Abstract
Lodging is one of the most important factors that reduce wheat grain yields. To improve lodging resistance, increasing the strength of wheat stems has been an important breeding objective. To assess the rate of genetic improvement of the character, 30 wheat accessions, varying from landraces to cultivars that have been widely grown in Henan Province, China during the last 70 years, were tested for several parameters of lodging resistance in a two-year experiment. Stem strength, morphological and anatomical traits, and chemical composition were measured. The results showed that stem strength, both at anthesis and 25 days thereafter, has increased significantly during this period with average annual genetic gains of 1.07 and 0.87 %, respectively. Compared with older wheat cultivars, stem walls of modern cultivars are thicker, and the lignin content is higher. Grain yield has been significantly increased over the last 70 years, and the results of multiple regression analysis indicated that the genetic improvement of grain yield was associated with the increase in stem strength and the decrease in plant height.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Wheat (Triticum aestivum L.) is one of the most important crops in the world. In 2013, global production amounted to 716 million tons harvested from 219 million hectares of land. The yield of wheat has shown a dramatic increase from 1.1 t ha−1 in 1961 to 3.2 t ha−1 in 2013 (FAO, http://faostat.fao.org/). Lodging is one of the most important factors that limits continued increase of wheat yields, as it not only destroys the canopy structure of plants thereby reducing photosynthetic capacity and photosynthate production, but also hinders the transportation of water and photosynthate, which influences grain filling (Berry et al. 2004; Acreche and Slafer 2011; Berry et al. 2015). Simultaneously, grain quality can be affected by lodging, which is associated with the reduction of Hagberg falling number, thousand grain weight and specific weight (Berry et al. 2004). In addition, lodging complicates mechanized harvesting. Research has shown that serious lodging can decrease wheat yield by 31–80 % (Easson et al. 1993; Berry and Spink 2012).
Lodging resistance is a major wheat breeding objective. Introduction of dwarfing genes (Rht1, Rht2, and Rht8) since the 1960s led to distinct decreases in plant height, and that resulted in a significant increase in lodging resistance and grain yield (Gooding and Davies 1997). However, if the plant height is too low, not only is the aboveground biomass decreased, but the microclimate of the plant population is degraded and leaf disease is aggravated, which results in a diminished output. Studies have shown that the height of modern wheat cultivars is close to the optimal height (Flintham et al. 1997; Sherman et al. 2014). Although the reduction of plant height has enhanced lodging resistance significantly, lodging still occurs in modern wheat cultivars grown under conditions of high soil fertility and grain yield. Therefore, genetic improvement of lodging resistance remains an important wheat breeding target.
Lodging can be of two types, namely root lodging and stem lodging. Root lodging generally occurs in poor soils or can be due to dysplastic root development (Baker et al. 1998), while stem lodging means that the stem is bent or broken by external force (Neenan and Spencer-Smith 1975). In practice, root lodging and stem lodging occur separately or simultaneously depending on differing cultivar characteristics and growing environments (Berry et al. 2003). Stem lodging, which is associated with plant height and stem strength, tends to occur in the first and second internodes of the stem base, especially the second internode (Kelbert et al. 2004; Peng et al. 2014). Chuanren et al. (2004) demonstrated that stem strength has significant correlation with stem diameter, wall thickness and the number of vascular bundles. Thicker stem wall and increased dry weight of the internodes were also associated with improved stem strength (Zuber et al. 1999; Islam et al. 2007; Feng et al. 2012; Kong et al. 2013). As main components of the cell wall, lignin and cellulose contents are also closely related to the stem strength (Esechie et al. 2004; Wang et al. 2006; Kong et al. 2013; Peng et al. 2014).
Henan Province is the largest wheat producing area in China, with a planted area of 5.32 million ha (Wu et al. 2014). Yield losses due to lodging are common and breeding for lodging resistance has been on-going for many years. This study sampled wheat varieties released in Henan Province since the 1940s to the present in order to compare their stem strength, morphological and anatomical traits, and chemical composition. Measurements were made at anthesis and 25 days after anthesis (25 DAA) using the second basal internode of the sampled cultivars. The aim of the study was to assess the rate of genetic improvement of stem strength and related traits during this period and to analyse the associations among traits. The results were expected to further understanding of the factors contributing to lodging resistance and to aid future strategies for its genetic improvement in China and elsewhere.
Materials and methods
Plant material and experimental design
Based on the maximum area on which a cultivar was planted annually, 30 of the most widely grow landraces and cultivars produced during the last 70 years in Henan Province, China, were selected (Table 1). During the 2013/2014 and 2014/2015 crop seasons, a field experiment was conducted at the Henan Research and Development Base for Modern Agriculture (Yuanyang, Henan, China; 35°00′N, 113°40′E, 77 m a.s.l.). The average annual temperature of the experimental area was 14.4°C and the rainfall was 556 mm. The soil type of the experimental field was highly organic and slightly alkaline sandy clay. Urea [ca. 13 kg ha−1 of nitrogen (N)], ammonium phosphate [ca. 170 kg ha−1 of phosphate (P), 67 kg ha−1 of N] and potassium chloride [ca. 75 kg ha−1 of potassium (K)] were applied prior to planting, and then an additional 120 kg ha−1 of N was top-dressed at stem elongation (GS31) (Zadoks et al. 1974). The trial was arranged in a completely randomized block design with six replications, of which three replications were used to determine the grain yield (yield plots) and the rest three replications were used for sample collection to evaluate other traits (sampling plots). Each plot consisted of six rows 4.25 m in long with a row width of 0.23 m, giving a total plot size of 6.67 m2. Seed was planted on 6 October in 2013 and on 7 October in 2014 at a seeding rate of 180 kernels m−2 using a plot row planter. Following local practices, furrow irrigations (60 mm each) were provided at the tillering (before winter) (GS25), stem elongation (GS31) and anthesis (GS60) stages. Fungicides and pesticides were applied at stem elongation (GS33) and grain filling (GS73) stages to prevent attack by diseases and pests. Because tall plants of the old landraces or cultivars are prone to lodging, bamboo sticks (2–3 m long) were used to prevent lodging so that maximum yield potential could be reached.
Plant sampling and determinations made
Plant sampling
The second basal internode of the stem was sampled at anthesis (GS64) and 25 DAA (GS77) in both the 2013/2014 and 2014/2015 seasons. Twenty samples (the whole second basal internode without node) randomly taken from each sampling plot were divided into four sub-samples (five stem cutting in each) and used for the determination of stem strength, morphological and anatomical traits, lignin content, and cellulose content.
Determination of stem strength
Stem strength was measured using the method of Kokubo et al. (1989) as described by Peng et al. (2014), with a plant lodging tester (Zhejiang TOP Instrument Co, YYD-1A, Hangzhou, China). Each internode, without the stem sheath, was put on the groove of the support pillars at a distance of 50 mm, respectively. The tester was set perpendicular to the internode at the middle, loading was increased gradually, and stem strength in bending was measured when the internode was pushed to breaking. The value displayed, representing the largest acting force, was the stem strength in Newtons.
Determination of morphological and anatomical traits of the stem
The morphological and anatomical traits of the stem were studied using the methodology of Wang et al. (2006). Sections were cut at the centre of each internode with a freezing microtome (Leica, CM1950, Wetzlar, Germany) and stained with phloroglucinol-HCl. The number of vascular bundles was determined using a microscope (Olympus, BX53, Tokyo, Japan). The stem diameter and stem wall thickness were measured using a statistical software package attached to the microscope.
Determination of the chemical composition of the stem
The internodes were ground to a fine powder in liquid nitrogen using a mortar and pestle, and 200 mg and 20 mg fine powder was collected to assay the lignin content and cellulose content of the stem using the methods described by Kirk and Obst (1988) and Updegraff (1969), respectively.
Determination of plant height and grain yield
Plant height was measured at 5 days after anthesis (GS71) from the soil surface to the top of the spike (excluding awns), and each plot was measured in three different positions and the mean was calculated. At the physiological maturity (GS92), each yield plot was hand-harvested, threshed, dried, and weighed to determine the grain yield at 13 % moisture content.
Statistical analysis
Analysis of variance (ANOVA) was done for the combined data over 3 years. Stem strength and related traits were analysed at anthesis and 25 DAA, respectively. The significance of each source of variation was determined by an F-test. The ANOVA, correlation analysis, and multiple linear regressions were calculated using the software Statistical Product and Service Solutions (SPSS Inc., Chicago, IL, USA).
The absolute (strength gains in Newtons per single-stalk per year) or exponential (the percentage strength gain per year) genetic gains of the stem strength and related traits were estimated using the respective equations:
where y i is the mean stem strength of cultivar i, ln(y i ) is the natural logarithm of y i , and x i is the year of release for cultivar i. The intercept of both equations was estimated by a, while the slope b measured absolute or exponential stem strength gains; the latter was converted to a percentage. The residual error was estimated by u (Ortiz-Monasterio et al. 1997).
Results
The ANOVA of the stem strength and related traits
The ANOVA results (Table 2) showed that there was no significant (P > 0.05) difference between replications for all of the traits while the genotypic differences were significant (P < 0.01) for all. The year effect was significant for most of the traits except stem strength and lignin content at anthesis, and a similar pattern was seen for the genotype × year interaction. The ANOVA indicated that the results of the experiment were reliable, that differences among genotypes were significant and that the environment had a strong effect on most of the traits.
Changes in the stem strength
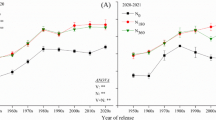
Stem strength at anthesis increased significantly over time, ranging from 1.89 Newtons for Florence that was introduced in 1951 to 5.24 Newtons for Zhoumai 22 released in 2007 (Fig. 1). On average, stem strength at anthesis had increased yearly by 1.07 % (R 2 = 0.68, P < 0.01) or 0.034 Newtons (R 2 = 0.64, P < 0.01) over the last 70 years. Stem strength at 25 DAA also increased significantly, and the average annual genetic gain was 0.87 % (R 2 = 0.71, P < 0.01). Stem strength at 25 DAA was reduced as compared to stem strength at anthesis; this effect was more pronounced for cultivars developed in the 21st century, which showed a reduction of 1.96 Newtons (42.7 %) (Fig. 2).
Changes in morphological and anatomical traits of the stem
Stem diameter at anthesis and 25 DAA increased significantly over the last 70 years and ranged from 2.65 and 1.79 mm for the landrace Hongheshangtou (1941) to 4.34 mm for Yumai 49 released in 1998 at anthesis and 3.66 mm for Zhengzhou 761 released in 1976 at 25 DAA, respectively. The average annual genetic gains in stem diameter were 0.37 % (R 2 = 0.45, P < 0.05) and 0.57 % (R 2 = 0.59, P < 0.01), respectively (Fig. 3a). Similar to the stem diameter, stem wall thickness at the two growth stages also increased significantly and the average annual genetic gains were 0.82 % (R 2 = 0.68, P < 0.01) and 0.58 % (R 2 = 0.51, P < 0.01), respectively (Fig. 3b).
No significant changes were observed in the average number of large and small vascular bundles for either growth stage (P > 0.05) (Fig. 3c, d), meaning that the numbers were either stable or changed only slightly.
Changes in lignin content and cellulose content of the stem
The results showed that the average stem lignin content increased significantly with the years of cultivars release, both at anthesis and 25 DAA (Fig. 4a). Lignin contents at anthesis ranged from 105.7 mg g−1 for the landrace Hulutou to 247.0 mg g−1 for Zhoumai 22 released in 2007, and the average annual genetic gain was 1.14 % (R 2 = 0.90, P < 0.01). The lignin content at 25 DAA was significantly higher than at anthesis, and the average annual genetic gain was 0.71 % (R 2 = 0.86, P < 0.01).
The cellulose content of the stem at anthesis also increased significantly with the years of cultivars release, while there was no significant variance in the cellulose content of the stem at 25 DAA (Fig. 4b). The cellulose content of the stem at anthesis ranged from 168.5 mg g−1 for Pingyuan 50 released in 1951 to 236.9 mg g−1 for Zhoumai 22 released in 2007, and the average annual genetic gain was 0.82 % (R 2 = 0.80, P < 0.01).
Relationship between stem strength and related traits
Associations among the stem strength and related traits are shown in Table 3. At anthesis, stem strength was significantly and positively correlated with stem diameter, wall thickness, number of large vascular bundles, lignin content and cellulose content, but not with the number of small vascular bundles. The values of the coefficients of determination showed that lignin content and stem wall thickness had the most relevance to stem strength. At 25 DAA, stem strength was significantly associated with all of the traits, and the lignin content, stem diameter and wall thickness had the strongest correlations with stem strength. The results of the correlation analysis therefore suggested that stem wall thickness and lignin content had the greatest effect on the stem strength, and improving these two traits should significantly increase stem strength.
Multivariate regression analysis among grain yield, plant height and the stem strength
The grain yields ranged between 4.48 t ha−1 (landrace Hulutou) and 9.49 t ha−1 (Zhengmai 7698 released in 2011), and the average annual genetic gain was calculated to be 1.03 % (R 2 = 0.90, P < 0.01) or 0.069 t ha−1 (R 2 = 0.92, P < 0.01) over the last 70 years (Fig. 5a). While the grain yield increased, plant height clearly decreased over the same period (Fig. 5b). Plant height average ranged from 1.37 m (landrace Dakoumai) to 0.67 m (Aikang 58 released in 2005), with the average annual genetic gain of −0.96 % (R 2 = 0.84, P < 0.01).
Multiple regression equations of yield on plant height (x 1) and stem strength (x 2) at anthesis and 25 DAA were derived. For measurements made at anthesis, the multiple regression equation was: \(y = \, - 0. 6 7 8x_{ 1}\,+\, 0. 3 4 4x_{ 2} \;\left( {R^{ 2} \,=\,0. 8 9,F\,=\,{ 1}0 4. 5 5 { } > F_{0.0 1} } \right)\). For measurements at 25 DAA the regression equation was: \(y = \, - 0. 5 5 9x_{ 1} + \, 0. 4 6 8x_{ 2} \;\left( {R^{ 2} \,= \, 0. 9 2,F = { 15}0. 6 5 { } > F_{0.0 1} } \right)\).
The equations showed that plant height had a strong negative association with grain yield, while stem strength had a smaller but positive association. Thus, since the 1940s, the reduction of plant height had a greater effect on raising yields than the improvement of stem strength. However, the height of modern wheat cultivars is probably close to the optimal height and it is unlikely that continued changes in plant height will achieve further yield improvement. Instead, more attention should now be paid to the improvement of stem strength and lodging resistance so that grain yield can be enhanced further.
Discussion
Lodging is a complicated phenomenon that is influenced by many factors including rain, wind, soil type, cropping system, disease and so on. It is one of the most common factors limiting grain yields, and lodging resistance has always been an important consideration in wheat breeding. There are two patterns of lodging resistance that are resistance to stem failure and anchorage failure. Resistance to stem failure is closely related to plant height and stem strength (Berry et al. 2004). In the process of wheat genetic improvement, breeders have focused for a long time on breeding for reduced height using dwarfing genes, and have made great progress in raising wheat yields worldwide (He et al. 2001; Mathews et al. 2006). The current study suggests that in Henan Province breeding for reduced height similarly led to considerable yield improvement over the last seven decades. In addition, a parallel improvement in stem strength occurred since the 1940s which also added to the higher productivity of modern cultivars. The selection of wheat cultivars with greater lodging resistance was the primary reason for improvement stem strength, which was facilitated the selection of other yield enhancing traits as well, such as large spikes that can accompany thicker stalks and higher fertilizer application. Lodging resistance remains one of the major objectives of wheat breeding in the future, on account of the increasing grain yield. However, it is unlikely that further yield improvement will be achieved through height reduction as the height of modern wheat cultivars is probably close to optimal (Horton 2000). Therefore, the genetic improvement of stem strength, specifically, has become more important.
The current study showed that compared with older wheat cultivars, the stem walls of modern cultivars are thicker, and the lignin content is higher. There are several studies have suggested that stem strength is closely associated with stem diameter, wall thickness and the number of vascular bundles (Jezowski 1981; Chuanren et al. 2004; Wang et al. 2006). In addition, it is also closely linked to the lignin content of the stem; in other words, increasing the lignin content of the stem can improve the stem mechanical strength significantly, which has a great effect on stem strength and lodging resistance (Esechie et al. 2004; Kong et al. 2013; Peng et al. 2014). The results of this study are consistent with these earlier findings. Lignin is difficult to be degraded because of its stable chemical structure, and this is one of the most important factors limiting the breakdown of the straw residue in the field (Bugg et al. 2011). The increased lignin content of stem could lead to slower breakdown of the straw, and that could be the reason of the straw residue breakdown become more difficult under minimal or no tillage. This issue, however, has been realized by culturist, and the cultivation technique of deep ploughing taken every 2 or 3 years were applied in China.
Future breeding for lodging resistance should continue to select for plants with greater stem strength, thicker stem wall and higher lignin content in stem tissue. However, there is little method to evaluate the stem strength and related traits in the large-scale field selection, and it should be explored in the future. Quantitative trait loci (QTL) associated with stem strength and related traits have been discovered in recent years (Hai et al. 2005; Ma et al. 2011; Ma and Liu 2014; Berry and Berry 2015), paving the way for marker-aided selection of increased stem strength, stem lignin content and thicker stem walls.
Lodging occurred during grain filling when the ears were heaviest and when the soil was wet (Crook and Ennos 1994). In addition, the competing for assimilate between lower internodes and grains was also could increase the risk of lodging at the grain filling stage. The current results showed that the stem strength of modern wheat cultivars is significantly reduced at 25 DAA compared with anthesis. This could be related to a large amount of photosynthate being shifted from the stem to the spike (Berry et al. 2007; Mu et al. 2010; Foulkes et al. 2011). This result suggests that if we want to maintain the stem strength at 25 DAA, the stem photosynthate should not be excessively transferred; otherwise it will seriously decrease lodging resistance in the late stage of grain filling. Therefore, improving the photosynthetic capacity of plant at the grain filling stage is not only a way to improve the grain yield, but also an important way to sustain lodging resistance. There are three approaches to improve the photosynthetic capacity at the grain filling stage: (1) improving the post-anthesis photosynthetic efficiency by crosses with the cultivars with high post-anthesis photosynthetic efficiency; (2) delaying leaf senescence to extend the duration of active photosynthesis at the grain filling stage; (3) improving wheat photosynthetic efficiency under strong light radiation and high temperature at the grain filling stage, when the dry hot wind occurs frequently.
References
Acreche MM, Slafer GA (2011) Lodging yield penalties as affected by breeding in Mediterranean wheats. Field Crops Res 122:40–48
Baker CJ, Spink PM, Sylvester-Bradley R, Griffin JM, Scott RK, Clare RW (1998) A method for the assessment of the risk of wheat lodging. J Theor Biol 194:587–603
Berry PM, Berry ST (2015) Understanding the genetic control of lodging-associated plant characters in winter wheat (Triticum aestivum L.). Euphytica 205:671–689
Berry PM, Spink J (2012) Predicting yield losses caused by lodging in wheat. Field Crops Res 137:19–26
Berry PM, Spink J, Sterling M, Pickett AA (2003) Methods for rapidly measuring the lodging resistance of wheat cultivars. J Agron Crop Sci 189:390–401
Berry PM, Sterling M, Spink JH, Baker CJ, Stylvester-Bradley R, Mooney SJ, Tams AR, Ennos AR (2004) Understanding and reducing lodging in cereals. Adv Agron 84:217–271
Berry PM, Sylvester-Bradley R, Berry S (2007) Ideotype for lodging resistant wheat. Euphytica 154:165–179
Berry PM, Kendall S, Rutterford Z, Orford S, Griffiths S (2015) Historical analysis of the effects of breeding on the height of winter wheat (Triticum aestivum) and consequences for loding. Euphytica 203:375–383
Bugg TDH, Ahmad M, Hardiman EM, Rahmanpour R (2011) Pathways for degradation of lignin in bacteria and fungi. Nat Prod Rep 28:1883–1896
Chuanren D, Bochu W, Pingqing W, Daohong W, Shaoxi C (2004) Relationship between the minute structure and the lodging resistance of rice stems. Colloid Surf B 35:155–158
Crook MJ, Ennos AR (1994) Stem and root characteristics associated with lodging resistance in four winter wheat cultivars. J Agr Sci 123:167–174
Easson DL, White EM, Pickles SJ (1993) The effects of weather, seed rate and cultivar on lodging and yield in winter wheat. J Agr Sci 121:145–156
Esechie HA, Rodriguez V, Al-Asmi H (2004) Comparison of local and exotic maize varieties for stalk lodging components in a desert climate. Eur J Agron 21:21–30
Feng S, Jiang X, Hu T, Niu L, Ru Z, Li X, Yin K (2012) Study on relationship between the stem microstructure and lodging resistance with different wheat varieties. Chin Agric Sci Bull 28:57–62
Flintham JE, Börner A, Worland AJ, Gale MD (1997) Optimizing wheat grain yield: effects of Rht (gibberellin-insensitive) dwarfing genes. J Agr Sci 128:11–25
Foulkes MJ, Slafer GA, Davies WJ, Berry PM, Sylvester-Bradley R, Martre P, Calderini DF, Griffiths S, Reynolds MP (2011) Raising yield potential of wheat. III. Optimizing partitioning to grain while maintaining lodging resistance. J Exp Bot 62:469–486
Gooding MJ, Davies WP (1997) Wheat production and utilization: systems, quality and the environment. CAB International Press, New York
Hai L, Guo H, Xiao S, Jiang G, Zhang X, Yan C, Xin Z, Jia J (2005) Quantitative trait loci (QTL) of stem strength and related traits in a doubled-haploid population of wheat (Triticum aestivum L.). Euphytica 141:1–9
He ZH, Rajaram S, Xin ZY, Huang GZ (2001) A history of wheat breeding in China. CIMMYT, Mexico
Horton P (2000) Prospects for crop improvement through the genetic manipulation of photosynthesis: morphological and biochemical aspects of light capture. J Exp Bot 51:475–485
Islam MJ, Peng SB, Visperas RM, Ereful N, Bhuiya MSU, Julfiquar AW (2007) Lodging-related morphological traits of hybrid rice in a tropical irrigated ecosystem. Field Crops Res 101:240–248
Jezowski S (1981) Variation correlation and heritability of characters determining lodging of spring barley (Hordeum vulgare L.). II. Analysis of relationship between lodging grade and some morphological characters of spring barely varieties. Genet Pol 22:45–61
Jin SB (1986) Records of Chinese wheat cultivars (1962–1982). China Agriculture Press, Beijing
Jin SB (1997) Records of Chinese wheat cultivars (1983–1993). China Agriculture Press, Beijing
Jin SB, Liu DA (1964) Records of Chinese wheat cultivars (1962). China Agriculture Press, Beijing
Kelbert AJ, Spaner D, Briggs KG, King JR (2004) The association of culm anatomy with lodging susceptibility in modern spring wheat genotypes. Euphytica 136:211–221
Kirk TK, Obst JR (1988) Lignin determination. Method Enzymol 161:87–101
Kokubo A, Kuraishi S, Sakurai N (1989) Culm strength of barley: correlation among maximum bending stress, cell wall dimensions and cellulose content. Plant Physiol 91:876–882
Kong E, Liu D, Guo X, Yang W, Sun J, Li X, Zhan K, Cui D, Liu J, Zhang A (2013) Anatomical and chemical characteristics associated with lodging resistance in wheat. Crop J 1:43–49
Ma Q, Liu Y (2014) TaDIR13, a dirigent protein from wheat, promotes lignan biosynthesis and enhances pathogen resistance. Plant Mol Biol Rep 33:143–152
Ma Q, Wang C, Zhu H (2011) TaMYB4 cloned from wheat regulates lignin biosynthesis through negatively controlling the transcripts of both cinnamyl alcohol dehydrogenase and cinnamoyl-CoA reductase genes. Biochimie 93:1179–1186
Mathews KL, Chapman SC, Trethowan R, Singh RP, Crossa J, Pfeiffer W, van Ginkel M, Delacy I (2006) Global adaptation of spring bread and durum wheat lines near-isogenic for major reduced height genes. Crop Sci 46:603–613
Mu M, Zhang R, Cui C, Yi S, Ma C, Xun Y (2010) Analysis of source-sink-translocation characteristics and indicators for high-yield colony of winter wheat. Chin J Eco-Agric 18:5–40
Neenan M, Spencer-Smith JL (1975) An analysis of the problem of lodging with particular reference to wheat and barley. J Agr Sci 85:495–507
Ortiz-Monasterio R, Sayre KD, Rajaram S, Mcmahon M (1997) Genetic progress in wheat yield and nitrogen use efficiency under four nitrogen rates. Crop Sci 37:898–904
Peng D, Chen X, Yin Y, Lu K, Yang W, Tang Y, Wang Z (2014) Lodging resistance of winter wheat (Triticum aestivum L.): lignin accumulation and its related enzymes activities due to the application of paclobutrazol or gibberellin acid. Field Crops Res 157:1–7
Sherman JM, Martin JM, Blake NK, Lanning SP, Talbert LE (2014) Genetic basis of agronomic differences between a modern and a historical spring wheat cultivar. Crop Sci 54:1–13
Updegraff DM (1969) Semimicre determination of cellulose in biological materials. Anal Biochem 32:420–424
Wang J, Zhu J, Lin Q, Li X, Teng N, Li Z, Li B, Zhang A, Lin J (2006) Effects of stem structure and cell wall components on bending strength in wheat. Chin Sci Bull 51:815–823
Wang SZ, Tian YF, Guo TC, Wang ZH (2010) Wheat cultivation in Henan Province. China Agricultural Science and Farming Techniques Press, Beijing
Wu W, Li C, Ma B, Shah F, Liu Y, Liao Y (2014) Genetic progress in wheat yield and associated traits in China since 1945 and future prospects. Euphytica 196:155–168
Zadoks JC, Chang TT, Konzak CF (1974) A decimal code for the growth stages of cereals. Weed Res 14:415–421
Zhuang QS (2003) Chinese wheat improvement and pedigree analysis. China Agricultural Press, Beijing
Zuber U, Winzeler H, Messmer MM, Keller M, Keller B, Schmid JE, Stamp P (1999) Morphological traits associated with lodging resistance of spring wheat (Triticum aestivum L.). J Agron Crop Sci 182:17–24
Acknowledgments
This study was supported by the China Agriculture Research System (Grant No. CARS-3-1-9) and the Public-Benefit Project of the Ministry of Agriculture of China (Grant No. 201203033).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, Y., Xu, W., Wang, H. et al. Progress in improving stem lodging resistance of Chinese wheat cultivars. Euphytica 212, 275–286 (2016). https://doi.org/10.1007/s10681-016-1768-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10681-016-1768-1