Abstract
The Amazon forests are under threat from multiple human land uses, but the effect of the different types of land uses on environmental heterogeneity and the α- and β-diversity of aquatic insects remains unclear. We studied how habitat features of streams and aquatic insect diversity in the orders Ephemeroptera, Plecoptera, and Trichoptera (hereafter, EPT) responded to different land uses in the Brazilian Amazon. By sampling and analyzing EPT community data from 83 streams distributed in multiple land uses and land covers, we found that the impact of forest conversion was mixed. Despite contiguous and fragmented forest streams presenting similar environmental conditions, they differed in insect diversity metrics. α-diversity was highest in contiguous forest streams and EPT β-diversity was higher in streams surrounded by livestock farming and primary oil palm plantations. The association between land use and habitat degradation may not be so direct, mainly when streams are inserted into or surrounded by forest fragments. This has important implications because politicians and policymakers often regard forest fragments as degraded landscapes, to justify their conversion to other land uses. Our study shows that forest fragments must be protected and restored to reduce the risks of degrading the ecological condition of Amazonian streams.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Environmental heterogeneity, which here we define as the variation in environmental conditions among sites (Heino et al., 2015), has long been associated with patterns of species distribution (e.g., Levin et al., 2010; Astorga et al., 2014; Massicotte et al., 2015; Zorzal-Almeida et al., 2017). Thus, it is expected that more environmentally heterogeneous sites support a more diverse biological community because species coexistence is facilitated in such environments due to a greater number of different resources (e.g., MacArthur & MacArthur, 1961; Ricklefs, 1977). Stream ecosystems are a good model system to study this relationship because they are highly heterogeneous (Stein et al., 2014; Heino et al., 2015), which contributes to high biological diversity owing to the accumulation of species with different environmental affinities (Southwood, 1977; Muotka et al., 2002; Brown, 2003; Stein et al., 2014). However, human activities modify the landscape around streams, which may alter environmental conditions and heterogeneity among streams and, consequently, affect the biotic communities (Levin et al., 2010; Giam et al., 2015; Cunha & Juen, 2017; Wilkinson et al., 2018).
The main human activities responsible for changing the environmental characteristics of the Amazonian streams are livestock farming, agriculture (e.g., soybean and oil palm), and logging (IMAZON, 2017). These activities result in the loss of riparian vegetation that in turn results in increased solar radiation, water temperature (Caissie, 2006), fine sediment loading in the streambed, greater bank instability that leads to increased erosion and loss of instream habitat, and consequently, a change in energy and organic matter dynamics (Allan, 2004; Roebuck et al. 2019). Furthermore, these activities can decrease stream depth and dissolved oxygen concentration (Dias et al., 2010; Nogueira et al., 2016; Prudente et al., 2017) and increase nutrient and pollutant concentrations (Belsky et al., 1999; García-García et al., 2017).
The importance of riparian vegetation for water quality and biodiversity is well recognized in headwater streams (e.g., Dala-Corte et al., 2019; Turunen et al., 2021); however, there are relatively few studies on the importance of secondary forest and forest fragments in conserving stream habitat and the biota therein (Wang et al., 2020). Under the current scenario of increased deforestation in the Amazon, which in 2021 alone destroyed an area of 1,195,723 km2 (Terra Brasilis, 2022), assessing the importance of forest fragments continuous to streams is essential for directing public policies for the conservation and potential restoration of these fragments.
Previous studies have shown that human land uses can alter riparian and instream conditions (Ligeiro et al., 2013; Murphy & Romanuk, 2014; de Paiva et al., 2017) and cause a decline in species richness (a measure of α-diversity; Whittaker, 1960). For instance, changes in pH, temperature, and turbidity of the water due to land use (Astorga et al., 2011; Cunha et al., 2015) and proximity to roads (de Paiva et al., 2017) decreased the richness of macroinvertebrate species in streams. More recent studies also assess differences in community composition among sites (β-diversity) (e.g., Alahuhta et al., 2017; Leão et al., 2020), but empirical support for the impact of human activities on environmental heterogeneity and β-diversity remains equivocal. Whereas some studies showed that streams in anthropic landscapes were more environmentally homogeneous (Cunha & Juen, 2017) and supported lower β-diversity (Siqueira et al., 2015), others found that β-diversity was greater in these streams (Hawkins et al., 2015; Fugère et al., 2016), likely because anthropically impacted streams were more environmentally heterogeneous (Leão et al., 2020). The relationship between environmental heterogeneity and β-diversity also varied among various studies. For example, some studies demonstrated a positive relationship between environmental heterogeneity and β-diversity of aquatic macroinvertebrates (Astorga et al., 2014) and diatoms (Zorzal-Almeida et al., 2017). In contrast, other studies found no such relationship (Heino et al., 2013; Bini et al., 2014; Grman et al., 2015).
The mechanisms by which human land uses affect freshwater biodiversity are multifaceted and complex, because different land use alterations result in different changes to the aquatic and riparian environment (Dala-Corte et al., 2019). Most of the previous studies conducted in Amazonian streams evaluated the effect of a single land use type, such as oil palm (Cunha & Juen, 2017; Carvalho et al., 2018) or logging (Calvão et al., 2016; Cardoso et al., 2018), whereas only a few have investigated the relative effect of different land uses within a single study with consistent sampling methods (e.g., Montag et al., 2018; Leão et al., 2020). There is an increasing demand for natural resources and an urgent need to implement effective environmental conservation and restoration measures, as natural landscapes are converted to different types of production lands. Therefore, it is important and timely to investigate the mechanisms underlying the impact of multiple human land uses on habitat heterogeneity and α and β-diversity in streams of the Amazon.
We studied how the Amazonian streams habitat features and aquatic diversity (insects of the orders Ephemeroptera, Plecoptera, and Trichoptera; hereafter, EPT) respond to different human land use types, based on four hypotheses: H1. Environmental characteristics of streams are altered in different ways by the different land uses; H2. Streams surrounded by human land uses are environmentally less heterogeneous than streams in continuous forest and forest fragments; H3. EPT α-diversity is lower in streams surrounded by human land uses; H4. EPT β-diversity is lower among streams surrounded by human land uses than among streams in continuous forest and forest fragments. To test our hypotheses, we recorded habitat features and performed EPT sampling in streams in continuous forest and forest fragments (control streams) and streams affected by reduced-impact logging, conventional logging, oil palm plantation, and livestock farming. As we have clarified throughout this introduction, many studies have tested these hypotheses individually. However, none tested them simultaneously, considering many land uses, and using a standardized sampling protocol. Thus, our approach can deepen the discussion and contribution to the field, providing important insights to the conservation and management of stream ecosystems.
Methods
Study site
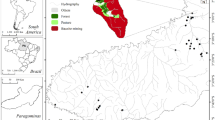
We sampled 83 streams of 1st to 3rd order (sensu Strahler, 1957), with a mean width of 3 m and mean depth of 0.28 m. The streams were distributed in the Northeastern and Southeastern mesoregions of the State of Pará (PA), in an area known as the ‘Arc of Deforestation’ that extends across four Brazilian states (Pará, Mato Grosso, Rondônia, and Acre). The Arc of Deforestation is a region on the Amazon where the agricultural frontier is advancing most rapidly at the expense of forest (INPE, 1997; IBGE, 2011) and this is facilitated by new infrastructure (Hansen et al., 2020). These landscape changes began in the 1970’s due to government financial incentives for farmers to establish plantations in the region (Mahar, 1979; Smith et al., 1995; Aldrich et al., 2012). The sampled streams were in two hydrological units (Capim and Acará basins) in the Atlantic Coast-Northeast hydrological region (ANA, 2010). This hydrological region was defined according to the homogeneity of its geophysiological characteristics (e.g., geomorphology, geology, hydrography, soils, and hydroclimatic factors) (SEMAS, Resolution No. 04 of September 2008). All streams in our study are located in the dystrophic yellow latosol soil type, with the predominant texture being clayey soil (IBGE, 2006).
We sampled streams in natural contiguous forest (n = 13; NCF), forest fragment (n = 8; FF), conventional logging (n = 9; CL), reduced-impact logging (n = 21; RIL), oil palm plantation (Elaeis guineensis Jacq.) (n = 22), and livestock farming areas (n = 10; LIV). We sampled streams in these land cover and land use types because they were the most predominant types in the studied region (Fig. 1). In reduced-impact logging (RIL) sites, logging cycles occur after 30 years of forest regeneration and riparian vegetation along streams is preserved. By contrast, in conventional logging (SL) sites, timber is removed without regard for the forest or riparian protection, which results in riparian vegetation impairment and accumulation of woody debris on the forest surface (for more details, see Calvão et al., 2016).
Geographic locations of the 83 stream sites sampled in the Northeastern and Southeastern mesoregions of the state of Pará (PA), Brazil. The streams were distributed in natural contiguous forest (NCF), forest fragment (FF), conventional logging (CL), reduced-impact logging (RIL), plantation of primary oil palm (PPP), plantation of secondary oil palm (PSP), and livestock farming (LIV). The geographical coordinates of each stream are available in the supporting information on Table S1
Oil palm streams were divided into two groups: plantation of primary oil palm (n = 17; PPP) and plantation of secondary oil palm (n = 5; PSP), based on differences in the historical context of the area at the time of planting the crop. In PPP sites, oil palm plantations were directly converted from the forest. In these sites, riparian vegetation is preserved according to Brazilian legislation (Forest Code, Law 12.651 and its amendments; Brasil, 2012), although the streams are used as an irrigation source for young plantations (Agropalma, 2015). In PSP sites, the plantations were converted from non-forest lands, such as lands planted with other crops, pastures for livestock farming or deforested areas (Biopalma, 2015), following the Brazilian federal proposal of the Ecological-Economic Zoning (EEZ) (Ramalho Filho et al., 2010). In these sites, riparian vegetation is sparsely present in some stream reaches. While chemical fertilizers and pesticides are used in both PPP and PSP sites, organic fertilizers of industrial co-products (e.g., empty fruit bunches and palm fibers) are also used in PSP sites. There were fewer streams sampled in PSP than in PPP and other human land uses because PSP is rarer in the landscape since the EEZ, which stipulated for oil palm to be planted only on non-forest lands, was only proposed in 2010. Lastly, in livestock farming sites, the cattle are typically managed according to rotational grazing techniques in which they are allowed to consume all the grass in a particular area before moving to the next area on the farm. The latter area is then left to rest until the grass recover (Silva & Barreto, 2014). In addition, livestock farmers use tanks, artificial dams, or the streams to provide drinking water to cattle. As a result, some streams in livestock farming have a small strip of riparian vegetation (~ 10 m wide) on both banks while, in others, the vegetation is absent.
Study design and physical and chemical factors
We measured instream habitat characteristics along each stream site in which we sampled the EPT community. Each 150 m long stream reach was subdivided into 10 longitudinal sections of 15 m by 11 transects, following the protocol of the United States Environmental Protection Agency (US-EPA, Peck et al., 2006) adapted to tropical areas (Callisto et al., 2014). At each of the 10 longitudinal sections, we measured the thalweg depth and % of riffle habitats. We also recorded % of the organic substrate on the streambed, which included partially decomposed leaf detritus and plant material between 0.053 mm and 2 mm in size. Lastly, the canopy density at the bank was measured at both sides (left and right) of the wetted channel margin with a spherical densiometer (convex type) positioned 0.30 cm above the water surface. These physical characteristics were calculated following Kaufmann et al. (1999). Four environmental characteristics were calculated based on these measurements: mean thalweg depth (cm), % of riffle habitats, % of organic detritus substrate and mean canopy density at stream bank. Dissolved oxygen (mg/L) and pH were measured with an U-50 series multi-parameter at three points along each stream site (downstream, middle, and upstream), and we considered the mean value. Chemical variables were measured before EPT sampling and physical habitat measurements to avoid affecting the water chemistry. The correlations among environmental characteristics were all low to moderate (pairwise Pearson correlation always with r ≤ 0.4, Figure S1), therefore allowing the use of all of them in the analyses.
EPT sampling and laboratory procedures
Each stream site was sampled once, always in the dry season in the region (August—December) to avoid seasonal effects on the environmental characteristics and the EPT communities. For EPT sampling, each longitudinal section was subdivided into three segments of 5 m. Only the first two segments of each section were sampled because the third segment of each section was used to access the environmental variables of the next stream section and to avoid disturbing the substrate of the next section to be sampled. In each segment, two portions of the substrate were randomly collected using an 18 cm diameter dipnet (250 μm mesh), forming one composite sample per segment. Thus, in each stream 40 portions of substrates are collected in 20 segments (more details see Juen et al., 2016). We sampled all available habitats present at the streams, including inorganic substrates, macrophytes, leaf litter, and parts of terrestrial vegetation immersed in the water. This methodology has effectively assessed environmental impacts on aquatic ecosystems in the Amazon and Brazilian Cerrado biomes (Shimano et al., 2013; Brasil et al., 2016; Faria et al., 2017; de Paiva et al., 2017).
The EPT specimens collected were fixed in 85% ethanol and identified at the genus level using the specialized keys of Pes et al. (2014), Hamada & Silva (2014), Domínguez et al. (2006), Salles & Domínguez (2012), and Salles et al. (2014). The genus level is considered sufficient to assess subtle ecological patterns using EPT assemblages (e.g., Godoy et al., 2019). The specimens sampled were deposited in the collection of Aquatic Insects of the Laboratory of Ecology and Conservation at the Federal University of Pará, Brazil.
Data analysis
Each stream site was considered as an independent sample unit in the analyses. To test our first hypothesis (H1), we applied a Permutational Multivariate Analysis of Variance (PERMANOVA; Anderson, 2001) to examine whether the environmental characteristics were different among the land use types, followed by pairwise comparisons between each pair of land use types. The coefficient of determination (R2) provided by PERMANOVA represented the magnitude of the land use effect on environmental conditions. In the PERMANOVA analysis, we used a Euclidean distance matrix calculated from the six standardized (mean = 0 and standard deviation = 1) instream habitat characteristics. We tested for the null hypothesis of no differences between streams using a Monte Carlo method with 10,000 random permutations. P-values ≤ 0.05 were regarded as statistically significant. We corrected the P-value using the sequential Bonferroni correction to minimize inflation of the type I error with multiple comparisons among land uses. We used Principal Coordinates Analysis (PCoA) to visualize differences among land use types. We calculated correlations between environmental characteristics and PCoA axes to quantify how each habitat variable contributed to individual PCoA axes (Legendre & Legendre, 2012).
We evaluated differences in spatial dispersion of streams among the land use types to examine the possibility of spatial dispersion affecting environmental heterogeneity and β-diversity (following Heino et al., 2013; Tonkin et al., 2016), since more clumped sites would be more prone to have lower habitat heterogeneity and beta diversity. We used Multivariate Dispersion Analysis (PERMDISP, Anderson et al., 2006) to evaluate the spatial dispersion among land use types. To calculate spatial dispersion, we used the Euclidean distance between all pairs of sites based on the geographic coordinates (lat-long) of the streams (see Supporting Information Table S1). We did not observe a significant difference in spatial dispersion among land use types (F(6,76) = 1.943; P = 0.09; Figure S2). Therefore, any differences found in environmental heterogeneity and β-diversity were unlikely to be confounded by the spatial dispersion of the streams.
To test our second hypothesis (H2) and fourth hypothesis (H4), we also used PERMDISP. We calculated, for each response matrix (environmental matrix and community matrix), the distance of each site to its group centroid (where each group is a land use type). The higher the average distance to the centroid, the greater the dispersion among sites within a given land use type (Anderson et al., 2006). Therefore, the average distance to the centroid at each land use type was used as a measure of environmental heterogeneity and EPT β-diversity. The significance of among-group differences was tested through the permutation of least-squares residuals (Anderson et al., 2006) running 10,000 permutations (P-values ≤ 0.05 were taken as statistically significant). The environmental characteristics were used for measuring environmental heterogeneity in each land use type. The six instream habitat variables were standardized (mean = 0, standard deviation = 1) and the dissimilarity matrix was constructed using Euclidean distance. β-diversity was calculated using the Jaccard dissimilarity index, which considers taxa occurrences (presence/absence data rather than taxa abundance data), since we were especially concerned about the changes in the taxonomic composition between assemblages. In each analysis, we used a sqrt(n/(n − 1)) F-statistic correction (O’Neill, 2000; Stier et al., 2013) to account for the unbalanced sample sizes (i.e., number of streams) between groups (Anderson and Walsh, 2013).
To test our third hypothesis (H3), we used the genera richness of EPT in each stream site as a measure of α-diversity, and we tested if there were differences between the land use types with a one-way analysis of variance (ANOVA, Zar, 2010), followed by a Tukey’s post hoc test. In addition, we fitted a negative-binomial generalized linear model (GLM) to identify which environmental characteristics were the most important in determining α-diversity (Zuur et al., 2009). The generalized linear model was validated by the residuals of the fitted linear mixed models (DHARMa, Hating, 2022; Figure S3). We performed all analyses in R v. 3.3.2 (R Core Team, 2016), using the "MASS" (Venables & Ripley, 2002), "mgcv" (Wood, 2006), “stats” (R Core Team, 2016), "pairwiseAdonis" (Arbizu, 2019), "vegan" packages (Oksanen et al., 2016), and “hnp” (Moral et al., 2017) for the multivariate analyses.
Results
Environmental characteristics of streams
There were differences in environmental characteristics between streams in different land use types (PERMANOVA pseudo-F(6,76) = 8.0; R2 = 0.386; P < 0.01, Table 1). Pairwise PERMANOVA comparisons (Table 1) indicate significant differences between contiguous forested streams (NCF) versus conventional logging (CL), secondary oil palm plantations (PSP), and livestock farming (LIV). Streams in forest fragments (FF) were significantly different from streams in both conventional (CL) and reduced-impact logging (RIL). As expected, there was no significant difference between our two types of control sites, contiguous (NCF) and fragmented forest (FF) streams.
Streams in livestock farming (LIV) were most different from other land uses, presenting high PC1 values and low PC2 values, which was driven by a higher water pH (5.28 vs. 4.6–5.1 in other land uses), higher dissolved oxygen (11.1 mg/L vs. 3.7–7.0 mg/L), and lower canopy cover (64.1% vs. 74.6–96.8%) (Fig. 2A and Table 2). There were also notable differences among the other land use types. Forested streams (NCF and FF) had greater canopy density (94.7–96.8%) than streams of human land uses (64.1–84.1%), except for RIL (96.8%) (Table 2). In addition, forested streams (NCF and FF) had intermediate values of mean thalweg depth (23.9 and 31.0 cm, respectively) and % of riffle habitats (18.6% and 32.9%, respectively) compared to streams in other land use types (depth: 21.0–38.6 cm; % riffle: 4.6–40.1%). There was more organic detritus in PSP, FF and PPP streams (24–42.5%) than in other streams (0.97–11.6%). The dissolved oxygen was lower in CL and PSP streams (3.7 and 4.4 mg/L, respectively) than in streams of other human land uses (6.1–11.1 mg/L).
Principal Coordinate Analysis (PCoA) plot of the first two PCoA axes summarizing environmental characteristics (A) and box plot of values of distances to group centroids calculated by PERMDISP representing the environmental heterogeneity (B). Summary statistics of environmental characteristics in each land use category and Pearson correlations between environmental characteristics and PCoA axes are shown in Table 2. The horizontal line represents the median; the box represents the interquartile range (IQR), and the whiskers represent values up to + / − 1.5 × IQR from the 75th and 25th percentiles, respectively. The letters over the box plots correspond to pairwise comparisons among land use categories (categories that do not share the same letter are significantly different; see Table 1 for results of statistical tests). Points in the PCoA plot are sampling sites color-coded according to land use types: natural contiguous forest (NCF), forest fragment (FF), conventional logging (CL), reduced-impact logging (RIL), plantation of primary oil palm (PPP), plantation of secondary oil palm (PSP), and livestock farming (LIV)
Environmental heterogeneity differed significantly among land use types (PERMDISP F(6,76) = 4.7, P = 0.002, Table 1). Streams in LIV, NCF, and FF were more environmentally heterogeneous than streams in other land uses. The difference between streams in LIV, NCF, and FF and streams in RIL and CL was statistically significant. CL streams were most environmentally homogeneous, followed by RIL and PSP streams (Fig. 2B, Table 1).
EPT diversity
We collected 16,038 EPT specimens, representing 51 genera distributed across 18 families (Table S3). The mean abundance of EPT was greater in the streams in PSP (472 ± 219, standard deviation) and LIV (287 ± 256) sites compared the other land use types (NCF, FF, CL, RIL, and PPP, Table S3). EPT α-diversity was significantly different among the land uses (ANOVA, F(6,76) = 5.7; P < 0.001, Fig. 3A and Table 1). Streams in the contiguous forest (NCF) had significantly higher α-diversity than CL, PPP, and LIV streams. Among anthropic land uses, RIL had significantly greater α-diversity than PPP (Table 1). The percentage of riffle habitat was positively associated with EPT α-diversity (Z value = 2.310, P = 0.020; Fig. 3B). Detailed results on the environmental correlates of EPT α-diversity are available in the Supporting Information, Table S4.
A Box plot of the EPT α-diversity in the natural contiguous forest (NCF), forest fragment (FF), conventional logging (CL), reduced-impact logging (RIL), plantation of primary oil palm (PPP), plantation of secondary oil palm (PSP), and livestock farming (LIV). The horizontal line represents the median; the box represents the interquartile range (IQR), and the whiskers represent values up to + / − 1.5 × IQR from the 75th and 25th percentiles, respectively. The letters over the box plots correspond to Tukey’s post hoc test among land use types (types that do not share the same letter are significantly different; see Table 1 for results of statistical tests). B Relationship between α-diversity and % of riffle habitats. The gray color represents the confidence interval of 0.95 for the linear model
EPT β-diversity differed between the land use types (PERMDISP, F(6,76) = 2.7, P = 0.023, Table 1). The β-diversity was highest in LIV and PPP streams than in RIL and forested streams (NCF, FF) (Fig. 4A and B). There was no correlation between β-diversity and environmental heterogeneity (Pearson correlation r = 0.356, Figure S4).
Principal Coordinate Analysis (PCoA) plot of the first two PCoA axes (A) and box plots of values of distances to group centroids calculated by PERMDISP representing EPT β-diversity (B). Points in the PCoA plot are sampling sites color-coded according to land use types: natural contiguous forest (NCF), forest fragment (FF), conventional logging (CL), reduced-impact logging (RIL), plantation of primary oil palm (PPP), plantation of secondary oil palm (PSP), and livestock farming (LIV). The horizontal line represents the median; the box represents the interquartile range (IQR), and the whiskers represent values up to + / − 1.5 × IQR from the 75th and 25th percentiles, respectively. The letters over the box plots correspond to pairwise comparisons among land use types (types that do not share the same letter are significantly different; see Table 1 for results of statistical tests)
Discussion
Effects of land uses on the environmental condition of streams
Our results supported our first hypothesis (H1), that different human land uses altered the environmental characteristics of streams in different ways. It is well known that land use changes in the riparian zone can negatively impact the physical habitats and stream water quality (Ramírez et al., 2008). This is because riparian vegetation acts as a buffer zone that protects streams from inputs of nonpoint source pollutants and from the erosion and subsequent deposition of fine sediments (Dosskey et al., 2010; Pinay et al., 2018). Riparian forests also provide canopy cover that shades the stream from direct shortwave solar radiation, thus moderating stream water temperatures (Caissie, 2006). However, the magnitude and nature of stream environmental impacts under different competing human land uses remain understudied in the Amazon. Here, we found that the impact of forest conversion was mixed; not every human land use was associated with detectable changes to the stream environment. Whereas streams affected by conventional logging differed significantly from streams in both continuous and fragmented forests, stream environments of primary oil palm plantations did not differ significantly from forested streams. Further, although contiguous and fragmented forest streams presented similar environmental conditions, both being considered forested areas, they presented differences in their ecological integrity. Therefore, some human land uses differed significantly from contiguous forest but not from fragmented forest streams. For example, the environmental conditions of streams affected by livestock farming were significantly different from contiguous forest streams but not from fragmented forest streams.
Expansion of areas with human land uses is expected to significantly reduce the landscape vegetation cover since vegetation suppression is still a traditional land preparation method for planting (Denich et al., 2004). However, vegetation suppression (i.e., land management) has important implications for the aquatic environment (Mello et al., 2020). For example, the indiscriminate cutting of trees of all sizes observed in conventional logging practices resulted in streams with lower canopy density, thalweg depth, riffle habitats, and substrate organic detritus (Table 2, Fig. 2A). By contrast, the environmental characteristics in reduced-impact logging did not differ significantly from contiguous forest streams (Table 1). This result suggests that different human land uses resulted in different environmental impacts on the aquatic ecosystems (e.g., Cruz et al., 2022), likely because the land use types have mixed effects on the ecological condition depending on the management techniques adopted. This finding reinforces the importance of conducting studies in streams that drain basins used for different land uses, seeking to understand the main changes that happen, as well as establishing measures to mitigate impacts and environmental restoration programs. However, the science of ecosystem restoration is relatively new and there are still many challenges in defining and applying restoration projects to aquatic ecosystems (Ciotti et al., 2021), mainly due to the complexity of the impacts in the stream catchments (Leitão et al., 2018) and the political-economic drivers underlying environmental change (Osborne et al., 2021).
Our results showed that the context of pre-conversion land cover might also influence the magnitude of human land use on the aquatic ecosystem. We observed that stream environments in oil palm plantations converted from forest areas did not differ significantly from those in the contiguous forest. By contrast, the environmental characteristics of streams in oil palm plantations established on non-forest land differed significantly from stream environmental characteristics in the contiguous forest (Table 1). This result is likely a consequence of land use legacies where deforestation prior to establishing oil palm plantations may have relatively long-lasting effects on the stream environment. A previous study showed that much of the contemporary variation in the physical and chemical characteristics of streams was correlated with historical agriculture (Maloney et al., 2008). Therefore, our results indicate that the relative magnitude of land use effects on aquatic habitats in the Amazon may depend upon the land use surrounding the streams (Sponseller et al., 2001), the intensity of management (Mello et al., 2020), land use legacies (Munteanu et al., 2015; Martin et al., 2017), and the condition of riparian vegetation (Giam et al., 2015; Cruz et al., 2022).
We found partial support for our second hypothesis; on average, forested streams had higher environmental heterogeneity, except for one human land use (livestock farming). However, this difference is only significant when forested streams are compared to streams affected by conventional and reduced-impact logging (Table 1, Fig. 2B). These results are consistent with a meta-analysis (Seiferling et al., 2014) that found that environmental heterogeneity in natural ecosystems is at intermediate levels, increasing or decreasing depending on the intensity of human disturbance. For example, remnant riparian forests adjacent to sugarcane plantations in Southeastern Brazil are structurally heterogeneous due to forest stratification, promoting local aquatic environmental heterogeneity on physical and chemical characteristics of the streams (Fernandes et al., 2014). We found that stream environments in the contiguous forest are statistically indistinguishable from stream environments in fragmented forests in terms of their characteristics and the level of heterogeneity among sites (Table 1). This has important implications because politicians and policymakers often regard forest fragments as degraded landscapes, and use this to justify their conversion to other land uses (e.g., Wang et al., 2020). For example, a recent study showed that the conversion of secondary forest to pastureland in the Brazilian Amazon increased by 282% between 2008 and 2014 (Wang et al., 2020). However, as our results show, these areas provide valuable aquatic habitats comparable to contiguous forests and should also be considered in conservation and zonation projects in the Amazon.
Streams surrounded by livestock farms were the most environmentally heterogeneous (Table 1, Fig. 2B). Significant differences in the amount of patchy canopy and vegetation cover drove the high environmental heterogeneity among livestock farming streams. Further, differences in canopy cover among these streams may lead to heterogeneity in habitat conditions that we did not investigate. For example, greater light input in streams where canopy cover is largely absent will lead to greater primary productivity and an increase in macrophyte cover, whereas streams with greater canopy cover may have more homogeneous benthic habitat, mostly composed by leaf litter (Giam et al. 2015; Fares et al., 2020). Therefore, particular human land uses may result in greater environmental variation among streams due to different intensities of impact on riparian vegetation, which generate greater habitat heterogeneity (e.g., Fugère et al., 2016; Dala-Corte et al., 2019). We would like to emphasize that a greater heterogeneity in livestock farming does not mean that streams have higher environmental quality since it simply measures the variation in environmental characteristics of habitat among streams, but not the water quality or the quality of the environment to stream biota. Controversial results on the effect of pasture on water quality have been found (Tanaka et al., 2016; Mello et al., 2018), possibly because the effect depends on the grazing intensity (Mello et al., 2020). Furthermore, in Brazil, there are farms with natural grasslands associated with low cattle density, which may not strongly affect water quality (Mello et al., 2020). It is important to highlight that our study area does not have large cattle farms, and some fields are in fact only abandoned grasslands. Thus, adverse impacts of the presence of animals on water quality may not be of the same magnitude found by other studies.
The intensification of human activities in highly modified ecosystems can lead to drastic reductions in environmental heterogeneity (Seiferling et al., 2014), likely due to significant changes in instream structure (e.g., Deegan et al., 2011). We observed that the amount of organic detritus is very much reduced in streams in conventional logging landscapes, where up to 26% of above-ground tree biomass can be lost (West et al., 2014). Therefore, the reduction of organic detritus will likely result from lower vegetation and canopy cover and, consequently, lower allochthonous input into the streams from the riparian zone. The dissolved oxygen concentration in conventional logging is below water quality standards allowed for human consumption established by the Brazilian legislation (MMA—CONAMA, Resolution No. 357 of March 17, 2005, and its amendments). This is plausibly a consequence of the intense disturbance associated with conventional logging and low flow velocity (% of rifle habitats) observed in these streams (Table 2), which reduces vertical aeration of the water column. Taken together, these results suggest that environmental heterogeneity is driven not only by the relatively natural characteristics of the preserved ecosystem, but also by dynamic processes derived from land use practices (sensu Leão et al., 2020).
Effects of land uses on alpha and beta diversity of streams
Our results indicated partial support for our third hypothesis. Mean α-diversity of aquatic invertebrates was highest in contiguous forest streams, significantly different from streams affected by conventional logging, livestock farming, and primary oil palm plantation. However, the α-diversity in streams within forest fragments was not significantly different from any of the human land uses (Table 1, Fig. 3). Rapid environmental change can lead to species loss (Suhonen et al., 2014) due to ecological requirements of individual taxa (Marques Peixoto et al., 2020). These differences in biodiversity compared to human land use environments can be explained by the dependence that aquatic systems have on their drainage basin. Environmental alterations and pollutants that may occur at the surrounding marginal vegetation or drainage basin, ending up being leached into the stream channels (Mello et al., 2018), resulting in the simplification of microhabitats and in the decrease of richness of aquatic insects (Sueyoshi et al., 2016). Mean α-diversity in fragmented forest streams was not different from that in contiguous forest streams and other human land uses. This indicates that fragmented forest areas are intermediate in quality for supporting aquatic invertebrate richness between contiguous forest and the other human land use types. Compared to streams in contiguous forest, streams in forest fragments are likely affected by human land uses adjacent to the fragments. This finding is consistent with studies that have found that greater catchment-level forest cover is correlated with higher macroinvertebrate taxa richness (Roy et al., 2003; Harding et al., 2006; Death & Collier, 2010).
Multiple environmental factors such as microclimate, allochthonous resources, vegetation cover and edaphic structure interact to determine biotic assemblages in streams (Marques Peixoto et al., 2020). In our study, the amount of riffle habitat was positively related to α-diversity (Table S4, Figure 3B). This relationship, which was also found in other studies (Roy et al., 2003; Amaral et al., 2015), probably arose as coarser substrate underlying fast riffles provide microhabitat to support riffle specialists such as stoneflies or some caddisfly (Ligeiro et al., 2010). Interestingly, despite livestock farming streams had the highest percent of riffle habitats, their mean α-diversity was lower than control streams (contiguous and fragmented forests), probably due to the lower proportion of organic detritus in the substrate, which provides shelter and food for shredder invertebrates (e.g., Firmino et al., 2021).
We did not find support for our fourth hypothesis that EPT β-diversity is lower among streams surrounded by human land uses than among streams in contiguous or fragmented forests. EPT β-diversity was significantly higher in streams surrounded by livestock farming and primary oil palm plantation than in forested streams (Table 1, Fig. 4). EPT β-diversity had a weak correlation with environmental heterogeneity (Figure S4), which indicates that habitat heterogeneity had little (if any) effect on compositional variation in our study system. It is more likely that under disturbance, taxa that require more pristine instream habitats (i.e., specialists) are negatively affected, whereas the more disturbance-tolerant taxa (i.e., generalists) become more dominant in these sites (Büchi & Vuilleumier, 2016). Our results suggest that streams in human-modified landscapes present assemblages comprising varied subsets of these generalist species. In addition, landscapes are influenced by various management practices, which alter different aspects of the environment and consequently support different sets of species (Sambhu et al., 2018). For example, some farms in our study area adopt environmentally friendly practices, such as maintaining riparian and headland vegetation, while other farms are much more destructive, which might generate high environmental and biological dissimilarities among sites of the same type of land use. Although some studies have found lower beta diversity in anthropized or contaminated areas (Leboucher et al., 2019; Ribeiro et al., 2022), recent studies have observed an opposite pattern, going against the common expectations (Simões et al., 2020; Paiva et al., 2021). However, in cases of greater intensification of anthropic activity and homogenization of the landscape by a certain type of land use, lower beta diversity is often the case (Siqueira et al., 2015; Li et al., 2020).
Conclusion
Our study examined changes in environmental heterogeneity and biological diversity in a region undergoing rapid land use change. We found greater environmental heterogeneity in forested streams than in those embedded in anthropic land uses, except for livestock farms. The responses of α- and β-diversity of EPT differed according to the type of land use. All land uses resulted in a decline of EPT α-diversity relative to streams in the contiguous forest. EPT β-diversity was higher in streams embedded in livestock farming areas and primary oil palm plantations than in forested streams, the former comprising varied sets of generalist taxa. Therefore, our study contributes to understanding the effects of multiple land uses on stream environment and insect diversity at both local and regional scales. We highlight that the association between land use and habitat degradation may not be so direct, mainly when streams are inserted into or surrounded by forest fragments. Even with the landscape altered, the impact may not be evident if the changes do not directly affect the stream or their banks. This has important implications because politicians and policymakers often regard forest fragments as degraded landscapes, to justify their conversion to other land uses. However, our study shows that forest fragments are important for the maintenance of the environmental condition of the streams and should be the focus of conservation and restoration actions, especially in highly biodiverse regions such as the Amazon.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Agropalma, 2015. Relatório de sustentabilidade. Retrieved February 20, 2018, from http://www.agropalma.com.br/.
Alahuhta, J., S. Kosten, M. Akasaka, D. Auderset, M. M. Azzella, R. Bolpagni, C. P. Bove, P. A. Chambers, E. Chappuis, J. Clayton, M. Winton, F. Ecke, E. Gacia, G. Gecheva, P. Grillas, J. Hauxwell, S. Hellsten, J. Hjort, E. H. Li, B. A. Lukács, M. Mjelde, A. Mikulyuk, R. P. Mormul, M. Søndergaard, T. Yamanouchi, Q. Yu, H. Wang, N. Willby, X. K. Zhang & J. Heino, 2017. Global variation in the beta diversity of lake macrophytes is driven by environmental heterogeneity rather than latitude. Journal of Biogeography 44: 1758–1769. https://doi.org/10.1111/jbi.12978.
Aldrich, S., R. Walker, C. Simmons, M. Caldas & S. Perz, 2012. Contentious land change in the Amazon’s arc of deforestation. Annals of the Association of American Geographers 102: 103–128. https://doi.org/10.1080/00045608.2011.620501.
Allan, J. D., 2004. Landscapes and riverscapes: the influence of land use on stream ecosystems. Annual Review of Ecology, Evolution, and Systematics 35: 257–284. https://doi.org/10.1146/annurev.ecolsys.35.120202.110122.
Al-Shami, S. A., J. Heino, M. R. Che Salmah, A. Abu Hassan, A. H. Suhaila & M. R. Madrus, 2013. Drivers of beta diversity of macroinvertebrate communities in tropical forest streams. Freshwater Biology 58: 1126–1137. https://doi.org/10.1111/fwb.12113.
Amaral, P. H. M. D., L. S. D. Silveira, B. F. J. V. Rosa, V. C. D. Oliveira & R. D. G. Alves, 2015. Influence of habitat and land use on the assemblages of Ephemeroptera, Plecoptera, and Trichoptera in Neotropical streams. Journal of Insect Science 15: 60. https://doi.org/10.1093/jisesa/iev042.
ANA - Agência Nacional de Águas, 2010. Ottobacias, Shapefiles para download. Retrieved February 20, 2018, from http://www.ana.gov.br
Anderson, M. J., 2001. A new method for non-parametric multivariate analysis of variance. Austral Ecology 26: 32–46. https://doi.org/10.1111/j.1442-9993.2001.01070.pp.x.
Anderson, M. J., K. E. Ellingsen & B. H. Mcardle, 2006. Multivariate dispersion as a measure of beta diversity. Ecology Letters 9: 683–693. https://doi.org/10.1111/j.1461-0248.2006.00926.x.
Anderson, M. J. & D. C. Walsh, 2013. PERMANOVA, ANOSIM, and the Mantel test in the face of heterogeneous dispersions: what null hypothesis are you testing? Ecological Monographs 83: 557–574. https://doi.org/10.1890/12-2010.1.
Arbizu, P. M., 2019. pairwiseAdonis: pairwise multilevel comparison using adonis. R package version 0.3
Astorga, A., J. Heino, M. Luoto & T. Muotka, 2011. Freshwater biodiversity at regional extent: determinants of macroinvertebrate taxonomic richness in headwater streams. Ecography 34: 705–713. https://doi.org/10.1111/j.1600-0587.2010.06427.x.
Astorga, A., R. Death, F. Death, R. Paavola, M. Chakraborty & T. Muotka, 2014. Habitat heterogeneity drives the geographical distribution of beta diversity: the case of New Zealand stream invertebrates. Ecology and Evolution 4: 2693–2702. https://doi.org/10.1002/ece3.1124.
Belsky, A. J., A. Matzke & S. Uselman, 1999. Survey of livestock influences on stream and riparian ecosystems in the Western United States. Journal of Soil Water Conservation 54: 419–431.
Bini, L. M., V. L. Landeiro, A. A. Padial, T. Siqueira & J. Heino, 2014. Nutrient enrichment is related to two facets of beta diversity for stream invertebrates across the United States. Ecology 95: 1569–1578. https://doi.org/10.1890/13-0656.1.
Biopalma, 2015. Relatório de sustentabilidade. Retrieved February 20, 2018, from http://www.biopalma.com.br/relatorio-sustentavel.
Brasil, 2012. Forest Code: Law 12.651 of May 25. Retrieved January, 2020, from http://www.planalto.gov.br/ccivil_03/_Ato2011-2014/2012/Lei/L12651.htm.
Brasil, L. S., J. Juen, N. F. S. Giehl & H. S. R. Cabette, 2016. Effect of environmental and temporal factors on patterns of rarity of ephemeroptera in stream of the Brazilian Cerrado. Neotropical Entomology 45: 1–7. https://doi.org/10.1007/s13744-016-0431-9.
Brown, B. L., 2003. Spatial heterogeneity reduces temporal variability in stream insect communities. Ecology Letters 6: 316–325. https://doi.org/10.1046/j.1461-0248.2003.00431.x.
Büchi, L. & S. Vuilleumier, 2016. Ecological strategies in stable and disturbed environments depend on species specialisation. Oikos 125: 1408–1420. https://doi.org/10.1111/oik.02915.
Callisto, M., C. B. M. Alves, J. M. Lopes & M. A. Castro, 2014. Condições ecológicas em bacias hidrográficas de empreendimentos hidrelétricos. Belo Horizonte: Cemig.
Calvão, L. B., D. S. Nogueira, L. F. A. Montag, M. A. Lopes & L. Juen, 2016. Are Odonata communities impacted by conventional or reduced impact logging? Forest Ecology and Management 382: 143–150. https://doi.org/10.1016/j.foreco.2016.10.013.
Caissie, D., 2006. The thermal regime of rivers: a review. Freshwater Biology 51: 1389–1406. https://doi.org/10.1111/j.1365-2427.2006.01597.x.
Cardoso, M. N., L. B. Calvão, L. F. A. Montag, B. S. Godoy & L. Juen, 2018. Reducing the deleterious effects of logging on Ephemeroptera communities through reduced impact management. Hydrobiologia 823: 191–203. https://doi.org/10.1007/s10750-018-3705-x.
Carvalho, F. G., F. O. Roque, L. Barbosa, L. F. A. Montag & L. Juen, 2018. Oil palm plantation is not a suitable environment for most forest specialist species of Odonata in Amazonia. Animal Conservation 21: 526–533. https://doi.org/10.1111/acv.12427.
Ciotti, D. C., J. Mckee, K. L. Pope, G. M. Kondolf & M. M. Pollock, 2021. Design criteria for process-based restoration of fluvial systems. BioScience 71: 831–845. https://doi.org/10.1093/biosci/biab065.
Cruz, G. M., A. P. J. Faria & L. Juen, 2022. Patterns and metacommunity structure of aquatic insects (Trichoptera) in Amazonian streams depend on the environmental conditions. Hydrobiologia. https://doi.org/10.1007/s10750-022-04901-0.
Cunha, E. J., L. F. A. Montag & L. Juen, 2015. Oil palm crops effects on environmental integrity of Amazonian streams and Heteropteran (Hemiptera) species diversity. Ecological Indicators 52: 422–429. https://doi.org/10.1016/j.ecolind.2014.12.024.
Cunha, E. J. & L. Juen, 2017. Impacts of oil palm plantations on changes in environmental heterogeneity and Heteroptera (Gerromorpha and Nepomorpha) diversity. Journal of Insect Conservation 21: 111–119. https://doi.org/10.1007/s10841-017-9959-1.
Dala-Corte, R. B., L. F. Sgarbi, F. G. Becker & A. S. Melo, 2019. Beta diversity of stream fish communities along anthropogenic environmental gradients at multiple spatial scales. Environmental Monitoring and Assessment 191: 288. https://doi.org/10.1007/s10661-019-7448-6.
Deegan, L. A., C. Neill, C. L. Haupert, M. V. R. Ballester, A. V. Krusche, R. L. Victoria, S. M. Thomas & E. Moor, 2011. Amazon deforestation alters small stream structure, nitrogen biogeochemistry and connectivity to larger rivers. Biogeochemistry 105: 53–74. https://doi.org/10.1007/s10533-010-9540-4.
Denich, M., K. Vielhauer, M. D. A. Kato, A. Block, O. R. Kato, T. D. Abreu Sá, W. Lucke & P. L. Vlek, 2004. Mechanized land preparation in forest-based fallow systems: the experience from Eastern Amazonia. Agroforestry Systems 61: 91–106. https://doi.org/10.1023/B:AGFO.0000028992.01414.2a.
de Paiva, C. K. S., A. P. J. Faria, L. B. Calvao & L. Juen, 2021. The anthropic gradient determines the taxonomic diversity of aquatic insects in Amazonian streams. Hydrobiologia 848: 1073–1085. https://doi.org/10.1007/s10750-021-04515-y.
de Paiva, C. K. S., A. P. J. Faria, L. B. Calvão & L. Juen, 2017. Effect of oil palm on the Plecoptera and Trichoptera (Insecta) assemblages in streams of eastern Amazon. Environmental Monitoring and Assessment 189: 393. https://doi.org/10.1007/s10661-017-6116-y.
Death, R. G. & K. J. Collier, 2010. Measuring stream macroinvertebrate responses to gradients of vegetation cover: when is enough enough? Freshwater Biology 55: 1447–1464. https://doi.org/10.1111/j.1365-2427.2009.02233.x.
Dias, M. S., W. E. Magnusson & J. Zuanon, 2010. Effects of reduced-impact logging on fish assemblages in Central Amazonia. Conservation Biology 24: 278–286. https://doi.org/10.1111/j.1523-1739.2009.01299.x.
Domínguez, E., C. Molineri, M. L. Pescador, M. D. Hubbard & C. Nieto, 2006. Ephemeroptera of South America. In: Adis, J. R., Rueda-Delgado, G., & Wantzen, K. M. (eds), Aquatic biodiversity in Latin American (ABLA). Pensoft, Sofia Moscow.
Dosskey, M. G., P. Vidon, N. P. Gurwick, C. J. Allan, T. P. Duval & R. Lowrance, 2010. The role of riparian vegetation in protecting and improving chemical water quality in streams. JAWRA Journal of the American Water Resources Association 46: 261–277. https://doi.org/10.1111/j.1752-1688.2010.00419.x.
Fares, A. L. B., L. B. Calvão, N. R. Torres, E. S. C. Gurgel & T. S. Michelan, 2020. Environmental factors affect macrophyte diversity on Amazonian aquatic ecosystems inserted in an anthropogenic landscape. Ecological Indicators 113: 106231. https://doi.org/10.1016/j.ecolind.2020.106231.
Faria, A. P. J., R. Ligeiro, M. Callisto & L. Juen, 2017. Response of aquatic insect assemblages to the activities of traditional populations in eastern Amazonia. Hydrobiologia 802: 39–51. https://doi.org/10.1007/s10750-017-3238-8.
Fernandes, J. D. F., A. L. de Souza & M. O. Tanaka, 2014. Can the structure of a riparian forest remnant influence stream water quality? A tropical case study. Hydrobiologia 724: 175–185. https://doi.org/10.1007/s10750-013-1732-1.
Firmino, V. C., L. S. Brasil, R. T. Martins, R. Ligeiro, A. Tonin, J. F. G. Júnior & L. Juen, 2021. Litter decomposition of exotic and native plant species of agricultural importance in Amazonian streams. Limnology 22: 289–297. https://doi.org/10.1007/s10201-021-00655-1.
Fugère, V., A. Kasangaki & L. J. Chapman, 2016. Land use changes in an afrotropical biodiversity hotspot affect stream alpha and beta diversity. Ecosphere 7: e01355. https://doi.org/10.1002/ecs2.1355.
García-García, P. L., G. Vázquez, R. Novelo-Gutiérrez & M. E. Favila, 2017. Effects of land use on larval Odonata assemblages in cloud forest streams in central Veracruz, Mexico. Hydrobiologia 785: 19–33. https://doi.org/10.1007/s10750-016-2900-x.
Giam, X., R. K. Hadiaty, H. H. Tan, L. R. Parenti, D. Wowor, S. Sauri, K. Y. Chong, D. C. J. Yeo & D. S. Wilcove, 2015. Mitigating the impact of oil-palm monoculture on freshwater fishes in Southeast Asia. Conservation Biology 29: 1357–1367. https://doi.org/10.1111/cobi.12483.
Grman, E., J. L. Orrock, C. W. Habeck, J. A. Ledvina & L. A. Brudvig, 2015. Altered beta diversity in post-agricultural woodlands: two hypotheses and the role of scale. Ecography 38: 614–621. https://doi.org/10.1111/ecog.01159.
Godoy, B. S., A. P. J. Faria, L. Juen, S. Lodi & L. G. Oliveira, 2019. Taxonomic sufficiency and effects of environmental and spatial drivers on aquatic insect community. Ecological Indicators 107: 105624. https://doi.org/10.1016/j.ecolind.2019.105624.
Hamada, N. & J. O. Silva, 2014. Ordem Plecoptera. In: Hamada, N., Nessimian, J. L., & Querino, R. B. (Eds), Insetos Aquáticos na Amazônia brasileira: taxonomia, biologia e ecologia. Manaus: Editora do INPA.
Hansen, M. C., L. Wang, X. P. Song, A. Tyukavina, S. Turubanova, P. V. Potapov & S. V. Stehman, 2020. The fate of tropical forest fragments. Science Advances 6: eaax8574. https://doi.org/10.1126/sciadv.aax857.
Harding, J. S., E. F. Benfield, P. V. Bolstad, G. S. Helfman & E. B. D. Jones, 1998. Stream biodiversity: the ghost of land use past. Proceedings of the National Academy of Sciences 95: 14843–14847. https://doi.org/10.1073/pnas.95.25.14843.
Harding, J. S., K. Claassen & N. Evers, 2006. Can forest fragments reset physical and water quality conditions in agricultural catchments and act as refugia for forest stream invertebrates? Hydrobiologia 568: 391–402. https://doi.org/10.1007/s10750-006-0206-0.
Hawkins, C. P., H. Mykrä, J. Oksanen & J. J. Vander Laan, 2015. Environmental disturbance can increase beta diversity of stream macroinvertebrate assemblages. Global Ecology and Biogeography 24: 483–494. https://doi.org/10.1111/geb.12254z.
Heino, J., M. Grönroos, J. Ilmonen, T. Karhu, M. Niva & L. Paasivirta, 2013. Environmental heterogeneity and β diversity of stream macroinvertebrate communities at intermediate spatial scales. Freshwater Science 32: 142–154. https://doi.org/10.1899/12-083.1.
Heino, J., A. S. Melo & L. M. Bini, 2015. Reconceptualising the beta diversity-environmental heterogeneity relationship in running water systems. Freshwater Biology 60: 223–235. https://doi.org/10.1111/fwb.12502.
IBGE - Instituto Brasileiro de Geografia e Estatística, 2011. Estado do Pará: cobertura e uso da terra no. Retrieved February 20, 2018, from http://mapas.ibge.gov.br/tematicos/uso-da-terra.
IBGE - Instituto Brasileiro de Geografia e Estatística, 2006. Mapas de Solos do Brasil. Retrieved August 23, 2022, from https://www.ibge.gov.br/geociencias/informacoes-ambientais/pedologia/15829-solos.html?=&t=downloads
IMAZON - Instituto do Homem e Meio Ambiente da Amazônia, 2017. Retrieved February 20, 2018, from https://imazon.org.br/?s=arco+do+desmatamento&submit-search=.
INPE - Instituto Nacional de Pesquisas Espaciais, 1997. Deforestation 1995–1997. Retrieved February 20, 2018, from http://www.obt.inpe.br/OBT/assuntos/programas/amazonia/prodes/pdfs/prodes1995-1997.pdf
Juen, L., E. J. Cunha, F. G. Carvalho, T. O. B. Ruffeil, M. C. Ferreira, A. L. Andrade, Y. Shimano, H. Leão, P. S. Pompeu & L. F. A. Montag, 2016. Effects of oil palm plantations on the habitat structure and biota of streams in Eastern Amazon. Rivers Research and Applications 32: 1996–2012. https://doi.org/10.1002/rra.3050.
Kaufmann, P. R., P. Levine, E. G. Robison, C. Seeliger & D. V. Peck, 1999. Quantifying Physical Habitat in Wadeable Streams, United States Environmental Protection Agency, Washington DC:
Leão, H., T. Siqueira, N. R. Torres & L. F. A. Montag, 2020. Ecological uniqueness of fish communities from streams in modified landscapes of Eastern Amazonia. Ecological Indicators 111: 106039. https://doi.org/10.1016/j.ecolind.2019.106039.
Leboucher, T., W. R. Budnick, S. I. Passy, S. Boutry, A. Jamoneau, J. Soininen, W. Vyverman & J. Tison-Rosebery, 2019. Diatom β-diversity in streams increases with spatial scale and decreases with nutrient enrichment across regional to sub-continental scales. Journal of Biogeography 46: 734–744. https://doi.org/10.1111/jbi.13517.
Legendre, P. & L. F. Legendre, 2012. Numerical Ecology, Elsevier, Amsterdam:
Leitão, R. P., J. Zuanon, D. Mouillot, C. G. Leal, R. M. Hughes, P. R. Kaufmann, S. Villéger, P. S. Pompeu, D. Kasper, F. R. Paula, S. F. B. Ferraz & T. A. Gardner, 2018. Disentangling the pathways of land use impacts on the functional structure of fish assemblages in Amazon streams. Ecography 41: 219–232. https://doi.org/10.1111/ecog.02845.
Levin, L. A., M. Sibuet, A. J. Gooday, C. R. Smith & A. Vanreusel, 2010. The roles of habitat heterogeneity in generating and maintaining biodiversity on continental margins: an introduction. Marine Ecology 31: 1–5. https://doi.org/10.1111/j.1439-0485.2009.00358.x.
Li, B., W. Tan, L. Wen, X. Zhao, B. Peng, J. Yang, C. Lu, Y. Wang & G. Lei, 2020. Anthropogenic habitat alternation significantly decreases α-and β-diversity of benthopelagic metacommunity in a large floodplain lake. Hydrobiologia 847: 293–307. https://doi.org/10.1007/s10750-019-04091-2.
Ligeiro, R., R. M. Hughes, P. R. Kaufmann, D. R. Macedo, K. R. Firmiano, W. R. Ferreira, D. Oliveira, A. S. Melo & M. Callisto, 2013. Defining quantitative stream disturbance gradients and the additive role of habitat variation to explain macroinvertebrate taxa richness. Ecological Indicators 25: 45–57. https://doi.org/10.1016/j.ecolind.2012.09.004.
Ligeiro, R., A. S. Melo & M. Callisto, 2010. Spatial scale and the diversity of macroinvertebrates in a Neotropical catchment. Freshwater Biology 55: 424–435. https://doi.org/10.1111/j.1365-2427.2009.02291.x.
MMA – Ministério do Meio Ambiente, 2005. CONAMA: Resolution No. 357 of March 17, 2005. Retrieved January, 2020, from http://www2.mma.gov.br/port/conama/.
MacArthur, R. H. & J. W. MacArthur, 1961. On bird species diversity. Ecology 42: 594–598. https://doi.org/10.2307/1932254.
Mahar, D. J., 1979. Frontier Development Policy in Brazil: A Study of Amazonia, Praeger, New York:
Maloney, K. O., J. W. Feminella, R. M. Mitchell, S. A. Miller, P. J. Mulholland & J. N. Houser, 2008. Landuse legacies and small streams: identifying relationships between historical land use and contemporary stream conditions. Journal of the North American Benthological Society 27: 280–294. https://doi.org/10.1899/07-070.1.
Marques Peixoto, G., R. De Fraga, M. C. Araújo, I. L. Kaefer & A. P. Lima, 2020. Hierarchical effects of historical and environmental factors on lizard assemblages in the upper Madeira River Brazilian Amazonia. PloS ONE 15: e0233881. https://doi.org/10.1371/journal.pone.0233881.
Martin, S. L., D. B. Hayes, A. D. Kendall & D. W. Hyndman, 2017. The land-use legacy effect: towards a mechanistic understanding of time-lagged water quality responses to land use/cover. Science of the Total Environment 579: 1794–1803. https://doi.org/10.1016/j.scitotenv.2016.11.158.
Massicotte, P., R. Proulx, G. Cabana & M. A. Rodriguez, 2015. Testing the influence of environmental heterogeneity on fish species richness in two biogeographic provinces. PeerJ 3: e760. https://doi.org/10.7717/peerj.760.
McKie, B. G. & B. Malmqvist, 2009. Assessing ecosystem functioning in streams affected by forest management: increased leaf decomposition occurs without changes to the composition of benthic assemblages. Freshwater Biology 54: 2086–2100. https://doi.org/10.1111/j.1365-2427.2008.02150.x.
Mello, K., R. H. Taniwaki, F. R. de Paula, R. A. Valente, T. O. Randhir, D. R. Macedo, C. G. Leal, C. B. Rodrigues & R. M. Hughes, 2020. Multiscale land use impacts on water quality: assessment, planning, and future perspectives in Brazil. Journal of Environmental Management 270: 110879. https://doi.org/10.1016/j.jenvman.2020.110879.
Mello, K., R. A. Valente, T. O. Randhir, A. C. A. Santos & C. A. Vettorazzi, 2018. Effects of land use and land cover on water quality of low-order streams in Southeastern Brazil: watershed versus riparian zone. Catena 167: 130–138. https://doi.org/10.1016/j.catena.2018.04.027.
Montag, L. F., K. O. Winemiller, F. W. Keppeler, H. Leão, N. L. Benone, N. R. Torres, B. S. Prudente, T. O. Begot, L. M. Bower, D. E. Saenz, E. O. Lopez-Delgado, Y. Quintana, D. J. Hoeinghaus & L. Juen, 2018. Land cover, riparian zones and instream habitat influence stream fish assemblages in the eastern Amazon. Ecology of Freshwater Fish 28: 317–329. https://doi.org/10.1111/eff.12455.
Muotka, T., R. Paavola, A. Haapala, M. Novikmec & P. Laasonen, 2002. Long-term recovery of stream habitat structure and benthic invertebrate communities from in-stream restoration. Biological Conservation 105: 243–253. https://doi.org/10.1016/S0006-3207(01)00202-6.
Murphy, G. E. & T. N. Romanuk, 2014. A meta-analysis of declines in local species richness from human disturbances. Ecology and Evolution 4: 91–103. https://doi.org/10.1002/ece3.909.
Munteanu, C., T. Kuemmerle, N. S. Keuler, D. Müller, P. Balázs, M. Dobosz, P. Griffiths, L. Halada, D. Kaim, G. Király, É. Konkoly-Gyuró, J. Kozak, J. Lieskovsky, K. Ostafin, K. Ostapowicz, O. Shandra & V. C. Radeloff, 2015. Legacies of 19th century land use shape contemporary forest cover. Global Environmental Change 34: 83–94. https://doi.org/10.1016/j.gloenvcha.2015.06.015.
Nogueira, D. S., L. B. Calvão, L. F. A. Montag, L. Juen & P. Jr De Marco, 2016. Little effects of reduced-impact logging on insect communities in eastern Amazonia. Environmental Monitoring and Assessment 188: 1–20. https://doi.org/10.1007/s10661-016-5431-z.
Osborne, T., S. Brock, R. Chazdon, S. Chomba, E. Garen, V. Gutierrez, R. Lave, M. Lefevre & J. Sundberg, 2021. The political ecology playbook for ecosystem restoration: principles for effective, equitable, and transformative landscapes. Global Environmental Change 70: 102320. https://doi.org/10.1016/j.gloenvcha.2021.102320.
Oksanen, J., F. G. Blanchet, M. Friendly, R. Kindt, P. Legendre, D. McGlinn, P. Minchin, R. B. O’Hara, G. L. Simpson, P. Solymos, M. H. H. Stevens, E. Szoecs & H. Wagner, 2016. vegan: Community Ecology Package. R package version 2.4–1. Retrieved, from https://CRAN.R-project.org/package=vegan
O’Neill, M. E., 2000. A weighted least squares approach to Levene’s test of homogeneity of variance. Australian & New Zealand Journal of Statistics 42: 81–100. https://doi.org/10.1111/1467-842X.00109.
Pes, A. M. O., A. P. M. Santos, P. Barcelos-Silva & L. M. Camargos, 2014. Ordem Trichoptera. In Hamada, N., J. L. Nessimian & R. B. Querino (eds), Insetos Aquáticos na Amazônia brasileira: taxonomia, biologia e ecologia Manaus, Editora do INPA: 391–433.
Peck, D. V., A. T. Herlihy, B. H. Hill, R. M. Hughes, P. R. Kaufmann, D. J. Klemm, J. M. Lazorchak, F. H. Mccormick, S. A. Peterson, P. L. Ringold, T. Magee & M. R. Cappaert, 2006. Environmental Monitoring and Assessment Program – Surface Waters Western Pilot Study: Field Operations Manual for 16 Wadeable Streams. Environmental Protection Agency, EPA 600/R-06/003. U.S., Office of Research and Development, Washington, DC.
Pinay, G., R. Bernal, B. W. Abbott, A. Lupon, E. Marti, F. Sabater & S. Krause, 2018. Riparian corridors: A new conceptual framework for assessing nitrogen buffering across biomes. Frontiers in Environmental Science 6: 47. https://doi.org/10.3389/fenvs.2018.00047.
Prudente, B. S., P. S. Pompeu, L. Juen & L. F. Montag, 2017. Effects of reduced-impact logging on physical habitat and fish assemblages in streams of Eastern Amazonia. Freshwater Biology 62: 303–316. https://doi.org/10.1111/fwb.12868.
R Core Team, 2016. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/.
Ramalho Filho, A., P. E. M. Motta, P. L. Freitas & W. G. Teixeira, 2010. Zoneamento agroecológico, produção e manejo da cultura de palma de óleo na Amazônia. Rio de Janeiro, Embrapa Solos, 216 p. il. ISBN 978-85-85864-34-7
Ramírez, A., C. M. Pringle & K. M. Wantzen, 2008. Tropical Stream Conservation, Academic Press, In Tropical Stream Ecology:, 285–304.
Hartig F., 2022. DHARMa: residual diagnostics for hierarchical (Multi-Level / Mixed) regression models. R package version 0.4.5. https://CRAN.R-project.org/package=DHARMa
Ribeiro, C., M. E. Rodrigues, G. Sahlén & F. O. Roque, 2022. Dragonflies within and outside a protected area: a comparison revealing the role of well-preserved Atlantic forests in the preservation of critically endangered, phytotelmatous species. Journal of Insect Conservation 26: 271–282. https://doi.org/10.1007/s10841-022-00385-4.
Ricklefs, R., 1977. Environmental heterogeneity and plant species diversity: a hypothesis. American Naturalist 111: 376–381. https://doi.org/10.1086/283169.
Roebuck, J. A., Jr., M. Seidel, T. Dittmar & R. Jaffé, 2019. Controls of land use and the river continuum concept on dissolved organic matter composition in an anthropogenically disturbed subtropical watershed. Environmental Science & Technology 54: 195–206. https://doi.org/10.1021/acs.est.9b04605.
Roy, A. H., A. D. Rosemond, D. S. Leigh, M. J. Paul & J. B. Wallace, 2003. Habitat-specific responses of stream insects to land cover disturbance: biological consequences and monitoring implications. Journal of the North American Benthological Society 22: 292–307. https://doi.org/10.2307/1467999.
Salles, F. F. & E. Domínguez, 2012. Systematics and phylogeny of ulmeritus-ulmeritoides revisited (ephemeroptera: leptophlebiidae). Zootaxa 3571: 49–65.
Salles, F. F., J. M. C. Nascimento, P. V. Cruz, R. Boldrini, & E. L. L. Belmont, 2014. Ordem Ephemeroptera. In: N. Hamada, J. L. Nessimian & R. B. Querino (Eds), Insetos Aquáticos na Amazônia brasileira: taxonomia, biologia e ecologia. Manaus, Editora do INPA.
Sambhu, H., A. Nankishore, S. M. Turton & T. D. Northfield, 2018. Trade-offs for butterfly alpha and beta diversity in human-modified landscapes and tropical rainforests. Ecology and Evolution 8: 12918–12928. https://doi.org/10.1002/ece3.4732.
Seiferling, I., R. Proulx & C. Wirth, 2014. Disentangling the environmental-heterogeneity–species-diversity relationship along a gradient of human footprint. Ecology 95: 2084–2095. https://doi.org/10.1890/13-1344.1.
SEMAS - Secretaria de Meio Ambiente e Sustentabilidade, 2008. Resolution No. 04 of September. Retrieved January 2019, from https://www.sema.pa.gov.br/imagens/RESOL%20N%c3%82%c2%ba%2004.pdf.
Shimano, Y., L. Juen, F. F. Salles, D. S. Nogueira & H. S. R. Cabette, 2013. Environmental and spatial processes determining Ephemeroptera (Insecta) structures in tropical streams. International Journal of Limnology 49: 31–41. https://doi.org/10.1051/limn/2013036.
Silva, D. S. & P. Barreto, 2014. O aumento da produtividade e lucratividade da pecuária bovina na Amazônia: o caso do Projeto Pecuária Verde em Paragominas. IMAZON, Belém-PA.
Siqueira, T., C. G. L. T. Lacerda & V. S. Saito, 2015. How does landscape modification induce biological homogenization in tropical stream metacommunities? Biotropica 47: 509–516. https://doi.org/10.1111/btp.12224.
Silva, D. R. O., A. T. Herlihy, R. M. Hughes, D. R. Macedo & M. F. Callisto, 2018. Assessing the extent and relative risk of aquatic stressors on stream macroinvertebrate assemblages in the neotropical savanna. Science of the Total Environment 633: 179–188. https://doi.org/10.1016/j.scitotenv.2018.03.127.
Simões, N. R., L. S. Braghin, G. A. Duré, J. S. Santos, S. L. Sonoda & C. C. Bonecker, 2020. Changing taxonomic and functional β-diversity of cladoceran communities in Northeastern and South Brazil. Hydrobiologia 847: 3845–3856. https://doi.org/10.1007/s10750-020-04234-w.
Smith, N. J. H., E. A. S. Serrão, P. T. Alvim & I. C. Falesi, 1995. Amazônia: Resiliency and Dynamism of the Land and Its People, United Nations University Press, Tokyo:
Sponseller, R. A., E. F. Benfield & H. M. Valett, 2001. Relationships between land use, spatial scale and stream macroinvertebrate communities. Freshwater Biology 46: 1409–1424. https://doi.org/10.1046/j.1365-2427.2001.00758.x.
Stein, A., K. Gerstner & H. Kreft, 2014. Environmental heterogeneity as a universal driver of species richness across taxa, biomes and spatial scales. Ecology Letters 17: 866–880. https://doi.org/10.1111/ele.12277.
Stier, A. C., S. W. Geange, K. M. Hanson & B. M. Bolker, 2013. Predator density and timing of arrival affect reef fish community assembly. Ecology 94: 1057–1068. https://doi.org/10.1890/11-1983.1.
Strahler, A. N., 1957. Quantitative analysis of watershed geomorphology. Eos, Transactions American Geophysical Union 38: 913–920. https://doi.org/10.1029/tr038i006p00913.
Southwood, T. R. E., 1977. Habitat, the templet for ecological strategies? Journal Animal. Ecology 46: 337–365. https://doi.org/10.2307/3817.
Sueyoshi, M., N. Ishiyama & F. Nakamura, 2016. β-diversity decline of aquatic insects at the microhabitat scale associated with agricultural land use. Landscape and Ecological Engineering 12: 187–196. https://doi.org/10.1007/s11355-015-0283-1.
Suhonen, J., E. S. A. Korkeamäki, J. Salmela & M. Kuitunen, 2014. Risk of local extinction of Odonata freshwater habitat generalists and specialists. Conservation Biology 28: 783–789. https://doi.org/10.1111/cobi.12231.
Tanaka, M. O., A. L. T. Souza, L. E. Moschini & A. K. Oliveira, 2016. Influence of watershed land use and riparian characteristics on biological indicators of stream water quality in southeastern Brazil. Agriculture, Ecosystems & Environment 216: 333–339. https://doi.org/10.1016/j.agee.2015.10.016.
Terra Brasilis, 2022. PRODES program. Retrieved from http://terrabrasilis.dpi.inpe.br/app/dashboard/deforestation/biomes/legal_amazon/increments
Tonkin, J. D., J. Heino, A. Sundermann, P. Haase & S. C. Jähnig, 2016. Context dependency in biodiversity patterns of central German stream metacommunities. Freshwater Biology 61: 607–620. https://doi.org/10.1111/fwb.12728.
Turunen, J., V. Elbrecht, D. Steinke & J. Aroviita, 2021. Riparian forests can mitigate warming and ecological degradation of agricultural headwater streams. Freshwater Biology 66: 785–798. https://doi.org/10.1111/fwb.13678.
Venables, W. N. & B. D. Ripley, 2002. Modern Applied Statistics with S, 4th ed. Springer, New York:
Zar, J. H., 2010. Biostatistical Analysis, Pearson Prentice-Hall, Upper Saddle River:
Zorzal-Almeida, S., L. M. Bini & D. C. Bicudo, 2017. Beta diversity of diatoms is driven by environmental heterogeneity, spatial extent and productivity. Hydrobiologia 800: 7–16. https://doi.org/10.1007/s10750-017-3117-3.
Zuur, A. F., E. N. Ieno, N. J. Walker, A. A. Saveliev & G. M. Smith, 2009. Mixed Effects Models and Extensions in Ecology with R, Springer Science & Business Media, Berlin:
Wang, Y., G. Ziv, M. Adami, C. A. de Almeida, J. F. G. Antunes, A. C. Coutinho, J. C. D. M. Esquerdo, A. R. Gomes & D. Galbraith, 2020. Upturn in secondary forest clearing buffers primary forest loss in the Brazilian Amazon. Nature Sustainability 3: 290–295. https://doi.org/10.1038/s41893-019-0470-4.
Wilkinson, C. L., D. C. J. Yeo, H. H. Tan, A. H. Fikri & R. Ewers, 2018. Land-use change is associated with a significant loss of freshwater fish species and functional richness in Sabah, Malaysia. Biological Conservation 222: 164–171. https://doi.org/10.1016/j.biocon.2018.04.004.
Whittaker, R. H., 1960. Vegetation of the Siskiyou Mountains, Oregon and California. Ecological Monographs 30: 279–338.
West, T. A. P., E. Vidal & F. E. Putz, 2014. Forest ecology and management forest biomass recovery after conventional and reduced-impact logging in Amazonian Brazil. Forest Ecology and Management 314: 59–63. https://doi.org/10.1016/j.foreco.2013.11.022.
Wood, S. N., 2006. Generalized Additive Models: An Introduction with R, Chapman and Hall/CRC, Boca Raton:
Acknowledgements
We are grateful to Agropalma Group and Conservation International Brazil for their financial and logistic support for sampling in oil palm areas. In addition, we would like to thank Cikel LTDA, 33 Forest and Instituto de Floresta Tropical (IFT) for financial and logistic support for sampling in Reduced-impact logging area. The Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for financing the projetcs entitleds "Influência dos diferentes tipos de uso do solo sobre a biodiversidade na Amazônia Oriental" (process 449315/2014-2) and "Tempo de resiliência das comunidades aquáticas após o corte seletivo de madeira na Amazônia Oriental’’ (process 481015/2011-6). APJF would also like to thank the Fundação Amazônia Paraense de Amparo à Pesquisa (FAPESPA) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) by scholarship (process 88882.176756/2018-01). LBC thanks CNPq for granting her a scholarship (process 154761/2018-4). RL and LJ would also like to thank CNPq for a research productivity fellowship (Grant # 312531/2021-4 and 304710/2019-9, respectively). We thank the PhD. Adolfo Calor, M.D. Carina KS. Paiva, PhD. Frederico F. Salles, PhD. Yulie Shimano, and M.D. Mylena Cardoso for their help with identification of Trichoptera and Ephemeroptera specimens.
Funding
Funding was provided by Agropalma Group, Conservation Internation Brazil, Cikel LTDA, Conselho Nacional de Desenvolvimento Científico e Tecnológico, Fundação Amazônia Paraense de Amparo à Pesquisa, Coordenação de Aperfeiçoamento de Pessoal de Nível Superior.
Author information
Authors and Affiliations
Contributions
APJF, LBC, RL, and LJ designed the study and analyzed the data and manuscript writing. XG and MAL contributed to the manuscript and revisions.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Guest editors: Luiz Ubiratan Hepp, Frank Onderi Masese & Franco Teixeira de Mello / Stream Ecology and Environmental Gradients
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Faria, A.P.J., Ligeiro, R., Calvão, L.B. et al. Land use types determine environmental heterogeneity and aquatic insect diversity in Amazonian streams. Hydrobiologia 851, 281–298 (2024). https://doi.org/10.1007/s10750-023-05190-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-023-05190-x