Abstract
The common grass yellow Eurema mandarina has a characteristic patch (sex brand) composed of specialized scales (androconia) and wing intermembranous cells on the ventral surface of its male forewing. This structure is specific to males and is thought to release compounds that induce female mate acceptance. However, no study has demonstrated that these compounds function as sex pheromones in the genus Eurema. Here we report the identification of sex pheromones in males of E. mandarina. Chemical analyses revealed that 6,10,14-trimethylpentadecan-2-one (TMP) and (E/Z)-3,7,11,15-tetramethylhexadec-2-enal [(E/Z)-phytal] were male-specific and abundant in particular regions of the male forewings. TMP was highest in the sex brand, whereas (E/Z)-phytal was concentrated in the anal cell (cell 2 A), lacking androconia and intermembranous cells. The content of these compounds increases with age in males after emergence. In bioassays, virgin females displayed a posture of bending their abdomens as mating acceptance in response to stimulation by fresh male forewings. However, solvent-washed male wings did not induce such female responses, suggesting that some compounds from male wings serve as triggers. When we examined female responses to compounds applied to solvent-washed male wings, authentic TMP and (E/Z)-phytal alone showed little activity. However, the mixture elicited abdomen-bending responses in one-third of the females. Therefore, TMP and (E/Z)-phytal were found to act synergistically as aphrodisiac sex pheromones for E. mandarina females, although these activities were weak.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Butterflies use multimodal signals for mate choice and sexual selection, in which visual and chemical signals play a central role (Constanzo and Monteiro 2007; Mérot et al. 2015; Papke et al. 2007; Vane-Wright and Boppré 1993). Many butterfly species have wing ornamentation such as colorful patterns, polarized iridescence, and UV reflectance, serving as long-range visual signals (Kemp and Rutowski 2011; Silberglied 1984). Moreover, several butterfly species have male-limited and specialized secretory organs called androconia on the surface of their wings and the tip of their abdomen (Kristensen and Simonsen 2003). When males approach mating partners, certain volatile compounds released from these structures function as short-range chemical signals (Boppré 1984). Males of several species possess structurally diverse and sex-specific volatiles that act as sex pheromones for females of the same species, such as pyrrolizidine alkaloid derivatives in several species of danaine and ithomine butterflies (Honda 2008; Nishida et al. 1996; Pliske 1975), oxygenated aliphatic and terpenoid compounds in Bicyclus and Heliconius spp. (Nierberding et al. 2008; Darragh et al. 2017), and terpenoid-derived cyclic lactones in Pieris spp. (Yildizhan et al. 2009).
The components, functions, and significance of visual and chemical mate signals have been intensively studied in the coliadine butterfly genera Colias and Eurema (Kemp 2006; Kemp and Rutowski 2011). Two closely related and nearly sympatric sulfur butterflies, Colias eurytheme and Colias philodice, use the presence or absence of UV coloration on male wings for mate recognition by females and reproductive isolation between species (Silberglied and Taylor 1973, 1978). In addition, their males emit sex-limited and species-specific volatiles from their wings, namely, n-hexyl esters of three aliphatic acids in C. philodice and 13-methylheptacosane in C. eurytheme (Grula et al. 1980). These volatiles are sex pheromones, and females of the same species respond to these odors by extending their abdomen ventrally from between the hindwings in a mate-acceptance posture (Grula et al. 1980). The hindwings of male C. philodice have sac-like intermembranous cells underneath the scale sockets with a basal bulge, which are thought to be scent-producing organs (Rutowski 1980). Characteristic volatiles and the same wing structures have been found in males of other sulfur butterflies (Nobre et al. 2021; Ômura and Yotsuzuka 2015).
Similar to the genus Colias, the yellow butterflies of the genus Eurema such as Eurema lisa and Eurema hecabe show a strong sexual dimorphism in the UV reflectance of their adult wings, which is a primary cue for mate discrimination by females (Kemp 2007; Rutowski 1977a). Moreover, E. lisa males have a specific patch (sex brand) on the ventral surface of the basal forewings, where specialized scales (androconia) and characteristic intermembranous cells connect to the sockets of these scales (Rutowski 1977b). In the early stages of mating behavior in E. lisa and Eurema mandarina, the male contacts or strikes the perching female with his wings, and the female responds to male buffeting by flapping her wings and performing abdomen-bending (Rutowski 1977a, 1978; Takanashi et al. 2001). This female response to male courtship, especially abdomen-bending behavior, is believed to be elicited by some volatiles emanating from male wings (Rutowski 1977a; Takanashi et al. 2001). However, active compounds (sex pheromones) have not yet been identified in the genus Eurema.
In this study, we attempted to elucidate the male sex pheromones of the Eurema butterflies. The common grass yellow E. mandarina (Lepidoptera: Pieridae) was used as research material. This species inhabits the Palearctic region from central and northern China to mainland Japan, except Hokkaido. It is a sister species of E. hecabe widely distributed in the Afrotropic, Indomalaya, and Australasia regions. Eurema mandarina also displays clear sexual dimorphism in visible and UV wing coloration (Fig. S1; Kato and Nakane 1989; Kemp 2007). Male adults have a light-gray sex brand along the cubitus (Cu) vein on the ventral surface of the forewing, extending near the origin of vein Cu2 (Fig. S2; Jeratthitikul et al. 2009; Khan and Sahito 2012). This study aimed to examine (1) the chemical structure of male sex pheromone of E. mandarina, (2) its quantitative change with male aging, and (3) its distribution on the insect body.
Methods and materials
Insects
Eureme mandarina adults used for the experiments were summer morph individuals collected in the field or reared in the laboratory. Wild individuals were captured in Higashihirohisma city (34.4 °N, 132.7 °E) from 2015 to 2021. Gravid females were allowed to lay eggs on host plants, Alibizia julibrissin and Lespedeza cuneata (Fabaceae). Newly hatched larvae were reared on fresh leaves at 25 °C under an LD 16:8 h photocycle in the laboratory. Emerged adults were used as laboratory-reared individuals without being allowed to mate. Several field-collected individuals were also used for chemical analyses, although their ages after emergence and mating experience were unknown. All adults were fed 15% sucrose solution daily and kept individually in transparent plastic containers (9 cm height, 12 cm diameter).
Extraction of volatiles from adult butterflies
Samples of volatiles from adult butterflies were prepared using conventional solvent extraction method. Adult butterflies were anesthetized by chilling to 4 °C and frozen to death at -20 °C. The following four types of samplings were conducted.
(1) To identify male-specific compounds, whole bodies of wild-caught butterflies were extracted individually. The study sample consisted of 19 males and 12 females.
(2) To reveal quantitative changes in male-specific compounds with male aging, whole bodies of laboratory-reared males were extracted individually on the day of emergence (day 0), three days after emergence (day 3), and five days after emergence (day 5). Five males were used at each time point.
(3) To examine the distribution of male-specific volatiles on the insect body, a field-collected male was divided into five parts: anterior forewings, posterior forewings (including sex brands), anterior hindwings, posterior hindwings, and body (see plate A in Fig. 5). Each part was extracted individually. The sampling was replicated for nine males.
(4) To determine the wing regions where male-specific compounds were concentrated, the posterior of two forewings, including a sex brand, were detached from a 3-day-old laboratory-reared male and divided into three parts: sex brand, anal cell (cell 2 A), and the remainder (see plate A in Fig. 6). When the sex brand was set as a unit area of one, the area of the anal cell was 17, and that of the remainder was 48. Moreover, the anal cells of the two forewings were separated from a 3-day-old laboratory-reared male and divided into three parts: basal third, middle third, and marginal third (see plate B in Fig. 6). The basal:middle:marginal area ratio was 4:6:7. Each part obtained from a single male individual was extracted separately. The sampling was replicated for five males.
Each individual’s whole body and divided parts were submerged in 1 ml purified (twice distilled) dichloromethane for 3 min. Subsequently, the crude extract was filtered to remove the body and scales and then concentrated to 100 µl under a gentle nitrogen stream at 20 °C. All extracts were stored at -20 °C until use.
Headspace sampling of volatiles from live males
To determine whether male-specific volatiles were emitted from the insect body, static headspace sampling was conducted using a Supelco solid-phase microextraction (SPME) syringe with a 100-mm polydimethylsiloxane fiber (Sigma-Aldrich, St. Louis, MO, USA). Five live wild-caught males were placed in a glass vessel (10 cm height, 6 cm diameter) without disturbance, and headspace volatiles were collected with SPME fiber for 20 h at 20 °C.
Chemical analyses
The concentrated extracts of E. mandarina and the headspace samples of live males were subjected to gas chromatography-electron impact mass spectrometry (GC-EIMS) and gas chromatography-high-resolution-time-of-flight mass spectrometry (GC-HR-TOFMS). GC-EIMS was carried out at an EI voltage of 70 eV using a Shimadzu QP5000 mass spectrometer and a Shimadzu GC-17 A gas chromatograph (Shimadzu, Kyoto, Japan) equipped with an Agilent J&W DB-1 capillary column (15 m × 0.25 mm ID, 0.25 μm film thickness: Santa Clara, CA, USA) or an Agilent J&W HP-5MS capillary column (30 m × 0.25 mm ID, 0.25 μm film thickness: Santa Clara, CA, USA). GC-HR-TOFMS was carried out at a field ionization voltage of -10 kV using a JEOL JMS-T100GCV time-of-flight mass spectrometer (JEOL, Akishima, Japan) equipped with a Phenomenex Zebron ZB-1MS column (30 m × 0.25 mm, 0.25 μm film thickness: Torrance, CA, USA). The splitless injection of 1 µl extract sample or the SPME fiber was operated with an injector temperature of 250 °C and a split opening 30 s after injection. The oven temperature was programmed from 50 °C (initial 1 min hold) to 300 °C (final 10 min hold) at 10 °C/min, or from 40 °C (initial 2 min hold) to 230 °C (final 5 min hold) at 10 °C/min. The linear retention index of the detected compound was calculated from the retention times of authentic n-alkanes under the same analytical conditions. Compounds were identified by comparing their retention times and mass spectra with those of authentic chemicals. The compounds were semi-quantified by comparing their base peak intensity with that of authentic n-pentacosane (Tokyo Chemical Industry, Tokyo, Japan) as an external standard.
Authentic chemicals
6,10,14-Trimethylpentadecan-2-one (synonym: hexahydrofarnesyl acetone or phytone; abbreviation: TMP) was synthesized by oxidation of (2E/Z,7R,11R)-phytol (Tokyo Chemical Industry, Tokyo, Japan) with sodium periodate in acetonitrile (Nieberding et al. 2008). The synthesized TMP had a (6R,10R)-configuration. (E/Z)-3,7,11,15-Tetramethylhexadec-2-enal [(E/Z)-phytal] was prepared by oxidation of the same authentic phytol with pyridinium dichromate in dry diethyl ether (Fürstenau et al. 2013). The synthesized (E/Z)-phytal had a (7R,11R)-configuration. After purification by silica-gel column chromatography, GC-MS analyses revealed that the purity of these products was over 97% and the E/Z ratio of the phytal was approximately 8.6/1.
Butterfly models
To examine whether male-specific volatiles induce behavioral responses in E. mandarina females, several butterfly models were prepared using 3-day-old to 5-day-old adults reared in the laboratory.
First, intact butterfly specimens were used as whole-body models. The ‘fresh’ models were male or female specimens that retained natural compounds and were prepared by freeze-killing at -20 °C. The ‘washed’ models were male or female specimens where almost all volatile and epicuticular components were removed by soaking the ‘fresh’ models five times with 1 ml dichloromethane for 5 min (Fig. S3). All models had opened wings, and an insect pin piercing the thorax.
Second, wings detached from one male were used to prepare three types of wing models: whole wings, wings from which the posterior forewings containing sex brands were removed, and the posterior forewings containing sex brands (Fig. S4). The wings were used as untreated ‘fresh’ or solvent-treated ‘washed’ as mentioned above and fixed in an open position by attaching the detached points with polyvinyl acetate glue. A wooden clip was used to pinch the glued point of the model, and a 10 cm vinyl strip attached to the clip was used as a handle (plate E in Fig. S4).
Bioassays
We examined whether virgin females exhibited an abdomen-bending posture as a mating acceptance in response to the butterfly, to the wing models, and to the samples applied to the models. Bioassays were performed under a mosquito net (75 × 105 cm, 90 cm height) in a greenhouse at Hiroshima University between 10:00 and 14:00 on sunny days within a temperature range of 26–31 °C. Within 2–7 days after emergence, the posterior margins of the hindwings of virgin females were removed to make the observation of abdomen-bending postures easier (Fig. S5). One female and one mature male were introduced into the net. When the male approached and started copulation attempts toward the female, the butterflies were trapped in a transparent cylindrical cup (9 cm height, 12 cm diameter), and the female was subjected to male courtship for approximately 30 s. Females that showed abdomen bending were subjected to the following three experiments, whereas females that showed no response were excluded.
(1) We examined female responses to the whole-body models of fresh males, fresh females, and washed males. In addition, the washed male models were treated with a concentrated crude extract from one male or one female butterfly and used for bioassays after the solvent had volatilized.
(2) Male wing models were used to examine female responses and to estimate wing regions where possible pheromone compounds may be present in high abundance. Bioassays were performed on five different models: fresh whole wings, washed whole wings, fresh wings excluding posterior forewing parts, fresh posterior forewing parts, and washed posterior forewing parts.
(3) The effects of male-specific compounds on female abdomen-bending were assessed using the washed model of whole male wings as a surrogate for chemical exposure. Dichloromethane solutions were produced from TMP, (E/Z)-phytal, and a mixture of the two. By applying 100 µl of each solution, the models contained 0.5 µg TMP, 10 µg (E/Z)-phytal, or 0.5 µg TMP and 10 µg (E/Z)-phytal, respectively. The ratio of TMP to (E/Z)-phytal was similar to that found in laboratory-reared males five days after emergence. TMP and (E/Z)-phytal amounts were approximately 6–15 times greater than the median values detected in 5-day-old male after emergence. A model treated with 100 µl of dichloromethane was used as a control. These models were used after the solvent had volatilized.
All models were prepared immediately prior to the experiment and tested in random order. When tested with multiple models, an interval of 2 min was allowed between the model exposures. After capturing and removing live males from the mosquito net, we picked up each test model with an insect pin or vinyl strip and held it close to the head or body side of the female. Subsequently, as in previous studies (Rutowski 1977b; Takanashi et al. 2001), we stimulated the female by rubbing the model on her antennae and body for 1 min and examined whether the female displayed an abdomen-bending posture. Females were tested once for each model. Experiments for each model were replicated using 16–25 different females and the percentage of the responding females was calculated.
Morphological observation
Forewings dissected from male and female butterflies were subjected to optical microscopy using an Olympus SZX-7 binocular microscope (Olympus Corp., Tokyo, Japan) and a Keyence BZ-9000 fluorescent microscope (Keyence, Osaka, Japan) under bright conditions and in digital Z-stacking mode. To examine the wing intermembrane structures, wing scales were gently removed from both the dorsal and ventral surfaces of the wings using a pair of precise forceps.
Statistical analyses
All statistical analyses were conducted using R version 3.6.1 (R Development Core Team 2019). Chemical analysis data were analyzed by Mann-Whitney U test and Steel-Dwass nonparametric pairwise comparisons using the R source code ‘Steel.Dwass’ (Aoki 2004). Nonparametric pairwise comparisons based on Fisher’s exact test were performed for the bioassay data using the ‘fisher.multcomp’ function in the R package ‘RVAideMemoire’ (Hervé 2019).
Results
Female responses to whole-body models
Virgin females of E. mandarina displayed abdomen-bending in response to the fresh whole-body model of male, but no response toward that of female (Table 1). The washed whole-body model of male, which contained few volatile and epicuticular components, elicited significantly fewer responses from females than the fresh male model. When the washed male model was treated with the extract from a male, females showed weak responses, which were not significantly different from the responses to the fresh male model. In contrast, the washed model treated with the female extract was significantly less efficient than the fresh male model in eliciting female responses. These results suggest that several male-derived compounds promote mate acceptance in females.
Identification of male-specific compounds
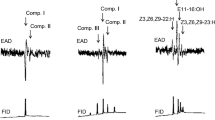
Although most of the components were shared by males and female butterflies, three compounds were identified as male-specific (peaks 1–3 in Fig. 1). These compounds were also detected in small amounts in the headspace sample from live males (Fig. 2) and were, therefore, determined to be volatiles emitted from males.
Typical total ion chromatograms obtained from crude dichloromethane extracts of male and female Euremamandarina (A) and enlarged views up to 25 min of retention time (B). Chromatograms were run on an Agilent J&W DB-1 capillary column (15 m × 0.25 mm ID), programmed from 50 ºC (initial 1 min hold) to 300 ºC (final 20 min hold) at a temperature increase of 10 ºC/min. Peaks 1, 2, and 3 correspond to 6,10,14-trimethylpentadecan-2-one, (Z)-3,7,11,15-tetramethylhexadec-2-enal, and (E)-3,7,11,15-tetramethylhexadec-2-enal, respectively
Total ion chromatograms obtained from crude dichloromethane extract (upper trace) and headspace SPME sample (lower trace) of male Euremamandarina with a 15–25 min retention time. Chromatograms were run on an Agilent J&W DB-1 capillary column (15 m × 0.25 mm ID), programmed from 40 ºC (initial 2 min hold) to 230 ºC (final 5 min hold) at a temperature increase of 10 ºC/min. Peaks 1, 2, and 3 correspond to 6,10,14-trimethylpentadecan-2-one, (Z)-3,7,11,15-tetramethylhexadec-2-enal, and (E)-3,7,11,15-tetramethylhexadec-2-enal, respectively
The EI mass spectrum of compound 1 exhibited a base peak at m/z 43 and diagnostic ions at m/z 250 and 58 with a retention index of 1826 on the DB-1 column (plate A in Fig. S6). The HR-MS results indicated a molecular ion [M]+ at m/z 268.27553 (calculated: 268.27661), corresponding to the molecular formula C18H36O. This compound was identified as TMP by comparing the mass spectrum and retention index with those of an authentic sample. Using the HP-5MS column, the retention index of this compound was 1850, the same as that previously reported for TMP (Sasaerila et al. 2003; Yildizhan et al. 2009). The field-collected males contained significantly more TMP than the field-collected females (Mann-Whitney U test, P < 0.001), and the median amount per individual was 130 ng for males and 13.5 ng for females (plate A in Fig. 3).
Contents of 6,10,14-trimethylpentadecan-2-one (A), (Z)-3,7,11,15-tetramethylhexadec-2-enal (B), and (E)-3,7,11,15-tetramethylhexadec-2-enal (C) per individual of Eurema mandarina. The sample sizes were 19 males and 12 females collected in the field. Box-and whisker plots show 10th, 25th, 50th (median), 75th, and 90th percentiles. Cross marks denote the average values. Asterisks indicate significant differences between males and females in the content of each compound (Mann-Whitney U test: P < 0.001)
Compounds 2 and 3 displayed similar EI mass spectra: the spectrum of compound 3 consisted of a base peak at m/z 84 and diagnostic ions at m/z 263 and 41 (plates B and C in Fig. S6). GC-HR-TOFMS revealed that the molecular ions [M]+ of compounds 2 and 3 were at m/z 294.29121 and 294.29129, respectively, indicating that these molecules had a molecular formula of C20H38O (calculated: 294.29226). (E/Z)-Phytal gave two peaks with similar EI-MS spectra at retention indices of 2087 and 2120 on the DB-1 column. The calculated retention index of 2153 for compound 3 on the HP-5MS column was identical to that previously reported for (E)-phytal (Yildizhan et al. 2009). Therefore, compounds 2 and 3 were identified as the Z and E isomers of phytal, respectively. Field-collected males contained significantly more (E/Z)-phytal than field-collected females (Mann-Whitney U test: P < 0.001). The median amounts of (Z)-phytal were 100 ng per male and 0 ng per female (plate B in Fig. 3), while those of (E)-phytal were 411 ng per male and 13.5 ng per female (plate C in Fig. 3).
Quantitative changes in male-specific compounds with male aging
These three compounds were barely detected in laboratory-reared males on the day of emergence (day 0 in Fig. 4). However, the content per male increased significantly with the number of days after emergence (Steel-Dwass test: P < 0.05), and the median values of TMP, (Z)-phytal, and (E)-phytal reached 33, 127, and 1552 ng, respectively, on the fifth day after emergence (day 5 in Fig. 4).
Age-dependent changes in the contents of 6,10,14-trimethylpentadecan-2-one (A), (Z)-3,7,11,15-tetramethylhexadec-2-enal (B), and (E)-3,7,11,15-tetramethylhexadec-2-enal (C) per Eurema mandarina male. The sample sizes were 5 laboratory-reared males each at 0, 3, and 5 days after emergence. Box-and whisker plots show 10th, 25th, 50th (median), 75th, and 90th percentiles. Cross marks denote the average values. Different letters indicate significant differences between male ages (Steel-Dwass test: P < 0.05)
Distribution of male-specific compounds on the insect body
Among the five divided parts of field-collected males, the posterior forewings contained a significantly larger amount of TMP than the other parts (plate B in Fig. 5). Although not statistically significant, (Z)- and (E)-phytal tended to be concentrated in the posterior forewings (plates C and D in Fig. 5). These results demonstrated that organs producing these compounds might be localized to the posterior parts of the male forewings, from which the compounds might be transferred to other parts by rubbing the wings.
Five-part segmentation of one Euremamandarina male (A) and the contents of 6,10,14-trimethylpentadecan-2-one (B), (Z)-3,7,11,15-tetramethylhexadec-2-enal (C), and (E)-3,7,11,15-tetramethylhexadec-2-enal (D) in each part. The sample sizes were 9 wild-caught males. Divided male parts are anterior forewing (FA), posterior forewing including a sex brand (FP), anterior hindwing (HA), posterior hindwing (HP), and body (B) (plate A). Box-and whisker plots show 10th, 25th, 50th (median), 75th, and 90th percentiles. Cross marks denote the average values. Different letters indicate significant differences between 5 divided parts (Steel-Dwass test: P < 0.05)
Wing regions where male-specific compounds are concentrated
Because the absolute amounts of compounds varied greatly among males and wing parts, the relative abundance of each compound in the wing parts was calculated as a percentage for each male. Based on the average relative abundance and area of each wing part, we estimated the male wing regions where the compounds were concentrated (Fig. 6). In the three divided parts of the male forewing posterior half, 42% of TMP was present in the sex brand, 34% in the anal cell, and 24% in the remainder (plate A in Fig. 6). However, TMP was estimated to be the most concentrated in the sex brand because the sex brand was greatly smaller than the other two parts. In contrast, (E)- and (Z)-phytal were absent in the sex brand, and 90% of the detected phytal was in the anal cell (plate A in Fig. 6). In the three divided parts of the anal cell, each compound showed 25–39% of the relative abundance (plate B in Fig. 6). Considering the area ratio of the three parts, (E/Z)-phytal tended to be relatively abundant in the basal third.
Segmentation of the posterior forewing (A) and anal cell (B) of one Eurema mandarina male and average relative abundance of male-specific compounds in each part. The sample sizes were 5 laboratory-reared males at 3 days after emergence. Divided parts of the posterior forewing are sex brand (SB), anal cell (AC), and the remainder (R) (plate A), while those of the anal cell are basal third (AC1), middle third (AC2), and marginal third (AC3) (plate B). Male-specific compounds are 6,10,14-trimethylpentadecan-2-one (TMP), (Z)-3,7,11,15-tetramethylhexadec-2-enal [(Z)-Phytal], and (E)-3,7,11,15-tetramethylhexadec-2-enal [(E)-Phytal]
Female responses to male wing models
The fresh model of whole male wings evoked abdomen-bending responses in 91% of the virgin females tested (Table 2). In contrast, females did not respond to the washed model of whole male wings, suggesting that sex pheromones that encourage females to mate are contained in male wings. In comparison with the whole fresh male wings, the activity induced by the posterior half of the fresh male forewings was almost the same. However, removal of the posterior half of the forewings significantly decreased the female responses to the fresh male wings (pairwise comparison with Fisher’s exact test: P < 0.05). This suggests that the active compounds are localized in the posterior half of the male forewings.
Effects of male-specific compounds on female abdomen-bending
When the models were treated with 0.5 µg TMP and 10 µg (E/Z)-phytal individually, 8% and 16% of the females responded, respectively (Table 3). However, the percentage of responding females was not significantly different between the model treated with each compound and the control model. In contrast, the mixture of 0.5 µg TMP and 10 µg (E/Z)-phytal elicited responses in 36% of the tested females and significantly increased the percentage of responding females compared to the control (pairwise comparison with Fisher’s exact test: P < 0.05).
Alar organs producing male-specific compounds
The sex brand on the male forewing, where TMP was relatively abundant, was covered by specialized scales (plate B in Fig. S2). Optical microscopy of the scale-removed wings revealed saccular structures within the wing membrane at the position of the sex brand (plates A-C in Fig. 7). This intermembranous structure was absent in the female forewings (plates D-E in Fig. 7). In contrast, sex-specific structures, such as specialized scales and intermembranous structures, were not found in the anal cells of male forewings, where (E/Z)-phytal was concentrated (Figs. S1, S2, and S7).
Optical micrographs of ventral area around the vein Cu on the forewing of Eurema mandarina butterflies. Male (A-C) and female (D-E) wing surfaces with scales removed. Plate C was an enlarged view within the dashed frame in plate B. In plate C, the location of intermembranous cells was indicated by black arrows
Discussion
This study revealed that E. mandarina conatins male-specific oxygenated terpenoids, TMP, and (E/Z)-phytal. Washing male wings with solvent prevented females from exhibiting abdomen-bending responses. When applied to the washed models, TMP or (E/Z)-phytal alone caused little abdomen-bending in females, whereas the mixture induced weak responses. Therefore, these compounds act synergistically as aphrodisiac sex pheromones that induce female mate acceptance. Small amounts of these compounds were emitted from the male insect body. However, females did not exhibit abdomen-bending unless approached and touched by fresh male wings and treated models. These findings agree with the results of previous studies on other Eurema and Colias species (Grula et al. 1980; Rutowski 1977b, 1980; Takanashi et al. 2001), suggesting that TMP and (E/Z)-phytal are short-range olfactory or contact chemical signals for E. mandarina females.
TMP has been identified in several lepidopteran species (Bacquet et al. 2014; Sasaerila et al. 2003; Schulz and Nishida 1996; Schulz et al. 1993). The white butterflies Pieris rapae and Pieris brassicae contain a larger amount of TMP in male wings than in female wings (Yildizhan et al. 2009). Male butterflies of nine Bicyclus species and Heliconius eleuchia have TMP as their alar androconial substance (Darragh et al. 2020; Hedenström et al. 2015). However, to the best of our knowledge, the use of TMP as a sex pheromone by lepidopteran insects has been revealed in the two Pieris butterflies and the bumblebee moth, Aphomia sociella (Pyralidae) (Wallin et al. 2020; Yildizhan et al. 2009). This compound acts as a contact sex pheromone in the bruchine beetle Callosobruchus rhodesianus (Coleoptera: Chrysomelidae) (Shimomura et al. 2016), a phagostimulant of Bermuda grass for larvae of Spodoptera frugiperda (Noctuidae) (Mohamed et al. 1992), a sex pheromone component collected by male euglossine bees from orchid flowers (Eltz et al. 2010), and a key volatile in nursery pollination between Greya moths (Prodoxidae) and Lithophragma plants (Saxifragaceae) (Schiestl et al. 2021).
Although pheromonal activity has not been determined, phytal has been reported as a wing substance in several butterflies, such as P. rapae males, P. brassicae males and females, and two Bicyclus males (Bacquet et al. 2014; Yildizhan et al. 2009). The Moroccan locust Dociostaurus maroccanus males use (E)-phytal as the most active sex pheromone (Fürstenau et al. 2013; Guerrero et al. 2019).
TMP and (E/Z)-phytal were absent or present in trace amounts in newly emerged males of E. mandarina but increased with age. Summer-morph males of E. mandarina do not perform courtship on the first day after emergence and start mating behavior after the third day when they become sexually mature (Kato 1989). The quantitative changes in TMP and (E/Z)-phytal coincided well with the changes in courtship activity in males, suggesting that both compounds may play a role in transmitting the sexual maturity of males to mating partners. Males of the African satyrid butterfly Bicyclus anynana have a sex pheromone consisting of (Z)-9-tetradecenol, hexadecanal, and 6,10,14-trimethylpentadecan-2-ol, the corresponding alcohol of TMP (Nieberding et al. 2008). The relative amount of each component changes with the age of the male after emergence. The pheromone composition indicates the sexual maturity of males and is used for sexual selection by females (Nieberding et al. 2008, 2012). It appears that the higher TMP and (E/Z)-phytal levels on the third days after emergence may be required for E. mandarina males to facilitate female mate acceptance. The dose-response curve of these pheromonal activities needs to be further investigated.
Phytophagous insects ingest chlorophyll contained in plants, which is broken down during digestion to form (E)-phytol (Schulz et al. 2011). Pieris brassicae and D. maroccanus synthesize TMP and (E)-phytal, respectively, from the oxidation of phytol (Ahlquist et al. 1978; Fürstenau et al. 2013; Schulz et al. 2011). Wild males of Bicyclus butterflies exclusively possess the (6R,10R)-isomer of TMP and the (2R,6R,10R)-isomer of 6,10,14-trimethylpentadecan-2-ol (Hedenström et al. 2015). Therefore, male E. mandarina is highly likely to produce (6R,10R)-TMP and (7R,11R)-phytal from plant-derived phytol. Moreover, phytol is likely to serve as a male sex pheromone in the two Pieris butterflies (Yildizhan et al. 2009). Interestingly, several phytophagous insects use phytol derivatives as sex pheromones. In the future, it will be necessary to determine the absolute configuration of TMP and (E/Z)-phytal in E. mandarina and examine whether these enantiomers have different pheromone activities.
The sex brand of male E. mandarina consists of specialized scales and distinct intermembranous cells just below the sockets of these scales. Similar intermembranous cells have been reported in the male wings of several pierid butterflies in the genera Eurema, Colias, and Anthocharis, and are thought to be involved in the emission of sex pheromones (Okumura et al. 2016; Ômura and Yotsuzuka 2015; Rutowski 1977b, 1980; Vetter and Rutowski 1978). In contrast, no distinctive structures were found in male forewing anal cells of E. mandarina. It is possible that the hindwing anterior region, where the second-largest amount of (E/Z)-phytal was detected, contains concealed secretory organs from which the released (E/Z)-phytal can be rapidly transferred to the friction surface, the anal cell, in the posterior forewing region. Males of several Eurema butterflies, such as Eurema laeta, have a sex brand in the basal area of cell Sc + R1 on the dorsal surface of the hindwing (Jeratthitikul et al. 2009).
While 86% of the females displayed abdomen-bending to the posterior part of fresh male forewings, only 36% of the females responded to the model treated with a mixture of TMP and (E/Z)-phytal. This result implies that there are other sex pheromone substances in male wings besides TMP and (E/Z)-phytal and that cuticular hydrocarbons may be candidates. Several Papilio swallowtail butterflies have distinct species specificity and sexual dimorphism in their cuticular hydrocarbon composition (Ômura et al. 2021, 2022). Males of the common Mormon swallowtail Papilio polytes use particular alkene components to distinguish mating partner females from homologous males and heterologous adults (Ômura et al. 2020). The sex pheromone titer of 13-methylheptacosane, a male-specific compound of C. eurytheme, varies with the ratio of two cuticular hydrocarbons, n-heptacosane and n-nonacosane, to this compound (Sappington and Taylor 1990a,b). Further research is needed to investigate sex differences in cuticular hydrocarbons in E. mandarina and their functions in mating behavior.
Data Availability
All the data used in the present genetic analyses are included in this published article and its supplementary information.
References
Ahlquist L, Bergström G, Liljenberg C (1978) Acyclic diterpene alcohols: occurrence and synthesis of geranylcitronellol, phytol and geranylgeraniol. Prog Chem Fats other Lipids 16:231–255
Aoki S (2004) Steel-Dwass nonparametric multiple comparison test (in Japanese). https://aoki2.si.gunma-u.ac.jp/R/Steel-Dwass.html Accessed 5 Mar 2021
Bacquet PMB, Brattström O, Wang H-L, Allen CE, Löfstedt C, Brakefield PM, Nieberding CM (2014) Selection on male sex pheromone composition contributes to butterfly reproductive isolation. Proc R Soc B 282:20142734
Boppré M (1984) Chemically mediated interactions between butterflies. In: Vane-Wright RI, Ackery PR (eds) The biology of butterflies. Academic Press, London, pp 259–275
Constanzo K, Monteiro A (2007) The use of chemical and visual cues in female choice in the butterfly Bicyclus anynana. Proc R Soc B 274:845–851
Darragh K, Vanjari S, Mann F, Gonzalez-Rojas MF, Morrison CR, Salazar C, Pardo-Diaz C, Merrill RM, McMillan WO, Schulz S, Jiggins CD (2017) Male sex pheromone components in Heliconius butterflies released by the androconia affect female choice. PeerJ 5:e3953
Darragh K, Montejo-Kovacevich G, Kozak KM, Morrison CR, Figueiredo CME, Ready JS, Salazar C, Linares M, Byers KJRP, Merrill RM, McMillan WO, Schulz S, Jiggins CD (2020) Species specificity and intraspecific variation in the chemical profiles of Heliconius butterflies across a large geographic range. Ecol Evol 10:3895–3918
Eltz T, Hedenström E, Bång J, Wallin EA, Andersson J (2010) (6R,10R)-6,10,14-Trimethylpentadecan-2-one, a dominant and behaviorally active component in male orchid bee fragrances. J Chem Ecol 36:1322–1326
Fürstenau B, Muñoz L, Coca-Abia M, Rosell G, Guerrero A, Quero C (2013) Phytal: a candidate sex pheromone component of the Moroccan locust Dociostaurus maroccanus. ChemBioChem 14:450–1459
Grula JW, McChesney JD, Taylor OR Jr (1980) Aphrodisiac pheromones of the sulfur butterflies Colias eurytheme and C. philodice (Lepidoptera, Pieridae). J Chem Ecol 6:241–256
Guerrero A, Ramos VE, López S, Alvarez JM, Domínguez A, Coca-Abia M, Bosch MP, Quero C (2019) Enantioselective synthesis and activity of all diastereoisomers of (E)-phytal, a pheromone component of the Moroccan locust, Dociostaurus maroccanus. J Agric Food Chem 67:72–80
Hedenström E, Wallin EA, Andersson J, Bång J, Wang H-L, Löfstedt C, Brattström O, Baquet P (2015) Stereoisomeric analysis of 6,10,14-trimethylpentadecan-2-ol and the corresponding ketone in wing extracts from African Bicyclus butterfly species. J Chem Ecol 51:44–51
Hervé M (2019) RVAideMemoire: Testing and Plotting Procedures for Biostatistics. R package version 0:9–73
Honda (2008) Addiction to pyrrolizidine alkaloids in male danaine butterflies: a quest for the evolutionary origin of pharmacophagy. In: Maes RP (ed) Insect Physiology: New Research. Nova Science Publishers, New York, pp 73–118
Jeratthitikul E, Lewvanich A, Butcher BA, Lekprayoon C (2009) A taxonomic study of the genus Eurema Hübner, [1819] (Lepidoptera: Pieridae) in Thailand. Nat Hist J Chulalongkorn Univ 9:1–20
Kato Y (1989) Differences in reproductive behavior among seasonal wing morphs of the butterfly Eurema hecabe. J Insect Behav 2:419–429
Kato Y, Nakane T (1989) Male approach to pupae in the yellow butterfly, Eurema hecabe. J Ethol 7:59–61
Kemp DJ (2006) Ultraviolet ornamentation and male mating success in a high-density assemblage of the butterfly Colias eurytheme. J Insect Behav 19:669–684
Kemp DJ (2007) Female mating biases for bright ultraviolet iridescence in the butterfly Eurema hecabe (Pieridae). Behav Ecol 19:1–8
Kemp DJ, Rutowski RL (2011) The role of coloration in mate choice and sexual interactions in butterflies. Adv Study Behav 43:55–92
Khan D, Sahito ZA (2012) Eurema hecabe (Linnaeus, 1758) [Lepidoptera: Pieridae], a serious seedling pest of Acacia stenophylla A. Cunn. Ex. Benth., in Karachi. Int J Biol Biotech 9:307–312
Kristensen NP, Simonsen TJ (2003) ‘Hairs’ and scales. In: Kristensen NP (ed) Lepidoptera, moths and butterflies. Volume 2: Morphology, physiology, and development. Walter de Gruyter, Berlin, pp 9–22
Mérot C, Frérot B, Leppik E, Joron M (2015) Beyond magic traits: Multimodal mating cues in Heliconius butterflies. Evolution 69:2891–2904
Mohamed MA, Quisenberry SS, Moellenbeck DJ (1992) 6,10,14-Trimethylpentadecan-2-one: a Bermuda grass phagostimulant to fall armyworm (lepidoptera: Noctuidae). J Chem Ecol 18:673–682
Nieberding CM, de Vos H, Schneider MV, Lassance J-M, Estramil N, Andersson J, Bång J, Hedenström E, Löfstedt C, Brakefield PM (2008) The male sex pheromone of the butterfly Bicyclus anynana: towards an evolutionary analysis. PLoS ONE 3:e2751
Nieberding CM, Fischer K, Saastamoinen M, Allen CE, Wallin EA, Hedenström E, Brakefield PM (2012) Cracking the olfactory code of a butterfly: the scent of ageing. Ecol Lett 15:415–424
Nishida R, Schulz S, Kim CS, Fukami H, Kuwahara Y, Honda K, Hayashi N (1996) Male sex pheromone of a giant danaine butterfly, Idea leuconoe. J Chem Ecol 22:949–972
Nobre CEB, Lucas LAS, Padilha RJR, Navarro DMAF, Alves LC, Maia ACD (2021) Specialized androconial scales conceal species-specific semiochemicals of sympatric sulphur butterflies (Lepidoptera: Pieridae: Coliadinae). Org Divers Evol 21:93–105
Okumura Y, Ozeki Y, Itoh T, Ohta S, Ômura H (2016) Volatile terpenoids from male wings lacking scent scales in Anthocharis scolymus (Lepidoptera: Pieridae). Appl Entomol Zool 51:385–392
Ômura H, Yotsuzuka S (2015) Male-specific epicuticular compounds of the sulfur butterfly Colias erate poliographus (Lepidoptera: Pieridae). Appl Entomol Zool 50:191–199
Ômura H, Noguchi T, Ohta S (2020) The male swallowtail butterfly, Papilio polytes, uses cuticular hydrocarbons for mate discrimination. Anim Behav 170:133–145
Ômura H, Morozumi Y, Noguchi T, Ohta S (2021) Variation in cuticular lipid profiles of black butterflies of the genus Papilio in Japan. Biochem Syst Ecol 96:104265
Ômura H, Noguchi T, Ohta S (2022) Chemical identity of cuticula lipid components in the mimetic swallowtail butterfly Papilio polytes. Chem Biodivers 19:e202100879
Papke RS, Kemp DJ, Rutowski RL (2007) Multimodal signalling: structural ultraviolet reflectance predicts male mating success better than pheromones in the butterfly Colias eurytheme L. (Pieridae). Anim Behav 73:47–54
Pliske TE (1975) Courtship behavior and use of chemical communication by males of certain species of ithomiine butterflies (Nymphalidae: Lepidoptera). Ann Entomol Soc Am 68:935–942
R Development Core Team (2019) R: A language and environment for statistical computing. Vienaa, Austria: R Foundation for Statistical Computing. https://www.R-project.org
Rutowski RL (1977a) The use of visual cues in sexual and species discrimination by males of the small sulphur butterfly Eurema lisa (Lepidoptera, Pieridae). J Comp Physiol A 115:61–74
Rutowski RL (1977b) Chemical communication in the courtship of the small sulphur butterfly Eurema lisa (Lepidoptera, Pieridae). J Comp Physiol A 115:75–85
Rutowski RL (1978) The courtship behaviour of the small sulphur butterfly, Eurema lisa (Lepidoptera: Pieridae). Anim Behav 26:892–903
Rutowski RL (1980) Male scent-producing structures in Colias butterflies–function, localization, and adaptive features. J Chem Ecol 6:13–26
Sappington TW, Taylor OR (1990a) Developmental and environmental sources of pheromone variation in Colias eurytheme butterflies. J Chem Ecol 16:2771–2786
Sappington TW, Taylor OR (1990b) Disruptive sexual selection in Colias eurytheme butterflies. Proc Natl Acad USA 87:6132–6135
Sasaerila Y, Gries R, Gries G, Khaskin G, King S, Takács S, Hardi(2003) Sex pheromone components of male Tirathaba mundella (Lepidoptera: Pyralidae). Chemoecology 13:89–93
Schiestl FP, Wallin EA, Beck JJ, Friberg M, Thompson JN (2021) Generalized olfactory detection of floral volatiles in the highly specialized Greya-Lithophragma nursery pollination system. Arthrop Plant Interact 15:209–221
Schulz S, Nishida R (1996) The pheromone system of the male danaine butterfly, Idea leuconoe. Bioorg Medic Chem 4:341–349
Schulz S, Boppré M, Vane-Wright RJ (1993) Specific mixtures of secretions from male scent organs of African milkweed butterflies (Danainae). Philos Trans R Soc Lond B 342:161–181
Schulz S, Yildizhan S, van Loon JJA (2011) The biosynthesis of hexahydrofarnesylacetone in the butterfly Pieris brassicae. J Chem Ecol 37:360–363
Shimomura K, Matsui S, Ohsawa K, Yajima S (2016) Saltational evolution of contact sex pheromone compounds of Callosobruchus rhodesianus (Pic). Chemoecology 26:15–23
Silberglied RE (1984) Visual communication and sexual selection among butterflies. In: Vane-Wright RI, Ackery PR (eds) The biology of butterflies. Academic Press, London, pp 207–223
Silberglied RE, Taylor OR(1973) Ultraviolet differences between sulfur butterflies, Colias eurytheme and C. philodice, and a possible isolating mechanism. Nature 241:406–408
Silberglied RE, Taylor OR(1978) Ultraviolet reflection and its behavioral role in the courtship of the sulphur butterflies Colias eurytheme and C. philodice (Lepidoptera, Pieridae). Behav Ecol Sociobiol 3:203–243
Takanashi T, Hiroki M, Obara Y (2001) Evidence for male and female sex pheromones in the sulfur butterfly, Eurema hecabe. Entomol Exp Appl 101:89–92
Vane-Wright RI, Boppré M (1993) Visual and chemical signalling in butterflies: functional and phylogenetic perspectives. Phil Trans R Soc Lond B 340:197–205
Vetter RS, Rutowski RL (1978) External sex brand morphology of three sulphur butterflies (Lepidoptera: Pieridae). Psyche 85:383–393
Wallin EA, Kalinová B, Kindl J, Hedenström E, Valterová I(2020) Stereochemistry of two pheromonal components of the bumblebee wax moth, Aphomia sociella. Sci Rep 10:2094
Yildizhan S, van Loon J, Sramkova A, Ayasse M, Arsene C, ten Broeke C, Schulz S(2009) Aphrodisiac pheromones from the wings of the small cabbage white and large cabbage white butterflies, Pieris rapae and Pieris brassicae. Chem Bio Chem 10:1666–1667
Acknowledgements
We are thankful to Dr. Tomoko Amimoto, at the Natural Science Center for Basic Research and Development (N-BARD), Hiroshima University, for the GC–HR–TOFMS measurements.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Chemical analyses and morphological observations were performed by C. Okuda, K. Yoshimori, and H. Ômura. Bioassays were performed by K. Yoshimori and H. Ômura. Statistical analyses were performed by H. Ômura. The first draft of the manuscript was written by H. Ômura, with editorial inputs from K. Yoshimori, C. Okuda, and S. Ohta. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yoshimori, K., Okuda, C., Ohta, S. et al. Sex pheromones from male forewings of the Common Grass Yellow Eurema mandarina. J Chem Ecol 48, 518–530 (2022). https://doi.org/10.1007/s10886-022-01368-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-022-01368-0