Abstract
Eucalyptus citriodora is one of the most widely used essential oils (EOs) because of its various antimicrobial, antioxidant and anti-inflammatory activities. However, its limited aqueous solubility restricts its use. The aim of this study was to develop supramolecular formulations able to retain E. citriodora EO in solution. For this purpose, new cyclodextrin (CD) polymers were synthesized and characterized using gel permeation chromatography, FTIR and NMR spectroscopies. Their retention ability toward E. citriodora in non-conventional green media (deep eutectic solvent (DES):water mixture), more precisely choline chloride:urea DES:water (70:30 wt%), was evaluated and compared to the corresponding native CD [β-cyclodextrin (β-CD)] or CD derivatives [hydroxypropylated-β-cyclodextrin (HP-β-CD) and low methylated-β-cyclodextrin (CRYSMEB)] using static headspace-gas chromatography (SH-GC). All the studied formulations showed a great capacity to retain and reduce the volatility of E. citriodora. The various polymers showed divergent retention efficiencies.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Essential oils (EOs) are a natural source of aromas and bioactive molecules with a wide range of properties including antioxidant, antimicrobial, anti-inflammatory and anti-carcinogenic. The incorporation of EOs in formulations has always attracted cosmetic, pharmaceutical and food industries. However, their use is challenging due to their volatile nature and their low aqueous solubility. Various methods could be envisaged to reduce their volatility and improve their solubility, including encapsulation in cyclodextrins (CDs) [1, 2] or cosolvency which means adding one or more solvents to the formulation [3, 4].
CDs are natural cyclic oligosaccharides possessing a truncated tridimensional cone shape characterized by a relatively hydrophobic internal cavity and a hydrophilic external surface [5]. Native CDs composed of 6, 7, and 8 α-d-glucose residues have different cavity dimensions and are called α-CD, β-CD, and γ-CD, respectively. β-CD is the most interesting host for the encapsulation of EOs components due to its perfect cavity size [6, 7], efficient drug complexation and loading [8, 9], and relatively low cost [10]. However, β-CD is poorly soluble in water (18 g L−1) [11]. Various β-CD derivatives with improved aqueous solubility, inclusion capacity and lower toxicity profiles have been synthesized and efficiently investigated in the literature [2]. The hydroxypropylated-β-CD (HP-β-CD) and the low methylated-β-CD (CRYSMEB) are two of the most studied CD derivatives [6, 9].
CD polymers can be also obtained, by copolymerizing CD molecules with other difunctionalized compounds. They can be synthesized using conventional strategies that require the use of organic solvents or sustainable procedures such as the mechanochemical approach [12]. In addition, their synthesis could be conducted in green media such as natural deep eutectic solvents (NADES) [13]. CD polymers also offer the advantages of superior solubilizing and complexing abilities. For example, an increase in the solubility of Lavandula angustifolia and Mentha piperita EOs was observed when using CD polymers as compared to native CDs (α-CD and γ-CD) [14, 15].
On the other hand, deep eutectic solvents (DESs) are neoteric class of green solvents defined as a mixture of two or more compounds that become liquid when mixed and heated at a particular molar ratio [16, 17]. A DES is characterized by a great depression of the melting point as compared to its starting materials and to their ideal mixture [18]. The first and most studied DES is reline, composed of choline chloride (ChCl) and urea (U) mixed at a 1:2 molar ratio [19]. DESs are characterized by low volatility, non-flammability and biocompatibility [20]. Few studies used DES to solubilize EOs and their components. A recent study showed a significant increase in the solubility of trans-anethole, as well as star anise and fennel EOs in DESs in comparison to water [21].
To the best of our knowledge, the literature lacks any research that considered combining DESs and CD polymers for the solubilization and retention of EOs. The aim of the present study is to develop new formulations composed of DES and CD polymers to solubilize one of the most used EOs, E. citriodora. Water-soluble polymers were synthesized using native β-CD or its derivatives (HP-β-CD and CRYSMEB) via the cross-linking method. Various crosslinking agents were used including four carboxylic acids (malonic acid, citric acid, glutaric acid and itaconic acid), poly(ethylene glycol) diglycidyl ether-640 (PEGDGE) and sodium trimetaphosphate (STMP). β-CD/malonic acid, HP-β-CD/glutaric acid, HP-β-CD/itaconic acid, HP-β-CD/PEGDGE, HP-β-CD/STMP and all CRYSMEB/based polymers were herein synthesized and reported for the first time. The synthesized polymers were submitted to physicochemical characterization. Different formulations combining ChCl:U (1:2 molar ratio) DES, water and the encapsulating agent (β-CD, CD derivative or CD polymer) were then prepared. Finally, the retention ability of these formulations towards E. citriodora was evaluated using static headspace-gas chromatography (SH-GC).
Experimental section
Materials
Choline chloride (ChCl, 98%), glutaric acid and trisodium trimetaphosphate were purchased from Sigma-Aldrich, China. Urea (U, 99%) and poly(ethylene glycol) diglycidyl ether (PEGDGE) Mw 640 were purchased from Sigma-Aldrich, USA. Citric acid and sodium hydroxide were purchased from Sigma-Aldrich, France. Itaconic acid was purchased from Sigma-Aldrich, Germany. Malonic acid was provided by Merck group, Germany. β-CD was provided by Wacker-Chemie, Lyon, France. CRYSMEB (DS = 4.9) and HP-β-CD (DS = 5.6) were provided by Roquette Frères, Lestrem, France. E. citriodora EO was provided by Herbes et Traditions, Comines, France. Choline chloride was dried at 60 °C for at least 2 weeks before use. All other compounds were used as received. Distilled water was used throughout this work.
Preparation of deep eutectic solvent
ChCl:U DES was prepared by heating ChCl and U in a 1:2 molar ratio under stirring at 60 °C until the formation of a clear and homogeneous liquid.
Synthesis and purification of CD polymers
Preparation of CD polymers
CD polymers were prepared by reticulation of either β-CD, HP-β-CD or CRYSMEB using several crosslinking agents.
Four acids (malonic acid, citric acid, glutaric acid and itaconic acid) were used following the synthetic procedure as previously reported [22]. The aqueous solutions were prepared by mixing the catalyst (KH2PO4), the CD, and the carboxylic acid with respective concentrations of 0.2, 0.09, and 0.45 mol L−1. These solutions were then concentrated by evaporation (at 50 °C and 5 mbar) and then treated at 140 °C for 30 min under reduced atmospheric pressure. The polymers were recovered after the addition of 50 mL of distilled water to the round-bottom flask. The flask was kept under agitation overnight to solubilize the reaction products. The fraction was then filtered off using sintered glass funnel (membrane porosity: 40 μm). Only trace amounts of insoluble products were obtained so these insoluble fractions were discarded.
Another crosslinking agent, the PEGDGE, was also used for polymer synthesis as previously described [23]. 5 g of CD was dissolved in 21 mL NaOH (1.42 mol L−1) and the mixture was stirred mechanically for 16 h at room temperature. PEGDGE was then added dropwise during 20 min under stirring, with a molar ratio of PEGDGE/CD equal to 3/1. The reaction mixture was heated to 30 °C for 5 h under stirring. After cooling for 20 min, the mixture was neutralized using HCl (6 mol L−1). The volume of water was reduced by evaporation using the rotary evaporator (at 5 mbar and 50 °C). Then polymers were precipitated in acetone three times and one time in ether. The solid was then collected and dissolved in distilled water.
The linker STMP was also used to prepare CD polymers as previously described [24]. 5–6.6 g of CD were dissolved in 10 mL of NaOH solution (2.5 mol L−1), under mechanical stirring (300 rpm) at 30 °C. After 210 min, the temperature was raised to 50 °C, and the desired amount of STMP (0.5, 3, 5 or 10 g with identifying codes of STMP1, STMP2, STMP3 or STMP4, respectively) was added to the solution under vigorous stirring (600 rpm). After 90 min, 50 mL of water were added, and the pH was neutralized by HCl addition (6 mol L−1).
Purification of CD polymers
The soluble fractions obtained with malonic, citric and glutaric acids were purified by several passages on a Sephadex gel column (SephadexTM G-25 Medium) using water as eluent. Polymers with itaconic acid, PEGDGE and STMP were purified by dialysis three times at room temperature for 24 h each time using a Spectra/Por® Standard grade regenerated cellulose membrane (molecular weight cut-off of 1 KDa). Solvent was then removed by a rotary evaporator and the obtained polymer fractions were dried under vacuum overnight.
Characterization of CD polymers
The purification process was followed by a physicochemical characterization of polymers using nuclear magnetic resonance spectroscopy (NMR), Fourier-transform infrared spectroscopy (FTIR) and gel permeation chromatography.
Nuclear magnetic resonnance spectroscopy
NMR spectra were recorded at 25 °C in D2O using a Bruker Avance III spectrometer, equipped with a multinuclear z-gradient BBFO probe head, operating at 400 MHz and 161 MHz for 1H and 31P NMR (when using STMP as the linker), respectively. Standard 5 mm NMR tubes were used.
In addition, using 1H-NMR spectra, the number of linker molecules (n) for one CD molecule was determined by integrating the respective protons peaks. Then, the wt% of CD in the polymer fraction was calculated using the following equation:
where MWCD and MWlinker stand for the molecular weight of the CD and the linker, respectively.
The synthesis yield was also calculated using the following equation:
where mpolymer stands for the experimentally obtained mass of polymer after purification, apparent MW stands for the calculated MW of the modified monomer unit using the number of linker molecules per one CD molecule from the 1H-NMR analysis, and nCD stands for the initially introduced CD number of moles. mpolymer over apparent MW represents the number of the modified monomer unit.
Results are presented in * MERGEFORMAT Table 1.
Infrared spectroscopy
Nicolet iS5 (Thermo Scientific) spectrophotometer was used to obtain the FTIR spectra of all compounds in the range of 600–4000 cm−1 using attenuated total reflection (iD7 ATR) diamond accessory. The spectra are the results of 16 scans with a resolution of 0.482 cm−1. Before performing the FTIR measurements on the compounds, background scan was carefully filtered.
Determination of the polydispersity index by gel permeation chromatography
Gel permeation chromatography (PL-GPC50 Plus, Agilent Technologies, Polymer Laboratories, A Varian Inc. Company) was carried out with PL-aquagel-OH 40 column at 40 °C and the flow rate was 1 mL min−1. Detection was conducted with a refractive index detector. Ultrapure water was used as eluent. The pressure during the analysis was set to 5.69 MPa. Calibration was made using standard pullulan polysaccharide samples from molecular weight: 180–708 × 103 g mol−1. Samples were used with concentration of 0.01% and filtered using a 0.2 mm Agilent captiva filter-syringe prior to use. Mw and Mn (weight averaged molecular weight and number averaged molecular weight, respectively) were determined and the polydispersity index (PDI) was calculated for all obtained polymers using the following equation [25]:
Static headspace gas chromatography
3 mL of ChCl:U (1:2 molar ratio) DES:water (70:30 wt%) mixed with different wt% of native CD, CD derivative or CD polymer were placed in 22 mL headspace vials. Then, an appropriate amount of E. citriodora EO (263 ppm) was added to all mixtures. Vials were sealed using silicone septa and aluminium foil and thermostated at 30 °C for 24 h under stirring to reach the equilibrium between liquid and gaseous phases. Subsequently, 1 mL of the headspace was withdrawn from the vial and injected in the chromatographic system for analysis via a heated transfer line (250 °C). Measurements were carried out using a Varian Genesis headspace sampler coupled to a Perkin Elmer Autosystem XL gas chromatography equipped with a flame ionization detector and a DB624 column using nitrogen as carrier gas. The GC column temperature conditions were set as follows: initial temperature of 50 °C for 2 min, increased to 200 °C at 5 °C min−1, then hold for 2 min, giving a total runtime of 34 min.
Retention studies
The volatility reduction (%) of E.citriodora was expressed as follows:
where ΣAi stands for the sum of the peak areas of the E. citriodora EO volatile components in DES:water mixture (70:30 wt%) alone or in the presence of 2 wt% β-CD, HP-β-CD, CRYSMEB or CD polymers and ΣA0 stands for the sum of the peak areas of the E. citriodora EO volatile components in water.
Titration method
Increasing content (wt%) of β-CD, HP-β-CD, CRYSMEB, P1, P8 or P16 was used while the EO concentration was maintained constant (263 ppm). Results were expressed as:
where ΣAi stands for the sum of the peak areas of the E. citriodora EO volatile components in the presence of β-CD, HP-β-CD, CRYSMEB, P1, P8 or P16 in DES:water mixture (70:30 wt%) and ΣA0 stands for the sum of the peak areas of the E. citriodora EO volatile components in ChCl:U (1:2 molar ratio) DES:water (70:30 wt%).
Results and discussion
Characterization of CD polymers
Different crosslinking agents were used to synthesize CD polymers of β-CD, HP-β-CD and CRYSMEB (Table 1). Among these studied polymers, β-CD/malonic acid, HP-β-CD/glutaric acid, HP-β-CD/itaconic acid, HP-β-CD/PEGDGE, HP-β-CD/STMP and all CRYSMEB/based polymers were synthesized and studied herein for the first time.
Polymers were characterized using 1H-NMR and FTIR spectroscopies. Also, their homogeneity was examined using gel permeation chromatography and the corresponding PDI values were calculated.
Figure 1 shows the superposition of 1H-NMR spectra of β-CD/based polymers. Results obtained for HP-β-CD and CRYSMEB/based polymers are illustrated in Supplementary information in Figures S1 and S2.
1H-NMR results depicted alterations in the form of the CDs peaks as well as modifications in the chemical shifts (δ). This confirmed the modification of all three CDs. Also, broad signals were observed in the spectra of the new polymers indicating the loss of the symmetry of the CD molecule and changes in the chemical environment due to the polymerization [26].
Polymers synthesized with STMP were investigated using 31P-NMR. Results for β-CD/based polymers are illustrated in Fig. 2. Results for HP-β-CD and CRYSMEB/based are presented in Supplementary information in Figure S3.
The peak that corresponds to inorganic phosphates (or unreacted STMP), usually located at − 21 ppm, was not detected in the spectra of eight polymers (Fig. 2, Figure S3). This proved that polymers were sufficiently washed before NMR measurements [24]. Peaks located between 2 and 5 ppm, − 2 to 2 ppm, − 10 to − 6 ppm as well as − 22 to − 20 ppm could be assigned to the grafted monophosphate groups, monophosphate linkages, pyrophosphates and grafted tripolyphosphates, respectively [27]. The corresponding structures of these moieties are found in the polymers and illustrated in \* MERGEFORMAT Fig. 2. This proved the reticulation and thus the formation of CD polymers.
Then, the FTIR spectra were recorded for all polymers and starting materials. The FTIR spectra of β-CD, citric acid and the corresponding polymer (P1) are shown, as an example, in \* MERGEFORMAT Fig. 3. Other examples are illustrated in supplementary information in Figures S4 and S5. Results showed the appearance of sharp peaks around 1700 cm−1 in all the polymers spectra. This proved the conversion of the carbonyl groups of the carboxylic acids into ester functions [22, 28].
Finally, gel permeation chromatography was carried out to determine the macromolecular mass of the soluble polymers and then to calculate the polydispersity index (PDI) values. Results are listed in \* MERGEFORMAT Table 1. Examples of the obtained chromatograms are illustrated in supplementary information in Figures S6–S8.
PDI values for all polymers ranged from 1.14 to 3.77 depicting the heterogeneity of the obtained samples.
Retention studies
Retention studies were carried out in ChCl:U DES:water (70:30 wt%) medium. Water was added to ChCl:U DES because it was shown that the addition of water decreases the viscosity of this DES [29]. Additionally, the presence of water allows CD experiencing an aqueous like environment and gives the CD macrocycle enough mobility to maintain its encapsulation ability [30].
Retention studies were conducted using 2 wt% of CD or polymer. The final molar concentration of CD in the formulations containing polymers was calculated based on the number of linkers per CD molecule obtained from NMR studies according to Eq. 1 and listed in \* MERGEFORMAT Table 1.
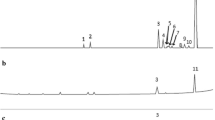
The retention ability of native CD (β-CD), CD derivatives (HP-β-CD and CRYSMEB) and the synthetized polymers towards E. citriodora was evaluated by measuring the reduction in the EO volatility according to Eq. 4. Figure 4 shows, as an example, the effect of β-CD/citric acid (P1) polymer on the chromatographic profile of E. citriodora EO in ChCl:U (1:2 molar ratio) DES:water (70:30 wt%). Results obtained with P8 and P16 polymers are illustrated in Supplementary information in Figures S9 and S10.
Results of the volatility reduction of E. citriodora EO in the different mixtures containing β-CD/based polymers are presented in Fig. 5. Results for HP-β-CD and CRYSMEB/based polymers are presented in Figure S11.
Firstly, it is worthy to note that ChCl:U (1:2 molar ratio) DES:water (70:30 wt%) itself significantly entrapped and reduced up to 58% the volatility of E. citriodora compared to water. This can be mainly due to the lower polarity of the mixture compared to water, which allowed higher solubilization of the EO [31]. As can be noted in Figures Fig. 5 and S11, the addition of all the encapsulating agents (β-CD, CD derivatives (HP-β-CD and CRYSMEB) or their corresponding polymers) led to an additional decrease in the volatility of the studied EO. The volatility reduction observed in the presence of the various hosts in DES:water 70:30 wt% was in the range of 77–94% as compared to water. This important decrease in the volatility directly implies a significant increase in the solubility of E. citriodora in the new formulations.
Additionally, from a mechanistic point of view, the better entrapment potential observed in the presence of β-CD, HP-β-CD, CRYSMEB and their synthetized polymers indicated that all these structures retained, at least partially, their inclusion capacity in this non-conventional medium (DES:water 70:30 wt%). In addition, results showed that β-CD and its two derivatives (HP-β-CD and CRYSMEB) possess a similar efficacy of volatility reduction (Fig. 5). This could be contributed to the fact that these CDs present the same cavity volume and thus pointed out the important role of the cavity in the entrapment potential of EOs components. However, as concerns the polymers, a large discrepancy in their performance was noticed (Fig. 5). This could not be attributed to the difference in the molar concentration of CD in the polymer/based mixtures since the variation in the CD concentration among mixtures was very slight ( \* MERGEFORMAT Table 1). For example, the HP-β-CD/glutaric acid polymer (P9) with a molar CD concentration of 13.56 mmol L−1 showed a better volatility reduction than the HP-β-CD/PEDGE polymer (P11) with a molar CD concentration of 16.02 mmol L−1, both in the ChCl:U DES:water 70:30 wt%.
Interestingly, some polymers presented significantly better entrapment ability than the parent macrocycle. β-CD/itaconic acid (P2), HP-β-CD/glutaric acid (P9), and CRYMEB/STMP1 (P18) polymers showed slightly higher retention ability towards E. citriodora EO, in comparison to the corresponding parent CD. All other polymers presented a similar or a less efficiency than the parent CD. This could be due to the highly-branched architecture of CD polymers, that can hinder the CD cavity and lower their encapsulation efficiency [32]. Generally, the lowest retention efficiency was obtained with PEGDGE/based CD polymers. PEGDGE presents a very long chain that could hinder the access to the CD cavity.
Static headspace-gas chromatography titration method
In order to check out the presence of an inclusion phenomenon, static headspace-gas chromatography SH-GC titration method was carried out. The evolution of the EO volatility as a function of P1, P8 and P16 content in comparison to the relative native β-CD and CD derivatives was evaluated. These polymers were chosen, because they were synthesized using a conventional cross-linking agent, citric acid, and they showed very good synthesis yields (Table 1). Results obtained with β-CD and P1 polymer are presented in \* MERGEFORMAT Fig. 6. Results obtained with HP-β-CD, CRYSMEB and their relative polymers are illustrated in Supplementary information in Figures S12 and S13, respectively.
A gradual decrease in the volatility of the EO was observed when increasing the wt% of all studied CDs and polymers. The shape of the titration curves obtained fits well with the typical curves generally observed for the complexation phenomenon [33, 34]. This suggests that the retention of the EO could be due to the encapsulation of the EO components within the CD cavity. In fact, the geometric fit of citronellal (Fig. 4), the major component of E. citriodora, with the cavity of β-CD and its derivatives was already confirmed in the literature [6].
Conclusion
CD polymers have long been considered as very promising materials targeting different applications including drug carriers, organic pollutants remedies and others in aqueous solutions. Herein, the ability of new ternary mixtures based on ChCl:U DES, water, and CD (β-CD, its derivatives (HP-β-CD and CRYSMEB), or CD polymers) to entrap and reduce the volatility of E. citriodora EO was investigated. Results showed that these novel mixtures efficiently reduced the volatility of EO. This consequently proved their ability to solubilize E. citriodora EO.
References
Sebaaly, C., Jraij, A., Fessi, H., Charcosset, C., Greige-Gerges, H.: Preparation and characterization of clove essential oil-loaded liposomes. Food Chem. 178, 52–62 (2015). https://doi.org/10.1016/j.foodchem.2015.01.067
Kfoury, M., Hădărugă, N.G., Hădărugă, D.I., Fourmentin, S.: Cyclodextrins as encapsulation material for flavors and aroma. In: Encapsulations, pp. 127–192. Elsevier, Amsterdam (2016)
Nakhle, L., Kfoury, M., Greige-Gerges, H., Fourmentin, S.: Effect of dimethylsulfoxide, ethanol, α- and β-cyclodextrins and their association on the solubility of natural bioactive compounds. J. Mol. Liq. 310, 113156 (2020). https://doi.org/10.1016/j.molliq.2020.113156
Kfoury, M., Geagea, C., Ruellan, S., Greige-Gerges, H., Fourmentin, S.: Effect of cyclodextrin and cosolvent on the solubility and antioxidant activity of caffeic acid. Food Chem. 278, 163–169 (2019). https://doi.org/10.1016/j.foodchem.2018.11.055
Crini, G.: Review: a history of cyclodextrins. Chem. Rev. 114, 10940–10975 (2014). https://doi.org/10.1021/cr500081p
Kfoury, M., Auezova, L., Greige-Gerges, H., Fourmentin, S.: Promising applications of cyclodextrins in food: improvement of essential oils retention, controlled release and antiradical activity. Carbohydr. Polym. 131, 264–272 (2015). https://doi.org/10.1016/j.carbpol.2015.06.014
Kfoury, M., Landy, D., Fourmentin, S.: Characterization of cyclodextrin/volatile inclusion complexes: a review. Molecules 23, 1204 (2018). https://doi.org/10.3390/molecules23051204
Kfoury, M., Auezova, L., Fourmentin, S., Greige-Gerges, H.: Investigation of monoterpenes complexation with hydroxypropyl-β-cyclodextrin. J. Incl. Phenom. Macrocycl. Chem. 80, 51–60 (2014). https://doi.org/10.1007/s10847-014-0385-7
Kfoury, M., Auezova, L., Ruellan, S., Greige-Gerges, H., Fourmentin, S.: Complexation of estragole as pure compound and as main component of basil and tarragon essential oils with cyclodextrins. Carbohydr. Polym. 118, 156–164 (2015). https://doi.org/10.1016/j.carbpol.2014.10.073
Crini, G., Fourmentin, S., Fenyvesi, É., Torri, G., Fourmentin, M., Morin-Crini, N.: Cyclodextrins, from molecules to applications. Environ. Chem. Lett. 16, 1361–1375 (2018). https://doi.org/10.1007/s10311-018-0763-2
McCune, J.A., Kunz, S., Olesińska, M., Scherman, O.A.: DESolution of CD and CB macrocycles. Chem. Eur. J. 23, 8601–8604 (2017). https://doi.org/10.1002/chem.201701275
Rubin Pedrazzo, A., Trotta, F., Hoti, G., Cesano, F., Zanetti, M.: Sustainable mechanochemical synthesis of β-cyclodextrin polymers by twin screw extrusion. Environ. Sci. Pollut. Res. 29, 251–263 (2022). https://doi.org/10.1007/s11356-021-15187-5
Cecone, C., Hoti, G., Krabicová, I., Appleton, S.L., Caldera, F., Bracco, P., Zanetti, M., Trotta, F.: Sustainable synthesis of cyclodextrin-based polymers by exploiting natural deep eutectic solvents. Green Chem. 22, 5806–5814 (2020). https://doi.org/10.1039/D0GC02247K
Ciobanu, A., Mallard, I., Landy, D., Brabie, G., Nistor, D., Fourmentin, S.: Inclusion interactions of cyclodextrins and crosslinked cyclodextrin polymers with linalool and camphor in Lavandula angustifolia essential oil. Carbohydr. Polym. 87, 1963–1970 (2012). https://doi.org/10.1016/j.carbpol.2011.10.005
Ciobanu, A., Mallard, I., Landy, D., Brabie, G., Nistor, D., Fourmentin, S.: Retention of aroma compounds from Mentha piperita essential oil by cyclodextrins and crosslinked cyclodextrin polymers. Food Chem. 138, 291–297 (2013). https://doi.org/10.1016/j.foodchem.2012.10.106
Shishov, A., Bulatov, A., Locatelli, M., Carradori, S., Andruch, V.: Application of deep eutectic solvents in analytical chemistry. A review. Microchem. J. 135, 33–38 (2017). https://doi.org/10.1016/j.microc.2017.07.015
El Achkar, T., Greige-Gerges, H., Fourmentin, S.: Basics and properties of deep eutectic solvents: a review. Environ. Chem. Lett. (2021). https://doi.org/10.1007/s10311-021-01225-8
Martins, M.A.R., Pinho, S.P., Coutinho, J.A.P.: Insights into the nature of eutectic and deep eutectic mixtures. J. Solution Chem. 48, 962–982 (2019). https://doi.org/10.1007/s10953-018-0793-1
Abbott, A.P., Capper, G., Davies, D.L., Rasheed, R.K., Tambyrajah, V.: Novel solvent properties of choline chloride/urea mixturesElectronic supplementary information (ESI) available: spectroscopic data. See http://www.rsc.org/suppdata/cc/b2/b210714g/. Chemical Communications. 70–71 (2003). https://doi.org/10.1039/b210714g
Smith, E.L., Abbott, A.P., Ryder, K.S.: Deep eutectic solvents (DESs) and their applications. Chem. Rev. 114, 11060–11082 (2014). https://doi.org/10.1021/cr300162p
El Achkar, T., Moura, L., Moufawad, T., Ruellan, S., Panda, S., Longuemart, S., Legrand, F.-X., Costa Gomes, M., Landy, D., Greige-Gerges, H., Fourmentin, S.: New generation of supramolecular mixtures: characterization and solubilization studies. Int. J. Pharm. (2020). https://doi.org/10.1016/j.ijpharm.2020.119443
Martel, B., Ruffin, D., Weltrowski, M., Lekchiri, Y., Morcellet, M.: Water-soluble polymers and gels from the polycondensation between cyclodextrins and poly(carboxylic acid)s: a study of the preparation parameters. J. Appl. Polym. Sci. 97, 433–442 (2005). https://doi.org/10.1002/app.21391
Luppi, F., Cavaye, H., Dossi, E.: Nitrated cross-linked β-cyclodextrin binders exhibiting low glass transition temperatures. Propellants, Explos., Pyrotech. 43, 1023–1031 (2018). https://doi.org/10.1002/prep.201800137
Wintgens, V., Dalmas, F., Sébille, B., Amiel, C.: Novel phosphorus-containing cyclodextrin polymers and their affinity for calcium cations and hydroxyapatite. Carbohydr. Polym. 98, 896–904 (2013). https://doi.org/10.1016/j.carbpol.2013.06.073
Ohno, K., Wong, B., Haddleton, D.M.: Synthesis of well-defined cyclodextrin-core star polymers. J. Polym. Sci. A Polym. Chem. 39, 2206–2214 (2001). https://doi.org/10.1002/pola.1197
Anand, R., Malanga, M., Manet, I., Manoli, F., Tuza, K., Aykaç, A., Ladavière, C., Fenyvesi, E., Vargas-Berenguel, A., Gref, R., Monti, S.: Citric acid–γ-cyclodextrin crosslinked oligomers as carriers for doxorubicin delivery. Photochem. Photobiol. Sci. 12, 1841 (2013). https://doi.org/10.1039/c3pp50169h
Wintgens, V., Lorthioir, C., Dubot, P., Sébille, B., Amiel, C.: Cyclodextrin/dextran based hydrogels prepared by cross-linking with sodium trimetaphosphate. Carbohydr. Polym. 132, 80–88 (2015). https://doi.org/10.1016/j.carbpol.2015.06.038
Li, Y.-F., Ha, Y.-M., Guo, Q., Li, Q.-P.: Synthesis of two β-cyclodextrin derivatives containing a vinyl group. Carbohydr. Res. 404, 55–62 (2015). https://doi.org/10.1016/j.carres.2014.11.012
Nakhle, L., Kfoury, M., Mallard, I., Greige-Gerges, H., Landy, D.: Solubilization of Eucalyptus citriodora essential oil and citronellal in deep eutectic solvents:water:cyclodextrins mixtures. J. Mol. Liq. 359, 119371 (2022). https://doi.org/10.1016/j.molliq.2022.119371
Dugoni, G.C., Di Pietro, M.E., Ferro, M., Castiglione, F., Ruellan, S., Moufawad, T., Moura, L., Costa Gomes, M.F., Fourmentin, S., Mele, A.: Effect of water on deep eutectic solvent/β-cyclodextrin systems. ACS Sustain. Chem. Eng. 7, 7277–7285 (2019). https://doi.org/10.1021/acssuschemeng.9b00315
Cao, J., Cao, J., Wang, H., Chen, L., Cao, F., Su, E.: Solubility improvement of phytochemicals using (natural) deep eutectic solvents and their bioactivity evaluation. J. Mol. Liq. 318, 113997 (2020). https://doi.org/10.1016/j.molliq.2020.113997
Morin-Crini, N., Winterton, P., Fourmentin, S., Wilson, L.D., Fenyvesi, É., Crini, G.: Water-insoluble β-cyclodextrin–epichlorohydrin polymers for removal of pollutants from aqueous solutions by sorption processes using batch studies: a review of inclusion mechanisms. Prog. Polym. Sci. 78, 1–23 (2018). https://doi.org/10.1016/j.progpolymsci.2017.07.004
Saokham, P., Muankaew, C., Jansook, P., Loftsson, T.: Solubility of cyclodextrins and drug/cyclodextrin complexes. Molecules 23, 1161 (2018). https://doi.org/10.3390/molecules23051161
Landy, D., Fourmentin, S., Salome, M., Surpateanu, G.: Analytical improvement in measuring formation constants of inclusion complexes between β-cyclodextrin and phenolic compounds. J. Incl. Phenom. Macrocycl. Chem. 38, 187–198 (2000). https://doi.org/10.1023/A:1008156110999
Acknowledgements
The authors would like to acknowledge the National Council for Scientific Research of Lebanon (CNRS-L) and Université du Littoral Côte d'Opale (ULCO) for granting a doctoral fellowship to Lamia Nakhle. This work is a contribution to the CPER (Contrat de Plan Etat-Région) research project IRenE (Innovation et Recherche en Environnement) and is supported by the French Ministère de l’Enseignement Supérieur, the region Hauts-de-France and the European Regional Development Fund.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nakhle, L., Kfoury, M., Mallard, I. et al. Cyclodextrin polymers in combination with water and deep eutectic solvent for the retention of Eucalyptus citriodora essential oil. J Incl Phenom Macrocycl Chem 102, 831–840 (2022). https://doi.org/10.1007/s10847-022-01161-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-022-01161-2