Abstract
While intermittent stream research has increased dramatically in recent decades, it remains difficult to predict traits that allow biota to persist in response to reduced flows and prolonged intermittent conditions. We quantified flow duration (FD) in ten reaches of six streams previously categorized as intermittent or perennial in north-central Arkansas. Macroinvertebrates were sampled in six streams in autumn and spring following a summer when six reaches did not maintain connected flow and four that maintained connected flow. Seasonal changes in discharge altered wetted habitat availability in all reaches and differences in FD influenced the magnitude of change in habitat availability. Richness increased in all reaches in spring due to more taxa that were poor dispersers, with traits such as low crawling rate and weak female dispersal. Richness was similar across reaches in both seasons but macroinvertebrate communities differed depending on FD. Perennial reaches were characterized by large predators and rheophilic taxa, while reaches with shorter FD were characterized by shredders and non-insect taxa. This study can inform conservation and restoration activities that aim to improve or maintain connectivity between intermittent and perennial habitats to increase local biodiversity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Intermittent streams make up over 50% of total stream length in the United States and represent a similar proportion of stream length worldwide (Arscott et al., 2010; Datry et al., 2014b). Stream hydrology is predicted to become more variable and intermittent from increased water withdrawals, dam construction, and global climate change (Stevenson & Sabater, 2010; Döll & Schmied, 2012; Steward et al., 2012). Higher air temperatures and rates of evapotranspiration are expected to increase the frequency of low-flow and drought conditions (Larned et al., 2010).
Intermittent stream research increased dramatically in the last 15 years, but it is still hard to predict traits that allow biota to persist in response to reduced flows and prolonged periods of drying (Robson et al., 2011; Leigh et al., 2015). The effect of increased intermittency on macroinvertebrate community structure is hard to predict due to variations across systems in climate, geomorphology, and antecedent disturbance regimes, as well as temporal variation in communities within freshwater systems (Beche & Resh, 2007; Robson et al., 2011; Leigh et al., 2016). In spite of these challenges, intermittent streams are major components of river networks worldwide, and it is important to understand how altered intermittent flow regimes will influence macroinvertebrate persistence and consequently biodiversity at the local scale (Zuellig & Schmidt, 2012; Dodds et al., 2015).
Stream drying is one of the one of the strongest drivers of macroinvertebrate community composition and structure (Poff & Ward, 1989; Arscott et al., 2010). Flow duration (FD), the average number of days per year a reach has surface water, is often used as a metric to quantify macroinvertebrate community response to the frequency and the extent of stream drying (Poff & Ward, 1989; Arscott et al., 2010; Datry et al., 2014a). When streams dry, available habitats for macroinvertebrates disappear, decrease, or change in water quality (Stanley et al., 1997; Lake, 2003; Bogan et al., 2015). Riffle habitats disappear during water drawdown and riparian and hyporheic habitats decrease in extent. As flow reduces in the hyporheic or pools become disconnected, water quality decreases as dissolved oxygen concentrations decrease and temperatures and conductivity increase (Lake, 2003; Bogan et al., 2015). These changes in habitat size and quality are expected to limit connectivity between riparian, hyporheic, and benthic habitats and influence macroinvertebrate community composition and structure (Poff, 1997; Larned et al., 2010).
The idea that variation in abiotic conditions and habitat availability will influence ecological traits of an organism was first described by the habitat template hypothesis, and following theories of ecological filtering which purport spatiotemporal variation in habitat is an evolutionary force that selects for traits that maximize survival and recruitment (Southwood, 1977; Poff et al., 2006). FD is a major driver of spatiotemporal habitat variation and streams with shorter FD often have greater densities of macroinvertebrates with traits adapted for resistance and resilience to stream drying (Stubbington & Datry, 2013; Datry et al., 2014a; Bogan et al., 2015). Resistance refers to the ability of macroinvertebrate communities to persist through a disturbance and resilience is the capacity to recover to pre-disturbance metrics of community structure (Stanley et al., 1994; Datry et al., 2014a). Traits that confer resistance (e.g., desiccation resistance stages, plastron/spiracle respiration, low rheophily), resilience (long adult lifespan, high female dispersal, strong fliers, strong swimmers), or a combination of both strategies have been found in intermittent reaches after rewetting (Datry et al., 2014a; Bogan et al., 2015). In addition, taxa with traits adapted to isolated pool habitats (e.g., aerial respiration, swim habit) and predator avoidance (e.g., high crawling rate, armoring) have been observed in greater abundances in intermittent and low-flow conditions (Bonada et al., 2007; Walters, 2011). However, the selection for traits associated with isolated pool habitats depends on the persistence of pool habitat throughout drying, which does not always occur or is not reported.
Metrics that characterize macroinvertebrate community structure such as richness and diversity generally decrease with decreased FD (Datry, 2012; Datry et al., 2014a; Leigh et al., 2015) except in one system with an extremely resilient community adapted to multiple forms of disturbance such as floods and droughts (Vander Vorste et al., 2016). The mechanism that explains decreased richness with decreased FD remains equivocal. One proposed mechanism is the proximity of intermittent reaches to perennial reaches (Fritz & Dodds, 2004; Robson et al., 2011). Macroinvertebrates recolonize intermittent reaches after rewetting through drift from upstream perennial habitat, aerial dispersal, redistribution from refuge (e.g., hyporheic habitats, debris dams, pools), or emergence from diapause (Robson et al., 2011; Bogan et al., 2015). Therefore, intermittent reaches with greater hydrologic connectivity or proximity to perennial habitats often have macroinvertebrate communities similar to perennial reaches, and community composition of intermittent streams is generally a subset of perennial streams (Feminella, 1996; Price et al., 2003; Bonada et al., 2007; Datry et al., 2014a). Another proposed mechanism that controls richness is the long- and short-term antecedent hydrologic conditions within a watershed (Arscott et al., 2010; Leigh et al., 2016). While the metrics used to describe antecedent conditions vary across studies, measures of FD and frequency of disturbance are usually included (Fritz & Dodds, 2005; Arscott et al., 2010; Rolls et al., 2012; Leigh et al., 2016).
In addition to richness and diversity, macroinvertebrate density and biomass are generally lower in streams with shorter FD (Grimm & Fisher, 1989; Chadwick & Huyrn, 2007). Density is likely lower due to loss of riffle habitat and low recruitment following summer drying (Miller & Golladay, 1996; Muñoz, 2003; Fritz & Dodds, 2004). Biomass is expected to be lower in streams with shorter FD as communities are likely dominated by short-lived, small-bodied taxa (Huryn & Wallace, 2000; Lytle, 2001), and large-bodied macroinvertebrates have been shown to decrease in abundance in streams with shorter FD (Beche et al., 2006; Chadwick & Huyrn, 2007; Arscott et al., 2010). Of the few studies that have measured macroinvertebrate density and biomass in intermittent streams, both metrics decreased with shorter FD, but statistical differences between intermittent and perennial macroinvertebrate biomass were rarely calculated (Grimm & Fisher, 1989; Muñoz, 2003; Chadwick & Huyrn, 2007). While density and biomass were generally correlated, biomass could differ from density in intermittent versus perennial habitats. For example, if recently rewetted reaches had more small-bodied taxa due to colonization from small, resilient taxa, then intermittent reaches would have greater density and less biomass than perennial reaches after rewetting.
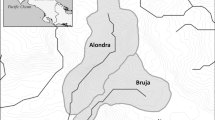
To try and identify traits that link macroinvertebrate resistance and resilience to stream drying, we characterized a range of FDs and community structure in six streams in two watersheds in north-central Arkansas. The watersheds span the Arkansas River valley and the Boston Mountains, a subregion of the Ozark Highlands. Four streams were classified as intermittent and two were classified as perennial by the United States Geological Survey (USGS) (Watershed Boundary Dataset, http://datagateway.nrcs.usda.gov) (Fig. 1). Our first objective was to quantify on a daily scale FD in six reaches of five streams where we observed extensive drying in summer (Low Cedar, High Cedar, Sis Hollow, Dry Hollow, Point Remove, and Powerline) and in four reaches of the South Fork Little Red River (SFLRR) that typically did not dry. Our goal was to characterize FD more precisely than intermittent versus perennial and describe FD in a conceptual diagram that explained how differences in FD translated into differences in available habitat.
Streams represented as solids lines were classified as perennial by the USGS Watershed Boundary Dataset and streams with dashed lines were classified as intermittent. Site symbols represent flow duration (FD) categories determined by the average number of days with connected flow and the presence or absence of isolated pools during stream drying. Reaches classified as perennial were along a 12-km section of the South Fork Little Red River (SFLRR), reaches with 7-month FD included Point Remove, Powerline, and Dry Hollow, and reaches with 9-month FD were low and high Cedar and Sis Hollow. The top inset shows the location of the 5 gages where low-flow and high-flow indices were calculated and their location relative to the 10 study reaches which were located within the black box
Our second objective had two parts: (1) quantify macroinvertebrate density, biomass, richness, and diversity in streams that differed by FD and (2) quantify densities of macroinvertebrates with traits adapted for resistance or resilience after stream drying. In reaches with shorter FD, we expected lower macroinvertebrate richness, diversity, density, and biomass due to reduced habitat availability and time for recruitment. We predicted that differences in FD would alter wetted habitat availability and result in greater densities of macroinvertebrates with traits that reflect adaptations for the use of refuge during drying and traits that reflect a capacity to disperse and recolonize streams upon rewetting. We used biological and ecological species traits as a framework to assess differences in community structure that would reflect responses to different habitat availability depending on FD (sensu Poff et al., 2006). We compared ten species traits hypothesized as advantageous for colonization (female dispersal ability, crawling rate, voltinism), persistence (desiccation resistance, armoring, functional feeding group), and ability to exploit refuge (mode of respiration, rheophily, ability to swim or burrow, size) in streams with shorter FD (Table 1).
Methods
Study area
The South Fork Little Red River (SFLRR) and Point Remove drainages are located in north-central Arkansas and span two ecoregions within the state, the Arkansas River valley and the Boston Mountains. Study reaches were all between 200- and 315-m elevations within the 60 km2 Gulf Mountain Wildlife Management Area which abuts the Ozark National Forest and is dominated by deciduous hardwoods (Quercus alba Linnaeus, Quercus rubra Linnaeus) and pines (Pinus taeda Linnaeus, Pinus palustris Linnaeus)(Birdsong, 2011). The wildlife management area was established by the state in 1970 and management practices since then have focused on maintaining grass and open forest for hunting through controlled burning. The remaining forested land is managed for timber production of loblolly and short leaf pine, and limited natural gas development started in 2010 (Austin et al., 2015).
Stream hydrology
Thirty-year average precipitation patterns in north-central Arkansas reflect low precipitation in summer (June–August, mean ± 1 SE: 86 ± 5 mm) and higher precipitation in autumn (September–November: 111 ± 7 mm) and spring (March–May: 117 ± 2 mm) (http://charts.srcc.lsu.edu/). Intermittent streams generally dry at some point in summer and resume connected flow in early autumn (Leasure et al., 2014). We sampled six headwater reaches in five streams that were previously observed to dry with varying duration in summer 2010. Four of the streams where we observed drying in summer were categorized as intermittent, and one was categorized as perennial by the USGS (Watershed Boundary Dataset, http://datagateway.nrcs.usda.gov). We sampled four reaches on the SFLRR that were characterized by the USGS as perennial (Fig. 1).
To highlight and identify long-term trends in seasonal changes in flow, a 90-day moving average was calculated for the gage with the longest record (1961–2012) in the SFLRR watershed, the most downstream USGS gage on the SFLRR in Clinton, Arkansas. FD curves (FDCs) and flow indices were calculated to characterize and compare antecedent conditions in the SFLRR and Point Remove drainages to calculate variation in flow frequency and magnitude within the study area. Mean daily discharge values were used to create FDCs, which result in cumulative frequency curves that represent the percent of time a given discharge was equaled or exceeded, and can be used to characterize the magnitude and frequency of average daily discharge over the historical period of record (Vogel & Fennessey, 1995).
A high-flow index of Q10 represented less frequently observed high flows and corresponded to discharges met or exceeded 10% of the year; low-flow indices of Q75 and Q90 identify discharges that met or exceeded 75 and 90% of the year, respectively (Smakhtin, 2001; Pastor et al., 2013). FDCs and flow indices were calculated for the most upstream SFLRR study reach (SFLRR Site1), the most downstream SFLRR reach (SFLRR Site4), and the most downstream gage on the SFLRR in Clinton, Arkansas (SFLRR Clinton). FDCs were also calculated for West Fork Point Remove, a perennial river, and Cypress Creek, an intermittent stream, to provide a context to compare discharge variability of the SFLRR to other USGS-categorized intermittent and perennial systems in the study region (Example Perennial, Example Intermittent). The percent of days discharge was above or below flow indices was used to characterize antecedent flow conditions 446 days prior to sampling. Antecedent flow conditions were characterized for the water year prior to sampling (Oct 1, 2009–Sep 30, 2010), plus the days prior to autumn sampling (Sep 30, 2010 to Dec 21, 2010), except for at Site4 where discharge measurements were not recorded until Oct 2, 2010. These 446 days encompassed the wet and the dry season prior to sampling.
To characterize FD in intermittent reaches, we used camera-monitored gages located at each intermittent reach. The number of days that reaches maintained connected flow from October 2010 to October 2011 was recorded using cameras installed March 2011. Connected flow was defined as the connection of flow between riffles and pools within a reach, and thus we considered connectivity strictly as a hydrologic attribute and did not consider ecological aspects of connectivity which include the flow of energy and materials (sensu Pringle, 2003). FD for a reach was then defined as the number of days with connected flow for the 2010 water year. Statistical differences in FD among reaches were tested with a type III one-way analysis of variance (ANOVA) followed by a Tukey’s honestly significant difference test. Type III ANOVAs were used to help account for an unbalanced statistical design (five intermittent and four perennial reaches in autumn, six intermittent and four perennial reaches in spring) (Herr, 1986). The models tested for a difference in the total number of days with connected flow and included stream reach as a random effect. ANOVAs were conducted using the R language and environment for statistical computing (v. 3.3.1; R Core Team, 2009).
We observed the presence of isolated pools in intermittent streams in the absence of connected flow in summer of August 2012, and recorded the presence or absence of isolated pools within 300 m of intermittent reaches. The point transect method was used to quantify benthic habitat in each stream reach during the summer of 2010 and summer of 2011 following the methods of Gordon et al. (2004). Wetted width transects were established perpendicular to flow every ten meters within a reach. At each transect, substrate size class was recorded every 0.5 m (Wentworth, 1922). The proportion of each substrate was calculated for each stream reach, and differences among reaches were tested with a type III one-way ANOVAs. We quantified stream discharge at least once in autumn and once in spring for all reaches using the velocity–area method as described by Gordon et al. (2004). We delineated and quantified stream catchment area using a 30-m resolution National Elevation Dataset (NED) and Arc Hydro Tools for ArcGIS 9.
Macroinvertebrate sampling
All reaches were sampled in December 2010 and March 2011, excluding one intermittent reach in December 2010 because the stream had not yet resumed connected flow. Autumn sampling did not begin until December 2010 since streams dried in summer and continuous flow did not resume in intermittent reaches until the last week of November 2010. Senesced leaf litter was observed in intermittent reaches through late November in dry streambeds and through December. Therefore, we describe December sampling as representative of “autumn” macroinvertebrate communities. We sampled macroinvertebrates with a 32-cm-diameter Hess sampler with 250-µm mesh. We took ten samples in intermittent reaches that were 200 m long, and five samples in perennial reaches that were 100 m long. Perennial reaches were shorter and had fewer samples because there was less accessible sample habitat due to dangerously high discharge in perennial versus intermittent reaches.
In the laboratory, we separated macroinvertebrates into 1-mm and 250-µm size classes using nested sieves. Macroinvertebrates larger than 1 mm were sorted by eye and those less than 1 mm using a sample splitter, with subsamples having at least 100 individuals (Waters, 1969). Macroinvertebrates were counted and measured to the nearest millimeter and identified to the genus or the lowest practical taxonomic resolution (Stewart & Stark, 1988; Wiggins, 1996; Merritt et al., 2008). Species traits were assigned at the genera or family level depending on the lowest taxonomic level of taxa trait information in Poff et al. (2006), Merritt et al. (2008), and Pennak (1989). Trait density, the number of individuals with a given trait per square meter of stream bottom, was calculated by summing the densities of all taxa with a given trait in each benthic core. We then calculated average trait density of all benthic cores within a reach. We calculated taxon-specific biomass using length–mass regression relationships provided in Benke et al. (1999).
Macroinvertebrate data analysis
Linear mixed effects regressions were used to analyze the relationship among FD, season, and response variables (macroinvertebrate density, biomass, richness, diversity, and trait densities). We evaluated a nested mixed effect model with four models. The null model only included the random effect of stream reach. All other models included stream reach as a random effect and included season, FD plus season, or the interaction between FD and season. Normality was tested with the Kolmogorov–Smirnov test, and we assessed the distribution of residuals with residual plots. We did not control for family-wise error with a Bonferroni adjustment for multiple comparisons because it is overly conservative when controlling for type I error (Benjamini & Hochberg, 1995; Verhoeven et al., 2005). Models were ranked using Akaike Information Criterion corrected for small sample size (AICc; Burnham & Anderson, 2002). Models were chosen if the change in AICC was greater than two compared to all other models. All regressions were done in the R language and environment for statistical computing (v. 3.3.1; R Core Team, 2009) using the nlme package (Pinheiro et al., 2016).
We used non-metric multidimensional scaling (NMS) in PC-ORD (version 6.0; MjM Software, Gleneden Beach, Oregon) to analyze differences in macroinvertebrate community structure between reaches with different FDs. We relativized taxa density and biomass as a percentage of total macroinvertebrate density and biomass to compare the proportion of each taxon within a reach to the proportion of taxa in all other reaches. Taxon density and biomass were considered significantly correlated with NMS axes if the Kendall’s tau coefficient coefficient (tau value) was less than −0.45 or greater than 0.45 (Rohlf & Sokal, 1994).
We used Multi-Response Permutation Procedure (MRPP) to test for differences in taxonomic communities based on FD. We used Indicator Species Analysis (ISA) to identify taxa that were exclusive to reaches grouped by hydrology and that occurred at all sites within a group (McCune & Grace, 2002). ISA produced an indicator value (IV) that was the product of the relative abundance and relative frequency of taxa within a group. The resulting IV ranged from 0 to 100 with 100 representing taxa that were both exclusive and fidelis to a group (McCune & Grace, 2002). We tested the significance of a taxon identified by ISA using Monte Carlo randomization with 1000 runs and taxa were considered indicative of each predetermined group at α = 0.05.
Results
Hydrology and reach characteristics
While categorized as a perennial river, the SFLRR had a minimum discharge of 0 cm in 31 out of 46 years of records (Fig. 2A). Average daily discharge for the SFLRR was progressively lower with distance upstream (Fig. 2B). For example, Site1, the most upstream gage on the SFLRR, had the lowest average daily discharge; the most downstream site (SFLRR Clinton) had the highest average discharge on the SFLRR. In addition, the magnitude and frequency of discharges at SFLRR Site1 were more similar to those at the example intermittent reach, and discharges at SFLRR Site4 were more similar to those at the example perennial reach (Fig. 2B). Thus, the SFLRR had flow regime characteristics intermediate between known intermittent and known perennial reaches in the study region (Fig. 2B).
Ninety-day moving average of mean daily discharge in cubic meters per second (CMS) at the lowest gaging station on the South Fork Little Red River (SFLRR Clinton) from 1961 to 2013 (A). Flow duration curves (FDC) for a known perennial (Example Perennial) and a known intermittent stream (Example Intermittent) in the study region, and for SFLRR Site1, SFLRR Site4, and SFLRR Clinton (B). Mean average daily discharge 464 days prior to sampling at SFLRR Site 1, and gray lines represent low-flow indices (Q90 = 0.01 CMS, Q75 = 0.06 CMS) and high-flow index (Q10 = 5.6 CMS)(C). Discharge exceedance probability represents the percent of time a given discharge is predicted to be met or exceeded over the period of record
Antecedent flow conditions characterized by high-flow and low-flow indices reveal that all streams experienced average magnitude and frequencies of daily discharges prior to sampling (Appendix 1—Supplementary material). In the 464 days prior to sampling, reaches exceeded low-flow indices, Q75 and Q90 on average 20% and 5% of the time (± 4.3 and ± 3.8 SD), and exceeded the high-flow index, Q10 on average 12% of the time (± 7.4 SD) (Appendix 1—Supplementary material). While perennial reaches maintained connected flow throughout the year, low flows (below Q75) were observed from July through November prior to sampling (SFLRR Site1 Fig. 2C), the same period intermittent reaches were dry. During this low-flow period, Site1 and Site4 had many riffles that contracted to very narrow, shallow channels between pools, resulting in shrinkage of riffle habitat.
Streams were classified into three FD groups based on the average days of connected flow per year and the presence of pools during stream drying. We observed three out of six intermittent reaches that did not maintain pools once connected flow ceased in summer, and had connected flow for 191, 218, and 249 days of the year between October 2010 and 2011 (Table 2). The remaining three intermittent reaches maintained pools during summer drying and had connected flow for 254, 295, and 307 days a year. All perennial reaches had connected flow for 365 days a year except for the uppermost reach which had connected flow for 350 days of the year. Reaches with 219-day average FD that did not retain pools throughout drying were categorized in the 7-month FD group. Those reaches with an average of 285-day FD and those that maintained isolated pools during summer were categorized in the 9-month FD group; reaches with 364-day average FD were classified as perennial. Reaches had statistically different days of FD based on these classifications (ANOVA F 2,7 = 54.8, P < 0.01, Tukey’s HSD, P < 0.01).
Catchment area was significantly larger between perennial reaches and reaches with 7- and 9-month FDs (hereafter intermittent reaches), but there was no difference in catchment area between intermittent reaches (ANOVA F 2,7 = 57.4, P < 0.01, Tukey’s HSD, P < 0.01) (Table 2). Similar to catchment area, discharge was significantly lower in intermittent than perennial reaches, but not different between intermittent reaches (ANOVA F 2,7 = 85.9, P < 0.01, Tukey’s HSD, P < 0.01).
Benthic substrate in all streams was composed primarily of gravel (avg. 22%), cobble (avg. 24%), and boulder (19%). Benthic substrate was not significantly different among reaches that differed by FD except for 23% more cobble substrate in reaches with 9-month FD compared to perennial reaches (ANOVA, F 2,7, P = 0.03, Tukey’s HSD, P = 0.03). There was no difference in cobble between intermittent reaches.
Macroinvertebrate community structure
A total of 93 taxa were identified. Five taxa were only collected in reaches with 7-month FD (Chrysops, Collembola, Hexagenia, Molophilus, and Rhyacophila); two taxa were collected exclusively in reaches with 9-month FD (Caloparyphus and Helichus). Ten taxa were collected exclusively in perennial reaches (Chauloides, Hagenius, Hansonoperla, Haploperla, Isonychia, Neoperla, Ordobrevia, Psychomyia, Sphaeriidae, Tabanus). There were 25 taxa collected in either classification of intermittent reaches. Taxa that were exclusive to reaches grouped by FD were not present at all reaches within a group, except Collembola which was present in all reaches with 7-month FD. Six taxa were present only in autumn and seven taxa were present only in spring. Fourteen taxa were non-insect, of which five were found exclusively in intermittent reaches (Collembola, Lirceus, Hyallela, Ostracoda, Caecidotea). One non-insect taxon, Sphaeriidae, was exclusive to perennial reaches. In autumn, non-insect taxa were 14% of total richness in reaches with 7- and 9-month FDs, respectively, and 10% of total richness in perennial reaches. In spring, non-insect taxa were 19% and 13% of total richness in reaches with 7- and 9-month FDs, respectively, and 8% of total richness in perennial reaches.
Macroinvertebrate richness increased in all reaches in spring, but density, biomass, and diversity did not differ significantly between seasons or among FDs (Fig. 3A–D, Table 3, Appendix 2—Supplementary material). Richness increased in spring by as much as five taxa in reaches with 7-month FD, and as little and two taxa in perennial reaches (Fig. 3C). A small change in AICc for the biomass and diversity mixed model from the null to season model suggested a trend in seasonal differences in biomass and diversity (Appendix 2—Supplementary material).
Box plots of macroinvertebrate density (A), biomass (B), richness (C), diversity (D), and tested trait densities (E–L). Gray bars represent Hess samples taken in autumn (December 2010) and white bars represent samples taken in spring (March 2011).Asterisks represent significant differences between autumn and spring samples (P = 0.05)
Of the ten species traits examined, eight modalities were explained by either season, FD, or by both factors (Table 3). Five modalities were different depending on season. Weak female dispersers, taxa with low crawling rate, taxa that prefer erosional habitat (i.e., rheophiles), swimmers, and collector-filterers increased in all reaches in spring (Table 3, Fig. 3E, G–I, K). The density of scrapers in each reach depended on season and FD, with greater densities of scrapers in intermittent reaches in both seasons and increased scraper density in all reaches in spring (Table 3, Fig. 3J). The densities of two trait modalities, taxa with sclerotization and shredders, differed depending on FD (Table 3, Fig. 3F, L). Taxa with sclerotization were greater in reaches with 7-month FD both seasons. In reaches with 7-month FD, Hydroptila, Lirceus, and Caecidotea made up 50% of sclerotized taxa density in autumn, while Caecidotea made up 50% of sclerotized taxa density in spring. Shredders had higher densities in intermittent reaches in both seasons (Fig. 3L). Non-insect taxa were 30% of shredder density, with the remainder composed of primarily stoneflies (Allocapnia, Amphinemura, Leuctra, Nemoura, and Zealeuctra). Average richness of each functional feeding group as a percentage of total richness for reaches grouped by FD was calculated to identify why richness did not differ depending on FD even though there were differences in community composition depending on FD. There tended to be more collector-filterers in perennial reaches versus more shredders in intermittent reaches in both seasons (Fig. 3K, L, Appendix 3—Supplementary material).
Non-metric multidimensional scaling ordinations resulted in stable solutions for macroinvertebrate community density and biomass. MRPP analysis indicated significant differences among macroinvertebrate density and biomass grouped by FD (density, A = 0.085, P < 0.001, biomass, A = 0.095, P < 0.001). The NMS of density and biomass separated reaches along two axes, but biomass separated reaches by FD more distinctly than taxa density, so only taxa biomass is presented (Fig. 4) (Appendix 4—Supplementary material). This NMS of taxa biomass resulted in a significant 2-axis solution. Axes 1 and 2 explained 21 and 49% of variation, respectively, in macroinvertebrate community composition. Macroinvertebrate communities were separated into 3 groups. Axis 2 separated intermittent from perennial reaches, while axis 1 separated reaches with 7-month FD from all other reaches. Reaches with 7-month FD had greater variation among sites especially in autumn (Fig. 4).
Non-metric multidimensional scaling (NMS) ordination of macroinvertebrate community biomass for reaches in autumn 2010 and spring 2011. Symbols with “A” represent reaches sampled in autumn and symbols with “S” represent spring samples. The proportion of variance explained by macroinvertebrate biomass for axes 1 and 2 was 21 and 49%, respectively (stress = 15.3)
The results of Kendall’s tau coefficient associated taxa on the positive side of axis 2 and the negative side of axis 1 with 7-month FD reaches and were represented by sclerotized shredders and scrapers (ordered by strength of association, Lirceus, Hyalella, Agapetus, Amphinemura, Collembola, Hydroporus) (Table 4). Taxa on the negative side of axis 2 were associated with perennial reaches and were represented by large-bodied predators, rheophiles, and collector-filterers (Stylogomphus, Argia, Neoperla, Sphaeriidae, Stenelmis, Cheumatopsyche) (Table 4). Stenonema and Caenis were clustered together in the center of the plot and were found at reaches with 9-month and perennial FD. Ectopria was also located in the center of the plot but was only found at perennial sites and was an indicator taxon of perennial sites.
Indicator species analysis
Twelve taxa were indicative of reaches with 7-month FD, seven taxa were indicative of reaches with 9-month FD, and ten taxa were indicative of perennial reaches. Indicator taxa in reaches with 7-month FD were non-insect taxa tolerant of low dissolved oxygen (e.g., Lirceus, Collembola, Ostracoda) and are known to reside in the hyporheic zone (Stubbington, 2012) (Table 5). From the ISA of taxa density, three large-bodied, semivoltine taxa were indicative of reaches that flowed 9-months (Gomphus, Psephenus, Tipula) and one small-bodied shredder (Allocapnia) (Table 5). Three additional predators (Nigronia, Ceratopogonidae, Tanypodinae) were identified as indicator taxa by biomass that were not identified by taxa density (Table 5). Taxa indicative of perennial reaches tended to be large-bodied, univoltine, collector-filterers and predators (e.g., Argia, Stylogomphus, Neoperla, Cheumatopsyche, Sphaeriidae). The only taxa with an IV score of 100 was Collembola because it was the only taxa exclusive to a hydrologic group and present at all sites in that group.
Discussion
As expected, differences in FD influenced the magnitude of change in habitat availability which shaped differences in species traits and taxonomic community composition. From observations and analyses, we outlined a conceptual diagram that summarizes changes in habitat availability depending on FD as discharge decreased in summer and increased in autumn (Fig. 5). Similar to the stream ecosystem expansion and contraction model (Stanley et al., 1997), changes in discharge from spring through summer and into autumn altered habitat availability. We identified transition periods in habitat availability according to the probability of drying for that period and habitat type (i.e., riffle, pool, hyporheic) (Fig. 5). To infer habitat availability, we were limited to field observations, discharge data collected prior to sampling, low-flow indices, and FD categories determined from our statistical analysis. We cannot know the exact dates and duration of time over which reaches lost longitudinal and lateral connectivity, or riffle, benthic, and hyporheic habitat decreased. We knew intermittent reaches that resumed connected flow in late autumn because we were not able to sample reaches until December 2010 due to lack of connected flow, and one intermittent reach was completely dry until February 2011. Based on the presence of isolated pools in some reaches and the persistence of connected flow between riffles and pools in perennial reaches, we assumed that reaches with 9-month FD had greater area and duration of suitable benthic habitat in summer than reaches with 7-month FD, and less suitable benthic habitat throughout the study period compared to perennial reaches (Fig. 5). Reaches with 9-month FD lost riffle habitat during summer, but perennial reaches did not, so we inferred that perennial reaches maintained riffle habitat for longer than intermittent reaches (Fig. 5). Reduced available benthic habitat influenced macroinvertebrate community structure by limiting recruitment of poor dispersers in all reaches from late summer through autumn. In addition, the persistence of riffle habitat in perennial reaches resulted in indicator taxa that were large-bodied rheophiles. Lastly, there were more non-insect taxa such as isopods and amphipods in reaches with 7-month FD because they likely could persist in disconnected flow habitats or use the hyporheic as refuge during drying.
Conceptual diagram of changes in habitat due to reduced flow or no flow in all reaches from July to December. Black represents full lateral and longitudinal connectivity, gray represents loss of lateral connectivity, vertical stripes represent a reduction in riffle and pool habitat, and dots represent when streams with 9-month FD were dry except for the presence of isolated pools. White represents when streams with 7-month FD were completely dry. The length of all bars from June to November is meant to convey the sequence over which habitat contraction or expansion occurred in one reach relative to another (After Bogan et al., 2015 and Stanley et al., 1997)
Contrary to what was expected, richness did not differ depending on FD and increased in all reaches in spring due to more rheophiles and poor dispersers that were likely limited by available colonization habitat during summer. For example, the density of taxa that were poor dispersers, with traits such as low crawling rate and weak female dispersal, increased from autumn to spring in all reaches. There is evidence that most stream taxa are limited to very short dispersal distances (< 1 km), and there is little difference in dispersal ability between intermittent and perennial streams (Chester et al., 2015). While perennial reaches did not experience complete drying, low mean daily discharges prior to sampling, as indicated by gages at Site1 and Site4, likely reduced riffle habitat (Stanley et al., 1997; Lake, 2003; Walters & Post, 2011). Additionally, weak dispersers are more likely affected by local differences in connected flow habitat than strong dispersers (Phillipsen & Lytle, 2013; Cañedo-Argüelles et al., 2015). Increased richness of rheophiles in spring is consistent with other studies that found rheophilic taxa increased just before dry periods in intermittent streams (Leigh, 2013) and loss of sensitive rheophiles during low flows (Boulton, 2003; Dewson et al., 2007a; Brooks et al., 2011; Walters & Post, 2011).
In general, macroinvertebrate richness is decreased with shorter FD (Arscott et al., 2010; Datry et al., 2014a). However, similar to this study, others have found instances of equal richness in streams that differed in FD, which was attributed to similar flood frequencies in both intermittent and perennial habitats (Fritz & Dodds, 2004; Leigh, 2013; Leigh et al., 2016). In this study, all reaches likely experienced a loss in richness or “reset” after large floods in spring, but differences in local habitat availability throughout summer and into autumn resulted in differences in community structure depending on FD. Taxon characteristics of perennial reaches were large predators (e.g., Argia, Stylogomphus, Sialis) and large collector-filterers (e.g., Isonychia, Cheumatopsyche, Sphaeriidae) due to the persistence of riffle habitat throughout summer. Intermittent reaches with 7-month FD were represented by sclerotized, non-insect taxa at all times of year such as Hyallela, Lirceus, and Caecidotea. Non-insect taxa such as isopods, copepods, ostracods, and mites are known residents of the hyporheic and possess traits such as a tolerance for low dissolved oxygen and the ability to burrow (Stubbington, 2012; Strachan et al., 2015). In reaches with 7-month FD, sclerotized taxa with the ability to persist in poorly oxygenated hyporheic conditions may have remained in the hyporheic throughout summer drying and migrated into benthic habitats in autumn, followed by increased recruitment throughout winter and spring (sensu Stubbington et al., 2016). Traits possessed by non-insect taxa allowed for their persistence in hyporheic habitats in reaches with 7-month FD, as described in other studies (Gibert et al., 1994; del Rosario & Resh, 2000; Vander Vorste et al., 2016).
Differences in community structure between reaches with 7- vs. 9-month flow duration were likely due to the presence or absence of isolated pools during stream drying. Non-insect taxa were more prevalent in reaches with 7- vs. 9-month FD, perhaps because insect shredders and/or predators persisted in isolated pools in reaches with 9-month FD, which displaced weak dispersers such as isopods, amphipods, mites, copepods, and ostracods. Descriptive evidence that disconnected pools serve as refuge for macroinvertebrates and may temporarily increase the density or richness of predators within temporary pool habitat was shown decades ago (Wiggins et al., 1980; Schneider & Frost, 1996). Evidence specific to disconnected pool habitat has also shown an increase in the richness of lentic taxa (Boulton & Lake, 1992; Bonada et al., 2006), but there is little evidence that taxa adapted to pool habitat influence community composition upon rewetting (Chester & Robson, 2011). Since we did not sample pool-specific habitat or disconnected pools, we cannot directly attribute the community composition of reaches with 9-month FD to isolated pools, but we conclude longer FD maintained more refuge habitat for a longer time period, which allowed for the persistence of predacious, indicator taxa such as Nigronia, Ceratopogonidae, and Tanypodinae.
More organic matter retention in summer and early autumn likely resulted in high densities of shredders throughout the year in intermittent reaches. While we did not measure particulate organic matter retention, increased intermittency was previously correlated with increased coarse particulate organic matter retention (Dewson et al., 2007b) and reduced leaf litter breakdown (Datry et al., 2011 and references within). Our results contrast with other studies that assessed shredder density in relation to flow permanence. Lower shredder density with decreased FD has been attributed to sensitivity of shredders to low dissolved oxygen concentrations and other stressful abiotic conditions associated with stream drying (Datry et al., 2011; Mariluan et al., 2015; Mora-Gómez et al., 2015). Shredder density has also been equal in perennial and intermittent reaches (Bogan et al., 2013). Our study system had relatively short periods of drying (3–5 months) compared to other studies (ca. 9-months), and sensitive shredders were likely not excluded due to harsh abiotic conditions in these intermittent reaches. As a result of relatively short periods of drying, many shredders could likely complete their lifecycle during the connected flow phase. Confounding factors such as position in the watershed, for example low-order versus high-order reaches, must also be considered since headwater streams generally have more shredders than high-order streams (Vannote et al., 1980). We believe that shorter FD was a major factor influencing shredder density because 30% of shredders in all intermittent reaches were sclerotized, non-insect taxa tolerant to low dissolved oxygen concentrations. Future studies are needed to untangle the effects of network position, flow duration, and organic matter retention on shredder density and diversity.
It is important to note macroinvertebrate biomass differentiated communities depending on FD to a greater degree than density. This could have been due to smaller average individual size of taxa in intermittent reaches because of short recruitment periods or due to more large-bodied taxa in perennial reaches. The latter is likely a factor considering taxa indicative of perennial reaches by biomass included large-bodied predators such as Argia, Stylogomphus, and Neoperla. Differences in density and biomass ISA reveal those taxa that were abundant in communities versus those that played greater roles in resource consumption and storage. Biomass versus density measurements can help elucidate differences in resource use since biomass measures macroinvertebrate mass per area, thus indicating how potential energy is stored at the community scale for a point in time (Grubaugh et al., 1996; O’Gorman et al., 2008). Secondary production was not measured so we cannot infer differences in the total energy flow among reaches, but we can identify taxa that maintained large standing stocks of biomass within communities that differed by FD. Nigronia, Ceratopogonidae, and Tanypodinae were indicative of reaches with 9-month FD by biomass but not density, suggesting there were enough resources or habitat to support these predators in reaches with 9-month flow duration, but not 7-month FD.
This study can inform conservation and restoration activities that aim to improve or maintain connectivity across intermittent and perennial aquatic habitats to increase local biodiversity. Twenty years ago, interest and research in restoring hydrologic and ecological habitat connectivity increased dramatically (Merenlender & Matella, 2013). Restorations that attempt to improve biodiversity by altering local habitat can sometimes improve richness, but not biotic diversity (Parkyn & Smith, 2011). Failure to increase diversity is generally attributed to restorations at inappropriately small spatial scales, and restorations that focus primarily on increasing flow without consideration of the timing and spatial distribution needed to provide refuge at critical time periods (Lake et al., 2007). In addition, restoration of connected flow when streams would normally be dry can be counterproductive if native species are specifically adapted to fragmented habitat during low-flow conditions (Merenlender & Matella, 2013). If more streams become intermittent in the future, there may be a loss in flow regime diversity at the regional scale, which could compromise regional biodiversity. While this was a short study and long-term patterns were hard to infer, we found taxa unique to intermittent reaches and associated with hyporheic habitat, suggesting that the loss of hyporheic habitat and more intense stream drying will result in local loss of taxa.
References
Arscott, D. B., S. Larned, M. R. Scarsbrook & P. Lambert, 2010. Aquatic invertebrate community structure along an intermittence gradient: Selwyn River, New Zealand. Journal of the North American Benthological Society 29: 530–545.
Austin, B. J., N. Hardgrave, E. Inlander, C. Gallipeau, S. Entrekin & M. A. Evans-White, 2015. Stream primary producers relate positively to watershed natural gas measures in north-central Arkansas streams. Science of the Total Environment 529: 54–64.
Beche, L. A., E. P. Mcelravy & V. H. Resh, 2006. Long term seasonal variation in the biological traits of benthic macroinvertebrates in two Mediterranean climate streams in California, USA. Freshwater Biology 51: 56–75.
Beche, L. A. & V. H. Resh, 2007. Short-term climatic trends affect the temporal variability of macroinvertebrates in California ‘Mediterranean’ streams. Freshwater Biology 52: 2317–2339.
Benjamini, Y. & Y. Hochberg, 1995. Controlling the false discovery rate: a practical and powerful approach to multiple testing. Journal of the Royal Statistical Society Series B (Methodological) 57: 289–300.
Benke, A. C., A. D. Huryn, L. A. Smock & J. B. Wallace, 1999. Length-mass relationships for freshwater macroinvertebrates in North America with particular reference to the southeastern United States. Journal of the North American Benthological Society 18: 308–343.
Birdsong, B., 2011. Wildlife Management Area Details. http://www.agfc.com.
Bogan, M. T., K. S. Boersma & D. A. Lytle, 2013. Flow intermittency alters longitudinal patterns of invertebrate diversity and assemblage composition in an arid-land stream network. Freshwater Biology 58: 1016–1028.
Bogan, M. T., K. S. Boersma & D. A. Lytle, 2015. Resistance and resilience of invertebrate communities to seasonal and supraseasonal drought in arid-land headwater streams. Freshwater Biology 60: 2547–2558.
Bonada, N., M. Rieradevall & N. Prat, 2007. Macroinvertebrate community structure and biological traits related to flow permanence in a Mediterranean river network. Hydrobiologia 589: 91–106.
Bonada, N., M. Rieradevall, N. Prat & V. H. Resh, 2006. Benthic macroinvertebrate assemblages and macrohabitat connectivity in Mediterranean-climate streams of northern California. Journal of the North American Benthological Society 25: 32–43.
Boulton, A. J., 2003. Parallels and contrasts in the effects of drought on stream macroinvertebrate assemblages. Freshwater Biology 48: 1173–1185.
Boulton, A. & P. Lake, 1992. The ecology of two intermittent streams in Victoria, Australia. III. Temporal changes in faunal composition. Freshwater Biology 27: 123–138.
Boulton, A. & P. Lake, 2008. Effects of drought on stream insects and its ecological consequences. Proceedings of the Royal Entomological Society’s 24th Symposium. CABI Publishing: 81–101.
Brooks, A. J., B. C. Chessman & T. Haeusler, 2011. Macroinvertebrate traits distinguish unregulated rivers subject to water abstraction. Journal of the North American Benthological Society 30: 419–435.
Burnham, K. P. & D. Anderson, 2002. Model selection and multi-model inference: a practical information-theoretic approach. Springer, New York.
Cañedo-Argüelles, M., K. S. Boersma, M. T. Bogan, J. D. Olden, I. Phillipsen, T. A. Schriever & D. A. Lytle, 2015. Dispersal strength determines meta-community structure in a dendritic riverine network. Journal of Biogeography 42: 778–790.
Chadwick, M. A. & A. D. Huyrn, 2007. Role of habitat in determining macroinvertebrate production in an intermittent stream system. Freshwater Biology 52: 240–251.
Chester, E. & B. Robson, 2011. Drought refuges, spatial scale and recolonisation by invertebrates in non-perennial streams. Freshwater Biology 56: 2094–2104.
Chester, E. T., A. D. Miller, I. Valenzuela, S. J. Wickson & B. J. Robson, 2015. Drought survival strategies, dispersal potential and persistence of invertebrate species in an intermittent stream landscape. Freshwater Biology 60: 2066–2083.
Datry, T., 2012. Benthic and hyporheic invertebrate assemblages along a flow intermittence gradient: effects of duration of dry events. Freshwater Biology 57: 563–574.
Datry, T., R. Corti, C. Claret & M. Philippe, 2011. Leaf litter decomposition along a gradient of flow permanence in a French temporary river: the memory of drying. Aquatic Sciences 73: 471–483.
Datry, T., S. Larned, K. Fritz, M. Bogan, P. J. Wood, E. Meyer & A. Santos, 2014a. Broad-scale patterns of invertebrate richness and community composition in temporary rivers: effects of flow intermittence. Ecography 37: 94–104.
Datry, T., S. T. Larned & K. Tockner, 2014b. Intermittent rivers: a challenge for freshwater ecology. Bioscience 64: 229–235.
del Rosario, R. B. & V. H. Resh, 2000. Invertebrates in intermittent and perennial streams: is the hyporheic zone a refuge from drying? Journal of the North American Benthological Society 19: 680–696.
Dewson, Z. S., A. B. James & R. G. Death, 2007a. A review of the consequences of decreased flow for instream habitat and macroinvertebrates. Journal of the North American Benthological Society 26: 401–415.
Dewson, Z. S., A. B. W. James & R. G. Death, 2007b. Stream ecosystem functioning under reduced flow conditions. Ecological Applications 17: 1797–1808.
Dodds, W. K., K. Gido, M. R. Whiles, M. D. Daniels & B. P. Grudzinski, 2015. The stream biome gradient concept: factors controlling lotic systems across broad biogeographic scales. Freshwater Science 34: 1–19.
Döll, P. & H. M. Schmied, 2012. How is the impact of climate change on river flow regimes related to the impact on mean annual runoff? A global-scale analysis. Environmental Research Letters 7: 014037.
Feminella, J. W., 1996. Comparison of benthic macroinvertebrate assemblages in small streams along a gradient of flow permanence. Journal of the North American Benthological Society 15: 651–669.
Fritz, K. M. & W. K. Dodds, 2004. Resistance and resilience of macroinvertebrate assemblages to drying and flood in a tallgrass prairie stream system. Hydrobiologia 527: 99–112.
Fritz, K. M. & W. K. Dodds, 2005. Harshness: characterisation of intermittent stream habitat over space and time. Marine and Freshwater Research 56: 13–23.
Gibert, J., J. A. Stanford, M.-J. Dole-Olivier & J. V. Ward, 1994. Basic attributes of groundwater ecosystems and prospects for research. In Gibert, J., D. L. Danielopol & J. A. Stanford (eds.), Groundwater Ecology. Academic Press, San Diego: 7–40.
Gordon, N. D., T. A. McMahon, B. L. Finlayson, C. J. Gippel & R. J. Nathan, 2004. Stream Hydrology: An Introduction for Ecologists. Wiley, Chichester.
Grubaugh, J., J. Wallace & E. Houston, 1996. Longitudinal changes of macroinvertebrate communities along an Appalachian stream continuum. Canadian Journal of Fisheries and Aquatic Sciences 53: 896–909.
Grimm, N. B. & S. G. Fisher, 1989. Stability of periphyton and macroinvertebrates to disturbance by flash floods in a desert stream. Journal of the North American Benthological Society 8: 293–307.
Herr, D. G., 1986. On the history of ANOVA in unbalanced, factorial designs: the first 30 years. The American Statistician 40: 265–270.
Huryn, A. D. & J. B. Wallace, 2000. Life history and production of stream insects. Annual Review of Entomology 45: 83–110.
Lake, P., 2003. Ecological effects of perturbation by drought in flowing waters. Freshwater Biology 48: 1161–1172.
Lake, P., N. Bond & P. Reich, 2007. Linking ecological theory with stream restoration. Freshwater Biology 52: 597–615.
Larned, S. T., T. Datry, D. B. Arscott & K. Tockner, 2010. Emerging concepts in temporary river ecology. Freshwater Biology 55: 717–738.
Leasure, D., D. D. Magoulick & S. Longing, 2014. Natural flow regimes of the Ozark-Ouachita interior highlands region. River Research and Applications 32: 18–35.
Leigh, C., 2013. Dry-season changes in macroinvertebrate assemblages of highly seasonal rivers: responses to low flow, no flow and antecedent hydrology. Hydrobiologia 703: 95–112.
Leigh, C., A. J. Boulton, J. L. Courtwright, K. Fritz, C. L. May, R. H. Walker & T. Datry, 2015. Ecological research and management of intermittent rivers: an historical review and future directions. Freshwater Biology 61: 1181–1199.
Leigh, C., N. Bonada, A. J. Boulton, B. Hugueny, S. T. Larned, R. Vander Vorste & T. Datry, 2016. Invertebrate assemblage responses and the dual roles of resistance and resilience to drying in intermittent rivers. Aquatic Sciences 78: 291–301.
Lytle, D. A., 2001. Disturbance regimes and life-history evolution. The American Naturalist 157: 525–536.
Mariluan, G. D., V. Díaz Villanueva & R. J. Albariño, 2015. Leaf litter breakdown and benthic invertebrate colonization affected by seasonal drought in headwater lotic systems of Andean Patagonia. Hydrobiologia 760: 171–187.
McCune, B. & J. B. Grace, 2002. Analysis of ecological communities, Vol. 28. MjM Software Design, Gleneden Beach, Oregon.
Merenlender, A. M. & M. K. Matella, 2013. Maintaining and restoring hydrologic habitat connectivity in mediterranean streams: an integrated modeling framework. Hydrobiologia 719: 509–525.
Merritt, R., K. Cummins & M. Berg, 2008. An Introduction to the Aquatic Insects of North America. Kendall Hunt, Dubuque.
Miller, A. M. & S. W. Golladay, 1996. Effects of spates and drying on macroinvertebrate assemblages of an intermittent and a perennial prairie stream. Journal of the North American Benthological Society 15: 670–689.
Mora-Gómez, J., A. Elosegi, E. Mas-Martí & A. M. Romaní, 2015. Factors controlling seasonality in leaf-litter breakdown in a Mediterranean stream. Freshwater Science 34: 1245–1258.
Muñoz, I., 2003. Macroinvertebrate community structure in an intermittent and a permanent Mediterranean streams (NE Spain). Limnetica 22: 107–116.
O’Gorman, E. J., R. A. Enright & M. C. Emmerson, 2008. Predator diversity enhances secondary production and decreases the likelihood of trophic cascades. Oecologia 158: 557–567.
Parkyn, S. M. & B. J. Smith, 2011. Dispersal constraints for stream invertebrates: setting realistic timescales for biodiversity restoration. Environmental Management 48: 602–614.
Pastor, A., F. Ludwig, H. Biemans, H. Hoff & P. Kabat, 2013. Accounting for environmental flow requirements in global water assessments. Hydrology and Earth System Sciences Discussions 10: 14987–15032.
Pennak, R. W., 1989. Freshwater invertebrates of the United States: Porifera to Crustacea, 3rd ed. Wiley, New York.
Phillipsen, I. C. & D. A. Lytle, 2013. Aquatic insects in a sea of desert: population genetic structure is shaped by limited dispersal in a naturally fragmented landscape. Ecography 36: 731–743.
Pinheiro, J., D. Bates, S. DebRoy & D. Sarkar, 2016. nlme: linear and nonlinear mixed effects models. http://CRAN.R-project.org/package=nlme.
Poff, N. L. R., 1997. Landscape filters and species traits: towards mechanistic understanding and prediction in stream ecology. Journal of the North American Benthological Society 16: 391–409.
Poff, N. L. & J. V. Ward, 1989. Implications of streamflow variability and predictability for lotic community structure: a regional analysis of streamflow patterns. Canadian Journal of Fisheries and Aquatic Sciences 46: 1805–1818.
Poff, N. L. R., J. D. Olden, N. K. M. Vieira, D. S. Finn, M. P. Simmons & B. C. Kondratieff, 2006. Functional trait niches of North American lotic insects: traits-based ecological applications in light of phylogenetic relationships. Journal of the North American Benthological Society 25: 730–755.
Price, K., A. Suski, J. McGarvie, B. Beasley & J. S. Richardson, 2003. Communities of aquatic insects of old-growth and clearcut coastal headwater streams of varying flow persistence. Canadian Journal of Forest Research 33: 1416–1432.
Pringle, C., 2003. What is hydrologic connectivity and why is it ecologically important? Hydrological Processes 17: 2685–2689.
R Development Core Team, 2009. R: A language and environment for statistical computing. R foundation for statistical computing, Vienna.
Robson, B., E. Chester & C. Austin, 2011. Why life history information matters: drought refuges and macroinvertebrate persistence in non-perennial streams subject to a drier climate. Marine and Freshwater Research 62: 801–810.
Rohlf, F. J. & R. R. Sokal, 1994. Statistical Tables. WH Freeman and Company, New York.
Rolls, R. J., C. Leigh & F. Sheldon, 2012. Mechanistic effects of low-flow hydrology on riverine ecosystems: ecological principles and consequences of alteration. Freshwater Science 31: 1163–1186.
Schneider, D. W. & T. M. Frost, 1996. Habitat duration and community structure in temporary ponds. Journal of the North American Benthological Society 15: 64–86.
Smakhtin, V., 2001. Low flow hydrology: a review. Journal of Hydrology 240: 147–186.
Southwood, T., 1977. Habitat, the templet for ecological strategies? Journal of Animal Ecology 46: 337–365.
Stanley, E. H., D. L. Buschman, A. J. Boulton, N. B. Grimm & S. Fisher, 1994. Invertebrate resistance and resilience to intermittency in a desert stream. American Midland Naturalist 131: 288–300.
Stanley, E. H., S. G. Fisher & N. B. Grimm, 1997. Ecosystem expansion and contraction in streams. BioScience 47: 427–435.
Stevenson, R. J. & S. Sabater, 2010. Understanding effects of global change on river ecosystems: science to support policy in a changing world. Hydrobiologia 657: 3–18.
Steward, A. L., D. von Schiller, K. Tockner, J. C. Marshall & S. E. Bunn, 2012. When the river runs dry: human and ecological values of dry riverbeds. Frontiers in Ecology and the Environment 10: 202–209.
Stewart, K. W. & B. P. Stark, 1988. Nymphs of North American Stonefly Genera (Plecoptera). Entomological Society of America, Lanham.
Strachan, S. R., E. T. Chester, & B. J. Robson, 2015. Freshwater invertebrate life history strategies for surviving desiccation. Spring Science Reviews 3: 57–75.
Stubbington, R., 2012. The hyporheic zone as an invertebrate refuge: a review of variability in space, time, taxa and behaviour. Marine and Freshwater Research 63: 293–311.
Stubbington, R. & T. Datry, 2013. The macroinvertebrate seedbank promotes community persistence in temporary rivers across climate zones. Freshwater Biology 58: 1202–1220.
Stubbington, R., J. Gunn, S. Little, T. P. Worrall & P. J. Wood, 2016. Macroinvertebrate seedbank composition in relation to antecedent duration of drying and multiple wet-dry cycles in a temporary stream. Freshwater Biology 61: 1293–1307.
Vander Vorste, R., A. Sagouis Corti & T. Datry, 2016. Invertebrate communities in gravel-bed, braided rivers are highly resilient to flow intermittence. Freshwater Science 35: 164–177.
Vannote, R. L., G. W. Minshall, K. W. Cummins, J. R. Sedell & C. E. Cushing, 1980. The river continuum concept. Canadian Journal of Fisheries and Aquatic Sciences 37: 130–137.
Verhoeven, K. J., K. L. Simonsen & L. M. McIntyre, 2005. Implementing false discovery rate control: increasing your power. Oikos 108: 643–647.
Vogel, R. M. & N. M. Fennessey, 1995. Flow duration curves II: a review of applications in water resources planning. Journal of American Water Resources Association 37: 1029–1039.
Walters, A. W., 2011. Resistance of aquatic insects to a low-flow disturbance: exploring a trait-based approach. Journal of the North American Benthological Society 30: 346–356.
Walters, A. W. & D. M. Post, 2011. How low can you go? Impacts of a low-flow disturbance on aquatic insect communities. Ecological Applications 21: 163–174.
Waters, T. F., 1969. Subsampler for dividing large samples of stream invertebrate drift. Limnology and Oceanography 14: 813–815.
Wentworth, C. K., 1922. A scale of grade and class terms for clastic sediments. The Journal of Geology 30: 377–392.
Wiggins, G. B., 1996. Larvae of North America caddisfly genera (Trichoptera). University of Toronto Press, Toronto.
Wiggins, G. B., R. J. Mackay & I. M. Smith, 1980. Evolutionary and ecological strategies of animals in annual temporary pools. Archiv für Hydrobiologie Supplement 58: 97–206.
Zuellig, R. E. & T. S. Schmidt, 2012. Characterizing invertebrate traits in wadeable streams of the contiguous US: differences among ecoregions and land uses. Freshwater Science 31: 1042–1056.
Acknowledgements
We thank Michelle Evans-White, Reid Adams, Hal Halvorson, and Richard Walker for their input and review of the manuscript. We thank Amanda Bates, Chris Fuller, Chelsea Miller, and Adam Musto for help in the field and laboratory. This work was funded by State Wildlife grant number T33-01 from the Arkansas Game and Fish Commission.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: Checo Colón-Gaud
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kelso, J.E., Entrekin, S.A. Intermittent and perennial macroinvertebrate communities had similar richness but differed in species trait composition depending on flow duration. Hydrobiologia 807, 189–206 (2018). https://doi.org/10.1007/s10750-017-3393-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-017-3393-y