Abstract
For the first time, a hierarchical phosphor Y2O3:Eu3+/MCM-41/YVO4:Eu3+, with a core–shell-core heterostructure, is presented in this study. Synergistically bridging the phosphors Y2O3:Eu3+ (as an inner core) and YVO4:Eu3+ (as an outer core) by amorphous SiO2, i.e., MCM-41 (with ordered mesoporous channels) leads to the generation of the core–shell-core heterostructure with enhanced red emission and tunable, broadened-band response to excitation. The novel structure of the core–shell-core hierarchical material is clarified through various characterization methods including X-ray diffraction analysis, transmission electron microscopy, selected-area electron diffraction and N2 adsorption–desorption measurements. Significantly, through temperature-dependent fluorescence investigation, it is found that our core–shell phosphor (Y2O3:Eu3+/MCM-41) exhibits impressive fluorescence stability against temperature variation (27–227 °C) due to the protective effect resulting from MCM-41. By contrast, lowered stability can be noted for the core–shell-core phosphor (Y2O3:Eu3+/MCM-41/YVO4:Eu3+), especially when the temperature is higher than 100 °C, owing to the outer core (YVO4:Eu3+ nanoparticles) that is directly exposed to heat. Such a kind of luminescent materials holds substantial promise for labeling the organisms that are vulnerable to short-wavelength UV light irradiation. Additionally, potential intelligent systems can be expected to be designed on the basis of the fluorescence mutation as triggered by the temperature of 100 °C.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Engineering a material into a core–shell hierarchical heterostructure has attracted extensive attention owing to the capability of fine-tuning physicochemical properties for various applications, including catalysis [1], photocatalysis [2], luminescence [3], photothermal conversion [4], lithium ion batteries [5], and ion separation [6], etc [7,8,9]. It is generally recognized that the formation of a shell on a core enables the protection of the core as an active center, improving the stability of core materials, especially active nanomaterials, e.g., a nanocatalyst [10]. Indeed, a large number of studies have demonstrated that direct synthesis of nanomaterials without a protection effect from a shell leads to the aggregation of nanomaterials and consequently performance degradation, e.g., in photocatalysis [11] and luminescence [12], of metal oxides involving calcination treatment.
Although the core–shell heterostructure engineering has been widely explored and recognized as an effective technology for synthesis of high-performance hierarchical structures, the core–shell-core hierarchical heterostructures with double cores are rarely investigated. For the first time, this paper presents the synthesis of a core–shell-core hierarchical heterostructure for luminescence applications [13,14,15,16,17,18,19,20,21,22,23,24]. The phosphor was effectively engineered into a well-constructed core–shell-core heterostructure Y2O3:Eu3+/MCM-41/YVO4:Eu3+, exhibiting enhanced red emission, along with tunable, broadened-band response to excitation. The broadening of band response of the phosphor could facilitate the fluorescent labeling applications involving organisms vulnerable to UV radiation at short wavelengths, e.g., smaller than 365 nm [25, 26]. Therefore, our phosphor shows great potential to replace conventional fluorescent organic dye and quantum dots (QDs), which thereby circumvents the insurmountable problem of the conventional bioimaging techniques, including the photobleaching and quenching of organic dyes and the toxicity of QDs [27]. Additionally, there will be many other potential applications for our prepared luminescent material, such as cathode ray tubes, plasma display panels, projection television screens, light-emitting diodes, and field emission displays [28,29,30,31,32,33,34,35,36,37,38,39].
The present synthesis of such a phosphor takes advantage of the fascinating properties (such as homogeneous mesopore distribution and satisfactory thermal stability) of a mesoporous molecule sieve working as a bridging material, in this case MCM-41. It possesses abundant hydroxyl groups on its surface, which offers an excellent platform for further modification and also facilitates the host–guest assembly chemistry [40,41,42]. Furthermore, its mesoporous structure endues tunable optical properties since the energy transfer of the emission activator [43] (namely Eu3+) of our phosphor can be varied with the extension direction along the pore walls and channels, providing a way for unraveling the photoluminescence mechanism. The hosts Y2O3 and YVO4 were selected because of suitable crystallographic sites for Eu3+ ion doping in the crystals [44]. This study aims to explore a novel core–shell-core hierarchical heterostructure Y2O3:Eu3+/MCM-41/YVO4:Eu3+, along with its photoluminescence properties. An understanding of the energy transfer and phosphorescence mechanism is investigated as well.
2 Experimental
2.1 Materials
Tetraethoxysilane (TEOS, Si(OC2H5)4), tetrabutyl ammonium bromide (TBAB, C16H36BrN) and cetyl trimethyl ammonium bromide (CTMAB, C16H33(CH3)3NBr), of AR grade, were supplied from Guanghua Chemical Factory (Shantou, China), Bodi Chemical Co., Ltd. (Tianjin, China), and Fuchen Chemical Reagents Factory (Tianjin, China), respectively. Y2O3 and Eu2O3 (high-purity grade, 99.999%) were obtained from Zhujiang Refinery Factory (Guangdong, China). All other chemicals were AR grade and used without further purification.
2.2 Synthesis of a (Y,Eu)(OH)3 hydrosol as a precursor of the Y2O3:Eu3+ core
According to a homogeneous co-precipitation method, the precursor (Y,Eu)(OH)3 was firstly synthesized using urea as a precipitation agent. A mixed solution (pH 4.3) of urea (2 mol/L), Y(NO3)3 (0.04 mol/L) and Eu(NO3)3 (0.002 mol/L) were agitated at 80 °C. When the solution turned out to be turbid, the reaction was stopped after a further 1 h reaction. The product was then collected by filtration, followed by through washing with deionized (DI) water and by re-dispersing, leading to the (Y,Eu)(OH)3-based hydrosol. The preparation of the Y3+(Eu3+)-based nitrates was described as follows: a given amount of Y2O3 and Eu2O3 was mixed to concentrated nitric acid in an excess quantity. The excessed nitric acid was volatilized by heating treatment. After cooling to room temperature, DI water was mixed to generate the final Y(NO3)3 (0.04 mol/L) and Eu(NO3)3 (0.002 mol/L) mixed nitrate solution.
2.3 Synthesis of core–shell heterostructure Y2O3:Eu3+/MCM-41
To prevent the dissolution of the core Y2O3:Eu3+ during imbedding YVO4:Eu3+ as the second core material, the surface of the nanoparticles in the (Y,Eu)(OH)3-based hydrosol was coated with a thin layer of SiO2, before further encapsulation with MCM-41, as schematically illustrated in Fig. 1. The precursor of Y2O3:Eu3+ (namely (Y,Eu)(OH)3, 1.0 g) was treated in 20 mL of ethanol by ultrasonication for 15 min, and then 3 mL of ammonia (25 wt%) was added as a catalyst. Under continuous stirring, 2.8 mL of TEOS was dropwise added, and a 1.5 h reaction at room temperature was conducted. After that, the product was collected by filtration, thoroughly washed with DI water three times, and finally dried at 90 °C for 12 h. The obtained dried product was then re-dispersed into 60.0 mL of DI water using 9.0 g of ammonia (25 wt%) as the catalyst. Separately, 0.6125 g of TBAB was dissolved into 10.0 mL of DI water. Similarly, CTMAB (2.7698 g) was mixed into 50.0 mL of DI water. All the three solutions were subsequently mixed together under stirring for 0.5 h, which was then agitated intensely, along with dropwise adding 10.48 g of TEOS. The time used for the addition of TEOS lasted for 1.5 h. Upon completion of the TEOS addition, the reaction further proceeded for 3 h at room temperature. The molar ratio of the reactants, namely CTMAB/TEOS/ammonia/TBAB, was calculated to be 0.152/1/2.8/141.2/0.038. The product was filtered, washed with DI water, and then dried at 363 °C for 12 h. The dried product in the powder form was employed as the precursor of Y2O3:Eu3+/MCM-41, which was placed in a muffle furnace for calcination treatment. The programming temperature was set as follows: a temperature ramp of 1.0 °C/min from room temperature to a final temperature of 823.0 °C, which was held for 9 h to produce the final Y2O3:Eu3+/MCM-41.
Schematic illustration of the synthesis of the present core–shell (Y2O3:Eu3+/MCM-41) and core–shell-core (Y2O3:Eu3+/MCM-41/YVO4:Eu3+) hierarchical heterostructures. The effective synthesis mainly includes the following three steps: (i) formation of protective SiO2 shell on (Y,Eu)(OH)3, (ii, iii) generation of the core–shell (ii) and core–shell-core (iii) phosphors
2.4 Synthesis of core–shell-core heterostructure Y2O3:Eu3+/MCM-41/YVO4:Eu3+
A given amount of NH4VO3 was dissolved into ammonia with a mass ratio of 1:1 by heating treatment, leading to the generation of a clear solution. A mixed solutions of Y(NO3)3 and Eu(NO3)3 was homogenized and then added into the solution of NH4VO3 (pH ~ 4), and agitated for 0.5 h to form a hydrosol in orange color. The as-synthesized Y2O3:Eu3+/MCM-41 was then added to the hydrosol under magnetic stirring for 2 h. The product was collected by filtration, drying treatment at 90 °C for 12 h, and calcination at 550 °C, resulting in the core–shell-core heterostructured phosphor Y2O3:Eu3+/MCM-41/YVO4:Eu3+.
2.5 Characterization
The X-ray diffraction (XRD) patterns of the synthesized samples in the powder form were inspected on a Dandong-aolong Y-2000 diffractometer using Cu Kα radiation at λ = 0.15418 nm with a single-crystal silicon powder as an external standard. The structure and morphology of the samples were examined using a high resolution transmission electron microscope (HR-TEM, JEOL JEM 2010). The excitation and emission spectra were recorded on a Hitachi F-2500 spectrofluorimeter equipped with a 150 W xenon lamp as the excitation source. All the measurements were carried out at room temperature. Low-temperature N2 adsorption–desorption isotherms were recorded on a Micromertics ASAP 2020 M system at liquid nitrogen temperature. Before the measurement, the sample was degassed in vacuum at 90 °C for 1 h, and then vacuum-dried at 350 °C for 8 h (less than 5 × 10−3 mmHg of vacuum degree).
3 Results and discussion
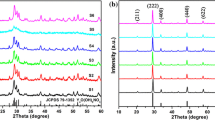
Figure 1 presents the schematic illustration of the synthesis process of core–shell and core–shell-core heterostructures Y2O3:Eu3+/MCM-41 and Y2O3:Eu3+/MCM-41/YVO4:Eu3+, respectively. For the synthesized heterostructures, their XRD patterns are shown in Figs. 2 and 3. For both core–shell (see Fig. 2a) and core–shell-core (see Fig. 2b) phosphors, the diffraction angles (2θ) appear at 28.9°, 33.7°, 48.3° and 57.2°, corresponding to (222), (400), (440), and (622) crystallographic planes of Y2O3 (in a body-centered cubic structure) respectively (JCPDS No. 25-1011) [45]. An amorphous broad peak at 22° can be an indication that the pore channels of the mesoporous molecule sieve are made from amorphous SiO2 [46, 47]. As for core–shell-core heterostructure Y2O3:Eu3+/MCM-41/YVO4:Eu3+, XRD peaks emerge at 2θ ≈ 24.6° and 49.5°. This characteristic of diffraction bands can be indexed to (200) and (312) crystallographic planes of YVO4 (with a quartet zircon structure) respectively (JCPDS No.17–0341) [48, 49], as displayed in Fig. 2b. It is also found that the characteristic XRD bands derived from Y2O3 remain almost unchanged after imbedding the second core YVO4:Eu3+ into Y2O3:Eu3+/MCM-41.
Small angle X-ray diffraction was also employed to further characterize the core–shell-core heterostructure (see Fig. 3). Apart from two small (110) and (200) diffraction peaks of MCM-41, a strong (100) diffraction band can be observed around 2.35° [50]. According to the Brag’s law, i.e., d 100 = λ/2sinθ, the spacing d 100 can be calculated as 3.76 nm, which is consistent with the literature [51]. Such a strong (100) plane diffraction band is characteristic of mesoporous materials with a hexagonal close-packed structure, which also reveals that the formation of periodically aligned liquid crystal and texture takes places through self-assembly interactions between inorganic silicate and surfactant ions. This result verifies that the formed inorganic skeleton of the shell possesses the hexagonal mesoporous structure typical of MCM-41 [50]. On the other hand, the incorporation of YVO4:Eu3+ into Y2O3:Eu3+/MCM-41 largely broadens the (100) diffraction peak (Fig. 3), suggesting a significant loss of the hexagonal mesostructure order due to the blocking of the mesopores as a result of deposition of the phosphor YVO4:Eu3+ [50, 52].
To further investigate the structural features of the single core–shell and double core–shell heterostructures, TEM images together with the selected-area electron diffraction (SAED) patterns are presented in Fig. 4. Both the core–shell (Fig. 4a) and core–shell-core (Fig. 4d) phosphors exhibit a sphere-like core–shell structure, with ordered hexagonal arrays of mesoporous channels. Such ordered hexagonal arrays can be more clearly observed as for the core–shell phosphor Y2O3:Eu3+/MCM-41 (Fig. 4c) relative to that for the core–shell-core phosphor Y2O3:Eu3+/MCM-41/YVO4:Eu3+ (Fig. 4f); this is because the deposition of the YVO4:Eu3+ enables the hexagonal mesoporous arrays to be covered to some extent, in good consistence with the small small-angle XRD patterns.
Microstructural analysis of the phosphors Y2O3:Eu3+/MCM-41 and Y2O3:Eu3+/MCM-41/YVO4:Eu3+by means of TEM and SAED. TEM (a, c) and SAED (b) analyses of Y2O3:Eu3+/MCM-41, TEM (d, f) and SAED (e) analyses of Y2O3:Eu3+/MCM-41. c and f are the TEM images at higher magnifications in comparison with (a) and (d), respectively
The crystalline phase (indexed to Y2O3:Eu3+) can be well evidenced by the SAED pattern of Y2O3:Eu3+/MCM-41 shown in Fig. 4b. Ordered diffraction spots can be unambiguously seen, indicating the high lattice ordering of Y2O3:Eu3+ crystalline particles that exist in the single-crystalline nature [53, 54]. On the other hand, the SAED pattern of Y2O3:Eu3+/MCM-41/YVO4:Eu3+features the concentric rings with dispersed spots (Fig. 4e), which is an indication of the polycrystalline nature of YVO4:Eu3+ nanoparticles strictly confined in the mesoporous channels of the MCM-41 shell [54]. The thickness of the bridging shell MCM-41 is approximately 20 nm. Such a nano-thin shell implies the imbedded YVO4:Eu3+ particles at a nanoscale even an atomic scale (Fig. 4d). Despite the deposition of YVO4:Eu3+ onto the core–shell phosphor Y2O3:Eu3+/MCM-41, the ordered mesoporous structure of the core–shell-core heterostructure Y2O3:Eu3+/MCM-41/YVO4:Eu3+ can still be noted, without observable large YVO4:Eu3+ aggregates (Fig. 4d), revealing uniform deposition of YVO4:Eu3+ nanoparticles into the mesopore channels of the MCM-41 shell. Furthermore, from the TEM images at a higher magnification (Fig. 4f), the lattice fringes of the crystalline phase (YVO4) are distinguishable; the distance between the adjacent lattice fringes can be calculated as approximately 0.35 nm, corresponding well to the interplanar distance of YVO4 (200) crystallographic planes [50]. This result is consistent with the values obtained from JCPDS 17–0341 and with our XRD analysis.
The surface area and pore size distribution of our prepared core–shell and core–shell-core heterostructures are further probed by N2 adsorption–desorption isotherms (Fig. 5). Both the isotherms feature Langmuir IV adsorption, with H1 hysteresis loop; this is a typical adsorption–desorption curve of mesoporous material, indicating an ordered mesoporous composition of the shell material [8, 55, 56]. At a low partial pressure range (P/P0 < 0.3), the adsorption amount of N2 exhibits a linear increase with increasing the partial pressure, which can be due to the monolayer adsorption of N2 on the mesopore walls. As the partial pressure further raises beyond P/P0 of 0.3, the adsorption amount is apparently increased, as a result of the capillary condensation as generated by the N2 adsorbed in the mesoporous skeleton. With ongoing increase of the partial pressure, the curve becomes slowly upward, indicating the saturation of the N2 adsorption in the capillary. When the P/P0 is close to 1, a steep raise of the N2 adsorption can be noted, implying the filling of large pores; this is associated with the capillary condensation of interparticle pores and channels. The main information read from the N2 adsorption–desorption isotherms is also summarized in Table 1. After embedding YVO4:Eu3+ into the core–shell heterostructure Y2O3:Eu3+/MCM-41, the surface area, and pore size and volume values decrease, revealing the well-dispersed YVO4:Eu3+ nanoparticles in the ordered pore channels of the mesopore SiO2 and consequent filling of a part of mesopore channels.
The luminescence properties of our prepared core–shell and core–shell-core heterostructures are finally investigated. Figure 6 presents the UV excitation spectra of the heterostructures Y2O3:Eu3+/MCM-41 and Y2O3:Eu3+/MCM-41/YVO4:Eu3+, as monitoired at 612 (Fig. 6a) and 620 (Fig. 6b) nm. For the sample Y2O3:Eu3+/MCM-41 with a core–shell structure, the absoprtion band at 256 nm can be ascribed to the charge transfer band (CBT) of Eu–O (Fig. 6a). By contrast, the sample Y2O3:Eu3+/MCM-41/YVO4:Eu3+ features the CBT-reated broad absorption in the range of 200–350 nm, centered at 254 nm. It also presents a typical VO4 3−-based CBT-related absorption at 307 nm, which is the main absorption characteristics of the spectrum. In the range of 350–450 nm, sharp absorption peaks can be noted, as generated by the f–f electron forbidden transition of Eu3+. These Eu3+-related peaks at 394, 363, 382, and 416 nm, can be attributed to 7F0→5L6, 7F0→5D4, 7F0→5G2, and 7F0→5D3 electron transitions, respectively. Concerning the sample Y2O3:Eu3+/MCM-41/YVO4:Eu3+, the excitation spectra shown in Fig. 6b display only a strong broad excitation band at 307 nm when monitored at 620 nm, suggesting that the excitation of Eu3+ mainly comes from the energy transfer from VO4 3− to Eu3+ that is embedded within the pore channels of the shell. This also reveals that VO4 3− can transfer energy to Eu3+ ions more efficiently in the mesopore channels.
Figure 7 shows the emission spectra of the samples Y2O3:Eu3+/MCM-41 and Y2O3:Eu3+/MCM-41/YVO4:Eu3+ at different excitation wavelengths of 255 and 303 nm. Upon excitation at 255 nm (Fig. 7a), the emission peak at 612 nm exhibits the highest intensity for the sample Y2O3:Eu3+/MCM-41, corresponding to the typical 5D0–7F2 electron transition of Eu3+. As for the core–shell-core sample Y2O3:Eu3+/MCM-41/YVO4:Eu3+, the biggest emission peaks are located at 616 and 620 nm (along with a shoulder peak at approximately 612 nm), corresponding to the main emission from YVO4:Eu3+. These fluorescence features can be primarily generated by the strong dependency of the emission-related ultrasensitive electron transition 5D0→7F2 on the surroundings and local symmetry of Eu3+ ions. Upon excitation at 303 nm, the emission intensity is much higher for Y2O3:Eu3+/MCM-41/YVO4:Eu3+ relative to Y2O3:Eu3+/MCM-41, as contributed by the emission from the phosphor YVO4:Eu3+. These results thus verify that embedding YVO4:Eu3+ into Y2O3:Eu3+/MCM-41 (Fig. 1) enhances the red emission performance under flexible excitation conditions. A schematic diagram is also given to show the luminescence routes upon excitation into both CTBs, i.e., Eu–O and VO4 3+, as well as the emission process of Eu3+ (Fig. 8).
To probe the impact of temperature on the fluorescence of the prepared core–shell and core–shell-core phosphors, temperature-dependent fluorescence properties were measured, with the results shown in Figs. 9 and 10. Note that the core–shell phosphor Y2O3:Eu3+/MCM-41 (Fig. 9) exhibits a significantly high fluorescence stability in the temparature range of 300–500 K, i.e., 27–227 °C. Such a stability can stem from the SiO2 and MCM-41 shells that protect Y2O3:Eu3+ from thermal quenching (Fig. 1), in addition to the high stability of Y2O3:Eu3+ as reported elsewhere [57]. The highly efficient fluorescence signal and a good thermal stability imply that our core–shell phosphor Y2O3:Eu3+/MCM-41 is suited for successful practical applications [58]. In contrast, the fluorescence stability against temperature variation is lowered when YVO4:Eu3+ nanoparticles are incorporated into the Y2O3:Eu3+/MCM-41 (Fig. 10a, b). The lowered stability can be due to the YVO4:Eu3+ nanoparticles that are directly exposed to heat (see Fig. 1). Despite the fluorescence degradation by heat, the fluorescence intensity still keeps a high level even at 500 K, i.e., 227 °C (Fig. 10b), revealing that it is also promising for applications of our phosphor Y2O3:Eu3+/MCM-41/YVO4:Eu3+under high-temperature conditions. It can be found that there exist around two stages of the temperature-dependent fluorescence spectra, i.e., stage I and stage II in the range of below and over 100 °C, respectively. This finding is expected to contribute to future research on the intelligent system that can sense temperatures of lower or higher than 100 °C .
Study of the impact of temperature (from 300 to 500 K) on the emission spectra at λex = 303 nm for the core–shell-core phosphor Y2O3:Eu3+/MCM-41/YVO4:Eu3+. a Temperature-dependent fluorescence spectra (the inset is the magnification of the box as marked, b plot of the characteristic fluorescence peak intensity as a function of temperature, clearly showing the impact of temperature on the fluorescence emission properties of the core–shell-core heterostructure Y2O3:Eu3+/MCM-41/YVO4:Eu3+
4 Conclusions
In this work, a novel core–shell-core heterostructure has been presented, which consists of the phosphor Y2O3:Eu3+ (as an inner core), a mesoporous amorphous SiO2 material (MCM-41) serving as a bridging shell, and the phosphor YVO4:Eu3+ (as an outer core). The mesoporous MCM-41 shell plays a pivotal role in forming the well-constructed core–shell-core hierarchical heterostructure. Highly-ordered mesoporous channels are evidenced in the MCM-41 shell, and the ultrasmall YVO4:Eu3+nanoparticles are effectively incorporated into the MCM-41 mesoporous channels. This heterostructure with double cores exhibits enhanced red emission and tunable, broadened-band response to excitation properties, which hold great potential for fluorescence labeling applications. Furthermore, our core–shell phosphor (with only an inner core) exhibits impressive fluorescence stability in the temperature range of 300–500 K, while the core–shell-core phosphor (with double cores) shows a lowered stability against temperature variation due to the outer core (namely YVO4:Eu3+ nanoparticles) that is directly explored to heat (Fig. 1). Interestingly, such temperature-dependent fluorescence changes may be useful for the future exploration of intelligent system that can detect temperatures of higher or lower than 100 °C. This work also opens up a new avenue for design and synthesis of core–shell-core hierarchical phosphors with many promising applications, even beyond fluorescence/luminescence. Additionally, our synthetic route can also be generalized to produce double core–shell materials for various applications. For instance, these double cores can either synergistically contribute to performance enhancement, or play completely different roles in generating multifunctional materials (such as a magnetic inner core and a catalytic outer core for producing magnetically separable catalysts).
References
H. Hu, J.H. Xin, H. Hu, X. Wang, D. Miao, Y. Liu, Synthesis and stabilization of metal nanocatalysts for reduction reactions: a review. J. Mater. Chem. A 3, 11157 (2015). doi:10.1039/c5ta00753d
H. Yu, Q. Dong, Z. Jiao et al., Ion exchange synthesis of PAN/Ag3PO4core–shell nanofibers with enhanced photocatalytic properties. J. Mater. Chem. A 2, 1668 (2014). doi:10.1039/c3ta14447j
J. Treu, M. Bormann, H. Schmeiduch et al., Enhanced luminescence properties of InAs–InAsP core–shell nanowires. Nano Lett. 13, 6070 (2013). doi:10.1021/nl403341x
G.Z. Jia, W.K. Lou, F. Cheng et al., Excellent photothermal conversion of core/shell CdSe/Bi2Se3 quantum dots. Nano Res. 8, 1443 (2015). doi:10.1007/s12274-014-0629-2
Y. Zhao, Y. Zhang, H. Zhao et al., Epitaxial growth of hyperbranched Cu/Cu2O/CuO core-shell nanowire heterostructures for lithium-ion batteries. Nano Res. 8, 2763 (2015). doi:10.1007/s12274-015-0783-1
N. Zhang, B. Zhu, F. Peng et al., Synthesis of metal–organic-framework related core–shell heterostructures and their application to ion enrichment in aqueous conditions. Chem. Commun. 50, 7686 (2014). doi:10.1039/c4cc00900b
X. Xia, J. Tu, Y. Zhang et al., High-quality metal oxide core/shell nanowire arrays on conductive substrates for electrochemical energy storage. ACS Nano 6, 5531 (2012). doi:10.1021/nn301454q
J. Wang, H. Huang, D. Zhang et al., Synthesis of gold/rare-earth-vanadate core/shell nanorods for integrating plasmon resonance and fluorescence. Nano Res. 8, 2548 (2015). doi:10.1007/s12274-015-0761-7
H. Hu, J.H. Xin, H. Hu, X. Wang, Y. Kong, Metal-free graphene-based catalyst—Insight into the catalytic activity: a short review. Appl. Catal. A 492, 1 (2015). doi:10.1016/j.apcata.2014.11.041
Y. Guo, L. Zhang, X. Liu et al., Synthesis of magnetic core–shell carbon dot@MFe2O4(M = Mn, Zn and Cu) hybrid materials and their catalytic properties. J. Mater. Chem. A 4, 4044 (2016). doi:10.1039/c5ta10708c
Y. Zhang, Y. Guo, Q. Du et al., Oxygen vacancies induced self-assembling synthesis of V4+-BiVO4/rGO core-shell nanorods with enhanced water splitting efficiency and superior sewage purification capability. Appl. Catal. A 526, 105 (2016). doi:10.1016/j.apcata.2016.08.012
X. Chen, D. Peng, Q. Ju, F. Wang, Photon upconversion in core–shell nanoparticles. Chem. Soc. Rev. 44, 1318 (2015). doi:10.1039/c4cs00151f
H. Hu, J. Xin, H. Hu, X. Wang, X. Lu, Organic liquids-responsive β-cyclodextrin-functionalized graphene-based fluorescence probe: label-free selective detection of tetrahydrofuran. Molecules 19, 7459 (2014). doi:10.3390/molecules19067459
H. Hu, M. Chang, M. Zhang, X. Wang, D. Chen, A new insight into PAM/graphene-based adsorption of water-soluble aromatic pollutants. J. Mater. Sci. 52, 8650 (2017). doi:10.1007/s10853-017-1090-x
I.P. Sahu, D.P. Bisen, N. Brahme, R.K. Tamrakar, Studies on the luminescence behavior of SrCaMgSi2O7:Eu3+ phosphor by solid state reaction method. J. Mater. Sci. 27, 1828 (2015). doi:10.1007/s10854-015-3961-8
R. Cao, T. Fu, Y. Cao, et al., Tunable emission, energy transfer, and charge compensation in the CaSb2O6:Eu3+, Bi3+ phosphor. J. Mater. Sci. 27, 3514 (2015). doi:10.1007/s10854-015-4186-6
H. Zhang, Z. Cheng, Y. Zhang, Z. Hu, J. Yu, N. Zou, Improved luminescence properties and thermal stability of SrSi2O2N2:Eu2+ phosphor with single phase via the formation of Eu3+ on surface structure. J. Mater. Sci 52, 7605 (2017). doi:10.1007/s10853-017-0992-y
W. You, Z. Xiao, F. Lai et al., Synthesis and photoluminescence properties of Ba3Al2O6:Eu3+ red phosphor. J. Mater. Sci 51, 5403 (2016). doi:10.1007/s10853-016-9843-5
H. He, R. Fu, F. Qian, X. Song, Luminescent properties of Li2CaSiO4:Eu2+ phosphor. J. Mater. Sci. 23, 599 (2011). doi:10.1007/s10854-011-0447-1
A. John Peter, I.B. Shameem Banu, J. Thirumalai, S.P. David, Enhanced luminescence in CaMoO4: Eu3+ red phosphor nanoparticles prepared by mechanochemically assisted solid state meta-thesis reaction method. J. Mater. Sci. 24, 4503 (2013). doi:10.1007/s10854-013-1433-6
M. Elsagh, M. Rajabi, E. Amini Characterization of SrAl2O4:Eu2+, Dy3+ phosphor nano-powders produced by microwave synthesis route. J. Mater. Sci. 25, 1612 (2014). doi:10.1007/s10854-014-1773-x
I.P. Sahu, The role of europium and dysprosium in the bluish-green long lasting Sr2Al2SiO7:Eu2+, Dy3+ phosphor by solid state reaction method. J. Mater. Sci. 26, 7059 (2015). doi:10.1007/s10854-015-3327-2
Y. Zhang, H. Hu, M. Chang, et al., Non-uniform doping outperforms uniform doping for enhancing the photocatalytic efficiency of Au-doped TiO2 nanotubes in organic dye degradation. Ceram. Int. 43, 9053 (2017). doi:10.1016/j.ceramint.2017.04.050
H. Hu, J.H. Xin, H. Hu, PAM/graphene/Ag ternary hydrogel: synthesis, characterization and catalytic application. J. Mater. Chem. A 2, 11319 (2014). doi:10.1039/c4ta01620c
L. Graziani, E. Quagliarini, M. D’Orazio, TiO2-treated different fired brick surfaces for biofouling prevention: Experimental and modelling results. Ceram. Int. 42, 4002 (2016). doi:10.1016/j.ceramint.2015.11.069
A. Shirai, T. Watanabe, H. Matsuki, Inactivation of foodborne pathogenic and spoilage micro-organisms using ultraviolet-a light in combination with ferulic acid. Lett. Appl. Microbiol. 64, 96 (2017). doi:10.1111/lam.12701
Y. Liang, K. Sun, P. Chui, S. Wang, X. Sun, Luminescence functionalization of magnetite/multiwalled carbon nanotubes by YVO4:Eu3+ phosphors. Solid State Sci. 15, 79. doi:10.1016/j.solidstatesciences.2012.09.009
Y.N. Zhu, W.W. Fu, PF Zhang, GH Zheng, JJ Mu, LY Zhang (2016) Self-assembled 3D micro-architectures of Sr3V2O8:xSm3+ ‘hydrothermal’ synthesis and luminescent properties. J. Mater. Sci. 27, 12772. doi:10.1007/s10854-016-5409-1
Q. Zhang, M. Rong, H. Tan, et al., Luminescent properties of the white long afterglow phosphors: Sr3Al2O5Cl2: Eu2+, Dy3+. J. Mater. Sci. 27, 13093 (2016). doi:10.1007/s10854-016-5453-x
L.-q. Yao, G.-h. Chen, T. Yang, C.-l. Yuan, C.-r. Zhou, Energy transfer, optical and luminescent properties in Tm3+/Tb3+/Sm3+ tri-doped borate glasses. J. Mater. Sci. 28, 553 (2016). doi:10.1007/s10854-016-5558-2
C. Yang, H. Zhou, J. Xu, et al. (2016) A series of highly quantum efficiency PMMA luminescent films doped with Eu-complex as promising light-conversion molecular devices. J. Mater. Sci. 27, 11284. doi:10.1007/s10854-016-5251-5
Y. Xing, Y. Zhu, C. Chang, Y. Wang, Y. Wang, New synthetic method and the luminescent properties of green-emitting β-Sialon: Eu2+ phosphors. J. Mater. Sci. (2017). doi:10.1007/s10854-017-6689-9
D. Thapa, J. Huso, K. Miklos, et al., UV-luminescent MgZnO semiconductor alloys: nanostructure and optical properties. J. Mater. Sci. 28, 2511 (2016). doi:10.1007/s10854-016-5825-2
M.M. Haque, H.-I. Lee, K.N. Hui, Investigation of luminescent properties of red-emitting Ba(Gd,Eu)B9O16 phosphors and the effect of Ca and Sr on its luminescent properties. J. Mater. Sci. 26, 4754 (2015). doi:10.1007/s10854-015-3161-6
J.M. Carvalho, L.C.V. Rodrigues, M.C.F.C. Felinto, L.A.O. Nunes, J. Hölsä, H.F. Brito, Structure–property relationship of luminescent zirconia nanomaterials obtained by sol–gel method. J. Mater. Sci 50, 873 (2014). doi:10.1007/s10853-014-8648-7
J. Cao, H. Niu, Z. Lu, P. Huo, Y. Yan, Green synthesis of highly luminescent ZnS:Mn2+ quantum dots. J. Mater. Sci. 27, 6175 (2016). doi:10.1007/s10854-016-4545-y
H. Hu, C.C.K. Allan, J. Li et al., Multifunctional organically modified graphene with super-hydrophobicity. Nano Res. 7, 418 (2014). doi:10.1007/s12274-014-0408-0
H.-W. Hu, J.H. Xin, H. Hu (2013) Highly efficient graphene-based ternary composite catalyst with polydopamine layer and copper nanoparticles. ChemPlusChem 78, 1483. doi:10.1002/cplu.201300124
H. Hu, M. Chang, X. Wang, D. Chen, Cotton fabric-based facile solar photocatalytic purification of simulated real dye wastes. J. Mater. Sci. 52, 9922 (2017). doi:10.1007/s10853-017-1107-5
E. Dündar-Tekkaya, Y. Yürüm, Mesoporous MCM-41 material for hydrogen storage: a short review. Int. J. Hydrog. Energy 41, 9789 (2016). doi:10.1016/j.ijhydene.2016.03.050
S. Loganathan, M. Tikmani, A.K. Ghoshal, Novel pore-expanded MCM-41 for CO2 capture: synthesis and characterization. Langmuir 29, 3491 (2013). doi:10.1021/la400109j
M Mandal, V Nagaraju, B Sarma, GV Karunakar, KK Bania (2015) Enantioselective epoxidation of styrene by manganese chiral schiff base complexes immobilized on MCM-41. ChemPlusChem 80, 749. doi:10.1002/cplu.201402446
D.J. Jovanović, Ž. Antić, R.M. Krsmanović et al., Annealing effects on the microstructure and photoluminescence of Eu3+-doped GdVO4 powders. Opt. Mater. 35, 1797 (2013). doi:10.1016/j.optmat.2013.03.012
Y. Jia, W. Lü, N. Guo, W. Lü, Q. Zhao, H. You, Utilizing Tb3+ as an energy transfer bridge to connect Eu2+–Sm3+ luminescent centers: realization of efficient Sm3+ red emission under near-UV excitation. Chem. Commun. 49, 2664 (2013). doi:10.1039/c3cc39277e
X. He, Y. Zhou, H. Liang, Cun+-assisted synthesis of multi- and single-hase yttrium oxide nanosheets. J. Mater. Chem. C 1, 6829 (2013). doi:10.1039/c3tc31321b
Y. Chi, Q. Yuan, Y. Li, et al., Magnetically separable Fe3O4@SiO2@TiO2–Ag microspheres with well-designed nanostructure and enhanced photocatalytic activity. J. Hazard. Mater. 262, 404 (2013). doi:10.1016/j.jhazmat.2013.08.077
H. Hu, J.H. Xin, H. Hu, A. Chan, L. He, Glutaraldehyde–chitosan and poly (vinyl alcohol) blends, and fluorescence of their nano-silica composite films. Carbohydr. Polym. 91, 305 (2013). doi:10.1016/j.carbpol.2012.08.038
D. Wawrzynczyk, M. Nyk, M. Samoc, Multiphoton absorption in europium(iii) doped YVO4 nanoparticles. J. Mater. Chem. C 1, 5837 (2013). doi:10.1039/c3tc31308e
Z. Jian, S. Huang, Y. Cao, Y. Zhang, Hydrothermal preparation and characterization of TiO2/BiVO4 composite catalyst and its photolysis of water to produce hydrogen. Photochem. Photobiol. 92, 363 (2016). doi:10.1111/php.12575
P. Yang, Z. Quan, L. Lu, S. Huang, J. Lin, H. Fu, MCM-41 functionalized with YVO4:Eu3+: a novel drug delivery system. Nanotechnology 18, 235703 (2007). doi:10.1088/0957-4484/18/23/235703
X. Xu, J. Wu, W. Xu et al., High-efficiency non-thermal plasma-catalysis of cobalt incorporated mesoporous MCM-41 for toluene removal. Catal. Today (2016). doi:10.1016/j.cattod.2016.03.036
V.Y. Gusev, X. Feng, Z. Bu, G.L. Haller, J.A. O’Brien, Mechanical stability of pure silica mesoporous MCM-41 by nitrogen adsorption and small-angle X-ray diffraction measurements. J. Phys. Chem. 100, 1985 (1996)
Y.-C. Chen, S.-C. Huang, Y.-K. Wang, Y.-T. Liu, T.-K. Wu, T.-M. Chen, Ligand-functionalization of BPEI-coated YVO4:Bi3+,Eu3+ nanophosphors for tumor-cell-targeted imaging applications. Chem. Asian J. 8, 2652 (2013). doi:10.1002/asia.201300570
J. Wang, M. Hojamberdiev, Y. Xu (2012) CTAB-assisted hydrothermal synthesis of YVO4:Eu3+ powders in a wide pH range. Solid State Sci. 14, 191. doi:10.1016/j.solidstatesciences.2011.10.019
Y. Du, Z. Hua, W. Huang et al., Mesostructured amorphous manganese oxides: facile synthesis and highly durable elimination of low-concentration NO at room temperature in air. Chem. Commun. 51, 5887 (2015). doi:10.1039/c5cc00269a
H. Hu, X. Wang, D. Miao et al., A pH-mediated enhancement of the graphene carbocatalyst activity for the reduction of 4-nitrophenol. Chem. Commun. 51, 16699 (2015). doi:10.1039/c5cc05826k
Y. Jiang, J.R. Smith, G.R. Odette (2009) Formation of Y–Ti–O nanoclusters in nanostructured ferritic alloys: a first-principles study. Phys. Rev. B. doi:10.1103/PhysRevB.79.064103
G. Liu, L. Fu, Z. Gao et al., Investigation into the temperature sensing behavior of Yb3+ sensitized Er3+ doped Y2O3, YAG and LaAlO3 phosphors. RSC Adv. 5, 51820 (2015). doi:10.1039/c5ra05986k
Acknowledgements
We greatly appreciate the Construction Project from Guangdong Engineering Technique Research Center (506302679076), High-Level Talent Start-Up Research Project of Foshan University (Gg040918), Start-Up Research Project of Foshan University (gg040948), Universities of Guangdong Province (2016GCZX008), the Project Funded by Engineering Technology Center of Foshan City (2014GA000355), and the Key Platform Financing Programs from the Education Department of Guangdong Province (gg041002).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Chang, M., Hu, H., Zhang, Y. et al. Core-shell-core heterostructural engineering of Y2O3:Eu3+/MCM-41/YVO4:Eu3+ for enhanced red emission and tunable, broadened-band response to excitation. J Mater Sci: Mater Electron 28, 16026–16035 (2017). https://doi.org/10.1007/s10854-017-7502-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-017-7502-5