Abstract
Ultrasonication has drawn an increasing attention as one of cell disruption methods for extracting cellular compounds or controlling algal blooms. However, the effects of biological and physical properties of microalgae on cell disruption were not well understood. In this work, cell disruption of six microalgae, namely, Chlamydomonas reinhardtii, Chlorella pyrenoidosa, Microcystis aeruginosa (three strains: PCC 7806, FACHB 469, and FACHB 1343), and Synechococcus elongatus, was compared mutually based on their characteristics induced by a low-frequency ultrasound (35 kHz, 0.043 W mL−1). Results showed that the most sensitive strain was C. reinhardtii which has a hydroxyproline-rich-glycoproteins cell wall and a larger cell size (normally 10 μm in diameter). More than 80% of the cells of C. reinhardtii were ruptured after sonication for 5 min. In comparison, C. pyrenoidosa, a cellulose-rich-wall algal species with a medium size of 4–6 μm, and M. aeruginosa FACHB 1343, a peptidoglycan-wall species with a smaller average size of 2.3 μm, were highly resistant to ultrasound. Only 7.5 and 7.7% of cell disruption were achieved for C. pyrenoidosa and M. aeruginosa FACHB 1343, respectively, when they were sonicated for 60 min. Declumping effect was dominant in these strains. This suggested that cellulose-rich-wall algal species might be much more resistant than hydroxyproline-rich glycoproteins, and peptidoglycan-wall species to sonication. It also revealed that the larger cell size was more susceptible to sonication the cell would be. This research provides useful insights into choosing the low-cost microalgae for extraction or controlling specific microalgal blooms in water systems using ultrasound.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Microalgae have been researched for a large range of fine chemicals and bulk products since the 1960s (i.e., fat, β-carotene, polyunsaturated fatty acids, natural dyes, antioxidants, and high-value bioactive compounds, etc.) (Spolaore et al. 2006; Raja et al. 2008; Borowitzka 2013a). Meanwhile, interest in using microalgae for production of renewable energy has increased since the first period of oil crisis (Mata et al. 2010; Borowitzka 2013b). However, these components, including most of the high-value compounds, are generally circumscribed by cell membranes and/or cell walls, but the cell boundaries are usually quite dense and intact. The result is that these materials are not readily available for extraction from the whole cells (Lee et al. 2012). The acquisition efficiency of cellular inclusions could be improved by up to several times using disrupted cells (Lee et al. 2010; Keris-Sen et al. 2014; Yap et al. 2014). Therefore, cell disruption is identified as an effective pretreatment for extraction (Lee et al. 2015).
Recently, ultrasonication has received an increasing attention as one of the cell disruption methods or an effective assistance for extraction because of its advantages (Furuki et al. 2003; Wang et al. 2014). Firstly, it does not require an extra chemical addition as compared to chemical processes, and secondly, it does not require the process of downstream separation as compared to bead milling. For low-frequency ultrasound, the shear force and free radicals produced from cavitation-induce cell disruption (Hao et al. 2004; Rajasekhar et al. 2012b). More specifically, when ultrasound passes through a cell suspension, its rapid and intense compression/expansion cycles generate small bubbles via overcoming the liquid’s tensile strength and then tearing it (Suslick 1989). These bubbles could result in transient cavitation when they undergo unsteady oscillations and eventually implode with localized, forced shear streams and “hot points”, which are points of extremely high temperature and high pressure (Lee et al. 2012). The transient cavitation can also trigger the production of free radical via decomposing water molecules (Luo et al. 2011).
Although ultrasound for cell disruption or controlling algal blooms has been extensively studied, most studies have focused on the optimization of ultrasound parameters, such as frequency and intensity (Tang et al. 2003; Ma et al. 2005; Joyce et al. 2010, 2014; Yamamoto et al. 2015; Kurokawa et al. 2016). The effect of biological and physical properties of microalgae on ultrasonic disruption is not yet well understood. Several reports have discovered that the susceptibility of cells to ultrasound varies with the species treated (Rajasekhar et al. 2012a). Tang et al. (2004) sonicated two cyanobacteria species, Microcystis aeruginosa (gas-vacuolated) and Synechococcus sp. (gas-vacuole negative), and found that only cells of M. aeruginosa were significantly damaged by ultrasound due to the presence of gas vacuoles in their cells. However, cells in these two cyanobacterial strains are also markedly distinct in terms of cell size, and the effect of cell size on ultrasonic damage was not discussed in their work. Similarly, Purcell et al. (2013) estimated the effectiveness of sonication on control of growth of four algal species which had a diversity of morphologies and were from three algal divisions. They revealed that Aphanizomenon flos-aquae (filamentous cyanobacterium) and Melosira sp. (chain-forming diatom) were much more susceptible to ultrasound than M. aeruginosa (unicellular cyanobacterium) and Scenedesmus subspicatus (colonial green alga). The difference in cell shape (filamentous/non-filamentous) was considered as one of the probable reasons for the different levels of susceptibility to sonication. In addition, Greenly and Tester (2015) suggested that cell size and wall composition might have a significant impact on cell lysis efficiency. However, it was difficult to find out which of these properties were the valid and working factors when numerous variables (different properties) were present synchronously in the treated strains. Moreover, explicit comparisons were not implemented between these different species in these documents above. Similar researches in bacteria were also ambiguous. Some reports showed that the larger and rod-shaped bacteria were more susceptible than smaller and coccus-shaped ones (Jacobs and Thornley 1954). Conversely, Cameron (2007) did not observe a direct influence of cell size and shape on ultrasonic disruption. Therefore, more investigations in this topic are needed to be conducted and to further serve the commercial ultrasonic application in algal utilization.
In order to study how ultrasonic disruption of algae cells might be affected by some of their biological (species, cell wall structure, or composition) and physical (cell shape, cell size, and arrangement) properties, six microalgae strains were treated in this work, which included two green algae (Chlamydomonas reinhardtii FACHB 359 and Chlorella pyrenoidosa FACHB 5) and four cyanobacterial strains (M. aeruginosa PCC 7806, FACHB 469 and FACHB 1343; Synechococcus elongatus PCC 7942). These microorganisms were used due to a variety of their comparable properties. In addition, M. aeruginosa is blooming in eutrophic freshwater lakes (Tan et al. 2009). It thereby could be a cheap feed stock for hydrogen or bioethanol products after ultrasonic pretreatment. Synechococcus also has been observed ubiquitously in freshwater and seawater (Scanlan et al. 2009; Callieri 2010). The two green algae have been investigated broadly as promising feed stocks for health products or biofuels production (Görs et al. 2010; Bigelow et al. 2014).
Materials and methods
Algal cultures and cell suspensions preparation
Microcystis aeruginosa PCC 7806 and Synechococcus elongatus PCC 7942 were kindly provided by the Nanjing Institute of Geography and Limnology, Chinese Academy of Sciences. Chlorella pyrenoidosa FACHB 5, Chlamydomonas reinhardtii FACHB 359, M. aeruginosa FACHB 469, and Microcystis aeruginosa FACHB 1343 were from the Freshwater Algae Culture Collection at the Institute of Hydrobiology (FACHB), Chinese Academy of Sciences. Cyanobacterial strains and C. pyrenoidosa were grown in 250-mL Erlenmeyer flasks with the standard BG-11 medium, whereas C. reinhardtii was cultured in flasks with SE medium (Bold and Parker 1962). Cultures were grown in a sterile illumination incubator at 25 ± 0.5 °C under 30 μmol photons m−2 s−1 light intensity with a light:dark cycle of 12 h:12 h. All algal cells were collected at stationary phase through centrifuging (5000×g for 10 min at 4 °C), and then were re-suspended in fresh medium. Well-mixed algal suspensions of M. aeruginosa, C. pyrenoidosa, and C. reinhardtii were prepared with similar cell concentrations at 1.25–1.33 × 107, 1.23 × 107, and 1.29 × 107 cells mL−1, respectively. The cell concentration was determined via cell counting under a light microscope using a hemocytometer. In comparison, the suspension of S. elongatus was prepared with an optical density of 1.0 at 680 nm (Ma et al. 2005). This measurement was not done using the microscope because of the limited accuracy to acquire the absolute cell number of such small cells.
Ultrasonic equipment
In this work, an ultrasonic apparatus (bath-type) was used, which was equipped with a disk-type transducer (35 kHz; DAS Corp., Hangzhou China) (Fig. 1). The transducer was fixed centrally at the bottom of the cylindrical stainless steel tank (with a maximal capacity of 200 mL). The tank was surrounded by a cooling jacket made of a glass to keep the temperature of the cell suspension below 25 °C. The calorimetric method was used to measure the accurate acoustic power which passed through the system (Mason et al. 1992). Briefly, in order to determine the stable power output of this device, 150 mL of distilled water was sonicated in the tank for 15 min with the cooling system not in operation, and the temperature was recorded every 5 min using a digital thermometer. The top of the device’s sample tank was covered using a lid during the measurement of the power output in order to minimize the heat loss. This measurement was carried out in triplicate and the data then were averaged. The acoustic power was computed using the equation below:
where, I is the ultrasonic intensity (W mL−1); T is the temperature of treated liquid (°C) and t is processing time (seconds); Cp indicates the heat capacity of water at 25 °C (J kg−1 °C−1); M is the mass of water (kg); and V is the volume of treated liquid (mL). After the measurement and calculation, it was confirmed that I was 0.043 W mL−1 in this system.
Experimental design
For M. aeruginosa and C. pyrenoidosa, the 150 mL of suspensions prepared above were sonicated for 60 min in the ultrasonic apparatus. In the course of treatment, 2 mL of the suspension was sampled at 0, 5, 10, 15, 20, 30, 40, 50, and 60 min, respectively, for cell counting and microscopic image analysis. Before sampling, the suspension was stirred to ensure better sampling. Three other 150-mL suspensions were treated for 0, 30, and 60 min, respectively, under the same ultrasonic conditions to assess the changes of the particle size distribution (PSD). In the case of C. reinhardtii, due to its high susceptibility to ultrasound, 150 mL of suspension was treated and analyzed as described above, but samples were taken at 0, 1, 2, 3, 4, 5, 10, 15, 20, 30, 40, 50, and 60 min, respectively. Other three suspensions of C. reinhardtii were treated for 0, 15, and 30 min, respectively, for the purpose of PSD analysis. Suspension of the S. elongatus was treated identically to the M. aeruginosa and the C. pyrenoidosa, but its cell reduction was measured by the decrease of OD680 of the suspension using a spectrophotometer. All experiments were conducted in triplicate.
Microscopic analysis and quantifying cell disruption
Morphological characteristics of these microalgae were observed while images were taken at different levels of disruption using a light imaging microscope (Carl Zeiss, Germany) which was equipped with a camera lens (AxioCam ICc 3) and connected with a computer. These microscopic images were analyzed through a software; AxioVision 4.8.2.0 (Carl Zeiss). The cell size of these strains except for S. elongatus was analyzed using the software. For S. elongatus, cell size was determined by PSD analysis and further referenced in literature.
Cell counting used to calculate cell disruption was conducted using a hemocytometer under an optical microscope (Olympus CX31, Japan). More than six replicate cell counts were made on each sample, and the average was computed. The cell disruption, CDt (%), was calculated using the following equation:
where, C 0 is the initial cell concentration, and C t is the cell concentration after sonication for t min (For S. elongatus, C indicates the value of OD680).
PSD analysis
A Malvern Mastersizer 2000 (Malvern Instruments Ltd., UK) was used to measure the particle size distribution of these algae. The dispersant was Milli-Q water with a refractive index of 1.33. A refractive index of 1.45 was used for the green algae (Spiden et al. 2013b) and 1.40 for the cyanobacterial strains (Li et al. 2014). A light absorption index of 0.1 was used for all the strains (Li et al. 2014). It should be noted that the Mastersizer 2000 software obtains the particle size value using a prerequisite assumption that the algal cells are spherical.
Statistical analysis
Significant differences in cell disruption among algae strains were compared through the Wilcoxon signed-rank test (* indicates P < 0.05, ** indicates P < 0.01). Pearson correlation analysis was conducted to describe the correlation between cell size and cell disruption (P < 0.05 was considered as significant). All statistical analyses were completed using SPSS 19.0. The values in this paper are expressed as mean ± standard deviation (SD).
Results
Some biological and physical properties of the treated microalgae
In this study, the major aim is to investigate the effect of several biological (species, major structures of the cell wall) and physical features (shape, cell size, and arrangement) of these microalgae on cell disruption induced by ultrasound. These features obtained by light microscopic analysis and literature review were displayed and in Table 1.
Cells of C. reinhardtii are spherical and unicellular with two flagella, whose diameters are approximately 10 μm (Table 1). They can also form large aggregates or colonies (up to 100 μm in size, Fig. 3a). Chlorella pyrenoidosa has a diameter which is about a half of the C. reinhardtii. Some aggregates or colonies were also observed in the suspension (Fig. 3c).
Four cyanobacterial strains of the genera of Microcystis and Synechococcus were employed in this case. This is because their characteristics were pronouncedly different from the green algae. Actually, they are Gram-negative microorganisms. Furthermore, they possessed a range of comparable properties. For example, the M. aeruginosa strains (PCC 7806, FACHB 469 and FACHB 1343) shared a similar coccoid shape (Figs. 2a, b and 3b) and biological features, but they were significantly different in cell size (ranging from 2.3 to 5.3 μm in diameter) (Table 1). In their axenic cultures, stains of PCC 7806 and FACHB 469 mainly presented as single cells with a few small aggregates or colonies, whereas large aggregates were observed in FACHB 1343 suspensions (Figs. 2a, b and 3b). In comparison, cells in the strain of S. elongatus PCC 7942 shared rod-like shapes with the smallest cell size (Table 1). They were able to form micro-colonies or aggregates of 5–100 cells (Fig. 2c).
Micrographs of algal cells with various ruptured extents. C. reinhardtii (control (a), sonication for 15 min (b), sonication for 30 min (c)); M. aeruginosa PCC 7806 (control (d), sonication for 30 min (e), sonication for 60 min (f)); and C. pyrenoidosa (control (g), sonication for 30 min (h); sonication for 60 min (i))
Ultrasonic disruption on algal cells
Disruption efficiency varied among algal species (Fig. 4). More than 80% of the cells in C. reinhardtii were ruptured after sonication for 5 min. When the suspension was treated for 15 min, most of the cells were no longer intact as observed in the micrographs (Fig. 3b). And a great number of collapsed cells and cell debris were captured. Moreover, there was no visible lumpy biomass when exposure time increased to 30 min (Fig. 3c).
However, the high efficiency of cell disruption observed in C. reinhardtii was not observed in M. aeruginosa and C. pyrenoidosa. In the case of M. aeruginosa, cell reduction increased in a linear model as a function of sonication time (R2 = 0.97, P < 0.01, n = 9; Fig. 4). After treatment for 30 min, 39.9% of Microcystis cells were raptured. The maximal cell disruption at the end of the treatment was 69.0%. Cells in the suspension had complete and clear cell edges at the beginning, but they turned out to be ambiguous and crippled after sonication (Fig. 3d–f). More fragmented cells were observed with increased exposure time.
Chlorella pyrenoidosa was highly resistant to sonication. Despite the fact that the suspension was treated for 60 min, the cell rupture was only found to be 7.5% (Fig. 4). Evidence from micrographs was well in agreement with the result of cell counting. The declumping effect rather than the rupturing effect would be dominant in the suspension because no visible fragmented cells were observed (Fig. 3h, i).
In this study, PSDs of microalgae with different disruption extents were presented in Fig. 5. The PSD of C. reinhardtii was mono-modal before sonication, then bimodal after exposure for 15 min with a new distribution of small sizes between 3.8–10 μm (accounted for 20.1% of the total mass of particles). On the other hand, D90, D50, and D10 of the suspension reduced significantly (P < 0.05), indicating that not only were aggregates of this microorganism dispersed, but also cells were fragmented in the course of sonication. These effects were reinforced in the continuing process. The D90 decreased to 1.3 μm when the suspension was treated for 30 min, indicating that almost all the cells were disrupted (Fig. 5a). The result of PSD analysis was highly consistent with those of cell counting and microscopy (Figs. 3a–c, and 4).
Effect of ultrasonic treatment on the particle size distributions in different microalgae suspensions. D x is particle size (μm) indicting that x% of the total mass of particles were smaller. Therefore, D10, D50, and D90 represent minimum, median, and maximum particle sizes in suspensions, respectively. Significant differences are marked with different letters (P < 0.05)
The PSD of M. aeruginosa PCC 7806 suspension had a bimodal distribution, indicating that there were aggregates (up to 17.4–363.1 μm, accounting for 25% of the total) at the beginning. During the first half of the exposure period, declumping effect coupled with cell disruption (39.9% of disruption was achieved according to the cell counting) was considered as the main reason for the significant decrease in D90 and D10 (Fig. 5b). The rupturing effect was predominant within the next 30 min, since the D90 reduced to 4.5 μm from 6.3 μm which is nearly the diameter of a single cell (Table 1, Fig. 5b). In the case of C. pyrenoidosa, in contrast, declumping effect was predominant during the whole period of treatment. The PSD and its parameters (D90, D50, and D10) of the C. pyrenoidosa suspension insignificantly changed when the suspension was sonicated from 30 to 60 min (Fig. 5c).
Effect of cell size and shape on disruption
To investigate the effect of cell size and shape on ultrasonic disruption, three M. aeruginosa stains (PCC 7806, FACHB 469 and FACHB 1343) with various diameters and a strain of S. elongatus (PCC 7942) were sonicated under the same conditions. Several of their characteristics are displayed in Table 1. Fig. 6 is a plot of the ultrasonic disruption of these microorganisms as a function of sonication time. The order of disruption efficiency was PCC 7806 > FACHB 469 > PCC 7942 > FACHB 1343, which was in line with the decreasing tendency of cell size except for PCC 7942 (Fig. 6). The cells of PCC 7942 are not only smaller in size but different in shape (rod-like) compared to the other strains (Table 1).
The PSD of treated cyanobacterial strains with different levels of rupture were drawn in Fig. 7. The dynamic trend of particle size in the suspension of FACHB 469 was similar to that of PCC 7806 described above (Figs. 5b, and 7a). Declumping effect and cell disruption were detected in this suspension during treatment. However, according to PSD analysis, only declumping effect was determined in suspensions of FACHB 1343 and PCC 7942, which was proved by their almost exact initial values of D10 (Fig. 7b–c). In other words, there were no additional smaller particles in suspensions of FACHB 1343 and PCC 7942 after sonication. In addition, PSD of PCC 7942 suspension displayed a bimodal distribution with two peaks at 1.0 and 0.18 μm, which could represent the long and short axes of the rod-like cell, respectively.
Effect of ultrasonic treatment on the particle size distributions in several cyanobacterial algae. D x is defined identically as in Fig. 5. Significant differences are marked with different letters (P < 0.05)
Discussion
Six algal strains were sonicated to investigate the effect of biological and physical properties of algae on ultrasonic disruption. Cell disruption or damage was estimated by four techniques: cell counting, microscopic analysis, PSD of suspensions, and spectrophotometry (the last was used only to quantify cell disruption of S. elongatus PCC 7942. In previous studies, cell counting was used extensively to examine cell damage induced by ultrasound (Ahn et al. 2003; Wu et al. 2012) and even was considered as the only reliable method for quantitative comparisons of all microalgae (Spiden et al. 2013b). As a visualisation tool, microscopic images are also usually used to analyze microorganisms under a variety of treatments (Hao et al. 2004; Gao et al. 2014b). In addition, particle sizes of Microcystis colonies and microorganisms have been determined by using PSD analysis (Spiden et al. 2013a; Gao et al. 2014a). However, Spiden et al. (2013b) reported that the PSD is derived from light scattering data using the assumption of spherical particles, and it needs to be interpreted and used with caution. In this study, all morphologies of the tested microalgal cells were spherical or nearly spherical except for S. elongatus PCC 7942 (Figs. 2 and 3 a, d and g). Furthermore, the result of PSD was well in line with those of microscopic analysis and cell counting (see in “Ultrasonic disruption on algal cells” and “Effect of cell size and shape on disruption” for details). This suggested that results acquired from this work could be reliable.
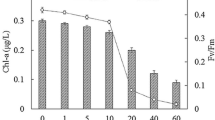
However, in the case of S. elongatus PCC 7942, the result of PSD was inconsistent with that of cell disruption (Figs. 6 and 7c). It should be noted that its cell disruption was calculated from the reduction of OD680 measured by spectrophotometry, since it was difficult to distinguish such small intact cell or tiny cell debris under a light microscope. Although many previous researchers have used this technique to quantify cell disruption, a discovery made by us (unpublished) showed that it was not ideal for all microalgae species due to the optical density’s complexity, which consists of light scattering, absorption, and refraction or reflection. Yamamoto et al. (2015) also reported that OD680 of treated suspension did not directly indicate the algae cell disruption because it interrelated with the chlorophyll content. In addition, the PSD of S. elongatus PCC 7942 should be further confirmed by other more advanced techniques such as TEM due to the inconsistency of its rod-like shape and the assumption of the Mastersizer 2000 mentioned above. Therefore, due to the complexity and uncertainty of the results of S. elongatus PCC 7942, the effect of its properties on ultrasonic cell disruption is not stated in the following discussion.
Effect of cell wall structures or compositions on ultrasonic disruption
The cell wall is the dominant barrier protecting microalgae from being damaged by external pressures. Structure or composition of the wall and its thickness are recognized as the two major factors that impact its strength. Generally the thicker the wall thickness is, the more the resistance of the cell wall could be to pressures (Joyce et al. 2014). Although the wall thickness of the strains tested in this work was not measured, it was estimated to account for approximately 5% of a cell size (Kurokawa et al. 2016). Accordingly, the cells of the C. reinhardtii should have the thickest wall and the highest resistance to sonication because of their larger cell size among all the treated algae (Table 1). The greatest disruption efficiency, however, was gained from the C. reinhardtii (Fig. 4). The thickest wall therefore fails to cause the greatest resistance to sonication if that evaluation on wall thickness is correct. Nevertheless, the effect of wall thickness on cell disruption merits further investigation.
Cell wall structures or compositions of microalgae used in this work have been reviewed briefly in Table 1. The two green algae, C. reinhardtii and C. pyrenoidosa, possess different cell walls in terms of structure or composition. Hydroxproline-rich glycoproteins and crystalline layers compose the major structure or composition of the C. reinhardtii wall, but there is no cellulose or other polysaccharides (Voigt 1988; Adair and Snell 1990; Domozych 2011). For instance, 17–30 glycoproteins were found in their wall complexes, arranged into interlocking fibrillar and granular elements. Crystalline layers that provide strength also made up their walls. On the other hand, C. pyrenoidosa generally consists of cellulose arranged in fibrils and a polysaccharide-rich matrix sheath (Northcote et al. 1958). Results of cell disruption and PSD analysis showed that C. reinhardtii was much more sensitive than C. pyrenoidosa to sonication (Figs. 4 and 5a, c). Specifically, the proportion of intact cells in the C. reinhardtii suspension decreased by 80% after sonication only for 5 min. Its D90 was smaller than 1.3 μm when the suspension was treated for 30 min. In comparison, even though the suspension of C. pyrenoidosa was sonicated for 60 min, the cell disruption was found to be only 7.5% (Fig. 4). These results suggested that cellulose-rich wall could be stronger than hydroxyproline-rich-glycoprotein wall in the ultrasonic field. A similar experiment conducted by Purcell et al. (2013), suggested that Melosira sp. (a diatom) has a higher vulnerability to the acoustic field than S. subspicatus (a green alga); probably due to differences in their wall structure. However, it should be noted that significant difference in cell sizes, ranging from 3.4 to 13 μm in diameter, between two green algal strains treated in this work was observed (Fig. 3a, g), which could be also responsible for the various disruption efficiencies.
In order to further investigate the effect of cell wall structure or composition on acoustic rupture, three strains (C. pyrenoidosa, M. aeruginosa PCC 7806, and M. aeruginosa FACHB 469) which shared a similar cell shape (coccoid-like) and cell size (4.4–5.3 μm in diameter), but were diverse in wall structure, were treated by the same ultrasound. Strains of M. aeruginosa are Gram-negative microorganisms and possess peptidoglycan and lipopolysaccharide wall (Tang et al. 2004). Results showed that C. pyrenoidosa was also more resistant than M. aeruginosa. The two M. aeruginosa strains achieved a similar cell disruption and a similar PSD model (Figs. 4; 5b, c; and 7a). Rajasekhar et al. (2012a) reported a similar finding that under the same sonication conditions, the order of decreasing growth inhibition of the two algae was: M. aeruginosa > Chlorella sp.. Therefore, a cellulose-rich wall might be highly more indestructible than a peptidoglycan-rich wall in the acoustic field. However, one still should be cautious to note such a conclusion because their internal cell structures could also be a reason to explain these different levels of susceptibility. For instance, gas vesicles in cyanobacterial cells (M. aeruginosa, Aphanizomenon flos-aquae, and Anabaena flos-aquae, etc.) resulted in high susceptibility of these species to ultrasound (Purcell 2009). Nevertheless, the cell wall structure or composition combined with the internal cell structure could be an important factor to explain the various levels of susceptibility of these algae to sonication.
Effect of cell size on ultrasonic disruption
The effect of cell size on disruption was investigated with three M. aeruginosa strains (PCC 7806, FACHB 469, and FACHB 1343) after eliminating interferences from other characteristics as far as possible. They had similar biological characteristics and cell shapes, but were significantly different in cell sizes and cell arrangements (Table 1, Figs. 2a, b and 3d). Despite larger aggregates or colonies observed in the strain FACHB 1343, declumping effect was completed without remarkable cell disruption in the first half of the sonicated period (Fig. 6), and there was no detectable cell debris in the suspension after treatment for 60 min (Fig. 7b), indicating ultrasonic disruption was insignificantly affected by the cell arrangement. Conversely, intense rupturing effect and declumping effect were shown synchronously in the suspensions of PCC 7806 and FACHB 469 during the first half of the period, and many smaller particles rather than intact single cells were detected in these suspensions after sonication for 60 min (Figs. 5a, 6, and 7b). These results demonstrated that the order of disruption efficiency was directly related to the cell size, which was PCC 7806 > FACHB 469 > FACHB 1343 (Table 1 and Fig. 6). Furthermore, even if numerous biological features were included (meaning that all of these algae here were taken into account except for PCC 7942 due to the reasons mentioned above), cell disruption was still positive correlated to cell size (r > 0.89, P < 0.05) (Fig. 8). This is in agreement with the report conducted by Greenly and Tester (2015), who demonstrated that Nannochloropsis oculata (CCMP 525), a 2-μm marine green alga, was more indestructible than the other bigger microalgae.
The relationship of cell size and ultrasonic disruption efficiency. a, b, and c indicate the suspensions which were sonicated for 15, 30, and 60 min, respectively. Symbols are white square C. reinhardtii; black square C. pyrenoidosa; white circle M. aeruginosa PCC 7806; gray circle M. aeruginosa FACHB 469; black circle M. aeruginosa FACHB 1343
In conclusion, six algal strains with a variety of biological and physical properties were sonicated, and cell disruption efficiency varied with the microalgae treated. Chlamydomonas reinhardtii was the most susceptible strain to ultrasound followed by M. aeruginosa PCC 7806, M. aeruginosa FACHB 469, and S. elongatus PCC 7942. C. pyrenoidosa and M. aeruginosa FACHB 1343 were not easily ruptured, and declumping effect, instead of cell disruption was the main result of sonication. Cell wall structures or compositions and cell sizes significantly affected the level of susceptibility to sonication. Cellulose-rich-wall algal species might be a lot more resistant than hydroxyproline-rich-glycoproteins, and peptidoglycan cell-walled species in the acoustic field. In addition, there was a positive correlation between ultrasonic cell disruption and cell size (P < 0.05, n = 5), that is the larger the cell size is, the more susceptible it would be to sonication.
References
Adair WS, Snell WJ (1990) The Chlamydomonas reinhardtii cell wall: structure, biochemistry, and molecular biology. In: Adair WS (ed) Organization and assembly of plant and animal extracellular matrix. Academic, San Diego, pp 15–84
Ahn C-Y, Park M-H, Joung S-H, Kim H-S, Jang K-Y, Oh H-M (2003) Growth inhibition of cyanobacteria by ultrasonic radiation: laboratory and enclosure studies. Environ Sci Technol 37:3031–3037
Bigelow TA, Xu J, Stessman DJ, Yao L, Spalding MH, Wang T (2014) Lysis of Chlamydomonas reinhardtii by high-intensity focused ultrasound as a function of exposure time. Ultrason Sonochem 21:1258–1264
Bold HC, Parker BC (1962) Some supplementary attributes in the classification of Chlorococcum species. Arch Mikrobiol 42:267–288
Borowitzka MA (2013a) High-value products from microalgae—their development and commercialisation. J Appl Phycol 25:743–756
Borowitzka MA (2013b) Energy from microalgae: a short history. In: Borowitzka MA, Moheimani NR (eds) Algae for biofuels and energy. Springer, Dordrecht, pp 1–15
Callieri C (2010) Single cells and microcolonies of freshwater picocyanobacteria: a common ecology. J Limnol 69:257–277
Cameron M (2007) Impact of low-frequency high-power ultrasound on spoilage and potentially pathogenic dairy microbes. PhD Thesis, Stellenbosch University, South Africa
Domozych DS (2011) Algal cell walls. In: Pettis G (ed) eLS. John Wiley & Sons, Ltd, Chichester
Furuki T, Maeda S, Imajo S, Hiroi T, Amaya T, Hirokawa T, Ito K, Nozawa H (2003) Rapid and selective extraction of phycocyanin from Spirulina platensis with ultrasonic cell disruption. J Appl Phycol 15:319–324
Gao S, Hemar Y, Ashokkumar M, Paturel S, Lewis GD (2014a) Inactivation of bacteria and yeast using high-frequency ultrasound treatment. Water Res 60:93–104
Gao S, Lewis GD, Ashokkumar M, Hemar Y (2014b) Inactivation of microorganisms by low-frequency high-power ultrasound: 1. Effect of growth phase and capsule properties of the bacteria. Ultrason Sonochem 21:446–453
Görs M, Schumann R, Hepperle D, Karsten U (2010) Quality analysis of commercial Chlorella products used as dietary supplement in human nutrition. J Appl Phycol 22:265–276
Greenly JM, Tester JW (2015) Ultrasonic cavitation for disruption of microalgae. Bioresource Technol 184:276–279
Hao H, Wu M, Chen Y, Tang J, Wu Q (2004) Cavitation mechanism in cyanobacterial growth inhibition by ultrasonic irradiation. Colloid Surface B 33:151–156
Jacobs SE, Thornley MJ (1954) The lethal action of ultrasonic waves on bacteria suspended in milk and other liquids. J Appl Bacteriol 17:38–56
Joyce EM, Wu X, Mason TJ (2010) Effect of ultrasonic frequency and power on algae suspensions. J Environ Sci Health A 45:863–866
Joyce EM, King PM, Mason TJ (2014) The effect of ultrasound on the growth and viability of microalgae cells. J Appl Phycol 26:1741–1748
Keris-Sen UD, Sen U, Soydemir G, Gurol MD (2014) An investigation of ultrasound effect on microalgal cell integrity and lipid extraction efficiency. Bioresource Technol 152:407–413
Kurokawa M, King PM, Wu X, Joyce EM, Mason TJ, Yamamoto K (2016) Effect of sonication frequency on the disruption of algae. Ultrason Sonochem 31:157–162
Lee J-Y, Yoo C, Jun S-Y, Ahn C-Y, Oh H-M (2010) Comparison of several methods for effective lipid extraction from microalgae. Bioresource Technol 101:S75–S77
Lee AK, Lewis DM, Ashman PJ (2012) Disruption of microalgal cells for the extraction of lipids for biofuels: processes and specific energy requirements. Biomass Bioenergy 46:89–101
Lee AK, Lewis DM, Ashman PJ (2015) Microalgal cell disruption by hydrodynamic cavitation for the production of biofuels. J Appl Phycol 27:1881–1889
Li M, Zhu W, Gao L (2014) Analysis of cell concentration, volume concentration, and colony size of Microcystis via laser particle analyzer. Environ Manag 53:947–958
Luo J, Fang Z, Richard L, Smith J, Qi X (2011) Fundamentals of acoustic cavitation and sonochemistry. In: Ashokkumar M (ed) Theoretical and experimental sonochemistry involving inorganic systems. Springer, Dordrecht, pp 1–29
Ma B, Chen Y, Hao H, Wu M, Wang B, Lv H, Zhang G (2005) Influence of ultrasonic field on microcystins produced by bloom-forming algae. Colloid Surface B 41:197–201
Mason T, Lorimer J, Bates D (1992) Quantifying sonochemistry: casting some light on a ‘black art’. Ultrasonics 30:40–42
Mata TM, Martins AA, Caetano NS (2010) Microalgae for biodiesel production and other applications: a review. Renew Sust Energ Rev 14:217–232
Northcote DH, Goulding KJ, Horne RW (1958) The chemical composition and structure of the cell wall of Chlorella pyrenoidosa. Biochem J 70:391–397
Purcell D (2009) Control of algal growth in reservoirs with ultrasound. PhD Thesis, Cranfield University, UK
Purcell D, Parsons SA, Jefferson B (2013) The influence of ultrasound frequency and power, on the algal species Microcystis aeruginosa, Aphanizomenon flos-aquae, Scenedesmus subspicatus and Melosira sp. Environ Technol 34:2477–2490
Raja R, Hemaiswarya S, Kumar NA, Sridhar S, Rengasamy R (2008) A perspective on the biotechnological potential of microalgae. Crit Rev Microbiol 34:77–88
Rajasekhar P, Fan L, Nguyen T, Roddick FA (2012a) Impact of sonication at 20 kHz on Microcystis aeruginosa, Anabaena circinalis and Chlorella sp. Water Res 46:1473–1481
Rajasekhar P, Fan L, Nguyen T, Roddick FA (2012b) A review of the use of sonication to control cyanobacterial blooms. Water Res 46:4319–4329
Scanlan DJ, Ostrowski M, Mazard S, Dufresne A, Garczarek L, Hess WR, Post AF, Hagemann M, Paulsen I, Partensky F (2009) Ecological genomics of marine picocyanobacteria. Microbiol Mol Biol Rev 73:249–299
Spiden EM, Scales PJ, Kentish SE, Martin GJO (2013a) Critical analysis of quantitative indicators of cell disruption applied to Saccharomyces cerevisiae processed with an industrial high pressure homogenizer. Biochem Eng J 70:120–126
Spiden EM, Yap BHJ, Hill DRA, Kentish SE, Scales PJ, Martin GJO (2013b) Quantitative evaluation of the ease of rupture of industrially promising microalgae by high pressure homogenization. Bioresource Technol 140:165–171
Spolaore P, Joannis-Cassan C, Duran E, Isambert A (2006) Commercial applications of microalgae. Biosci Bioeng 101:87–96
Suslick KS (1989) The chemical effects of ultrasound. Sci Am 260:80–86
Tan X, Kong FX, Zeng QF, Cao HS, Qian SQ, Zhang M (2009) Seasonal variation of Microcystis in Lake Taihu and its relationships with environmental factors. J Environ Sci 21:892–899
Tang J, Wu Q, Hao H, Chen Y, Wu M (2003) Growth inhibition of the cyanobacterium Spirulina (Arthrospira) platensis by 1.7 MHz ultrasonic irradiation. J Appl Phycol 15:37–43
Tang JW, Wu QY, Hao HW, Chen Y, Wu M (2004) Effect of 1.7 MHz ultrasound on a gas-vacuolate cyanobacterium and a gas-vacuole negative cyanobacterium. Colloid Surface B 36:115–121
Voigt J (1988) The lithium-chloride-soluble cell-wall layers of Chlamydomonas reinhardii contain several immunologically related glycoproteins. Planta 173:373–384
Wang M, Yuan W, Jiang X, Jing Y, Wang Z (2014) Disruption of microalgal cells using high-frequency focused ultrasound. Bioresource Technol 153:315–321
Wu X, Joyce EM, Mason TJ (2012) Evaluation of the mechanisms of the effect of ultrasound on Microcystis aeruginosa at different ultrasonic frequencies. Water Res 46:2851–2858
Yamamoto K, King PM, Wu X, Mason TJ, Joyce EM (2015) Effect of ultrasonic frequency and power on the disruption of algal cells. Ultrason Sonochem 24:165–171
Yap BHJ, Crawford SA, Dumsday GJ, Scales PJ, Martin GJO (2014) A mechanistic study of algal cell disruption and its effect on lipid recovery by solvent extraction. Algal Res 5:112–120
Acknowledgements
This work was supported by the Fundamental Research Funds for the Central Universities (2013B32414), National Natural Science Foundation of China (31470507), Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD) and Top-notch Academic Programs Project of Jiangsu Higher Education Institutions (TAPP).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Duan, Z., Tan, X., Guo, J. et al. Effects of biological and physical properties of microalgae on disruption induced by a low-frequency ultrasound. J Appl Phycol 29, 2937–2946 (2017). https://doi.org/10.1007/s10811-017-1178-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-017-1178-3