Abstract
Purpose
A significant proportion of patients undergoing catheter ablation for atrial fibrillation (AF) experience arrhythmia recurrence. This is mostly due to pulmonary vein reconnection (PVR). Whether mapping using High-Density Wave (HDW) technology is superior to standard bipolar (SB) configuration at detecting PVR is unknown. We aimed to evaluate the efficacy of HDW technology compared to SB mapping in identifying PVR.
Methods
High-Density (HD) multipolar Grid catheters were used to create left atrial geometries and voltage maps in 36 patients undergoing catheter ablation for AF (either due to recurrence of an atrial arrhythmia from previous AF ablation or de novo AF ablation). Nineteen SB maps were also created and compared. Ablation was performed until pulmonary vein isolation was achieved.
Results
Median time of mapping with HDW was 22.3 [IQR: 8.2] min. The number of points collected with HDW (13299.6±1362.8 vs 6952.8±841.9, p<0.001) and used (2337.3±158.0 vs 1727.5±163.8, p<0.001) was significantly higher compared to SB. Moreover, HDW was able to identify more sleeves (16 for right and 8 for left veins), where these were confirmed electrically silent by SB, with significantly increased PVR sleeve size as identified by HDW (p<0.001 for both right and left veins). Importantly, with the use of HDW, the ablation strategy changed in 23 patients (64% of targeted veins) with a significantly increased number of lesions required as compared to SB for right (p=0.005) and left veins (p=0.003).
Conclusion
HDW technology is superior to SB in detecting pulmonary vein reconnections. This could potentially result into a significant change in ablation strategy and possibly to increased success rate following pulmonary vein isolation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Recurrence of atrial fibrillation (AF) following catheter ablation procedures remains an important clinical problem, with reported rates of 24–55% [1, 2] and pulmonary vein reconnection (PVR) being the most common reason for low success rates of treatment [3, 4]. The causes of PVR appear to be multifactorial, with limitations in mapping to identify substrates for ablation being a major reason.
Improvement in mapping to identify pulmonary vein (PV) sleeves and effectiveness of complete pulmonary vein isolation (PVI) could be the key to prevent AF recurrence. This is currently achieved with incremental procedures [5], improved ablation techniques [6,7,8,9] confirming bidirectional block [10], prolonged waiting times after PVI, and uncovering dormant PVR with agents such as adenosine [11]. Improvements in mapping technology may lead to higher freedom from AF [1]. Despite promising advances made in PVI techniques, PVR still rates remain high. The concept of manipulating wavefront directionality to map PV ostia for isolation has not been utilized independently to identify PVR.
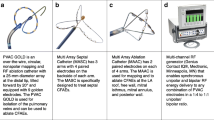
The High-Density (HD) Grid catheter consists of 16 electrodes mounted on four-splines within a closed frame (Fig. 1). The HD Grid mapping catheter has 4 splines, each with 4 small (1 mm) equally spaced (3 mm) electrodes. Unlike standard multipolar mapping, the HD Grid is the only available system that enables simultaneous assessment of both adjacent and orthogonal electrograms (EGMs). The spline electrodes are thin, fixed but highly flexible, mounted on a bi-directional deflectable catheter. The High-Density Wave (HDW) algorithm enables EGM data acquisition to be less direction-dependent to create voltage maps. It automatically selects the EGM with the largest peak-to-peak voltage, comparing orthogonal planes of activation, whereas standard bipolar (SB) mapping uses a single direction spline bipoles. This has the distinct advantage of detecting larger EGM signals, independent of electrode orientation to wavefront direction, as shown in previous experimental studies [12].
HD Grid catheter design
Simultaneously adjacent electrograms (EGMs) detect along the Grid splines (in this example B2-B3) and orthogonal EGMs across Grid spline (in this example B2-C2). HD wave algorithm selects the largest peak-to-peak voltage obtained from either vector (in this example B2-C2) and this is assigned as the local EGM at this point on the map. Standard bipolar algorithm defaults to select EGMs obtained from along the spline bipoles (in this example B2-B3) regardless of which vector produced the largest peak-to-peak voltage.
The HD Grid mapping catheter has 4 splines, each with 4 small (1 mm) equally spaced (3 mm) electrodes. The grid design offers the simultaneous orthogonal bipolar electrogram recording (along and across the spline)
In the present study, we aim to evaluate the efficacy of HDW technology in identifying PVR compared to SB mapping in PVI procedures.
2 Methods
2.1 Study population
Thirty-six consecutive patients undergoing AF catheter ablation for PV isolation with the HD Grid catheter (either due to recurrence of an atrial arrhythmia from previous AF ablation or de novo AF ablation) were recruited at Barts Heart Centre, London, UK, between December 2017 and November 2019.
Patients without PVR were excluded from the study. Each patient acted as their own control due to the HDW electroanatomical map and SB electroanatomical map being taken simultaneously. The study was approved by the local Hospital Ethics Committee. Patients gave written informed consent.
2.2 Procedural characteristics
All procedures were performed under conscious sedation or general anaesthesia. Right femoral venous access was obtained using ultrasound guidance in all cases. A decapolar catheter (6F Cournand fixed curve, Boston Scientific) was placed in the coronary sinus for pacing manoeuvers and used as reference for electroanatomical mapping and activation timings. Single or double trans-septal puncture was performed with a 71-cm Brockenbrough needle under fluoroscopy guidance or under transoesophageal echocardiogram guidance where general anaesthesia was used. The HD Grid and TactiCathTM (Abbott) irrigated ablation catheter were introduced into the left atrium through a non-steerable sheath (8.5F SL0 or SL1, Abbott) and a steerable sheath (Agilis; Abbott). Unfractionated Heparin was given to maintain activation clotting time above 300 throughout the procedure.
2.3 Creation of left atrial HDW map
Left atrial (LA) geometry and voltage maps were created using the HD Grid catheter and EnSite Precision mapping system (Abbott) both in sinus rhythm and in atrial fibrillation (Fig. 2). Myocardial voltages < 0.07mV were considered to be scar and voltages > 0.5mV were considered to be healthy active myocardium. Voltages between 0.07–0.5 were still considered to be low, but corresponding to viable tissue, as seen in many patients with diseased atrium.
Example of left atrial maps with a right lateral projection of SB data and the same projection using HDW in AF
A small anterior right upper pulmonary vein sleeve is seen on the SB map, but a larger more extensive PVR is detected on the HDW map. The same voltage settings of 0.07–0.5mV were used to create the maps. The corresponding electrograms around the RUPV are shown on the right of each map. Much larger and active electrograms are seen with the HDW wave
The HDW algorithm selects the largest peak to peak voltage EGMs obtained from adjacent orthogonally orientated electrode pairs acquired simultaneously, automatically assigning the largest detected EGM as activation points to create high density voltage map. Sufficient points coverage of the entire LA was collected, and in particular, higher density points were taken around each PV ostium.
2.4 Identification of pulmonary vein reconnections
Left atrial voltage maps completed with the HDW were used to guide ablation. Signals were annotated to the peak deflection (on local EGM) using a EGM voltage >0.07mV, while scar was denoted for voltages <0.07mV. Threshold of 0.07 was used for both catheters of the study (HD Grid and SB). A PVR was denoted where there was detectable voltage above threshold and channels of sequential activation proceeding from the atrium into the vein confirmed. The number of PVRs which were measured was identified as distinct channels surrounded by electrically silent tissue (scar). Activation points were re-annotated to exclude far-field signals. Differential pacing was utilized, if there was uncertainty of local or far-field signals. Following completion of the procedure, the number of PVRs was recorded for each vein as well as the maximal width of each PVR. The presence of PVR was re-confirmed by a second blinded experienced electrophysiologist and cardiac physiologist, who were blinded to each other’s assessment.
2.5 Radiofrequency ablation
PVR sleeves were targeted for ablation using a force sensing TactiCath catheter, aiming for 10- to 40-g contact. Segmental ablation lesions were delivered at 35–40 W energy along the wide antral circumferential ablation (WACA)/PV ring for the anterior wall and lesions at 30 W were delivered on the posterior wall. Energy titration was guided by lesion size index (LSI), aiming for a target of 4 in the posterior wall and 6 in the anterior wall. The LSI is a multi-parametric index, which has been validated by both experimental and clinical studies [13,14,15], incorporating time, power, contact-force, and impedance data recorded during radiofrequency ablation in a weighted formula that describes ablation biophysics. Catheter irrigation was set between 17 and 30ml/min, depending on power settings used.
2.6 Confirmation of pulmonary vein isolation
Entry and exit block were assessed in ablated veins and repeat voltage maps were made with the HD Grid to assess for PVI. As part of the validation protocol for this new catheter, we aimed to compare the data obtained from a circular mapping catheter (CMC). The choice for an OptimaTM or a Reflexion SpiralTM CMC catheter was left to operator’s discretion in order to confirm PVI after ablation guided by the HD Grid. This was performed in 53% of patients with 100% correlation, demonstrating concordance with current practice for PVI.
2.7 Creation of left atrial standard bipolar map
After completion of procedure, the HDW voltage map was converted into a SB map offline using the TurboMap feature. Unlike the HDW where the largest peak-to-peak voltage was automatically selected between adjacent and orthogonal vectors, the SB map was created from electrogram data taken from a fixed electrode orientation. The same surface points were used and voltage settings were applied with a range between 0.07 and 0.5mV for consistency. AutoMap settings were kept the same for both SB and HDW map for all patients (interior projection 7mm, exterior projection 7mm and interpolation setting 7) (Fig. 2). In our study, PVR was confirmed by a combination of activation and voltage points extending from the antral chamber leading into the vein, with adjacent electrogram that showed split potentials of >20msec, considered a line of block if recorded in sinus rhythm. Pulmonary vein reconnections were analyzed and identified in the same manner as described for the HDW map. The number of PVRs was recorded by two experienced electrophysiologists and electrophysiology technicians.
2.8 Statistical analysis
Descriptive data are presented as numbers and percentages or mean (± standard deviation) or median and inter-quartile range. The Chi-square test was used for comparing ratios and categorical variables. Comparison of continuous variables was performed using the paired or unpaired t-test, or their non-parametric equivalents, when appropriate. Results with p<0.05 were considered significant. PASW Statistics (SPSS Inc., Chicago, IL) version 18.0 was used for statistical analysis.
3 Results
Thirty-six (36) patients undergoing catheter ablation for PVI were enrolled into our study. Of these, 33 had previously undergone catheter ablation for paroxysmal/persistent atrial fibrillation or atrial tachycardia. PVI was performed as part of the procedure in 5 cases of relevant atrial tachycardia.
The mean age of patients was 66.3±8.1 years and 39% were female. The mean LA size was 4.10±0.60 cm. The median duration of AF history was 38 months. Baseline characteristics of the patients are presented in Table 1.
3.1 Mapping time, points collected and used
Creating detailed maps of the left atrium with the HD Grid required a median of 22.3 [IQR: 8.2] min. There was a significant difference between HDW and SB in the number of points collected and used (Table 2). More specifically, the number of points collected and used with the HDW was significantly higher compared to SB (13299.6±1362.8 vs 6952.8±841.9, p<0.001 and 2337.3±158.0 vs 1727.5±163.8 p<0.001, respectively) (Figure S1).
3.2 Pulmonary veins: number and size of sleeves
HDW detected 139 PV sleeves, while SB detected 123 PV sleeves (Table 2). HDW was able to identify 24 sleeves which were not detected by SB mapping. There were a few instances where SB was better, identifying 9 vein sleeves which were not detected by HDW. In general, not only were more PVRs identified by HDW compared to SB but also the ostial width of reconnections was substantially larger when mapped by HDW compared to SB (right veins: 23.0±3.53mm vs 12.7±2.33mm, p<0.001 and left veins: 37.4±6.1mm vs 23.0±5.46mm, p<0.001), (Figs. 2 and 3) (Table 2).
a Example of right lower pulmonary vein reconnections mapped during sinus rhythm. Comparative maps using HDW on the left are compared to SB on the right. Electrogams recorded at the RLPV are displayed on each right panel. More extensive reconnections are seen by the HDW map, with the vein almost totally connected posteriorly, extending into the inter-venous ridge. b Example of voltage maps of the left upper pulmonary (LUPV) comparing standard bipolar (SB) settings (left panel) compared to HD wave (right panel).
Scar is seen as grey <0.07mV. Sampled electrograms from the circled area are shown on the panel on the right of each map. Timing markers are seen as single sharp deflections. A CS reference electrogram is seen at the top in green. The SB map has hardly any electrogram, while the HD wave shows active reconnections leading into the LUPV. c Example of left atrial map with roof view projection, created by HDW and SB during sinus rhythm from LUPV. Electrograms taken from the left upper pulmonary vein (within the area denoted by the elliptical black line) are shown on the right of each map. The reference CS electrogram is shown in green. The left superior pulmonary vein is silent according to the SB map, but the whole vein is clearly connected on the HDW map, confirmed by the electrograms
3.3 HD Grid: role in ablation strategy, AF recurrence, and safety profile
Allowing for a 5-mm ablation lesion size to treat PVR, HDW mapping resulted in a significantly higher number of lesions for both left (10.9±1.55 vs 7.7±1.13, p=0.005) and right (8.56±1.16 vs 6.62±1.27, p=0.003) PV applications compared to SB (Fig. 4), which can actually be interpreted as a change in ablation strategy in 23 (64%) patients of the study population.
Twenty-nine patients had a minimum of 3months (only one patient, the rest greater than 5months) follow-up. Over a median period of 10 months post-mapping with HD Grid and ablation, 24 (83%) of the patients remained free of AF. Recurrence of AF was documented in 1 patient with paroxysmal AF, 2 patients undergoing repeat ablation for paroxysmal AF, 1 patient with persistent AF, and 1 patient having repeat ablation for persistent AF.
There were 2 cases (5.6%) of acute cardiac tamponade. These were evident during CMC manipulation, associated with difficulties in CMC navigation causing high forces. Although both cases were evident while using the CMC, a slowly evolving tamponade because of the previous use of HD Grid cannot be excluded, and we therefore do not ascribe both tamponade cases to CMC. Both cases required pericardial drainage, but no surgical intervention.
4 Discussion
The cornerstone of AF therapy remains PVI. There have been a number of technological advances over the last 2 decades, with the aim of achieving complete electrical isolation of pulmonary veins. These include the development of irrigated ablation catheters, application of higher energies for ablation, use of high resolution mapping, and contact-force sensing catheters. These have achieved single procedure success rates for catheter ablation procedures generally around 70%. Arrhythmia recurrence post-ablation is generally considered to be due to reconnection of PV sleeves. The current challenge is to develop more accurate mapping combined with effective ablation therapy, to create more complete and permanent PVI.
There are many variables that affect the resolution of bipolar EGMs including electrode numbers, spacing, and size [16,17,18]. In this study, wavefront direction technology was used to independently show its effect on the resolution of bipolar EGMs clinically and therefore alter the resolution of atrial electroanatomical maps.
We have demonstrated that wavefront directionality significantly influences the detection of PVR. Using HDW technology, orthogonal electrogram processing (2 different orientations of electrodes-down and across the spline) results in not only more voltage points being taken and used but also a significantly higher identification of PVRs compared to SB configuration (use of electrodes only down the spline-collection only along the longitudinal axis).
Previous research has demonstrated that bipolar EGM amplitude is directly affected by wavefront direction, with the wavefront detection being largest when propagating parallel to the bipole electrode orientation, and significantly reduced when it is perpendicular to electrode pair [19, 20]. If a wavefront crosses a bipole pair simultaneously, it may show little or no potential difference. This is of particular importance in PVI procedures, where the end point of isolation is determined by absence of ostial electrograms recorded by a circular catheter and this is in turn dependent on the orientation of the catheter electrodes to the activation wavefront.
By its very nature, SB map is limited by its orientation to wavefront progression. This may lack the sensitivity to detect myocardial sleeves connecting PVs, if paired electrodes are aligned perpendicular to wavefront activation, thus leading some authors to use the term “bipolar blindness” [21]. In this study, a number of PVs were considered to be silent and isolated, when in fact clear PVR sleeves were evident when mapped by HDW. In 16 right and 8 left PVs were considered isolated, but had clear PVRs. The HD wave algorithm also detected greater numbers of PVR and the overall width was significantly larger compared to SB settings, resulting in a change in ablation strategy (delivering more lesions) in 64% of PVRs.
Improving map resolution could increase detection of PVR [16, 21] and long-term freedom from atrial arrhythmias [16]. This study using the HD Grid catheter demonstrates the advantage of improving mapping resolution and how this technology could possibly overcome the limitations of current SB recordings.
The Grid shape, with thin flexibility splines, can be easily adapted and conformed to complex PV anatomy and ridges. In fact, the only complication we had was some difficulty in getting the CMC to sit ostially on small calibre PV, whereas the HD Grid had no problems achieving mapping nor access. The HD Grid catheter in this study was used by 10 different operators and was described as easy to manoeuvre by all users.
4.1 Clinical implications
Recurrence of AF remains a significant challenge in ablation therapy and PVRs are present in 92% of patients undergoing repeat AF ablation procedures [3]. The mechanism responsible for PVR includes incomplete linear lines leaving anatomic gaps or failure to produce transmural lesions, leading to PV conduction [22, 23]. This study also highlights the complex role of wavefront direction as well as the limitations of SB mapping. Development of HD wave technology allows mapping and identifying PVRs with greater resolution. This can change the ablation strategy and potentially result to improvement in clinical outcomes.
4.2 Limitations
This is a small pilot study and involves a relatively limited number of patients without a randomized control arm. Only short-term follow-up data is available; longer-term clinical outcomes in larger numbers are still awaited to assess the efficacy of HDW mapped for PVI. It is worth mentioning that not all HD Grid electrodes are always in contact with atrial tissues, which could potentially affect data acquisition, but this is common across all commercially available catheters.
In the present study, we have recorded the time required for mapping the left atrium with the HD Grid; however, we have not recorded the amount of time used for annotation and analysis. Each case was not done by the same operator, each case had its own challenges, and it was approached in a different way, while a difficult case would require more time compared to an easy one. Cases were analyzed offline and in retrospect. Again, the same principles applied, as such it would not be safe/accurate to provide timings, and therefore, these were not recorded.
Finally, we acknowledge that a CMC should have been used in all our cases; however, due to time and clinical reasons, we did not. We tried to avoid extending cases by additional time at patients’ best interest.
5 Conclusions
The simultaneous detection of orthogonal and adjacent EGMs by the HD Grid using HDW algorithm is significantly superior compared to configuration along spline bipoles at detecting PVRs. The greater resolution which is achieved by HD Grid is a promising development in PVI therapy for AF, but this requires larger-scale evaluation.
References
Nery PB, Belliveau D, Nair GM, Bernick J, Redpath CJ, Szczotka A, et al. Relationship Between Pulmonary Vein Reconnection and Atrial Fibrillation Recurrence: A Systematic Review and Meta-Analysis. JACC Clin Electrophysiol. 2016;2:474–83.
Morillo CA, Verma A, Connolly SJ, Kuck KH, Nair GM, Champagne J, et al. Radiofrequency ablation vs antiarrhythmic drugs as first-line treatment of paroxysmal atrial fibrillation (RAAFT-2): a randomized trial. JAMA. 2014;311:692–700.
Lin D, Santangeli P, Zado ES, et al. Electrophysiologic findings and long-term outcomes in patients undergoing third or more catheter ablation procedures for atrial fibrillation. J Cardiovasc Electrophysiol. 2015;26:371–7.
Kuck KH, Hoffmann BA, Ernst S, et al. Impact of Complete Versus Incomplete Circumferential Lines Around the Pulmonary Veins During Catheter Ablation of Paroxysmal Atrial Fibrillation: Results From the Gap-Atrial Fibrillation-German Atrial Fibrillation Competence Network 1 Trial. Circ Arrhythm Electrophysiol. 2016;9:e003337.
Nanthakumar K, Plumb VJ, Epstein AE, Veenhuyzen GD, Link D, Kay GN. Resumption of electrical conduction in previously isolated pulmonary veins: rationale for a different strategy? Circulation. 2004;109:1226–9.
Hussein A, Das M, Riva S, et al. Use of Ablation Index-Guided Ablation Results in High Rates of Durable Pulmonary Vein Isolation and Freedom From Arrhythmia in Persistent Atrial Fibrillation Patients. Circ Arrhythm Electrophysiol. 2018;11:e006576.
Hutchinson MD, Garcia FC, Mandel JE, Elkassabany N, Zado ES, Riley MP, et al. Efforts to enhance catheter stability improve atrial fibrillation ablation outcome. Heart Rhythm. 2013;10:347–53.
Park CI, Lehrmann H, Keyl C, et al. Mechanisms of pulmonary vein reconnection after radiofrequency ablation of atrial fibrillation: the deterministic role of contact force and interlesion distance. J Cardiovasc Electrophysiol. 2014;25:701–8.
Piorkowski C, Eitel C, Rolf S, Bode K, Sommer P, Gaspar T, et al. Steerable versus non steerable sheath technology in atrial fibrillation ablation: a prospective, randomized study. Circ Arrhythm Electrophysiol. 2011;4:157–65.
Chen S, Meng W, Sheng He D, et al. Blocking the pulmonary vein to left atrium conduction in addition to the entrance block enhances clinical efficacy in atrial fibrillation ablation. Pacing Clin Electrophysiol. 2012;35:524–31.
Anter E, Contreras-Valdes FM, Shvilkin A, Tschabrunn CM, Josephson ME. Acute pulmonary vein reconnection is a predictor of atrial fibrillation recurrence following pulmonary vein isolation. J Interv Card Electrophysiol. 2014;39:225–32.
Porta-Sánchez A, Magtibay K, Nayyar S, Bhaskaran A, Lai PFH, Massé S, et al. Omnipolarity applied to equi-spaced electrode array for ventricular tachycardia substrate mapping. Europace. 2019;21:813–21.
Kanamori N, Kato T, Sakagami S, Saeki T, Kato C, Kawai K, et al. Optimal lesion size index to prevent conduction gap during pulmonary vein isolation. J Cardiovasc Electrophysiol. 2018;29:1616–23.
Whitaker J, Fish J, Harrison J, et al. Lesion Index-Guided Ablation Facilitates Continuous, Transmural, and Durable Lesions in a Porcine Recovery Model. Circ Arrhythm Electrophysiol. 2018;11:e005892.
Mattia L, Crosato M, Indiani S, Causin E, Licciardello C, Maria Squasi PA, et al. Prospective Evaluation of Lesion Index-Guided Pulmonary Vein Isolation Technique in Patients with Paroxysmal Atrial Fibrillation: 1-year Follow-Up. J Atr Fibrillation. 2018;10:1858.
Lin C-Y, Te ALD, Lin Y-J, et al. High-resolution mapping of pulmonary vein potentials improved the successful pulmonary vein isolation using small electrodes and inter-electrode spacing catheter. Int J Cardiol. 2018;272:90–6.
Anter E, Tschabrunn CM, Josephson ME. High-Resolution Mapping of Scar-Related Atrial Arrhythmias Using Smaller Electrodes With Closer Interelectrode Spacing. Circ Arrhythm Electrophysiol. 2015;8:537–45.
Masuda M, Fujita M, Iida O, Okamoto S, Ishihara T, Nanto K, et al. The identification of conduction gaps after pulmonary vein isolation using a new electroanatomic mapping system. Heart Rhythm. 2017;14:1606–14.
Tedrow UB, Stevenson WG. Recording and interpreting unipolar electrograms to guide catheter ablation. Heart Rhythm. 2011;8:791–6.
Iso K, Watanabe I, Kogawa R, Okumura Y, Nagashima K, Takahashi K, et al. Wavefront direction and cycle length affect left atrial electrogram amplitude. J Arrhythm. 2017;33:269–74.
Garcia-Bolao I, Ballesteros G, Ramos B, et al. Identification of pulmonary vein reconnection gaps with high-density mapping in redo atrial fibrillation procedures. Europace. 2018;20:f351–8.
McGarry TJ, Narayan SM. The Anatomical Basis of Pulmonary Vein Reconnection After Ablation for Atrial Fibrillation: Wounds That Never Felt a Scar? J Am Coll Cardiol. 2012;59:939–41.
Kowalski M, Grimes MM, Perez FJ, Kenigsberg DN, Koneru J, Kasirajan V, et al. Histopathologic Characterization of Chronic Radiofrequency Ablation Lesions for Pulmonary Vein Isolation. J Am Coll Cardiol. 2012;59:930–8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr Chow receives research grant funding from Abbott and Boston Scientific Inc.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Figure S1.
(DOCX 1714 kb)
Rights and permissions
About this article
Cite this article
Papageorgiou, N., Karim, N., Williams, J. et al. Initial experience of the High-Density Grid catheter in patients undergoing catheter ablation for atrial fibrillation. J Interv Card Electrophysiol 63, 259–266 (2022). https://doi.org/10.1007/s10840-021-00950-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-021-00950-y