Abstract
This study aimed to characterize species composition and abundance of fish assemblages of low-order streams of the Machado River Basin and to identify the main environmental factors associated to fish assemblages along the elevational gradient. Fishes were collected by seine and dip nets, along an 80-m stretch of 71 streams. In each site, we recorded physical, chemical, and land–water ecotone variables. We collected 22,702 specimens of 91 species. Species richness and abundance varied negatively with elevation. Ordination and similarity analyses detected that streams located in higher and middle elevation ranges differed in species composition from those in the lower elevation range. Almost one-third of the recorded species were selected as indicator species. Species of the families Trichomycteridae, Cichlidae, Gymnotidae, and Characidae were closely associated with the land–water ecotone and structural characteristics of lower elevation range, whereas several other Characidae species were more abundant in middle elevation range, where aquatic vegetation cover and conductivity values were higher. By assuming that composition and relative abundance of fish respond to environmental changes, hydrological characteristics may be viewed as environmental filters for stream assemblages by ultimately selecting the pool of species able to reach for specific elevation ranges and meet their biological requirements.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The turnover in composition and the increase in species richness, which occur from the headwaters to the river mouth, are widely known in fluvial ecology (Sheldon, 1968; Lomolino, 2001). Those processes occur in organisms with highly different life history and body size. Hence, the organization of fish assemblages in higher order watercourses results from recurrent processes of species addition, despite simultaneous (but generally less effective) replacement along the longitudinal gradient (Ormerod et al., 1994; Jacobsen, 2004; Lujan et al., 2013). For some organisms, such as producers (diatoms) and decomposers (bacteria), the variation in species richness with elevation is closely related to resource availability (e.g., nutrients and carbon supply; Wang et al., 2011). In fish assemblages, on the one hand, structural characteristics of the habitat regulate species variations along the elevational gradient, such as tributaries adjoining the main stem, the increases in microhabitat diversity, channel width, and flow. On the other hand, the distance from the source of immigrants and the permeability of the landscape are, in general, negatively correlated with elevation (Sheldon, 1968; Rahel & Hubert, 1991; Belliard et al., 1997). The spatial structure of river networks coupled with time-varying watershed disturbances generates habitat heterogeneity and, thus, potentially promotes biological diversity and productivity (i.e., network dynamics hypothesis sensu Benda et al., 2004).
The river continuum concept (hereafter RCC) is a conceptual framework that explains the functional composition of aquatic assemblages as a function of the longitudinal riverine gradient (Vannote et al., 1980). Although originally developed for macroinvertebrates, the predictions of the RCC have been also tested in plants and vertebrates in the past decades (Roper & Scarnecchia, 2001; Heino et al., 2005; Terra et al., 2016). According to the RCC, species distribution follows from the lower to the higher order of the main stem, mainly due to the maximization of organic matter processing and different trophic patterns throughout the longitudinal riverine gradient. However, the relative importance of stream order to species richness has been questioned, as anywhere in the river basin rivers of the same order show a pattern of monotonic decreases in the number of species from the headwaters to the mouth (Roper & Scarnecchia, 2001; Grant et al., 2007; Carvajal-Quintero et al., 2015). In this sense, additional effects of elevation may be related to environmental factors of physical and chemical gradients, such as temperature, current, and conductivity (Tejerina-Garro et al., 2005).
The predictors proposed to explain changes in diversity associated with elevation are (i) reduction in the inundated area and environmental complexity, (ii) increase in climatic harshness due to a higher influence of adjacent terrestrial ecosystems, (iii) decrease in diversity and availability of feeding resources, and (iv) decrease in primary productivity with increasing altitude (Huston, 1994). Studies dealing with these aspects were mostly carried out in low-order streams of high altitudes (Sheldon, 1968; Poff, 1997; Bistoni & Hued, 2002; Peres-Neto, 2004; Gerhard, 2005; Hoeinghaus et al., 2007). In tropical streams, most studies are limited to highlands (Jaramillo-Villa et al., 2010; Albert et al., 2011; Lujan et al., 2013; Carvajal-Quintero et al., 2015). Conversely, due to the short and smooth elevational gradient, the complex hydrographic network of the biogeographically diverse tropical lowland areas results in a high local diversity (see Abell et al., 2008), even in low-order water courses (Fu et al., 2004; Albert et al., 2011; Súarez et al., 2011; Lujan et al., 2013).
In the Amazon Basin, a myriad of low-order streams, regionally known as igarapés, constitute the most important landscape components (Anjos & Zuanon, 2007). The relevance of those streams follows from their high rates of species endemism, mostly small-sized fish species, which correspond to approximately 50% of the Amazonian fish diversity (Carvalho et al., 2008). Such aquatic diversity is now threatened in Brazil as the legal protection of forests up to 1,800 m a.s.l. is no longer mandatory (amendment to the Brazilian Forestry Law, Federal Law 12651 of 25 May 2012). This change may lead to the loss of unique species, deterioration of resources downstream, and disruption of ecological processes that are still poorly known (Casatti, 2010). Fish assemblages of low-order Amazonian streams may hide complementary factors other than those established as determinant for species composition in tropical rivers (Goulding, 1980; Finn et al., 2011).
On the northwestern extension of the Brazilian Shield, the Machado River originates at 600 m a.s.l. and flows 972 km north, until draining into the middle course of the Madeira River, the main tributary of the right bank of the Amazon River. Excepting the upper and lowermost sections, most of the Machado River Basin is between 200 and 300 m of elevation. Within that range and on the second half of the course, the Machado River drains on Tertiary Lowlands, where some tributaries and the main stem cascade through several cataracts. On the last 100 km, the river flows sinuously within the floodplain (Goulding, 1980). We assessed the structure of fish assemblages of 71 low-order clearwater streams of the Machado River Basin aiming to characterize species composition and abundance and to identify the main environmental factors responsible for their variation along the transition from the border of the Brazilian Shield to the Amazonian Lowlands. We hypothesized that fish assemblages respond more closely to physical and land–water ecotone variables of the altitudinal gradient even in the subtle elevation observed in southwestern Amazonia.
Materials and methods
Study area
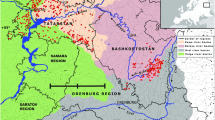
The present study was carried out in low-order streams of the Machado River (commonly listed as Ji-Paraná River) Basin, which covers 75,400 km2 of the State of Rondônia, Brazil (9–13°30′S, 63–61°30′W; Fig. 1). This seventh-order river is formed by the Pimenta Bueno and Comemoração rivers, and annually drains, on average, 700 m3 s−1 into the middle course of the Madeira River (Fernandes & Guimarães, 2002; Krusche et al., 2005). The Machado River Basin has different soil covers and land-use levels. It comprises from mostly unaltered habitats on the uppermost reach, near the two main tributaries, to a high level of alteration due to land use, as it reaches up to 500 m in channel width in the middle course. At this reach, the Machado River receives several tributaries, such as Rolim de Moura, Urupá, and Jaru Rivers (Krusche et al., 2005).
In Rondônia, old soils predominate, characterized as acids, weathered, nutrient poor, and having low cationic exchange capacity (Krusche et al., 2005). The regional climate is characterized by temperatures ranging from 19 to 33°C. The average annual rainfall is 2500 mm (Krusche et al., 2005), with two distinct seasons: the dry season, from late May to September, and the rainy season, from October to April (Fernandes & Guimarães, 2002).
Records of environmental data and fish sampling
Streams (up to third-order reaches sensu Horton–Strahler classification) were identified on hydrographic maps (at scales of 1:100.000) and included in the sampling design. Samplings were concentrated between June and September (dry season) of the years 2011, 2012, and 2013 and involved three collectors for up to 1 h of an intensive survey using seine and dip nets (2 mm mesh size) during daytime along an 80-m-long stretch, previously blocked with a net (0.5 cm mesh size) to prevent fish from escaping. All collected specimens were euthanized with an overdose of Eugenol, immediately fixed in 10% formalin, and transferred to 70% alcohol. Fish identification was carried out with the assistance of specialists and voucher specimens were deposited in the fish collection of the Department of Zoology and Botany, São Paulo State University, São José do Rio Preto, Brazil (DZSJRP 14335 to DZSJRP 19916).
We recorded six physical and chemical variables: dissolved oxygen, conductivity, temperature, mean width, mean depth, and current velocity. Immediately before fish sampling, the same collector (I. D. da Costa) visually estimated three other variables related to the structure of the land–water ecotone along the 80-m stretch: riparian vegetation (the relative cover of the stream bank, with roots, grasses, shrubs, and angiosperms), aquatic vegetation (the relative cover of the stream channel, with rooted and floating vegetation), and plant litter (the relative cover of the stream channel, with dead terrestrial plant material). Those variables and two other expressing micro-basin characteristics (the relative cover of mature forest and elevation) formed the environmental data matrix. Dissolved oxygen, electrical conductivity, and temperature were recorded with a YSI-85m. The average depth was calculated from 40 measurements of depth, randomly obtained in each reach, whereas the average width was calculated from four equidistant measurements across each sampling site. The water velocity was determined by measuring the time a floating object displaced through a known distance in the middle of the channel. The proportion of mature forest in the micro-basin and the elevation (m a.s.l.) of each sampling site were obtained from digital landscape elevation models (Table 1).
Data analysis
Sampling sites were categorized within three elevation ranges as follows: lower (13 streams up to 164 m a.s.l.), middle (49 streams between 175 and 258 m a.s.l.), and higher (9 streams higher than 286 m a.s.l.). Streams in the lower range of elevation drain to the Machado River mostly on its lowermost reach, whereas streams in the higher range of elevation drain mainly from the uppermost to the middle reach. Streams in the middle range of elevation are located from the middle to the lowermost reaches (Fig. 1; Appendix 1—Electronic Supplementary Material).
We used a matrix containing all environmental data to ordinate streams based on their physical, chemical, and structural characteristics. A principal component analysis (PCA) summarized this environmental matrix. Before the PCA, relative data were arcsine transformed, and all variables were standardized. We applied an unifactorial analysis of similarity (ANOSIM; Clarke & Green, 1988) to verify the differences in abundance along the elevation range, considering the range (lower, middle, and higher) as the predictor variable.
We determined indicator species for the higher, middle, and lower elevation ranges of low-order streams of the Machado River Basin on the dry season based on the occurrence and abundance of a given species considering each sample (INDVAL analysis; Dufrêne & Legendre, 1997). Species with probability of type I error < 5% (P ≤ 0.05; resulted from the random Monte Carlo reallocation procedure, based on 1,999 permutations) were considered potential indicators of a given elevation range. The null hypothesis assumed that the value (I I) for a given species i within a given elevation range j could be found at random (Legendre et al., 2009).
We used a redundancy analysis (RDA) to characterize the associations between fish assemblage structure and potential abiotic and biotic drivers at each site. RDA is a canonical ordination technique that uses PCA to explain variations in attributes through a matrix of environmental variables while preserving the Euclidean distance between the objects (Legendre & Legendre, 1998). For this analysis, fish abundance and environmental data were, respectively, log 10(x + 1) transformed and standardized. We exclude from the analyses those species that represented less than 1% of the total abundance. All analyses were performed using statistical packages in the R environment (version 3.2.5; R Core Team, 2016). PCA, RDA, and ANOSIM were performed using the “vegan” package (2.3-0; Oksanen et al., 2015), and INDVAL was performed in the “labdsv” (version 1.8-0; Roberts, 2016) package. Results were considered significant when P ≤ 0.05.
Results
Streams in the lower range of elevation showed low values of dissolved oxygen, electrical conductivity, and depth, but were wider than those located at higher altitudes. Streams in the middle range of elevation showed the highest values of electrical conductivity, whereas streams in the higher range of elevation showed the lowest temperature and the highest current velocity, and were much narrower than those in the middle and lower ranges of elevation (Table 1). Plant litter and mature forest cover predominated in streams up to 258 m a.s.l., especially in the lower range of elevation, whereas aquatic vegetation predominated on rivers of the middle and higher ranges of elevation (Table 1). The first two axes of the PCA summarized 42% of the variation in the environmental matrix (PC1 = 25% and PC2 = 17%; Table 1). The PCA ordinated samples according to the elevational gradient; the streams in the lower range of elevation were associated with high values of mature forest cover and gross litter, whereas those located in the higher range of elevation were more influenced by aquatic plants (Fig. 2).
We collected 22,702 specimens of 91 species. Species richness and abundance varied according to elevation. Streams in the lower range of elevation showed 5,360 specimens of 66 species (meanabundance = 412 ± 244 SD; meanrichness = 20 ± 5 SD), whereas those in the middle range showed 15,974 specimens of 77 species (meanabundance = 326 ± 322 SD; meanrichness = 19 ± 6 SD), and streams in the higher range of elevation showed 1,368 specimens of 45 species (meanabundance = 152 ± 150 SD; meanrichness = 11 ± 7 SD; Fig. 3; Appendix 1—Electronic Supplementary Material). The ANOSIM performed with abundance data confirmed significant differences between streams regarding the ranges of elevation (r = 0.46, P = 0.001; 9,999 permutations).
Almost one-third of the 91 species recorded showed significant indicator values for the dry season (Appendix 2—Electronic Supplementary Material). Characiformes [Astyanax cf. bimaculatus (Linnaeus, 1758)] were associated with rivers in the higher range of elevation. Two Characiformes (Cheirodon microdon Eigenmann, 1915, Microschemobrycon guaporensis Eigenmann, 1915) and a Siluriformes [Farlowella cf. oxyrryncha (Kner, 1853)] were associated with rivers in the lower range of elevation. A total of 22 species including Characiformes (ten species), Siluriformes (six species), Gymnotiformes (five species), and Perciformes (one species) were associated with rivers in the middle range of elevation. The remaining 65 species had the same likelihood of being found in streams of more than one elevation range and, therefore, are not indicator species in the sense of the INDVAL (Appendix 2—Electronic Supplementary Material).
The RDA explained 20% of the total variance in species composition and abundance (Fig. 4). Species of the families Trichomycteridae, Cichlidae, Gymnotidae, and Characidae were closely associated with land–water ecotone and structural characteristics represented mainly by the cover of mature forest and plant litter of streams in the lower range of elevation. On the other hand, several other Characidae species were more abundant in streams in the middle range of elevation, where aquatic vegetation cover and conductivity values were higher than those in other elevation ranges. Most species were associated with intermediate values of environmental variables and showed intermediate scores along the first two axes of the RDA (Appendix 2—Electronic Supplementary Material; unidentified points in the core of Fig. 4b).
Ordination of 71 low-order streams of the Machado River Basin (a) and 91 species (b) studied during dry season, along the first two axes of the redundancy analysis (RDA) relating species composition and abundance to environmental data. Arrow lengths correspond to the strength of relationships between the environmental variables (only those with correlations > 0.30 are shown) and the axes. Species scores and codes are presented in the Appendix—Electronic Supplementary Material. Streams in the lower (< 164 m a.s.l., white circle), middle (175–258 m a.s.l., gray circle), and higher ranges of elevation (> 286 m a.s.l., black circle)
Discussion
The composition and abundance of fish species in low-order streams of the Machado River Basin differed between the higher and middle ranges and the lower range of elevation. Species of the families Trichomycteridae, Cichlidae, Gymnotidae, and Characidae responded closely to the cover of mature forest and plant litter in the lower range, and several other species of Characidae were associated with aquatic vegetation cover and conductivity in the middle range of elevation.
Regardless of latitude, along the elevational gradient streams and rivers change from non-turbulent, turbid, and warm in the lowest range to very swift, turbulent, cold, and highly oxygenated in the highest range of elevation (Uieda & Castro, 1999; Jacobsen, 2008). Considering that fish composition and relative abundance respond to these environmental alterations (Jaramillo-Villa et al., 2010), hydrological characteristics may act as environmental filters for riverine assemblages, by selecting the pool of species able to reach specific elevation ranges according to their biological traits (Belliard et al., 1997; Poff, 1997; Martin-Smith, 1998).
The general pattern of species richness increasing toward downstream (Jacobsen, 2004; Albert et al., 2011; Lujan et al., 2013) might extend to southwestern Amazon, with variations according to elevation range. The fish fauna inhabiting low-order tributaries of the Machado River Basin varied from diverse, at low elevation (< 175–258 m a.s.l.), to less-specious assemblages, dominated by few species, at high elevations (> 286 m a.s.l.). However, a similar ratio of exclusive species was found among elevation ranges (seven species in lower, six species in middle and higher ranges).
In the 71 low-order streams of southwestern Amazon, the variables forest cover and gross litter best explained fish abundance and species richness of the streams located up to 164 m a.s.l. The association between those variables is reasonable, being the role played by the riparian forest in providing the input of leaves and branches to the streambed widely recognized. Besides preventing water heating and dissolved oxygen loss to the atmosphere by shade, forest provides a high contribution of allochthonous inputs of detritus to clearwater streams (Goulding, 1980). That terrestrial-derived litter increases the availability of nutritional resources and microhabitats, which promotes the coexistence of species with differential feeding requirements, ultimately resulting in increased species richness (Teresa & Casatti, 2010, 2012; Casatti et al., 2015). In addition, by being located on the lower elevation range, species richness of those streams might reflect the greater rates of species exchanges with the main stem, the floodplain, and the Madeira River (Goulding, 1981; Taylor & Warren, 2001; Tondato & Súarez, 2010). Low-order streams might contribute to enhance mainstream fish diversity when providing differential conditions for growth or survival, such as providing shelter for early life stages of rheophilic species (Benda et al., 2004), and this seems to be the case of the low-order streams of the Machado River Basin (Goulding, 1981). In an influential book detailing the movements of fish between the Madeira River main channel and clearwater tributaries, Goulding (1980) referred to the Machado River as one of the most important routes for upstream migration of recruits.
Low-order streams located above 286 m a.s.l. were associated with high cover of aquatic plants and showed the lowest values of fish abundance and species richness. The grassy cover of riverbanks and instream habitat may provide additional microhabitats that favor the colonization, reproduction, and feeding of aquatic insects, playing a role like that of aquatic macrophytes (Casatti et al., 2015). Macroinvertebrate availability may increase the persistence of opportunistic feeding fish species (Casatti et al., 2015) and seems reasonable to explain the strong association of some characins with middle and higher ranges of elevation.
By comparing fish assemblages among reaches defined according to elevation, we detected major differences in species richness, abundance, and composition between the streams located above 286 m a.s.l. and those located at lower elevations (< 258 m a.s.l.). We acknowledge that local composition might have been underestimated by the use of only two active sampling methods within a blocked stretch. However, due to logistic circumstances (i.e., accessibility, differences in water conductivity which might affect the effectiveness of electrical fishing) and for comparative purposes, we opted for the sampling effort similar to that employed in other studies on the region (e.g., Casatti et al., 2013). Fish assemblages responded more to elevational differences among ranges than within ranges, thus corroborating other studies, regardless of their location at low (Belliard et al., 1997; Súarez et al., 2011) or highland watersheds (Lujan et al., 2013). The fish fauna inhabiting the low-order streams of the Machado River Basin seems to display elevation preference, like species of other neotropical systems (Carvajal-Quintero et al., 2015). Almost one-third of the species were restricted and relatively abundant at a specific elevation range. The main patterns evidenced in the present study were the high number of species in streams at intermediate elevation (175–258 m) and the high species turnover from > 286 m a.s.l. to low-elevation streams. According to the literature, such changes can reflect differences in species habitat use (Jaramillo-Villa et al., 2010; Finn et al., 2011; Carvajal-Quintero et al., 2015). However, the role played by the middle reach cataracts as dispersal barriers and therefore reinforcing isolation mechanisms to populations of small- to medium-sized species should not be underestimated and deserves further investigations.
The variation in attributes of the structure of fish assemblages responded to the physical, chemical, land–water ecotone, and substrate composition characteristics that varied according to the elevational gradient within the Machado River Basin. Even though the studies about the neotropical fish fauna increased exponentially in the past decades, there are insufficient life history studies for most of the 91 species recorded herein.
In summary, there are significant longitudinal differences in fish composition of low-order streams within a river basin on the border of the Brazilian Shield and Amazonian Lowlands. Almost one-third of the species were restricted and relatively abundant in a specific elevation range. Seasonal studies of fish distribution and movement are needed to verify whether this pattern holds throughout the wet season. Nevertheless, the data provided a longitudinal insight on the importance of small streams and their high fish diversity and the needs to consider entire river basins when planning conservation initiatives.
References
Abell, R., M. L. Thieme, C. Revenga, M. Bryer, M. Kottelat, N. Bogutskaya, B. Coad, N. Mandrak, S. C. Balderas, W. Bussing, M. L. J. Stiassny, P. Skelton, G. R. Allen, P. Unmack, A. Naseka, R. Ng, N. Sindorf, J. Robertson, E. Armijo, J. V. Higgins, T. J. Heibel, E. Wikramanayake, D. Olson, H. L. Lopez, R. E. Reis, J. G. Lundberg, M. H. S. Perez & P. Petry, 2008. Freshwater ecoregions of the world: a new map of biogeographic units for freshwater biodiversity conservation. Bioscience 58: 403–414.
Albert, J. S., P. Petry & R. E. Reis, 2011. Major biogeographic and phylogenetic patterns. In Albert, J. S. & R. E. Reis (eds), Historical Biogeography of Neotropical Freshwater Fishes. University of California Press, Berkeley: 21–57.
Anjos, M. B. D. & J. Zuanon, 2007. Sampling effort and fish species richness in small terra firme forest streams of central Amazonia, Brazil. Neotropical Ichthyology 5: 45–52.
Belliard, J., P. Boët & E. Tales, 1997. Regional and longitudinal patterns of fish community structure in the Seine River Basin, France. Environmental Biology of Fishes 50: 133–147.
Benda, L. E. E., N. L. Poff, D. Miller, T. Dunne, G. Reeves, G. Pess & M. Pollock, 2004. The network dynamics hypothesis: how channel networks structure riverine habitats. Bioscience 54: 413–427.
Bistoni, M. A. & A. C. Hued, 2002. Patterns of fish species richness in rivers of the central region of Argentina. Brazilian Journal of Biology 62: 753–764.
Carvajal-Quintero, J. D., F. Escobar, F. Alvarado, F. A. Villa-Navarro, Ú. Jaramillo-Villa & J. A. Maldonado-Ocampo, 2015. Variation in freshwater fish assemblages along a regional elevation gradient in the northern Andes, Colombia. Ecology and Evolution 5: 2608–2620.
Carvalho, L. N., J. Zuanon & I. Sazima, 2008. Natural history of Amazon fishes. In Claro, P. S. O. & V. Rico-Gray (eds), Tropical Biology and Conservation Management. Eolss Publishers, Oxford: 1–32.
Casatti, L., 2010. Alterações no Código Florestal Brasileiro: impactos potenciais sobre a ictiofauna. Biota Neotropica 10: 1–31.
Casatti, L., M. A. Pérez-Mayorga, F. R. Carvalho, G. L. Brejão & I. D. Costa, 2013. The stream fish fauna from the Rio Machado Basin, Rondônia State, Brazil. Check List 9: 1496–1504.
Casatti, L., F. B. Teresa, Z. J. Oliveira, M. D. Ribeiro, G. L. Brejao & M. Ceneviva-Bastos, 2015. More of the same: high functional redundancy in stream fish assemblages from tropical agroecosystems. Environmental Management 55: 1300–1314.
Clarke, K. R. & R. H. Green, 1988. Statistical design and analysis for a “biological effects” study. Marine Ecology Progress Series 46: 213–226.
Dufrêne, M. & P. Legendre, 1997. Species assemblages and indicator species: the need for a flexible asymmetrical approach. Ecological Monographs 67: 345–366.
Fernandes, L. C. & S. C. P. Guimarães, 2002. Atlas geoambiental de Rondônia. SEDAM, Porto Velho.
Finn, D. S., N. Bonada, C. Múrria & J. M. Hughes, 2011. Small but mighty: headwaters are vital to stream network biodiversity at two levels of organization. Journal of the North American Benthological Society 30: 963–980.
Fu, C., J. Wu, X. Wang, G. Lei & J. Chen, 2004. Patterns of diversity, altitudinal range and body size among freshwater fishes in the Yangtze River Basin, China. Global Ecology and Biogeography 13: 543–552.
Gerhard, P., 2005. Comunidade de peixes de riachos em função da paisagem da bacia do rio Corumbataí, estado de São Paulo. USP, São Paulo.
Goulding, M., 1980. The Fishes and the Forest: Explorations in Amazonian Natural History. University of California Press, Los Angeles.
Goulding, M., 1981. Man and Fisheries on an Amazon Frontier. Kluwer Academic Publishers, Boston.
Grant, E. H. C., W. H. Lowe & W. F. Fagan, 2007. Living in the branches: population dynamics and ecological processes in dendritic networks. Ecology Letters 10: 165–175.
Heino, J., J. Parviainen, R. Paavola, M. Jehle, P. Louhi & T. Muotka, 2005. Characterizing macroinvertebrate assemblage structure in relation to stream size and tributary position. Hydrobiologia 539: 121–130.
Hoeinghaus, D. J., K. O. Winemiller & J. S. Birnbaum, 2007. Local and regional determinants of stream fish assemblage structure: inferences based on taxonomic vs. functional groups. Journal of Biogeography 34: 324–338.
Huston, A. H., 1994. Biological Diversity, the Coexistence of Species on Changing Landscapes. Cambridge University Press, London.
Jacobsen, D., 2004. Contrasting patterns in local and zonal family richness of stream invertebrates along an Andean altitudinal gradient. Freshwater Biology 49: 1293–1305.
Jacobsen, D., 2008. Tropical high-altitude streams. In Dudgeon, D. (ed.), Tropical Stream Ecology. Academic, London: 219–256.
Jaramillo-Villa, U., J. A. Maldonado-Ocampo & F. Escobar, 2010. Altitudinal variation in fish assemblage diversity in streams of the central Andes of Colombia. Journal of Fish Biology 76: 2401–2417.
Krusche, A. V., M. V. R. Ballester, R. L. Victoria, M. C. Bernardes, N. K. Leite, L. Hanada & B. M. Gomes, 2005. Efeitos das mudanças do uso da terra na biogeoquímica dos corpos d’água da bacia do rio Ji-Paraná, Rondônia. Acta Amazonica 35: 197–205.
Legendre, P. & L. Legendre, 1998. Numerical Ecology. Elsevier Science, Oxford.
Legendre, P., X. Mi, H. Ren, K. Ma, M. Yu, I. Sun & F. He, 2009. Partitioning beta diversity in a subtropical broad-leaved forest of China. Ecology 90: 663–674.
Lomolino, M. V., 2001. Elevation gradients of species-density: historical and prospective views. Global Ecology and Biogeography 10: 3–13.
Lujan, N. K., K. A. Roach, D. Jacobsen, K. O. Winemiller, V. M. Vargas, V. R. Ching & J. A. Maestre, 2013. Aquatic community structure across an Andes-to-Amazon fluvial gradient. Journal of Biogeography 40: 1715–1728.
Martin-Smith, K. M., 1998. Relationships between fishes and habitat in rainforest streams in Sabah, Malaysia. Journal of Fish Biology 52: 458–482.
Oksanen, J., F. G. Blanchet, R. Kindt, P. Legendre, P. R. Minchin, R. B. O’Hara, G. L. Simpson, P. Solymos, M. H. H. Stevens & H. Wagner, 2015. Vegan: Community Ecology Package. R package Version 2.3-0. https://CRAN.R-project.org/package=vegan.
Ormerod, S. J., S. D. Rundle, S. M. Wilkinson, G. P. Daly, K. M. Dale & I. Juttner, 1994. Altitudinal trends in the diatoms, bryophytes, macroinvertebrates and fish of a Nepalese river system. Freshwater Biology 32: 309–322.
Peres-Neto, P. R., 2004. Patterns in the co-occurrence of fish species in streams: the role of site suitability, morphology and phylogeny versus species interactions. Oecologia 140: 352–360.
Poff, N. L., 1997. Landscape filters and species traits: towards mechanistic understanding and prediction in stream ecology. Journal of the North American Benthological Society 16: 391–409.
R Core Team, 2016. R: A Language and Environment for Statistical Computing. R Core Team, Vienna. http://www.R-project.org.
Rahel, F. J. & W. A. Hubert, 1991. Fish assemblages and habitat gradients in a Rocky Mountain–Great Plains stream: biotic zonation and additive patterns of community change. Transactions of the American Fisheries Society 120: 319–332.
Roberts, D. W., 2016. Ordination and Multivariate Analysis for Ecology. R Package Version 1.8-0. http://ecology.msu.montana.edu/labdsv/R.
Roper, B. B. & D. L. Scarnecchia, 2001. Patterns of diversity, density, and biomass of ectothermic vertebrates in ten small streams along a North American river continuum. Northwest Science 75: 168–175.
Sheldon, A. L., 1968. Species diversity and longitudinal succession in stream fishes. Ecology 49: 193–198.
Súarez, Y. R., M. M. D. Souza, F. S. Ferreira, M. J. Pereira, E. A. D. Silva, L. Q. L. Ximenes & S. E. Lima Júnior, 2011. Patterns of species richness and composition of fish assemblages in streams of the Ivinhema River Basin, Upper Paraná River. Acta Limnologica Brasiliensia 23: 177–188.
Taylor, C. M. & M. L. Warren Jr., 2001. Dynamics in species composition of stream fish assemblages: environmental variability and nested subsets. Ecology 82: 2320–2330.
Tejerina-Garro, F. L., M. Maldonado, C. Ibañez, D. Pont, N. Roset & T. Oberdorff, 2005. Effects of natural and anthropogenic environmental changes on riverine fish assemblages: a framework for ecological assessments of rivers. Brazilian Archives of Biology and Technology 48: 91–108.
Teresa, F. B. & L. Casatti, 2010. Importância da vegetação ripária em região intensamente desmatada no sudeste do Brasil: um estudo com peixes de riacho. Pan-American Journal of Aquatic Sciences 5: 444–453.
Teresa, F. B. & L. Casatti, 2012. Influence of forest cover and mesohabitat types on functional and taxonomic diversity of fish communities in neotropical lowland streams. Ecology of Freshwater Fish 21: 1–10.
Terra, B. F., R. M. Hughes & F. G. Araújo, 2016. Fish assemblages in Atlantic Forest streams: the relative influence of local and catchment environments on taxonomic and functional species. Ecology of Freshwater Fish 25: 527–544.
Tondato, K. K. & Y. R. Súarez, 2010. Temporal changes in fish species composition of headwater streams of the upper Paraguay and Paraná Basins, Brazil. Acta Scientiarum Biological Sciences 32: 179–284.
Uieda, V. S. & R. M. C. Castro, 1999. Coleta e fixação de peixes de riacho. In Caramaschi, E. P. (ed.), Ecologia de peixes de riachos. Série Oecologia Brasiliensis, Rio de Janeiro: 1–22.
Vannote, R. L., G. W. Minshall, K. W. Cummins, J. R. Sedell & C. E. Cushing, 1980. The river continuum concept. Canadian Journal of Fisheries and Aquatic Sciences 37: 130–137.
Wang, J., J. Soininen, Y. Zhang, B. Wang, X. Yang & J. Shen, 2011. Contrasting patterns in elevational diversity between microorganisms and macroorganisms. Journal of Biogeography 38: 595–603.
Acknowledgements
This study is a part of the Thesis of I. D. da Costa in the Programa de Pós-Graduação em Ecologia e Evolução, Universidade do Estado do Rio de Janeiro. Monica P. de Araújo of the Universidade Federal do Rio de Janeiro and undergraduate students of the Fundação Universidade Federal de Rondônia assisted with designing Fig. 1 and the fieldwork, respectively. We are grateful to Dr. Francisco Langeani of the Universidade Estadual Paulista, campus de São José do Rio Preto, MSc. Willian Ohara of the Universidade de São Paulo, and Dr. João Alves de Lima Filho of the Universidade Federal de Rondônia for their help with taxonomic identification of the specimens, Dr. Lilian Casatti of the Universidade Estadual Paulista, campus de São José do Rio Preto, for providing suggestions on an earlier version of the manuscript and financial support (FAPESP: 2010/17494-8), and two anonymous reviewers for their helpful comments and suggestions. License for fish collections was provided by the Instituto Brasileiro do Meio Ambiente e dos Recursos Naturais Renováveis (IBAMA # 4355-1).
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: John M. Melack
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Costa, I.D., Petry, A.C. & Mazzoni, R. Responses of fish assemblages to subtle elevations in headwater streams in southwestern Amazonia. Hydrobiologia 809, 175–184 (2018). https://doi.org/10.1007/s10750-017-3463-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-017-3463-1