Abstract
The silvertip shark, Carcharhinus albimarginatus, is a coral reef-associated shark with a wide distribution across the Indo-Pacific. Yet, unlike common reef shark species, limited knowledge exists on its movement patterns. Here, we tracked 28 individuals for 4 years with acoustic telemetry in New Caledonia to estimate home range sizes and to investigate individual and seasonal patterns of space use. Comparisons were made with grey reef sharks, C. amblyrhynchos, a closely related but more documented species, tagged on the same acoustic network during the same period. We report similar home range (HR) for both species, with adult males displaying greater HR than females and juveniles. An increased mobility of adult males was observed during the austral summer for the silvertip shark, and the austral winter for the grey reef shark, corresponding to putative mating seasons. Our study brings new insight on the ecology of the silvertip shark and provides essential material to inform targeted conservation measures.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sharp declines in reef shark populations have been observed worldwide in response to human pressure such as fishing and habitat degradation (Robbins et al. 2006; Juhel et al. 2017; MacNeil et al. 2020). This in turn has prompted an increased focus on targeted conservation policies (Chapman et al. 2013; Dulvy et al. 2017; MacKeracher et al. 2019). Efficient protection measures such as Marine Protected Areas (MPAs) implementation require an in-depth understanding of animal behaviour and movement patterns (Green et al. 2015; Di Franco et al. 2018; Dwyer et al. 2020). For instance, estimating the home range of species can help define the minimum MPA size suitable for their protection (Abecasis et al. 2014; Weeks et al. 2017; Krueck et al. 2018).
While the movement patterns of common shark species, such as the grey reef shark (Carcharhinus amblyrhynchos) and the blacktip reef shark (C. melanopterus), have been widely documented (Papastamatiou et al. 2010; Heupel and Simpfendorfer 2014; Espinoza et al. 2015a; Bonnin et al. 2019, 2021), other less commonly observed species, whose conservation status may be of even greater concern (Green et al. 2015), remain poorly described. The main reasons for such lack of data lie in factors such as species lower abundance, avoidance behaviour to sampling (Juhel et al. 2019), large home range, and the associated difficulties in gathering sufficient numbers of individuals.
Among such species, the silvertip shark, C. albimarginatus, is a large-bodied coral reef-associated species distributed throughout the Indo-Pacific (Compagno 1984), but with highly fragmented populations (González-Medina et al. 2015), which increase its vulnerability to overexploitation. Due to significant population decline across its range, this species has been classified as ‘Vulnerable’ by the IUCN Red List of Threatened Species (González-Medina et al. 2015; Ferretti et al. 2018).
Few studies have focused on silvertip shark movement patterns. They showed high levels of residency and site fidelity (Barnett et al. 2012; Espinoza et al. 2015b), and larger space use than common reef shark species (Espinoza et al. 2015c; Carlisle et al. 2019; Tickler 2021). These studies revealed high levels of individual variations in space use and emphasized the need for estimating differences across sexes and ontogeny. Seasonal patterns of silvertip shark activity have been suggested (Espinoza et al. 2015b; Williamson et al. 2021), without, however, revealing any underlying biological mechanism. For instance, in the closely related grey reef shark (Dunn et al. 2020), adult male expand their home range seasonally, presumably for mating purpose, thus with important implications for the species management (Bonnin et al. 2019, 2021).
In this study, 28 silvertip sharks were tracked with acoustic telemetry for 4 years within an array of 55 receivers in New Caledonia, Southwestern Pacific Ocean. Home range sizes were estimated and individual and seasonal patterns of space use were investigated, as were variations across sexes and ontogeny. To assess how the silvertip sharks behaved relative to a closely related but more documented species, comparisons were made with 63 grey reef sharks tracked on the same network and at the same time. Our study provides a better understanding of the ecology of this relatively understudied species and provides valuable insight to inform targeted conservation management measures.
Material and methods
Acoustic array and shark tagging
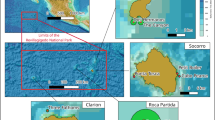
Fifty-five VR2W acoustic receivers (VEMCO Ltd., Halifax, Canada) were deployed from July 2015 to September 2019 in New Caledonia, most densely set in two regions: D’Entrecasteaux atolls (32 receivers) and the Great Northern Lagoon (12). Receivers were also deployed less densely along the west coast of the main land (8) and at the most northeastern corner of the Great Northern Lagoon (3) (Fig. 1). Range tests were performed over 27 receivers of the array and revealed decreasing detection probability with distance, following a sigmoidal curve with 50% of emissions detected at 172 m (Fig. ESM1). Thirty silvertip sharks and 81 grey reef sharks were captured and tagged along the outer slope of barrier reefs of D’Entrecasteaux atolls and the Great Northern Lagoon, at locations where some of the receivers were deployed. Detailed information on tagged individuals is available in Table ESM1. Sharks were caught with barbless circle hooks attached to a floating drum line and baited with large pieces of fish (mostly skipjack and albacore tuna, collected from local fisheries waste). Animals were processed immediately after capture, in a tonic immobility state alongside a small runabout, where total length (TL), sex and maturity stage were determined. Maturity stage was determined for males based on the extension and calcification of the claspers. For females it was extrapolated from total length, using average values of length at maturity for silvertip (Lm = 209 cm, Smart et al. 2017) and grey reef sharks (Lm = 136 cm, Robbins 2006). Sharks were internally fitted with V16 acoustic coded transmitters (68 × 16 mm; frequency: 69 kHz; high power output; VEMCO Ltd., Halifax, Canada), with transmission delay times ranging from 30 to 90 s.
Acoustic array and reef shark sampling in New Caledonia, Southwestern Pacific. Red dots indicate the location of VR2W acoustic receivers, deployed on the outer barrier reef slope. Silvertip and grey reef sharks were sampled and equipped with V16 acoustic transmitter in D’Entrecasteaux atolls (lower left panel) and in the Great Northern Lagoon (lower right panel). Numbers indicate the individual silvertip (S) and grey reef (GR) sharks retained for analyses
Among the 111 tagged individuals, 20 showed no detection after a two-week post-capture period and were therefore discarded from analyses. Analyses were thus performed on 28 silvertip sharks, including 14 juvenile females (total length range: 117–199 cm), 7 juvenile males (119–170 cm), 2 adult females (213–222 cm) and 5 adult males (172–209 cm), and 63 grey reef sharks, including 11 juvenile females (77–128 cm), 9 juvenile males (87–133 cm), 17 adult females (141–169 cm) and 26 adult males (141–169 cm). Monitoring duration ranged from 73 to 1494 days for silvertip sharks (median duration = 1012 days), and from 15 to 1502 days for grey reef sharks (median duration = 898 days). Raw acoustic data were filtered out using the FDA analyser tool from the Vemco VUE software in order to limit false detections resulting from code collisions (Steckenreuter et al. 2015). Individual residency indices (RI) were calculated as the proportion of days one individual was detected at its tagging site receiver.
Two other arrays of acoustic receivers in other parts of the New Caledonian Archipelago, Nouméa reefs (southwest New Caledonia, 12 receivers) and the Chesterfield atoll (8 receivers) were also deployed during the same period but were not included in the analysis as no silvertip sharks were tagged in these regions, and no sharks from D’Entrecasteaux atolls and the Great Northern Lagoon were detected on these arrays (Fig. ESM2).
Analysis of individual home range
Individual utilization distributions (UD) were calculated as one-dimensional convex hulls defined by the portions of outer reef slope delimited by the locations of visited receivers. The 95th and 100th percentile of daily positions were used to determine UD boundaries, then the surface of outer reef slope encompassed by the corresponding reef portions were calculated. This resulted in two different metrics of home range, UD95 and UD100, corresponding to core home range and total home range (Bonnin et al. 2021). Habitat maps from (Andréfouët et al. 2004) were used to define the outer reef slope, the preferred habitat for both species (Compagno 1984; Robbins 2006).
Variations of home range with species, sex and ontogeny were investigated with permutation linear model using the lmp function from the lmPerm R package (R Core Team 2020). The effect of species, sex and maturity stage over UD95 and UD100 was assessed using the Anscombe criterion (Anscombe 1953), which stops sampling when the estimated standard error of the p-value is less than a fraction (here 0.01) of the estimated p-value. If significant interactions were identified, pairwise tests between estimated marginal means of the linear model were then computed with the emmeans R package to identify which groups were significantly different from the others (Lenth et al. 2022). Significant differences between groups were assessed using 1000 permutations of samples, providing distributions of T-ratios associated with each pairwise test and enabling to compute permutational p-values. In order to prevent the false interpretation of significant differences associated with the testing of multiple pairs of marginal means, p-values were then adjusted using the False Discovery Rate approach (FDR; Benjamini and Hochberg 1995) and the p.adjust R function. The potential bias induced by local array geometry was assessed through an alternative model with the tagging site included as an additive factor.
Home range estimates for the different species, sex and maturity stages were provided using estimated marginal means of the linear model and their associated 95% confidence interval.
Analysis of seasonal mobility
To investigate seasonal variations in individual mobility, minimum travelled distance was calculated on a monthly basis as the sum of straightline distances between successively visited acoustic receivers. A permutation linear mixed-effect model was used to investigate variations of mobility associated with species, sex, maturity stage and calendar month. To this purpose, monthly mobility was modelled with individual as a random effect and with the interaction of sex, maturity stage, calendar month and species as fixed effects. The local geometry of the array was not formally included in the model since already accounted for in the individual random effect.
The model was computed with the lme function from the nlme R package (R Core Team 2020), using the Restricted Maximum Likelihood method (REML). Permutational statistics were calculated using 1000 permutations of samples, providing distributions of F-values associated with each factor and enabling us to compute permutational p-values. If significant interactions were identified, pairwise permutation tests between marginal means of the linear mixed-effect model were then used to identify which groups were significantly different from the others, using the same methodology as described in the “Analysis of individual home range” section.
Results and discussion
The 91 tracked individuals were detected a total of 3,310,933 times on the acoustic receivers during the duration of the study, with 78,621 detections of silvertip sharks and 3,232,312 detections of grey reef sharks (Fig. ESM3). Tagging site residency indices (RI) ranged between 0 and 0.81 for silvertip sharks (average RI = 0.2), and between 0 and 0.98 for grey reef sharks (average RI = 0.31). Diel patterns of detections showed that silvertip sharks were mostly detected on the reef during the day, while grey reef sharks were detected during both day and night (ESM4). This suggests that silvertip sharks are moving offshore (Williamson et al. 2021) and/or to deeper waters (Bond et al. 2015) at night.
No silvertip sharks nor grey reef sharks tagged in D’Entrecasteaux atolls or the Great Northern Lagoon were detected on Nouméa reefs or Chesterfield atoll acoustic receivers, located at more than 400 km from the tagging sites, respectively, along both a continuous reef habitat and oceanic habitat (Fig. ESM2). Boussarie et al. (2022) recently reported that deep oceanic areas act as strong barriers to genetic dispersal in grey reef sharks, supporting that direct movement from D’Entrecasteaux or the Great Northern Lagoon to Chesterfield is unlikely. In contrast, in a previous study of 147 tagged grey reef sharks, Bonnin et al. (2019) reported six animals moving up to 340 km from their tagging site along the continuous reef habitat of the main island’s west coast. Given the larger size of silvertip sharks compared to grey reef sharks, long-range movements may be expected, albeit rare, along areas of continuous habitat.
Home range estimates and variability
Permutation linear modelling showed a significant Sex x Maturity stage interaction effect over both home range metrics, UD95 and UD100 (UD95 p-value = 0.028, UD100 p-value = 0.012; Table 1). The model including the tagging site factor showed the same results, with no significant effect of the tagging site (Table ESM2). Pairwise comparisons of estimated marginal means showed that adult males of both species had greater home range values than other groups, with UD95 values significantly different from adult females, and juvenile males and females; while UD100 values significantly differed from adult males to juvenile males (Fig. 2; Table ESM3). Silvertip sharks showed an overall UD95 of 4.88 km2 (95% CI: 1.3–8.5 km2) and an overall UD100 of 7.9 km2 (95% CI: 2.9–12.8 km2), while grey reef sharks showed an overall UD95 of 1,95 km2 (95% CI: 0–4.0 km2) and an overall UD100 of 5.5 km2 (95% CI: 2.7–8.4 km2).
Comparison of home range values of sharks between sexes and maturity stages. Dots represent the estimated marginal means of UD95 and UD100 from the linear models for juvenile and adult males and females, while vertical bars indicate 95% confidence intervals. Significant differences (p < 0.05) between groups are represented with different lower case letter indicators
Interestingly, the permutation linear model showed no significant differences between home range values of the two species, suggesting similar requirements with respect to MPA size. In New Caledonia, MPAs with more than 100 km2 of suitable reef habitats (corresponding to an MPA size generally larger than 3500 km2) are considered suitable to protect the grey reef sharks (Bonnin et al. 2021). Our results suggest that silvertip sharks would benefit from MPA’s of similar size. However, this result has to be interpreted with caution as the home range values reported here are based on a small sample size of mature silvertip sharks, with only five adult males and two adult females, and depend to some extent on the network geometry, making comparison with other studies difficult. A study combining satellite and acoustic tracking in the Chagos Archipelago, Indian Ocean, reported an order of magnitude higher home range values for this species, while also reporting larger space use than the grey reef shark (Carlisle et al. 2019).
Seasonal mobility
Permutation linear mixed-effect modelling showed a significant effect of the quadruple interaction Species x Sex x Maturity x Month on monthly travelled distance (P = 0.004, Table 2). Pairwise comparisons of estimated marginal means further showed a significant change in distance travelled by adult males of both species during two specific months (Fig. 3; Table ESM5). Silvertip shark adult males showed increased mobility during the months of November and December, at the beginning of austral summer (Fig. 3; Table ESM5). Grey reef adult males displayed the same pattern during the months of July and August, in austral winter. For both species, juveniles of both sexes and adult females did not show any significant change in travelled distance in any month of the year.
Effect of species, sex, maturity stage and calendar month on monthly travelled distance. A permutation linear mixed-effect model was fitted to monthly travelled distances with the interaction between species, sex, maturity stage and calendar month as a fixed effect, and individuals as a random effect. Dots represent estimated marginal means of the model, while vertical bars indicate 95% confidence intervals. Within each group defined by species, sex and maturity stage, pairwise permutation tests were performed to assess significant differences of travelled distance between pairs of calendar months. Within each group, significant differences (p < 0.05) between pairs of calendar months are represented with different lower case letter indicators. No significant differences between months were found for juveniles and adult females
Although only five adult male silvertip sharks were considered here to establish this pattern, the similarity with grey reef sharks, albeit with a different timing, suggests that reproduction might be the driver of the seasonal increase in space use. The fact that only adult males were observed to increase their movements during these specific months suggest that this phenomenon is unlikely linked to a response to prey dynamics, which would affect both sexes. Moreover, in grey reef sharks, mating has been reported to start around the month of August in the region (Robbins 2006), thus coinciding with the increase in mobility reported in our study. Evidence of the existence and potential timing of a mating season in silvertip sharks is limited, yet Stevens (1984) suggested it may occur around the month of November in the Aldabra atoll, southern Indian Ocean. Parturition is indicated to occur between December and January in the Indian Ocean, after a gestation period of 12 months, which is consistent with a mating season around this time of the year (Bass et al. 1973; Stevens 1984). The increased mobility of adult silvertip sharks in November–December in our study provides further support for the existence of a summer mating season, and may also explain the reduced detections of tagged silvertip sharks within a receiver array on the Great Barrier Reef between September and January (Espinoza et al. 2015b).Our results also suggest female philopatry and male-biased dispersal for both species, a strategy common to many shark species (Chapman et al. 2015; Portnoy et al. 2015), where males assume the function of dispersing genes across large spatial scales.
While bringing new insight on the movements of the silvertip shark, a poorly documented species, our results also provide valuable information to inform protection measures. Marine Protected Areas sized appropriately for species such as grey reef sharks appear broadly compatible with silvertip sharks, however the higher vulnerability associated with seasonal increases in mobility indicates that this species would benefit from seasonal protection measures, such as fishing gear restrictions (Shiffman and Hammerschlag 2016) or seasonal fishing closures to reduce the risk of bycatch.
References
Abecasis D, Afonso P, Erzini K (2014) Combining multispecies home range and distribution models aids assessment of MPA effectiveness. Mar Ecol Prog Ser 513:155–169
Andréfouët S, Torres-Pulliza D, Dosdane M, Kranenburg C, Murch B (2004) Atlas des récifs coralliens de Nouvelle-Calédonie. IFRECOR Nouvelle-Calédonie IRD Nouméa 26:325
Anscombe FJ (1953) Sequential estimation. J Roy Stat Soc Ser B (Methodol) 15:1–21
Barnett A, Abrantes KG, Seymour J, Fitzpatrick R (2012) Residency and spatial use by reef sharks of an isolated seamount and its implications for conservation. PLoS ONE 7:e36574
Bass AJ, D’Aubrey JD, Kistnasamy N (1973) Sharks of the East Coast of Southern Africa: the genus Carcharhinus (Carcharhinidae) I. Oceanogr Res Inst 33:1–168
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J Roy Stat Soc Ser B (Methodol) 57:289–300
Bond ME, Tolentino E, Mangubhai S, Howey LA (2015) Vertical and horizontal movements of a silvertip shark (Carcharhinus albimarginatus) in the Fijian archipelago. Anim Biotelemetry 3:1
Bonnin L, Robbins WD, Boussarie G, Kiszka JJ, Dagorn L, Mouillot D, Vigliola L (2019) Repeated long-range migrations of adult males in a common Indo-Pacific reef shark. Coral Reefs 38:1121
Bonnin L, Mouillot D, Boussarie G, Robbins WD, Kiszka JJ, Dagorn L, Vigliola L (2021) Recent expansion of marine protected areas matches with home range of grey reef sharks. Sci Rep 11:14221
Boussarie G, Momigliano P, Robbins WD, Bonnin L, Cornu J-F, Fauvelot C, Kiszka JJ, Manel S, Mouillot D, Vigliola L (2022) Identifying barriers to gene flow and hierarchical conservation units from seascape genomics: a modelling framework applied to a marine predator. Ecography 253:e06158
Carlisle AB, Tickler D, Dale JJ, Ferretti F, Curnick DJ, Chapple TK, Schallert RJ, Castleton M, Block BA (2019) Estimating space use of mobile fishes in a large marine protected area with methodological considerations in acoustic array design. Front Mar Sci 6:256
Chapman DD, Frisk MG, Abercrombie DL, Safina C, Gruber SH, Babcock EA, Feldheim KA, Pikitch EK, Ward-Paige C, Davis B, Kessel S, Heithaus M, Worm B (2013) Give shark sanctuaries a chance. Science 339:757–757
Chapman DD, Feldheim KA, Papastamatiou YP, Hueter RE (2015) There and back again: a review of residency and return migrations in sharks, with implications for population structure and management. Ann Rev Mar Sci 7:547–570
Compagno LJ (1984) FAO species catalogue. Sharks of the world. In: An annotated and illustrated catalogue of shark species known to date. Part 2. Carcharhiniformes. FAO, vol 4
Di Franco A, Plass-Johnson JG, Di Lorenzo M, Meola B, Claudet J, Gaines SD, García-Charton JA, Giakoumi S, Grorud-Colvert K, Hackradt CW, Micheli F, Guidetti P (2018) Linking home ranges to protected area size: the case study of the mediterranean sea. Biol Cons 221:175–181
Dulvy NK, Simpfendorfer CA, Davidson LNK, Fordham SV, Bräutigam A, Sant G, Welch DJ (2017) Challenges and priorities in shark and ray conservation. Curr Biol 27:R565–R572
Dunn N, Johri S, Curnick D, Carbone C, Dinsdale E, Chapple T, Block B, Savolainen V (2020) Complete mitochondrial genome of the gray reef shark, Carcharhinus amblyrhynchos (Carcharhiniformes: Carcharhinidae). Mitochondrial DNA 5:2080–2082
Dwyer RG, Krueck NC, Udyawer V, Heupel MR, Chapman D, Pratt HL, Garla R, Simpfendorfer CA (2020) Individual and population benefits of marine reserves for reef sharks. Curr Biol 30:480
Espinoza M, MichelleR H, Tobin AJ, Simpfendorfer CA (2015a) Residency patterns and movements of grey reef sharks (Carcharhinus amblyrhynchos) in semi-isolated coral reef habitats. Mar Biol 162:343–358
Espinoza M, Heupel MR, Tobin AJ, Simpfendorfer CA (2015b) Movement patterns of silvertip sharks (Carcharhinus albimarginatus) on coral reefs. Coral Reefs 34:807–821
Espinoza M, Lédée EJI, Simpfendorfer CA, Tobin AJ, Heupel MR (2015) Contrasting movements and connectivity of reef-associated sharks using acoustic telemetry: implications for management. Ecol Appl 25:2101
Ferretti F, Curnick D, Liu K, Romanov EV, Block BA (2018) Shark baselines and the conservation role of remote coral reef ecosystems. Sci Adv 4:eaaq0333
González-Medina E, Richard Pillans (CSIRO Q, Mario Espinoza (James Cook University Q, Nicholas Dulvy (Simon Fraser University C/ ISSSG (2015) IUCN red list of threatened species: Carcharhinus albimarginatus. IUCN Red List of Threatened Species
Green AL, Maypa AP, Almany GR, Rhodes KL, Weeks R, Abesamis RA, Gleason MG, Mumby PJ, White AT (2015) Larval dispersal and movement patterns of coral reef fishes, and implications for marine reserve network design. Biol Rev 90:1215–1247
Heupel MR, Simpfendorfer CA (2014) Importance of environmental and biological drivers in the presence and space use of a reef-associated shark. Mar Ecol Prog Ser 496:47–57
Juhel J-B, Vigliola L, Mouillot D, Kulbicki M, Letessier TB, Meeuwig JJ, Wantiez L (2017) Reef accessibility impairs the protection of sharks. J Appl Ecol 55:673–683
Juhel J-B, Vigliola L, Wantiez L, Letessier TB, Meeuwig JJ, Mouillot D (2019) Isolation and no-entry marine reserves mitigate anthropogenic impacts on grey reef shark behavior. Sci Rep 9:2897
Krueck NC, Legrand C, Ahmadia GN, Estradivari GA, Jones GP, Riginos C, Treml EA, Mumby PJ (2018) Reserve sizes needed to protect coral reef fishes: reserve sizes to protect coral reef fishes. Conserv Lett 11:e12415
Lenth RV, Buerkner P, Herve M, Love J, Miguez F, Riebl H, Singmann H (2022) emmeans: Estimated marginal means, aka least-squares means. R package version 1.8.2. https://cran.r-project.org/web/packages/emmeans/index.html
MacKeracher T, Diedrich A, Simpfendorfer CA (2019) Sharks, rays and marine protected areas: a critical evaluation of current perspectives. Fish Fish 20:255–267
MacNeil MA, Chapman DD, Heupel M, Simpfendorfer CA, Heithaus M, Meekan M, Harvey E, Goetze J, Kiszka J, Bond ME, Currey-Randall LM, Speed CW, Sherman CS, Rees MJ, Udyawer V, Flowers KI, Clementi G, Valentin-Albanese J, Gorham T, Adam MS, Ali K, Pina-Amargós F, Angulo-Valdés JA, Asher J, Barcia LG, Beaufort O, Benjamin C, Bernard ATF, Berumen ML, Bierwagen S, Bonnema E, Bown RMK, Bradley D, Brooks E, Brown JJ, Buddo D, Burke P, Cáceres C, Cardeñosa D, Carrier JC, Caselle JE, Charloo V, Claverie T, Clua E, Cochran JEM, Cook N, Cramp J, D’Alberto B, de Graaf M, Dornhege M, Estep A, Fanovich L, Farabough NF, Fernando D, Flam AL, Floros C, Fourqurean V, Garla R, Gastrich K, George L, Graham R, Guttridge T, Hardenstine RS, Heck S, Henderson AC, Hertler H, Hueter R, Johnson M, Jupiter S, Kasana D, Kessel ST, Kiilu B, Kirata T, Kuguru B, Kyne F, Langlois T, Lédée EJI, Lindfield S, Luna-Acosta A, Maggs J, Manjaji-Matsumoto BM, Marshall A, Matich P, McCombs E, McLean D, Meggs L, Moore S, Mukherji S, Murray R, Kaimuddin M, Newman SJ, Nogués J, Obota C, O’Shea O, Osuka K, Papastamatiou YP, Perera N, Peterson B, Ponzo A, Prasetyo A, Quamar LMS, Quinlan J, Ruiz-Abierno A, Sala E, Samoilys M, Schärer-Umpierre M, Schlaff A, Simpson N, Smith ANH, Sparks L, Tanna A, Torres R, Travers MJ, van Zinnicq BM, Vigliola L, Ward J, Watts AM, Wen C, Whitman E, Wirsing AJ, Wothke A, Zarza-Gonzâlez E, Cinner JE (2020) Global status and conservation potential of reef sharks. Nature 583:801–806
Papastamatiou YP, Friedlander AM, Caselle JE, Lowe CG (2010) Long-term movement patterns and trophic ecology of blacktip reef sharks (Carcharhinus melanopterus) at Palmyra Atoll. J Exp Mar Biol Ecol 386:94–102
Portnoy DS, Puritz JB, Hollenbeck CM, Gelsleichter J, Chapman D, Gold JR (2015) Selection and sex-biased dispersal in a coastal shark: the influence of philopatry on adaptive variation. Mol Ecol 24:5877–5885
R Core Team (2020) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Robbins WD, Hisano M, Connolly SR, Choat JH (2006) Ongoing collapse of coral-reef shark populations. Curr Biol 16:2314–2319
Robbins WD (2006) Abundance, demography and population structure of the grey reef shark (Carcharhinus amblyrhynchos) and the white tip reef shark (Triaenodon obesus) (Fam. Charcharhinidae). Ph.D. thesis, James Cook University
Shiffman DS, Hammerschlag N (2016) Shark conservation and management policy: a review and primer for non-specialists. Anim Conserv 19:401–412
Smart JJ, Chin A, Baje L, Tobin AJ, Simpfendorfer CA, White WT (2017) Life history of the silvertip shark Carcharhinus albimarginatus from Papua New Guinea. Coral Reefs 36:577–588
Steckenreuter A, Simpfendorfer C, Tattersall K, Hoenner X (2015) Imos animal tracking database review and quality control analysis report. https://imos.org.au/fileadmin/user_upload/shared/AATAMS/IMOS_Animal_Tracking_Database_QC_and_FDAA_Analysis_Report_Oct_2015.pdf
Stevens JD (1984) Life-history and ecology of sharks at Aldabra Atoll, Indian Ocean. Proc R Soc Lond B 222:79–106
Tickler DM (2021) Roles, risks and refugia: spatial ecology, ecosystem function and vulnerability of reef sharks in the central Indian Ocean. Ph. D thesis. The University of Western Australia
Weeks R, Green AL, Joseph E, Peterson N, Terk E (2017) Using reef fish movement to inform marine reserve design. J Appl Ecol 54:145–152
Williamson MJ, Tebbs EJ, Dawson TP, Curnick DJ, Ferretti F, Carlisle AB, Chapple TK, Schallert RJ, Tickler DM, Harrison XA, Block BA, Jacoby DMP (2021) Analysing detection gaps in acoustic telemetry data to infer differential movement patterns in fish. Ecol Evol 11:2717–2730
Acknowledgements
We thank the crew of the research vessel Amborella (Government of New Caledonia). This work is an outcome of the APEX project co-funded by the Total Foundation, the Pew Charitable Trusts and the Government of New Caledonia. Animals were handled according to current legislation of the New Caledonian Government (Permit no. 2015-1351/GNC), the South Province (Permit no. 479-2016/ARR/DENV) and the North Province (Permit no. 60912-1508-2015/JJC). We thank the Millenium Coral Reef Mapping Project for providing shapefiles used for the map fonts.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, L. Bonnin states that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bonnin, L., Boussarie, G., Kiszka, J.J. et al. Individual and seasonal variations of space use in silvertip sharks, Carcharhinus albimarginatus. Coral Reefs 42, 261–269 (2023). https://doi.org/10.1007/s00338-022-02332-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00338-022-02332-w