Abstract
Knowledge of the spatial ecology and movement of animals contributes to our understanding of intra- and inter-specific interactions and ecosystem dynamics, and can inform conservation actions. Here we assessed the space use and activity levels of a marine predator, the Caribbean reef shark (Carcharhinus perezi), in coastal regions of Eleuthera, The Bahamas over a 60-day period using acoustic telemetry. Of the 14 adult sharks (eight males, six females) tagged with acoustic transmitters (equipped with accelerometer sensor), nine were detected in a 14 km2 gridded receiver array. Male sharks were significantly less likely to be detected over time relative to females. Given post-release survival is typically high in C. perezi, this finding may indicate that males have larger home ranges and may exhibit lower site fidelity compared to females. Patterns of space use indicated C. perezi primarily occupied the outer reef shelf and were rarely detected on the interior of the reef. Shark activity levels (inferred from acceleration profiles) were highest in close proximity to the reef shelf. Our findings indicate C. perezi individuals frequently occupy deeper water habitats, but make forays into reef shelf habitats where high activity levels are likely related to foraging.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Patterns of animal space use and activity have important implications for fundamental ecology by informing species interactions, population and ecosystem dynamics (Tilman and Kareiva 1997; Morales et al. 2010), as well as conservation through management of habitats and human activities (Sutherland 1998; Buchholz 2007). Elasmobranch fishes exhibit diverse patterns in space use and behaviour, playing important ecological roles in marine ecosystems (Ferretti et al. 2010; Schlaff et al. 2014). However, they also face numerous threats to their conservation (Knip et al. 2010; Dulvy et al. 2014). Recent advances in biotelemetry and biologging devices have enabled much deeper insights into the movement, behaviour, and physiology of elasmobranch fishes (Hammerschlag et al. 2011; Hussey et al. 2015). Acceleration sensors are particularly useful for characterizing patterns in activity, as well as bioenergetics (Whitney et al. 2007, 2010; Gleiss et al. 2009, 2010; Wilson et al. 2015; Cooke et al. 2016). When paired with positioning technology, acceleration patterns can inform behavioural ecology and habitat function (Payne et al. 2011; Brownscombe et al. 2017).
The Caribbean reef shark (Carcharhinus perezi) is a medium-bodied requiem shark, found across the tropical and sub-tropical western Atlantic (Rosa et al. 2006). C. perezi forms a considerable proportion of predator biomass in this region (Brooks et al. 2011, 2013), and is integral to the health and vitality of coral reef systems (Rosa et al. 2006; Maljković and Côté 2011). The Bahamas is one nation particularly reliant upon healthy C. perezi populations, due to the significant economic return from shark diving ecotourism (Maljković and Côté 2011; Haas et al. 2017), as well as ecosystem benefits (i.e. top-down control) critical to maintaining ecosystem health (Roff et al. 2016). Despite exhibiting high site fidelity to distinct bioregions (Garla et al. 2006; Chapman et al. 2007), C. perezi individuals are known to associate with multiple habitats such as coral reefs and open-ocean/deepwater (Chapman et al. 2007; Shipley et al. 2017). Although C. perezi is well known to The Bahamas (Brooks et al. 2011, 2013), key space-use patterns are poorly documented compared to other locales such as Brazil (Garla et al. 2006) and Belize (Chapman et al. 2005). Furthermore, there is currently no information pertaining to the activity levels of C. perezi in the scientific literature, nor how activity varies with diel period.
The waters of South Eleuthera, The Bahamas, are characterized by large coral heads and a narrow continental shelf, which runs adjacent to a deep-water inlet of the Atlantic Ocean, the Exuma Sound. The walls of the Sound drop rapidly to approximately 400 m shallow before sloping gradually to roughly 1600 m at its centre (Ball et al. 1969). The unique bathymetry of the Exuma Sound and surrounding neritic waters allow for the examination of elasmobranch behaviour occurring at the interface between shallow and open-ocean/deep-water habitats. As such, we quantified short-term movements and activity patterns of C. perezi in south Eleuthera, The Bahamas, to provide the first indication of key space-use areas, and diel patterns in activity for individuals at this locale.
Methods
Animal capture and tagging
This study was conducted between June and August 2009 in the waters surrounding South Eleuthera, The Bahamas (24.837° N, 76.342° W).
Thirty-two VR2 acoustic receivers (VEMCO, Nova Scotia, Canada) were deployed in a 14km2 grid in the waters off southwest Eleuthera, The Bahamas (24.77° N, 76.21° W). All receivers were placed no further than 500 m apart, in areas were obstruction by large coral heads would not cause significantly variability in detection ranges; however, the use of sentinal tags and range testing were not performed. C. perezi individuals were captured using stationary mid-water longlines equipped with 30–50 gangions baited with bonito (Sarda sarda) (see Brooks et al. 2013 for detailed methodology). Animals were secured alongside the research vessel, and a V9-AP-2 L coded sensor acceleration-pressure sensing transmitter (45 s average delay (range = 30–60 s); 82 day duration; VEMCO Inc., Canada), was attached to the dorsal fin prior to release. Each V9AP was secured to an acrylic mount. Two small holes were then made through the middle of the dorsal fin using a sterile scalpel, and the mount secured either side of the dorsal fin using surgical suture. The entire workup procedure lasted no longer than 15 min per animal. The accelerometer sensor had a 5 Hz sampling frequency and measured acceleration (g) in three axes (x,y,z). Prior to transmission, the route mean square (RMS) was calculated as \( RMS=\sqrt{A_x^2+{A}_y^2+{A}_z^2} \). The mean RMS was calculated for 27 s sampling periods and stored on board prior to transmission. The pressure sensor was rated to a depth of 200 m. The transmitters were programmed to transmit a single pressure sensor reading for every two acceleration readings.
Statistical analysis
Data were analysed using RStudio version 0.99.896 (R Core Team 2013). C. perezi post-release detection probability was modelled with Cox proportional hazards regression with sex and length as predictors and days detected post-release as a response variable. Data were checked for the assumption of proportional hazards prior to analysis. To identify spatial preference across the array, the log-transformed numbers of detections and mean acceleration values were examined for each individual VR2 receiver. A generalized additive mixed effects model was fit to log-transformed shark acceleration with time of day as a predictor, days post-release as a smoother, and individual as a random effect. The model was validated following the protocol outlined in Zuur et al. (2009). After deploying these tags a parallel study on Caribbean reef sharks in this region revealed that they make sporadic excursions into deep-water (> 200 m) (Shipley et al. 2017). Therefore all depth data collected during this study were removed from analyses due to the malfunctioning of integrated depth sensors after all tagged animals exceeded the depth rating of sensors (max depth = 200 m).
Results
Fourteen (eight male, six female) C. perezi individuals encompassing immature and mature individuals (139–217 cm STL) were captured and tagged over the course of this study. Five were not detected after tagging (Table 1). The remaining nine sharks generated a total of 10,862 detections across the array, with days at liberty ranging from 1 to 61 between individuals respectively (Table 1). Only three sharks exhibited significant presence around the array for the entire deployment duration (1,074,441, 1,074,437 and 1,074,447) despite that all sharks were captured and released within the footprint of the array. The time between release and first detection was highly variable between animals (7:20 h to 614:28 h, Table 1), however all animals remained undetected for at least 7 h post-release before being detected.
There was no significant effect of body size on the probability of being detected post release (Cox Proportional Hazards; z = 0.17, p = 0.86). There was a significant effect of sex (z = 2.2, p = 0.03), whereby males were significantly less likely to be detected in the array post-release than females (Fig. 1a). For those sharks that were detected within the array (n = 9), there was a significant effect of diel period on mean acceleration (GAMM; F1,10,860 = 9.2, p < 0.001). C. perezi individuals were more active at night, with activity levels peaking around 12 am (Fig. 1b).
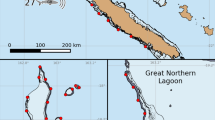
Examining spatial distributions, C. perezi individuals primarily occupied the edge of the Exuma Sound throughout the entire deployment duration and did not move onto the shallower regions of the shelf closer to South Eleuthera (Fig. 2). The majority of detections occurred northwest of Cape Eleuthera in the northeast region of the Exuma Sound, and in the southern region of the array. C. perezi activity levels were generally higher in close proximity to the reef shelf, with high activity in diverse regions along the shelf at night (Fig. 2). Sharks were also active in particular regions along the shelf during the day, with especially high levels of activity in the southern portion of the array where the shelf is close to shore (Fig. 2).
Discussion
Acoustic telemetry with accelerometer transmitters provided novel information regarding space-use and activity of C. perezi in south Eleuthera, The Bahamas. Time to first detection was highly variable between individuals, who all remained absent from the array for at least seven hours post-release. This behaviour could indicate stress-induced behavioural modification (Hoolihan et al. 2011), whereby animals may flee a considerable distance from the sampling area to recover from physical and physiological stress induced during the initial capture and tagging event. Females had a greater detection probability over the deployment duration than males (Fig. 2). C. perezi is known to be resilient to experimental longline capture (Brooks et al. 2012), and previous work has demonstrated low levels of post-release mortality regardless of sex (Shipley et al. 2017). It is therefore most likely that the majority of undetected fish exited the study region. Based on this assumption, male C. perezi could have larger home ranges than females, however rigorously testing this hypothesis this would require quantifying movements across a larger number of individuals and life history stages, and significantly increasing array coverage, which extended beyond the scope of this study. Further, extending sampling across an increased temporal window may elucidate seasonal changes in habitat use, as some individuals may move further offshore, and away from south Eleuthera during summer and fall (Shipley et al. 2017). Despite these caveats, sexual segregation in space-use is known for many elasmobranchs; for example spottail sharks (Carcharhinus sorrah; Knip et al. 2012), blue sharks (Prionace glauca), and shortfin mako sharks (Isurus oxyrinchus; Mucientes et al. 2009). These observations therefore imply that when designing and designating MPAs, the potentially variable home range sizes between male and female C. perezi should be considered. Although alternate tracking methodologies, such as satellite telemetry may offer insight into broad-scale movements and the interaction of animals with national boundaries, finer scale observations are of particular importance when informing current national conservation strategies in the Caribbean. The Caribbean challenge initiative, for example, focuses on the protection of distinct habitat-types, in which participating nations including The Bahamas, are aiming to protect 20% of coastal resources by 2020 (Moultrie 2012). In such cases, an understanding of organismal fine-scale habitat use is paramount to developing the most efficient, and informed placement of future MPAs.
C. perezi individuals were exclusively detected along continental drop-off for the entire deployment duration, and were not detected on receivers located further inshore. This likely represents ontogenetic segregation in habitat-use between differing size-class individuals; whereby smaller individuals reside further inshore, compared to larger sharks, which foray along escarpments and drop-offs (Pikitch et al. 2005; Brooks et al. 2013; Shipley et al. 2017). Specific depth estimates could not be retrieved from the integrated pressure sensors, as individuals moved to depths (max = 200 m), which exceeded the maximum capacity of the sensor. This suggests animals were active well below 200 m; behaviour documented in some C. perezi individuals (Chapman et al. 2007; Shipley et al. 2017). Sharks were commonly detected in two distinct areas along the drop-off of the northeast Exuma Sound. Firstly, a large number of detections were observed in the northern region of the array, as well as a peak in detections on a single receiver in the southern region, near Deals Point. Further, animals did not differentiate space use in relation to time of day, as high space use along the drop-off was homogenous during day and night time periods. These observations are in support of previous findings, as C. perezi individuals tagged at other locales have been observed to associate with highly specific areas of reef crest, and exhibit limited horizontal displacement (Chapman et al. 2005; Brooks et al. 2013). This suggests that patterns of detections observed in this study are largely driven by site fidelity exhibited by a single individual.
Importantly, spatial and temporal variability in detection frequency with acoustic telemetry systems can bias ecological interpretation of data (Kessel et al. 2014). This study did not employ range testing or sentinel tags to track detection range or efficiency, which must be considered when interpreting findings. Detection efficiency is generally lower and more temporally variable (due primarily to tides, wind, reef noise) in shallow water habitats (Gjelland and Hedger 2013; Kessel et al. 2014). We received zero detections of Caribbean reef sharks on the shallow reef region, which regardless of detection efficiency, suggests limited use of this area. These movement patterns are also consistent with data from other locales, such as Belize (e.g. Chapman et al. 2005). Detection efficiency also often decreases in reef habitats at night (Kessel et al. 2014), which may have resulted in reduced detections at night in our study, especially at receivers in close proximity to the reef.
Diel-variability in the activity of C. perezi was observed, as individuals exhibited greater activity during the night compared to the day, with a peak activity occuring around 12 am. Although species- and habitat-specific, some elasmobranch species are known to increase activity during the night in association with increased feeding (Gleiss et al. 2013; Papastamatiou et al. 2015; Barnett et al. 2016). Activity levels were also generally highest in close proximity to the reef shelf, which likely represents important foraging habitat for this species (Chapman et al. 2007). The proximity of the study location to open-ocean, and mesophotic reef habitat adjacent to the drop-off, may provide C. perezi access to a diverse number of prey-rich resource pools, as reef sharks are known to exploit both pelagic and mesophotic habitats to sustain energetic requirements (McCauley et al. 2012; Papastamatiou et al. 2015). C. perezi individuals are also known to perform deep vertical excursions (> 200 m), with a high frequency during the night (Chapman et al. 2007; Shipley et al. 2017), which is supportive of increased nighttime activity of individuals observed in this study. We therefore conclude such observations are likely driven by increased foraging behaviour during this period. Although beyond the scope of this study, 12 am could represent the time of highest lunar intensity, which may dictate periods of high activity, as seen in other reef-associated fishes (Koenig et al. 2017).
Novel insights from accelerometers highlight the distinct movement and activity patterns of C. perezi off South Eleuthera, The Bahamas. Based on our findings, male C. perezi may exhibit larger home ranges than females, but this hypothesis requires further testing. Regardless, such behavior illustrates the inherent complexity of shark behaviour, and may complicate the designation of future MPAs. Fringing coral reefs and continental drop-offs were identified as key habitat-use areas by immature and mature sharks, which may represent key foraging habitats, and may be exploited at higher frequency during the night. These findings provide insight into the spatial and behavioural ecology of C. perezi, which may have important implications for design and placement MPAs, as well as characterizing species interactions and community ecology in coastal marine systems.
References
Ball MM, Harrison CGA, Hurley RJ, Leist CE (1969) Bathymetry in the vicinity of the northeastern scarp of the great Bahama Bank and Exuma sound. Bull Mar Sci 19:243–252
Barnett A, Payne NL, Semmens JM, Fitzpatrick R (2016) Ecotourism increases the field metabolic rate of whitetip reef sharks. Biol Conserv 199:132–136. doi:10.1016/j.biocon.2016.05.009
Brooks EJ, Sloman KA, Sims DW, Danylchuk AJ (2011) Validating the use of baited remote underwater video surveys for assessing the diversity, distribution and abundance of sharks in the Bahamas. Endanger Species Res 13:231–243. doi:10.3354/esr00331
Brooks EJ, Mandelman JW, Sloman KA, Liss S, Danylchuk AJ, Cooke SJ, Skomal GB, Phillip DP, Sims DW, Suski CD (2012) The physiological response of the Caribbean reef shark (Carcharhinus perezi) to longline capture. Comp Biochem Physiol A 162:94–100. doi:10.1016/j.cbpa.2011.04.012
Brooks EJ, Sims DW, Danylchuk AJ, Sloman KA (2013) Seasonal abundance, philopatry and demographic structure of Caribbean reef shark (Carcharhinus perezi) assemblages in the north-east Exuma sound, the Bahamas. Mar Biol 160:2535–2546. doi:10.1007/s00227-013-2246-0
Brownscombe JW, Cooke SJ, Danylchuk AJ (2017) Spatiotemporal drivers of energy expenditure in a coastal marine fish. Oecologia 183:689–699. doi:10.1007/s00442-016-3800-5
Buchholz R (2007) Behavioural biology: an effective and relevant conservation tool. Trends Ecol Evol 22:401–407. doi:10.1016/j.tree.2007.06.002
Chapman DD, Pikitch EK, Babcock E, Shivji MS (2005) Marine reserve design and evaluation using automated acoustic telemetry: a case-study involving coral reef-associated sharks in the Mesoamerican Caribbean. Mar Technol Soc J 39:42–55. doi:10.4031/002533205787521640
Chapman DD, Pikitch EK, Babcock EA, Shivji MS (2007) Deep-diving and diel changes in vertical habitat use by Caribbean reef sharks Carcharhinus perezi. Mar Ecol Prog Ser 344:271–275. doi:10.3354/meps06941
Cooke SJ, Brownscombe JW, Raby GD, Broell F, Hinch SG, Clark TD, Semmens JM (2016) Remote bioenergetics measurements in wild fish: opportunities and challenges. Comp Biochem Physiol A 202:23–37. doi:10.1016/j.cbpa.2016.03.022
Dulvy NK, Fowler SL, Musick JA, Cavanagh RD, Kyne PM, Harrison LR et al (2014) Extinction risk and conservation of the world’s sharks and rays. elife 3:e00590. doi:10.7554/eLife.00590
Ferretti F, Worm B, Britten GL, Heithaus MR, Lotze HK (2010) Patterns and ecosystem consequences of shark declines in the ocean. Ecol Lett 13:1055–1071. doi:10.1111/j.1461-0248.2010.01489.x
Garla RC, Chapman DD, Wetherbee BM, Shivji M (2006) Movement patterns of young Caribbean reef sharks, Carcharhinus perezi, at Fernando de Noronha archipelago, Brazil: the potential of marine protected areas for conservation of a nursery ground. Mar Biol 149:189–199. doi:10.1007/s00227-005-0201-4
Gjelland KØ, Hedger RD (2013) Environmental influence on transmitter detection probability in biotelemetry: developing a general model of acoustic transmission. Methods Ecol Evol 4:665-674
Gleiss AC, Gruber SH, Wilson RP (2009) Multi-channel data-logging: towards determination of behaviour and metabolic rate in free-swimming sharks. In Tagging and tracking of marine animals with electronic devices. Springer, Netherlands, p 211-228.
Gleiss AC, Dale JJ, Holland KN, Wilson RP (2010) Accelerating estimates of activity-specific metabolic rate in fishes: testing the applicability of acceleration data-loggers. J Exp Mar Biol Ecol 385:85-91
Gleiss AC, Wright S, Liebsch N, Wilson RP, Norman B (2013) Contrasting diel patterns in vertical movement and locomotor activity of whale sharks at Ningaloo reef. Mar Biol 160:2981–2992
Haas AR, Fedler T, Brooks EJ (2017) The contemporary economic value of elasmobranchs in the Bahamas: reaping the rewards of 25 years of stewardship and conservation. Biol Conserv 207:55–63. doi:10.1016/j.biocon.2017.01.007
Hammerschlag N, Gallagher AJ, Lazarre DM (2011) A review of shark satellite tagging studies. J Exp Mar Biol Ecol 398:1–8. doi:10.1016/j.jembe.2010.12.012
Hoolihan JP, Luo J, Abascal FJ, Campana SE, De Metrio G, Dewar H et al (2011) Evaluating post-release behaviour modification in large pelagic fish deployed with pop-up satellite archival tags. ICES J Mar Sci 68:880–889. doi:10.1093/icesjms/fsr024
Hussey NE, Kessel ST, Aarestrup K, Cooke SJ, Cowley PD, Fisk AT, Harcourt RG, Holland KN, Iverson SJ, Kocik JF, Flemming JEM, Whoriskey FG (2015) Aquatic animal telemetry: a panoramic window into the underwater world. Science 348:1255642. doi:10.1126/science.1255642
Kessel ST, Cooke SJ, Heupel MR, Hussey NE, Simpfendorfer CA, Vagle S, Fisk AT (2014) A review of detection range testing in aquatic passive acoustic telemetry studies. Rev Fish Biol Fish 24:199–218
Knip DM, Heupel MR, Simpfendorfer CA (2010) Sharks in nearshore environments: models, importance, and consequences. Mar Ecol Prog Ser 402:1–11. doi:10.3354/meps08498
Knip DM, Heupel MR, Simpfendorfer CA (2012) Habitat use and spatial segregation of adult spottail sharks Carcharhinus sorrah in tropical nearshore waters. J Fish Biol 80:767–784. doi:10.1111/j.1095-8649.2012.03223.x
Koenig CC, Bueno LS, Coleman FC, Cusick JA, Ellis RD, Kingon K, Locascio JV, Malinowski C, Murie DJ, Stallings CD (2017) Diel, lunar, and seasonal spawning patterns of the Atlantic goliath grouper, Epinephelus itajara, off Florida, United States. Bull Mar Sci 93:391–406. doi:10.5343/bms.2016.1013
Maljković A, Côté IM (2011) Effects of tourism-related provisioning on the trophic signatures and movement patterns of an apex predator, the Caribbean reef shark. Biol Conserv 144:859–865. doi:10.1016/j.biocon.2010.11.019
McCauley DJ, Young HS, Dunbar RB, Estes JA, Semmens BX, Micheli F (2012) Assessing the effects of large mobile predators on ecosystem connectivity. Ecol Appl 22:1711–1717. doi:10.1890/11-1653.1
Morales JM, Moorcroft PR, Matthiopoulos J, Frair JL, Kie JG, Powell RA et al (2010) Building the bridge between animal movement and population dynamics. Proc R Soc B 365:2289–2301. doi:10.1098/rstb.2010.0082
Moultrie S (2012) Master plan for the Bahamas national protected area system. The Nature Conservancy, Northern Caribbean Office, Nassau
Mucientes GR, Queiroz N, Sousa LL, Tarroso P, Sims DW (2009) Sexual segregation of pelagic sharks and the potential threat from fisheries. Biol Lett rsbl-2008. doi:10.1098/rsbl.2008.0761
Papastamatiou YP, Watanabe YY, Bradley D, Dee LE, Weng K, Lowe CG, Caselle JE (2015) Drivers of daily routines in an ectothermic marine predator: hunt warm, rest warmer? PLoS One 10:e0127807
Payne NL, Gillanders BM, Seymour RS, Webber DM, Snelling EP, Semmens JM (2011) Accelerometry estimates field metabolic rate in giant Australian cuttlefish Sepia Apama during breeding. J Anim Ecol 80:422–430. doi:10.1111/j.1365-2656.2010.01758.x
Pikitch EK, Chapman DD, Babcock EA, Shivji MS (2005) Habitat use and demographic population structure of elasmobranchs at a Caribbean atoll (Glover’s reef, Belize). Mar Ecol Prog Ser 302:187–197. doi:10.3354/meps302187
R Core Team (2013) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna URL http://www.R-project.org/
Roff G, Doropoulos C, Rogers A, Bozec YM, Krueck NC, Aurellado E, Priest M, Birrell C, Mumby PJ (2016) The ecological role of sharks on coral reefs. Tr Ecol Evol 31:395–407. doi:10.1016/j.tree.2016.02.014
Rosa RS, Mancini P, Caldas JP, Graham RT (2006) Carcharhinus perezi. The IUCN Red List of Threatened Species 2006 doi:https://doi.org/10.2305/IUCN.UK.2006.RLTS.T60217A12323052.en
Rollin BE, Kessel ML (1998) Guidelines for the treatment of animals in behavioural research and teaching. Anim Behav 55:251–257
Schlaff AM, Heupel MR, Simpfendorfer CA (2014) Influence of environmental factors on shark and ray movement, behaviour and habitat use: a review. Rev Fish Biol Fish 24:1089–1103. doi:10.1007/s11160-014-9364-8
Shipley ON, Howey LA, Tolentino ER, Jordan LK, Ruppert JL, Brooks EJ (2017) Horizontal and vertical movements of Caribbean reef sharks (Carcharhinus perezi): conservation implications of limited migration in a marine sanctuary. R Soc Open Sci 4:160611. doi:10.1098/rsos.160611
Sutherland WJ (1998) The importance of behavioural studies in conservation biology. Anim Behav 56:801–809. doi:10.1006/anbe.1998.0896
Tilman D, Kareiva PM (1997) Spatial ecology: the role of space in population dynamics and interspecific interactions, vol 30. Princeton University Press, Princeton
Whitney NM, Papastamatiou YP, Holland KN, Lowe CG (2007) Use of an acceleration data logger to measure diel activity patterns in captive whitetip reef sharks, Triaenodon obesus. Aquat Living Resour 20:299–305. doi:10.1051/alr:2008006
Whitney NM, Pratt HL Jr, Pratt TC, Carrier JC (2010) Identifying shark mating behaviour using three-dimensional acceleration loggers. Endanger Species Res 10:71–82. doi:10.3354/esr00247
Wilson AD, Wikelski M, Wilson RP, Cooke SJ (2015) Utility of biological sensor tags in animal conservation. Conserv Biol 29:1065-1075
Zuur A et al (2009) Mixed effects models and extensions in ecology with R. Springer Science & Business Media, New York
Acknowledgements
This project was funded by the Save Our Seas Foundation (SOSF-2009-94). The authors would like to acknowledge the Cape Eleuthera Foundation for providing project funding, and students and staff of the Cape Eleuthera Institute and Island School for invaluable field support. Permission to capture sharks within the Bahamian Shark Sanctuary was established in accordance with Bahamian Department of Marine Resources Form 20A, Regulation 36D (3), permitting fishing, possession, and exportation of sharks or shark parts (permits MAF/FIS/17 and MAF/FIS/34 issued to the Cape Eleuthera Institute). Animal sampling protocols were aligned with The UK Home Office Animals (Scientific Procedures) Act regulations, and guidelines of the Association for the study of Animal Behaviour, and Animal Behaviour Society (Rollin and Kessel 1998).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shipley, O.N., Brownscombe, J.W., Danylchuk, A.J. et al. Fine-scale movement and activity patterns of Caribbean reef sharks (Carcharhinus perezi) in the Bahamas. Environ Biol Fish 101, 1097–1104 (2018). https://doi.org/10.1007/s10641-017-0656-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-017-0656-4