Abstract
In this meta-analysis, we evaluated overall strengths of relation between beliefs about pain, health, or illness and problems in functioning (i.e., functional impairment, affective distress, pain severity) in osteoarthritis and rheumatoid arthritis samples as well as moderators of these associations. In sum, 111 samples (N = 17,365 patients) met inclusion criteria. On average, highly significant, medium effect sizes were observed for associations between beliefs and problems in functioning but heterogeneity was also inflated. Effect sizes were not affected by arthritis subtype, gender, or age. However, pain belief content emerged as a significant moderator, with larger effect sizes for studies in which personal incapacity or ineffectiveness in controlling pain was a content theme of belief indices (i.e., pain catastrophizing, helplessness, self-efficacy) compared to those examining locus of control and fear/threat/harm beliefs. Furthermore, analyses of longitudinal study subsets supported the status of pain beliefs risk factors for later problems in functioning in these groups.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Arthritis is the leading cause of disability for people over age 15 in the United States (e.g., O’Donnell et al., 2010) as well as a common source of emotional distress among the affected (Margaretten et al., 2011; Murphy et al., 2012). Osteoarthritis (OA), a disease characterized by degeneration of cartilage in joints, is the most common subtype with a lifetime prevalence of about 9 % in the U.S. (Johannes et al., 2010; Neogi, 2013) and an estimated annual cost of $185.5 billion in healthcare expenditures alone (Kotlarz et al., 2009; White et al., 2007). OA is particularly widespread among adults over age 60 and those who are overweight or obese (Garver et al., 2014). Rheumatoid arthritis (RA), an autoimmune disease involving extreme joint tissue inflammation (Chang & Wei, 2011), is far less pervasive and affects 0.80–1.10 % of U.S. adults, typically with an onset at around age 40 (e.g., Silman & Hochberg, 2009). Regardless, RA is among the most potentially painful and crippling arthritis subtypes and can spread eventually to all body sites, including the heart, blood vessels, and lungs (Chang & Wei). Despite differences in symptoms, prevalence, and causes, OA and RA have shared psychological costs.
In line with World Health Organization QOL group (1993) recommendations regarding the assessment of health-related quality of life as well as key areas of functioning evaluated on popular multidimensional measures of disability (e.g., Kerns et al., 1985; Lurie, 2000; Nanda et al., 2003), pain severity, suffering (i.e., emotional distress), and interference with daily activity are widely-recognized, overlapping psychological consequences of ongoing pain that have been emphasized in pain assessment protocols. For example, following the onset of OA, substantial numbers of patients experience pain-related disability (Neogi, 2013), ongoing pain (Dray & Read, 2007; Jakobsson & Hallberg, 2002), and depression or anxiety (Memel et al., 2000; Sale et al., 2008). Similarly, aside from pain, long-term effects of RA include severe disability and reduced life expectancy (Villeneuve, 2013) as well as elevations in emotional distress. For example, 13–42 % of RA patients are estimated to experience comorbid depression (Bruce, 2008; Margaretten et al., 2011).
Notwithstanding the critical role that biological factors have for onsets and courses of OA and RA (e.g., Kourilovitch et al., 2014; Lafeber & Van Spil, 2013), cognitive social learning models have highlighted the importance of social relationships, interactions of people’s behavior with situations in their lives, and subjective expectancies, interpretations, and beliefs as factors that affect functioning. Regarding the latter, various authors have underscored the critical role of conceptually-overlapping beliefs reflecting locus of control (LOC), self-efficacy (SE), and/or helplessness as key influences on outcomes of OA and RA (e.g., Hewlett et al., 2008; Lorig et al., 1989; Stein et al., 1988a; Wallston et al., 1994).
To elaborate, research on locus of control (LOC) emerged from Rotter’s (1966) view that people rely on general beliefs about their ability to influence or control events, particularly when they are confronted with new situations. As applied to health and illness, Wallston and colleagues (e.g., Wallston, 2005; Wallston et al., 1994) contended that patients who believe good health results from taking care of themselves typically have stronger internal or personal health locus of control (HLOC) orientations and do what they can to maintain health and/or recover from illness. Conversely, patients who believe their health is mainly due to luck or efforts of health professionals often have stronger external HLOC orientations and may see little reason to make personal efforts vis a vis their health. Prospective studies of general population samples have linked stronger internal HLOC to better health (Gale et al., 2008) and reduced risk for heart attack and cancer (Sturmer & Hasselbach, 2006) at long-term follow-up.
In relation to OA and RA, patients with weaker internal LOC or HLOC orientations have been found to report more interference with activities of daily living (e.g., Cross et al., 2006; Graves et al., 2009; Lopez-Olivo et al., 2011; Zyrianova et al., 2011), depression (e.g., Rezaei et al., 2014; Smith et al., 1991; Treharne et al., 2005; Wallston et al., 1994; Zyrianova et al., 2011), and pain (e.g., Baker et al., 2008; Cross et al., 2006; Lopez-Olivo et al., 2011; Olchowska-Kotala et al., 2014; Rezaei et al., 2014; Tennen et al., 1992; Wallston et al., 1994) than peers who have stronger internal LOC orientations. However, modest or non-significant associations have been observed between internal LOC and impairment (e.g., Olchowska-Kotala et al., 2014; Tennen et al., 1992), depression (Ziarko et al., 2014) or other forms of distress such as anxiety (Treharne et al., 2005), and reported pain severity (Lopez-Olivo et al., 2011; Treharne et al., 2005) in some samples. While early research also found correlations between stronger external LOC orientations and elevations in pain and depression (Wallston et al., 1994), subsequent work suggested high scores on external LOC indices have weak associations with impaired daily functioning (Cross et al., 2006; Lopez-Olivo et al., 2011) and reported pain (e.g., Baker et al., 2008; Cross et al., 2006; Lopez-Olivo et al., 2011). In explaining non-significant effects of HLOC, Wallston (1992) pointed out that simply believing one’s actions affect health is not enough to maintain or improve health. Rather, good health and quality of life must also be highly valued for related efforts to be made.
Related social learning accounts contended that repeated experiences of unpredictable, uncontrollable aversive events result in learned helplessness, a dispositional tendency to believe highly desirable outcomes will not occur or highly aversive outcomes will arise, regardless of what one does (e.g., Abramson et al., 1978; Seligman, 1975). Such beliefs are typically accompanied by emotional, motivational, and cognitive deficits reflecting passivity (Stein et al., 1988a, b). While LOC beliefs and helplessness beliefs are moderately correlated (Wallston et al., 1994), the former reflect appraisals of where control over outcomes reside while the latter also tap expectations that one’s efforts will be ineffective. Learned helplessness models were applied initially to depression but later extended to the seemingly unpredictable and uncontrollable waxing and waning of RA symptoms (e.g., Nicassio et al., 1985). Within RA samples, elevations in helplessness beliefs have been linked to functional impairment (e.g., Bhat et al., 2010; Cadena et al., 2003; Covic et al., 2000, 2003; Englbrecht et al., 2012; Schoenfeld-Smith et al., 1996; Sinclair & Wallston, 2010; Smith et al., 1990, 1994), depression (e.g., Anderson et al., 1988; Cadena et al., 2003; Sinclair & Wallston, 2010; Smith et al., 1994) or general psychological distress (e.g., Englbrecht et al., 2012; Schoenfeld-Smith et al., 1996; Smith & Wallston, 1992), and reported pain (e.g., Bhat et al., 2010; Cadena et al., 2003; Covic et al., 2003; Smith & Wallston, 1992). Within OA samples, moderate positive associations have also been observed between helplessness beliefs and measures of reported pain (e.g., Creamer et al., 1999; Engel et al., 2004) and impairment (e.g., Creamer et al., 2000). Despite relatively consistent results, select arthritis studies have reported mixed or non-significant associations between helplessness beliefs and impaired daily functioning (e.g., Engel et al., 2004) as well as depression (Smith et al., 1990) and other forms of emotional distress (e.g., Anderson et al., 1988; Engel et al., 2004).
Bandura’s (1982, 1997) cognitive social learning model is a third, highly influential perspective that posits behavior can be affected by outcome expectations or beliefs that certain actions will lead to particular outcomes. However, self-efficacy (SE) expectations—confidence in one’s capacity to successfully perform specific actions that produce desired outcomes in given situations—are even stronger influences on behavior according to Bandura. While early work indicated SE beliefs are an important determinant of effort and perseverance shown in the face of stressors including pain, Bandura (1997) also hypothesized SE beliefs influence the performance of actions needed to manage or control pain itself and situations associated with pain. Extending Bandura’s “situation-specific” formulation of SE, pain researchers, including those whose work focuses on arthritis patients (e.g., Lorig et al., 1989), developed multidimensional domain-specific measures of SE beliefs related to the capacity to control pain, maintain daily activities despite pain, and manage experiences that accompany pain such as emotional distress.
Among patients with RA or OA, SE beliefs have had negative correlations with functional impairment (e.g., Baruth et al., 2013; Brekke et al., 2001; Cadena et al., 2003; Cross et al., 2006; Harrison, 2004; Marcum et al., 2014; Morone et al., 2009; Pells et al., 2008; Shelby et al., 2008; Somers et al., 2014), sometimes over and above the contributions of illness-related variables (e.g., Cadmus et al., 2010). Within some samples, patients who report weaker SE beliefs also report more severe pain (e.g., Brekke et al., 2001; Cadena et al., 2003; Harrison, 2004; Pells et al., 2008; Shelby et al., 2008; Somers et al., 2014) and heightened emotional distress (e.g., Barlow et al., 1997; Brekke et al., 2001; Cadena et al., 2003; Cross et al., 2006; Lowe et al., 2008; Pells et al., 2008; Rayahin et al., 2014; Shelby et al., 2008). Once again, however, modest and/or non-significant correlations with impaired functioning (e.g., Maly et al., 2006a, b; McKnight et al., 2010; Regan et al., 1988; van den Akker-Scheek et al., 2007; Wright et al., 2008), pain severity (e.g., Barlow et al., 2002; Lefebvre et al., 1999; Lopez-Olivo et al., 2011; Wylde et al., 2012) and/or emotional distress (e.g., Barlow et al., 1997; Lefebvre et al., 1999; Lopez–Lopez et al., 2008) have been documented in the SE studies of OA and RA.
Each cognitive social learning account outlined above was developed first as a general explanation of behavior and later applied to illuminate individual differences in functioning within chronic pain samples. In contrast, fear-avoidance (F-A) paradigms are pain-specific approaches designed to explain how cognitive factors including attentional focus and beliefs about pain as a signal for current bodily harm or potential future tissue damage influence the development and course of chronic pain (e.g., Leeuw et al., 2007; Vlaeyen and Linton 2000; Waddell et al., 1993). F-A models contend, in part, that pain-fearful people appraise pain sensations as signals for potential injury or ongoing harm (Vlaeyen & Linton, 2000) that can result in pain catastrophizing or exaggerated interpretations of pain as an extreme threat, even after healing is evident (Leeuw et al., 2007). Beliefs about pain as a source of threat, harm or impending catastrophe can exacerbate avoidance of physical movement. In turn, the resulting inactivity may contribute to physical deconditioning, long-term disability, depression, and increased pain (Vlaeyen & Linton, 2000). F-A models were used initially to explain how acute back pain can become chronic but subsequent extensions focused on other pain conditions, including arthritis (e.g., Somers et al., 2009; Sullivan et al., 2011).
Within RA and OA samples, patients who strongly endorse beliefs that pain is a source of actual or potential issue damage may be more susceptible to functional impairment (e.g., Heuts et al., 2004; Morone et al., 2009; Scopaz et al., 2009; Sullivan et al., 2011), emotional distress (e.g., Downe-Wamboldt & Melanson, 1995; Regan et al., 1988; Schiaffino & Revenson, 1995a; Scopaz et al., 2009; Somers et al., 2009; Sullivan et al., 2011), and more severe pain (e.g., Regan et al., 1988; Sullivan et al., 2011). Similarly, patients who report more pain catastrophizing can report more interference with daily activities (e.g., Costa et al., 2014; Edwards et al., 2010; Marcum et al., 2014; Morone et al., 2009; Shelby et al., 2008; Somers et al., 2009, 2014; Sullivan et al., 2011), distress (e.g., Edwards et al., 2010; Keefe et al., 2004; Lopez–Lopez et al., 2008; Rayahin et al., 2014; Shelby et al., 2008; Somers et al., 2009), and pain (e.g., Costa et al., 2014; Edwards et al., 2010; Keefe et al., 2004; Shelby et al., 2008; Somers et al., 2009, 2014; Sullivan et al., 2011) than peers who catastrophize less about pain. Nonetheless, within at least some samples, responses on measures of threat/harm/F-A beliefs or catastrophizing have had weak correlations with impaired functioning (e.g., Dekker et al., 1993; Downe-Wamboldt & Melanson, 1995; Rayahin et al., 2014; Regan et al., 1988; Schiaffino & Revenson, 1995a; Somers et al., 2009, 2014) and pain severity (e.g., Dekker et al., 1993; Kwissa-Gajewska et al., 2014; Schiaffino & Revenson, 1995b).
Taken together, the preceding overview underscores specific belief constructs based on cognitive social learning perspectives and F-A models as potentially important correlates of disturbances in functioning among patients with OA and RA. Regardless, each of these perspectives has also generated at least some conflicting evidence of weak associations between belief constructs and functioning. Hence, it is not clear whether these belief categories have strong overall strengths of relation with problems in functioning, nor is it evident that beliefs based on a particular cognitive social learning model or the F-A account have more value in informing problematic functioning among arthritis patients. Finally, it is not clear whether particular content themes of belief categories that cut across theoretical perspectives have stronger links with disturbances in OA and RA samples than other themes do. Meta- analysis, a procedure that relies on synthesized data from multiple studies instead of potentially idiosyncratic results of specific studies (Chan & Arvey, 2012), may clarify each of these issues. Given variable results in this literature, meta-analysis might also illuminate moderators that explain why belief-functioning relations have been robust in some studies and weak in others.
Regarding possible methodological moderators, indirect evidence suggests certain types of pain beliefs are more relevant than others in predicting arthritis outcomes. Recent meta-analyses on general chronic benign pain samples have found small to medium average effect sizes between measures of threatening pain appraisals or F-A beliefs and functional impairment (Jackson et al., 2014a; Zale et al., 2013) while a large mean effect size was reported for SE- impairment relations (Jackson et al., 2014b). These general patterns do not necessarily apply to OA and RA but imply effect sizes might be larger in studies of helplessness beliefs or SE beliefs rather than those that examine fear, threat, or harm beliefs salient to F-A models. On the other hand, exaggerated pain catastrophizing beliefs also appear to have robust associations with poorer functioning in narrative reviews (e.g., Leeuw et al., 2007). To date, meta-analyses on health/illness/pain LOC beliefs have not been conducted within chronic pain or arthritis samples. In contrast, following Wallston (1992), overall effect sizes for health or illness LOC beliefs could be comparatively weak because such measures are less typically task- or pain-specific than SE scales or those reflecting features of F-A models.
Operationalizations of functional impairment, content of emotional distress indices, and control of measures in prospective research can also influence effect sizes as well. Within general chronic pain samples, threat appraisals of pain and SE beliefs have had stronger associations with self-report than behavior performance indices of impairment (Jackson et al., 2014a, b). Moreover, beliefs related to threat or F-A should also have stronger conceptual links with fear or anxiety than other forms of emotional distress such as depression. Finally, within longitudinal studies, both logically and empirically, effect sizes between baseline beliefs and follow-up outcomes are more likely attenuated when baseline levels of the corresponding outcome have been statistically-controlled rather than left uncontrolled (Jackson et al., 2014b, 2016). However, significant average effect sizes under more stringent conditions of first controlling for baseline responses on an outcome would support the status of initial pain beliefs as a risk/protective factor for, not merely a correlate of, subsequent responses on that outcome.
Regarding possible sample characteristics that moderate belief-functioning relations, OA and RA differ in symptoms, prevalence, onset, causes, and severity (e.g., Axford et al., 2008; Silman & Hochberg, 2009); hence, it is possible that effect sizes also vary between these conditions. Past meta-analyses produced contradictory conclusions regarding moderating effects of pain site/condition on belief-outcome associations (Hinrichs-Rocker et al., 2009; Jackson et al., 2014a, b, 2016) though only small subsets of arthritis samples were assessed in these reviews. In terms of demographics, recent meta-analyses found SE beliefs and challenge appraisals had comparatively stronger relations with impairment or distress in older chronic pain samples, those having longer pain durations, and those comprising proportionately more women than men (Jackson et al., 2014a, b). Nonetheless, this pattern did not extend across all outcomes and analyses were based on numerous pain conditions, not just OA or RA. Hence, because details related to diagnosis, age, gender, and pain duration are typically reported in arthritis research, evaluating their moderating effects may help to identify specific arthritis patient subgroups in which beliefs correspond more strongly to outcomes.
Based on the preceding overview, this meta-analysis assessed the overall impact of belief constructs related to cognitive social learning models (i.e., health/illness LOC, helplessness, SE) and F-A models (i.e., pain as a source of threat, harm, or catastrophe) on problems in functioning that are directly relevant to health-related quality of life of OA and RA patients (i.e., functional impairment, affective distress, pain severity). While beliefs were expected to have significant overall associations with each area of functioning, significant effect size heterogeneity was also anticipated. Therefore, we explored the extent to which relations of beliefs with problems in functioning were moderated by methodological factors (i.e., belief measure content, impairment index content, affective distress measure content, research design) and sample characteristics (age, gender composition, pain duration, arthritis subtype).
Materials and methods
Search strategy
To identify relevant studies, PubMed, PsychINFO, Web of Science, Google Scholar, and Science Direct database searches were performed between dates of inception and March, 2015. Search terms were “arthritis” OR “rheumatism” AND “beliefs” OR “pain beliefs” OR “attitudes” OR “health” OR “illness” OR “appraisal” OR “control” OR “locus of control” OR “helplessness” OR “self-efficacy” OR “fear” OR “threat” OR “challenge” OR “harm” OR “catastrophizing”. Pain/health beliefs measures including the Arthritis Self-Efficacy Scale (ASES; Lorig et al., 1989), Rheumatoid Arthritis Self-Efficacy Scale (RASE; Hewlett et al., 2008), Multidimensional Health Locus of Control Questionnaire (MHLC; Wallston et al., 1994), Illness Perception Questionnaire (IPQ; Weinman et al., 1996), Survey of Pain Attitudes (SOPA; Jensen et al., 1987), Beliefs About Pain Control Questionnaire (BPCQ; Skevington, 1990), Rheumatology Attitudes Index (RAI; Callahan et al., 1988), Arthritis Helplessness Index (AHI; Nicassio et al., 1985), Tampa Scale for Kinesiophobia (TSK; Vlaeyen et al., 1995), Fear Avoidance Belief Questionnaire (FABQ; Waddell et al., 1993), and Pain Catastrophizing Scale (PCS; Sullivan et al., 1995) were also search terms. The broad search field, “anywhere” was used to identify citations. Reference lists of obtained articles were also reviewed to identify relevant papers.
Selection criteria
Abstracts of all potentially eligible studies were independently screened by the authors to exclude papers whose content was not salient. Subsequently, full-text versions of relevant papers were retrieved and reviewed to determine if they met the following 11 inclusion/exclusion criteria:
-
1.
Studies included adult human participant samples with an average age of 18 years or older.
-
2.
Studies were comprised principally of patients with ongoing OA and/or RA. Studies of laboratory pain, osteoporosis, other arthritis conditions such as fibromyalgia and gout, chronic pain unrelated to arthritis, and cancer pain were excluded.
-
3.
Belief measures had to reflect the domains of health, illness, or pain and tapped LOC, SE, helplessness, or F-A model belief constructs. Studies that assessed “general” LOC orientation, general SE, general competence, physical SE, or other pain beliefs related to other issues (e.g., Jensen et al., 1987) were excluded.
-
4.
Included studies reported at least one association between a measure of health, illness, or pain beliefs and an index reflecting disability/functional impairment, affective distress, or pain severity.
-
5.
Both cross-sectional and longitudinal studies were eligible for inclusion. When prospective studies only reported baseline assessment findings in sufficient detail, effect size calculations were limited to the initial assessment, per cross-sectional studies. Longitudinal studies that reported only associations between change scores for beliefs and change scores in outcomes were excluded. However, baseline belief-follow-up problems in functioning effect sizes were assessed in the moderator analyses examining effects of analytic approach within longitudinal study subsets (i.e., baseline functioning levels controlled or not controlled) to clarify the status of initial beliefs as correlates versus risk/protective factors for later problem outcomes, independent of corresponding baseline levels of problem functioning.
-
6.
Studies were included when belief measures were treated as independent variables or predictors rather than dependent measures.
-
7.
Regarding pain severity as an outcome, only studies that used continuous intensity measures or continuous measures re-coded into groups (e.g., median splits) were included for analyses. Conversely, research using less sensitive dichotomous categorical indices such as “presence of pain” (yes or no) or “presence of moderate to severe pain” (yes or no) was excluded.
-
8.
Reliability and validity data for all measures of pain beliefs and problems in functioning were reported or available.
-
9.
A minimum sample N of 50 was required because mean scores are more easily distorted by extreme responses in small samples.
-
10.
Only papers from peer-reviewed, English-language journals were retained for analyses. Reviews and studies that reported results based on secondary analyses of data in other articles were excluded.
-
11.
Because large-scale meta-analyses are time-consuming, resource-depleting, and labor intensive, we followed other published pain meta-analyses (Grossman et al., 2004; Jackson et al., 2014b) by including only papers that provided effect size information in a transparent manner or sufficient data for effect size calculations.
The formal assessment of study quality was also forgone due to the lack of consensus about criteria that best define study quality (Crombez et al., 2013a, b). However, several inclusion criteria (i.e., use of reliable, valid, sensitive measures, minimum sample N’s, publication in a peer-reviewed journal) ensured that all included studies had key methodological strengths and received favorable external editorial responses. All potentially relevant papers were reviewed independently by the authors. Disagreements in study selection and coding were resolved through discussion until consensus was achieved.
Search results
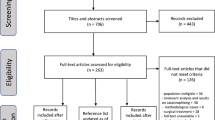
Possible studies were found in databases by key words (n = 7050), and health/pain belief questionnaire names (n = 136), emails to authors who published in the area (n = 12), and reference lists of obtained papers (n = 54). After removing duplicates, 6483 studies were retained for initial consideration. Figure 1 summarizes the screening process. Ultimately, 106 studies comprising 111 samples (N = 17,365 patients) fulfilled all selection criteria. Rates of inter-rater agreement were satisfactory for study inclusion in the meta-analysis (k = .89) and coding of variables used in the meta-analyses (k = .91).
Coding study characteristics
For each included study, data were extracted regarding first author, year of publication, sample size, gender composition (percentage of women per sample), mean age, arthritis type (RA vs. OA vs. mixed RA and OA), pain duration in years, research design (cross-sectional vs. longitudinal), health/illness/pain belief measure content (internal/personal LOC vs. external LOC vs. SE vs. fear/threat/harm vs. helplessness vs. catastrophizing) and problem area (i.e., disability/impairment vs. affective distress vs. pain severity). Impairment indices were coded as either self-report or behavioral performance measures in moderator analyses. Affective distress scales were coded as measures of depressive symptoms, anxiety/fear, or general emotional distress. Levels of inter-rater agreement in classifying measures of pain beliefs and functioning based on these categories were 100 %.
Characteristics of included studies
Table 1 summarizes features of studies included for meta-analysis. Papers published from 1988 to March, 2015 were included for analysis; more than half (53.77 %) were published after 2005, thus highlighting pain beliefs as a pertinent focus in recent arthritis studies. On average, samples included 156.44 patients (SD = 120.68, range 50–815), were late-middle-aged (M = 59.94 years, SD = 7.08, range 44.12–75.00 years) and had more women than men (M = 75.16 %, SD = 16.50 %, range 0–100 %). For arthritis type, 58 samples comprised RA patients, 38 included OA patients, and 15 evaluated both RA and OA patients. The mean pain duration of samples was 11.28 years (SD = 5.25 years, range 1.92 years to 25.9 years). Regarding longitudinal study subsets, follow-up impairment effect sizes were reported in 25 studies, an average of 10.21 months after baseline impairment assessments (SD = 8.29 months). Follow-up affect distress effect sizes were presented in 24 papers, an average of 10.10 months after initial distress evaluations (SD = 10.04 months). Follow-up pain severity ratings were reported in 18 studies, an average of 8.27 months after baseline assessments (SD = 5.77 months). In relation to belief categories of focus based on cognitive social learning models, 18 studies assessed internal and/or external health/illness LOC beliefs, 16 examined helplessness beliefs, and 62 evaluated pain SE beliefs based on measures listed in Table 1. Regarding belief constructs reflected in F-A models of pain, 10 studies assessed fear/threat/harm beliefs related to pain or illness and 18 studies evaluated pain catastrophizing beliefs. Finally, functional impairment, affective distress, and pain severity were assessed in 84, 74, and 61 samples, respectively (Table 1).
Effect size computations
Comprehensive Meta-Analysis Version 2.0 (CMAV2; Borenstein et al., 2005) was used for analyses. Bivariate correlations (r) represented individual effect sizes of included studies. Papers that reported regression coefficients within multiple regression models rather than bivariate associations were retained for analyses. However, such correlations may be attenuated within multiple regression models that include multiple predictors. More occasionally, measures in multiple regression models act as “suppressors” of error variance, artificially inflating effect sizes of other predictor measures. Therefore, where relevant, overall effect sizes for each outcome were also computed and presented in the text, after excluding data from multiple regression studies.
Effect sizes were weighted by the inverse of a study’s variance. Cochran’s Q test evaluated effect size heterogeneity for each outcome. When Q values reflected significant heterogeneity, subgroup analyses and meta-regression assessed sources of variability in effects. I square values represented amount of observed heterogeneity. Following from Higgins and Thompson (2002), I square values of 25, 50 and 75 % were low, moderate, and high, respectively. Overall effect sizes were based on random effects models as recommended by Hoffman et al. (2007). Drawing upon Cohen (1992), effect sizes of r = .10, r = .30, and r = .50 were interpreted as small, medium, and large, respectively.
Outlier analysis
Outlier analyses were conducted on the overall data set based on effect size magnitudes and sample sizes (Avolio et al., 2009). Hunter and Schmidt (2004) contended that extreme values can cause significant within-group heterogeneity of individual effect sizes that do not reflect reality. In the process of considering outliers, values more than three standard deviations from the mean were operationalized as outliers (Kline, 1998).
Moderator analyses
Categorical moderators operationalized above, including (a) pain/health/illness belief measure content (b) arthritis subtype, (c) analytic approach used within longitudinal studies of each outcome, (d) type of impairment scale, and (e) content of affective distress were assessed with Cochran’s Q values with effect sizes based on correlation coefficients. Moderating effects of continuous measures of sample age, gender composition, and pain duration were assessed via method of moments analyses (Higgins & Thompson, 2002).
Evaluation of publication bias
Publication bias was estimated by inspecting effect size funnel plots for asymmetrical distributions around mean effect sizes via trim and fill analyses (Duval & Tweedie, 2000). Typically, larger samples (top of effect size plot) provide the most accurate estimates of effect size; the spread should increase symmetrically with smaller samples toward the bottom of the plot. Hence, when there is no evidence of publication bias, the plot should resemble an inverted funnel. Bias against the publication of studies having non-significant effects is suggested by funnel plots that include fewer effects sizes on the left than the right side of the mean effect size for an outcome. This method provides an estimate of the nature and number of studies missing from a distribution and adjusted effect sizes based on estimated contributions of missing studies.
Results
Outlier analysis
One study with a very large sample (i.e., Brekke et al., 2001) was identified as an outlier but retained for analyses because overall effect sizes did not change as a function of its inclusion or exclusion. However, three effect sizes identified as outliers in the belief-affective distress analyses—Kwan (r = −0.900), Rezaei (r = 0.380), Zyrianova (r = −0.896)—were omitted to be conservative because their retention resulted in a slightly stronger average effect size (r = -.377, p < .001, 95 % CI −.319 to −.432) compared to their omission.
Overall associations between pain beliefs and outcomes of arthritis
Highly significant, medium average effect sizes were found for overall relations between pain beliefs and each facet of problem functioning. Specifically, beliefs reflecting elevations in SE or internal/personal control beliefs as well as reductions in external control, helplessness, fear/threat/harm, and catastrophizing were related to moderately less functional impairment, affective distress, and pain severity in OA and RA samples (Table 2). Omitting studies that reported odds-ratios, standard regression or partial correlation coefficients within regression models, average effect sizes for belief-impairment relations (r = −.358, P < .001, 95 % CI −.317 to −.399), belief-distress associations (r = −.368, P < .001, 95 % CI −.329 to −.405), and belief-pain severity relations (r = −.349, P < .001, 95 % CI −.303 to −.393) were slightly larger than those obtained from retaining regression coefficient data. Table 2 also indicates medium to high heterogeneity was evident in each overall effect size analysis. In light of such variability, moderator analyses assessed effects of select methodological factors and sample characteristics on belief-functioning effect sizes.
Moderator analyses for belief-impairment associations
Table 3 presents subgroup analyses of potential categorical moderators of belief-impairment relations. The significant moderating effect of pain belief subtype revealed significant, medium average effect sizes in studies of associations between SE, helplessness, or catastrophizing beliefs and impairment. In contrast, small, though significant average effect sizes were found for studies that assessed internal/personal health LOC and threat/fear/harm beliefs. Finally, small, non-significant effect sizes were observed in studies that assessed beliefs reflecting external LOC (e.g., powerful others, chance, luck). Effect size heterogeneity was substantially lower after controlling for belief subtype but was still considerable and highly significant (Q within value = 573.637, p < .001).
Overall belief-impairment relations did not differ much as a function of having RA or OA or use of self-report versus behavior performance measures of impairment (Table 3). Within the prospective study subset, belief-impairment relations were not moderated by analysis approach: a medium average effect size was observed for prospective studies that did not control for initial impairment levels but a small, significant average effect size was also found for longitudinal studies controlling for baseline impairment, supporting the status of pain beliefs as risk/protective factors for impairment. Finally, age (point of slope = .00371, p = .239), pain duration (point of slope = .00438, p = .408), and gender composition (point of slope = .00072, p = .566) did not moderate belief-impairment relations.
Moderator analyses for belief-affective distress associations
Belief subtype also moderated belief-distress effect sizes (Table 4). Effect sizes for catastrophizing beliefs had medium to large confidence intervals while those for SE, helplessness and/or threat/fear-avoidance beliefs were medium in strength. Conversely, studies assessing LOC beliefs had small, significant mean effect sizes. Effect size heterogeneity remained highly significant after controlling for belief subtype (Q within value = 250.126, p < .001). Moderating effects of arthritis subtype and affective distress measure content were not significant (see Table 4). However, in the longitudinal study subset, analysis approach was a significant moderator; effect sizes were small and medium, respectively, for studies that controlled and did not control for baseline distress levels. Heterogeneity was substantially lower but remained significant after controlling for analytic approach in longitudinal studies (Q within values = 43.530, p < .01). Sample age (point of slope = .00255, p = .426), gender composition (point of slope = .00095, p = .506), and pain duration (point of slope = .00004, p = .993) did not moderate belief-distress effect sizes.
Moderator analyses for belief-pain severity associations
Once again, belief subtype moderated belief-pain severity effect sizes (Table 5). Significant, medium effect sizes were found in studies that assessed SE, helplessness, and catastrophizing beliefs, small to medium effect size confidence intervals emerged in studies that tapped internal/personal health LOC or threat/fear beliefs, and a small, non-significant mean effect size was observed in studies of external LOC beliefs. Substantial heterogeneity remained after controlling for belief subtype (Q within values = 293.604, p < .001). Belief-pain severity effect sizes did not differ as a result of arthritis subtype (Table 5). However, in the prospective study subset, medium and small average effect sizes emerged, respectively, for studies that did not control for baseline pain severity and those that did; covarying initial pain severity levels, the effect size was reduced but significant, indicating pain beliefs served as risk/protective factors related to follow-up pain severity levels. Belief-pain severity effect sizes did not vary due to sample age (point of slope = .00220, p = .551), gender (point of slope = -.00024, p = .855), or pain duration (point of slope = .00297, p = .548).
Effects of publication bias
Trim and fill analyses indicated that symmetrical effect sizes distributions were observed in study plots assessing relations of pain beliefs with each facet of functioning (see Supplementary Figures 2-4). Consequently, no studies were estimated to be missing from any of the three distributions and no effect size adjustments were made.
Discussion
In this meta-analysis, we assessed overall associations of beliefs about pain, health, and illness with functional impairment, affective distress, and pain severity in published accounts of RA and OA samples. Highly significant, medium, average effect sizes highlighted how belief/appraisal constructs based on select cognitive social learning perspectives as well as F-A models of pain have robust links with key problems in functioning in these groups. Overall findings bolstered recent meta-analyses implicating SE beliefs (Jackson et al., 2014b), F-A beliefs (Zale et al., 2013), and threat appraisals of pain (Jackson et al., 2014a) as correlates of functioning in samples having diverse pain conditions. However, in line with variable results between individual studies, overall belief-outcome effect sizes were accompanied by medium to high levels of heterogeneity.
Towards informing the potential utility of different theoretical models, the nature of pain beliefs assessed was a significant moderator across all three problem areas and partially explained effect size variability for each analysis. The longstanding recognition of SE beliefs as important influences on functioning in OA and RA samples is reflected in the development of arthritis-specific SE measures (Hewlett et al., 2008; Lorig et al., 1989) and interventions to increase SE in these groups (e.g., Barlow et al., 2002; Smarr et al., 1997). Dovetailing with reviews linking high SE levels with beneficial health outcomes in non-clinical samples (Holden, 1992) and those with various chronic benign pain conditions (Jackson et al., 2014b), medium effect sizes for SE in this meta-analysis underscored how confidence in one’s capacity to perform particular actions corresponds to reduced impairment, distress, and pain intensity among RA and OA patients. While analyses could not demonstrate causal effects of SE beliefs on these problems, in the context of evidence showing how treatment-based improvements in SE contribute to reductions in disability, reported pain, and/or emotional distress (e.g., Lorig & Holman, 2003), graded mastery experiences, vicarious learning of mastery, physiological arousal reinterpretation, and social persuasion may enhance SE (Bandura, 1997; Lorig et al., 1989) and reduce negative psychological consequences of OA and RA.
Aside from SE, medium average effect sizes for helplessness beliefs indicated patients who believed they were incapable and/or unable to influence the course of their illness were especially prone to elevations in impairment, affective distress, and pain severity. Such results may not elucidate underlying mechanisms but some authors have argued helplessness beliefs are part of a vicious cycle that includes passive coping and impairment which, in turn, perpetuate helplessness (e.g., Covic et al., 2003; Smith & Wallston, 1992). Respectively, helplessness beliefs and SE reflect dispositional tendencies and highly specific actions. Nonetheless, beliefs that one is ineffective in or incapable of undertaking actions that affect functioning are a common element of helplessness and low SE. Consequently, interventions designed to increase SE (e.g., graded mastery) may help to reduce helplessness beliefs as well.
Average effect sizes were more modest and/or not significant in studies drawing from alternate social cognitive paradigms that evaluated relations between health, illness, or pain LOC beliefs and functioning. More precisely, small, statistically significant average effect sizes were found for relations between internal LOC and each problem in functioning while mean effect sizes for powerful others’ and chance/luck LOC were small to very small and not reliably significant. One potential limitation of using health LOC scales in groups with pain conditions is the possibility that LOC orientation related to one’s general health status is not the same as LOC beliefs about one’s illness (Wallston et al., 1994). Relatedly, patients with chronic pain may not know how to interpret or respond to health LOC queries such as, “If I do the right things, I can stay healthy”. While the development of illness- or pain-specific LOC scales has helped to address these limitations, a strong internal LOC orientation towards health or illness may have more limited benefits for people who feel incapable of carrying out behaviors that improve functioning (Schoenfeld-Smith et al., 1996). On this basis, assessments and interventions that focus on increasing SE and/or reducing helplessness may be more useful in working with OA and RA patients than evaluating and/or attempting to change LOC beliefs.
F-A models emphasize beliefs about pain as a threat for potential future tissue damage or ongoing harm and pain catastrophizing as precipitants of activity avoidance, physical disuse, disability, and increases in pain (Vlaeyen & Linton, 2000). Even though avoidance can occur both in the presence and absence of fear or threat appraisals (Wideman et al., 2013), significant small to medium average effect sizes for relations between problems in functioning and beliefs reflecting F-A, threat, and/or harm supported conclusions of meta-analyses on sundry pain conditions (Jackson et al., 2014a; Zale et al., 2013) and contentions that F-A model features apply to OA and RA patients (Heuts et al., 2004; Steultjens et al., 2002). Medium average effect sizes found for catastrophizing and each focus of problem functioning were even stronger than those based on F-A, threat or harm beliefs. Pain catastrophizing scales have substantial associations with F-A belief and threat appraisal indices but these constructs are also somewhat distinct (Jackson et al., 2014a). For example, content reflecting pain as a potential threat is present in the two most popular pain catastrophizing scales used in this meta-analysis (Rosentiel & Keefe, 1983; Sullivan et al., 1995), yet both scales include items that tap perceived helplessness. As noted above, beliefs about one’s incapacity or ineffectiveness in making changes have powerful repercussions for functioning among RA and OA patients.
In contrast to pervasive moderating effects of pain belief content, there was no evidence that belief-problem area effect sizes were moderated by age, gender composition, arthritis subtypes, or pain durations. Jackson et al. (2014b) reported SE-impairment relations were comparatively stronger in older samples and those having pain for longer periods of time. However, samples assessed in that meta-analysis were over 12.5 years younger on average, had a substantially shorter mean pain duration, and were far more variable in relation to pain conditions assessed compared to samples included in this review. Hence, beliefs about pain and health may have similar implications for difficulties in functioning regardless of age, pain duration, gender composition or arthritis subtype of samples. The non-significant moderator effect for impairment measure type highlighted how average strengths of relation between pain beliefs and self-reported impairment did not differ markedly from those based on performance-based impairment indices. Consequently, methodological concerns such as common method variance (i.e., exclusive use of self-report measures) do not provide a plausible explanation for belief-impairment findings. Similarly, effect sizes for belief-affective distress relations were comparable regardless of whether distress content reflected anxiety/fear, depression, or general emotional distress. Hence, at least when certain types of appraisals are considered, subject matter overlaps in the measurement of pain beliefs and emotional distress (e.g., fear) do not account for associations.
Finally, moderator analyses of longitudinal study subsets related to follow-up levels of impairment, distress, and pain severity indicated, as expected, that average effect sizes were stronger when baseline levels of impairment, distress, and pain severity were not controlled. Nonetheless, all average effect sizes for relations between baseline pain beliefs and follow-up adjustment difficulties were significant even when corresponding baseline responses on measures of adjustment problems had been statistically controlled. Significant effect sizes based on this more stringent analytic approach supported the proposition that beliefs about pain, health and illness are risk/protective factors for, not mere correlates of, later problems in functioning in these patient groups. As such, interventions that change less adaptive pain beliefs would seem to have important implications for later outcomes. While several prospective studies featured follow-up assessments 2–4 years after baseline (e.g., Brekke et al., 2001; Miller and Cronan (1998); Rayahin et al., 2014; Smith et al., 1994), these results were based on average baseline to follow-up intervals of 8–10 months. Consequently, additional long-term longitudinal research is needed to gauge the impact of initial pain beliefs on problems in functioning over more extended periods.
A particular strength of this meta-analysis was the evaluation of a large number of relevant studies, all of which featured methodological strengths including reliable, valid, sensitive measures, minimum sample sizes, and candid reporting of effect size information. Nonetheless, several limitations of the review must also be acknowledged. First, due to the focus on RA and OA samples, generalizations cannot be made to other types of arthritis such as fibromyalgia or gout or non-arthritic chronic pain syndromes. Second, findings may not apply to pain beliefs (e.g., pain as a cause of disability, others’ solicitude when one is in pain) and facets of functioning (e.g., anger as the expression of distress, fatigue, medication use, health care visits) outside the scope of this review. Third, given that effect size variability was significant after moderating effects of pain belief content were controlled, other factors may have affected heterogeneity. While age and gender were nearly always reported in selected studies, other key demographics including employment status, socioeconomic status (SES), and relationship status were examined less often; assessing these factors as a matter of course within individual studies would facilitate evaluations of possible moderating effects of SES indicators in future meta-analyses. Another plausible methodological influence on heterogeneity was the wide variety of measures used to assess impairment, distress, and pain severity. For example, 44 different impairment indices were used including those that reflected perceived physical functioning, psychosocial functioning, and global quality of life. While the inclusion of a broad array of impairment measures bolstered the external validity of studies from this domain, it likely contributed to effect size heterogeneity as well.
Fourth, some included studies, particularly those that relied on stepwise regression analyses, selectively reported only statistically significant effect sizes and excluded such details for non-significant predictors. This practice is problematic because it can inflate risk for type I errors and distort the scientific record to seem more robust than it is in reality. In light of this concern, we urge researchers and those who evaluate their work to ensure all relevant effects are reported transparently and in sufficient detail, including those that are not statistically significant. Doing so can help to ensure sober conclusions are drawn about this and other literatures. Finally, causal effects of pain beliefs on problems in functioning could not be tested because neither experimental manipulations of pain beliefs nor random assignment to groups was characteristic of included studies. As intervention-based studies accumulate, causal effects of modifying pain beliefs on changes in functioning may be clarified.
Conclusion
In sum, this meta-analysis indicated beliefs about health, illness and/or pain have significant moderate overall associations with impairment, distress and pain severity in RA and OA samples. Across all three problem areas, effect sizes were comparatively larger in studies that assessed beliefs related to SE, pain catastrophizing, and/or helplessness. Given personal incapacity or ineffectiveness is a content theme that each of these belief categories has in common, the use of belief indices that reflect this focus within assessment and interventions that increase subjective appraisals of effectiveness would seem to be critically important in these patient groups. In contrast to the impact of pain belief content of results, overall effect sizes were not moderated by arthritis subtype, pain duration, age, and gender composition of samples, use of subjective versus behavior performance measures of impairment or content of affective distress indices (i.e., depression, anxiety-fear, or general psychological distress). However, moderator analyses of longitudinal study subsets established beliefs about health, illness, and pain not only as correlates of subsequent functioning but also risk/protective factors that may predict the likelihood that adjustment will be more or less disturbed an average of 8–10 months later. Future work should assess the relative impact of beliefs reflecting one’s own ineffectiveness in dealing with pain and/or accompanying concerns versus other types of pain beliefs on problems in functioning within other chronic pain conditions. Furthermore, prospective studies with follow-up durations beyond 1 year would help to clarify the long-term impact of baseline pain beliefs on functioning of chronic pain samples.
References
* References with asterisks were included in the meta-analysis
Abraido-Lanza, A. (1997). Latinas with arthritis: Effects of illness, role identity, and competence on psychological well-being. American Journal of Community Psychology, 25, 601–627.
*Abramson, L. Y., Seligman, M. E. P., & Teasdale, J. D. (1978). Learned helplessness in humans: Critique and reformulation. Journal of Abnormal Psychology, 87, 49–74.
*Affleck, G., Tennen, I. I., Urrows, S., & Higgins, P. (1992). Neuroticism and the pain-mood relation in rheumatoid arthritis: Insights from a prospective daily study. Journal of Consulting and Clinical Psychology, 60, 119–126.
*Allen, K. D., Oddone, E. Z., Coffman, C. J., Keefe, F. J., Lindquist, J. H., & Bosworth, H. B. (2010). Racial differences in osteoarthritis pain and function: Potential explanatory factors. Osteoarthritis and Cartilage, 18, 160–167.
*Anderson, K. O., Keefe, F. J., Bradley, L. A., McDaniel, L. K., Young, L. D., Turner, R. A., et al. (1988). Prediction of pain behavior and functional status of rheumatoid arthritis patients using medical status and psychological variables. Pain, 33, 25–32.
Avolio, B. J., Reichard, R. J., Hannah, S. T., Walumbwa, F. O., & Chan, A. (2009). A meta-analytic review of leadership impact research: Experimental and quasi-experimental studies. The Leadership Quarterly, 20, 764–784.
Axford, J., Heron, C., Ross, F., & Victor, C. R. (2008). Management of knee osteoarthritis in primary care: Pain and depression are the major obstacles. Journal of Psychosomatic Research, 64, 461–467.
*Baker, T. A., Buchanan, N. T., & Corson, N. (2008). Factors influencing chronic pain intensity in older black women: Examining depression, locus of control, and physical health. Journal of Women’s Health, 17, 869–878.
Bandura, A. (1982). Self-efficacy mechanism in human agency. American Psychologist, 37, 122–147.
Bandura, A. (1997). Self-efficacy: The exercise of control. New York: Macmillan.
Barlow, J. H., Cullen, L. A., & Rowe, I. F. (2002). Educational preferences, psychological well-being and self-efficacy among people with rheumatoid arthritis. Patient Education and Counseling, 46, 11–19.
*Barlow, J. H., Williams, B., & Wright, C. C. (1997). The reliability and validity of the arthritis self-efficacy scale in a UK context. Psychology, Health and Medicine, 2, 3–17.
*Baruth, M., Wilcox, S., Schoffman, D. E., & Becofsky, K. (2013). Factors associated with disability in a sample of adults with arthritis. Disability and Health Journal, 6, 377–384.
*Benka, J., Nagyova, I., Rosenberger, J., Macejova, Z., Lazurova, I., der Klink, V., et al. (2014). Is coping self-efficacy related to psychological distress in early and established rheumatoid arthritis patients? Journal of Developmental and Physical Disabilities, 26, 285–297.
*Bhat, A., DeWalt, D., Zimmer, C., Fried, B., & Callahan, L. F. (2010). The role of helplessness, outcome expectation for exercise and literacy in predicting disability and symptoms in older adults with arthritis. Patient Education and Counseling, 81, 73–78.
Borenstein, M., Hedges, L., Higgins, J., & Rothstein, H. (2005). Comprehensive meta-analysis version 2. Englewood, NJ: Biostat.
*Brekke, M., Hjortdahl, P., & Kvien, T. K. (2001). Self-efficacy and health status in rheumatoid arthritis: A two-year longitudinal observational study. Rheumatology, 40, 387–392.
Bruce, T. O. (2008). Comorbid depression in rheumatoid arthritis: Pathophysiology and clinical implications. Current Psychiatry Reports, 10, 258–264.
*Cadena, J., Vinaccia, S., Pérez, A., Rico, M. I., Hinojosa, R., & Anaya, J. M. (2003). The impact of disease activity on the quality of life, mental health status, and family dysfunction in Colombian patients with rheumatoid arthritis. Journal of Clinical Rheumatology, 9, 142–150.
*Cadmus, L. I. S. A., Patrick, M. B., Maciejewski, M. L., Topolski, T. A. R. I., Belza, B. A. S. I. A., & Patrick, D. L. (2010). Community-based aquatic exercise and quality of life in persons with osteoarthritis. Medicine and Science in Sports and Exercise, 42, 8–15.
Callahan, L. F., Brooks, R. H., & Pincus, T. (1988). Further analysis of learned helplessness in rheumatoid arthritis using a rheumatology attitudes index. The Journal of Rheumatology, 15, 418–426.
*Carlisle, A. C. S., John, A. M. H., & Lloyd, M. (2005). The self-regulatory model in women with rheumatoid arthritis: Relationships between illness representations, coping strategies, and illness outcome. British Journal of Health Psychology, 10, 571–587.
Chan, M. E., & Arvey, R. D. (2012). Meta-analysis and the development of knowledge. Perspectives on Psychological Science, 7, 79–92.
Chang, X., & Wei, C. (2011). Glycolysis and rheumatoid arthritis. International Journal of Rheumatic Diseases, 14, 217–222.
Cohen, J. (1992). A power primer. Psychological Bulletin, 112, 155–159.
*Costa, J., Pinto-Gouveia, J., & Maroco, J. (2014). Pain related catastrophizing on physical limitation in rheumatoid arthritis patients: Is acceptance important? The Spanish Journal of Psychology, 17, 1–13.
*Covic, T., Adamson, B., & Hough, M. (2000). The impact of passive coping on rheumatoid arthritis pain. Rheumatology, 39, 1027–1030.
*Covic, T., Adamson, B., Spencer, D., & Howe, G. (2003). A biopsychosocial model of pain and depression in rheumatoid arthritis: A 12-month longitudinal study. Rheumatology, 42, 1287–1294.
*Covic, T., Tyson, G., Spencer, D., & Howe, G. (2006). Depression in rheumatoid arthritis patients: demographic, clinical, and psychological predictors. Journal of Psychosomatic Research, 60, 469–476.
*Creamer, P., Lethbridge-Cejku, M., & Hochberg, M. C. (1999). Determinants of pain severity in knee osteoarthritis: Effect of demographic and psychosocial variables using 3 pain measures. Journal of Rheumatology, 26, 1785–1792.
*Creamer, P., Lethbridge-Cejku, M., & Hochberg, M. C. (2000). Factors associated with functional impairment in symptomatic knee osteoarthritis. Rheumatology, 39, 490–496.
Crombez, G., Van Ryckeghem, D. M., Eccleston, C., & Van Damme, S. (2013a). Attentional bias to pain-related information: A meta-analysis. Pain, 154, 497–510.
Crombez, G., Viane, I., Eccleston, C., Devulder, J., & Goubert, L. (2013b). Attention to pain and fear of pain in patients with chronic pain. Journal of Behavioral Medicine, 36, 371–378.
*Cross, M. J., March, L. M., Lapsley, H. M., Byrne, E., & Brooks, P. M. (2006). Patient self-efficacy and health locus of control: Relationships with health status and arthritis-related expenditure. Rheumatology, 45, 92–96.
*Dekker, J., Tola, P., Aufdemkampe, G., & Winckers, M. (1993). Negative affect, pain and disability in osteoarthritis patients: The mediating role of muscle weakness. Behavior Research and Therapy, 31, 206–303.
*Dirik, G., & Karanci, A. N. (2010). Psychological distress in rheumatoid arthritis patients: An evaluation within the conservation of resources theory. Psychology and Health, 25, 617–632.
*Downe-Wamboldt, B., & Melanson, P. M. (1995). Emotions, coping, and psychological well-being in elderly people with arthritis. Western Journal of Nursing Research, 17, 250–265.
Dray, A., & Read, S. J. (2007). Arthritis and pain: Future targets to control osteoarthritis pain. Arthritis Research and Therapy, 9, 212–225.
Duval, S., & Tweedie, R. (2000). Trim and fill: A simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics, 56, 455–463.
*Edwards, R. R., Giles, J., Bingham, C. O, 3rd, Campbell, C., Haythornthwaite, J. A., & Bathon, J. (2010). Moderators of the negative effects of catastrophizing in arthritis. Pain Medicine, 11, 591–599.
*Engel, C., Hamilton, N. A., Potter, P. T., & Zautra, A. J. (2004). Impact of two types of expectancy on recovery from total knee replacement surgery (TKR) in adults with osteoarthritis. Behavior Medicine, 30, 113–123.
*Englbrecht, M., Gossec, L., DeLongis, A., Scholte-Voshaar, M., Sokka, T., Kvien, T. K., et al. (2012). The impact of coping strategies on mental and physical well-being in patients with rheumatoid arthritis. Seminars in Arthritis and Rheumatism, 41, 545–555.
Gale, C. R., Batty, G. D., & Deary, I. J. (2008). Locus of control at age 10 years and health outcomes and behaviors at age 30 years: The 1970 British Cohort Study. Psychosomatic Medicine, 70, 397–403.
Garver, M. J., Focht, B. C., Dials, J., Rose, M., Lucas, A. R., Devor, S. T., et al. (2014). Weight status and differences in mobility performance, pain symptoms, and physical activity in older, knee osteoarthritis patients. Arthritis. doi:10.1155/2014/375909
*Graves, H., Scott, D., Lempp, H., & Weinman, J. (2009). Illness beliefs predict disability in rheumatoid arthritis. Journal of Psychosomatic Research, 67, 417–423.
*Greene, B. L., Haldeman, G. F., Kaminski, A., Neal, K., Lim, S. S., & Conn, D. L. (2006). Factors affecting physical activity behavior in urban adults with arthritis who are predominantly African-American and female. Physical Therapy, 86, 510–519.
Grossman, P., Niemann, L., Schmidt, S., & Walach, H. (2004). Mindfulness-based stress reduction and health benefits: A meta-analysis. Journal of Psychosomatic Research, 57, 35–43.
*Harrison, A. L. (2004). The influence of pathology, pain, balance, and self-efficacy on function in women with osteoarthritis of the knee. Physical Therapy, 84, 822–831.
*Heuts, P. H., Vlaeyen, J. W., Roelofs, J., de Bie, R. A., Aretz, K., van Weel, C., et al. (2004). Pain-related fear and daily functioning in patients with osteoarthritis. Pain, 110, 228–235.
*Hewlett, S., Cockshott, Z., Almeida, C., Richards, P., Lowe, R., Greenwood, R., et al. (2008). Sensitivity to change of the rheumatoid arthritis self-efficacy scale (RASE) and predictors of change in self-efficacy. Musculoskeletal Care, 6, 49–67.
Higgins, J. P., & Thompson, S. G. (2002). Quantifying heterogeneity in a meta-analysis. Statistics in Medicine, 21, 1539–1558.
Hinrichs-Rocker, A., Schulz, K., Järvinen, I., Lefering, R., Simanski, C., & Neugebauer, E. (2009). Psychosocial predictors and correlates for chronic post-surgical pain (CPSP): A systematic review. European Journal of Pain, 13, 719–730.
Hoffman, B. M., Papas, R. K., Chatkoff, D. K., & Kerns, R. D. (2007). Meta-analysis of psychological interventions for chronic low back pain. Health Psychology, 26, 1–9.
Holden, G. (1992). The relationship of self-efficacy appraisals to subsequent health related outcomes: A meta-analysis. Social Work in Health Care, 16, 53–93.
*Holm, M. B., Rogers, J. C., & Kwoh, C. K. (1998). Predictors of functional disability in patients with rheumatoid arthritis. Arthritis and Rheumatism, 11, 346–355.
Hunter, J. E., & Schmidt, F. L. (2004). Methods of meta-analysis: Correcting error and bias in research findings (2nd ed.). Thousand Oaks, CA: Sage.
Jackson, T., Tian, P., Wang, Y., Iezzi, T., & Xie, W. (2016). Towards identifying moderators of associations between pre-surgery emotional distress and post-operative pain outcomes: A meta-analysis of longitudinal studies. The Journal of Pain, 17, 874–888. doi:10.1016/j.jpain.2016.04.003.
Jackson, T., Wang, Y., & Fan, H. (2014a). Associations between pain appraisals and pain outcomes: Meta-analyses of laboratory pain and chronic pain literatures. The Journal of Pain, 15, 586–601.
Jackson, T., Wang, Y., Wang, Y., & Fan, H. (2014b). Self-efficacy and chronic pain outcomes: A meta-analytic review. The Journal of Pain, 15, 800–814.
Jakobsson, U., & Hallberg, I. R. (2002). Pain and quality of life among older people with rheumatoid arthritis and/or osteoarthritis: A literature review. Journal of Clinical Nursing, 11, 430–443.
*James, N. T., Miller, C. W., Brown, K. C., & Weaver, M. (2005). Pain disability among older adults with arthritis. Journal of Aging and Health, 17, 56–69.
Jensen, M. P., Karoly, P., & Huger, R. (1987). The development and preliminary validation of an instrument to assess patients’ attitudes toward pain. Journal of Psychosomatic Research, 31, 393–400.
Johannes, C. B., Le, T. K., Zhou, X., Johnston, J. A., & Dworkin, R. H. (2010). The prevalence of chronic pain in United States adults: Results of an internet-based survey. The Journal of Pain, 11, 1230–1239.
*Jordan, M. S., Lumley, M. A., & Leisen, J. C. (1998). The relationships of cognitive coping and pain control beliefs to pain and adjustment among African-American and Caucasian women with rheumatoid arthritis. Arthritis Care and Research, 11, 80–88.
Keefe, F. J., Affleck, G., France, C., Emery, C., Waters, S., Caldwell, D., et al. (2004). Gender differences in pain, coping, and mood in individuals having osteoarthritic knee pain: A within-day analysis. Pain, 110, 571–577.
*Keefe, F. J., Affleck, G., Lefebvre, J. C., Starr, K., Caldwell, D. S., & Tennen, H. (1997). Pain coping strategies and coping efficacy in rheumatoid arthritis: A daily process analysis. Pain, 69, 35–42.
*Keefe, F. J., Brown, G. K., Wallston, K. A., & Caldwell, D. S. (1989). Coping with rheumatoid arthritis pain: Catastrophizing as a maladaptive strategy. Pain, 37, 51–56.
*Keefe, F. J., Lefebvre, J. C., Egert, J., Affleck, G., Sullivan, M. J., & Caldwell, D. S. (2000). The relationship of gender to pain, pain behavior, and disability in osteoarthritis patients: The role of catastrophizing. Pain, 87, 325–334.
Kerns, R. D., Turk, D. C., & Rudy, T. E. (1985). The west haven-yale multidimensional pain inventory (WHYMPI). Pain, 23, 345–356.
Khan, C. M., Iida, M., Stephens, M. A., Fekete, E. M., Druley, J. A., & Greene, K. A. (2009). Spousal support following knee surgery: Roles of self-efficacy and perceived emotional responsiveness. Rehabilitation Psychology, 54, 28–32.
Kline, R. B. (1998). Principles and practice of structural equation modeling. New York: Guilford Press.
*Knittle, K. P., De Gucht, V., Hurkmans, E. J., Vlieland, T., Peeters, A. J., Ronday, H. K., et al. (2011). Effect of self-efficacy and physical activity goal achievement on arthritis pain and quality of life in patients with rheumatoid arthritis. Arthritis Care and Research, 63, 1613–1619.
Kotlarz, H., Gunnarsson, C. L., Fang, H., & Rizzo, J. A. (2009). Insurer and out-of-pocket costs of osteoarthritis in the U.S.: Evidence from national survey data. Arthritis and Rheumatism, 60, 3546–3553.
Kourilovitch, M., Galarza-Maldonado, C., & Ortiz-Prado, E. (2014). Diagnosis and classification of rheumatoid arthritis. Journal of Autoimmunity, 48, 26–30.
Kwan, Y., Koh, E., Leong, K. P., & Wee, H. L. (2014). Association between helplessness, disability, and disease activity with health-related quality of life among rheumatoid arthritis patients in a multiethnic Asian population. Rheumatology International, 34, 1085–1093.
*Kwissa-Gajewska, Z., Olesińska, M., & Tomkiewicz, A. (2014). Optimism, pain coping strategies and pain intensity among women with rheumatoid arthritis. Reumatologia, 52, 166–171.
Lafeber, F. P. J. G., & Van Spil, W. E. (2013). Osteoarthritis year 2013 in review: Biomarkers; reflecting before moving forward, one step at a time. Osteoarthritis and Cartilage, 21, 1452–1464.
*Lee, Y. C., Cui, J., Lu, B., Frits, M. L., Iannaccone, C. K., Shadick, N. A., et al. (2011). Pain persists in DAS28 rheumatoid arthritis remission but not in ACR/EULAR remission: A longitudinal observational study. Arthritis Research and Therapy, 13, 173–174.
Leeuw, M., Goossens, M. E., Linton, S. J., Crombez, G., Boersma, K., & Vlaeyen, J. W. (2007). The fear-avoidance model of musculoskeletal pain: Current state of scientific evidence. Journal of Behavioral Medicine, 30, 77–94.
*Lefebvre, J. C., Keefe, F. J., Affleck, G., Raezer, L. B., Starr, K., Caldwell, D. S., et al. (1999). The relationship of arthritis self-efficacy to daily pain, daily mood, and daily pain coping in rheumatoid arthritis patients. Pain, 80, 425–435.
*Lopez-Lopez, A., Montorio, I., Izal, M., & Velasco, L. (2008). The role of psychological variables in explaining depression in older people with chronic pain. Aging and Mental Health, 12, 735–745.
*Lopez-Olivo, M. A., Landon, G. C., Siff, S. J., Edelstein, D., Pak, C., Kallen, M. A., et al. (2011). Psychosocial determinants of outcomes in knee replacement. Annals of the Rheumatic Diseases, 70, 1775–1781.
Lorig, K., Chastain, R. L., Ung, E., Shoor, S., & Holman, H. R. (1989). Development and evaluation of a scale to measure perceived self-efficacy in people with arthritis. Arthritis and Rheumatism, 32, 37–44.
Lorig, K., & Holman, H. (2003). Self-management education: History, definitions, outcomes and mechanisms. Annals of Behavioral Medicine, 26, 1–7.
*Lowe, R., Cockshott, Z., Greenwood, R., Kirwan, J. R., Almeida, C., Richards, P., et al. (2008). Self-efficacy as an appraisal that moderates the coping-emotion relationship: Associations among people with rheumatoid arthritis. Psychology and Health, 23, 155–174.
Lurie, J. (2000). A review of generic health status measures in patients with low back pain. Spine, 25, 3125–3129.
*Mäkeläinen, P., Vehviläinen-Julkunen, K., & Pietilä, A. (2008). A survey of rheumatoid arthritis patients’ self-efficacy. Internet Journal of Advanced Nursing Practice, 9, 2058–2065.
*Maly, M. R., Costigan, P. A., & Olney, S. J. (2005). Contribution of psychosocial and mechanical variables to physical performance measures in knee osteoarthritis. Physical Therapy, 85, 1318–1328.
*Maly, M. R., Costigan, P. A., & Olney, S. J. (2006a). Determinants of self-report outcome measures in people with knee osteoarthritis. Archives of Physical Medicine and Rehabilitation, 87, 96–104.
*Maly, M. R., Costigan, P. A., & Olney, S. J. (2006b). Role of knee kinematics and kinetics on per-formance and disability in people with medial compartment knee osteoarthritis. Clinical Biomechanics, 21, 1051–1059.
*Maly, M. R., Costigan, P. A., & Olney, S. J. (2007). Self-efficacy mediates walking performance in older adults with knee osteoarthritis. Journals of Gerontology, 62, 1142–1146.
*Marcum, Z. A., Zhan, H. L., Perera, S., Moore, C. G., Fitzgerald, G. K., & Weiner, D. K. (2014). Correlates of gait speed in advanced knee osteoarthritis. Pain Medicine, 15, 1334–1342.
Margaretten, M., Julian, L., Katz, P., & Yelin, E. (2011). Depression in patients with rheumatoid arthritis: Description, causes and mechanisms. International Journal of Clinical Rheumatology, 6, 617–623.
*McKnight, P. E., Afram, A., Kashdan, T. B., Kasle, S., & Zautra, A. (2010). Coping self-efficacy as a mediator between catastrophizing and physical functioning: Treatment target selection in an osteoarthritis sample. Journal of Behavioral Medicine, 33, 239–249.
Memel, D. S., Kirwan, J. R., Sharp, D. J., & Hehir, M. (2000). General practitioners miss disability and anxiety as well as depression in their patients with osteoarthritis. British Journal of General Practice, 50, 645–650.
*Mielenz, T., Kubiak-Rizzone, K., Alvarez, K., Hlavacek, P., Freburger, J., Giuliani, C., et al. (2013). Association of self-efficacy and outcome expectations with physical activity in adults with arthritis. Arthritis. doi:10.1155/2013/621396
*Miller, C., & Cronan, T. (1998). The effects of coping style and self-efficacy on health status and health care costs. Anxiety, Stress and Coping, 11, 311–325.
*Morone, N. E., Karp, J. F., Lynch, C. S., Bost, J., El Khoudary, S., & Weiner, D. K. (2009). Impact of chronic musculoskeletal pathology on older adults: a study of differences between knee OA and low back pain. Pain Medicine, 10, 693–701.
Murphy, H., Dickens, C., Creed, F., & Bernstein, R. (1999). Depression, illness perception and coping in rheumatoid arthritis. Journal of Psychosomatic Research, 46, 155–164.
Murphy, L. B., Sacks, J. J., Brady, T. J., Hootman, J. M., & Chapman, D. P. (2012). Anxiety and depression among US adults with arthritis: Prevalence and correlates. Arthritis Care and Research, 64, 968–976.
*Nadrian, H., Morowatisharifabad, M. A., & Bahmanpour, K. (2011). Development of a rheumatoid arthritis education program using the precede-proceed model. Health Promotion Perspectives, 1, 118–129.
Nanda, U., McLendon, P. M., Andresen, E. M., & Armbrecht, E. (2003). The SIP68: An abbreviated sickness impact profile for disability outcomes research. Quality of Life Research, 12, 583–595.
Neogi, T. (2013). The epidemiology and impact of pain in osteoarthritis. Osteoarthritis and Cartilage, 21, 1145–1153.
Nicassio, P. M., Wallston, K. A., Callahan, L. F., Herbert, M., & Pincus, T. (1985). The measurement of helplessness in rheumatoid arthritis. The development of the arthritis helplessness index. The Journal of Rheumatology, 12, 462–467.
O’Donnell, S., Lagacé, C., McRae, L., & Bancej, C. (2010). Life with arthritis in Canada: A personal and public health challenge. Chronic Diseases and Injuries in Canada, 31, 135–136.
*Olchowska-Kotala, Z., Olisinska, M., & Tomkiewicz, A. (2014). Optimism, pain coping strategies and pain intensity among women with rheumatoid arthritis. Reumatologia, 52, 166–171.
*Pells, J. J., Shelby, R. A., Keefe, F. J., Dixon, K. E., Blumenthal, J. A., LaCaille, L., et al. (2008). Arthritis self-efficacy and self-efficacy for resisting eating: Relationships to pain, disability, and eating behavior in overweight and obese individuals with osteoarthritic knee pain. Pain, 136, 340–347.
*Penhoat, M., Saraux, A., Goff, B. L., Augereau, P., Maugars, Y., & Berthelot, J. (2014). High pain catastrophizing scores in one-fourth of patients on biotherapy for spondylarthritis or rheumatoid arthritis. Joint Bone Spine, 81, 235–239.
*Prior, K. N., & Bond, M. J. (2004). The roles of self-efficacy and abnormal illness behaviour in osteoarthritis self-management. Psychology, Health and Medicine, 9, 177–192.
*Rayahin, J. E., Chmiel, J. S., Hayes, K. W., Almagor, O., Belisle, L., Chang, A. H., et al. (2014). Factors associated with pain experience outcome in knee osteoarthritis. Arthritis Care and Research, 66, 1828–1835.
*Regan, C. A., Lorig, K., & Thoresen, C. E. (1988). Arthritis appraisal and ways of coping. Scale development. Arthritis and Rheumatism, 1, 139–150.
*Reinseth, L., Uhlig, T., Kjeken, I., Koksvik, H. S., Skomsvoll, J. F., & Espnes, G. A. (2011). Performance in leisure-time physical activities and self-efficacy in females with rheumatoid arthritis. Scandinavian Journal of Occupational Therapy, 18, 210–218.
*Rejeski, W. J., Craven, T., Ettinger, W. H., McFarlane, M., & Shumaker, S. (1996). Self-efficacy and pain in disability with osteoarthritis of the knee. Journal of Gerontology, 51B, P24–P29.
*Rejeski, W. J., Martin, K. A., Miller, M. E., Ettinger, W. H., & Rapp, S. (1998). Perceived difficulty, importance and satisfaction with physical function in patients with knee osteoarthritis. Annals of Behavioral Medicine, 20, 141–148.
*Rejeski, W. J., & Mihalko, S. (2001). Physical activity and quality of life in older adults. Journal of Gerontology, 56A, 23–35.
*Rezaei, F., Neshat-Doost, H. T., Molavi, H., Abedi, M. R., & Karimifar, M. (2014). Depression and pain in patients with rheumatoid arthritis: Mediating role of illness perception. The Egyptian Rheumatologist, 36, 57–64.
Rosentiel, A. K., & Keefe, F. J. (1983). The use of coping strategies in chronic low back pain patients: relationship to patient characteristics and current adjustment. Pain, 17, 33–44.
Rotter, J. B. (1966). Generalized expectancies for internal versus external control of reinforcement. Psychological Monographs, 80, 1–28.
Sale, J. E., Gignac, M., & Hawker, G. (2008). The relationship between disease symptoms, life events, coping and treatment, and depression among older adults with osteoarthritis. The Journal of Rheumatology, 35, 335–342.
*Scharloo, M., Kaptein, A. A., Weinman, J., Hazes, J. M., Willems, L. N. A., Bergman, W., et al. (1998). Illness perceptions, coping and functioning in patients with rheumatoid arthritis, chronic obstructive pulmonary disease and psoriasis. Journal of Psychosomatic Research, 44, 573–585.
*Schiaffino, K. M., & Revenson, T. A. (1992). The role of perceived self-efficacy, perceived control, and causal attributions in adaptation to rheumatoid arthritis: Distinguishing mediator vs. moderator effects. Personality and Social Psychology Bulletin, 18, 709–718.
*Schiaffino, K. M., & Revenson, T. A. (1995a). Relative contribution of spousal support and illness appraisals to depressed mood in arthritis patients. Arthritis Care and Research, 8, 80–87.
*Schiaffino, K. M., & Revenson, T. A. (1995b). Why me? The persistence of positive and negative appraisals over the course of illness. Journal of Applied Social Psychology, 25, 601–618.
*Schiaffino, K. M., Revenson, T. A., & Gibofsky, A. (1991). Assessing the impact of self-efficacy beliefs on adaptation to rheumatoid arthritis. Arthritis Care and Research, 4, 150–157.
*Schoenfeld-Smith, K., Petroski, G. F., Hewett, J. E., Johnson, J. C., Wright, G. E., Smarr, K. L., et al. (1996). A biopsychosocial model of disability in rheumatoid arthritis. Arthritis and Rheumatism, 9, 368–375.
*Scopaz, K. A., Piva, S. R., Wisniewski, S., & Fitzgerald, G. K. (2009). Relationships of fear, anxiety, and depression with physical function in patients with knee osteoarthritis. Archives of Physical Medicine and Rehabilitation, 90, 1866–1873.
Seligman, M. E. P. (1975). Helplessness: On depression, development and death. San Francisco: W. H. Freeman.
*Šerbo, B., & Jajić, I. (1991). Relationship of the functional status, duration of the disease and pain intensity and some psychological variables in patients with rheumatoid arthritis. Clinical Rheumatology, 10, 419–422.
*Sharma, L., Cahue, S., Song, J., Hayes, K., Pai, Y. C., & Dunlop, D. (2003). Physical functioning over three years in knee osteoarthritis: role of psychosocial, local mechanical, and neuromuscular factors. Arthritis and Rheumatism, 48, 3359–3370.
*Shelby, R. A., Somers, T. J., Keefe, F. J., Pells, J. J., Dixon, K. E., & Blumenthal, J. A. (2008). Domain specific self-efficacy mediates the impact of pain catastrophizing on pain and disability in overweight and obese osteoarthritis patients. The Journal of Pain, 9, 912–919.
*Shifren, K., Park, D. C., Bennett, J. M., & Morrell, R. W. (1999). Do cognitive processes predict mental health in individuals with rheumatoid arthritis? Journal of Behavioral Medicine, 22, 529–547.
Silman, A., Hochberg, M. C., Smolen, J. S., Weinblatt, M. E., & Weisman, M. H. (2009). Descriptive epidemiology of rheumatoid arthritis. In M. C. Hochberg & A. J. Silman (Eds.), Rheumatoid arthritis (pp. 15–22). Philadelphia: Elsevier.
Sinclair, V. G., & Wallston, K. A. (2010). Psychological vulnerability predicts increases in depressive symptoms in individuals with rheumatoid arthritis. Nursing Research, 59, 140–146.
Skevington, S. M. (1990). A standardised scale to measure beliefs about controlling pain (BPCQ): A preliminary study. Psychology and Health, 4, 221–232.
Smarr, K. L., Parker, J. C., Wright, G. E., Stucky-Ropp, R. C., Buckelew, S. P., Hoffman, R. W., et al. (1997). The importance of enhancing self-efficacy in rheumatoid arthritis. Arthritis Care and Research, 10, 18–26.
*Smith, T. W., Christensen, A. J., Peck, J. R., & Ward, J. R. (1994). Cognitive distortion, helplessness, and depressed mood in rheumatoid arthritis: A four-year longitudinal analysis. Health Psychology, 13, 213–217.
*Smith, C. A., Dobbins, C. J., & Wallston, K. A. (1991). The mediational role of perceived competence in psychological adjustment to rheumatoid arthritis. Journal of Applied Social Psychology, 21, 1218–1247.
*Smith, T. W., Peck, J. R., & Ward, J. R. (1990). Helplessness and depression in rheumatoid arthritis. Health Psychology, 9, 377–389.
*Smith, C. A., & Wallston, K. A. (1992). Adaptation in patients with chronic rheumatoid arthritis: Application of a general model. Health Psychology, 11, 151–162.
*Somers, T. J., Keefe, F. J., Pells, J. J., Dixon, K. E., Waters, S. J., Riordan, P. A., et al. (2009). Pain catastrophizing and pain-related fear in osteoarthritis patients: Relationships to pain and disability. Journal of Pain and Symptom Management, 37, 863–872.
*Somers, T. J., Shelby, R. A., Keefe, F. J., Godiwala, N., Lumley, M. A., Mosley-Williams, A., et al. (2010). Disease severity and domain-specific arthritis self-efficacy: Relationships to pain and functioning in patients with rheumatoid arthritis. Arthritis Care and Research, 62, 848–856.
Somers, T., Wren, A. A., Blumenthal, J. A., Caldwell, D., Huffman, K. M., & Keefe, F. J. (2014). Pain, physical functioning, and overeating in obese rheumatoid arthritis patients: Do thoughts about pain and eating matter? Journal of Clinical Rheumatology, 20, 244–250.
Stein, M. J., Wallston, K. A., & Nicassio, P. M. (1988a). Factor structure of the arthritis helplessness index. The Journal of Rheumatology, 15, 427–432.
Stein, M. J., Wallston, K. A., Nicassio, P. M., & Castner, N. M. (1988b). Correlates of a clinical classification schema for the arthritis helplessness subscale. Arthritis and Rheumatism, 31, 876–881.
Steultjens, M. P. M., Dekker, J., & Bijlsma, J. W. J. (2002). Avoidance of activity and disability in patients with osteoarthritis of the knee: The mediating role of muscle strength. Arthritis and Rheumatism, 46, 1784–1788.
*Strahl, C., Kleinknecht, R. A., & Dinnel, D. L. (2000). The role of pain anxiety, coping, and pain self-efficacy in rheumatoid arthritis patient functioning. Behaviour Research and Therapy, 38, 863–873.
*Sturgeon, J. A., Zautra, A. J., & Arewasikporn, A. (2014). A multilevel structural equation modeling analysis of vulnerabilities and resilience resources influencing affective adaptation to chronic pain. Pain, 155, 292–298.
Sturmer, T., & Hasselbach, P. (2006). Personality, lifestyle, and risk of cardiovascular disease and cancer: Follow-up of population based cohort. British Medical Journal, 332, 1359.
Sullivan, M. J., Bishop, S. R., & Pivik, J. (1995). The pain catastrophizing scale: Development and validation. Psychological Assessment, 7, 524–532.
*Sullivan, M., Tanzer, M., Reardon, G., Amirault, D., Dunbar, M., & Stanish, W. (2011). The role of presurgical expectancies in predicting pain and function one year following total knee arthroplasty. Pain, 152, 2287–2293.
*Taal, E., Rasker, J. J., Seydel, E., & Wiegman, O. (1993). Health status, adherence with health recommendations, self-efficacy and social support in patients with rheumatoid arthritis. Patient Education and Counseling, 20, 63–76.
*Tennen, H., Affleck, G., Urrows, S., Higgins, P., & Mendola, R. (1992). Perceiving control, construing benefits, and daily processes in rheumatoid arthritis. Canadian Journal of Behavioral Sciences, 24, 186–203.
*Treharne, G., Kitas, G. D., Lyons, A. C., & Booth, D. A. (2005). Well-being in rheumatoid arthritis: The effects of disease duration and psychosocial factors. Journal of Health Psychology, 10, 457–474.
*van den Akker-Scheek, I., Stevens, M., Groothoff, J. W., Bulstra, S. K., & Zijlstra, W. (2007). Preoperative or postoperative self-efficacy: Which is a better predictor of outcome after total hip or knee arthroplasty? Patient Education and Counseling, 66, 92–99.
*Van Liew, C., Santoro, M. S., Chalfant, A. K., Gade, S., Casteel, D. L., Tomita, M., et al. (2013). The good life: Assessing the relative importance of physical, psychological, and self-efficacy statuses on quality of well-being in osteoarthritis patients. Arthritis. doi:10.1155/2013/914216
Villeneuve, E. (2013). To screen or not to screen: How to find and identify very early arthritis. Best Practice and Research Clinical Rheumatology, 27, 487–497.
Vlaeyen, J. W. S., Kole-Snijders, A. M. J., Boeren, R. G. B., & van Eek, H. (1995). Fear of movement/(re)injury in chronic low back pain and its relation to behavioural performance. Pain, 62, 363–372.
Vlaeyen, J. W., & Linton, S. J. (2000). Fear-avoidance and its consequences in chronic musculoskeletal pain: A state of the art. Pain, 85, 317–332.
Waddell, G., Newton, M., Henderson, I., Somerville, D., & Main, C. J. (1993). A Fear-Avoidance Beliefs Questionnaire (FABQ) and the role of fear-avoidance beliefs in chronic low back pain and disability. Pain, 52, 157–168.
Wallston, K. A. (1992). Hocus-pocus, the focus isn’t strictly on locus: Rotter’s social learning theory modified for health. Cognitive Therapy and Research, 16, 183–199.
Wallston, K. A. (2005). The validity of the multidimensional health locus of control scales. Journal of Health Psychology, 10, 623–631.
*Wallston, K. A., Stein, M. J., & Smith, C. A. (1994). Form C of the MHLC scales: A condition-specific measure of locus of control. Journal of Personality Assessment, 63, 534–553.
Weinman, J., Petrie, K. J., Moss-Morris, R., & Horne, R. (1996). The illness perception questionnaire: A new method for assessing the cognitive representation of illness. Psychology and Health, 11, 431–445.
White, A. G., Birnbaum, H. G., Buteau, S., Janagap, C., & Schein, J. R. (2007). Cost of pain therapy for osteoarthritis in a privately insured population in the United States. Value in Health, 10, A117.
Wideman, T. H., Asmundson, G. G., Smeets, R. J., Zautra, A. J., Simmonds, M. J., Sullivan, M. J., et al. (2013). Rethinking the fear avoidance model: Toward a multidimensional framework of pain-related disability. Pain, 154, 2262–2265.
*Wilcox, S., Schoffman, D. E., Dowda, M., & Sharpe, P. A. (2014). Psychometric properties of the 8-item English arthritis self-efficacy scale in a diverse sample. Arthritis. doi:10.1155/2014/385256
World Health Organization. (1993). Report of WHOQOL focus group work. Geneva: WHO.
*Wright, G. E., Parker, J. C., Smarr, K. L., Schoenfeld-Smith, K., Buckelew, S. P., Slaughter, J. R., et al. (1996). Risk factors for depression in rheumatoid arthritis. Arthritis and Rheumatism, 9, 264–272.
*Wright, L. J., Zautra, A. J., & Going, S. (2008). Adaptation to early knee osteoarthritis: The role of risk, resilience, and disease severity on pain and physical functioning. Annals of Behavioral Medicine, 36, 70–80.
*Wylde, V., Dixon, S., & Blom, A. W. (2012). The role of preoperative self-efficacy in predicting outcome after total knee replacement. Musculoskeletal Care, 10, 110–118.
Zale, E., Lange, K., Fields, S., & Ditre, J. (2013). The relation between pain-related fear and disability: A meta-analysis. The Journal of Pain, 14, 1019–1030.
*Ziarko, M., Mojs, E., Piasecki, B., & Samborski, W. (2014). The mediating role of dysfunctional coping in the relationship between beliefs about the disease and the level of depression in patients with rheumatoid arthritis. Scientific World Journal. doi:10.1155/2014/585063
*Zyrianova, Y., Kelly, B., Sheehan, J., McCarthy, C., & Dinan, T. G. (2011). The psychological impact of arthritis: the effects of illness perception and coping. Irish Journal of Medical Science, 180, 203–210.
Funding
This study was funded by the National Natural Science Foundation of China (#31371037), Education Ministry of China, and a Chongqing 100 Persons Fellowship to the corresponding author.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Xiaojun Jia and Todd Jackson declare that they do not have any conflict of interest.
Human and animal rights and Informed consent
All procedures followed were in accordance with ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jia, X., Jackson, T. Pain beliefs and problems in functioning among people with arthritis: a meta-analytic review. J Behav Med 39, 735–756 (2016). https://doi.org/10.1007/s10865-016-9777-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10865-016-9777-z