Summary
Bone morphogenetic protein 2 (BMP2), a pluripotent factor, is a member of the transforming growth factor-beta (TGF-β) superfamily and is implicated in embryonic development and postnatal homeostasis in tissues and organs. Experimental research in the contexts of physiology and pathology has indicated that BMP2 can induce macrophages to differentiate into osteoclasts and accelerate the osteolytic mechanism, aggravating cancer cell bone metastasis. Emerging studies have stressed the potent regulatory effect of BMP2 in cancer cell differentiation, proliferation, survival, and apoptosis. Complicated signaling networks involving multiple regulatory proteins imply the significant biological functions of BMP2 in cancer. In this review, we comprehensively summarized and discussed the current evidence related to the modulation of BMP2 in tumorigenesis and development, including evidence related to the roles and molecular mechanisms of BMP2 in regulating cancer stem cells (CSCs), epithelial-mesenchymal transition (EMT), cancer angiogenesis and the tumor microenvironment (TME). All these findings suggest that BMP2 may be an effective therapeutic target for cancer and a new marker for assessing treatment efficacy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The transforming growth factor β (TGF-β) superfamily is an evolutionarily conserved group of proteins with similar structural characteristics that are considered to appear from the early life history stage of multicellular (metazoan) evolution [1, 2]. The TGF-β superfamily has more than 30 members, including TGF-1, activin, nodal, and bone morphogenetic proteins (BMPs), and they can bind to different receptors, including activin-like kinases (ALKs), TGF-beta receptors (TGFBRs), and BMP receptors (BMPRs) [3]. BMPs were first discovered in 1965 for their ability to cause ectopic bone formation [4]. Subsequently, several studies indicated that BMP signaling is established as an organism develops and plays an important role in early embryogenesis, neural development, and retinal development [5,6,7]. A large amount of recent evidence emphasizes the relationship between cancer and BMP family members, BMP antagonists, and BMP receptors [8,9,10]. In addition, BMPs widely regulate various signaling molecules that may regulate crucial events in tumor growth, metastasis, and tumor angiogenesis via diverse signaling pathways [11, 12]. Based on gene homology, protein structure, and function, BMP members have been further divided into seven subgroups: BMP-2/4, BMP-3/3b, BMP-5/6/7/8/8b, BMP-9/10, BMP-11/growth, BMP-12/13/14 and BMP-15/GDF9 [13,14,15]. In mechanistic analysis, disulfide bonds covalently link two monomers of BMP molecules to form mature molecules that regulate normal physiological development and several diseases [16, 17]. Within the BMP family, BMP2 has been widely studied for its functions in osteogenic differentiation, chondrogenic differentiation, and endochondral ossification in stem cells and mesenchymal tissue [18,19,20]. In oncology research, bone marrow‐derived mesenchymal stem cells (BM-MSCs) are vital precursors of tumor stromal cells [21]. These results suggest that the potential role of BMP2 in cancer development deserves further elucidation [22].
The functions of the other family members have been extensively investigated. The detailed information is shown in Table 1. The function of TGF-β family members is highly associated with the regulation of fibrosis, immune responses, chondrogenesis, osteogenic differentiation, and cancer. Throughout history, cancer has been one of the biggest threats to human health and life and one of the major causes of death [23]. Fortunately, cancer therapy research has made great progress in recent years, providing patients with various new treatments. Molecular targeted therapy has been a real breakthrough in the treatment of cancer patients in recent years. Genes and proteins involved in cancers could be potential targets for the development of molecular targeted therapy. The functions and molecular mechanisms underlying the anticancer potential of BMP2 are included in this review.
There are two types of BMPRs: type I BMP receptors (BMPIRs: BMPRIA, BMPRIB, and ActRI) and type II BMP receptors (BMPIIRs: BMPRII, ActRII, and ActRIIB). Studies have indicated that the expression of BMPRIA, BMPRIB, and BMPRII is related to tumor grade in human prostate cancer tissues [126]. In addition, the protein expression levels of BMPRIA, BMPRIB, and BMPRII were significantly higher in B-cell chronic lymphocytic leukemia (B-CLL) cells than in normal cells. In particular, BMPRIA and BMPRIB were significantly upregulated in B-CLL patients with the advanced-stage disease [127]. BMP or BMPR mutations have been demonstrated in neoplasms in additional studies [128]. Thus, fully addressing the effects of BMPs in cancer is urgently needed. BMP2 is a candidate growth factor that has been approved by the Food and Drug Administration (FDA) for bone and cartilage repair and regeneration [129]. Abnormal activation of BMP2 can be detected in osteoarticular diseases [130]. It is worth noting that BMP2 signaling promotes non-small cell lung cancer bone metastases [131]. Recent studies have also highlighted the possible relationships of BMP2 with malignant cancer transformation, growth, and metastasis. We discuss the relevant findings next.
BMP2 signaling
TGF-β signal transduction is usually divided into SMAD (small mother against decapentaplegic)-dependent classical pathways and nonclassical pathways that are independent of SMAD in most instances [131]. The TGF-β family comprises several members, and we used the TGF-β subfamily as an example to demonstrate the process of the TGF-β/SMAD signaling cascade. The initiation of TGF-β/SMAD signaling is mediated by the binding of TGF-β ligands to its transmembrane TGF-β receptors, which include TGF-β type 1 receptor (TGF-βR1) and type 2 receptor (TGF-βR2) [132]. First, TGF-β ligands binding to TβRII form a heterodimeric that recruits and activates TβRI by phosphorylating specific serine and threonine residues. Then, activated TβRI phosphorylates SMAD proteins (SMAD2 and SMAD3) at C-terminal serine residues and forms phosphorylated heterodimers. Extracellular signals are transmitted intracellularly. SMAD proteins act as the intracellular transducers of TGF-β signals and are grouped into three major classes: receptor-regulated SMADs (R-SMADs, e.g., SMAD1, SMAD2, SMAD3, SMAD5, SMAD8, and SMAD9), common SMADs (Co-SMAD, e.g., SMAD4), and inhibitor SMADs (I-SMAD, e.g., SMAD6, SMAD7) [133]. In the cytosol, SMAD4 is a common binding target for SMAD2/SMAD3, which stabilizes the structure of SMAD2/SMAD3 phosphorylated heterodimers by forming a trimeric complex. Subsequently, the trimeric complex is transported into the nucleus, cooperating with other DNA-binding transcription factors, to regulate target gene expression [134, 135]. Additionally, I-SMADs may inhibit R-SMAD function by competing with SMAD4 for R-SMAD binding [136]. Similar to TGF-β/SMAD signaling, BMPs also activate SMAD proteins by relying on the binding of BMP ligands to type I and II BMP receptors in transmembrane signaling. Notably, the downstream targets of BMP receptors are SMAD1/SMAD5/SMAD8, which form multimers with SMAD4 for transport into the nucleus and regulate the transcription of target genes by BMP responsive elements (BREs) [137, 138] (Fig. 1). The target genes of BMP2/SMADs include some key proteins that induce osteogenic differentiation, angiogenesis, and signaling molecules to regulate other signaling pathways and networks [139, 140]. BMPRIA is the most effective receptor for BMP2 among the three types of I receptors [141]. Studies have shown that BMP2/BMPRIA/SMAD signaling can upregulate the expression of the Wnt inhibitors Dkk1 and Sost in osteoblasts [142]. In addition to canonical SMAD-dependent signaling, several SMAD-independent downstream signaling pathways, including classic mitogen-activated protein kinase (MAPK) pathways and the phosphatidylinositol 3-kinase (PI3K)/Akt pathway [143,144,145], have also been reported.
BMP2 functions and molecular mechanisms in cancer
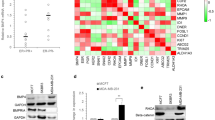
Early studies of BMP2/SMAD signaling mainly focused on the effect of inducing bone and cartilage formation [146, 147]. With the in-depth study of BMP2 and the molecular biological mechanism of tumorigenesis, the role of BMP2 in tumorigenesis has attracted increasing attention. Gene Expression Profiling Interactive Analysis (GEPIA, http://gepia.cancer-pku.cn/index.html) is a web server for cancer and normal gene expression profiling and interactive analyses based on t samples from the TCGA and GTEx databases. Differential expression analysis of BMP2 was performed on cancerous and para-cancerous tissues from the GEPIA database, as presented in Fig. 2, implicating an important role for BMP2 in cancer. Further investigations will be reviewed to clarify the detailed mechanism.
Cancer stem cells (CSCs)
CSCs constitute a special subpopulation in cancer and are inherently present in cancer cell populations. With the ability to self-renew and produce heterogeneous cancer cells, CSCs are key drivers of processes of malignant cancer progression, such as metastasis, chemoresistance, and recurrence [148]. BMP2 can be used as a marker and promote the differentiation of stem cells into chondrogenic and osteogenic tissues [129]. Studies have demonstrated that BMP-2 can decrease the tumorigenicity of CSCs with high aldehyde dehydrogenase activity in the human osteosarcoma cell line OS99-1 by reducing the expression of embryonic stem cell markers (Oct3/4, Nanog, and Sox-2) and inducing the transcription of osteogenic markers (Runx-2 and Collagen Type I) [149, 150]. BMP‑2 also weakened the tumor‑initiating ability of human renal cancer stem cells by initiating bone formation [151]. These results suggest that BMP-2 may provide an opportunity for cancer treatments by inducing differentiation along an osteogenic pathway. Additionally, BMP2 maintains the regulation of stemness features in cancer development. Glioblastoma multiforme (GBM) is characterized by special hypoxic microenvironment centers and partially necrotic cores enriched with stem-like cancer cells that increase the formation of resistant populations. Studies have demonstrated that BMP2 can increase glioblastoma stem-like cell responsiveness to chemotherapy by downmodulating the HIF-1α/MGMT axis [152]. Although intriguing, BMP2 appears to regulate CSCs in a contradictory manner. BMP2 signals also contribute to the emergence of cancer stem cells. In breast cancer, BMP2 induces epithelial-mesenchymal transition (EMT) and stemness through Rb and CD44 [153, 154]. In colon cancer, BMP-2 induces cancer cell metastasis by regulating STAT3-mediated EMT and/or CSC marker expression [155]. In addition, BMP2 signaling activity can be detected in mesenchymal stem cells (MSCs) [156]. Bone MSCs have the ability to promote cancer growth and stem cell niche formation [157]. The regulatory mechanism is summarized in Fig. 3.
EMT
EMT refers to the process by which epithelial cells transform into mesenchymal cells [158]. Physiologically, epithelial cells are normally associated tightly with their adjacent cells via adherent junctions, tight junctions, and desmosomes. They also maintain apical-basal polarity and contact that inhibits their potential to dissociate from the epithelial layer [159]. Although mesenchymal cells are located adjacent to epithelial cells, they are loosely packed and lack polarity and intercellular junctions, which allows them to migrate through the extracellular matrix [160]. Epithelial cells transformed into mesenchymal cells may lose their connection and polarity, change their morphology, and enhance their migration ability, thus gaining invasion and metastasis abilities [161]. Early studies pointed to the fact that BMP2 induces EMT and invasion in colon cancer by activating the PI3K/Akt pathway [145, 162]. Similarly, BMP2 induces the mTORC1 pathway to promote nasopharyngeal carcinoma cell proliferation and invasion [163]. In human pancreatic cancer PANC-1 cells, BMP2 increases EMT-associated protein matrix metalloproteinase-2 (MMP-2) levels through the activation of ROS and ERK signaling pathways [164]. The regulatory mechanism is summarized in Fig. 1.
Angiogenesis
Angiogenesis is essential to maintain the supply of nutrients and oxygen required to support cancer growth. It is difficult for cancers to grow more than 2 mm in diameter without an increased supply of oxygen and nutrients [165]. Studies have indicated that BMP2 expression can be detected in several cancer tissues, and BMP receptors are overexpressed in circulating endothelial progenitor cells (EPCs) and MSCs; all of these factors are involved in cancer angiogenesis [166]. BMP2 overexpression was demonstrated to activate angiogenesis in cancer by inducing the phosphorylation of SMAD 1/5/8 and ERK-1/2. ERK-1/2 activation increased the expression levels of epidermal growth factor (EGF), vascular endothelial growth factor (VEGF), fibroblast growth factor (FGF), and angiogenin, which all mediate cellular responses in endothelial cells. The inhibitor of DNA binding 1 (Id1) protein, which promotes angiogenesis, is the direct downstream effector of SMAD 1/5/8 [167]. In addition, a positive correlation between VEGF and BMP2 has been confirmed in lung cancer patients [168]. In an in vitro experiment, coculture with the hepatocellular carcinoma cell line HepG2 enhanced the angiogenic ability of endothelial cells via the BMP2-mediated MAPK/p38 signaling pathway [169]. Given its pivotal role in cancer angiogenesis, BMP2 has been termed a potential target for the inhibition of cancer angiogenesis. Thus, BMP2 also has an anticancer effect on some natural compounds. For instance, linalool inhibited the angiogenic activity of endothelial cells by activating ERK-mediated BMP2 deregulation [170]. An arabinogalactan from flowers of Panax notoginseng may reduce the migratory activity and tube formation ability of endothelial cells by inducing BMP2/SMAD/Id1 signaling [171]. The heparan sulfate (HS) mimetic WSS25 also inhibits cancer angiogenesis by blocking BMP2/SMAD/Id1 signaling [172]. The regulatory mechanism is summarized in Fig. 1.
Microenvironment
The influence of the TME on tumor cell behavior emphasizes the important relationship between the environment and cancer cell origin, growth, or metastasis. TME factors not only include carcinoma tissue structure, function, and metabolism but also include the intrinsic environment of cancer cells [173]. Overexpression of various growth factors and cytokines can be detected in the TME, and this phenotype is mediated by cancer cells via autocrine, paracrine, and juxtracrine mechanisms [174]. An increase in BMP2 protein levels in diverse TME has been detected, particularly in luminal cancers. Studies have indicated that high levels of BMP2 signaling mediated by the receptor BMPR1B promote the transformation of immature human mammary epithelial cells into luminal cancer-like cells, which could be related to the effect of BMP2 in controlling the maintenance and differentiation of early luminal progenitors [175].
Recent studies have found that several carcinoma-associated mesenchymal stem cells (CA-MSCs) are recruited to and aggregate in the TME and promote cancer growth by increasing the number of CSCs. Notably, on the basis of expressing traditional MSC markers, CA-MSCs have a characteristic expression profile distinguished from MSCs in healthy individuals, and this profile includes BMP2. In a human ovarian carcinoma model in vitro, BMP2 augmented the effects of CA-MSCs on tumorigenesis and cancer stem cells [176]. In the bone microenvironment, BMP2 upregulated the expression of osteogenic markers that facilitated MSC transformation into osteosarcoma with the help of WNT signaling [177]. The underlying mechanism of high BMP2 expression in the TME may be attributed to intratumoural acidosis [178]. The regulatory mechanism is summarized in Fig. 1.
As innate immune cells of the myeloid lineage, macrophages have diverse capacities, including phagocytosis (such as pathogens, cell debris, foreign substances, microbes, and cancer cells), antigen presentation, and immunomodulation [179]. Tumor-associated macrophages (TAMs) are macrophages that infiltrate the TME and are major components of the tumor immune system [180]. Immune function regulation by BMP2 signaling is dependent on the microenvironment. In the inflammatory response, BMP2 is known as a chemoattractant for lymphocytes, monocytes, and macrophages [181]. In liver cancer, high levels of BMP2 can aggravate cancer growth by regulating immune cells in the TME [182]. In turn, several immune cell types are important regulators of BMP2. Studies have indicated that macrophages produce BMP2 in the process of bone healing [183]. A similar mechanism was detected in TAMs. In breast cancer, BMP2 has been demonstrated to be a driving force implicated in breast microcalcification formation; it is secreted by TAMs in the TME but not by the breast cancer cells themselves [184]. Therefore, BMP2 acts as a linker between the microenvironment and carcinogenesis, and cancer patients may benefit from treatments targeting BMP2.
Conclusions and perspective
Among the BMP family members, BMP2 is one of the most heavily studied. BMP2 protein can promote bone formation, similar to other BMPs that are well known as osteogenic growth factors. The biology of BMP2 has recently gained attention in a wide range of research fields, especially in cancer, resulting in the expansion of scientific understanding around this protein. In this review, we discuss the roles of BMP2 in cancer, which may be associated with transcriptional activation and signal transduction. Studies have also suggested that BMP2 plays roles in several characteristic processes that contribute to cancer progressions, such as those related to CSCs, EMT, cancer angiogenesis, and the TME. Moreover, BMP2 mediates multiple signaling pathways, including SMAD-dependent and SMAD-independent pathways, many of which are carcinogenic. The most noteworthy finding is that BMP2 has dual roles in cancer development. On the one hand, BMP2 can inhibit the expansion of several malignant cancer stem cell subpopulations by inducing chondrogenic differentiation, osteogenic differentiation, and endochondral ossification. On the other hand, BMP2 can promote tumorigenesis and development by mediating cancer-related gene regulation and signal activation. Nevertheless, a range of cancer patients may benefit from BMP2 inhibition.
BMP2 and BMP2-associated signaling pathways play a variety of roles in cancer based on current studies in molecular biology and model organisms. It is highlighted that the conflicting data on BMP2 indicate a demand for additional meticulous studies. For example, BMP2 acts as a tumor suppressor that inhibits CSC expansion by reducing the expression of embryonic stem cell markers and inducing the transcription of osteogenic markers in cancer progression. In contrast, BMP2 acts as an oncogene by inducing EMT and CSC formation. BMP2 also augments the effects of CA-MSCs on tumorigenesis and CSCs. In summary, published studies have suggested that BMP2 is essential for tumor development, and paradoxical effects have been observed. A therapeutic strategy aimed at BMP2 can only be developed after the role of BMP2 has been clarified.
References
Huminiecki L, Goldovsky L, Freilich S et al (2009) Emergence, development and diversification of the TGF-beta signalling pathway within the animal kingdom. BMC Evol Biol 9:28
Moustakas A, Heldin CH (2009) The regulation of TGF beta signal transduction. Development 136(22):3699–3714
Wheeler SE, Lee NY (2017) Emerging Roles of Transforming Growth Factor beta Signaling in Diabetic Retinopathy. J Cell Physiol 232(3)486–489
Russow G, Jahn D, Appelt J et al (2018) Anabolic Therapies in Osteoporosis and Bone Regeneration. Int J Mol Sci 20(1)
Begum S (2019) Engineering renal epithelial cells: programming and directed differentiation towards glomerular podocyte's progenitor and mature podocyte. Am J Transl Res 11(2):1102–1115
Shimmi O, Matsuda S, Hatakeyama M (2014) Insights into the molecular mechanisms underlying diversified wing venation among insects. Proc Biol Sci 281(1789):20140264
Wagstaff PE, Heredero BA, Boon C et al (2021) The Role of Small Molecules and Their Effect on the Molecular Mechanisms of Early Retinal Organoid Development. Int J Mol Sci 22(13)
Ihle CL, Straign DM, Provera MD et al (2020) Loss of Myeloid BMPR1a Alters Differentiation and Reduces Mouse Prostate Cancer Growth. Front Oncol 10:357
Lu X, Jin EJ, Cheng X et al (2017) Opposing roles of TGFbeta and BMP signaling in prostate cancer development. Genes Dev 31(23–24):2337–2342
Bonetti J, Corti A, Lerouge L et al (2021) Phenotypic Modulation of Macrophages and Vascular Smooth Muscle Cells in Atherosclerosis-Nitro-Redox Interconnections. Antioxidants (Basel) 10(4)
Han O, Pak B, Jin SW (2021) The Role of BMP Signaling in Endothelial Heterogeneity. Front Cell Dev Biol 9:673396
Nakashima K, Yanagisawa M, Arakawa H et al (1999) Synergistic signaling in fetal brain by STAT3-Smad1 complex bridged by p300. Science 284(5413)479–482
Reddi AH (2005) BMPs: from bone morphogenetic proteins to body morphogenetic proteins. Cytokine Growth Factor Rev 16(3):249–250
Wagner DO, Sieber C, Bhushan R et al (2010) BMPs: from bone to body morphogenetic proteins. Sci Signal 3(107):r1
Wang EA, Rosen V, Cordes P et al (1988) Purification and characterization of other distinct bone-inducing factors. Proc Natl Acad Sci USA 85(24):9484–9488
Bessa PC, Casal M, Reis RL (2008) Bone morphogenetic proteins in tissue engineering: the road from the laboratory to the clinic, part I (basic concepts). J Tissue Eng Regen Med 2(1):1–13
Bessa PC, Casal M, Reis RL (2008) Bone morphogenetic proteins in tissue engineering: the road from laboratory to clinic, part II (BMP delivery). J Tissue Eng Regen Med 2(2–3):81–96
Jia Q, Xu B, Zhang Y et al (2021) CCN Family Proteins in Cancer: Insight Into Their Structures and Coordination Role in Tumor Microenvironment. Front Genet 12:649387
Damiati LA, El-Messeiry S (2021) An Overview of RNA-Based Scaffolds for Osteogenesis. Front Mol Biosci 8:682581
Krajewska-Wlodarczyk M, Owczarczyk-Saczonek A, Placek W et al (2017) Role of Stem Cells in Pathophysiology and Therapy of Spondyloarthropathies-New Therapeutic Possibilities. Int J Mol Sci 19(1)
Tsai CL, Tsai CN, Lin CY et al (2012) Secreted stress-induced phosphoprotein 1 activates the ALK2-SMAD signaling pathways and promotes cell proliferation of ovarian cancer cells. Cell Rep 2(2):283–293
Ishay-Ronen D, Diepenbruck M, Kalathur R et al (2019) Gain Fat-Lose Metastasis: Converting Invasive Breast Cancer Cells into Adipocytes Inhibits Cancer Metastasis. Cancer Cell 35(1):17–32
Yu J, Wang Q, Zhang X et al (2021) Mechanisms of Neoantigen-Targeted Induction of Pyroptosis and Ferroptosis: From Basic Research to Clinical Applications. Front Oncol 11:685377
Kim KK, Sheppard D, Chapman HA (2018) TGF-beta1 Signaling and Tissue Fibrosis. Cold Spring Harb Perspect Biol 10(4)
Taylor RA, Chang CF, Goods BA et al (2017) TGF-beta1 modulates microglial phenotype and promotes recovery after intracerebral hemorrhage. J Clin Invest 127(1)280–292
Delaney K, Kasprzycka P, Ciemerych MA et al (2017) The role of TGF-beta1 during skeletal muscle regeneration. Cell Biol Int 41(7)706–715
Hadaschik EN, Enk AH (2015) TGF-beta1-induced regulatory T cells. Hum Immunol 76(8)561–564
Yu SN, Miao YY, Zhang BT et al (2021) MicroRNA-1269a promotes the occurrence and progression of osteosarcoma by inhibiting TGF-beta1 expression. Eur Rev Med Pharmacol Sci 25(7):2824
Fezza M, Moussa M, Aoun R et al (2019) DKK1 promotes hepatocellular carcinoma inflammation, migration and invasion: Implication of TGF-beta1. PLoS ONE 14(9):e223252
Zhang M, Meng QC, Yang XF et al (2020) TGF-beta1/WISP1/Integrin-alpha interaction mediates human chondrocytes dedifferentiation. Eur Rev Med Pharmacol Sci 24(17):8675–8684
Yu H, Ma S, Sun L et al (2019) TGFbeta1 upregulates the expression of lncRNAATB to promote atherosclerosis. Mol Med Rep 19(5)4222–4228
Gomes FC, Sousa VO, Romao L (2005) Emerging roles for TGF-beta1 in nervous system development. Int J Dev Neurosci 23(5):413–424
Rosenkranz S (2004) TGF-beta1 and angiotensin networking in cardiac remodeling. Cardiovasc Res 63(3):423–432
Caraci F, Spampinato S, Sortino MA et al (2012) Dysfunction of TGF-beta1 signaling in Alzheimer's disease: perspectives for neuroprotection. Cell Tissue Res 347(1):291–301
Sun F, Yu PF, Wang D et al (2019) MicroRNA-488 regulates diabetic nephropathy via TGF-beta1 pathway. Eur Rev Med Pharmacol Sci 23(10):4333–4340
Manuyakorn W, Kamchaisatian W, Atamasirikul K et al (2008) Serum TGF-beta1 in atopic asthma. Asian Pac J Allergy Immunol 26:185–189
Takahashi H, Alves C, Stanford KI et al (2019) TGF-beta2 is an exercise-induced adipokine that regulates glucose and fatty acid metabolism. Nat Metab 1(2):291–303
Dropmann A, Dooley S, Dewidar B et al (2020) TGF-beta2 silencing to target biliary-derived liver diseases. Gut 69(9)1677–1690
Futakuchi A, Inoue T, Wei FY et al (2018) YAP/TAZ Are Essential for TGF-beta2-Mediated Conjunctival Fibrosis. Invest Ophthalmol Vis Sci 59(7):3069–3078
Zhang C, Zhang X, Xu R et al (2017) TGF-beta2 initiates autophagy via Smad and non-Smad pathway to promote glioma cells’ invasion. J Exp Clin Cancer Res 36(1):162
Hau P, Jachimczak P, Schlaier J et al (2011) TGF-beta2 signaling in high-grade gliomas. Curr Pharm Biotechnol 12(12):2150–2157
Hibino T, Nishiyama T (2004) Role of TGF-beta2 in the human hair cycle. J Dermatol Sci 35(1):9–18
Lv Y, Zhang Z, Xing X et al (2020) lncRNA TGFbeta2-AS1 promotes ECM production via TGF-beta2 in human trabecular meshwork cells. Biochem Biophys Res Commun 527(4):881–888
Balzar S, Chu HW, Silkoff P et al (2005) Increased TGF-beta2 in severe asthma with eosinophilia. J Allergy Clin Immunol 115(1):110–117
Duan M, Wang Q, Liu Y et al (2021) The role of TGF-beta2 in cartilage development and diseases. Bone Joint Res 10(8):474–487
Tian L, Sun S, Li W et al (2020) Down-regulated microRNA-141 facilitates osteoblast activity and inhibits osteoclast activity to ameliorate osteonecrosis of the femoral head via up-regulating TGF-beta2. Cell Cycle 19(7):772–786
Smith P, Mosiello G, Deluca L et al (1999) TGF-beta2 activates proliferative scar fibroblasts. J Surg Res 82(2):319–323
Gottanka J, Chan D, Eichhorn M et al (2004) Effects of TGF-beta2 in perfused human eyes. Invest Ophthalmol Vis Sci 45(1):153–158
Xue L, Xiong C, Li J et al (2020) miR-200–3p suppresses cell proliferation and reduces apoptosis in diabetic retinopathy via blocking the TGF-beta2/Smad pathway. Biosci Rep 40(11)
Okamura T, Morita K, Iwasaki Y et al (2015) Role of TGF-beta3 in the regulation of immune responses. Clin Exp Rheumatol 33(4 Suppl 92):S63-S69
Laverty HG, Wakefield LM, Occleston NL et al (2009) TGF-beta3 and cancer: a review. Cytokine Growth Factor Rev 20(4):305–317
Yang Y, Chen L, Si J et al (2020) TGF-beta3/Smad3 Contributes to Isoflurane Postconditioning Against Cerebral Ischemia-Reperfusion Injury by Upregulating MEF2C. Cell Mol Neurobiol 40(8):1353–1365
Tang QO, Shakib K, Heliotis M et al (2009) TGF-beta3: A potential biological therapy for enhancing chondrogenesis. Expert Opin Biol Ther 9(6):689–701
Li Y, Qiao Z, Yu F et al (2019) Transforming Growth Factor-beta3/Chitosan Sponge (TGF-beta3/CS) Facilitates Osteogenic Differentiation of Human Periodontal Ligament Stem Cells. Int J Mol Sci 20(20)
Herath CB, Yamashita M, Watanabe G et al (2001) Regulation of follicle-stimulating hormone secretion by estradiol and dimeric inhibins in the infantile female rat. Biol Reprod 65(6):1623–1633
Ball EM, Mellor SL, Risbridger GP (2004) Cancer progression: is inhibin alpha from Venus or Mars? Cytokine Growth Factor Rev 15(5):291–296
Kim YI, Park SW, Kwon HS et al (2017) Inhibin-alpha gene mutations and mRNA levels in human lymphoid and myeloid leukemia cells. Int J Oncol 50(4):1403–1412
Schmitt JF, Millar DS, Pedersen JS et al (2002) Hypermethylation of the inhibin alpha-subunit gene in prostate carcinoma. Mol Endocrinol 16(2)213–220
Huang M, Cheng YL, Zeng JT et al (2018) Inhibin alpha-subunit inhibits BMP9-induced osteogenic differentiation through blocking BMP/Smad signal and activating NF-kappaB signal in mesenchymal stem cells. J Cell Biochem 119(10):8271–8281
He Z, Liang J, Wang B (2021) Inhibin, beta A regulates the transforming growth factor-beta pathway to promote malignant biological behaviour in colorectal cancer. Cell Biochem Funct 39(2)258–266
Kleeff J, Ishiwata T, Friess H et al (1998) Concomitant over-expression of activin/inhibin beta subunits and their receptors in human pancreatic cancer. Int J Cancer 77(6)860–868
Sawchenko PE, Plotsky PM, Pfeiffer SW et al (1988) Inhibin beta in central neural pathways involved in the control of oxytocin secretion. Nature 334(6183):615–617
Sjoholm K, Palming J, Lystig TC et al (2006) The expression of inhibin beta B is high in human adipocytes, reduced by weight loss, and correlates to factors implicated in metabolic disease. Biochem Biophys Res Commun 344(4)1308–1314
Cross JC, Baczyk D, Dobric N et al (2003) Genes, development and evolution of the placenta. Placenta 24(2–3):123–130
Luo L, Ye G, Nadeem L et al (2012) MicroRNA-378a-5p promotes trophoblast cell survival, migration and invasion by targeting Nodal. J Cell Sci 125(13)3124–3132
Sharma M, McFarlane C, Kambadur R et al (2015) Myostatin: expanding horizons. IUBMB Life 67(8):589–600
Wei Q, Holle A, Li J et al (2020) BMP-2 Signaling and Mechanotransduction Synergize to Drive Osteogenic Differentiation via YAP/TAZ. Adv Sci (Weinh) 7(15):1902931
Park SY, Kim KH, Kim S et al (2019) BMP-2 Gene Delivery-Based Bone Regeneration in Dentistry. Pharmaceutics 11(8)
Skovrlj B, Koehler SM, Anderson PA et al (2015) Association Between BMP-2 and Carcinogenicity. Spine (Phila Pa 1976) 40(23):1862–1871
Matzelle MM, Shaw AT, Baum R et al (2016) Inflammation in arthritis induces expression of BMP3, an inhibitor of bone formation. Scand J Rheumatol 45(5):379–383
Gamer LW, Cox K, Carlo JM et al (2009) Overexpression of BMP3 in the developing skeleton alters endochondral bone formation resulting in spontaneous rib fractures. Dev Dyn 238(9):2374–2381
Yu X, Gu P, Huang Z et al (2017) Reduced expression of BMP3 contributes to the development of pulmonary fibrosis and predicts the unfavorable prognosis in IIP patients. Oncotarget 8(46):80531–80544
Kisiel JB, Li J, Zou H et al (2013) Methylated Bone Morphogenetic Protein 3 (BMP3) Gene: Evaluation of Tumor Suppressor Function and Biomarker Potential in Biliary Cancer. J Mol Biomark Diagn 4(145):1000145
Kawabata T, Otsuka T, Fujita K et al (2017) Suppression by HSP90 inhibitors of BMP4stimulated osteoprotegerin synthesis in osteoblasts: Attenuation of p70 S6 kinase. Mol Med Rep 16(6)8507–8512
Yokoyama Y, Watanabe T, Tamura Y et al (2017) Autocrine BMP-4 Signaling Is a Therapeutic Target in Colorectal Cancer. Cancer Res 77(15)4026–4038
Son JW, Jang EH, Kim MK et al (2011) Serum BMP-4 levels in relation to arterial stiffness and carotid atherosclerosis in patients with Type 2 diabetes. Biomark Med 5(6)827–835
Mailhot G, Yang M, Mason-Savas A et al (2008) BMP-5 expression increases during chondrocyte differentiation in vivo and in vitro and promotes proliferation and cartilage matrix synthesis in primary chondrocyte cultures. J Cell Physiol 214(1):56–64
Beck HN, Drahushuk K, Jacoby DB et al (2001) Bone morphogenetic protein-5 (BMP-5) promotes dendritic growth in cultured sympathetic neurons. BMC Neurosci 2:12
Bramlage CP, Muller GA, Tampe B et al (2011) The role of bone morphogenetic protein-5 (BMP-5) in human nephrosclerosis. J Nephrol 24(5):647–655
Gruber R, Graninger W, Bobacz K et al (2003) BMP-6-induced osteogenic differentiation of mesenchymal cell lines is not modulated by sex steroids and resveratrol. Cytokine 23(4–5):133–137
Verhamme FM, De Smet EG, Van Hooste W et al (2019) Bone morphogenetic protein 6 (BMP-6) modulates lung function, pulmonary iron levels and cigarette smoke-induced inflammation. Mucosal Immunol 12(2):340–351
Hu F, Zhang Y, Li M et al (2016) BMP-6 inhibits the metastasis of MDA-MB-231 breast cancer cells by regulating MMP-1 expression. Oncol Rep 35(3):1823–1830
Toma K, Otsuka F, Oguni K et al (2016) BMP-6 modulates somatostatin effects on luteinizing hormone production by gonadrotrope cells. Peptides 76:96–101
Liu L, Wang Y, Yan R et al (2019) BMP-7 inhibits renal fibrosis in diabetic nephropathy via miR-21 downregulation. Life Sci 238:116957
Aluganti NC, Singla DK (2020) The Role of Bone Morphogenetic Protein 7 (BMP-7) in Inflammation in Heart Diseases. Cells 9(2)
Mathavan N, Raina DB, Tagil M et al (2020) Longitudinal in vivo monitoring of callus remodeling in BMP-7- and Zoledronate-treated fractures. J Orthop Res 38(9):1905–1913
Tasli PN, Aydin S, Yalvac ME et al (2014) Bmp 2 and bmp 7 induce odonto- and osteogenesis of human tooth germ stem cells. Appl Biochem Biotechnol 172(6):3016–3025
Aoki M, Ishigami S, Uenosono Y et al (2011) Expression of BMP-7 in human gastric cancer and its clinical significance. Br J Cancer 104(4):714–718
Manson SR, Austin PF, Guo Q et al (2015) BMP-7 Signaling and its Critical Roles in Kidney Development, the Responses to Renal Injury, and Chronic Kidney Disease. Vitam Horm 99:91–144
Urisarri A, Gonzalez-Garcia I, Estevez-Salguero A et al (2021) BMP8 and activated brown adipose tissue in human newborns. Nat Commun 12(1):5274
Wang XJ, Lian TY, Jiang X et al (2019) Germline BMP9 mutation causes idiopathic pulmonary arterial hypertension. Eur Respir J 53(3)
Tang N, Rao S, Ying Y et al (2020) New insights into BMP9 signaling in organ fibrosis. Eur J Pharmacol 882:173291
Viallard C, Audiger C, Popovic N et al (2020) BMP9 signaling promotes the normalization of tumor blood vessels. Oncogene 39(14):2996–3014
Xiao H, Wang X, Wang C et al (2020) BMP9 exhibits dual and coupled roles in inducing osteogenic and angiogenic differentiation of mesenchymal stem cells. Biosci Rep 40(6)
Neuhaus H, Rosen V, Thies RS (1999) Heart specific expression of mouse BMP-10 a novel member of the TGF-beta superfamily. Mech Dev 80(2):181–184
Ye L, Bokobza S, Li J et al (2010) Bone morphogenetic protein-10 (BMP-10) inhibits aggressiveness of breast cancer cells and correlates with poor prognosis in breast cancer. Cancer Sci 101(10):2137–2144
Karkera JD, Lee JS, Roessler E et al (2007) Loss-of-function mutations in growth differentiation factor-1 (GDF1) are associated with congenital heart defects in humans. Am J Hum Genet 81(5):987–994
Li Q, Ling Y, Yu L (2012) GDF3 inhibits the growth of breast cancer cells and promotes the apoptosis induced by Taxol. J Cancer Res Clin Oncol 138(6):1073–1079
Andersson O, Bertolino P, Ibanez CF (2007) Distinct and cooperative roles of mammalian Vg1 homologs GDF1 and GDF3 during early embryonic development. Dev Biol 311(2):500–511
Mounier R, Chazaud B (2017) [PPARgamma transcription factor controls in anti-inflammatory macrophages the expression of GDF3 that stimulates myogenic cell fusion during skeletal muscle regeneration]. Med Sci (Paris) 33(5):466–469
Li X, Zheng Y, Zheng Y et al (2018) Circular RNA CDR1as regulates osteoblastic differentiation of periodontal ligament stem cells via the miR-7/GDF5/SMAD and p38 MAPK signaling pathway. Stem Cell Res Ther 9(1):232
Zhang W, Wu X, Pei Z et al (2019) GDF5 Promotes White Adipose Tissue Thermogenesis via p38 MAPK Signaling Pathway. DNA Cell Biol 38(11):1303–1312
Lettre G (2017) The osteoarthritis and height GDF5 locus yields its secrets. Nat Genet 49(8):1165–1166
Sullivan AM, O'Keeffe GW (2005) The role of growth/differentiation factor 5 (GDF5) in the induction and survival of midbrain dopaminergic neurones: relevance to Parkinson's disease treatment. J Anat 207(3):219–226
Clendenning DE, Mortlock DP (2012) The BMP ligand Gdf6 prevents differentiation of coronal suture mesenchyme in early cranial development. PLoS ONE 7(5):e36789
Asai-Coakwell M, French CR, Berry KM et al (2007) GDF6, a novel locus for a spectrum of ocular developmental anomalies. Am J Hum Genet 80(2):306–315
Becker J, May A, Gerges C et al (2016) The Barrett-associated variants at GDF7 and TBX5 also increase esophageal adenocarcinoma risk. Cancer Med 5(5):888–891
Zhou Y, Liu S, Wang W et al (2021) The miR-204-5p/FOXC1/GDF7 axis regulates the osteogenic differentiation of human adipose-derived stem cells via the AKT and p38 signalling pathways. Stem Cell Res Ther 12(1):64
Belli M, Shimasaki S (2018) Molecular Aspects and Clinical Relevance of GDF9 and BMP15 in Ovarian Function. Vitam Horm 107:317–348
Li S, Nie EH, Yin Y et al (2015) GDF10 is a signal for axonal sprouting and functional recovery after stroke. Nat Neurosci 18(12):1737–1745
Zhou T, Yu L, Huang J et al (2019) GDF10 inhibits proliferation and epithelial-mesenchymal transition in triple-negative breast cancer via upregulation of Smad7. Aging (Albany NY) 11(10):3298–3314
Rochette L, Mazini L, Meloux A et al (2020) Anti-Aging Effects of GDF11 on Skin. Int J Mol Sci 21(7)
Simoni-Nieves A, Gerardo-Ramirez M, Pedraza-Vazquez G et al (2019) GDF11 Implications in Cancer Biology and Metabolism. Facts and Controversies. Front Oncol 9:1039
Zhang YH, Pan LH, Pang Y et al (2018) GDF11/BMP11 as a novel tumor marker for liver cancer. Exp Ther Med 15(4):3495–3500
Rochette L, Malka G (2019) Neuroprotective Potential of GDF11: Myth or Reality?" Int J Mol Sci 20(14)
Emmerson PJ, Duffin KL, Chintharlapalli S et al (2018) GDF15 and Growth Control. Front Physiol 9:1712
Baek SJ, Eling T (2019) Growth differentiation factor 15 (GDF15): A survival protein with therapeutic potential in metabolic diseases. Pharmacol Ther 198:46–58
Zhang Y, Jiang M, Nouraie M et al (2019) GDF15 is an epithelial-derived biomarker of idiopathic pulmonary fibrosis. Am J Physiol Lung Cell Mol Physiol 317(4):L510-L521
Ahmed DS, Isnard S, Lin J et al (2021) GDF15/GFRAL Pathway as a Metabolic Signature for Cachexia in Patients with Cancer. J Cancer 12(4):1125–1132
Li C, Wang J, Kong J et al (2016) GDF15 promotes EMT and metastasis in colorectal cancer. Oncotarget 7(1):860–872
Liu H, Huang Y, Lyu Y et al (2021) GDF15 as a biomarker of ageing. Exp Gerontol 146:111228
Dewailly D, Laven J (2019) AMH as the primary marker for fertility. Eur J Endocrinol 181(6):D45-D51
Saijoh Y, Adachi H, Mochida K et al (1999) Distinct transcriptional regulatory mechanisms underlie left-right asymmetric expression of lefty-1 and lefty-2. Genes Dev 13(3):259–269
Alowayed N, Salker MS, Zeng N et al (2016) LEFTY2 Controls Migration of Human Endometrial Cancer Cells via Focal Adhesion Kinase Activity (FAK) and miRNA-200a. Cell Physiol Biochem 39(3):815–826
Zabala M, Lobo NA, Antony J et al (2020) LEFTY1 Is a Dual-SMAD Inhibitor that Promotes Mammary Progenitor Growth and Tumorigenesis. Cell Stem Cell 27(2):284–299
Yang S, Zhong C, Frenkel B et al (2005) Diverse biological effect and Smad signaling of bone morphogenetic protein 7 in prostate tumor cells. Cancer Res 65(13):5769–5777
Dzietczenia J, Wrobel T, Jazwiec B et al (2010) Expression of bone morphogenetic proteins (BMPs) receptors in patients with B-cell chronic lymphocytic leukemia (B-CLL). Int J Lab Hematol 32(6 Pt 1):e217-e221
Tang N, Song WX, Luo J et al (2008) Osteosarcoma development and stem cell differentiation. Clin Orthop Relat Res 466(9):2114–2130
Zhou N, Li Q, Lin X et al (2016) BMP2 induces chondrogenic differentiation, osteogenic differentiation and endochondral ossification in stem cells. Cell Tissue Res 366(1):101–111
Qi D, Tian X, Wang Y et al (2019) BMP2 variants in the risk of ankylosing spondylitis. J Cell Biochem
Huang F, Cao Y, Wu G et al (2020) BMP2 signalling activation enhances bone metastases of non-small cell lung cancer. J Cell Mol Med 24(18):10768–10784
Xu F, Liu C, Zhou D et al (2016) TGF-beta/SMAD Pathway and Its Regulation in Hepatic Fibrosis. J Histochem Cytochem 64(3):157–167
Bobik A (2006) Transforming growth factor-betas and vascular disorders. Arterioscler Thromb Vasc Biol 26(8):1712–1720
Shi Y, Massague J (2003) Mechanisms of TGF-beta signaling from cell membrane to the nucleus. Cell 113(6):685–700
Massague J, Seoane J, Wotton D (2005) Smad transcription factors. Genes Dev 19(23):2783–2810
Miyazawa K, Miyazono K (2017) Regulation of TGF-beta Family Signaling by Inhibitory Smads. Cold Spring Harb Perspect Biol 9(3)
Chen G, Deng C, Li YP (2012) TGF-beta and BMP signaling in osteoblast differentiation and bone formation. Int J Biol Sci 8(2):272–288
Abe E (2006) Function of BMPs and BMP antagonists in adult bone. Ann N Y Acad Sci 1068:41–53
Javed A, Bae JS, Afzal F et al (2008) Structural coupling of Smad and Runx2 for execution of the BMP2 osteogenic signal. J Biol Chem 283(13):8412–8422
Chen X, Xiao F, Wang Y et al (2012) Structure-activity relationship study of WSS25 derivatives with anti-angiogenesis effects. Glycoconj J 29(5–6)389–398
Keller S, Nickel J, Zhang JL et al (2004) Molecular recognition of BMP-2 and BMP receptor IA. Nat Struct Mol Biol 11(5):481–488
Kamiya N, Kobayashi T, Mochida Y et al (2010) Wnt inhibitors Dkk1 and Sost are downstream targets of BMP signaling through the type IA receptor (BMPRIA) in osteoblasts. J Bone Miner Res 25(2):200–210
Wang J, Guo J, Liu J et al (2014) BMP-functionalised coatings to promote osteogenesis for orthopaedic implants. Int J Mol Sci 15(6):10150–10168
Kiwanuka E, Junker JP, Eriksson E (2017) Transforming growth factor beta1 regulates the expression of CCN2 in human keratinocytes via Smad-ERK signalling. Int Wound J 14(6):1006–1018
Kang MH, Kim JS, Seo JE et al (2010) BMP2 accelerates the motility and invasiveness of gastric cancer cells via activation of the phosphatidylinositol 3-kinase (PI3K)/Akt pathway. Exp Cell Res 316(1):24–37
Noth U, Tuli R, Seghatoleslami R et al (2003) Activation of p38 and Smads mediates BMP-2 effects on human trabecular bone-derived osteoblasts. Exp Cell Res 291(1):201–211
Gersbach CA, Guldberg RE, Garcia AJ (2007) In vitro and in vivo osteoblastic differentiation of BMP-2- and Runx2-engineered skeletal myoblasts. J Cell Biochem 100(5):1324–1336
Keyvani-Ghamsari S, Khorsandi K, Rasul A et al (2021) Current understanding of epigenetics mechanism as a novel target in reducing cancer stem cells resistance. Clin Epigenetics 13(1):120
Wang L, Park P, La Marca F et al (2013) Bone formation induced by BMP-2 in human osteosarcoma cells. Int J Oncol 43(4):1095–1102
Wang L, Park P, Zhang H et al (2011) BMP-2 inhibits the tumorigenicity of cancer stem cells in human osteosarcoma OS99–1 cell line. Cancer Biol Ther 11(5):457–463
Wang L, Park P, La Marca F et al (2015) BMP-2 inhibits tumor-initiating ability in human renal cancer stem cells and induces bone formation. J Cancer Res Clin Oncol 141(6):1013–1024
Persano L, Pistollato F, Rampazzo E et al (2012) BMP2 sensitizes glioblastoma stem-like cells to Temozolomide by affecting HIF-1alpha stability and MGMT expression. Cell Death Dis 3:e412
Huang P, Chen A, He W et al (2017) BMP-2 induces EMT and breast cancer stemness through Rb and CD44. Cell Death Discov 3:17039
Zhang G, Huang P, Chen A et al (2018) How BMP-2 induces EMT and breast cancer stemness through Rb and CD44? Cell Death Dis 9(2):20
Kim BR, Oh SC, Lee DH et al (2015) BMP-2 induces motility and invasiveness by promoting colon cancer stemness through STAT3 activation. Tumour Biol 36(12):9475–9486
Takada I, Yogiashi Y, Kato S (2012) Signaling Crosstalk between PPARgamma and BMP2 in Mesenchymal Stem Cells. PPAR Res 2012:607141
Abolarinwa BA, Ibrahim RB, Huang YH (2019) Conceptual Development of Immunotherapeutic Approaches to Gastrointestinal Cancer. Int J Mol Sci 20(18)
Saitoh M (2018) Involvement of partial EMT in cancer progression. J Biochem 164(4):257–264
Gabryanczyk A, Klimczak S, Szymczak-Pajor I et al (2021) Is Vitamin D Deficiency Related to Increased Cancer Risk in Patients with Type 2 Diabetes Mellitus? Int J Mol Sci 22(12)
Singh M, Yelle N, Venugopal C et al (2018) EMT: Mechanisms and therapeutic implications. Pharmacol Ther 182:80–94
Aiello NM, Kang Y (2019) Context-dependent EMT programs in cancer metastasis. J Exp Med 216(5):1016–1026
Kang MH, Kang HN, Kim JL et al (2009) Inhibition of PI3 kinase/Akt pathway is required for BMP2-induced EMT and invasion. Oncol Rep 22(3):525–534
Wang MH, Zhou XM, Zhang MY et al (2017) BMP2 promotes proliferation and invasion of nasopharyngeal carcinoma cells via mTORC1 pathway. Aging (Albany NY) 9(4):1326–1340
Liu J, Ben QW, Yao WY et al (2012) BMP2 induces PANC-1 cell invasion by MMP-2 overexpression through ROS and ERK. Front Biosci (Landmark Ed) 17(7):2541–2549
Wu CJ, Sundararajan V, Sheu BC et al (2019) Activation of STAT3 and STAT5 Signaling in Epithelial Ovarian Cancer Progression: Mechanism and Therapeutic Opportunity. Cancers (Basel) 12(1)
Raida M, Heymann AC, Gunther C et al (2006) Role of bone morphogenetic protein 2 in the crosstalk between endothelial progenitor cells and mesenchymal stem cells. Int J Mol Med 18(4):735–739
Langenfeld EM, Langenfeld J (2004) Bone morphogenetic protein-2 stimulates angiogenesis in developing tumors. Mol Cancer Res 2(3):141–149
Bieniasz M, Oszajca K, Eusebio M et al (2009) The positive correlation between gene expression of the two angiogenic factors: VEGF and BMP-2 in lung cancer patients. Lung Cancer 66(3):319–326
Feng PC, Ke XF, Kuang HL et al (2019) BMP2 secretion from hepatocellular carcinoma cell HepG2 enhances angiogenesis and tumor growth in endothelial cells via activation of the MAPK/p38 signaling pathway. Stem Cell Res Ther 10(1):237
Becker V, Hui X, Nalbach L et al (2021) Linalool inhibits the angiogenic activity of endothelial cells by downregulating intracellular ATP levels and activating TRPM8. Angiogenesis 24(3):613–630
Wang P, Zhang L, Yao J et al (2015) An arabinogalactan from flowers of Panax notoginseng inhibits angiogenesis by BMP2/Smad/Id1 signaling. Carbohydr Polym 121:328–335
Qiu H, Yang B, Pei ZC et al (2010) WSS25 inhibits growth of xenografted hepatocellular cancer cells in nude mice by disrupting angiogenesis via blocking bone morphogenetic protein (BMP)/Smad/Id1 signaling. J Biol Chem 285(42):32638–32646
Chen M, Liu H, Li Z et al (2021) Mechanism of PKM2 affecting cancer immunity and metabolism in Tumor Microenvironment. J Cancer 12(12):3566–3574
Smith MG, Hold GL, Tahara E et al (2006) Cellular and molecular aspects of gastric cancer. World J Gastroenterol 12(19):2979–2990
Chapellier M, Bachelard-Cascales E, Schmidt X et al (2015) Disequilibrium of BMP2 levels in the breast stem cell niche launches epithelial transformation by overamplifying BMPR1B cell response. Stem Cell Reports 4(2):239–254
McLean K, Gong Y, Choi Y et al (2011) Human ovarian carcinoma-associated mesenchymal stem cells regulate cancer stem cells and tumorigenesis via altered BMP production. J Clin Invest 121(8):3206–3219
Rubio R, Abarrategi A, Garcia-Castro J et al (2014) Bone environment is essential for osteosarcoma development from transformed mesenchymal stem cells. Stem Cells 32(5):1136–1148
Avnet S, Di Pompo G, Chano T et al (2017) Cancer-associated mesenchymal stroma fosters the stemness of osteosarcoma cells in response to intratumoral acidosis via NF-kappaB activation. Int J Cancer 140(6):1331–1345
Engin AB, Nikitovic D, Neagu M et al (2017) Mechanistic understanding of nanoparticles’ interactions with extracellular matrix: the cell and immune system. Part Fibre Toxicol 14(1):22
Chouaib S, Messai Y, Couve S et al (2012) Hypoxia promotes tumor growth in linking angiogenesis to immune escape. Front Immunol 3:21
Zara JN, Siu RK, Zhang X et al (2011) High doses of bone morphogenetic protein 2 induce structurally abnormal bone and inflammation in vivo. Tissue Eng Part A 17(9–10):1389–1399
Wu G, Huang F, Chen Y et al (2020) High Levels of BMP2 Promote Liver Cancer Growth via the Activation of Myeloid-Derived Suppressor Cells. Front Oncol 10:194
Chen Z, Wu C, Gu W et al (2014) Osteogenic differentiation of bone marrow MSCs by beta-tricalcium phosphate stimulating macrophages via BMP2 signalling pathway. Biomaterials 35(5):1507–1518
Wang S, Jiang H, Zheng C et al (2022) Secretion of BMP-2 by tumor-associated macrophages (TAM) promotes microcalcifications in breast cancer. BMC Cancer 22(1):34
Acknowledgements
We thank all participants involved in this research.
Funding
This work was supported by the National Natural Science Foundation of China (No. 21777060), the Horizontal Project of Jiangsu Medical Vocational College (jcyxhxkt2021-01), and the Biology Discipline Team (jdxktd-2019003).
Ethics declarations
Ethics approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
For this type of study, informed consent was not needed.
Conflicts of interest
All authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, Tt., Lai, Yw., Han, X. et al. BMP2 as a promising anticancer approach: functions and molecular mechanisms. Invest New Drugs 40, 1322–1332 (2022). https://doi.org/10.1007/s10637-022-01298-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-022-01298-4