Abstract
For cross-pollination trees, the optimal breeding method is hybridization. Tree heterosis is commonly present and is the main research focus in tree crossbreeding. Salt stress and interspecific hybridization may lead to DNA methylation changes. The study crossed Fraxinus mandshurica (female parent) with Fraxinus velutina (male parent) to obtain interspecific F1 hybrid progenies that could obtain the good characters of parents. The results showed that growth and survival rate of the interspecific hybrid progenies (F1 hybrids of F. mandshurica × F. velutina) were significantly higher than those of intraspecific open pollinated plants from parental F. mandshurica and F. velutina. Salt tolerance and cytosine methylation in interspecific F1 hybrids and the intraspecific open pollinated plants from parents were examined. Membrane permeability, ROS and antioxidant activity, malondialdehyde, and photosynthesis were measured after salt treatment and genomic methylation was analyzed using a methylation-sensitive amplified polymorphism protocol. F1 hybrids exhibited heterosis for growth in normal as well as high salt conditions. DNA methylation in the F1 hybrids was lower than the intraspecific open pollinated plants from parents. Salt treatments changed DNA methylation patterns in F1 hybrids. Genomic DNA of the intraspecific open pollinated plants from parents had internal cytosine methylation (average of 13.22 %), whereas F1 hybrid seedlings had external cytosine methylation (average of 7.34 %). Such changes in DNA methylation patterns in F1 hybrids suggest a connection between salt tolerance and epigenetic mechanisms in plants. We observed that alteration of DNA methylation was closely correlated with the adaptation to the salt stress and provided epigenetic mechanisms of salt tolerance in the interspecific hybridization of trees.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Plants respond to severe environmental changes or stresses, such as high salt, low temperature, or drought, via physiological and developmental changes. Salt stress affects plant metabolism by reducing water potential, perturbing ion balance, reducing CO2 assimilation, and production of reactive oxygen species (ROS) (Bohnert and Jensen 1996; Hasegawa et al. 2000; Zhu 2001, 2002, Munns and Tester 2008). Plants have developed various protective strategies to minimize saline stress, such antioxidant systems to quench ROS (Apel and Hirt 2004; Kočová et al. 2009). For example, superoxide dismutase (SOD) is a major scavenger of superoxide (O2 −), producing H2O2 and O2. Hydrogen peroxide is then scavenged by catalase (CAT) and various peroxidises (PODs) (Misra and Gupta 2006). Enzymes involved in the ascorbate–glutathione (GSH) cycle include ascorbate peroxidase, dehydroascorbate reductase, monodehydroascorbate reductase, and GSH reductase also contribute to stress response (Noctor and Foyer 1998; Hernández et al. 2000; Farmer and Mueller 2013).
Fraxinus mandshurica (Manchurian ash) is a dioecious, wind-pollinated, cold-adapted, and broad-leaved tree species that is susceptible to drought and saline stress (Hu et al. 2008). Even though it comprises one of the most important components of cool-temperate forest ecosystems in Northeast China, F. mandshurica has been declared an endangered tree species in China due to its extensive overexploitation and widespread deforestation (Hu et al. 2008). Fraxinus velutina is a species native to southwestern North America, and it is widely distributed in coastal areas because of its rapid growth rate and tolerance to alkaline soils (Griffin and Critchfield 1972; Liu et al. 2003). Fraxinus velutina does not adapt well to cold weather in Northeast China, such as in the Heilongjiang Province. Thus, we crossed F. mandshurica (female parent) with F. velutina (male parent) to obtain interspecific F1 hybrid progenies that could obtain the good characters of parents. Heterosis refers to the phenomenon in which the hybrid F1 offspring exhibit phenotypic characteristics that are superior to the mean of the two parents (mid-parent heterosis), or the better of the two parents (better parent heterosis) (Springer and Robert 2007). Heterosis for various defense-related enzymes and physiological changes depends on the susceptibility or tolerance of a plant to saline stress (Noctor and Foyer 1998; Hernández et al. 2000). Hybridization between cultivated and wild lettuce (Lactuca serriola) resulted in a moderate to high heritability for the vigour traits and many of the hybrids showed improved vigour over the wild parent under non-stress and stress conditions (Hooftman et al. 2007, 2009). King et al. (1997) reported the crosses between letraploid wheat, Triticum durum, and Th. bessurabicum and demonstrated that they are more tolerant to high salt concentrations than their wheat parents. Heterosis was apparent under saline (NaCl) conditions in the elongation of stems in hybrids of Lycopersicon esculentum produced with three wild species (L. cheesmanii, L. peruvianum, and L. pennellii = Solanum pennellii) by Tal and Shannon (1983). Total dry matter production of another F1 hybrid, between L. esculentum and L. pennellii, showed hybrid vigour (SarangaY et al. 1991) under saline conditions. Tree heterosis is commonly present and is the main research focus in tree crossbreeding. For cross-pollination trees, the optimal breeding method is hybridization (Shepherd et al. 2008; Li et al. 1998). However, the mechanisms of heterosis have not been fully elucidated. In-depth analysis of heterosis in response to saline stress is essential for the production of new salt-tolerant interspecific progenies of F. mandshurica and F. velutina.
Previous studies suggest that cytosine DNA methylation is critical for orchestrating gene expression during plant development (Rangwala and Richards 2004; Chan et al. 2005; Yan et al. 2010) and in maintaining genome integrity (Cao and Jacobsen 2002; Rapp and Wendel 2005). Recent studies implicate cytosine DNA methylation in plant salt-stress responses (Boyko and Kovalchuk 2011; Mirouze and Paszkowski 2011; Karan et al. 2012; Bilichak et al. 2012; Song et al. 2012; Bräutigam et al. 2013). Interspecific hybridization may lead to chromosomal rearrangements (Shivaprasad et al. 2011), transposable element mobilization (Liu and Wendel 2000; Shan et al. 2005), and DNA methylation changes (Salmon et al. 2005). The objective of this study was to obtain further insights into the relationship of salt tolerance and DNA methylation of interspecific hybrids between F. mandshurica and F. velutina. The alterations of different parameters (growth and survival rate, photosynthetic parameters, defense-related enzymes, and physiological functions) of Fraxinus hybrids grown under optimal and salt stress conditions were analyzed. Moreover, we investigated cytosine methylation patterns using a methylation-sensitive amplified polymorphism (MSAP) analysis in F1 hybrids and parents after salt treatment. We conclud that a connection exists between salt tolerance and altered epigenetic mechanisms in plants. Studies of salt tolerance and DNA methylation variation of interspecific hybrids at salt stress may provide information on epigenetic mechanisms of salt tolerance in the interspecific hybridization of trees. Such information also could be useful to breeders in developing appropriate selection and breeding strategies.
Materials and methods
Plant materials
Male flowering branches of F. velutina were collected in mid-April in Tianjin City (China). The branches were cultivated in water, and their pollen was collected in test tubes and stored at 4 °C. Hybridization was performed in May using F. mandshurica (Dailing, Heilongjiang Province) and F. velutina as the female and male parents, respectively. Four individuals of F. mandshurica were used and labelled as follows: F. m 2, F. m 5, F. m 7 and F. m 8; four individuals of F. velutina were used and labelled as follows: F. v 3, F. v 6, F. v 9 and F. v 10. We made six combinations for crossing: F. m 2 × F. v 3; F. m 5 × F. v 3; F. m 5 × F. v 9; F. m 7 × F. v 3; F. m 7 × F. v 10; F. m 8 × F. v 6. Prior to pollination, F. velutina pollen was treated with a high-voltage electrostatic field (HVEF) for 30 min. The interspecific hybrid progenies and the intraspecific open pollinated plants from parental F. mandshurica and F. velutina were planted in the Maoershan experimental forest farm of the Northeast Forestry University under natural conditions (45.14°N, 127.55°E) in a randomized complete block design. Each region had 30 plants with 5 replicates. After 2 years, the tree height, ground diameter and survival rate were analyzed.
Salt treatment
Healthy 2 year-old plants were grown in the plastic buckets containing humus-sand mixture (3:1) in greenhouse with nature light. The seedlings were randomly divided into control and treatment groups. Treatment groups received 200 mmol/L salt and three replicates were taken from each group. Seedlings were administered salt solution twice a day, then irrigated daily with water. Physiological parameters of each plant were measured on the tenth day after treatment. Seedling height was measured before and after treatment; survival percentages and symptoms of stress were recorded everyday after treatment. Stress symptoms included the time and degree of leaf wilting, withering, and shedding. The same plants were given water without salt for 3 d after 10 d of salt treatment.
Photosynthesis
Net photosynthetic rate (Pn), stomatal conductance (Gs), intercellular CO2 concentration (Ci), and transpiration rate (Tr) were measured with a LI-6400XT portable photosynthesis system (Li-COR, USA) using the following measuring cell (6 cm2) parameters: 26 °C, 800 µmol photons m−2 s−1, and an air flow of 300 µl s−1, as previously described (Mittler et al. 2001; Pnueli et al. 2003).
MDA, Electrolyte leakage and ROS steady-state measurement
Thiobarbituric acid (TBA)-reactive substance-MDA adducts were was extracted as described previously (Meir et al. 1992) by homogenization of 0.5 g of tissue in 5 mL of solution containing 20 % trichloroacetic acid (TCA) and 1.5 mM EDTA. TBA reactive substance-MDA adducts was assayed via the TBA testing (Kosugi and Kikugawa 1985), according to modifications of Meir et al. (1992). One milliliter of 0.67 % TBA was added to 3 mL aliquot of the supernatant and the solution was incubated at 100 °C for 1 h. The solution was then cooled to room temp and centrifuged (11, 000×g) for 10 min. The volume of the resultant supernatant was increased to 10 mL with distilled water, and the absorbance was read at 532 nm (A532) to measure MDA. Three replicates were used for each assay. Electrolyte leakage (EL) was determined according the method of Lutts et al. (1996) and Maribel and Dionisio-Sese (1998). Leaf segments (5-mm) were measurement for each treatment. Briefly, samples were washed with de-ionized water to remove surface adherent electrolytes, and then samples were placed in closed vials containing 10 mL de-ionized water and incubated at 25 °C on a rotary shaker for 24 h. The electrical conductivity of the solution (L1) was subsequently determined. Samples were then autoclaved at 120 °C for 20 min and the final electrical conductivity (L2) was determined after equilibration at 25 °C. The EL was defined as follows: EL (%) = (L1/L2) × 100. Total indirect ROS was measured twice using four replicates as described by Causevic et al. (2006) using a Bioxytech H2O2-560 assay kit (TEBU-BIO, Le Perray en Yvelines, France) according to the manufacturer’s instructions.
Enzyme assays
Enzyme extracts were prepared by homogenizing leaves in pre-chilled 50 mM sodium phosphate buffer (pH 7.0) containing 1.0 mM (EDTA), 0.5 % (v/v) Triton X-100 and 1 % (w/v) polyvinyl-pyrrolidone (PVPP). Ascorbate (5 mM) was added to the extraction buffer for determination of ascorbate peroxidase (APX), monodehydroascorbate reductase (MDHAR) and dehydroascorbate reductase (DHAR). Homogenates were centrifuged at 18,000×g for 20 min at 4 °C, and their total soluble protein content was measured by Bradford’s assay (Bradford 1976). Supernatants were immediately used for standard enzyme assays and three replicates were measured for each enzyme assay. DHAR activity was measured according to the protocol by Doulis et al. (1997). The assay mixture contained 90 mM phosphate buffer (pH 7.0), 0.1 mM EDTA, 5.0 mM reduced GSH, and approximately 30 μg extracted protein. The reaction was initiated with the addition of freshly made 0.2 mM dehydroascorbate (DAsA). Activity was measured by the reduction of DAsA at 265 nm after accounting for the non‐enzymatic reduction of DAsA by GSH. MDHAR activity was assayed in a reaction mixture consisting of 90 mM potassium phosphate (pH 7.5), 0.01 mM EDTA, 0.0125 % Triton X‐100, 2.5 mM ascorbate, 0.25 units ascorbate oxidase (units as defined by Sigma Chemical Co.), 0.2 mM NADH, and up to 50 μg protein extract (Hossain et al. 1984). The reaction was monitored by measuring the decrease in absorbance at 340 nm due to NADH oxidation. GR activity was determined according to the procedure by Foyer and Halliwell (Foyer and Halliwell 1976). The reaction mixture contained 80 mM Tris‐HCl (pH 8.5), 1.5 mM EDTA, 2.5 mM oxidized GSH (GSSG), and up to 100 μg extracted protein. The reaction was initiated with the addition of 0.5 mM NADPH in 1 % NaHCO3 and then oxidation of NADPH was read at 340 nm. APX was measured using methods described by Nakano and Asada (1987). Briefly, the assay mixture containing 90 mM potassium phosphate (KP) buffer (pH 7.0), 0.1 mM EDTA, 0.65 mM ascorbate, and 1.0 mM H2O2 was initiated with the addition of approximately 40 μg extracted protein. Activity was measured via H2O2‐dependent decomposition of ascorbate at 290 nm. CAT activity was assayed in a reaction mixture containing 100 mM KP buffer (pH 6.5), 1.0 mM EDTA, 60.0 mM H2O2, and approximately 40 μg extracted protein according to the method described by Aebi (1983). Activity was measured by following the decomposition of H2O2 at 240 nm. Gamma glutamyl transferase (indicator of GSH) was measured according to Smith (1985). Briefly, 1 g (fresh weight) of leaves were homogenized in 5 ml 5 % sulfosalicylic acid; homogenates were centrifuged at 1000×g for 10 min and the supernatant was neutralized with 1.5 ml 0.5 M potassium phosphate buffer (pH 7.5) to assay total GSH. The standard incubation mixture contained: 0.5 ml 0.1 M sodium phosphate buffer (pH 7.5) containing 5 mM EDTA, 0.2 ml 6 mm 5, 5′-dithiobis-(2-nitrobenzoic acid), 0.1 ml of 2 mM NADPH, and 0.1 ml (1 unit) of yeast GSH reductase type III (Sigma Chemical Co.). The reaction was initiated by the addition of 0.1 ml GSH standard (50–400 ng) or extract. The change in absorbance at 412 nm (A412) was monitored for 650 s at 25 °C or until A412 reached 0.5.
The enzyme extract was used for the AsA-POD (POD activity toward ascorbate) and G-POD (POD activity toward guaiacol) assays. AsA-POD activity was assayed according to the method of Amako et al. (1994). The reaction was started by adding H2O2. The G-POD assay mixture contained 0.1 M phosphate buffer (pH 6.1), 4 mM guaiacol as donor, 3 mM H2O2 as substrate, and 1.0 ml crude enzyme extract. The total reaction volume was 3.0 ml. The rate of change in absorbance at 420 nm was measured, and the level of enzyme activity was expressed as the difference in absorbance (OD).
Nucleic acid isolation and MSAP analysis
Genomic DNA was isolated from fresh leaves using the cetyl trimethyl ammonium bromide (CTAB) method described by Porebski et al. laboratory (1997). MSAP analysis was conducted as reported previously (Reyna-Lopez et al. 1997; Xiong et al. 1999). The restriction enzymes EcoRI, HpaII, and MspI were purchased from New England Biolabs Inc (location). Overall, one pair of pre-selective primers and 20 pairs of selective primers were used for amplifications (Supplementary Table 1). Silver-stained sequencing PAGE was used to resolve and visualize amplification products. HpaII and MspI recognize the same sequence (CCGG) but have different sensitivities to DNA methylation. HpaII cleaves sequences with hemi-methylated external cytosines (mCCGG), whereas, MspI can only cleave sequences that are fully methylated at internal cytosines (CmCGG) (Ashikawa 2001). Only clear and reproducible bands that appeared in two independent PCR amplifications were scored. The scored MSAP bands represented three major cytosine methylation states: (1) hemi-methylation of the external cytosine, which were bands present in HpaII-digest but absent in the corresponding MspI-digest, such as pattern H/M = ±; (2) full methylation of the internal cytosine, which were bands absent in HpaII-digest but present in the corresponding MspI-digest, such as pattern H/M = ∓; and (3) non-methylation of both cytosines, which were bands absent in both HpaII- and MspI-digest, such as pattern H/M = −/−.
Overall evaluation of salt resistance and statistical analysis
Fuzzy subordination method was adopted to evaluate salt resistance and measure greater hybrid vigor. The subordinate function adopts the method of membership functions in fuzzy mathematics for different combinations to determine their average index subordinate function values and identify their resistance. If a positive correlation exists between the index and resistance, then the formula is X (u) = (X−Xmin)/(Xmax−Xmin). If a negative correlation exists between the index and resistance, then the formula is X (u) = 1−(X−Xmin)/(Xmax−Xmin). In the two formulas, X is the index determination value, Xmax is the maximum index determination value, and Xmin is the minimum index determination value.
Statistical analyses of data were performed using the SPSS statistical software package (SPSS version 19.0). Data were expressed as mean ± SE, and were compared by ANOVA (significance p ≤ 0.05). Enzyme assays and physiological parameters were all measured using three replicates.
Results
Adaptability and growth of F1 hybrids in field under natural conditions
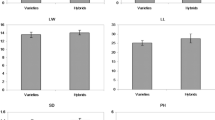
To evaluate the adaptability and overwintering survival of F1 hybrids under natural conditions, F1 generation plants were grown in the forest field for 2 years. The results showed that growth and survival rate of the interspecific hybrid progenies (F1 hybrids of F. mandshurica × F. velutina) were significantly higher than those of intraspecific open pollinated plants from parents. F. velutina died in the winter and was regenerated from the root in the spring, suggesting that F. velutina cannot live through the winter (Fig. 1). These findings suggest that interspecific hybrid progenies (F1 hybrids of F. mandshurica × F. velutina) exhibit heterosis for growth and survival rate.
Growth and survival rate of different F1 generation in the field F. m 2, F. m 5, F. m 8, F. v 3, F. v 10, F. v 6 represented the intraspecific open pollinated plants from parental F. mandshurica 2, F. mandshurica 5, F. mandshurica 8, F. velutina 3, F. velutina 10, F. velutina 6, respectively. Data are mean ± SE from three independent replicates. The different normal letters indicate significant difference among variance ration after salt stress of F1 at 0.05 levels as determined by one-way ANOVA
Effect of salt stress on seedling growth of F1 hybrids and intraspecific open pollinated plants from parents
To study the effect of salt stress on the growth of F1 generation plants, healthy 2 year-old plants were grown in greenhouse with nature light in a randomized complete block design. The plants were treated with salt stress. Seedling growth was greatly affected by stress, and this may reflect plant adaptation to their environments. Two days after treatment with 200 mmol/L salt solution, F1 hybrid seedling leaves gradually curled, wilted, and discolored (yellow), and salt crystals appeared on the leaf surfaces. Five days after treatment, the top leaves of all F1 hybrids gradually recovered their normal color; however, the bottom leaves of F. m 2, F. m 5, F. m 7 and F. m 8 had brownish patches and were shedding. Ten days after treatment, the top leaves of F. m 8 × F. v 6 and the intraspecific open pollinated plants from parents had brownish patches, none of the other F1 hybrids had these characteristics. The relative growth of F1 hybrids and the intraspecific open pollinated plants from parents under no salt stress (controls) and 200 mmol/L salinity is shown in Table 1. Except for F. m 8 × F. v 6, the relative growth of the other F1 hybrids was greater than growth of the the intraspecific open pollinated plants from parents when seedlings were not salt treated. F. m 7 × F. v 10 had the highest relative growth (164.83 %), followed by F. m 2 × F. v 3 (150.49 %), F. m 8 × F. v 6 had the lowest relative growth (95.17 %). Ten days after salt treatment, F1 hybrid growth decreased compared with untreated controls. Specifically, F. m 8 × F. v 6 growth was only 29.95 % of that of the untreated control. With the exception of F. m 8 × F. v 6 and F. m 5 × F. v 3, the relative growth of all F1 hybrids was higher than those of their respective parents, whether they were treated with salt or not, indicating increased F1 hybrid vigor compared to their parents.
Effect of salt stress on cell membrane permeability, MDA content, and steady-state ROS in seedlings
Relative conductivity, MDA content, and steady-state ROS are important physiological indices for examining injured/stressed cells as they reflect the variety and degree of cell membrane injury (Puckette et al. 2007). Conductivity measurements of cell membrane revealed that different F1 hybrids had significant differences in permeability in response to salt stress (Fig. 2). Compared with their untreated controls, cell membrane permeability increased in all hybrid F1 progeny. Cell membrane permeability of F. m 7 × F. v 10 increased only 4.17 % compared with its untreated control. Similarly, salt stress did not significantly affect membrane permeability of F. m 5 × F. v 9, indicating that F. m 7 × F. v 10 and F. m 5 × F. v 9 had greater hybrid vigor with respect to resisting cell injury after salt stress. The cell membrane permeability of F. m 5 × F. v 3 increased 304.15 % compared with its untreated control. MDA content and ROS followed similar trends, with differences between treatment and control groups in the same combination being significantly different (Fig. 2).
Cell membrane permeability, MDA content, and steady-state ROS in seedlings treated with/without salt. Data are mean ± SE of three independent replicates (n = 3). * (0.01) and ** (0.05) indicate significant differences between treatment and control in the same combination. Ten days after treatment with/without salt, MDA, electrolyte leakage and steady-state ROS in the seedlings were measured as described in Materials and methods. Dw dry weight, fw fresh weight
Salt treatment increases antioxidant activity
APX, GR, DHAR, and MDHAR are essential antioxidants involved in the ascorbate-GSH pathway. APX, GR, MDHAR, and DHAR increased in plants treated with 200 mM salt compared to controls (Fig. 3). POD and CAT activity and GSH content also increased after salt treatment (Fig. 4). POD activity in F. m 7 × F. v 10 was the highest of the groups, increasing 58.92 % from 16.82 U/g before treatment to 26.73 U/g after treatment. POD activity was lowest in F. m 5. GSH and CAT activity followed similar trends. We compared antioxidant activity between interspecific hybrid progenies and the intraspecific open pollinated plants from parental F. mandshurica and F. velutina. With the exception of F. m 8 × F. v 6, antioxidant activity in interspecific hybrid progenies of all other families was greater than that measured in the intraspecific open pollinated plants from female parent F. mandshurica. Antioxidant activity of F. m 7 × F. v 10 hybrid exceeded their respective parents after saline stress and normal growth conditions. These findings suggest that F. m 7 × F. v 10 hybrid exhibits heterosis for antioxidant enzymes.
Effects of salt stress on MDAR, DHAR, GH, APX activity in seedlings treated with/without salt. Data are mean ± SE (n = 3). * (0.01) and ** (0.05) indicate significant differences between treatment and control in the same combination. Ten days after treatment with/without salt MDAR, DHAR, GH, APX activity in seedlings were measured as described in Materials and Methods. Dw dry weight; fw fresh weight, prot protein
Effects of salt stress on CAT, POD activity and GSH in seedlings treated with/without salt. Data are mean ± SE (n = 3). * (0.01) and ** (0.05) indicate significant differences between treatment and control in the same combination. Ten days after treatment with/without salt, CAT, POD activity and GSH in seedlings were measured as described in Materials and methods. Dw dry weight; fw fresh weight, prot protein
Effect of salt treatment on photosynthesis
Significant differences in photosynthetic parameters were observed between untreated controls and treated groups. Compared with other seedlings, F. m 5 × F. v 3 had a lower net photosynthetic rate (Pn), but the intercellular CO2 concentration (Ci) was high under stress (Fig. 5). Salt stress significantly decreased Pn and stomatal conductance (Gs), indicating an inhibitory effect on these parameters. The most significant reduction in Pn and Gs was observed in F. m 7 × F. v 10, which decreased by 84.39 and 89.88 % compared with its untreated control. F. m2, F. m 2 × F. v 3, F. m 7 × F. v10, F. v10 had a lower Ci rate compared to their untreated controls, illustrating their more efficient CO2 utilization. The seedlings consumed more water to restrain their growth and increase their Tr. Data indicate that Pn and Gs were reduced in all of the interspecific hybrid progenies in response to salt stress. Furthermore, with the exception of F. m 2 × F. v 3 and F. m 5 × F. v 9, other F1 hybrids had a lower Tr than those of their intraspecific open pollinated plants from parental F. mandshurica and F. velutina seedlings under salt stress. These results reveal that the interspecific F1 hybrid progenies underwent heterosis for their photosynthetic functions under salt stress.
Effect of salt treatment on photosynthesis in seedlings treated with/without salt. Data are mean ± SE (n = 3). * (0.01) and ** (0.05) indicate significant differences between treatment and control in the same combination. Ten days after treatment with/without salt, Net photosynthetic rate, stomatal conductance, intercellular CO2 concentration, and Tr were measured as described in Materials and methods
Overall evaluation of salt resistance
We adopted the most widely used method “fuzzy subordination method” to evaluate salt resistance and measure greater hybrid vigor. The subordinate function adopts the method of membership functions in fuzzy mathematics for different combinations to determine their average index subordinate function values and identify their resistance. Based on fuzzy subordination method, the average index subordinate function values (\(\Delta\)) with salt treatment for each line were calculated. The higher the total index subordinate function values, the stronger the salt resistance of a particular line. Figure 6 showed overall evaluation of salt resistance in seedlings by fuzzy subordination method. F1 hybrids F. m 7 × F. v 10, F. m 2 × F. v 3, and F. m 7 × F. v 3 had the highest salt resistance, and their subordinate function values were 10 % greater than average, whereas F. m 8 × F. v 6 had the least salt tolerance in F1 hybrids.
Alterations in cytosine methylation in the interspecific hybrids revealed by MSAP
Using 20 pairs of EcoRI + HpaII/MspI selective primer combinations (Supplementary Table 1), 394–726, clear and reproducible bands were amplified by MSAP. Based on these MSAP patterns, various bands representing non-methylation, hemi-methylation of external cytosine, and full methylation of internal cytosine were tabulated. Among the intraspecific open pollinated plants from parental F. m 7, F. v 10 and F. m 5, total methylation (calculated by adding up the various patterns) was 25.04, 23.02 and 28.05 %, respectively. Total DNA methylation levels in interspecific hybrid progenies F. m 7 × F. v 3, F. m 7 × F. v 10, and F. m 5 × F. v 9 ranged from 18.51 to 20.79 %; the hemi-methylation of the external cytosine ranged from 10.16 to 13.36 %, and the full methylation of the internal cytosine ranged from 6.23 to 8.35 % (Fig. 7; Table 2). These results suggested that the methylation patterns between interspecific hybrid progenies and the intraspecific open pollinated plants from parental F. mandshurica and F. velutina were different. The level of DNA methylation in interspecific hybrid progenies F. m 7 × F. v 3, F. m 7 × F. v10, and F. m 5 × F. v 9 was significantly lower than those of the intraspecific open pollinated plants from parents. Differences in DNA methylation level and patterns were observed between untreated controls and salt-treated plants. On average, DNA methylation of the intraspecific open pollinated plants F. m 7, F. v10 and F. m5 increased from 31.97 to 35.19 % 5 d after salt treatment, whereas those of interspecific hybrid progenies F. m 7 × F. v 3, F. m 7 × F. v10, and F. m 5 × F. v 9 increased from 27.11 to 28.85 %. Methylation of F. m 7 reached 35.19 % after 5 d of salt treatment, whereas that of F. m 7 × F. v10 was only 26.09 %. DNA methylation decreased slightly 10 d after salt treatment. After rehydration for 3 d, cytosine methylation remained high (22.83–25.14 % in F1 hybrid progenies), suggesting that salt treatment can increase methylation of the plant genome.
Discussion
Compared with the intraspecific open pollinated plants from parents under normal growth conditions, F. m 7 × F. v10 had the greatest growth (164.83 %). Among hybrid combinations, family F. m 7 had the highest relative growth rate. Significant differences were observed among interspecific F1 hybrids of F. velutina and F. mandshurica and the intraspecific open pollinated plants from parents after salt treatment. Generally, growth suppression is one of the most significant symptoms of plants suffering saline stress (Tozlu et al. 2000). Our results showed that seedling growth was inhibited by salt stress. Leaves had brown patches and some were shed; however, with the exception of F. m 8 × F. v 6, F1 hybrids had less growth reduction than their respective parents. Our results indicate that F1 hybrids had better hybrid vigor under both saline stress and normal growth conditions.
Malondialdehyde (MDA) content is a commonly used measurement for assessing lipid peroxidation and oxidative damage in plants (Queiroz et al. 1998; Zhou and Zhao, 2004), and its maintenance of low levels has been associated with increased stress resistance in many plant species (Lima et al. 2002; DaCosta and Huang, 2007). Cell membrane stability plays a critical role in maintaining cell turgor and physiological functions, particularly during plant dehydration, and electrolyte leakage (EL)has been widely used to estimate cell membrane stability (Blum and Ebercon 1981; Xu et al. 2011). In this study, membrane permeability and MDA content increased to varying degrees under salt stress. whereas, cell membrane permeability and MDA content of F. m 7 × F. v 10 increased only 4.17 and 12.19 % compared with its untreated control, which indicated that F. m 7 × F. v 10 had greater hybrid vigor with respect to resisting cell injury after salt stress. The antioxidant system in plants may be a result of sequential and simultaneous actions by several antioxidant enzymes, including CAT and POD. Both CAT and POD scavenge the accumulated H2O2 to non-toxic levels by converting it into water and oxygen (Apel and Hirt 2004). Our experiments showed that activities of these enzymes in salt-stressed plant leaves were heightened over time when compared with the controls. Peak activities of CAT and POD were founded in F. m 7 × F. v 10 after salt treatment. Again, these increases in activities were more pronounced in our salt-tolerant hybtids, a finding in accordance with those reported with other studies (Hernandez et al. 2010; Zlatev et al. 2006; El-Mashad and Mohamed 2012). With the exception of F. m 8 × F. v 6, antioxidant activity in interspecific hybrid progenies was greater than that in the intraspecific open pollinated plants from F. mandshurica,which demonstrated that they are more tolerant to salt stress than their female parent. We also found here that when enzymes in the AsA-GSH cycle (APX, GR, DHAR, and MDHAR) were induced in some hybrids (F. m 7 × F. v 10 and F. m 7 × F. v 3) under salt stress, oxidative damage (EL and MDA content) could be minimized. This observation is consistent with previous research on the ‘Gala’ cultivar in Malus domestica Borkh (Ma et al. 2008). Photosynthesis is one of the physiological processes that are most sensitive to many environmental stresses, including salt stress (Zhu et al. 2012). Salinity reduced leaf photosynthesis in all species, which was consistent with other reports (Parida et al. 2003; Wang et al. 2007). Our data indicate that Pn and Gs were reduced in all of the interspecific hybrid progenies in response to salt stress. The positive relationship between stomatal conductance and net photosynthesis indicates that the primary limiting factor for net photosynthesis upon exposure to salinity is stomatal closure (Tavakkoli et al. 2010).
Based on the subordinate function method, the average index subordinate function values of all physiological parameters after salt treatment were calculated. F. m 8 × F. v 6 had weakest salt resistance among all F1 lines tested. The strongest salt tolerance was observed in family F. m 7, whereas F. m 7 × F. v10 had the greatest salt tolerance among all plants. The results showed that growth and survival rate of the interspecific hybrid progenies (F1 hybrids of F. mandshurica × F. velutina) were significantly higher than those of intraspecific open pollinated plants from parental F. mandshurica and F. velutina. Paternal F. velutina has not yet been successfully introduced in the Heilongjiang Province of China; this species has wilted shoot tips after year of growth, and these shoots re-sprout in spring. Fraxinus velutina cannot withstand winter in the Heilongjiang Province. Therefore, introducing these varieties into this region does not permit exploitation of their salt tolerance and growth characteristics. Interspecific hybrids could survive winter after 2 years of field trials. We compared interspecific hybrids with their parents and observed that interspecific Fl hybrids gained the advantage of salt tolerance.
Analysis of plant genomic methylation with MSAP revealed a relationship between salt tolerance and DNA methylation. Using a well-studied model of TMV infection, Boyko et al. (2006) established a correlation between stress treatment, loci-specific epigenetic changes, and genome stability in exposed plants and their progenies. Stress can induce changes in gene expression through DNA hypomethylation or hypermethylation (Boyko and Kovalchuk 2011; Karan et al. 2012; Bilichak et al. 2012; Song et al. 2012). Osmotic stress induces transient DNA hypermethylation in two heterochromatic loci in tobacco cell-suspension cultures (Kovarik et al. 1997), and this DNA hypermethylation was also induced by drought stress in pea (Labra et al. 2002). Gene expression is influenced by chromatin structure, which in turn is governed by processes often associated with epigenetic regulation, namely, histone variants, histone post-translational modifications, and DNA methylation (Chinnusamy and Zhu 2009). Epigenetic modifications occur during the plant’s exposure to stress, and initiate numerous changes in gene expression that can persist over several generations (Zhu 2008). Therefore, the epigenetic control of plant responses to stress is a complex phenomenon.
Tsaftaris and Kafka (1998) studied DNA methylation of maize hybrids and their parents and reported that cytosine methylation of the two parents was 31.4 and 28.3 %, respectively, whereas methylation of F1 hybrid was 27.4 %. Hepburn et al. (1985) studied the effect of plant DNA methylation on gene expression to analyze the relationship between hybridization and methylation. They found that the extent of genomic methylation self-hybridization led to the gradual accumulation of genomic methylation, whereas genomic methylation sites could be removed or rearranged by hybridization. In the present study, total methylation in interspecific hybrid progenies ranged from 18.51 to 20.79 %, whereas hemi-methylation of external cytosine ranged from 10.16 to 13.36 %, and full methylation of internal cytosine ranged from 6.23 to 8.35 %. DNA methylation of F1 hybrids was significantly lower than those of corresponding controls in our study, which corroborates results from previous publications (Xiong et al. 1999). Furthermore, our studies also showed that salt stress can increase the methylation degree and alter the methylation pattern in both hybrid and parental intraspecific progenies. Our results for DNA methylation patterns under salt stress in tree plants were similar to those reported in a previous model plant study (Kovalchuk et al. 2003). Dyachenko et al. (2006) studied genomic methylation changes in the Mesembryanthemum crystallinum plant under saline stress conditions and reported that the methylation of CCWGG (where W is A or T) sequence CpNpG (where N is any nucleotide) was twice that of control. This is probably due to the induction of DNA methylation modification and gene expression by stress during the process of plant growth (Hua et al. 2005; Ruiz et al. 2005; Portis et al. 2004). Sensing environmental changes and initiating a gene expression response is most important for plants as sessile autotrophs. Epigenetic systems must be part of the relay from sensing a change in the environment to a change in gene expression (Grant-Downton and Dickinson 2006). The cytoplasmic DNA of the progeny in sexual hybridization is mainly from the maternal chromosome, which results in the formation of a new nucleoplasm relationship and intracellular environment (Natcheva and Cronberg 2007). In the hybrids, DNA methylation patterns are largely adjusted in order to coordinate the expression of the cytoplasmic and nucleus genes and ensure the optimal status of both genetic systems (Grant-Downton and Dickinson 2005). Therefore, our results suggest an association between salt tolerance and methylation changes in plants.
Conclusion
Fraxinus velutina does not adapt well to cold weather in Northeast China, such as in the Heilongjiang Province. The study crossed F. mandshurica (female parent) with F. velutina (male parent) to obtain interspecific F1 hybrid progenies that could obtain the good characters of parents. The results showed that growth and survival rate of the interspecific hybrid progenies were significantly higher than those of intraspecific open pollinated plants from parental F. mandshurica and F. velutina in natural conditions. The F1 hybrids have also increased hybrid vigor with respect to salt stress resistance as demonstrated by measurement of photosynthesis, antioxidant enzymes, and other physiological functions. DNA methylation of F1 hybrids was lower than that measured in the intraspecific open pollinated plants from parents. Furthermore, the DNA methylation patterns of F1 hybrids can be changed by salt treatment. Thus, heterosis of F1 hybrids is associated with genomic methylation status. The study provided epigenetic mechanisms of salt stress resistance in the interspecific hybridization of trees.
Abbreviations
- APX:
-
Ascorbate peroxidase
- CAT:
-
Catalase
- DHAR:
-
Dehydroascorbate reductase
- GR:
-
Glutathione reductase
- GSH:
-
Reduced glutathione
- mC:
-
Methylcytosine
- MDAR:
-
Monodehydroascorbate reductase
- POD:
-
Peroxidises
- ROS:
-
Reactive oxygen species
- SE:
-
Standard error
- SOD:
-
Superoxide dismutase
- Pn:
-
Net photosynthetic rate
- Gs:
-
Stomatal conductance
- Ci:
-
Intercellular CO2 concentration
- Tr:
-
Transpiration rate
- MSAP:
-
Methylation-sensitive amplified polymorphism
References
Aebi HE (1983) Catalase. In: Bergmeyer HU (ed) Methods of enzymatic analysis. Verlag Chemie, Weinhem, pp 273–286
Amako K, Chen GX, Asada K (1994) Separate assay specific for ascorbate peroxidase and guaiacol peroxidase and for chloroplastic and cytosolic isoenzymes of ascorbate peroxidase in plants. Plant Cell Physiol 35:497–504
Apel K, Hir H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399
Ashikawa I (2001) Surveying CpG methylation at 5′-CCGG in the genomes of rice cultivars. Plant Mol Biol 45:31–39
Bilichak A, Ilnystkyy Y, Hollunder J, Kovalchuk I (2012) The progeny of Arabidopsis thaliana plants exposed to salt exhibit changes in DNA methylation, histone modifications and gene expression. PLoS ONE 7(1):e30515. doi:10.1371/journal.ppat.1004735
Blum A, Ebercon A (1981) Cell membrane stability as a measure of drought and heat tolerance in wheat. Crop Sci 21:43–47
Bohnert HJ, Jensen RG (1996) Metabolic engineering for increased salt tolerance. Aust J Plant Physiol 23:661–667
Boyko A, Kovalchuk I (2011) Genome instability and epigenetic modification -heritable responses to environmental stress? Curr Opin Plant Biol 14:260–266
Boyko A, Hudson D, Bhomkar P, Kathiria P, Kovalchuk I (2006) Increase of homologous recombination frequency in vascular tissue of Arabidopsis plants exposed to salt stress. Plant Cell Physiol 47:736–742
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Bräutigam K, Vining KJ, Lafon-Placette C et al (2013) Epigenetic regulation of adaptive responses of forest tree species to the environment. Ecol Evol 3:399–415
Cao X, Jacobsen SE (2002) Role of the Arabidopsis DRM methyltransferases in de novo DNA methylation and gene silencing. Curr Biol 12:1138–1144
Causevic A, Gentil MV, DelaunayA El-SoudWA, GarciaZ Pannetier C, Brignolas F, Hagège D, Maury S (2006) Relationship between DNA methylation and histone acetylation levels, cell redox and cell differentiation states in sugarbeet lines. Planta 224:812–827
Chan SW, Henderson IR, Jacobsen SE (2005) Gardening the genome: DNA methylation in Arabidopsis thaliana. Nat Rev Genet 6:351–360
Chinnusamy V, Zhu JK (2009) Epigenetic regulation of stress responses in plants. Curr Opin Plant Biol 12:133–139
DaCosta M, Huang BR (2007) Changes in antioxidant enzyme activities and lipid peroxidation for bentgrass species in response to drought stress. J Am Soc Hortic Sci 132:319–326
Doulis AG, Debian N, Kingston-Smith AH, Foyer CH (1997) Differential localization of antioxidants in maize. Plant Physiol 114:1031–1037
Dyachenko OV, Zakharchenko NS, Shevchuk TV, Bohnert HJ, Cushman JC, Buryanov YI (2006) Effect of hypermethylation of CCWGG sequences in DNA of Mesembryanthemum crystallinum plants on their adaptation to salt stress. Biochemistry (Moscow) 71:461–465
El-Mashad AA, Mohamed HI (2012) Brassinolide alleviates salt stress and increases antioxidant activity of cowpea plants (Vigna sinensis). Protoplasma 249(3):625–635
Farmer EE, Mueller MJ (2013) ROS-mediated lipid peroxidation and RES-activated signaling. Annu Rev Plant Biol 64:429–450
Foyer CH, Halliwell B (1976) The presence of glutathione and glutathione reductase in chloroplasts: a proposed role in ascorbic acid metabolism. Planta 133:21–25
Grant-Downton R, Dickinson H (2005) Epigenetics and its implications for plant biology. 1. The epigenetic network in plants. Ann Bot 96:1143–1164
Grant-Downton RT, Dickinson HG (2006) Epigenetics and its implications for plant biology 2. The ‘epigenetic epiphany’: epigenetics, evolution and beyond. Ann Bot 97(1):11–27
Griffin JR, Critchfield WB (1972) The distribution of forest trees in California. Research paper PSW-82. USDA Forest Service, Pacific Southwest Forest and Range Experiment Station, Berkeley, CA
Hasegawa PM, Bressan RA, Zhu JK, Bohnert HJ (2000) Plant cellular and molecular responses to high salinity. Annu Rev Plant Physiol Plant Mol Biol 51:463–499
Hepburn AG, Wade M, Fraser RSS (1985) Present and future prospects for exploitation of resistance in crop protection by novel means. Mech Resist Plant Dis Adv Agric Biotechnol 17:425–452
Hernandez M, Fernandez-Garcia N, Diaz-Vivancos P, Olmos E (2010) A different role for hydrogen peroxide and the antioxidative system under short and long salt stress in Brassica oleracea roots. J Exp Bot 61(2):521–535
Hernández JA, Jiménez A, Mullineaux PM, Sevilla F (2000) Tolerance of pea (Pisum sativum L.) to long-term salt stress is associated with induction of antioxidant defences. Plant Cell Environ 23:853–862
Hooftman DAP, De Jong MJ, Oostermeijer JGB, Den Nijs HCM (2007) Modelling the long-term consequences of crop–wild relative hybridization: a case study using four generations of hybrids. J Appl Ecol 44:1035–1045
Hooftman DAP, Hartman Y, Oostermeijer JGB, Den Nijs HCM (2009) Existence of vigorous lineages of crop–wild hybrids in lettuce under field conditions. Environ Biosaf Res 4:203–217
Hossain MA, Nakano Y, Asada K (1984) Monodehydroascorbate reductase in spinach chloroplasts and its participation in regeneration of ascorbate for scavenging hydrogen peroxide. Plant Cell Physiol 25:385–395
Hu LJ, Uchiyama K, Shen HL, Saito Y, Tsuda Y, Ide Y (2008) Nuclear DNA microsatellites reveal genetic variation but a lack of phylogeographical structure in an endangered species, Fraxinus mandshurica, across North-east China. Ann Bot 102:195–205
Hua Y, Chen XF, Xiong JH, Zhang YP, Zhu YG (2005) Isolation and analysis of differentially methylated fragment CIDM7 in rice induced by cold stress. Heredits 27(4):595–600
Karan R, DeLeon T, Biradar H, Subudhi PK (2012) Salt stress induced variation in DNA methylation pattern and its influence on gene expression in contrasting rice genotypes. PLoS ONE 7(6):e40203. doi:10.1371/journal.ppat.1004735
King IP, Law CN, Cant KA, Orford SE, Reader SM, Miller TE (1997) Tritipyrum, a potential new salt-tolerant cereal. Plant Breed 116:127–132
Kočová M, Holá D, Wilhelmová N, Rothová O (2009) The influence of low-temperature on the photochemical activity of chloroplasts and activity of antioxidant enzymes in maize leaves. Biol Plant 53:475–483
Kosugi H, Kikugawa K (1985) Thiobarbituric acid reaction of aldehydes and oxidized lipids in glacial acetic acid. Lipids 20(12):915–921
Kovalchuk I, Kovalchuk O, Kalck V, Boyko V, Filkowski J, Heinlein M, Hohn B (2003) Pathogen-induced systemic plant signal triggers DNA rearrangements. Nature 423:760–762
Kovarik A, Koukalova B, Bezdek M, Opatrn Z (1997) Hypermethylation of tobacco heterochromatic loci in response to osmotic stress. Theor Appl Genet 95:301–306
Labra M, Ghiani A, Citterio S, Sgorbati S, Sala F, Vannini C, Ruffini-Castiglione M, Bracale M (2002) Analysis of cytosine methylation pattern in response to water deficit in pea root tips. Plant Biol (Stuttgart) 4:694–699
Li B, Howe GT, Wu R (1998) Developmental factors responsible for heterosis in aspen hybrids (Populus tremuloides × P. tremula. Tree Physiol 18(1):29–36
Lima ALS, DaMatta FM, Pinheiro HA, Totola MR, Loureiro ME (2002) Photochemical responses and oxidative stress in two clones of Coffea canephora under water deficit conditions. Environ Exp Bot 47:239–247
Liu B, Wendel JF (2000) Retrotransposon activation followed by rapid repression in introgressed rice plants. Genome 43:874–880
Liu H, Bauer LS, Gao R, Zhao T, Petrice TR, Haack RA (2003) Exploratory survey for the emerald ash borer, Agrilus planipennis (Coleoptera: Buprestidae), and its natural enemies in China. Great Lakes Entomol 36:191–204
Lutts S, Kinet JM, Bouharmont J (1996) NaCl-induced senescence in leaves of rice (Oriza sativa L.) cultivar differing in salinity resistance. Ann Bot 78:389–398
Ma YH, Ma FW, Zhang JK, Li MJ, Wang YH, Liang D (2008) Effects of high temperature on activities and gene expression of enzymes involved in ascorbate–glutathione cycle in apple leaves. Plant Sci 157:761–766
Maribel L, Dionisio-Sese Satoshi T (1998) Antioxidant responses of rice seedlings to salinity stress. Plant Sci 135:1–9
Meir S, Philosoph-hadas S, Aharoni N (1992) Ethylene-increased accumulation of fluorescent lipid-peroxidation products detected during senescence of parsley by a newly developed method. J Am Soc Hortic Sci 117:128–132
Mirouze M, Paszkowski J (2011) Epigenetic contribution to stress adaptation in plants. Curr Opin Plant Biol 14:267–274
Misra N, Gupta AK (2006) Effect of salinity and different nitrogen sources on the activity of antioxidant enzymes and indole alkaloid content in Catharanthus roseus seedlings. J Plant Physiol 163:11–18
Mittler R, Merquiol E, Hallak-Herr E, Rachmilevitch S, Kaplan A, Cohen M (2001) Living under a “dormant” canopy: a molecular acclimation mechanism of the desert plant Retama raetam. Plant J 25:407–416
Munns R, Tester M (2008) Mechanisms of salinity tolerance. Annu Rev Plant Biol 59:651–681
Nakano Y, Asada K (1987) Purification of ascorbate peroxidase in spinach chloroplasts; its inactivation in ascorbate-depleted medium andreactivation by monodehydroascorbate radical. Plant Cell Physiol 28:131–140
Natcheva R, Cronberg N (2007) Maternal transmission of cytoplasmic DNA in interspecific hybrids of peat mosses, Sphagnum (Bryophyta). J Evol Biol 20:1613–1616
Noctor G, Foyer CH (1998) Ascorbate and glutathione: keeping active oxygen under control. Annu Rev Plant Physiol Plant Mol Biol 49:249–279
Parida AK, Das AB, Mittra B (2003) Effect of NaCl stress on the structure, pigment complex composition, and photosynthetic activity of mangrove Bruguiera parviflora chloroplasts. Photosynthetica 41:191–200
Pnueli L, Hongjian L, Mittler R (2003) Growth suppression, altered stomatal responses, and augmented induction of heat shock proteins in cytosolic ascorbate peroxidase (Apx1)-deficient Arabidopsis plants. Plant J 34:187–203
Porebski S, Bailey LG, Baum BR (1997) Modification of a cTAB DNA extraction protocol for plants containing high polysaccharide and polyphenol components. Plant Mol Biol Rep 15:8–15
Portis E, Acquadro A, Comino C, Lanteri S (2004) Analysis of DNA methylation during germination of peper (Capsicum annuum L) seeds using ethylation-sensitive amplification polymorphism (MSAP). Plant Sci 166(1):169–178
Puckette MC, Weng H, Mahalingam R (2007) Physiological and biochemical responses to acute ozone-induced oxidative stress in Medicago truncatula. Plant Physiol Biochem 45(1):70–79
Queiroz CGS, Alonso A, Mares-Guia M, Magalhaes AC (1998) Chilling-induced changes in membrane fluidity and antioxidant enzyme activities in Coffea arabica L. roots. Biol Plant 41:403–413
Rangwala SH, Richards EJ (2004) The value-added genome: building and maintaining genomic cytosine methylation landscapes. Curr Opin Genet Dev 14:686–691
Rapp RA, Wendel JF (2005) Epigenetics and plant evolution. New Phytol 168:81–91
Reyna-Lopez GE, Simpson J, Ruiz-Herrera J (1997) Differences in DNA methylation patterns are detectable during the dimorphic transition of fungi by amplification of restriction polymorphisms. Mol Gen Genet 253:703–710
Ruiz GL, Cervera MT, Martínez-Zapater JM (2005) DNA methylation increases throughout Arabidopsis development. Planta 222(2):301–306
Salmon A, Ainouche ML, Wendel JF (2005) Genetic and epigenetic consequences of recent hybridization and polyploidy in Spartina (Poaceae). Mol Ecol 14:1163–1175
SarangaY Zamir D, Marani A, Rudich J (1991) Breeding tomatoes for salt tolerance—field-evaluation of Lycopersicon germplasm for yield and dry-matter production. J Am Soc Hortic Sci 116:1067–1071
Shan X, Liu Z, Dong Z, Wang Y, Chen Y, Lin X, Long L, Han F, Dong Y, Liu B (2005) Mobilization of the active MITE transposons mPing and Pong in rice by introgression from wild rice (Zizania latifolia Griseb.). Mol Biol Evol 22:976–990
Shepherd M, Kasem S, Lee DJ, Henry R (2008) Mapping species differences for adventitious rooting in a Corymbia torelliana × Corymbia citriodora subspecies variegata hybrid. Tree Genet Genomes 4(4):715–725
Shivaprasad PV, Dunn RM, Santos BACM, Bassett A, Baulcombe DC (2011) Extraordinary transgressive phenotypes of hybrid tomato are influenced by epigenetics and small silencing RNAs. The EMBO J 31:257–266
Smith I (1985) Stimulation of glutathione synthesis in photorespiring plants by catalase inhibitors. Plant Physiol 79:1044–1047
Song Y, Ji D, Li S, Wang P, Li Q, Xiang F (2012) The dynamic changes of DNA methylation and histone modifications of salt responsive transcription factor genes in soybean. PLoS ONE 7(7):e41274
Springer NM, Robert M (2007) Stupar Allelic variation and heterosis in maize: how do two halves make more than a whole? Genome Res 17:264–275
Tal M, Shannon MC (1983) Salt tolerance in two wild relatives of the cultivated tomato: responses of Lycopersican esculentum, L. cheesmani, L. peruvianum, Solanum pennelli, and F1 hybrids of high salinity. Aust J Plant Physiol 10:109–117
Tavakkoli E, Rengasamy P, McDonald GK (2010) High concentrations of Na+ and Cl− ions in soil solution have simultaneous detrimental effects on growth of faba bean under salinity stress. J Exp Bot 61(15):4449–4459
Tozlu I, Moore GA, Guy CL (2000) Regulation of growth and differential tissue dry mass accumulation by Citrus grandis, Poncirus trifoliata, and their F1 under salinized and non-salinized environments. Aust J Plant Physiol 27:27–33
Tsaftaris AS, Kafka M (1998) Mechanisms of heterosis in crop plants. J Crop Prod 1:95–111
Wang R, Chen S, Deng L, Fritz E, Hüttermann A, Polle A (2007) A Leaf photosynthesis, fluorescence response to salinity and the relevance to chloroplast salt compartmentation and anti-oxidative stress in two poplars. Trees-Struct Funct 21:581–591
Xiong LZ, Xu CG, Shagi-Maroof MA, Zhang Q (1999) Patterns of cytosine methyaltion in an elite rice hybrid and its parental lines, detected by a methylation-sensitive amplification polymorphism technique. Mol Genet Genomics 261:439–446
Xu L, Han L, Huang B (2011) Antioxidant enzyme activities and gene expression in drought-stressed Kentucky bluegrass. J Am Soc Hortic Sci 136:247–255
Yan H, Kikuchi S, Neumann P, Zhang W, Wu Y, Chen F, Jiang J (2010) Genome-wide mapping of cytosine methylation revealed dynamic DNA methylation patterns associated with genes and centromeres in rice. Plant J 63:353–365
Zhou R, Zhao H (2004) Seasonal pattern of antioxidant enzyme system in the roots of perennial forage grasses grown in alpine habitat, related to freezing tolerance. Physiol Plant 121:399–408
Zhu JK (2001) Plant salt tolerance. Trends Plant Sci 6(2):66–71
Zhu JK (2002) Salt and drought stress signal transduction in plants. Annu Rev Plant Biol 53:247–273
Zhu JK (2008) Epigenome sequencing comes of age. Cell 133:395–397
Zhu Z, Chen J, Zheng HL (2012) Physiological and proteomic characterization of salt tolerance in a mangrove plant, Bruguiera gymnorrhiza (L.) Lam. Tree Physiol 32(11):1378–1388
Zlatev ZS, Lidon FC, Ramalho JC, Yordanov IT (2006) Comparison of resistance to drought of three bean cultivars. Biol Plant 50:389–394
Acknowledgments
This work was financially supported by The National Forestry Science and Technology Support Program (2012BAD01B0503), The Innovation Project of State Key Laboratory of Tree Genetics and Breeding (Northeast Forestry University) (2013B04) and the National Natural Science Foundation of China (NO: 31270697) and Harbin Science and Technology Innovation Funds (RC2011QN002051). We also thank the editor and anonymous reviewers for many detailed and helpful comments that improved the quality of this manuscript.
Conflict of interest
The authors declare that they have no conflict of interest.
Data archiving statement
The research performed in the present study did not produce any data on nucleic acid or protein sequences, genetic maps, SNPs or gene expression that could be deposited in the public databases. Data of enzyme assays and physiological parameters is stored at Northeast Forestry University, Harbin (http://www.nefu.edu.cn/).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zeng, FS., Li, LL., Liang, NS. et al. Salt tolerance and alterations in cytosine methylation in the interspecific hybrids of Fraxinus velutina and Fraxinus mandshurica . Euphytica 205, 721–737 (2015). https://doi.org/10.1007/s10681-015-1432-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10681-015-1432-1