Abstract
Cladocerans are increasingly used in palaeolimnological studies as their community composition is sensitive to both anthropogenic and natural forces in lakes. We present the results of a palaeolimnological investigation of three Turkish shallow lakes located in cold dry steppe and semi-dry Mediterranean climatic regions. The aim was to elucidate historical changes in environmental conditions by analysing sub-fossil cladocerans in 210Pb-dated sediment cores. Sub-fossil cladoceran remains from the surface sediment of 40 Turkish lakes were analysed to examine the environmental factors that most correlated with variation in the cladoceran assemblage. Redundancy analysis showed that salinity, macrophyte abundance, fish density, depth and total phosphorus were the most correlated with change in cladoceran assemblage composition with eigenvalues for the first and the second axes being λ 1 = 0.312 and λ 2 = 0.061, respectively. Sedimentary cladoceran assemblages from three cores were placed passively within the framework of the surface sediment ordination. The results reveal a prevalent impact of salinity, fish abundance and water level changes from the past to present. Thus, using cladoceran-based inferences, we traced key environmental changes related to variation in climate change, restoration and water level regulation over the last century.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Mediterranean shallow lakes presently face serious pressures due to increasing nutrient loading and water level changes, the latter resulting from changes in climate as well as increasing water abstraction (Coops et al., 2003; Papastergiadou et al., 2010), with implications also for salinity and further eutrophication (Beklioglu et al., 2011). Water quality and biodiversity are therefore increasingly threatened. Turkey, with its large variation in climate and topography, has numerous shallow lakes, ponds and reservoirs with very rich freshwater biodiversity (Şekercioğlu et al., 2011; Çakıroğlu et al., 2014; Levi et al., 2014; Boll et al., in revision). The lakes are mostly shallow with large surface areas and often eutrophic with many being impacted by changes in water level due to enhanced water abstraction and climate variability (Beklioğlu et al., 2006; Beklioglu & Tan, 2008). Following the Industrial and Green Revolutions of the nineteenth and the twentieth centuries, agriculture began to have negative impacts on the environment (Rehber, 1991). Agricultural land use has changed in the Mediterranean region, particularly since the 1950s, through increasing agricultural intensification with use of fertilisers along with industrialisation, urbanisation, and mass tourism (Tanrıvermiş, 2003). Between 1927 and 1957, many woodlands were replaced by arable agriculture in Turkey and by the 1950s, the population of farmers had increased threefolds (Anaç & Esetlili, 2012). Information on the long-term dynamics and past trophic state of Turkish lakes is, however, limited due to lack of established long-term monitoring programme.
The predicted changes in climate are expected to have adverse impacts on ecosystem structure, function and on many individual species and communities of lakes (IPCC, 2007, 2014). Being mostly part of the Mediterranean climatic region as well as predominance of cold dry steppe climates, Turkey is highly vulnerable to impacts of climate change. Many studies have revealed an ongoing warming trend from the early 1990s (Türkeş et al., 1995, 2002; Kadıoğlu 1997). Furthermore, recent studies have reported a trend of decreasing precipitation in the western and southern parts of Turkey (Türkeş et al., 2009), which affects the hydrology of shallow lakes, especially in Central Anatolia where lakes are highly susceptible to changes in hydraulic residence time and water level (Beklioğlu et al., 2006; Özen et al., 2010; Özkan et al., 2010; Bucak et al., 2012). Hydrological alterations and regional climate changes have led to salinization and further eutrophication of many Turkish shallow lakes (Beklioğlu et al., 2006, 2011; Beklioglu & Tan, 2008; Özen et al., 2010; Bucak et al., 2012; Çakıroğlu et al., 2014; Levi et al., 2014).
To be able to predict the future effects of these multiple stressors on Turkish lakes, it is useful to place current conditions in a long-term perspective. Palaeolimnological techniques may provide a broader perspective on ecological changes, and sub-fossil cladocerans have been shown to have a great potential in elucidating nutrient- and climate-change-driven effects on lake ecosystems (Davidson et al., 2011a; Nevalainen et al., 2013). Cladocera are particularly sensitive to change as they hold an intermediate position in the food web, linking the bottom (algae) and the top (zooplanktivorous fish) of the food web (Jeppesen et al., 1997; Moss, 1998; Abrantes et al., 2006; Jeppesen et al., 2011). Consequently, cladocerans are strongly controlled by bottom-up changes in resources, lateral changes in habitat structure (e.g., macrophyte abundance) and a top-down impact of predation by fish and invertebrates (Jeppesen et al., 2001). Remains of most cladocerans are well-preserved in sediment and their relative abundance in the sediment largely reflects the original size of the population in the water column (Korhola & Rautio, 2001; Jeppesen et al., 2003; Szeroczynska & Sarmaja-Korjonen, 2007; Çakıroğlu et al., 2014). Cladocera remains have been used in different systems to infer, both quantitatively and qualitatively, trophic structure, plant composition, fish population, climatic change, eutrophication, salinity, temperature and water level fluctuation (Jeppesen et al., 1996; Hofmann, 1998; Lotter et al., 1998; Bos et al., 1999; Jeppesen et al., 2003; Amsinck et al., 2005; Johansson et al., 2005; Kattel et al., 2008; Nevalainen et al., 2011).
In the present study, 40 shallow Turkish lakes were studied to establish the key controls of cladoceran community composition. The study aimed to elucidate historical changes in environmental conditions, such as trophic state and salinity, in three shallow lakes (Lakes Mogan, Eymir and Gölhisar) located in cold dry steppe and semi-dry Mediterranean climatic regions of central to western Turkey using sub-fossil cladoceran taxa in 210Pb-dated short sediment cores. Besides being affected by climate, two of the lakes (Lakes Mogan and Eymir) have been subjected to increasing nutrient loading as well as diversion of external nutrient loadings through sewage effluent diversions (Altınbilek et al., 1995). Moreover, a lake-restoration programme at Lake Eymir using biomanipulation approach has been carried out following the sewage effluent diversion for further enhancement of the water quality. Lakes Mogan and Eymir have had nearly 20 years of fortnightly-to-monthly monitoring programme for physical, chemical and biological variables making a ground true evaluation of palaeolimnological data for the recent period feasible. The third lake (Lake Gölhisar) has also been affected by hydrological change, chiefly by water level change because of irrigation as well as a major decrease in precipitation.
We hypothesised that (i) sub-fossil cladoceran remains retrieved from the cores would provide a good indication of key environmental changes such as, water level changes, eutrophication and salinization over the last century and that (ii) sedimentary cladocerans would support the historical observations and monitoring data from Lakes Mogan and Eymir such as fish biomanipulation, salinity and nutrient changes.
Materials and methods
Study sites
Lakes Mogan, Eymir and Gölhisar, have different climate, trophic status and lake-management histories. They are therefore representing different tests of using Cladocera sub-fossils to infer past change.
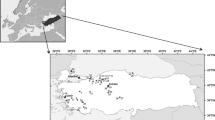
Lake Mogan (39°46′N, 39°47′E) and Lake Eymir (39°49′N, 39°49′E) are connected shallow lakes located in the Ankara province in the Central Anatolia region (Fig. 1). Lake Mogan lies upstream of Lake Eymir and has an area of 5.4 km2, an elevation of 973 m, a mean depth of 2.8 m and a maximum depth of 4 m. Downstream Lake Eymir is comparatively small, has a surface area of 1.25 km2, an elevation of 969 m, a mean depth of 3.1 m and a maximum depth of 6 m.
The two lakes are situated in a cold dry steppe climate (Central Anatolia) characterised by hot and dry summers, the driest period being from June to September but also with cold and snowy winters. Data between 1980 and 2011 from the Gölbaşı Meteorological Station situated near the lakes show an average precipitation of 405 ± 80 mm and an average temperature 11.9 ± 8.4°C (Turkish State Meteorological Service, 2011). The water level fluctuations reveal two dry periods in both lakes (Özen et al., 2010)—low water level (LWL) periods in the dry year 2001 and between 2005 and 2010, and a high water level period during the years in between.
Previous studies have characterised Lake Mogan as a clear-water lake with high macrophyte coverage which is sensitive to water level fluctuations (Burnak & Beklioğlu, 2000; Zhang et al., 2003a, b). Lake Mogan received a relatively less important quantity of sewage effluent, only from the neighbouring restaurants and houses along the shore that was also diverted, though, and it was not as high as for Lake Eymir (Altınbilek et al., 1995). After 2000, the fish community in Lake Mogan has shifted from dominance by Esox lucius Linnaeus, 1758, Tinca tinca (Linnaeus, 1758), Cyprinus carpio Linnaeus, 1758, and Siluris glanis Linnaeus, 1758, to dominance by exotic and invasive species such as Pseudorasbora parva (Temminck & Schlegel, 1846), accompanied by complete disappearance of E. lucius (Beklioğlu et al., unpublished data; DSI, 1993; Akbulut & Akbulut, 2002; ÖÇKK, 2002; Manav & Yerli, 2008).
Lake Eymir received raw sewage effluents for more than 25 years from the neighbouring town until sewage was diverted in 1995 (Altınbilek et al., 1995; Beklioğlu et al., 2003). The diversion reduced the external loading of total phosphorus (TP) by 88%, but water clarity did not improve accordingly as in-lake nutrient concentrations remained very high (Beklioğlu et al., 2003). As an attempt to restore, the lake biomanipulation was conducted from 1998 to 1999, where 50% of the planktivorous fish stock (T. tinca, and C. carpio) was removed, and E. lucius fishing was banned (Beklioğlu et al., 2003). Following this fish removal, water clarity improved, and submerged macrophyte coverage increased from 2.5 to 40–90%. However, 5 years after the first fish removal, the ecological state deteriorated again, coinciding with a period of extended drought, which led to increased nutrient concentrations that reached to the pre-biomanipulation levels, and at the same time, fish biomass increased (Beklioglu & Tan, 2008; Özen et al., 2010). A second biomanipulation was initiated in 2006 and continued until the end of 2014.
Lake Gölhisar (37°8′N, 29°36′E) is a 400-ha shallow lake located in the Burdur province in south western part of Turkey and has an elevation of 930 m (Fig. 1). Maximum depth is 1.6 m, and the hydrological catchment covers ~300 km2. Small springs around the margin of the lake flow into and out of the lake through the groundwater discharge (Jones et al., 2002). Average annual precipitation is low, 560 mm, with 50% falling during winter and 12% during summer (Eastwood et al., 1999). During the last 15 years, seven dams and ponds have been established in the area for irrigation purposes (DSI, 2000), and there are several number of illegal irrigation wells in the catchment of the lake. Moreover, a sluice gate was built in 1993 in order to regulate the water level and drain water into the nearby Dalaman River (Alp, 1997).
For Lake Gölhisar, the data on August maximum temperature (°C) and average precipitation (mm) used in this study were retrieved from the European climate assessment and dataset project and average evaporation (mm) data derived from the Turkish State Meteorological Service.
Field sampling
Snapshot sampling
Surface sediments were sampled once in each of the 40 lakes during the peak growing season of 2006–2012 (Moss et al., 2003). The 40 study lakes were all shallow and small and spanned over five latitudes (Çakıroğlu et al., 2014; Levi et al., 2014) between the warm and dry temperate, arid cold steppe in a Mediterranean climate according to the Köppen–Geiger classification system (Peel et al., 2007; Fig. 1). Depth-integrated, mixed water samples covering the entire water column were taken at the deepest part of the lake and analysed to determine concentrations of TP, soluble reactive phosphate (SRP), nitrite–nitrate, total nitrogen (TN) and chlorophyll a (Chla). Lake water temperature (°C), conductivity (±1 µS cm−1), salinity (‰), maximum depth (m), dissolved oxygen (mg O2 l−1) and pH were determined in situ using a YSI 556 MPS multiprobe, and Secchi depth was measured. The composition and abundance of fish (catch per unit effort, CPUE, number net−1) were determined by overnight (12 h) fishing with gill nets with 12 multiple mesh sizes ranging from 5 to 55 mm. Per cent plant volume inhabited (PVI%) of the lakes was calculated based on data on submerged plants sampled at evenly spaced intervals along parallel transects. From each lake, seven surface sediment (0–2 cm) samples were taken using a KC-Denmark Kajak Corer (5.2-cm diameter) at the deepest point of the lake and mixed. For further details about sampling and chemical analyses, see Çakıroğlu et al. (2014) and Levi et al. (2014).
Monitoring of Lakes Mogan and Eymir
Since 1997, Lakes Mogan and Eymir have been sampled fortnightly-to-monthly to determine nutrients (TN, NH4-N, NO3 + NO2-N, TP, SRP), zooplankton and phytoplankton abundances and Chla concentration. The samples were collected at the deepest point of the lakes, and Secchi depth, dissolved oxygen, conductivity and salinity were measured in situ using multiprobe field equipment. Macrophyte abundance was estimated using PVI% in samples taken along parallel transects, and data on submerged macrophytes were collected once a year in September. In Lake Eymir, fish surveys were conducted between 1997 and 2007 using multi-mesh gill nets (100 m long and 3.5 m high nets with mesh sizes of 7, 9, 12, 16, 22, 25, 36, 42, 55 and 65 mm). After 2007, fish samplings have been undertaken in both lakes by the end of the summer using gill nets with 12 mesh sizes (length 30 m, height 1.5 m, mesh sizes 5.0, 6.25, 8.0, 10.0, 12.5, 15.5, 19.5, 24.0, 29.0, 35.0, 43.0 and 55.0 mm), as used in the 40 lakes survey also.
Palaeolimnological sampling and analyses
In 2008, sediment coring was conducted using a KC-Denmark Kajak Corer in Lakes Mogan and Eymir and 55- and 60-cm-long cores were collected, respectively, at the deepest point of the lakes. The cores were sectioned at 1-cm intervals in the field. Sub-fossil cladocerans were sampled for every 5-cm and at 1-cm intervals in the top 10 cm. In 2007 in Lake Gölhisar, a 51-cm-long sediment core was collected and sectioned in the field at 1-cm intervals. Sub-fossil cladocerans were determined at 5-cm intervals.
For sub-fossil cladoceran taxonomic analysis, 5 g wet homogenised sediment was boiled in 100 ml 10% KOH for 50 min, and all materials were subsequently kept in Lugol’s solution, and the samples were sieved prior to microscopic analysis. For the counting process, sub-fossils were separated into two size fractions: >140 and 45–140 µm. All remains >140 µm were counted, while remains in the 45–140-µm size fraction were subsampled; approximately 20% was counted using a stereomicroscope (Leica MZ 16) and an inverted light microscope (Leica DMI 4000). For species identification, the keys developed by Frey (1959), Flöβner (2000) and Szeroczynska & Sarmaja-Korjonen (2007) were used. Carapaces, head shields, post abdomens, post-abdominal claws and resting eggs (ephippia) of each sub-fossil cladoceran were counted, and the most abundant body part was used in the analyses. We adjusted the counts of remains to be representative of the Cladocera as individuals; thus, one individual is composed of one head shield, two carapace halves and one post-abdomen (Korhola & Rautio, 2001). For Daphnia, Ceriodaphnia, Simocephalus (Simo) and Moina, the abundance of individuals was based on ephippia because they were dominant body part encountered in the samples. Taxonomic species’ separations of Alona rectangula Sars, 1862 and Alona guttata Sars, 1862 from the head pores were occasionally not possible if no post abdomens were present. These two Alona species are consequently used as A. rectangula/guttata (Arecgut) in the statistical analyses.
Laboratory analyses
Dating
The sediment cores were dated by means of 210Pb-dating. The sediment samples from Lakes Mogan and Eymir were analysed for activities of 210Pb, 226Ra and 137Cs via gamma spectrometry at the Gamma Dating Center, Department of Geosciences and Natural Resource Management, University of Copenhagen. The measurements were carried out on a Canberra ultra low-background germanium well-detector. 210Pb was measured via its gamma-peak at 46,5 keV, 226Ra via the granddaughter 214Pb (peaks at 295 and 352 keV) and 137Cs via its peak at 661 keV. The Lake Gölhisar sedimental samples were analysed for activities of 210Pb, 226Ra and 137Cs via gamma spectrometry at Flett Research, Canada.
Data analyses
Ordination
Environmental data from the 40 lakes were tested for normality using the Kolmogorov–Smirnov test in SigmaStat 3.5 (Justel et al., 1997). Environmental variables that did not pass the normality test were either log10, log10(x + 1) or square root transformed to obtain the best approximation to normal distribution (Table 1), and species data were Hellinger transformed (Legendre & Gallagher, 2001).
Ordination is an effective method to investigate the patterns and causes of variation in multivariate datasets (ter Braak & Prentice, 1988). Detrended correspondence analysis (DCA) was first performed on Hellinger-transformed cladoceran data in order to determine whether unimodal or linear ordination techniques were suitable for the data of the current study. The length of the first axis, 2.21 standard deviation units, in DCA supported use of the linear ordination method, redundancy analysis (RDA; ter Braak, 1995). Non-parametric Spearman’s correlation was applied to all environmental variables in Table 1. Highly correlated environmental variables were identified using the Spearman’s rank correlation and by means of variance inflation factors (VIFs). Those with VIF >5 were deleted from the environmental dataset (DeSellas et al., 2008). The remaining variables controlled by eutrophication and salinity also had a very strong impact on cladoceran community composition (Bos et al., 1999; Jeppesen et al., 2001; Amsinck et al., 2005; Davidson et al., 2010a) and were chosen as input environmental variables in the RDA—these were: macrophytes (PVI%), fish (fish CPUE), TP, depth and salinity. The unique effects of each environmental factor on species distribution of Cladocera and joint effects were partitioned and quantified using variation partitioning analysis (Borcard et al., 1992).
The linear ordination method, principal component analysis (PCA), was performed to summarise change in the sub-fossil cladoceran assemblages for each record since the gradient lengths for DCA were 1.85 for Lake Mogan, 0.95 for Lake Eymir and 1.63 for Lake Gölhisar. Constrained clustering analysis CONISS (Grimm, 1987) was conducted on the cladoceran data for the three cores in order to identify key points of change, zones, in the cladoceran assemblage. The significance of the CONISS zones was determined with the broken stick model (Bennett, 1996). Ordinations were carried out using the vegan (Oksanen et al., 2010) and rioja (Juggins, 2009) packages in R (The R Foundation for Statistical Computing version 2.12.2).
RDA was also used to estimate the historical changes in environmental variables. The analysis used the framework established by applying the RDA of the surface sediment cladoceran assemblages calibrated against the selected environmental variables impacting cladoceran community composition (selection, see above). The samples from the short sediment cores of the three lakes were then passively placed within this framework. Therefore, the position of the historical sediment samples may be used to estimate the past environmental conditions of the lakes. For each RDA analysis, the site for which the conditions were reconstructed was removed from the calibration set. All ordination analyses were completed using CANOCO version 4.5 with Hellinger-transformed cladoceran and transformed environmental data.
Results
Environmental data
The 40 study lakes were generally small and mostly shallow with a maximum depth ranging from 0.55 to 17.4 m (Table 1). The summer surface water temperature of the 40 lakes averaged 24.4°C, and the lakes were well oxygenated (average 6.7 mg O2 l−1). Most of the lakes had pH values close to neutral. The lakes were generally eutrophic (average TP 138.1 μg l−1, TN 1,024.7 μg l−1, Chla 19.2 μg l−1, Secchi depth 130 cm; Table 1). The mean abundance of fish—CPUE—was 107 and PVI% was 20.2% (Table 1).
There were a number of statistically significant correlations among some of the environmental variables in the data set (Table 2). Spearman’s correlation revealed strong and significant positive relationships between TP and TN, Chla, and temperature and a significant negative relationship between TP and Secchi disc depth. There were also positive significant correlations between salinity and TP, TN and Chla, whereas a significant negative correlation appeared between salinity and depth.
Species data
A total of 25 taxa were identified in the surface sediments of the 40 lakes (Table 3). Bosmina longirostris (O.F. Müller, 1785) and Chydorus sphaericus (Chyd; O.F. Müller, 1776) were the most widespread and also the most abundant species. The Chydoridae family with 18 taxa exhibited the highest species richness (see Çakıroğlu et al., 2014, for details).
Ordination
Salinity, PVI%, fish CPUE, depth and TP explained 41.1% of the total variation in the species composition of the sub-fossil cladocerans (Fig. 2). The eigenvalues of the RDA for the first and the second axis were λ 1 = 0.312 and λ 2 = 0.061, respectively. According to the biplot scores for constraining variables, axis 1 was correlated with salinity (0.54), PVI (0.33) and TP (0.16), and negatively correlated with depth (−0.52). Axis 2 was positively correlated with fish CPUE (0.35), salinity (0.35) and TP (0.14), and negatively correlated with PVI (−0.21) as well as depth (−0.05) (Fig. 2). Variance partitioning analysis identified the variances in the cladoceran data, which were uniquely attributable to salinity and depth as 10.6 and 10.9%, respectively, with lower proportions explained by fish, PVI% and TP; 6.6, 4.6 and 1.3%, respectively. The variance uniquely explained by a combination of depth and salinity combined was higher (23.1%) when comparing this with the individual unique effects (10.9 + 10.6 = 21.5%); it shows the shared effect was 1.6%. For fish and TP together, the unique and total effects were 8.3 and 7.9%, respectively; thus, the shared variance was 0.4%. For salinity and PVI% together, the unique and total effects were 16.3 and 15.2%, respectively, with 1.1% shared effect. For salinity and TP together, the unique and total effects were 11.9 and 11.5%, respectively, with 0.4% interaction.
RDA of the 40 study lakes and sedimentary cladoceran samples with environmental variables. The lakes were numbered relative to a salinity gradient, 1 for the lowest and 40 for the highest salinity. Lake sites are shown as circles. For species abbreviations, see Table 3
Chydorus sphaericus (Chyd), Leydigia leydigi (Schoedler, 1863) (Lleyd), Leydigia acanthocercoides (Fischer, 1854) (Lacanth), Pleuroxus spp. (Pleuspp) and Simo were positively associated with salinity, while most cladocerans, in particular B. longirostris (Bosm), were negatively related to salinity but positively related to water depth (Fig. 2). The vegetation- and sediment-associated taxa Alona rectangula/guttata (Arecgut) and Oxyurella tenuicaudis (Sars, 1862) (Oxyu) exhibited a close relationship with PVI%. Large-bodied cladoceran taxa, especially Daphnia spp. (Daph), Leptodora (Leptod) and Graptoleberis testudinaria (Fischer, 1848) (Grapto), correlated with fish CPUE (Fig. 2). Lakes 18, 15 and 35 had the highest number of fish but lakes 15 and 35 also the highest abundance of Daphnia (Fig. 2).
Core chronology, cladoceran stratigraphy and historical changes
Lake Mogan
The core showed surface contents of the unsupported 210Pb of about 370 Bq kg−1, and the activity generally decreased exponentially with depth. The calculated flux of the unsupported 210Pb was 164 Bq m−2 year−1, which was higher than the estimated local atmospheric supply (based on data in Appleby, 2001). This indicates that sediment was moved from shallower to deeper zones by sediment focussing. Significant activity of 137Cs was found down to a depth of about 40 cm, and the activity above 30 cm was also relatively high but without a distinct peak. Precise dating of the core was hampered by the irregular profile of the unsupported 210Pb. The sedimentation rate was calculated to be 2.1 kg m−2 year−1. CRS-modeling (Appleby, 2001) was applied, and 210Pb dates were calculated; for the bottom of the core (55 cm), and the date was calculated to be as the year 1,860 ± 18.
Twelve cladoceran taxa were identified in the core. Based on the cluster analysis, the record can be split into three zones (Fig. 3). Zone 1 (55–44 cm, ca. 1860–1960) was dominated by macrophyte-sediment-associated taxa of which C. sphaericus and A. rectangula/guttata were the most abundant species. Pelagic taxa were rare at the onset of zone 1, while the abundances of B. longirostris and Moina sp. increased in zone 2 indicating more open water conditions. Chydorids dominated the cladoceran community in zone 1, with the decreasing abundance in zone 2 (Fig. 3). The abundance of macrophyte-sediment-associated taxa remained relatively high except that Camptocercus rectirostris Schoedler, 1862, and Alonella exigua (Lilljeborg, 1853) disappeared after 30 cm (Fig. 3). In zone 3 (5–0 cm), the total cladoceran abundance increased sharply at 5 cm (Fig. 3) where Simocephalus sp. was first identified and Pleuroxus spp. reappeared.
When plotting the Lake Mogan core sample scores from RDA ordination by means of cladoceran abundance data from each depth with environmental variables from the calibration set, a clear differentiation after ca. 1950 (25–26 cm) was observed as captured in a change of PCA axis 1 scores (Figs. 3, 4). The distribution of the Lake Mogan core samples relative to the environmental vectors in the RDA ordination indicated higher salinity in zone 1 and in the first half of zone 2 (Fig. 4). Furthermore, in zone 2, PVI% seemed to have reached relatively high levels with a decreasing trend towards the present day. Zone 2 was characterised by high TP. Moreover, in zone 3, salinity and fish seemed to affect the cladoceran community.
Lake Eymir
The core showed surface contents of the unsupported 210Pb of about 140 Bq kg−1, and the activity generally decreased exponentially with depth. The calculated flux of the unsupported 210Pb was 277 Bq m−2 year−1, which was higher than the estimated local atmospheric supply (based on data in Appleby, 2001), indicating sediment focussing. Significant 137Cs activity was found in all samples; however, the profile of 137Cs showed clear signs of leaching, and the chronology based on 210Pb could not be verified using this isotope. The sedimentation rate was calculated to be 1.7 kg m2 year−1.
A total of 11 cladoceran taxa were identified. Three zones were determined by cluster analysis (Fig. 5). Zone 1 (60–36 cm, ca. 1860–1930) was dominated by the pelagic taxa B. longirostris and Moina sp. as well as by the macrophyte-sediment-associated C. sphaericus, A. rectangula/guttata and Pleuroxus spp. and the sedimentary species L. leydigi, whereas true macrophyte-associated taxa were absent. Furthermore, at the bottom of the core, the pelagic cladoceran Daphnia spp. was found but disappeared; but reappeared in zone 2 from around 1900 to the 1980s (35–15 cm). Among the pelagic cladocerans, B. longirostris abundance decreased in zone 2. For the first time, the macrophyte-associated taxon Graptoleberis testudinaria (Fischer, 1848) appeared in zone 2. The levels of macrophyte-sediment and sediment-associated species remained the same as in zone 1, and Ceriodaphnia sp. and L. acanthocercoides appeared for the first time in zone 2 (Fig. 5). In zone 3, from ca. 1995 and onwards, the abundance of small-bodied B. longirostris remained low, whereas the abundance of large-sized Daphnia spp. appeared to be high.
In Lake Eymir, fish CPUE had relatively high levels and showed an increasing trend from zone 1 to the onset of zone 3. The pre-biomanipulation years, corresponding to 7 to 8-, 9 to 10- and 15 to 16-cm core depth, showed high fish CPUE followed by a decline; however, it increased again towards 0–1 cm. From zone 1 to zone 3, TP increased except for the post-biomanipulation years. Unlike Lake Mogan, the RDA ordination did not indicate a shift towards higher salinity (Fig. 6).
Lake Gölhisar
In accordance with the constant rate of 210Pb supply found by CRS modelling, the sedimentation rate was calculated to be 3.4 kg m−2 year−1. The deepest point of the core for which the last 210Pb signal was determined was 46 cm, corresponding to 1942.
A total of 14 cladoceran taxa were recorded based on chitinous and ephippial remains along the sediment core (Fig. 7), B. longirostris having the highest abundance. Other abundant taxa were A. rectangula/guttata, C. sphaericus and L. leydigi. Cluster analysis identified three different zones, zone 1 ranging from the bottom of the core to 35 cm. Macrophyte-associated C. rectirostris was only found in zone 1. Zone 2 ranged from 35 to 5 cm and was characterised by high relative abundances of B. longirostris and occurrence of all other taxa except Acroperus harpae (Baird, 1835), C. rectirostris and A. exigua. In zone 3, which covered the recent period from 4 cm to the top of the core, the pelagic species B. longirostris and Moina sp. decreased in abundance towards the present day, and A. exigua reappeared.
For Lake Gölhisar, core sample scores from RDA ordination showed depth-related changes (Fig. 8). From the onset of zone 3 to the top, the effect of water depth was apparently less prominent and the lake became more saline. Zone 2 was also characterised by relatively high fish CPUE levels (Fig. 8).
Discussion
Cladoceran assemblages are structured by multiple forces (Davidson et al., 2010a). The surface sediment dataset presented here reflects this, with a relatively large amount of variance (41.1%) shared amongst five variables (depth, salinity, fish CPUE, PVI%, and TP). There was some amount of shared variance between correlated variables; for example, the negatively correlated depth and salinity—but the proportions were relatively small. It was notable that there was only a small proportion of variance shared by fish CPUE and TP, given that CPUE has been shown to increase with TP (Jeppesen et al., 2000). Passive placing of sedimentary cladoceran assemblages in an ordination space, constructed from surface sediment data, allows for the qualitative inference of past change in these multiple variables. This approach has been used alone and in combination with multivariate regression trees (Davidson et al., 2010b). Although the approach is more flexible than a transfer function, as it is not limited to a single variable, its weakness is that it is more difficult to interpret. This is because a movement in ordination space can, for example, represent an increase in fish CPUE and/or a decline in macrophyte abundance, or perhaps both. Here we have combined monitoring data, where available, and also used the known ecology of the cladoceran taxa to identify the most plausible drivers of past change in the cladoceran assemblage and therefore to infer past conditions.
The core data, placed within the framework established using the surface sediment data, suggest that the ecology of all three lakes was altered after ca. 1950, almost certainly as a result of increased anthropogenic pressures such as irrigation for intensive farming and waste water discharge. The core data from Lake Mogan clearly showed the shift from a benthic to a pelagic assemblage after ca. 1960, this likely being due to domestic wastewater input from the catchment as well as carp stocking which stirred up the bottom sediments (Tanyolaç & Karabatak, 1974) and likely enhanced predation pressure on zooplankton as indicated by a high proportion of Bosmina among the pelagic species (Fig. 3). At Lake Eymir, the core data identified the effect of biomanipulation by reflecting, not only the lower fish CPUE post 1996 until 2000, but also the resultant improvement of the water quality (Fig. 6). Several other studies have also demonstrated effects of fish biomanipulation and fish stockings on lake ecosystems by means of cladoceran remains in the sediment as proxies (Skov et al., 2010; Buchaca et al., 2011; Davidson et al., 2011b; Jeppesen et al., 2011). The core data from Lake Gölhisar indicated a lake level decrease from the 1940s to the beginning of the 1990s and then again a relative increase until the present day (Fig. 8), possibly as a result of water level regulation after 1993 (15 cm), leading to a somewhat deeper lake (10 cm).
The changes suggested by the sub-fossil Cladocera assemblages (Figs. 4, 5) agree with historical observations and the existing monitoring data. For example, in Lake Mogan marked changes occurred in the 1960s, with the increasing relative contribution of pelagic taxa and among the benthic taxa of species indicative of eutrophication, C. sphaericus and A. rectangula/guttata (Gasiorowski & Szeroczynska, 2004; Chen et al., 2010; Davidson et al., 2010a). C. sphaericus can invade the pelagic zone and become super-abundant in the chydorid assemblage in the sediment of nutrient-enriched lakes (Hofmann, 1987). In addition, the abundance of daphniids increased, which may suggest increased food abundance (Davidson et al., 2011a) or decreased predation through turbidity, which has been shown to be an effective refuge for large-bodied zooplankton in Turkish shallow lakes when fish CPUE are at intermediate levels (Tavşanoğlu et al., 2015). In the RDA, the large-bodied cladoceran taxa, especially Daph, Leptod and Grapto correlated positively with fish CPUE likely reflecting that lakes 15, 18, 23, 32, 33, and 35 were turbid, with Secchi disc depth:maximum depth ratio being less than 0.3 (Fig. 2). Other studies have also shown that higher turbidity in a eutrophic lake may, to some extent, act as a refuge for daphniids from fish predation (Horppila et al., 2004; Nurminen et al., 2008) although they are substituted by small cladocerans at high fish densities irrespective of turbidity (Jeppesen et al., 2003). A gradual increase in nutrient concentrations since 1975, due to domestic wastewater input from the catchment, has been reported for Lake Mogan (Tanyolaç & Karabatak, 1974; Yerli et al., 2012). Accordingly, Burnak & Beklioğlu (2000) found grazing pressure from Daphnia spp. to be relatively low in 1997 and 1998, corresponding well with the core data showing low abundance of Daphnia spp. at 10 cm (ca. 1997). Above 4 cm, Daphnia spp. became more abundant (Fig. 3), concurring with a LWL between 2005 and 2008, which resulted in increased macrophyte coverage and PVI, as shown by monitoring data. This increase in PVI may have offered a refuge (Mangıt & Yerli, 2010; Tavşanoğlu, 2012), but also recorded eutrophication-related fish kills (Beklioğlu et al., unpublished data) have likely contributed.
An observed gradual eutrophication expected from the change in external nutrient loading and the monitoring data is also reflected by the changes in the sub-fossil cladoceran assemblage of the Lake Eymir core (until year 2008; Fig. 6). From the onset of zone 2, pelagic species, especially Daphnia spp., appeared in relatively high abundances, while the abundance of benthic species remained unaltered (Fig. 5), suggesting that Lake Eymir became more eutrophic during this period (36–19 cm). The eutrophication continued in zone 3 with a possible ecosystem shift from the dominance of benthic to pelagic taxa (Vadeboncoeur et al., 2003; Davidson et al., 2010a) followed by some recovery beginning after ca. 1996 (6–7 cm). These results fit well with the monitoring data. The lake received sewage effluents from ca. 1970 until sewage diversion in 1995, followed by an 88 and a 95% reduction in the loadings of TP and dissolved organic nitrogen, respectively (Altınbilek et al., 1995; Beklioğlu et al., 2003). During the eutrophication period, the highest peak of Daphnia spp. was observed (15–16 cm, ca. 1980s), reflecting either the observed fish kills during the 1980s (Altınbilek et al., 1995; Beklioğlu et al., 2003) or more abundant food resources. The additional Daphnia spp. peak in the sediment record (at 3–4 cm, ca. 2000) corresponds to the post-first biomanipulation as well as during the second biomanipulation period characterised by lower abundance of the dominant fish species T. tinca and C. carpio, a twofold lower Chla concentration and a fourfold higher Secchi depth and 40–80% macrophyte coverage than in the years before manipulation (Beklioğlu et al., 2003; Beklioglu & Tan, 2008). However, 5 years after biomanipulation, perhaps as a result of severe drought, resulting in a water level drop of 2 m and enhanced nutrient concentrations, the lake became eutrophic with high turbidity and fish biomass increased (Beklioğlu et al., 2003; Beklioglu & Tan, 2008; Özen et al., 2010). This is reflected in the cladoceran sediment record between 2 to 3- and 1 to 2-cm depths (Fig. 6). The increase in C. sphaericus, sometimes an indicator of eutrophication (Brodersen et al., 1998), in the top 0–1 cm coincides in time (2008) with the lowest water level that led to severe eutrophication (Özen et al., 2010). These changes suggest that sedimentary cladocerans reflect the long-term monitoring observations of eutrophication and the subsequent environmental deterioration of Lake Eymir.
The RDA data also reflected the historical changes in Lake Gölhisar. Both the historical observations and the cladoceran record suggest that up to ca. 1940, Lake Gölhisar was affected by hydrological changes, but not eutrophicated. The sediment record indicates that fish CPUE increased towards the early 1990s (Fig. 8). The dominant taxa in lakes with high fish abundance are often the small-bodied B. longirostris, C. sphaericus and A. rectangula/guttata, and they were indeed abundant throughout the core (Fig. 7). Average yearly precipitation was approx. 590 mm and evaporation 664 mm in the region, and a regional drought analysis has shown increased aridity from 1960 to the present day (Turkish State Meteorological Service, 2014). In addition to this high evaporation, seven dams and ponds have been built in the catchment for irrigation during the past 15 years (DSI, 2000), and an unknown number of illegal irrigation wells have extracted groundwater as well (Tübitak Marmara Research Center, 2013). Several studies on Mediterranean lakes have pointed that a reduced water table and altered hydrology often result in eutrophication through up concentration and enhanced internal loading (Brucet et al., 2009; Özen et al., 2010; Jeppesen et al., 2015). In accordance with this, after 1960, the number and abundance of pelagic species, especially B. longirostris, increased (Fig. 7). In contrast, prior to 1960, the benthic and macrophyte-associated taxa such as G. testudinaria and A. harpae were abundant. A water gauge was built at the outflow of the lake in 1993 in order to regulate the water level. Following this regulation, in the mid-1990s, pelagic taxa started to decline in the core, whereas A. harpae appeared again and the abundance of benthic species such as L. leydigi, L. acanthocercoides and A. rectangula/guttata increased. The sediment record suggests that the lake partly returned to the pre-1960 state following this water level regulation (Figs. 7, 8).
According to the projections in the 2014 IPCC report, the Mediterranean climatic regions will be markedly affected by climatic change, with significant implications for major decrease in lake water levels and salinity. Furthermore, these climate change effects may be intensified by the increased use of water for anthropogenic purposes, especially irrigation (Jeppesen et al., 2015). Warmer and drier periods with long water residence time enhance eutrophication and salinization (Beklioğlu et al., 2007, 2011; Özen et al., 2010). Cladocerans are sensitive to salinity as they are largely restricted to freshwater habitats (Dodson & Frey, 2001). Accordingly, it has been shown repeatedly that salinity influences both the richness and the composition of cladocerans (Hobaek et al., 2002; Green et al., 2005; Çakıroğlu et al., 2014). However, impact of salinity increase did not appear in the sediment records from oligo-saline Lakes Mogan and Eymir (Figs. 4, 6) despite the fact the monitoring data showed a salinity increase during the drought period from 1.1 to 2.9 ppt for Lake Mogan and from 1.0 to 2.4 ppt for Lake Eymir during the years from 1995 to 2008 (Beklioglu et al., 2011). The effect of changes in nutrients and fish on the cladoceran taxa in these lakes was apparently more pronounced than that of salinity, reflecting the relatively modest changes in salinity; stronger effects on species richness and composition are to be expected at higher salinities (Brucet et al., 2009; Nevalainen, 2010; Çakıroğlu et al., 2014). Only in Lake Mogan, the more salinity-tolerant taxa C. sphaericus, L. leydigi and Alona quadrangularis (O.F. Müller, 1785) increased in abundance towards the top of the core (Fig. 3) (Çakıroğlu et al., 2014). No clear salinity effects were detected in Lake Gölhisar either, despite the fact that a regional drought analysis has shown increased aridity from 1960 to the present day in the area (Turkish State Meteorological Service, 2014). The lack of response likely reflects the overriding effect of the major changes in water management and water level regulation in the lake and its catchment described above, although it cannot be excluded that the relatively strong correlation between depth and salinity in the training set has played a role.
In summary, the three Turkish shallow lakes started to change after ca. 1950, which was reflected by the cladoceran community structure. The cladoceran distribution of the core samples within the RDA space revealed an overall shift towards higher salinity concentrations and fish abundance in Lakes Mogan and Eymir together with hydrological alterations in Lake Gölhisar from ca. 1950 to 2008. Moreover, a transient reduction in these variables followed restoration by fish manipulation in Lake Eymir and by water level regulation in Lake Gölhisar. It appears that sub-fossil Cladocera successfully traced key environmental changes related to variation in climate change, restoration and water level regulation over the last century.
References
Abrantes, N., S. C. Altunes, M. J. Pereira & F. Gonçalves, 2006. Seasonal succession of cladocerans and phytoplankton and their interactions in shallow eutrophic lake (Lake Vela, Portugal). Acta Oecologica 29(1): 54–64.
Akbulut (Emir), N. & A. Akbulut, 2002. The plankton composition of Lake Mogan in Central Anatolia. Zoology in the Middle East 27: 107–116.
Alp, A., 1997. The research of economic fish populations (Cyprinus carpio L., 1758 and Stizostedion lucioperca L., 1758) in Gölhisar Lake. PhD Thesis, Ege University: 68 pp.
Altınbilek, D., N. Usul, H. Yazıcıoğlu, Y. Kutoğlu, N. Merzi, M. Göğüş, V. Doyuran & A. Günyaktı, 1995. Gölbaşı Mogan-Eymir Gölleri için su kaynakları ve çevre yönetim planı projesi. Technical Report No. 93-03-03-04-01. Middle East Technical University, Ankara (in Turkish).
Amsinck, S. L., E. Jeppesen & F. Landkildehus, 2005. Inference of past changes in zooplankton community structure and planktivorous fish abundance from sedimentary subfossils – a study of a coastal lake subjected to major fish kill incidents during the past century. Archiv für Hydrobiologie 162: 363–382.
Anaç, D. & B. Ç. Esetlili, 2012. Türkiye’de Toprak Verimliliğinin Gelişimi ve Tarihçesi. Journal of Soil Science and Plant Nutrition 1: 20–22. (in Turkish).
Appleby, P. G., 2001. Chronostratigraphic techniques in recent sediments. Tracking environmental change using lake sediments. In Basin Analysis, Coring, and Chronological Techniques, Vol. 1. Kluwer Academic Publishers, Dordrecht.
Beklioglu, M. & C. O. Tan, 2008. Drought complicated restoration of a shallow Mediterranean Lake by biomanipulation. Archiv für Hydrobiologie/Fundamentals of Applied Limnology 171: 105–118.
Beklioğlu, M., Ö. İnce & İ. Tüzün, 2003. Restoration of eutrophic Lake Eymir, Turkey, by biomanipulation undertaken following a major external nutrient control I. Hydrobiologia 489: 93–105.
Beklioğlu, M., G. Altınayar & C. O. Tan, 2006. Water level control over submerged macrophyte development in five shallow lakes of Mediterranean Turkey. Archiv für Hydrobiologie 166: 535–556.
Beklioğlu, M., S. Romo, I. Kagalou, X. Quintana & E. Becares, 2007. State of the art in the functioning of shallow Mediterranean lakes: workshop conclusions. Hydrobiologia 584: 317–326.
Beklioglu, M., M. Meerhoff, M. Søndergaard & E. Jeppesen, 2011. Eutrophication and restoration of shallow lakes from a cold temperate to a warm Mediterranean and a (sub) tropical climate. In Ansari, A. A., S. Singh Gill, G. R. Lanza &W. Rast (eds), Eutrophication: Causes, consequences and control. Springer, Netherlands: 91–108.
Bennett, K., 1996. Determination of the number of zones in a biostratigraphic sequence. New Phytologist 132: 155–170.
Boll T., E. E. Levi, G. Bezirci, M. Özuluğ, Ü. N. Tavşanoğlu, A. İ. Çakıroğlu, S. Özcan, S. Brucet, E. Jeppesen & M. Beklioğlu, in revision. Fish assemblage and diversity in lakes of western and central Turkey - role of geo-climatic and other environmental variables. Hydrobiologia.
Borcard, D., P. Legendre & P. Drapeau, 1992. Partialling out the spatial component of ecological variation. Ecology 73: 1045–1055.
Bos, D. G., B. F. Cumming & J. P. Smol, 1999. Cladocera and Anostraca from the interior plateau of British Columbia, Canada, as paleolimnological indicators of salinity and lake level. Hydrobiologia 392: 129–141.
Brodersen, K. P., M. C. Whiteside & C. Lindegaard, 1998. Reconstruction of trophic state in Danish lakes using subfossil chydorid (Cladocera) assemblages. Canadian Journal of Fisheries and Aquatic Sciences 55: 1093–1103.
Brucet, S., D. Boix, S. Gascon, J. Sala, X. D. Quintana, A. Badosa, M. Søndergaard, T. L. Lauridsen & E. Jeppesen, 2009. Species richness of crustacean zooplankton and trophic structure of brackish lagoons in contrasting climate zones: north temperate Denmark and Mediterranean Catalonia (Spain). Ecography 32: 692–702.
Bucak, T., E. Saraoğlu, E. E. Levi, Ü. N. Tavşanoğlu, Aİ. Çakıroğlu, E. Jeppesen & M. Beklioğlu, 2012. The role of water level for macrophyte growth and trophic interactions in eutrophic Mediterranean shallow lakes: a mesocosm experiment with and without fish. Freshwater Biology 57: 1631–1642.
Buchaca, T., T. Skov, S. L. Amsinck, V. Gonçalves, J. M. N. Azevedo, T. J. Andersen & E. Jeppesen, 2011. Rapid ecological shift following piscivorous fish introduction to increasingly eutrophic and warmer Lake Furnas (Azores Archipelago, Portugal): a paleoecological approach. Ecosystems 14: 458–477.
Burnak, L. & M. Beklioğlu, 2000. Macrophyte dominated clear-water state of Lake Mogan. Turkish Journal of Zoology 24: 305–313.
Çakıroğlu, Aİ., Ü. N. Tavşanoğlu, E. E. Levi, T. A. Davidson, T. Bucak, A. Özen, G. K. Akyıldız, E. Jeppesen & M. Beklioğlu, 2014. Relatedness between contemporary and 123 subfossil cladoceran assemblages in Turkish lakes. Journal of Paleolimnology 52: 367–383.
Chen, G., C. Dalton & D. Taylor, 2010. Cladocera as indicators of trophic state in Irish lakes. Journal of Paleolimnology 44: 465–481.
Coops, H., M. Beklioglu & T. L. Crisman, 2003. The role of water-level fluctuations in shallow lake ecosystems – workshop conclusions. Hydrobiologia 506: 23–27.
Davidson, T. A., C. D. Sayer, M. Perrow, M. Bramm & E. Jeppesen, 2010a. The simultaneous inference of zooplanktivorous fish and macrophyte density from sub-fossil cladoceran assemblages: a multivariate regression tree approach. Freshwater Biology 55: 546–564.
Davidson, T. A., C. D. Sayer, P. Langdon, A. Burgess & M. Jackson, 2010b. Inferring past zooplanktivorous fish and macrophyte density in a shallow lake: application of a new regression tree model. Freshwater Biology 55: 584–599.
Davidson, T. A., S. L. Amsinck, O. Bennike, F. Landkildehus, T. L. Lauridsen & E. Jeppesen, 2011a. Inferring a single variable from an assemblage with multiple controls: getting into deep water with cladoceran lake-depth transfer functions. Hydrobiologia 676: 129–142.
Davidson, T. A., H. Bennion, E. Jeppesen, G. H. Clarke, C. D. Sayer, D. Morley, B. V. Odgaard, P. Rasmussen, R. Rawcliffe, J. Salgado, G. L. Simpson & S. L. Amsinck, 2011b. The role of cladocerans in tracking long-term change in shallow lake trophic status. Hydrobiologia 676: 299–315.
DeSellas, A. M., A. M. Paterson, J. M. Sweetman & J. P. Smol, 2008. Cladocera assemblages from the surface sediments of south- central Ontario (Canada) lakes and their relationships to measured environmental variables. Hydrobiologia 600: 105–119.
Dodson, S. I. & D. G. Frey, 2001. Cladocera and other Branchiopoda. In Ecology and Classification of North American Freshwater Invertebrates. Academic, San Diego: 849–913.
DSİ, 1993. Mogan Gölü limnolojik etüt raporu. Ankara: 212 pp (in Turkish).
DSİ, 2000. XVIII.Bölge Müdürlüğü 2001 yılı program bütçe toplantısı takdim raporu. Isparta (in Turkish).
Eastwood, W. J., N. J. G. Pearce, J. A. Westgate, W. T. Perkins, H. F. Lamb & N. Roberts, 1999. Geochemistry of Santorini tephra in lake sediments from Southwest Turkey. Global and Planetary Change 21: 17–29.
European climate assessment and dataset project [available on internet at http://eca.knmi.nl/], August, 2013.
Flöβner, D., 2000. Die Haplopoda und Cladocera (ohne Bosminidae) Mitteleuropas. Backhuys Publishers, Leiden.
Frey, D. G., 1959. The taxonomic and phylogenetic significance of the head pores of the Chydoridae (Cladocera). International Review of Hydrobiology 44: 27–50.
Gasiorowski, M. & K. Szeroczynska, 2004. Abrupt changes in Bosmina (Cladocera, Crustacea) assemblages during the history of the Ostrowite Lake (northern Poland). Hydrobiologia 526: 137–144.
Green, A. J., C. Fuentes, E. Moreno-Ostos & S. L. Rodrigues da Silva, 2005. Factors influencing cladoceran abundance and species richness in brackish lakes in Eastern Spain. Annales de Limnologie: International Journal of Limnology 41: 73–81.
Grimm, E. C., 1987. CONISS – a FORTRAN-77 program for stratigraphically constrained cluster-analysis by the method of incremental sum of squares. Computers and Geosciences 13: 13–35.
Hobaek, A., M. Manca & T. Andersen, 2002. Factors influencing species richness in lacustrine zooplankton. Acta Oecologica 23: 155–163.
Hofmann, W., 1987. Cladocera in space and time: analysis of lake sediments. Hydrobiologia 145: 315–321.
Hofmann, W., 1998. Cladocerans and chironomids as indicators of lake level changes in north temperate lakes. Journal of Paleolimnology 19: 55–62.
Horppila, J., A. Liljendahl-Nurminen & T. Malinen, 2004. Effects of clay turbidity and light on the predator–prey interaction between smelts and chaoborids. Canadian Journal of Fisheries and Aquatic Sciences 61(1): 862–1870.
IPCC, 2007. Climate change 2007: impacts, adaptation and vulnerability. In Parry, M. L., O. F. Canziani, J. P. Palutikof, P. J. van der Linden & C. E. Hanson (eds), Contribution of Working Group II to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge.
IPCC, 2014. Summary for policymakers. In Field, C. B., V. R. Barros, D. J. Dokken, K. J. Mach, M. D. Mastrandrea, T. E. Bilir, M. Chatterjee, K. L. Ebi, Y. O. Estrada, R. C. Genova, B. Girma, E. S. Kissel, A. N. Levy, S. MacCracken, P. R. Mastrandrea & L. L. White (eds), Climate Change 2014: Impacts, Adaptation, and Vulnerability. Part A: Global and Sectoral Aspects. Contribution of Working Group II to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge: 1–32.
Jeppesen, E., E. A. Madsen, J. P. Jensen & N. J. Anderson, 1996. Reconstructing the past density of planktivorous fish and trophic structure from sedimentary zooplankton fossils: a surface sediment calibration data set from shallow lake. Freshwater Biology 36: 115–127.
Jeppesen, E., J. P. Jensen, M. Søndergaard, T. Lauridsen, L. J. Pedersen & L. Jensen, 1997. Top-down control in freshwater lakes: the role of nutrient state, submerged macrophytes and water depth. Hydrobiologia 342(343): 151–164.
Jeppesen, E., T. L. Lauridsen, S. F. Mitchell, K. Christoffersen & C. W. Burns, 2000. Trophic structure in the pelagial of 25 shallow New Zealand lakes: changes along nutrient and fish gradients. Journal of Plankton Research 22: 951–968.
Jeppesen, E., J. P. Jensen, H. Skovgaard & C. B. Hvidt, 2001. Changes in the abundance of planktivorous fish in Lake Skanderborg during the past two centuries – a palaeoecological approach. Palaeogeography, Palaeoclimatology, Palaeoecology 172: 143–152.
Jeppesen, E., T. L. Lauridsen, S. L. Amsinck, K. Christoffersen & S. F. Mitchell, 2003. Sub-fossils of cladocerans in the surface sediments of 135 lakes as proxies for community structure of zooplankton, fish abundance and lake temperature. Hydrobiologia 491: 321–330.
Jeppesen, E., P. Noges, T. A. Davidson, J. Haberman, T. Noges, K. Blank, T. L. Lauridsen, M. Søndergaard, C. Sayer, R. Laugaste, L. S. Johansson, T. Bjerring & S. L. Amsinck, 2011. Zooplankton as indicators in lakes: a scientific-based plea for including zooplankton in the ecological quality assessment of lakes according to the European Water Framework Directive (WFD). Hydrobiologia 676: 279–297.
Jeppesen, E., S. Brucet, L. Naselli-Flores, E. Papastergiadou, K. Stefanidis, T. Noges, P. Noges, J. L. Attayde, T. Zohary, J. Coppens, T. Bucak, R. Fernandes Menezes, F. R. S. Freitas, M. Kernan, M. Søndergaard & M. Beklioğlu, 2015. Ecological impacts of global warming and water abstraction on lakes and reservoirs due to changes in water level and related changes in salinity. Hydrobiologia 750: 201–227.
Johansson, L. S., S. L. Amsinck, R. Bjerring & E. Jeppesen, 2005. Mid- to Late-Holocene land-use change and lake development at Dallund Sø, Denmark: trophic structure inferred from cladoceran subfossils. The Holocene 5: 1143–1151.
Jones, M. D., M. J. Leng, W. J. Eastwood, D. H. Keen & C. S. M. Turney, 2002. Interpreting stable-isotope records from freshwater snail-shell carbonate: a Holocene case study from Lake Gölhisar, Turkey. The Holocene 12: 629–634.
Juggins, S., 2009. Rioja: Analysis of Quaternary Science Data, R Package Version 0.5-6, http://cran.r-project.org/package=rioja, May, 2012.
Justel, A., D. Pena & R. Zamar, 1997. A multivariate Kolmogorov–Smirnov test of goodness of fit. Statistics and Probability Letters 35(3): 251–259.
Kadıoğlu, M., 1997. Trends in surface air temperature data over Turkey. International Journal of Climatology 17: 511–520.
Kattel, G. R., R. W. Battarbee, A. W. Mackay & H. J. B. Birks, 2008. Recent ecological change in remote mountain loch: an evaluation of Cladocera-based temperature transfer function. Palaeogeography, Palaeoecology, Palaeooceanography 259: 51–76.
Korhola, A. & M. Rautio, 2001. Cladocera and other branchiopod crustaceans. In Smol, J. P., H. J. B. Birks & W. M. Last (eds), Tracking Environmental Change Using Lake Sediments, Vol. 4., Zoological Indicators Kluwer Academic Publishers, Dordrecht: 173–180.
Legendre, P. & E. D. Gallagher, 2001. Ecologically meaningful transformations for ordination of species data. Oecologia 129: 271–280.
Levi, E. E., Aİ. Çakıroğlu, T. Bucak, B. V. Odgaard, T. A. Davidson, E. Jeppesen & M. Beklioğlu, 2014. Similarity between contemporary vegetation and plant remains in the sediment surface in Mediterranean lakes. Freshwater Biology 59: 724–736.
Lotter, A. F., H. J. B. Birks, W. Hofmann & A. Marchetto, 1998. Modern diatom, Cladocera, chironomid, and chrysophyte cyst assemblages as quantitative indicators for the reconstruction of past environmental conditions in the Alps. II. Nutrients. Journal of Paleolimnology 19: 443–463.
Manav, E. & S. V. Yerli, 2008. An assessment on the trophic status of Lake Mogan, Turkey. Fresenius Environmental Bulletin 17: 3–8.
Mangıt, F. & S. V. Yerli, 2010. An approach for trophic gradient in Lake Mogan (Turkey): a shallow eutrophic lake. Hacettepe Journal of Biology and Chemistry 38(1): 41–45.
Moss, B., 1998. The Ecology of Freshwaters, Man and Medium Past to Future, 3rd ed. Blackwell Science, Oxford: 557 pp.
Moss, B., D. Stephen, C. Alvarez, E. Becares, W. van de Bund, S. E. Collings, E. van Donk, E. de Eyot, T. Feldmann, C. Fernandez-Alaez, M. Fernandez-Alaez, R. J. M. Franken, F. Garcıa-Criado, E. M. Gross, M. Gyllström, L. A. Hansson, K. Irvine, A. Jarvalt, J. P. Jensen, E. Jeppesen, T. Kairesalo, R. Kornijow, T. Krause, H. Künnap, A. Laas, E. Lill, B. Lorens, H. Luup, M. R. Miracle, P. Noges, T. Noges, M. Nykanen, I. Ott, W. Peczula, E. T. H. M. Peeters, G. Phillips, S. Romo, V. Russell, J. Salujoe, M. Scheffer, K. Siewertsen, H. Smal, C. Tesch, H. Timm, L. Tuvikene, I. Tonno, T. Virro, E. Vicente & D. Wilson, 2003. The determination of ecological status in shallow lakes – a tested system (ECOFRAME) for implementation of the European Water Framework Directive. Aquatic Conservation: Marine and Freshwater Ecosystems 13: 507–549.
Nevalainen, L., 2010. Evaluation of microcrustacean (Cladocera, Chydoridae) biodiversity based on sweep net and surface sediment samples. Ecoscience 17: 356–364.
Nevalainen, L., K. Sarmaja-Korjonen & T. P. Luoto, 2011. Sedimentary Cladocera as indicators of past water level changes in shallow northern lakes. Quaternary Research 75: 430–437.
Nevalainen, L., T. P. Luoto, S. Kultti & K. Sarmaja-Korjonen, 2013. Spatio-temporal distribution of sedimentary Cladocera (Crustacea: Branchiopoda) in relation to climate. Journal of Biogeography 40: 1548–1559.
Nurminen, A. L., J. Horppila, L. Uusitalo & J. Niemisto, 2008. Spatial variability in the abundance of pelagic invertebrate predators in relation to depth and turbidity. Aquatic Ecology 42: 25–33.
ÖÇKK, 2002. Mogan Gölü Havzası Biyolojik Zenginlikleri ve Ekolojik Yönetim Planı. Çevre Bakanlığı Özel Çevre Koruma Kurumu Başkanlığı, Ankara. (in Turkish).
Oksanen, J., F. G. Blanchet, R. Kindt, P. Legendre, R. B. O’Hara, G. L. Simpson, P. Solymos, M. H. H. Stevens & H. Wagner, 2010. Vegan: Community Ecology Package. R Package Version 1.17-4, http://CRAN.R-project.org/package=vegan, May, 2012.
Özen, A., B. Karapınar, İ. Küçük, E. Jeppesen & M. Beklioğlu, 2010. Drought-induced changes in nutrient concentrations and retention in shallow Mediterranean lakes subjected to different degrees of management. Hydrobiologia 646: 61–72.
Özkan, K., E. Jeppesen, L. S. Johansson & M. Beklioğlu, 2010. The response of periphyton and submerged macrophytes to nitrogen and phosphorus loading in shallow warm lakes: a mesocosm experiment. Freshwater Biology 55: 463–465.
Papastergiadou, E., I. Kagalou, K. Stefanidis, A. Retalis & I. Leonardos, 2010. Effects of anthropogenic influences on the trophic state, land uses and aquatic vegetation in a shallow Mediterranean lake: implications for restoration. Water Resources Management 24: 415–435.
Peel, M. C., B. L. Finlayson & T. A. McMahon, 2007. Updated world map of the Köppen–Geiger climate classification. Hydrology and Earth System Sciences Discussions 4: 439–473.
Rehber, E., 1991. Alternatif Tarım Üzerine Bir Tartışma (A Review on Alternative Agriculture). Uludağ Üniversitesi Ziraat Fakültesi Dergisi No: 8: 153–160 (in Turkish).
Şekercioğlu, Ç. H., S. Anderson, E. Akçay, R. Bilgin, Ö. E. Can, G. Semiz, Ç. Tavşanoğlu, M. B. Yokeş, A. Soyumert, K. Ipekdal, I. K. Sağlam, M. Yücel & H. N. Dalfes, 2011. Turkey’s globally important biodiversity in crisis. Biological Conservation 144: 2752–2769.
Skov, T., T. Buchaca, S. L. Amsinck, F. Landkildehus, B. V. Odgaard, J. Azevedo, V. Gonçalves, P. M. Raposeiro, T. J. Andersen & E. Jeppesen, 2010. Using invertebrate remains and pigments in the sediment to infer changes in trophic structure after fish introduction in Lake Fogo: a crater lake in the Azores. Hydrobiologia 654: 13–25.
Szeroczynska, K. & K. Sarmaja-Korjonen, 2007. Atlas of Subfossil Cladocera from Central and Northern Europe. Friends of the Lower Vistula Society, Świecie: 84 pp.
Tanrıvermiş, H., 2003. Agricultural land use change and sustainable use of land resources in the Mediterranean region of Turkey. Journal of Arid Environments 54: 553–564.
Tanyolaç, J. & M. Karabatak, 1974. Mogan Gölü’nün Biyolojik ve Hidrolojik Özelliklerinin Tespiti. TÜBİTAK Proje no: VHAG-91 (in Turkish).
Tavşanoğlu, Ü. N., 2012. Zooplankton adaptation strategies against fish predation in Turkish shallow lakes. PhD Thesis.
Tavşanoğlu, Ü. N., S. Brucet, E. E. Levi, T. Bucak, G. Bezirci, A. Özen, L. S. Johansson, E. Jeppesen & M. Beklioğlu, 2015. Size-based diel migration of zooplankton in Mediterranean shallow lakes assessed from in situ experiments with artificial plants. Hydrobiologia 753: 47–59.
ter Braak, C. J. F., 1995. Ordination. In Jongman, R. H. G., C. J. F. ter Braak & O. F. R. van Tongeren (eds), Data Analysis in Community and Landscape Ecology. Cambridge University Press, Cambridge: 91–173.
ter Braak, C. J. F. & I. C. Prentice, 1988. A theory of gradient analysis. Advances in Ecological Research 18: 271–317.
Tübitak Marmara Research Center, 2013. Havza Koruma Eylem Planlarının Hazırlanması-Burdur Havzası: 466 pp (in Turkish).
Türkeş, M., U. M. Sümer & G. Kılıç, 1995. Variations and trends in annual mean air temperatures in Turkey with respect to climatic variability. International Journal of Climatology 15: 557–569.
Türkeş, M., U. M. Sümer & I. Demir, 2002. Evaluation of trends and changes in mean, maximum and minimum temperatures of Turkey for the period 1929–1999. International Journal of Climatology 22: 947–977.
Türkeş, M., T. Koç & F. Saris, 2009. Spatiotemporal variability of precipitation total series over Turkey. International Journal of Climatology 29: 1056–1074.
Turkish State Meteorological Service, 2011. http://www.meteoroloji.gov.tr/index.aspx, January, 2011.
Turkish State Meteorological Service, 2014. http://www.meteoroloji.gov.tr/index.aspx, March, 2014.
Vadeboncoeur, Y., E. Jeppesen, M. J. Vander Zanden, H. Schierup, K. Christoffersen & D. Lodge, 2003. From Greenland to green lakes: cultural eutrophication and the loss of benthic pathways in lakes. Limnology and Oceanography 48: 1408–1418.
Yerli, S. V., E. Kıvrak, H. Gürbüz, E. Manav, F. Mangıt & O. Türkecan, 2012. Phytoplankton community, nutrients and chlorophyll a in Lake Mogan (Turkey); with comparison between current and old data. Turkish Journal of Fisheries and Aquatic Sciences 12: 95–104.
Zhang, J., S. E. Jørgensen, C. O. Tan & M. Beklioğlu, 2003a. A structurally dynamic modelling – Lake Mogan, Turkey as a case study. Ecological Modelling 164: 103–120.
Zhang, J., S. E. Jørgensen, M. Beklioğlu & Ö. Ince, 2003b. Hysteresis in vegetation shift – Lake Mogan prognoses. Ecological Modelling 164: 227–238.
Acknowledgments
This study was supported by The Scientific and Technological Research Council Of Turkey (TÜBİTAK-ÇAYDAG, Projects Nos. 105Y332 and 110Y125); the Middle East Technical University (METU)-BAP Program of Turkey (BAP.07.02.2009–2012); and FP-7 REFRESH (Adaptive strategies to Mitigate the Impacts of Climate Change on European Freshwater Ecosystems, Contract No. 244121) and the MARS Project (Managing Aquatic ecosystems and water Resources under multiple Stress) funded under the 7th EU Framework Programme, Theme 6 (Environment including Climate Change)—Contract No. 603378 (http://www.mars-project.eu). AİÇ, EEL, UNT and GB were also supported by Tübitak (Project Nos. 105Y332 and 110Y125). ŞE was supported by BAP Research Grant and METU-DPT ÖYP Programme of Turkey (DPT-2011-1786). NF was supported by the TÜBİTAK-BİDEB 2211 National Research Scholarship. TAD’s contribution was supported by the CIRCE, funded by the AUFF–AU Ideas Program. EJ was further supported by the CRES (Danish Strategic Research Council), CLEAR (a Villum Kann Rasmussen Centre of Excellence Project). We thank Arda Özen, Korhan Özkan, Mukadder Arslan, Jan Coppens, Semra Yalçın, Damla Beton, and Thomas Boll Kristensen for assistance in the field and laboratory and Anne Mette Poulsen for valuable editing of the manuscript. We also thank for Jan Coppens for bathymetric maps of Lakes Eymir and Mogan.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Guest editors: M. Beklioğlu, M. Meerhoff, T. A. Davidson, K. A. Ger, K. E. Havens & B. Moss / Shallow Lakes in a Fast Changing World
Rights and permissions
About this article
Cite this article
Çakıroğlu, A.İ., Levi, E.E., Tavşanoğlu, Ü.N. et al. Inferring past environmental changes in three Turkish lakes from sub-fossil Cladocera. Hydrobiologia 778, 295–312 (2016). https://doi.org/10.1007/s10750-015-2581-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-015-2581-x