Abstract
The conversion of riparian vegetation into monocultures alters the input of allochthonous energy and the heterotrophy of streams. Here, we aimed to assess the implication of leaf litter inputs from plant species of agricultural importance on decomposition in Amazonian streams. We hypothesized that decomposition is low in litter with high concentrations of lignin and cellulose, regardless of the origin of plant species (exotic or native). We incubated leaf litter from plant species of agricultural importance, such as cocoa (native) and oil palm (exotic), as a control, leaf litter of Hymenaea courbaril (native) in four streams for 60 days. Contrary to our initial expectations, the leaf litter from oil palm, which was of higher quality and had lower concentrations of structuring compounds (lignin and cellulose), did not differ in decomposition rates from the lower quality leaf litter of the control species, which presented higher concentrations of structuring compounds. We recorded higher decomposition rate in cocoa, which had intermediate concentrations of structural compounds. Our results indicate a negative effect of the conversion of riparian vegetation into oil palm monocultures (Elaeis guineensis) for the processing of organic matter in Amazonian streams, due to the slower decomposition rate of the leaf litter of this species.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Agriculture is one of the human activities that most threaten freshwater ecosystems worldwide, causing biodiversity loss and impacts on ecosystem processes (Carpenter et al. 2011). Removal of riparian vegetation is an agricultural practice that also involves deforestation and plantations of exotic species. This can alter the spatial and temporal distribution of organic matter entering into streams, changing the quality and quantity of suitable inputs for ecosystem functioning (Graça et al. 2002; Leite-Rossi and Trivinho-Strixino 2012; Boyero et al. 2012, 2014).
Leaf litter from riparian vegetation is the main energy resource that enters into the stream (Wallace et al. 1997; Tank et al. 2010; Benfield et al. 2017). When leaf litter enters the stream, decomposition begins as a process usually driven by aquatic microbial communities (bacteria and fungi) and shredder invertebrates (Gonçalves et al. 2013; Graça et al. 2015). Nevertheless, shredders are generally scarce in tropical streams, and microbial decomposition becomes predominant on leaf litter (Rezende et al. 2015, 2018; Boyero et al. 2017). Bacterial communities act in the early stages of decomposition because of their high capacity to degrade soluble compounds, such as carbohydrates and phenols (Gonçalves et al. 2006; Alvim et al. 2015a). On the other hand, aquatic hyphomycetes contribute to the final decomposition stages by degrading structural compounds, such as lignin and cellulose (Graça et al. 2015, 2016).
The quality of leaf litter is a controlling factor of decomposition due to its direct effects on decomposers (Tank et al. 2010). Leaf litter quality can be defined as a set of physical and chemical characteristics that give nutritive and energetic value to microorganisms and invertebrates, leading to enhanced decomposition (Ardón and Pringle 2008; Wantzen et al. 2008). In tropical streams, leaf litter with higher concentrations of nutrients (e.g., nitrogen and phosphorus) and labile compounds tend to decompose more rapidly compared to leaf litter with higher concentrations of structural compounds, such as lignin and cellulose (Rezende et al. 2014; Gonçalves et al. 2017; Kiffer et al. 2018).
In the Amazon region, the exotic African species Elaies guineensis Jacq. (oil palm tree) and the native Amazonian species Theobroma cacao L. (cacao tree) are ostensibly cultivated, and the state of Pará is among the largest producers of palm oil and cocoa beans in Brazil (Fapespa 2017; Benami et al. 2018; Montag et al. 2021). Only in 2014, were planted in Pará state 256,000 ha of the oil palm tree, and it is estimated that ~ 30 million ha are suitable for cultivation in this region (Brandão and Schoneveld 2015). Also, the Pará state currently has a total area of 175,000 ha of cocoa cultivation (Melo et al. 2017).
Oil palm plantations often provide smaller quantity, less diverse and lower quality leaf litter and in consequence, result in slower decomposition (Chellaiah and Yule 2018). In lowland humid Ghana, forests recently converted to cocoa crops (3 years old) have a reduction in the supply of organic matter and slowed decomposition compared to secondary forests (Dawoe et al. 2010). Some studies have shown that oil palm monocultures negatively affect the diversity and taxonomic composition of aquatic insects in the Amazon streams (Cunha and Juen 2017; Paiva et al. 2017; Mendes et al. 2019), but no study has evaluated how palm oil leaf litter decomposes in Amazonian streams. Besides, one study performed in the Indian subcontinent evaluated colonization by aquatic hyphomycetes and cocoa leaves decomposition. In this study, cocoa leaves were rapidly colonized by aquatic hyphomycetes and decomposed rapidly compared to other crop leaf litter, such as coffee and cashew (Sridhar et al. 1992).
Considering the expansion of cultivation of E. guineensis and T. cacao in the Amazon region (Fapespa 2017; Benami et al. 2018; Montag et al. 2021), and due to the lack of information on the decomposition of leaf litter from both species in Amazonian streams, we aimed to evaluate the decomposition of leaf litter from exotic and native plant species used in economic exploitation in Amazon. We hypothesized that litter with high lignin and cellulose concentrations have reduced decomposition, due to a higher concentration of refractory compounds which decompose slowly, regardless of plant species origin (exotic or native species).
Material and methods
Study area
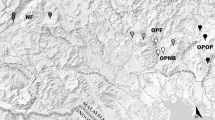
The study was conducted in streams inserted in forest remnants with predominant dense ombrophile forest. We conducted the experiments in forested streams to simulate possible ecological implications of the input of agricultural plant species on the leaf litter decomposition. We selected four streams in two micro basins of the Acará river drainage network, two streams in each micro basin, located in the municipality of Tailândia, northeast Pará State (Fig. 1). We used a hierarchical design to control for possible spatial autocorrelation effects on leaf litter decomposition. The region's climate is classified as tropical humid (Af), according to the Köopen-Geiger classification (Peel et al. 2007), with an average annual temperature of 26 °C, relative air humidity around 85% and an average annual rainfall of 2344 mm. The streams had an average of water temperature of 25.5 °C (± 0.5, standard deviation), pH of 4.9 (± 0.4), electrical conductivity of 0.16 µS/cm (± 0.002), and dissolved oxygen of 6.8 mg/L (± 2.8). The average stream width was 2.9 m (± 0.5) and depth was 38.2 cm (± 11.5).
Sampling design
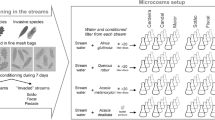
The decomposition experiment was conducted from September to November 2018 (60 days), during the dry season. We evaluated the leaf litter of two native species (Hymenaea courbaril and Theobroma cacao) and one exotic species (Elaeis guineensis). Both T. cacao and E. guineensis are of agricultural importance, while H. courbaril (Control) is found widely distributed in both dryland forests and riparian vegetation (Leite 2001, 2007). We collected senescent leaves of all species that recently fallen to the ground. Due to their high humidity, they were all dried for 72 h at 60 °C.
We used litter bags (26 × 16 cm) with coarse (10 mm) and fine (0.5 mm) mesh sizes. We selected four stream segments from first to third order (Strahler 1957), located at two micro basins, and in each segment, we selected three pool sites of similar physical conditions (see Fig. 2). The distance between pools was approximately 40 m and, in each one, one litter bags of each mesh size (coarse and fine) were incubated for each plant species studied. Thus, 24 litter bags containing 4.00 ± 0.05 g were installed for each species, totalling 72 litter bags. At the occasion of installation (incubation), the following physical–chemical parameters of the water were measured in each pool: pH, conductivity, width, depth, temperature, dissolved oxygen (Horiba model U-50, Japan) (see Supplementary material—Table S1).
After 60 days of incubation, litter bags were retrieved from the streams. In the laboratory, leaf litter were cleaned with running water using a 0.045 mm mesh sieve. The invertebrates retained in the sieve were preserved in 90% ethyl alcohol and identified at the lowest possible taxonomic level, according to the taxonomic key for the Amazonian region (Hamada et al. 2014). Later, invertebrates were classified into five functional trophic groups (filters, collectors, shredders, predators, and scrapers) according to specific literature for tropical environments (e.g., Cummins et al. 2005; Hamada et al. 2014) (see Table S2). We tested difference of abundance between leaf species only of the shredders and scrapers functional feeding groups, as they act more directly in litter decomposition (Gonçalves et al. 2017). However, we used the entire aquatic invertebrate assemblage to verify the colonization of decomposing leaf litter. The remaining leaf litter was dried for 72 h at 60 °C and weighed.
We estimated litter decomposition for each plant species (k day−1) using the negative exponential model, where mass was expressed in grams and time in days (e.g., Petersen and Cummins 1974), considering the following equation: k = − (ln [final mass/initial mass]/time). The rates of total and microbial decomposition (k total and k microbial) were determined from the coarse and fine mesh size bags, respectively. The contribution of invertebrates to the decomposition of leaf litter was estimated by the difference between the remaining mass in fine mesh and coarse mesh bags for each leaf litter type and then a new value of k was calculated (k invertebrates; Tonin et al. 2018).
Chemical analysis of leaf litter
To determine lignin and cellulose concentrations, leaf litter samples from each species were blade milled and then in a ball mill until obtaining a homogeneous and fine powder. All analyses were performed in triplicates. The concentrations of lignin and cellulose were obtained by gravimetric determination using the acid fibre detergent procedure (Gessner 2020).
Leaf litter from E. guineensis showed the lowest concentrations of lignin (32.5 ± 0.49%) and cellulose (33.5 ± 0.15%), followed by T. cacao (lignin = 36.1 ± 0.53% and cellulose = 35.6 ± 0.68%). Leaf litter of H. courbaril had the highest concentrations of these compounds (lignin = 40.5 ± 0.56% and cellulose = 35.9 ± 1.35%).
Data analysis
To test our hypothesis, we used Linear Mixed Effects models (LME), considering the decomposition rates of the total (k total), microbial (k microbial) and invertebrate-mediated (k invertebrates) as response variables. Species were used as a fixed effect and the spatial levels were replicated and nested hierarchically (micro basins: stream segments: pool sites) as a random effect. Litter bags were considered as the sample unit. The assumptions of normality and homogeneity of residuals were checked for all models. The adjusted models were subjected to an analysis of variance (ANOVA), and then we performed pairwise comparisons using Tukey’s test. Finally, we performed a direct ordination of the relative abundance of aquatic invertebrates according to the decomposition rate of each leaf litter (k day−1). All analyses were performed in R, version 3.6.2 using the ‘lme4’ and ‘multcomp’ packages (R Core Team 2019; Bates et al. 2015; Hothorn et al. 2008).
Results
We recorded lower percentage of remaining mass in leaf litter of T. cacao (62 ± 5%; mean ± standard deviation), followed by E. guineensis (74 ± 5%) and H. courbaril (74 ± 3%) (Fig. 3). Higher decomposition rates were recorded in T. cacao (total and microbial k = 0.008 ± 0.001 day−1). Total and microbial decomposition rates were similar in E. guineensis (total k = 0.006 ± 0.001; k Microbial = 0.005 ± 0.001 day−1) and H. courbaril (total k = 0.005 ± 0.001; k Microbial = 0.004 ± 0.004 day−1).
There was no effect of leaf litter species on invertebrate decomposition rates. Fixed effects explained 63% of the variance (marginal R2) of total k model. The marginal R2 for k microbial and k invertebrate models were 68% and 9%, respectively (Table 1, Fig. 4). Random effects had little influence on the response variables (see Table S3).
A total of 59 individuals belonging to 18 genera of aquatic invertebrates colonized the leaf litter bags (Fig. 5). There was no difference in the mean abundance of invertebrates in the leaf litter of all species (T. cacao = 3 ± 3; E. guineensis = 2 ± 2; H. courbaril = 2 ± 1) individuals/ bag. The shredders and scrapers were recorded in the detritus of all plant species, with relative abundances of 4 ± 4% (mean ± standard deviation) and 6 ± 7%, respectively.
Discussion
The total and microbial decomposition rates of native (T. cacao and H. courbaril) and exotic (E. guineensis) species can be classified as intermediate (k = 0.0041–0.0173), based on the classification for tropical species (Gonçalves et al. 2013). The chemical composition of leaf litter affected total and microbial decomposition, giving a better explanation microbial decomposition variance. For this reason, our results corroborate that the species of leaf litter influence the microbial community and the litter decomposition in tropical streams (Irons et al. 1994; Gonçalves et al. 2006; Boyero et al. 2015, 2017).
Although the litter from T. cacao showed intermediate concentrations of lignin and higher concentrations of cellulose than H. courbaril leaf litter, microbial communities were already recorded as able to efficiently process the cellulose of detritus in tropical rivers and streams (Gonçalves et al. 2017; Tiegs et al. 2019). Also, leaf litter characteristics may change over the time immersed in the streams (Graça et al. 2001). For example, Alvim et al. (2015b) observed that cellulose and lignin concentrations might increase or decrease over time depending on the species evaluated. Thus, the entry of T. cacao leaf litter into Amazonian streams will increase the availability of fine particulate organic matter, as this litter decomposes faster. Sridhar et al. (1992) observed that the rapid decomposition of cocoa leaves in streams is due to the non-use of pesticides in crops, enabling colonization by aquatic hyphomycetes. In addition, a rapid rate of decomposition of T. cacao leaves was observed in a river under the influence of cocoa cultivation associated with other plant species (that is, in agroforestry systems (Rojas et al. 2017).
Contrary to our initial expectations, the leaf litter from E. guineensis, which was of higher quality and had lower concentrations of structuring compounds, did not differ in total and microbial decomposition rates from the lower quality leaf litter of the control species H. courbaril, which presented higher concentrations of lignin and cellulose. This result contrasts with previous studies that found a positive relationship between litter quality and decomposition rates, regardless of the origin of leaf litter (Rezende et al. 2010; Gonçalves et al. 2017), indicating that litter quality is a controlling factor for organic matter decomposition in streams. Therefore, our results indicate a potential negative effect of leaf litter from the exotic species E. guineensis on the processing of organic matter and the functioning of Amazonian streams. Chellaiah and Yule (2018), in a study performed in Malaysia, found that the native leaves of Macaranga sp. decomposed five times faster than the leaf litter of E. guineensis, both in streams with natural riparian vegetation and in streams with direct and indirect effect from palm monocultures.
Unlike T. cacao leaf litter, the higher concentration of lignin in H. courbaril indicates that this compound could be a controlling factor for decomposition because it is a polymer with high resistance to enzymatic degradation by microorganisms (Kirk and Farrell 1987; Jabiol et al. 2019). Thus, leaf litter rich in lignin and cellulose tends to be intractable and tends to decompose slowly (Gessner 2020), corroborating our hypothesis that detritus with higher concentrations of lignin and cellulose decompose more slowly. Similarly, Gonçalves et al. (2017) found a negative correlation between lignin concentrations and the decomposition rates of native and exotic species.
Invertebrate-mediated decomposition did not differ between leaf litter sources. This can be explained by the low frequency of shredders and scrapers in our study, on average 4 and 6%, respectively. Our results are in line with Gonçalves et al. (2017), who found low frequencies of shredders (on average 6%) and scrapers (on average 5%) colonizing the leaf litter in Amazonian streams. Besides, some studies have shown that shredders are scarce and not remarkably diverse in tropical streams, suggesting that they have little influence on the decomposition process (Boyero et al. 2012, 2017; Gonçalves et al. 2012; Rezende et al. 2018).
The low abundance of invertebrates colonising the litter bags may reflect the low quality of leaf litter used in this study. Janke and Trivinho-Strixino (2007) observed that litter with high concentrations of structural compounds (e.g., lignin and cellulose) and secondary compounds might result in low colonization by aquatic invertebrates. This is exacerbated by the fact that low abundances of macroinvertebrates are usually found in many Amazonian streams (Montag et al. 2019). Also, high densities of invertebrates colonizing high-quality leaf litter, i.e., with high concentrations of nitrogen and phosphorus, had already been reported in previous studies (Rezende et al. 2010; Gonçalves et al. 2012, 2017).
Conclusions
Our hypothesis was partially corroborated, and the leaf litter from H. courbaril, with lower leaf quality, had decomposition values like those found in E. guineensis, a best quality leaf litter, both with high remaining mass. The leaf litter from T. cacao had intermediate lignin concentrations and high cellulose concentrations, and a lower remaining mass, suggesting that high cellulose concentrations can compensate for a high lignin content. Our results indicate that streams dominated by E. guineensis monocultures and T. cacao crop may potentially have their energy dynamics altered due to the low decomposition values to E. guineensis and high for T.cacao, which may impact the ecosystem functioning of streams in the Amazon region. Considering the constant conversion of native Amazonian forests to other types of land use, in addition to the palm oil and cocoa investigated here, it is important to continuously assess the impacts of land use changes on the processing of organic matter at Amazonian streams, since the input of leaf litter from exotic crops with low-quality leaves can compromise essential ecosystem processes.
References
Alvim EACC, Oliveira Medeiros A, Rezende RS, Goncalves JF (2015a) Small leaf breakdown in a Savannah headwater stream. Limnologica 51:131–138. https://doi.org/10.1016/j.limno.2014.10.005
Alvim EACC, Rezende RS, Gonçalves JF, Medeiros AO (2015b) Leaf breakdown in a natural open tropical stream. J Limnol 74(2):248–260. https://doi.org/10.4081/jlimnol.2014.982
Ardón M, Pringle CM (2008) Do secondary compounds inhibit microbial-and insect-mediated leaf breakdown in a tropical rainforest stream, Costa Rica? Oecologia 155(2):311–323. https://doi.org/10.1007/s00442-007-0913-x
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67(1):1–48. https://doi.org/10.18637/jss.v067.i01
Benami E, Curran LM, Cochrane M, Venturieri A, Franco R, Kneipp J, Swartos A (2018) Oil palm land conversion in Pará, Brazil, from 2006–2014: evaluating the 2010 Brazilian sustainable palm oil production program. Environ Res Lett 13(3):034037. https://doi.org/10.1088/1748-9326/aaa270
Benfield EF, Fritz KM, Tiegs SD (2017) Leaf-litter breakdown. In: Lamberti GA and Hauer FR (eds) Methods in stream ecology. Academic Press, pp 71–82
Boyero L, Barmuta LA, Ratnarajah L, Schmidt K, Pearson RG (2012) Effects of exotic riparian vegetation on leaf breakdown by shredders: a tropical–temperate comparison. Freshw Sci 31(2):296–303. https://doi.org/10.1899/11-103.1
Boyero L, Cardinale BJ, Bastian M, Pearson RG (2014) Biotic vs. abiotic control of decomposition: a comparison of the effects of simulated extinctions and changes in temperature. PLoS ONE 9(1):e87426. https://doi.org/10.1371/journal.pone.0087426
Boyero L, Pearson RG, Swan CM, Hui C, Albariño RJ, Arunachalam M, Cornejo A (2015) Latitudinal gradient of nestedness and its potential drivers in stream detritivores. Ecography 38(9):949–955. https://doi.org/10.1111/ecog.00982
Boyero L, Graça MA, Tonin AM, Pérez J, Swafford AJ, Ferreira V, Albariño RJ (2017) Riparian plant litter quality increases with latitude. Sci Rep 7(1):1–10. https://doi.org/10.1038/s41598-017-10640-3
Brandão F, Schoneveld G (2015) The state of oil palm development in the Brazilian Amazon: trends, value chain dynamics, and business models, vol 198. CIFOR.
Carpenter SR, Stanley EH, Vander Zanden MJ (2011) State of the world’s freshwater ecosystems: physical, chemical, and biological changes. Annu Rev Environ Resour 36:75–99. https://doi.org/10.1146/annurev-environ-021810-094524
Chellaiah D, Yule CM (2018) Litter decomposition is driven by microbes and is more influenced by litter quality than environmental conditions in oil palm streams with different riparian types. Aquat Sci 80(4):43. https://doi.org/10.1007/s00027-018-0595-y
Cummins KW, Merritt RW, Andrade PC (2005) The use of invertebrate functional groups to characterize ecosystem attributes in selected streams and rivers in south Brazil. Stud Neotrop Fauna Environ 40(1):69–89. https://doi.org/10.1080/01650520400025720
Cunha EJ, Juen L (2017) Impacts of oil palm plantations on changes in environmental heterogeneity and Heteroptera (Gerromorpha and Nepomorpha) diversity. J Insect Conserv 21(1):111–119. https://doi.org/10.1007/s10841-017-9959-1
Dawoe EK, Isaac ME, Quashie-Sam J (2010) Litterfall and litter nutrient dynamics under cocoa ecosystems in lowland humid Ghana. Plant Soil 330(1):55–64. https://doi.org/10.1007/s11104-009-0173-0
Fundação Amazônia de Amparo a Estudos e Pesquisas do Pará—Fapespa (2017) Boletim Agropecuário do Pará 2017. http://www.fapespa.pa.gov.br/upload/Arquivo/anexo/1383.pdf?id=1533567716. Accessed 6 Dec 2016
Gessner MO (2020) Lignin and cellulose. In Bärlocher F, Gessner M, Graça M (eds) Methods to study litter decomposition. Springer, Cham, pp 179–185
Gonçalves JF, França JS, Medeiros AO, Rosa CA, Callisto M (2006) Leaf breakdown in a tropical stream. Int Rev Hydrobiol 91(2):164–177. https://doi.org/10.1002/iroh.200510826
Gonçalves JF, Rezende RS, França J, Callisto M (2012) Invertebrate colonisation during leaf processing of native, exotic and artificial detritus in a tropical stream. Mar Freshw Res 63(5):428–439
Gonçalves JF, Martins RT, Ottoni BMP, Couceiro SRM (2013) Uma visão sobre a decomposição foliar em sistemas aquáticos brasileiros. In: Insetos Aquáticos: Biologia, Ecologia e Taxonomia. pp 1–41.
Gonçalves JF, Couceiro SR, Rezende RS, Martins RT, Ottoni-Boldrini BM, Campos CM, Hamada N (2017) Factors controlling leaf litter breakdown in Amazonian streams. Hydrobiologia 792(1):95–207. https://doi.org/10.1007/s10750-016-3056-4
Graça MS, Cressa CMOG, Gessner TO, Feio MJ, Callies KA (2001) Food quality, feeding preferences, survival and growth of shredders from temperate and tropical streams. Freshw Biol 46(7):947–957. https://doi.org/10.1046/j.1365-2427.2001.00729.x
Graça MA, Pozo J, Canhoto C, Elosegi A (2002) Effects of Eucalyptus plantations on detritus, decomposers, and detritivores in streams. Sci World J 2:1173–1185. https://doi.org/10.1100/tsw.2002.193
Graça MA, Ferreira V, Canhoto C, Encalada AC, Guerrero-Bolaño F, Wantzen KM, Boyero L (2015) A conceptual model of litter breakdown in low order streams. Int Rev Hydrobiol 100(1):1–12. https://doi.org/10.1002/iroh.201401757
Graça MA, Hyde K, Chauvet E (2016) Aquatic hyphomycetes and litter decomposition in tropical–subtropical low order streams. Fungal Ecol 19:182–189. https://doi.org/10.1016/j.funeco.2015.08.001
Hamada N, Nessimian JL, Querino RB (2014) Insetos Aquáticos na Amazônia Brasileira: Taxonomia. Biologia e Ecologia. Editora INPA, Manaus
Hothorn T, Bretz F, Westfall P (2008) Simultaneous inference in general parametric models. Biom J J Math Methods Biosci 50(3):346–363. https://doi.org/10.1002/bimj.200810425
Irons JG, Oswood MW, Stout RJ, Pringle CM (1994) Latitudinal patterns in leaf litter breakdown: is temperature really important? Freshw Biol 32(2):401–411. https://doi.org/10.1111/j.1365-2427.1994.tb01135.x
Jabiol J, Lecerf A, Lamothe S, Gessner MO, Chauvet E (2019) Litter quality modulates effects of dissolved nitrogen on leaf decomposition by stream microbial communities. Microb Ecol 77(4):959–966. https://doi.org/10.1007/s00248-019-01353-3
Janke H, Trivinho-Strixino S (2007) Colonization of leaf litter by aquatic macroinvertebrates: a study in a low order tropical stream. Acta Limnologica Brasiliensia 19(1):109–115. https://www.ablimno.org.br/acta/pdf/acta19_vol1_10.pdf.
Kiffer WP, Mendes F, Casotti CG, Costa LC, Moretti MS (2018) Exotic Eucalyptus leaves are preferred over tougher native species but affect the growth and survival of shredders in an Atlantic Forest stream (Brazil). PLoS ONE 13(1):1–17. https://doi.org/10.1371/journal.pone.0190743
Kirk TK, Farrell RL (1987) Enzymatic “combustion”: the microbial degradation of lignin. Ann Rev Microbiol 41(1):465–501. https://doi.org/10.1146/annurev.mi.41.100187.002341
Leite EJ (2001) Spatial distribution patterns of riverine forest taxa in Brası́lia, Brazil. For Ecol Manag 140(2–3):257–264. https://doi.org/10.1016/S0378-1127(00)00316-9
Leite EJ (2007) State-of-knowledge on Hymenaea courbaril L. var. Stilbocarpa (Hayne), Lee and Lang. (Leguminosae: Caesalpinioideae) for genetic conservation in Brazil. Res J For 1:1–26. https://doi.org/10.3923/rjf.2007.1.26
Leite-Rossi LA, Trivinho-Strixino S (2012) Are sugarcane leaf-detritus well colonized by aquatic macroinvertebrates? Acta Limnol Bras 24(3):303–313. https://doi.org/10.1590/S2179-975X2012005000048
Melo ADS, Batista SR, da Costa CA, Vilar JJ, França P, Augusto SG, Pereira DL (2017) Cocoa production systems with emphasis on aspects that improve production in the state of Pará Brazil. Amazonian J Plant Res 1(2):69–75. https://doi.org/10.26545/b00007x
Mendes TP, Benone NL, Juen L (2019) To what extent can oil palm plantations in the Amazon support assemblages of Odonata larvae? Insect Conserv Divers 12(5):448–458. https://doi.org/10.1111/icad.12357
Montag LF, Leão H, Benone NL, Monteiro-Júnior CS, Faria APJ, Nicacio G, Winemiller KO (2019) Contrasting associations between habitat conditions and stream aquatic biodiversity in a forest reserve and its surrounding area in the Eastern Amazon. Hydrobiologia 826(1):263–277. https://doi.org/10.1007/s10750-018-3738-1
Montag LFA, Begot TO, Prudente BS, Ferreira MC, Cunha EJ, Shimano Y, Paiva CKS, Carvalho FG, Mendes TP, Andrade AL, Barbosa LCF, Juen L (2021) A monocultura de palma de dendê Elaeis guineenses Jacq. e a biodiversidade de riachos amazônicos. Recurso Água, pp 131–161. https://doi.org/10.4322/978-65-86819-07-6-05.
Paiva CKS, de Faria APJ, Calvão LB, Juen L (2017) Effect of oil palm on the Plecoptera and Trichoptera (Insecta) assemblages in streams of eastern Amazon. Environ Monit Assess 189(8):393. https://doi.org/10.1007/s10661-017-6116-y
Peel MC, Finlayson BL, McMahon TA (2007) Updated world map of the Köppen-Geiger climate classification. Hydrol Earth Syst Sci 11:1633–1644. www.hydrol-earth-syst-sci.net/11/1633/2007/.\
Petersen RC, Cummins KW (1974) Leaf processing in a woodland stream. Freshw Biol 4(4):343–368. https://doi.org/10.1111/j.1365-2427.1974.tb00103.x
R Core Team (2019) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/. Accessed 28 Mar 2018.
Rezende RS, Gonçalves JF, Petrucio MM (2010) Leaf breakdown and invertebrate colonization of Eucalyptus grandis (Myrtaceae) and Hirtella glandulosa (Chrysobalanaceae) in two Neotropical lakes. Acta Limnol Bras 22(1):23–34. https://doi.org/10.4322/actalb.02201004
Rezende RS, Petrucio MM, Gonçalves JF (2014) The effects of spatial scale on breakdown of leaves in a tropical watershed. PLoS ONE 9(5):e97072. https://doi.org/10.1371/journal.pone.0097072
Rezende RDS, Leite GFM, de Lima AKS, Silva Filho LABD, Chaves CVC, Prette ACH, Gonçalves JF (2015) Effects of density and predation risk on leaf litter processing by Phylloicus sp. Austral Ecol 40(6):693–700. https://doi.org/10.1111/aec.12236
Rezende RS, Leite GFM, Ramos K, Torres I, Tonin AM, Gonçalves JF (2018) Effects of litter size and quality on processing by decomposers in a tropical savannah stream. Biotropica 50(4):578–585. https://doi.org/10.1111/btp.12547
Rojas J, Caicedo V, Jaimes Y (2017) Biomass decomposition dynamic in agroforestry systems with Theobroma cacao L. in Rionegro, Santander (Colombia). Agronomía Colombiana 35(2):182–189. https://doi.org/10.15446/agron.colomb.v35n2.60981
Sridhar KR, Chandrashekar KR, Kaveriappa KM (1992) Research on the Indian subcontinent. In: Bärlocher F (ed) The ecology of aquatic hyphomycetes. Springer, Berlin, pp 182–211
Strahler AN (1957) Quantitative analysis of watershed geomorphology. EOS Trans Am Geophys Union 38(6):913–920. https://doi.org/10.1029/TR038i006p00913
Tank JL, Rosi-Marshall EJ, Griffiths NA, Entrekin SA, Stephen ML (2010) A review of allochthonous organic matter dynamics and metabolism in streams. J N Am Benthol Soc 29(1):118–146. https://doi.org/10.1899/08-170.1
Tiegs SD, Costello DM, Isken MW, Woodward G, McIntyre PB, Gessner MO, Albariño R (2019) Global patterns and drivers of ecosystem functioning in rivers and riparian zones. Sci Adv 5(1):eaav0486. https://doi.org/10.1126/sciadv.aav0486
Tonin AM, Hepp LU, Gonçalves JF (2018) Spatial variability of plant litter decomposition in stream networks: from litter bags to watersheds. Ecosystems 21(3):567–581. https://doi.org/10.1007/s10021-017-0169-1
Wallace JB, Eggert SL, Meyer JL, Webster JR (1997) Multiple trophic levels of a forest stream linked to terrestrial litter inputs. Science 277(5322):102–104. https://doi.org/10.1126/science.277.5322.102
Wantzen KM, Yule CM, Mathooko JM, Pringle CM (2008) Organic matter processing in tropical streams. In: Dudgeon D (ed) Tropical stream ecology. Academic Press, pp 43–64
Acknowledgements
We thank the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) for the VF Master's grant. We acknowledge Grupo Agropalma and Denpasa for providing the necessary structure for the experiment development. We would like to thank the Graduate Program in Zoology (UFPA/ MPEG) at Federal University of Pará and the Laboratory of Ecology and Conservation (UFPA). The authors thank Continental Decomposition Experiment (CONDEX/AquaRiparia) for making the development of the experiment in Amazonian streams viable. We are grateful to Conselho Nacional de Desenvolvimento Científico e Tecnológico—CNPq) for scholarships to LJ (process 304710/2019-9). RTM was supported by Programa de Apoio à Fixação de Doutores no Amazonas-FIXAM/AM (FAPEAM) and INCT ADAPTA II funded by CNPq—Brazilian National Research Council (465540/2014-7), FAPEAM—Amazonas State Research (062.1187/2017). The authors thank Dr. Erlane Cunha, Dr. Fernando Carvalho and Jorge Pereira for support in the field and contributions.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Luz Boyero.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Firmino, V.C., Brasil, L.S., Martins, R.T. et al. Litter decomposition of exotic and native plant species of agricultural importance in Amazonian streams. Limnology 22, 289–297 (2021). https://doi.org/10.1007/s10201-021-00655-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10201-021-00655-1