cladocerans are known to be important. Here we compared contemporary Cladocera assemblages, sampled in summer, from both littoral and pelagic zones, with their sub-fossil remains from surface sediment samples from 40 Turkish, mainly shallow, lakes. A total of 20 and 27 taxa were recorded in the contemporary and surface sediment samples, respectively. Procrustes rotation was applied to both the principal components analysis (PCA) and redundancy analysis (RDA) ordinations in order to explore the relationship between the cladoceran community and the environmental variables. Procrustes rotation analysis based on PCA showed a significant accord between both littoral and combined pelagic–littoral contemporary and sedimentary assemblages. RDA ordinations indicated that a similar proportion of variance was explained by environmental variation for the contemporary and fossil Cladocera data. Total phosphorus and salinity were significant explanatory variables for the contemporary assemblage, whereas salinity emerged as the only significant variable for the sedimentary assemblage. The residuals from the Procrustes rotation identified a number of lakes with a high degree of dissimilarity between modern and sub-fossil assemblages. Analysis showed that high salinity, deep water and high macrophyte abundance were linked to a lower accord between contemporary and sedimentary assemblages. This low accord was, generally the result of poor representation of some salinity tolerant, pelagic and macrophyte-associated taxa in the contemporary samples. This study provides further confirmation that there is a robust relationship between samples of modern cladoceran assemblages and their sedimentary remains. Thus, sub-fossil cladoceran assemblages from sediment cores can be used with confidence to track long-term changes in this environmentally sensitive group and in Mediterranean lakes, subjected to large inter-annual variation in water level, salinity and nutrients.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Zooplankters are important indicators of the structure and function of freshwater lake ecosystems and their ecological status (Jeppesen et al. 2011a). They are a key component of the trophic web and are sensitive to anthropogenic impacts (Caroni and Irvine 2010). Cladocerans are sensitive to increasing salinity because of their limited osmoregulating capacity (Aladin 1991; Frey 1993; Bezirci et al. 2012), and cladoceran species richness and diversity decrease with increasing salinity (Frey 1993; Jeppesen et al. 1994; Bos et al. 1999; Boronat et al. 2001; Brucet et al. 2009; Jeppesen et al. 2011a).
Cladoceran sedimentary remains provide information on ecosystem state and change through time (Jeppesen et al. 2001; Davidson et al. 2011a). Cladocerans grow by periodically shedding their exoskeletons at the end of each instar. Molting rates differ among the species and are influenced by temperature, predation and food availability (Vijverberg 1980; Boersma et al. 1996; Hülsmann and Mehner 1997). Each cladoceran taxon is preserved in the sediment differently (Anderson and Battarbee 1994; Leavitt et al. 1994), but they are generally well preserved in the sediment, in particular benthic taxa (Rautio et al. 2000). The chitinous outer body parts, such as carapaces, head shields, ephippia, post-abdomens and claws, preserve selectively in lake sediments, whereas soft tissues decompose (Frey 1986; Szeroczyńska and Sarmaja-Korjonen 2007). Mechanical, chemical and biological agents can all decompose or damage exoskeleton fragments and chitin, thus affecting their preservation (Kerfoot 1995). Despite this, chitinous remains in surface sediments have generally been shown to provide a good representation of benthic and macrophyte-associated cladoceran species and generally show higher species richness and diversity than contemporary samples (Frey 1964; Davidson et al. 2007; Nevalainen 2010). Further, ephippia of many species preserve well (Frey 1986; Hofmann 1987; Hann 1989; Rautio et al. 2000). For some pelagic taxa, such as Daphnia, Moina and Ceriodaphnia, resting eggs can be used to estimate abundance where chitinous remains are sparse (Jeppesen et al. 1996).
Interpretation of the sub-fossil cladoceran data has typically relied on either relationships obtained by analysis of contemporary assemblages and environmental data or on relationships between surface fossil data with environmental variables (Müeller 1964; Frey 1986; Jeppesen et al. 2001). In case of the former, the reliability of relationship between contemporary and sub-fossil specimens is critical since several processes determine the production and distribution of cladoceran remains (Davidson et al. 2007; Nykänen et al. 2009).
The Mediterranean climatic region is particularly sensitive to changes in climate that may affect salinity. Lakes in the region are likely to receive reduced inputs of water due to shorter wet seasons and associated lengthening of warm dry periods (Sánchez et al. 2004; Jeppesen et al. 2011b). Recent studies in the region have reported decreases in precipitation (Türkeş 1996; Türkeş et al. 2009) affecting hydraulic residence time, water level and salinity (Beklioğlu and Tan 2008; Ozan et al. 2010; Bucak et al. 2012) with strong effects on the lake ecosystem, water turbidity and salinity (Beklioglu et al. 2011; Beklioğlu and Tan 2008).
Analysis of sub-fossil cladocerans from sediment cores in Mediterranean lakes would provide a means to track ecosystem response to past climate change. Thus, allowing the current status to be placed in the context of past condition but also to compare the recent and current ecological change with those that occurred in the past. There is, however, very little information on how the ecology of Mediterranean lakes changed in the past. In the current study we focused on the comparison of the contemporary pelagic and littoral cladoceran assemblages with surface sediment remains in forty shallow Mediterranean lakes. Furthermore, we compared their relationships with key environmental variables. It was our hypothesis that sedimentary and contemporary assemblages would closely resemble each other and that their environmental controls would be similar.
Materials and methods
Study sites and field sampling
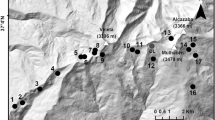
During the main growing period (August–September) from 2006 to 2011, environmental variables and surface sediments were collected in forty Anatolian shallow lakes spanning over 5° of latitude between the warm and dry temperate, arid cold steppe, and Mediterranean climate according to the Köppen-Geiger classification system (Peel et al. 2007) (Fig. 1).
A snapshot sampling protocol developed by Moss et al. (2003) was employed to take depth-integrated, mixed water samples from the whole water column in the deepest part of the lake. Lake water temperature (°C), conductivity (±1 µS cm−1), salinity (‰), maximum depth (m), dissolved oxygen (mg L−1) and pH were determined in situ using YSI 556 MPS multi-probe.
Cladoceran samples were taken from 40 to 20 L depth-integrated water samples, which were collected both from the pelagic and littoral zones, respectively. Water samples were filtered through a 20 μm mesh size filter and the remains were fixed using 4 % Lugol’s solution.
The composition and abundance of fish (Catch per unit effort—CPUE—number.net−1) were determined by overnight (12 h) fishing with Lundgren gill nets consisting of twelve multiple mesh sizes (from 5 to 55 mm) each section being 3 m long and 1.5 m deep. The nets were set parallel to each other and left overnight for an average duration of 12 h in the littoral and pelagic part of the lake. Number of nets used depended on the size of the lake.
Percent plant volume inhabited (PVI%) was calculated based on data on submerged plants sampled at evenly spaced intervals along parallel transect lines (Canfield et al. 1984). Details can be found in Levi et al. (2014).
From each lake, seven surface sediment (0–2 cm) samples with close proximity to each other were taken using a KC-Denmark Kajak Corer (5.2 cm diameter) at the deepest part of each lake. These seven samples were mixed and kept frozen (−18 °C) prior to analysis for sub-fossil cladoceran remains.
Laboratory analyses
All water samples were kept frozen prior to chemical analyses in the laboratory. For analysis of total phosphorus (TP) and soluble reactive phosphate (SRP) we used the acid hydrolysis and molybdate reaction methods, respectively (Mackereth et al. Mackereth et al. 1978). Total nitrogen (TN) was analysed by the Scalar Autoanalyzer Method (San++ Automated Wet Chemistry Analyzer, Skalar Analytical, B.V., Breda, The Netherlands). For chlorophyll a determination, the ethanol extraction method by Jespersen and Christoffersen (1987) was employed and suspended solids were determined by weighing the dried GF/C Whatman filters after filtering ~500 mL water through weighted filters.
For sub-fossil cladoceran taxonomical analysis 5 g wet homogenised sediment were boiled in 100 mL 10 % KOH for 50 min and all material was subsequently kept in Lugol’s solution and the samples were sieved prior to microscopic analysis. Remains larger than 45 µm were all identified and counted using a stereomicroscope (LEICA MZ 16) and an inverted light microscope (LEICA DMI 4000). For the counting process, sub-fossils were separated into two size fractions: >140 and 45–140 µm. All remains >140 µm were counted, while remains in the 45–140 µm size fraction were subsampled and approximately 20 % was counted. For species identification, the keys developed by Frey (1959), Flöβner (2000) and Szeroczyńska and Sarmaja-Korjonen (2007) were used. Carapaces, head shields, post abdomens, post abdominal claws and resting eggs (ephippia) of each sub-fossil cladocerans were counted and the most abundant body part was used in the analyses. For Daphnia, Ceriodaphnia, Simocephalus and Moina, the abundance of individuals were based on ephippia because they were the only body parts encountered in the samples. Taxonomic species separation of Alona rectangula Sars, 1862 and Alona guttata Sars, 1862 was occasionally not possible from the head pores if no post abdomens were present. These two Alona species are consequently used as Alona rectangula/guttata in the statistical analyses.
Contemporary cladoceran species were identified to species level and individuals were counted at 40× magnification using a LEICA MZ 16 stereomicroscope (Tavşanoğlu 2012). For taxonomical identification, the keys in Scourfield and Harding (1966), Ruttner-Kolisko (1977), Pontin (1978), Einsle (1993), Segers (1995), Smirnov (1996), Flöβner (2000) and Smith (2001) were used. Littoral and pelagic contemporary cladoceran abundance (individual L−1) data were used both separately and combined, the latter dataset referred to as “joint contemporary” assemblage.
Data analyses
Environmental data were tested for normality using the Kolmogorov–Smirnov test in SigmaStat 3.5 (Justel et al. 1997). Environmental variables that did not pass the normality test were either log10, log10(x + 1) or square root transformed to obtain the best approximation to normal distribution, and species data were either log or Hellinger transformed depending on the ordination analysis (Legendre and Gallagher 2001).
A range of ordination techniques including principal component analysis (PCA), detrended correspondence analysis (DCA) and redundancy analysis (RDA) were employed for investigating variation. In PCA, cladoceran assemblage data were used to identify the distribution of the species between lakes. RDA or Canonical Correspondance Analysis (CCA) was employed when the gradient length of axis 1 in DCA was lower than or higher than 3 standard deviation (SD) units, respectively (ter Braak 1995). In order to choose the environmental variables explaining the maximum variation in the species data, highly co-linear environmental variables were identified by their high variance inflation factors (VIF). Variables with VIF more than 20 were deleted from the environmental dataset. Monte Carlo permutation tests were applied in order to test the significance of each environmental variable used in the ordination analyses. Environmental variables which did not explain a significant portion of species variance after Monte Carlo permutations (p < 0.05; 999 random permutations) were removed from the ordination analyses. A series of RDA ordinations with forward selection were run to determine the subset of significant environmental parameters.
In order to explore the relationship between the cladoceran community and the environmental variables for both contemporary and surface-sediment data and to test the significance of the identified relationships, Procrustes rotation and the related PROTEST permutation test were applied to the PCA and RDA ordination results (Jackson 1995; Peres-Neto and Jackson 2001). Procrustes rotation considers the general degree of correlation between two or more ordination results by using a procrustean superimposition approach in which the results of the ordinations are scaled and rotated to find an optimal superimposition for maximising their fit (Peres-Neto and Jackson 2001). The sum of the squared residuals between configurations in their optimal fit can be used as a metric of correlation (Gower 1971). PROTEST is a permutation-based procedure and measures the significance of the concordance (Jackson 1995) established by the Procrustes rotation. PROTEST produces a correlation-like statistic referred to as “correlation in a symmetric Procrustes rotation (m 12 ) and an associated significance (p value)” (Jackson 1995), indicating the likelihood of the relationship occurring by chance (Peres-Neto and Jackson 2001). Procrustes rotation and PROTEST were applied a number of times to assess the significance of the relationship of the fossil PCA with: (a) joint contemporary PCA, (b) pelagic contemporary PCA and (c) littoral contemporary PCA. It was also applied to compare the fossil RDA with joint contemporary RDA ordinations. All analyses were done in R version 2.12.2 (R Core Development Team 2011) using the vegan (Oksanen et al. 2008) package.
Shannon-Wiener diversity and Pielou’s evenness were calculated from the abundance data using the vegan package (Oksanen et al. 2008) in R Statistical Software (R Development Core Team 2011). Species richness was measured as the total number of species of cladocerans in each lake. One-way analyses of variance (ANOVA) were used to test the differences in diversity, evenness and richness between high salinity (>4 ‰) and low salinity (<4 ‰) lakes. Analyses were performed using Sigma Stat 3.5.
Multiple linear regression analysis was employed in order to investigate the role of environmental variables in the accordance between contemporary and sedimentary cladoceran assemblages. Procrustes residuals from PCA-Procrustes analysis were used as a response variable and environmental variables were used as explanatory variable. Analyses were performed using Sigma Stat 3.5.
Results
The 40 lakes studied were small and mostly shallow with a maximum depth ranging from 0.55 to 17.4 m and a mean depth of 4 m (Table 1). Surface water temperature averaged 24.4 °C and the lakes were well oxygenated (mean: 6.7 mg L−1). With the exception of four lakes with salinities ranging between 3.1 and 5.8 ‰, all were fresh waters. Most of the lakes had a pH close to neutral, were eutrophic with an average chlorophyll a concentration of 19 µg L−1 and an average Secchi depth of 1.3 m. Omnivorous fish dominated the fish catches, average number of catch per net per night being 107. The PVI% varied from 0 to 80 % (Table 1).
A total of 11 and 21 taxa were identified in the pelagic and littoral water column, respectively; whereas 27 taxa were identified in the surface sediment samples (Table 2; Fig. 2). The maximum, mean and median values of the relative abundance of all identified taxa are represented in Table 2. 87 % of the taxa found in the joint contemporary samples were represented by the sedimentary remains. Some of the taxa frequently occurring in the water column were not found in the sediment assemblages, including Diaphanosoma sp., Macrothrix sp., Scapholeberis mucronata (Müller) and Polyphemus pediculus (Linnaeus, 1761) (Table 2; Fig. 2). However, Disparalona rostrata (Koch, 1841), Graptoleberis testudinaria (Fischer, 1848), Alonella excisa (Fischer, 1854), Leptodora kindtii (Focke, 1844), Kurzia latissima Kurz, 1874, Leydigia acanthocercoides (Fischer, 1854), Oxyurella tenuicaudis (Sars, 1862), Sida crystallina (O.F.Müller, 1776), Camptocercus spp. and Eurycercus lamellatus (O.F. Müller, 1785) were found in the sediment but not in contemporary samples (Fig. 2).
Venn diagram showing cladoceran species (big diagram) and number of species (small diagram) distributions in contemporary pelagic and littoral, and surface sediment dataset (for abbreviations see Table 2)
Lakes Hamam, Pedina and Baldımaz did not contain any cladoceran taxa in their contemporary assemblages and were therefore not included in ordination analyses.
Ordinations
Detrended correspondence analysis (DCA) had a first axis length of 1.41 SD units for the joint datasets. The axis length was 2.03 SD units for the surface fossil data. Thus, we used linear methods for both the contemporary and the sedimentary assemblages and PCA and RDA were employed for both datasets (ter Braak and Prentice 1988).
The Monte Carlo permutation tests showed that TP and salinity were significant for the joint contemporary assemblage (p < 0.05), while only salinity (p < 0.01) was significant for the sub-fossil assemblage (Fig. 3). Thus, in both RDAs salinity was the most significant variable and was negatively correlated with axis 1 (Fig. 3). Furthermore, a similar percentage of variance was explained for contemporary (16.7 %) and surface sediment fossil cladocerans (13.6 %) when all environmental variables were included in RDA (Table 3). When using single variables in each analysis, salinity again had the highest percentage of variance explained in both contemporary and sedimentary datasets (Table 3).
The distribution of the most common species was similar between the two RDA plots. For instance, Bosmina longirostris (O. F. Müller, 1785), Chydorus sphaericus (O.F. Müller, 1776) and Leydigia leydigi (Schoedler, 1863) distributions in ordination space were similar and exhibited a similar response to the same environmental variables in both ordinations (Fig. 3). C. sphaericus was positively associated with salinity; in contrast, B. longirostris was negatively associated with salinity in the comparison of contemporary and sedimentary RDAs. Moina spp. was positively linked with TP in the contemporary dataset. Moreover, vegetation and sediment associated taxa Pleuroxus spp., Simocephalus sp., Alona rectangula/guttata, Alonella nana (Baird, 1850) and Alonella exigua (Lilljeborg, 1853) were in close relationship with PVI% in both RDAs. Although fish (CPUE number.net−1) was not a significant variable, the large bodied cladoceran, Daphnia spp. were negatively associated with fish in both RDAs.
Species diversity, evenness and richness
Significant differences were identified in the Shannon-Wiener diversity, evenness and species richness for two cladoceran communities (Table 4). Species richness and diversity were higher in the sedimentary cladoceran community and evenness was higher in the joint contemporary community (Table 4).
According to one-way ANOVA, there was a clear significant (p < 0.05) response to salinity in the Shannon-Wiener diversity, evenness and richness patterns of the joint contemporary cladoceran data, whereas only richness showed significant (p < 0.05) response to salinity for the sub-fossil cladoceran data. Diversity, evenness and richness in modern cladoceran community increased and richness of sub-fossil cladoceran community increased with decreasing salinity (Fig. 4).
Species diversity (Shannon-Wiener index H’), evenness (Pielou index) and species richness between contemporary and sedimentary communities and the effects of salinity. Lakes were separated according to salinity. Lakes with salinities >0.7 ‰ are termed high salinity lakes and the rest low salinity lakes
Procrustes rotation and PROTEST
For the Procrustes rotation analysis and PROTEST, the PCA scores of the species data and the RDA scores of 37 lakes were employed and the results are shown in Table 5. The correlation diagnostic (m 12 , which is the equivalent of r2) had values 0.528 and 0.537 when using joint contemporary and sedimentary fossil, and littoral contemporary and sedimentary fossil PCA scores, respectively. Furthermore, the significant (p: 0.001) Procrustes rotation test suggested that using joint contemporary and sedimentary fossil PCA scores, littoral and sedimentary fossil PCA scores, and joint contemporary and sub-fossil RDA scores gave an optimal fit and a high degree of correspondence between contemporary and sedimentary cladoceran assemblages (Table 5). However, pelagic contemporary and sub-fossil Cladocera assemblages did not show significant accord (p = 0.147) and had a low m 12 value (0.255) (Table 5).
Only “joint contemporary and sedimentary fossil PCA” and “joint contemporary and sedimentary fossil RDA” Procrustes analyses plots are presented (Figs. 5, 6). Black dots in Figs. 5 and 6 represent contemporary and arrow heads the sedimentary data. The distance between the dots and arrow heads shows the Procrustes residuals. Low residuals (short arrows) indicated good agreement between datasets. The direction of the movement between the base and the end of the arrows and the length of the arrows were related to the distribution of the sedimentary Cladocera. The Procrustes rotation plot using PCA scores identified most of the lakes displaying a high degree of similarity (short arrows) especially Lakes 11, 13, 26, 28 and 31, which had low residuals, indicating a high degree of concordance between contemporary and sedimentary assemblages. However, some of the study sites such as Lakes 2, 3, 5, 20, 30, and 35 displayed a low degree of similarity (long arrows) (Fig. 5). As shown as an overlying PCA plot, the direction of the movement and the length of the arrows were related to the distribution of the sedimentary taxa, (Fig. 5). For instance, the longer arrow in Lake 5 resulted from B. longirostris, Ilyocryptus spp., Kurzia latissima and Eurycercus lamellatus which dominated the sedimentary community, whereas the remaining lakes (2, 3, 20, 30, and 35) either did not exhibit, or had only very low abundances of, Ilyocryptus spp., K. latissima and E. lamellatus (Fig. 5). Low concordance was also found in Lake 2 judged from the PCA of sedimentary fossil data, D. rostrata, Camptocercus sp., and S. crystallina were responsible for this difference and these taxa were only found in sediment samples in this lake. A similar pattern was observed for Lake 20, where Simocephalus sp. was only found in sediment samples but not in water column. Furthermore, for Lake 35 Ceriodaphnia spp. and Daphnia spp. were abundant taxa that were found only in the sediment samples not in contemporary samples at that site. A. rectangula/guttata and C. sphaericus were only present in Lake 30’s sediment sample resulting in the high residuals between contemporary and sedimentary assemblages. Finally, Lake 3 was the only lake where A. nana had high abundance in the sediment but it was completely absent from the water column (Fig. 5). In order to investigate reason for the low degree of similarity in some lakes, multiple linear regression analyses were run using PCA-Procrustes residuals and environmental variables. Water depth emerged as the only significant (p < 0.05) variable explaining the high residuals or low degree of similarity between the data sets. In accordance, the lakes with water depth higher than 4.5 m had mostly low degree of similarity between contemporary and sub-fossil assemblages.
PROTEST plot generated from PCA scores. Dots represent joint contemporary samples and arrow points represent sedimentary samples, the distance between the two showing Procrustes residuals. The overlaid species PCA plot was retained from sedimentary cladoceran PCA. Lake numbers followed a salinity gradient (see legend of Fig. 1)
PROTEST plot generated from RDA scores. Dots represent contemporary samples and arrow points sedimentary samples, the distance between the two showing Procrustes residuals. Environmental variables were retained from fossil RDA and inserted passively. Lake numbers followed a salinity gradient (see legend of Fig. 1)
The Procrustes rotation plot using RDA scores were examined to investigate the environmental variables important in causing a discrepancy between the sub-fossil and contemporary assemblages. Several sites with low residuals were identified (Fig. 6). For instance, Lakes 11, 17, 22, and 27 exhibited high correlation between the two datasets. However, a number of lakes such as Lakes 9, 20, 25, 26, 34, 35, 36, 38, 39, and 40 displayed high Procrustes residuals (Fig. 6). The direction of movement between the dot and arrowhead is related to the environmental variables from the ordinations. Accordingly, the point of the arrow (sub-fossil assemblage) for Lakes 35, 36, 38 and 39 reflects higher salinity concentrations since salinity from the inserted RDA plot and direction of movement were parallel to each other (Fig. 6). Also, the sub-fossil communities in these lakes were dominated by C. sphaericus which was positively associated with salinity, whereas in the contemporary data C. sphaericus was absent. Thus, direction of movement was not the only indication of the environmental relationship between cladocerans in a lake, also the absence/presence of taxa can result in the discrepancy between contemporary and sub-fossil data. Another similar example was the high Procrustes residuals of Lakes 9, 34 and 40 in which the movement between the base and the point of the arrow extended towards the high PVI% and high Secchi:Max depth ratio (Fig. 6). A. rectangula/guttata was related to the high PVI% and Secchi:Max depth ratio in the sedimentary RDA (Fig. 3). The sub-fossil cladoceran assemblages of these three lakes included high abundances of A. rectangula/guttata but in their contemporary assemblages it had low abundance.
Discussion
Higher cladoceran species richness and diversity were found in the surface sediment samples compared to the contemporary samples (27 and 21 species, respectively). There was a significant concordance between sedimentary assemblages and contemporary littoral and also joint contemporary (pelagic and littoral) assemblages. The good agreement between littoral and surface sediment assemblages was driven by taxa such as B. longirostris and C. sphaericus which are abundant and well-represented in littoral water column samples and in the sediments. In contrast, contemporary pelagic and sedimentary communities did not show significant agreement. Previous studies indicated that littoral cladocerans are primarily deposited close to their shallow habitats, while planktonic cladocerans accumulate principally at deep-water locations (Nevalainen 2011; Nevalainen and Luoto 2012). However, the number of species in the mid-lake surface sediment was twice as high as in the contemporary samples (Fig. 2) reflecting that sediment samples collected from the deepest part of the lake includes both cladocerans transported from shallow habitats and those deposited from deep habitats (Frey 1988; Smol 2008). Our sampling method used in the contemporary cladoceran sampling could potentially create a bias toward pelagic species since attached species are more difficult to sample (Vandekerkhove et al. 2005). Different sampling strategies using sweep net along the shore and inside plant beds could prevent such bias. However, Davidson et al. (2007) found a similar bias towards pelagic taxa despite using series of sweep nets in plant beds. Some species were better represented in the contemporary assemblages, including the open water associated Moina spp. Additionally, Diaphanosoma sp., Macrothrix sp., Scapholeberis mucronata and Polyphemus pediculus were all absent from the sediment record but well-represented in the contemporary samples in several of the lakes, probably due to inadequate preservation of them in lake sediment because of their soft-shelled chitinous body parts (Davidson et al. 2007; Kattel et al. 2007). By contrast, the contemporary cladoceran sample lacked large-bodied taxa, such as L. kindtii and S. crystallina which were represented in the sediment in some of the lakes.
The key environmental variables structuring the cladoceran assemblages explained similar proportions of the variance in the joint contemporary and sub-fossil cladoceran assemblages (Table 3). Salinity and TP were of key importance for the joint contemporary cladoceran assemblage whereas only salinity was significant for the sub-fossil assemblage. Diversity, evenness and richness of joint contemporary community data and richness of the sedimentary cladoceran were all negatively related to salinity. The decline in richness and diversity reflect the limited salinity tolerance of cladocerans, apart from a few species (Aladin 1991; Frey 1993; Jeppesen et al. 1994; Dodson and Frey 2001; Boix et al. 2008; Brucet et al. 2009). Salinity influences survival, abundance, fecundity, size and growth rate (Williams 1987; Schuytema et al. 1997; Bezirci et al. 2012). Others (Bjerring et al. 2009; Hobaek et al. 2002; Green et al. 2005; Nevalainen 2010) also found conductivity to be a strong predictor of both cladoceran species richness and species. Most of the cladoceran taxa in our sedimentary and joint contemporary datasets were negatively correlated with salinity, especially B. longirostris and Daphnia spp. (Fig. 3). However, we found that L. leydigi, C. sphaericus and A. quadrangularis were positively related to salinity likely reflecting their higher osmoregulatory capacity (Bos et al. 1999; Amsinck et al. 2005; Brucet et al. 2009; Jensen et al. 2010). In support, Ustaoğlu et al. (2012) found L. leydigi, and C. sphaericus in lagoons with salinity up to 22 and 33 ‰, respectively. Furthermore, a study on the plant macrophyte remains from surface sediments in the same set of lakes also showed conductivity to be key environmental factors for macrophytes (Levi et al. 2014). It appears to be that salinity is a key driver of richness of cladoceran and other species in semi-dry Mediterranean lakes which may become even more important in a future warmer and drier climate (Beklioğlu et al. 2011).
Our study indicates that similar environmental forces shape surface sediment and joint contemporary cladoceran assemblages in a similar way. There were, however, exceptions (Fig. 6), Lakes 35, 36, 38 and 39 exhibited high salinity (>1.5 ‰) and had high Procrustes residuals. The joint contemporary assemblage included only one cladoceran taxon in each of the lakes, whereas the sediment samples had 4, 6, 10 and 9 taxa, respectively. The long term monitoring data series of Lakes 35 and 36 revealed high seasonal and annual salinity fluctuations (Beklioğlu and Tan, 2008; Beklioğlu et al. unpublished data), which may explain the high number of cladoceran taxa with contrasting salinity tolerances found in the sediment. The contemporary assemblages only represent late summer whereas the surface sediment assemblages are a temporal integration of the cladoceran community representing several years (Vandekerkhove et al. 2005; Amsinck et al. 2006). This may be of particular importance when key environmental variables, such as salinity in Mediterranean lakes, show large inter- or intra-annual variations. Moreover, salinity could enhance formation of resting eggs, increasing the representation of sedimentary ephippia. For instance, the ratio of ephippia to the sum of ephippia and carapaces of Bosmina have been found to be higher in brackish lakes, which may reflect temporary harsh environments due to high salinity (Jeppesen et al. 2003), although higher temperatures (leading to increased salinity) may have the opposite effect (Bjerring et al. 2009).
The Lakes 9, 28, 34, and 40 were also characterised by high residuals in the Procrustes rotation of the RDAs. This stems from the disparity in the occurrence of macrophyte associated taxa in the contemporary and sediment assemblages. A number of macrophyte associated species such as S. crystallina, E. lamellatus, K. latissima, Camptocercus sp., O. tenuicaudis and G. testudinaria were recorded in the surface sediments but not in the contemporary samples even though PVI% were high (>35 %) in these lakes. Many of the species associated with plants are found mainly within the plant beds (Lauridsen et al. 1996) and therefore may go undetected by pelagic sampling. The lowest agreement between contemporary and sub-fossils therefore occurred in lakes where salinity or PVI were high.
In addition to salinity, total phosphorus was important in shaping the joint contemporary cladoceran assemblages (Fig. 3). Nutrient enrichment indirectly affects cladocerans by changing their food quantity, quality, predation pressure and habitats (Gliwicz 2003; Gyllström et al. 2005; Jeppesen et al. 2011a). Increases in TP concentration may be associated with enhanced fish predation on Cladocera and large-bodied cladocerans may take day-time refuge near the sediment and therefore be absent from the contemporary samples sampled during day (Tavşanoğlu et al. 2012) but not necessarily from the surface sediment cladoceran assemblages. Decreases in cladoceran species richness with an increase in TP have also been observed in Mediterranean and cold temperate lakes (Jeppesen et al. 2000; Romo et al. 2005; Declerck et al. 2005). Contrary to other studies (Bjerring et al. 2009; Chen et al. 2010), we did not find any effect of TP on the sub-fossil cladoceran assemblages.
In summary, notwithstanding differential preservation of taxa and the fact that contemporary and sedimentary samples represent different time periods, we found good correspondence between the contemporary community sampled in late summer and the sub-fossil assemblages of surface sediment samples gathered from the deepest part of the lakes. Furthermore, diversity and richness of cladocerans in both datasets decreased with increasing salinity. The data demonstrated that sedimentary fossil Cladocera could be used instead of contemporary assemblages to reflect assemblage composition (87 % of the taxa recorded in water column were represented by the sedimentary remains) and that sedimentary remains show the controlling environmental factors shaping them in a similar way to the modern assemblages. In addition, the higher diversity and richness for the sedimentary assemblages suggest that a composite surface sediment sample from the deepest point of the lakes may provide reasonable sample material for evaluation of cladoceran diversity in shallow lakes, as in other studies (Nevalainen 2010; Davidson et al. 2013). However, it should be noted that sedimentary remains do not provide important information on seasonality or other aspects of population dynamics and are thus not equivalent to modern sampling. What is apparent is that sub-fossil cladoceran assemblages can be reliably used to track past change in Mediterranean lake ecosystems and, provided that the problems of the multiple structuring forces are addressed (Davidson et al. 2011b), can be used to reconstruct past environmental change in these highly dynamic (e.g. in terms of water level and salinity) lakes.
References
Aladin NV (1991) Salinity tolerance and morphology of the osmoregulation organs in Cladocera with special reference to Cladocera from the Aral Sea. Hydrobiologia 225:291–299
Amsinck SL, Jeppesen E, Landkildehus F (2005) Relationships between environmental variables and zooplankton subfossils in the surface sediments of 36 shallow coastal brackish lakes with special emphasis on the role of fish. J Paleolimnol 33:39–51
Amsinck SL, Strzelczak A, Bjerring R, Landkildehus F, Lauridsen TL, Søndergaard M, Jeppesen E (2006) Lake depth rather than fish planktivory determine cladoceran community structure in Faroese lakes—evidence from contemporary data and sediments. Freshw Biol 51:2124–2142
Anderson NJ, Battarbee RW (1994) Aquatic community persistence and variability: a palaeolimnological perspective. In: Giller PS, Hildrew AG, Raffelli D (eds) Aquatic ecology: scale. Press, Oxford, Pattern and Process. Blackwell Sci, pp 233–259
Beklioglu M, Meerhoff M, Søndergaard M, Jeppesen E (2011) Eutrophication and restoration of shallow lakes from cold temperate to a warm Mediterranean and a (sub) tropical climate. In: Ansari AA, Singh Gill S, Lanza GR, Rast W (eds) Eutrophication: causes, consequences and control, vol 1. Springer, New York, pp 91–108
Bezirci G, Akkas SB, Rinke K, Yildirim F, Kalaylioglu Z, Severcan F, Beklioglu M (2012) Impacts of salinity and fish-exuded kairomone on the survival and macromolecular profile of Daphnia pulex. Ecotoxicology 21:601–614
Bjerring R, Becares E, Declerck S, Gross EM, Hansson LA, Kairesalo T, Nykanen M, Halkiewic A, Kornijow R, Conde-Porcuna JM, Seferlis M, Noges T, Moss B, Amsinck SL, Odgaard BV, Jeppesen E (2009) Subfossil Cladocera in relation to contemporary environmental variables in 54 Pan-European lakes. Freshw Biol 54:2401–2417
Boersma M, van Tongeren OFR, Mooij WM (1996) Seasonal patterns in the mortality of Daphnia species in a shallow lake. Can J Fish Aquat Sci 53:18–28
Boix D, Gascon S, Sala J, Badosa A, Brucet S, Lopez-Flores R, Martinoy M, Gifre J, Quintana XD (2008) Patterns of composition and species richness of crustaceans and aquatic insects along environmental gradients in Mediterranean water bodies. Hydrobiologia 597:53–69
Boronat L, Miracle MR, Armengol X (2001) Cladoceran assemblages in a mineralization gradient. Hydrobiologia 442:75–88
Bos DG, Cumming BF, Smol JP (1999) Cladocera and Anostraca from the Interior Plateau of British Columbia, Canada, as paleolimnological indicators of salinity and lake level. Hydrobiologia 392:129–141
Brucet S, Boix D, Gascón S, Sala J, Quintana XD, Badosa A, Søndergaard M, Lauridsen TL, Jeppesen E (2009) Species richness of crustacean zooplankton and trophic structure of brackish lagoons in contrasting climate zones: north temperate Denmark and Mediterranean Catalonia (Spain). Ecography 32:692–702
Bucak T, Saraoğlu E, Levi EE, Tavşanoğlu UN, Çakıroğlu Aİ, Jeppesen E, Beklioğlu M (2012) The influence of water level on macrophyte growth and trophic interactions in eutrophic Mediterranean shallow lakes: a mesocosm experiment with and without fish. Freshw Biol 57(8):1631–1642
Canfield DE, Shireman JV, Colle DE, Haller WT, Watkins CE, Maceina MJ (1984) Prediction of chlorophyll-a concentrations in Florida lakes: importance of aquatic macrophytes. Can J Fish Aquat Sci 41:497–501
Caroni R, Irvine K (2010) The potential of zooplankton communities for ecological assessment of lakes: redundant concept or political oversight? Biol Environ Proc R Ir Acad 110B:35–53
Chen G, Dalton C, Taylor D (2010) Cladocera as indicators of trophic state in Irish lakes. J Paleolimnol 44:465–481
Davidson TA, Sayer CD, Perrow MR, Bramm M, Jeppesen E (2007) Are the controls of species composition similar for contemporary and fossil cladoceran assemblages? A study of 39 shallow lakes of contrasting trophic status. J Paleolimnol 38:117–134
Davidson TA, Bennion H, Sayer C, Jeppesen E, Clarke GH, Morley D, Odgaard BV, Rasmussen P, Rawcliffe R, Salgado J, Amsinck SL (2011a) The role of cladocerans in tracking long-term in shallow lake trophic status. Hydrobiologia 676:129–142
Davidson TA, Amsinck SL, Bennike O, Landkildehus F, Lauridsen TL, Jeppesen E (2011b) Inferring a single variable from an assemblage with multiple controls: getting into deep water with cladoceran lake-depth transfer functions. Hydrobiologia 676:129–142
Davidson TA, Reid MA, Sayer CD, Chilcott S (2013) Palaeolimnological records of shallow lake biodiversity change: exploring the merits of single versus multi-proxy approaches. J Paleolimnol 49:431–446
Declerck S, Vandekerkhove J, Johansson L, Muylaert K, Conde-Porcuna JM, Van der Gucht K, Perez-Martinez C, Lauridsen T, Schwenk K, Zwart G, Rommens W, Lopez-Ramos J, Jeppesen E, Vyverman W, Brendonck L, De Meester L (2005) Multi-group biodiversity in shallow lakes along gradients of phosphorus and water plant cover. Ecology 86:1905–1915
Dodson SI, Frey DG (2001) Cladocera and other Branchiopoda. In: Ecology and classification of North American freshwater invertebrates, p 849–913
Einsle U (1993) Crustacea Copepoda, Calanoida und Cyclopoida. Gustav Fisher Verlag, Stuttgart, p 208
Flöβner D (2000) Die Haplopoda und Cladocera (ohne Bosminidae) Mitteleuropas. Backhuys Publishers, Leiden
Frey DG (1959) The taxonomic and phylogenetic significance of the head pores of the Chydoridae (Cladocera). Int Rev Ges Hydrobiol 44:27–50
Frey DG (1960) On the occurrence of cladoceran remains in lake sediments. Proc Natl Acad Sci USA 46(6):917–920
Frey DG (1964) Remains of animals in Quaternary lake and bog sediments and their interpretation. Ergeb der Limnol 2:1–114
Frey DG (1986) Cladocera analysis. In: Berglund BE (ed) Handbook of Holocene palaeoecology and palaeohydrology. John Wiley & Sons Ltd, Hoboken, pp 667–692
Frey DG (1988) What is paleolimnology? J Paleolimnol 1:5–8
Frey DG (1993) The penetration of Cladocera into saline waters. Hydrobiologia 267:233–248
Gliwicz ZM (2003) Between hazards of starvation and risk of predation: the ecology of offshore animals. In: Kinne O (ed) Excellence of ecology, Book 12. International Ecology Institute, Oldendorf/Luhe
Gower JC (1971) Statistical methods of comparing different multivariate analyses of the same data. In: Hodson FR, Kendall DG, Tautu P (eds) Mathematics in the archaeological and historical sciences. Edinburgh University Press, Edinburgh, pp 138–149
Green AJ, Fuentes C, Moreno-Ostos E, Rodrigues da Silva SL (2005) Factors influencing cladoceran abundance and species richness in brackish lakes in Eastern Spain. Ann Limnol Int J Limnol 41(2):73–81
Gyllström M, Hansson LA, Jeppesen E, Garcia-Criado F, Gross E, Irvine K, Kairesalo T, Kornijow R, Miracle MR, Nykänen M, Noges T, Romo S, Stephen D, Van Donk E, Moss B (2005) Zooplankton community structure in shallow lakes: interaction between climate and productivity. Limnol Oceanogr 50:2008–2021
Haberman J, Laugaste R (2003) On characteristics reflecting the trophic state of large and shallow Estonian lakes (L. Peipsi, L. Vortsjarv). Hydrobiologia 506:737–744
Hann BJ (1989) Methods in quaternary ecology #6 Cladocera. Geosci Can 16:17–26
Hobaek A, Manca M, Andersen T (2002) Factors influencing species richness in lacustrine zooplankton. Acta Oecol 23:155–163
Hofmann W (1987) Cladocera in space and time: analysis of lake sediments. Hydrobiologia 145:315–321
Hülsmann S, Mehner T (1997) Predation by under yearling perch (Perca fluviatilis) on a Daphnia galeata population in a short-term enclosure experiment. Freshw Biol 38:209–219
Jackson DA (1995) PROTEST: a Procrustean randomization test of community environment concordance. Ecoscience 2:297–303
Jensen E, Brucet S, Meerhoff M, Nathansen L, Jeppesen E (2010) Community structure and diel migration of zooplankton in shallow brackish lakes: role of salinity and predators. Hydrobiologia 646:215–229
Jeppesen E, Sondergaard M, Kanstrup E, Petersen B, Eriksen RB, Hammershoj M, Mortensen E, Jensen JP, Have A (1994) Does the impact of nutrients on the biological structure and function of brackish and freshwater lakes differ? Hydrobiologia 275(276):15–30
Jeppesen E, Madsen EA, Jensen JP, Anderson NJ (1996) Reconstructing the past density of planktivorous fish and trophic structure from sedimentary zooplankton fossils: a surface sediment calibration data set from shallow lake. Freshw Biol 36:115–127
Jeppesen E, Lauridsen TL, Mitchell SF, Christoffersen K, Burns CW (2000) Trophic structure in the pelagial of 25 shallow New Zealand lakes: changes along nutrient and fish gradients. J Plankton Res 22:951–968
Jeppesen E, Leavitt P, De Meester L, Jensen JP (2001) Functional ecology and palaeolimnology: using cladoceran remains to reconstruct anthropogenic impact. TREE 16(4):191–198
Jeppesen E, Jensen JP, Lauridsen TL, Amsinck SL, Christoffersen K, Søndergaard M, Mitchell SF (2003) Sub-fossils of cladocerans in the surface sediment of 135 lakes as proxies for community structure of zooplankton, fish abundance and lake temperature. Hydrobiologia 491:321–330
Jeppesen E, Noges P, Davidson TA, Haberman J, Noges T, Blank K, Lauridsen TL, Søndergaard M, Sayer C, Laugaste R, Johansson LS, Bjering R, Amsink SL (2011a) Zooplankton as indicators in lakes: a scientific-based pleafor including zooplankton in the ecological qualityassessment of lakes according to the European WaterFramework Directive (WFD). Hydrobiologia 676:279–297
Jeppesen E, Kronvang B, Olesen JE, Audet J, Søndergaard M, Hoffman CC, Andersen HE, Lauridsen TL, Liboriussen L, Larsen SE, Beklioğlu M, Meerhoff M, Özen A, Özkan K (2011b) Climate change effects on nitrogen loading from cultivated catchments in Europe: implications for nitrogen retention, ecological state of lakes and adaptation. Hydrobiologia 663(1):1–21
Jespersen AM, Christoffersen K (1987) Measurements of chlorophyll a from phytoplankton using ethanol as axtraction solvent. Arch Hydrobiol 109:445–454
Justel A, Peña D, Zamar R (1997) A multivariate Kolmogorov–Smirnov test of goodness of fit. Stat Probab Lett 35(3):251–259
Kattel G, Battarbee R, Mackay A, Birks HJB (2007) Are cladoceran fossils in lake sediment samples a biased reflection of the communities from which they are derived? J Paleolimnol 38:157–181
Kerfoot WC (1995) Bosmina remains in Lake Washington sediments: qualitative heterogeneity of bay environments and qualitative correspondence to production. Limnol Oceanogr 40:211–225
Leavitt PR, Sanford PR, Carpenter SR, Kitchell JF (1994) An annual fossil record of production, planktivory and piscivory during whole-lake manipulations. J Paleolimnol 11:133–149
Legendre P, Gallagher ED (2001) Ecologically meaningful transformations for ordination of species data. Oecologia 129:271–280
Levi EE, Çakıroğlu Aİ, Bucak T, Odgaard BV, Davidson TA, Jeppesen E, Beklioğlu M (2014) Similarity between contemporary vegetation and plant remains in the sediment surface in Mediterranean lakes. Freshw Biol. doi:10.1111/fwb.12299
Mackereth FJH, Heron J, Talling JF (1978) Water analysis: some revised methods for limnologists. Freshwater Biological Association, Ambleside, p 36
Moss B, Stephen D, Alvarez C, Becares E, van de Bund W, Collings SE, van Donk E, de Eyto E, Feldmann T, Fernández-Aláez C, Fernández-Aláez M, Franken RJM, García-Criado F, Gross EM, Gyllström M, Hansson LA, Irvine K, Järvalt A, Jensen JP, Jeppesen E, Kairesalo T, Kornijów R, Krause T, Künnap H, Laas A, Lill E, Lorens B, Luup H, Miracle MR, Nõges P, Nõges T, Nykänen M, Ott I, Peczula W, Peeters ETHM, Phillips G, Romo S, Russell V, Salujõe J, Scheffer M, Siewertsen K, Smal H, Tesch C, Timm H, Tuvikene L, Tonno I, Virro T, Vicente E, Wilson D (2003) The determination of ecological status in shallow lakes—a tested system (ECOFRAME) for implementation of the European Water Framework Directive. Aquat Conserv Mar Freshw Ecosyst 13:507–549
Müeller WP (1964) The distribution of cladoceran remains in surficial sediments from three Northern Indiana Lakes. Invest Indiana Lakes Streams 6:1–63
Nevalainen L (2010) Evaluation of microcrustacean (Cladocera, Chydoridae) biodiversity based on sweep net and surface sediment samples. Ecoscience 17:356–364
Nevalainen L (2011) Intra-lake heterogeneity of sedimentary cladoceran (Crustacea) assemblages forced by local hydrology. Hydrobiologia 676:9–22
Nevalainen L, Luoto TP (2012) Intralake training set of fossil Cladocera for paleohydrological inferences: evidence for multicentennial drought during the Medieval Climate Anomaly. Ecohydrology 5:834–840
Nykänen M, Vakkilainen K, Liukkonen M, Kairesalo T (2009) Cladoceran remains in lake sediments: a comparison between plankton counts and sediment records. J Paleolimnol 4:551–570
Oksanen J, Kindt R, Legendre P, O’Hara B, Simpson GL, Solymos P, Stevens MHH, Wagner H (2008) Vegan: community ecology package. R-Package version 1.15-1
Peel MC, Finlayson BL, McMahon TA (2007) Updated world map of the Köppen-Geiger climate classification. Hydrol Earth Syst Sci Discuss 4:439–473
Peres-Neto PR, Jackson DA (2001) How well do multivariate data sets match? The advantages of a Procrustean superimposition approach over the Mantel test. Oecologia 129:169–178
Pontin RM (1978) A key to British freshwater planktonic Rotifera. Freshwater Biological Association, Ambleside, pp 5–15
R Development Core Team (2011) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Rautio M, Sorvari S, Korhola A (2000) Diatom and crustacean zooplankton communities, their seasonal variability and their representation in the sediment of subarctic Lake Saanajarvi. J Limnol 59(Suppl. 1):81–96
Romo S, Villena MJ, Sahuquillo M, Soria JM, Gimenez M, Alfonso T, Vicente E, Miracle MR (2005) Response of a shallow Mediterranean lake to nutrient diversion: does it follow similar patterns as in northern shallow lakes? Freshw Biol 50:1706–1717
Ruttner-Kolisko A (1977) Suggestions for biomass calculations of plankton rotifers. Arch Hydrobiol Beih Ergebn Limnol 8:71–76
Sánchez E, Gallardo C, Gaertner MA, Arribas A, Castro M (2004) Future climate extreme events in the Mediterranean simulated by a regional climate model: a first approach. Glob Planet Chang 44:163–180
Schuytema GS, Nebeker AV, Stutzman TW (1997) Salinity tolerance of Daphnia magna and potential use for estuarine sediment toxicity tests. Arch Environ Contam Toxicol 33:194–198
Scourfield DJ, Harding JP (1966) A key to the British freshwater Cladocera with notes on their ecology, 3rd edn. Freshwater Biological Association, Ambleside, p 5
Segers H (1995) Rotifera. Vol. 2, The Lecanidae (Monogononta). In: Dumont HJF, Nogrady T (eds) Guides to the identification of the microinvertebrates of the continental waters of the World 6. SPB Academic Publishing, The Hague, pp 142–167
Smirnov NN (1996a) Cladocera: the Chydorinae and Sayciinae (Chydoridae) of the world, guides to the identification of the microinvertebrates of the continental waters of the world. SPB Academic Publishing, Netherlands, pp 1–197
Smirnov NN (1996b) Cladocera: the Chydorinae and Sayciinae (Chydoridae) of the world. Guides to the identification of the microinvertebrates of the continental waters of the world. SPB Academic Publishing, Netherlands, pp 1–197
Smol JP (2008) Pollution of lakes and rivers—a paleoenvironmental perspective, 2nd edn. Blackwell Publishing, Oxford, p 383
Szeroczyńska K, Sarmaja-Korjonen K (2007) Atlas of Subfossil Cladocera from Centraland Northern Europe. wyd. Towarzystwo Przyjaciół Dolnej Wisły, Świecie, p 1–84
Tavşanoğlu UN (2012) Zooplankton adaptation strategies against fish predation in Turkish shallow lakes. PhD Thesis
Tavşanoğlu UN, Çakıroğlu Aİ, Erdoğan Ş, Meerhoff M, Jeppesen E, Beklioğlu M (2012) Sediment—not plants—is the preferred refuge for Daphnia against fish predation in Mediterranean shallow lakes: an experimental approach. Freshw Biol 57:795–802
ter Braak CJF (1995) Ordination. In: Jongman RHG, ter Braak CJF, van Tongeren OFR (eds) Data analysis in community and landscape ecology. Cambridge University Press, Cambridge, pp 91–173
ter Braak CJF, Prentice IC (1988) A theory of gradient analysis. Adv Ecol Res 18:271–317
Ustaoğlu MR, Mis DÖ, Aygen C (2012) Observations on zooplankton in some lagoons in Turkey. J Black Sea/Mediterr Environ 18(2):208–222
Vandekerkhove J, Declerck SAJ, Jeppesen E, Conde-Porcuna J, Brendonck L, De Meester L (2005) Dormant propagule banks integrate spatio-temporal heterogeneity in cladoceran communities. Oecologia 142:109–116
Vijverberg J (1980) Effect of temperature in laboratory studies on development and growth of Cladocera and Copepoda from Tjeukemeer, The Netherlands. Freshw Biol 10:317–340
Williams WD (1987) Salinization of rivers and streams: an important environmental hazard. Ambio 16:180–185
Acknowledgments
We are grateful to S. L. Amsinck, L. S. Johansson and K. Jensen for help with the identification of cladoceran remains. This study was supported by Middle East Technical University (METU)-BAP programme of Turkey (BAP.07.02.2009-2012), TÜBİTAK-ÇAYDAG (Projects Nos: 105Y332 and 110Y125) and FP-7 REFRESH (Adaptive strategies to Mitigate the Impacts of Climate Change on European Freshwater Ecosystems, Contract No.: 244121) and the MARS project (Managing Aquatic ecosystems and water Resources under multiple Stress) funded under the 7th EU Framework Programme, Theme 6 (Environment including Climate Change), Contract No.: 603378 (http://www.mars-project.eu). AİÇ, ÜNT and EET were also supported by TÜBİTAK (Project Nos.: 105Y332 and 110Y125), TB was supported by TÜBİTAK 2211 Scholarship Programme, AÖ was supported by METU-ÖYP Programme, and TAD’s contribution was supported by CIRCE, funded by the AUFF–AU Ideas program. AİÇ is also thankful to the EU-Erasmus Student Exchange Programme for the fellowship during her stay in Denmark. The authors are grateful to A. M. Poulsen for editing the manuscript. We also want to thank Lisa Doner, Korhan Özkan, Damla Beton, Şeyda Erdoğan, Gizem Bezirci, Nur Filiz, Burcu Yeşilbudak, Seval Özcan, Ceran Şekeryapan, Mengü Türk, Semra Yalçın and Mukadder Arslan for their invaluable support for this work both in the field and in the laboratory.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Çakıroğlu, A.İ., Tavşanoğlu, Ü.N., Levi, E.E. et al. Relatedness between contemporary and subfossil cladoceran assemblages in Turkish lakes. J Paleolimnol 52, 367–383 (2014). https://doi.org/10.1007/s10933-014-9799-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10933-014-9799-x