Abstract
Atlantic sturgeon (Acipenser oxyrinchus oxyrinchus) is a large, anadromous fish native to the Atlantic Coast of North America. Although this species once supported important fisheries, centuries of exploitation and habitat degradation have resulted in dramatic declines, presumed extirpation in some rivers, and ultimately listing under the U.S. Endangered Species Act (ESA). Under the ESA, Atlantic sturgeon are listed as five separate Distinct Population Segments (DPSs), which form the basis for federal management. Despite state and federal protections Atlantic sturgeon still face significant threats to their recovery, including fisheries bycatch mortality, marine construction, dredging, dams, and vessel strikes. However, because subadult and adult Atlantic sturgeon migrate extensively across estuarine and marine environments and frequently form mixed-stock aggregations in non-natal habitats, it can be difficult to determine how these threats impact specific populations and DPSs. To better understand ontogenetic shifts in habitat use and stock-specific exposure to anthropogenic threats, we performed a mixed-stock analysis of 1704 Atlantic sturgeon encountered across the U.S. Atlantic Coast. Collections made north of Cape Cod, MA and south of Cape Hatteras, NC were dominated by individuals from regional stocks; however, we found extensive stock mixing in the mid-Atlantic region, particularly in coastal environments where individuals from all five DPSs were commonly observed. Subadults and adults that were encountered in offshore environments had moved, on average, 277 km from their natal source; however, 23% were sampled over 500 km from their natal river suggesting long-distance movements are relatively common in these age classes. Overall, our work highlights that Atlantic sturgeon populations are vulnerable to threats over vast areas and emphasizes the need for continued genetic monitoring to track recovery progress.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Atlantic sturgeon (Acipenser oxyrinchus oxyrinchus) is a large, long-lived, anadromous fish native to the Atlantic Coast of North America. Historically, this species was very abundant (Secor 2002) and supported important commercial fisheries for caviar and meat in rivers and estuaries along the coast. However, centuries of exploitation and habitat degradation resulted in dramatic population declines and extirpation in some rivers (ASSRT 2007). Following nearly a century of modest harvest on remaining populations, states from Maine to Florida instituted a coastwide harvest moratorium in the United States in 1998 (ASMFC 1998), which was ultimately followed by their listing under the U.S. Endangered Species Act (ESA) in 2012 (NMFS 2012a, b).
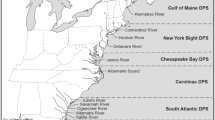
Under the ESA, Atlantic sturgeon populations were divided into five Distinct Population Segments (DPSs; USFWS and NMFS 1996), with each DPS comprised of populations that were thought to share genetic and physiological properties and located in similar geophysical habitats (ASSRT 2007). The decision to list Atlantic sturgeon by DPSs was based on available mitochondrial DNA and microsatellite genetic data, which indicated that each river supported a discrete spawning population and that populations within geographic regions formed genetic clusters that were significant to the species (Waldman et al. 2002; ASSRT 2007; Grunwald et al. 2008). At the time of ESA listing, four DPSs were classified as endangered including the South Atlantic, Carolina, Chesapeake Bay, and New York Bight DPSs. The Gulf of Maine DPS was classified as threatened due to limited perceived threats to population persistence (ASSRT 2007). Atlantic sturgeon populations in Canadian rivers are outside the purview of the ESA; however, they are listed as threatened by the Committee on the Status of Endangered Wildlife in Canada (COSEWIC 2011) and may co-occur with United States stocks (hereinafter, we use the term ‘stock’ to refer to either DPSs or populations, as the DPS framework is a hierarchical organization wherein DPSs aggregate rivers that broadly share similar characteristics, but rivers within a DPS are genetically distinct and may have different demographic properties that dictate population-specific management goals).
Federal management of Atlantic sturgeon in the United States is legally predicated on the DPS framework, with each DPS managed separately to minimize the impacts of anthropogenic activities (e.g., dredging, offshore wind development, and commercial fisheries) on recovery. The Atlantic States Marine Fisheries Commission also seeks to evaluate stock status at coastwide, DPS, and population-specific levels (ASMFC 2017). However, the need to monitor and manage recovery threats to specific stocks presents significant challenges to Atlantic sturgeon conservation. While juveniles are generally thought to remain in natal rivers (Fox and Peterson 2019), subadults and adults migrate extensively across estuarine and marine environments where they commonly form mixed-stock aggregations (Dunton et al. 2012; Wirgin et al. 2012; Waldman et al. 2013; O’Leary et al. 2014). Thus, the mosaic of habitats used by Atlantic sturgeon to complete their life cycle makes populations vulnerable to anthropogenic threats at both local and coast-wide scales (Erickson et al. 2011; Wirgin et al. 2015a) including bycatch mortality (Dunton et al. 2015), marine construction, dredging, vessel strikes, dams, and potential predation by invasive species (ASMFC 2017). Consequently, an enhanced understanding of stock-specific habitat use can help determine how anthropogenic disturbances differentially impact each population, which can support recovery and management activities (ASSRT 2007; ASMFC 2017).
The primary method used to determine the origin of Atlantic sturgeon encountered in non-natal habitats is genetic assignment testing. Previous efforts have used assignment tests to characterize mixed-stock aggregations in specific areas and to understand the stock composition of marine bycatch. However, these efforts have largely been concentrated to Atlantic sturgeon encountered in offshore environments in more northern extents of the species’ range, particularly off the cost of New York and in the Bay of Fundy (Wirgin et al. 2012, 2015b; Waldman et al. 2013). Less attention has been given to individuals captured in inshore areas (rivers and estuaries). Likewise, little is known about the stock composition of Atlantic sturgeon encountered offshore of the southeastern United States. This presents a critical uncertainty, as anthropogenic activities may disproportionately impact different stocks, depending on the region and whether they occur in sheltered inshore waters or on the continental shelf.
In this study, we performed individual-based assignment tests and mixture analyses to characterize the stock composition of Atlantic sturgeon encountered in offshore and riverine/estuarine environments across the U.S. Atlantic Coast. We first evaluated broad patterns in stock composition across all samples collected from Maine to Florida and estimated ontogenetic differences in marine dispersal from natal environments. Due to differences in sample size and stock composition, we then divided the data into three latitudinal regions and quantified differences in stock composition. Finally, we investigated whether stock composition varied between riverine/estuarine and offshore habitats within each region. Overall, this analysis provides an enhanced understanding of the life history of this species, helps identify stock-specific exposure to anthropogenic threats, and can be used to support efforts to understand the relative abundance of specific stocks in different geographic regions.
Materials and methods
Sample collection and genotyping
Samples used in our mixed-stock analyses (herein referenced as “unknown samples”, as their natal origins were unknown prior to this analysis) were from Atlantic sturgeon collected by partners at other institutions in both riverine/estuarine and offshore environments and were selected by the National Marine Fisheries Service to ensure the analysis had broad geographic coverage across the U.S. Atlantic coast. Samples represented all life stages, with n = 129 juveniles (defined as individuals with a total length (TL) < 500 mm), 1186 subadults (500–1500 mm), and 147 adults (> 1500 mm). There were also 242 individuals for which size data were not collected. These individuals were excluded from analyses that were age-specific; however, they were included in comparisons where individuals were pooled across size classes. Thus, our total sample size for the mixed-stock analysis was 1704 individuals. The majority of these (79.6%) were collected within the last 10 years, which represents less than a single generation time for Atlantic sturgeon.
Each sample was preserved upon capture, typically via immersion in 95% nondenatured ethanol or RNALater (Invitrogen). Whole genomic DNA was extracted from tissue samples using Puregene reagents (Qiagen). All samples were screened for 12 Atlantic sturgeon microsatellite loci (LS19, LS39, LS54, LS68, Aox12, Aox23, Aox45, AoxD170, AoxD188, AoxD165, AoxD44, AoxD241; described in May et al. 1997; King et al. 2001; and Henderson-Arzapalo and King 2002) at the U.S. Geological Survey Leetown Science Center (USGS-LSC) in Kearneysville, WV. The mean number of loci scored across all samples was 11.7 (range 7–12; individuals that were scored at fewer than seven loci were omitted from analysis, as assignments based on fewer loci are more likely to be spurious).
Individual assignment tests and mixture analyses
We used two different approaches to characterize the stock composition of Atlantic sturgeon. In the first, we performed individual-based assignment tests in the program GeneClass2 (Piry et al. 2004) using the criterion of Rannala and Mountain (1997). In this analysis, allele frequency distributions were used to assign each unknown individual to one of 15 Atlantic sturgeon populations represented in a reference genetic baseline (described below). For each unknown individual, the population that had the highest relative likelihood was considered that individual’s assigned population, which subsequently determined the individual’s assigned DPS.
We also conducted a mixture analyses using ONCOR (Kalinowski et al. 2007). In contrast to individual assignment testing, this approach estimated the likely proportional contribution of each of the 15 populations to the sample of unknown individuals. Ninety-five percent confidence intervals on the inferred mixture proportions were calculated with 1000 bootstraps using the method of Rannala and Mountain (1997). Results of the mixture analysis were visualized using the ggplot package (Wickham 2016) within R.
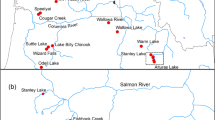
The reference genetic baseline used in individual-based assignment tests and mixture analyses was comprised of 2569 Atlantic sturgeon captured in targeted surveys by outside agencies and other permitted parties and incidental captures by fisheries, dredging, and other permitted activities [see Acknowledgements for a list of contributors, some samples were included in a previous publication by Wirgin et al. (2015b)]. Together, samples represented 14 populations plus one estuarine aggregation (Table 1, Fig. 1). The estuarine aggregation, herein the Albemarle Complex, included samples that likely represented several different spawning populations; however, these populations were grouped into a single population due to limited sample sizes. Those populations were assumed to be genetically similar to one another, and we considered their combined allele frequencies to be the best available representation of these populations given the data available. Within the South Atlantic DPS, genetically discrete spring- and fall-spawning populations have been documented in the Edisto River (Farrae et al. 2017), so these populations were included in the baseline separately. Although several other rivers are suspected to support separate spring- and fall-spawning populations, these were not included in the genetic baseline as insufficient samples were available to meet our criteria. In total, the baseline included 15 populations which represented all five DPSs, as well as populations from two Canadian rivers (Saint Lawrence and Saint John) that are beyond the purview of the ESA. To minimize the likelihood of using non-natal individuals to characterize a population’s genetic signature, individuals were only included in the baseline if they were river-resident juveniles (RRJ) or adults (see Table 1 for a description of the age classes).
Geographic distribution of 15 reference populations representing five Distinct Population Segments (DPSs) and Canadian rivers which were used as potential source populations for genetic assignment testing and mixture analyses. Note: there are two discrete populations in the baseline from the Edisto River which spawn separately in the spring and fall
Most Atlantic sturgeon used in the baseline were genotyped at the USGS-LSC using the same methods described above for unknown samples. However, a subset of individuals (n = 434; 16.9% of the total baseline) used to characterize the Carolina and South Atlantic DPSs were genotyped by the South Carolina Department of Natural Resources (SCDNR). A previous effort to standardize allele calls between USGS-LSC and SCDNR found very strong concordance between the two laboratories (99.7% of calls were identical across 94 individuals screened independently at both facilities). However, there were occasional differences at Aox12 that were difficult to reconcile. As a result, we omitted (i.e., treated as missing data) the scores generated by SCDNR at that locus for most populations. However, since limited USGS data was available to characterize the spring-spawning population of Edisto River (and GeneClass2 requires allelic data for all loci in all reference populations), we included Aox12 scores from SCDNR at this population. Overall, we anticipated this to have a negligible impact on the overall analysis, as the allele that we were unable to standardize occurred at a low frequency in this population. The final genetic baseline included only individuals scored at seven or more loci (mean = 11.67; range = 7–12).
The genetic baseline we used in this study included a greater number of reference individuals which represented a larger number of populations than previous mixed-stock analyses for Atlantic sturgeon. Moreover, as described above, all reference individuals were vetted against strict inclusion criteria in order to prevent inclusion of non-natal individuals in a population. While we expected this refined baseline to increase the accuracy of assignment tests on our unknown samples, we evaluated the efficacy of the baseline for assignment testing by using GeneClass2 to perform an individual-based assignment test using a leave-one-out approach. Overall, 83.1% of reference individuals assigned to the correct source population, and 96.1% of individuals to the correct DPS of origin (Table 2). In every population except those located in the Carolina DPS, > 90% of individuals were assigned to the correct DPS (Table 2). To evaluate accuracy of the mixture analyses in ONCOR, we performed 100% simulations in which we simulated samples composed entirely from a single population (200 individuals × 100 independent simulations per population; Anderson et al. 2008) and then quantified the average proportion of each sample attributed to the correct source. In general, populations with average assignment > 90% are often considered well-defined stocks that are good candidates for inclusion in mixed-stock analyses (Miralles et al. 2016). The 100% simulations analyses suggested that average estimated mixture proportions were > 88% and > 93% for populations and DPSs, respectively (Table 3). Taken together, these results show considerable power to resolve the composition of mixed-stock aggregations of Atlantic sturgeon, particularly at the DPS-level.
Spatial distribution of Atlantic sturgeon stocks
Results of individual-based assignment tests of unknown samples were visualized in ArcMap 10.6.1 (Environmental Systems Research Institute, Redlands, California) and R (R Core Team 2019). We used simple one-way ANOVAs to test for differences in the latitudinal distribution of populations and DPSs and considered P < 0.05 to reflect statistical significance.
To evaluate ontogenetic changes in movement and habitat use, we used the raster package (Hijmans 2020) in R to measure the shortest waterway distance between the capture location of each subadult and adult encountered in offshore habitats and the mouth of its assigned natal river. Juveniles were excluded from this analysis, as only three fish < 500 mm were captured offshore, which is expected given limited movement at this life stage (Hilton et al. 2016; Fox and Peterson 2019). Additionally, because the exact location of spawning ground(s) is unknown in most rivers, we only measured distance to the river mouth. However, the distance between spawning habitat and river mouth is minor relative to the spatial scale of this analysis and this source of measurement error is held relatively constant across all individuals. We acknowledge that this analysis cannot account for non-linear movement and is biased by non-random temporal and spatial sampling. Thus, results of this analysis provide only a minimum estimate of individual movement and should be viewed as a conservative estimate of the relative ability of each size class to disperse to distant locations.
Our initial visual review of the results suggested there was spatial heterogeneity in sample size and stock composition, and so we divided the data into three latitudinal regions, “NORTH”, “MID”, and “SOUTH” for further analysis. When deciding on the breakpoint for each region, we also considered the location of important coastal features, namely Cape Hatteras and Cape Cod, which have been shown to be important geophysical features in the distribution and genetic structure of other species (Wilkinson et al. 2009; Pappalardo et al. 2015). We used Chi-squared tests to quantify differences in stock composition (population- and DPS-level) among the three latitudinal regions, with stock identity determined by results of the individual-based assignment tests. Using the same methods described above for the range-wide dataset, we also conducted a mixture analysis in ONCOR to estimate the proportional contribution of each population and DPS to the sample collected in each region.
Within each region, we used a Chi-square test to assess for differences in stock composition between fish caught in riverine/estuary habitats and those caught offshore. Here, we define riverine/estuarine encounters as all individuals that were captured in inland waters and sounds. We recognize that the difference between riverine/estuarine and offshore habitats is not necessarily dichotomous as individuals may frequently move between habitats across short temporal scales; however, this classification system was the most objective way to classify individuals by habitat type.
Results
Average individual-based assignment probability was 87.7%, suggesting most individuals assigned strongly to a single population (Supplemental Material 1; Supplemental Fig. 1). In the rare cases where affinity towards a specific natal population was weak (i.e., < 50%), the next most probable population was frequently within the same DPS (40/68 instances).
Across all 1704 unknown samples collected on the U.S. Atlantic Coast, we observed a mixture of Atlantic sturgeon from all five DPSs and two Canadian rivers. Individual-based assignment tests indicated that the majority of individuals likely originated from populations in the South Atlantic (38.2%), New York Bight (25.5%), and Carolina DPSs (22.6%) with smaller contributions from the Chesapeake Bay DPS (9.8%) and the Gulf of Maine DPS and Canada (< 3%). Results from mixture analyses were consistent with those from individual-based analyses (Supplemental Fig. 2; see Supplemental Material 2 for mixture proportions). However, we note that estimates reflect the spatial distribution of samples considered in this study, as most samples (1663 of 1704) were taken in the MID and SOUTH latitudinal regions.
Based on one-way ANOVAs, there were statistically significant differences in the latitudinal distribution of each population (P < 0.001; r2 = 0.711) and DPS (P < 0.001; r2 = 0.678). This was largely because individuals were most likely to assign to a population/DPS located in the same general region in which they were collected (Fig. 2). However, subadults and adults from all five DPSs and the two Canadian populations were frequently found far from their natal habitats (Supplemental Fig. 3). For example, eight sturgeon that assigned to populations in the South Atlantic DPS were collected from the coastal waters between Long Island, NY and Cape Cod, MA including one subadult that was collected near Martha’s Vineyard—over 1300 km from its inferred natal source. Conversely, two individuals from the Kennebec River (Gulf of Maine DPS) were captured off the Outer Banks of North Carolina (approximately 1040 km from the Kennebec River) and a subadult from the Saint John River (Canada) was encountered 913 km away off the coast of Virginia.
Latitudinal distribution of samples collected in riverine/estuarine and offshore environments that assigned to each of the 15 populations in the genetic baseline. Populations are color-coded by Distinct Population Segment (DPS). The boxes span the interquartile range and the horizontal bar denotes the median latitude observed for each population. Yellow diamonds indicate the approximate latitude of the mouth of each natal river for populations within the United States
Overall, while movements outside of non-natal rivers were seldom observed in juveniles, subadults and adults were frequently captured in offshore habitats that were thousands of kilometers from their source (Fig. 3). Many subadults were captured within 200 km of their natal river; however, 21.5% of subadults had moved at least 500 km from their natal river at the time of capture (median distance to source location = 123 km, range = 5 to 1868 km). Adults were generally captured further from their natal source (median = 190 km, range = 28 to 1879); however, a large proportion of adults were also detected near their source river.
Due to differences in stock composition and sampling intensity among regions, we present regional results separately below. This decision was supported by a significant Chi-squared test result (P < 0.001).
Stock composition in the NORTH region
Of the 41 individuals captured in the NORTH region, 87.8% assigned to the Kennebec River population, which is the only population in the Gulf of Maine DPS. Smaller contributions (12.2%) of Atlantic sturgeon from Canadian Rivers were also detected. There was no indication that Atlantic sturgeon from other stocks were present in our samples from the NORTH region, nor did we detect any differences in stock composition between individuals collected in riverine/estuarine habitats and offshore (P = 0.09 at the population-level and P = 0.06 at the DPS-level). Results from mixture analyses (Fig. 4) supported results from individual-based assignments (Fig. 5) and also suggested that samples from the NORTH region were represented exclusively by fish from the Gulf of Maine DPS and the Canadian rivers (see Supplemental Material 2 for mixture proportions). However, we note that inferences should be drawn with caution given our limited sample size.
Individual-based assignment tests for individuals that were captured in the NORTH region on the U.S. Atlantic Coast. Points are symbolized by river and color-coded by Distinct Population Segment (DPS) of origin. Bar graphs on the right show the proportion of individuals that were captured in riverine/estuarine (top) and offshore (bottom) that assigned to each of the 15 populations
Stock composition in the MID region
In the MID region, individual-based assignment tests suggested a highly mixed assemblage of Atlantic sturgeon from all five DPSs which included 14 populations from across the U.S. and Canada (the only population not represented in the sample was the Saint Lawrence). Individual-based assignment testing suggested 37.5% of individuals assigned to populations in the New York Bight DPS and 30.7% of individuals to populations in the Carolina DPS (Fig. 4). At the population-level, the Albemarle Complex (27.3%) and Hudson River (26.2%) populations were the most prevalent in the sample. However, there were notable contributions from all DPSs and the Canadian rivers (Fig. 6). Mixture analyses suggested similar contributions from each population and DPS in the sample (see Supplemental Material 2 for mixture proportions).
Individual-based assignment tests for individuals that were captured in the MID region on the U.S. Atlantic Coast. Points are symbolized by river and color-coded by Distinct Population Segment (DPS) of origin. Bar graphs on the right show the proportion of individuals that were captured in riverine/estuarine (top) and offshore (bottom) that assigned to each of the 15 populations
We also observed differences in the stock composition of individuals captured in riverine/estuarine habitats versus offshore (P < 0.001 at both the population- and DPS-level; Fig. 5). Of the individuals captured in the MID region in riverine/estuarine environments, 60.9% assigned to the Carolina DPS. Conversely, only 6.0% of individuals captured offshore assigned to the Carolina DPS, and individuals from the New York Bight (54.0%) and Chesapeake Bay DPSs (21.6%) were more prevalent (Fig. 6). Results from the mixture analysis were similar and also suggested that samples collected offshore likely represented individuals from more populations and DPSs than samples collected in riverine/estuarine environments (Supplemental Material 2).
Stock composition in the SOUTH region
Individual-based assignment testing indicated that Atlantic sturgeon from the SOUTH region were primarily from the South Atlantic (91.2%) and Carolina (6.2%) DPSs. At a population-level, individual-based assignment tests suggested the Altamaha River population was the most prevalent in the sample (35.7%), with lesser contributions from the fall-spawning population of the Edisto River (21.4%), Savannah River (18.9%) and Ogeechee River (7.2%). Minor contributions were attributed to the Satilla River (5.5%), Pee Dee River (3.7%), Edisto River spring-run (2.0%), and Albemarle Complex (2.5%; Fig. 7). These results were consistent with those obtained via mixture analyses (see Supplemental Material 2 for mixture proportions).
Individual-based assignment tests for individuals that were captured in the SOUTH region on the U.S. Atlantic Coast. Points are symbolized by river and color-coded by Distinct Population Segment (DPS) of origin. Bar graphs on the right show the proportion of individuals that were captured in riverine/estuarine (top) and offshore (bottom) that assigned to each of the 15 populations
Despite the predominance of individuals from South Atlantic DPS populations in the SOUTH region, we did observe some individuals from more northern populations. Some of these individuals were observed offshore at the boundary between the MID and SOUTH regions, including sturgeon collected offshore of Cape Hatteras, NC and that assigned to the Chesapeake Bay DPS (approximately 235 km from the source population) and New York Bight DPS (651 km from the source population). However, individuals from the Chesapeake Bay DPS collected in riverine/estuarine habitats spanned nearly the entire latitudinal range of the SOUTH region, with seven individuals captured in the Neuse River, NC and one individual as far south as the East River, GA. Thus, while the stock structure in the coastal and marine habitats in the SOUTH is less complex than the MID latitude region, multiple DPSs were present with varying contributions from many different populations in both offshore and riverine/estuarine habitats.
At the DPS-level, there were no differences in stock composition between individuals captured in riverine/estuarine habitats and offshore (P = 0.13). At the population-level, there were significant differences in stock composition between the two habitats (P < 0.001), which reflects the relatively larger proportion of individuals from the Edisto River fall-run, Savannah River, and Ogeechee River populations sampled from riverine/estuarine environments. As discussed below, this difference may reflect the relatively low abundance of these populations compared to the Altamaha, which could make them difficult to capture in offshore environments. Results of the mixture analysis supported the relatively large contribution of individuals from the South Atlantic DPS in both riverine/estuarine and offshore habitats and indicated that the Altamaha was also the most numerically present population in samples from the SOUTH region (Supplemental Material 2).
Discussion
It is evident from our results that Atlantic sturgeon move and mix extensively along the U.S. Atlantic Coast. In this general respect, our results are consistent with previous mixed-stock analyses (Dunton et al. 2012; Wirgin et al. 2012; Waldman et al. 2013; O’Leary et al. 2014) and telemetry studies (Erickson et al. 2011; Taylor et al. 2016) which have documented long-distance movement patterns that allow Atlantic sturgeon to move among the geographic regions that are associated with the five DPSs. However, our study is the first to rigorously expand our understanding to the southeastern United States, which provides new insight into broad-scale patterns of stock composition across the coast and allows spatial comparisons among latitudinal regions to be made. The substantial spatial heterogeneity in stock composition and uneven sampling intensities among regions suggest that the stock composition we observed at coast-wide scales should not be interpreted as the overall relative abundance of each sturgeon stock. Rather, our data indicate that stock composition is more accurately assessed at a regional level.
In our visual assessment, we saw clear changes in stock composition at Cape Cod, MA and Cape Hatteras, NC and subsequently used these latitudinal landmarks to demarcate our regional comparisons. These prominent coastal landmarks are associated with important ecological breaks for many other marine taxa (Wilkinson et al. 2009; Pappalardo et al. 2015), potentially due to significant shifts in circulation patterns, thermal regimes, and habitat (Pappalardo et al. 2015). Although our study lacks the ability to comment which mechanisms may be shaping the observed distributions of Atlantic sturgeon stocks, it does suggest that stock distribution may not simply reflect random dispersal from natal habitats. Rather, stock-specific distribution patterns may be dependent on regional environmental conditions (Di Santo 2016; Neuheimer et al. 2018) and physiological constraints imposed by local adaptation to natal rivers. Thus, while seasonal changes in abiotic environmental conditions likely allow individuals to at least temporarily occupy distantly located habitats, it is noteworthy that we did not observe Atlantic sturgeon from the Gulf of Maine DPS and Canadian rivers in the warmer waters south of Cape Hatteras, and those from populations which spawn south of Cape Hatteras were not observed in the colder waters of the Gulf of Maine.
In the MID region (between Cape Hatteras and Cape Cod), we observed a highly mixed assemblage of Atlantic sturgeon from all five DPSs, which included 14 populations distributed across the U.S. Atlantic Coast as well as Canada (Fig. 6). This is congruent with previous genetic (Dunton et al. 2012; Waldman et al. 2013; O’Leary et al. 2014; Wirgin et al. 2015a; Wirgin et al. 2015b) and telemetry studies (Rulifson et al. 2020), which assigned Atlantic sturgeon in the marine waters of this region to many different stocks. However, even within this well-mixed region, patterns were still apparent. Consistent with previous studies in the region (Dunton et al. 2012; Waldman et al. 2013; O’Leary et al. 2014; Wirgin et al. 2015a; Wirgin et al. 2015b), the majority of individuals sampled offshore assigned to the Hudson River (New York Bight DPS) and James River (Chesapeake Bay DPS) populations, with lesser contributions from other stocks (Fig. 6). However, within the sheltered waters of Albemarle Sound and Delaware Bay, the proportion of individuals that assigned to distant stocks was smaller, with the majority of samples assigning to the Albemarle Complex (Carolina DPS) and Delaware River (New York Bight DPS) populations. Importantly, given that we sampled a large number of individuals from the sounds of North Carolina, it is likely that our analysis overestimated the total proportion of Atlantic sturgeon from the Carolina DPS that occur within the MID region. Along these lines, Wirgin et al. (2018) examined the composition of subadults collected within the Hudson River estuary (an area where we have limited samples) and found that the majority were part of the Hudson River population. However, even samples that were taken in close proximity to spawning habitats in the Hudson River, which is thought to host one of the most robust extant population of Atlantic sturgeon (ASSRT 2007; Waldman et al. 2019; Kazyak et al. 2020), Wirgin et al. (2018), still included individuals that originated from as far away as the Ogeechee and Kennebec rivers.
In the coastal waters of the SOUTH region, we observed less mixing among DPSs. Most individuals in this area assigned to populations within the South Atlantic DPS, with moderate contributions from the Carolina DPS. Among the samples that assigned to the South Atlantic DPS, the relative proportion of individuals that assigned to each population in the DPS appears to roughly correspond to the overall abundance of the population. This pattern was particularly apparent in offshore habitats, likely because Atlantic sturgeon are more difficult to capture in offshore environments and so capture probability is likely a function of abundance. For example, we saw a proportionally large contribution of individuals from the Altamaha River population in offshore samples, which is thought to host one of the largest contemporary populations of Atlantic sturgeon (Peterson et al. 2008). Populations from the Edisto (fall-spawning), Savannah, and Ogeechee rivers are likely intermediate in abundance, as inferred from side-scan sonar surveys (Vine et al. 2019) and the relative ease at which samples are collected, and contributed moderate proportions to the observed stock composition in the SOUTH region. Conversely, we observed few individuals from the Satilla or Edisto (spring spawning) rivers, and both of these populations are likely small [as suggested by the amount of effort required to capture individuals from these systems (Fritts et al. 2016; Farrae et al. 2017)]. However, assignments to specific populations within the South Atlantic DPS should be viewed with caution, as populations within this area have the lowest levels of genetic differentiation observed in the species (ASSRT 2007) and our ability to discriminate these populations using assignment tests is not as strong as in other regions (Table 2). Moreover, given the heterogeneous spatial and temporal distribution of sampling effort, the proportion of individuals that assigned to each population is likely not static across the region and over time. However, given that our assignments to DPS within this region were still very accurate (> 96% correct; Table 2) and individuals are most commonly found in close proximity to their natal river, our conclusion that the majority of Atlantic sturgeon observed in this region belonged to populations in the South Atlantic DPS is likely robust.
In the NORTH region, most of the sturgeon we analyzed were associated with the Kennebec River population (> 87%), with lesser contributions from Canadian populations (Saint Lawrence and Saint John rivers; Fig. 7). A previous study in the Bay of Fundy (hundreds of kilometers to the northeast of our sampling locations) determined the stocks in that area were primarily from the Saint John River (> 60%) and the Kennebec River (34–36%; Wirgin et al. 2012). In conjunction with our results, this suggests that Atlantic sturgeon in the Gulf of Maine are primarily from the Kennebec River and Canadian populations. This seems reasonable, as these are the only two known spawning populations within the region (ASSRT 2007; ASMFC 2017). However, given the small number of samples considered in our study it is possible that populations from other regions use this area but were not detected. Notably, Erickson et al. (2011) reported that a large adult female Atlantic sturgeon tagged in the Hudson River was positioned in the Bay of Fundy, suggesting, along with Wirgin et al. (2012) that at least some sturgeon from populations to the south occasionally utilize this area. However, as previously noted, stock-specific habitat use may vary seasonally and potentially reflects physiological differences among stocks.
Across all regions, many stocks appeared to be most common near their natal habitats (Fig. 2) with, for example, clusters of fish that assigned to the New York Bight and Chesapeake Bay DPSs near the primary spawning rivers found in those geographic areas. Consistent with the patterns of ontogenetic habitat use, juveniles (< 500 mm) were generally sampled in their natal river. However, we did detect three juveniles in offshore habitats, including one juvenile that assigned to the Hudson River (New York Bight DPS) and was captured nearly 900 km away off the coast of North Carolina. Compared to subadults, adult Atlantic sturgeon were captured further from their natal river (123 km and 190 km median distance from natal source, respectively). However, it should be noted that many subadults were also captured thousands of kilometers from their natal source, suggesting that this life stage is also capable of long-distance movements in offshore environments. Moreover, our movement estimates represent a minimum distance between capture location and natal source, and so significantly underestimate the total distance an individual moved between its spawning habitat and the point of capture.
Taken together, our analyses suggest that many Atlantic sturgeon use habitats far from their natal river, and in moving to these they pass the spawning rivers for other populations/DPSs and cross between state and international boundaries. For example, Atlantic sturgeon from the South Atlantic DPS were observed near Martha’s Vineyard, MA, along with individuals from many other stocks. While our study detected very few sturgeon in the SOUTH region from the New York Bight DPS, telemetry data suggest these fish use these coastal waters at least to some extent (Erickson et al. 2011). Arendt et al. (2017) reported that 224 Atlantic sturgeon which had been tagged at coastal locations from Virginia to Connecticut were detected on a telemetry array off the coast of South Carolina, and presented genetic data to suggest that many of these fish represented populations spawning in distant areas to the north. This suggests that our current mixed-stock analysis may underestimate the extent to which northern populations are using southern habitats, particularly if they are staying offshore in areas where they are less likely to be physically captured.
Although the general patterns are compelling, appropriate caution should be exercised in interpreting the results. Collections occurred sporadically along the coast and were typically part of research projects that were targeting known aggregation sites during specific seasons. Thus, the current mixed-stock analysis presents a snapshot in time, and the actual stock composition of Atlantic sturgeon in coastal habitats almost certainly varies seasonally and annually. However, despite considerable telemetry research on Atlantic sturgeon, broad-scale movement patterns are still poorly understood (Dunton et al. 2010; Melnychuk et al. 2017). Migration patterns may relate to changing conditions in the marine environment (Breece et al. 2016), and presumably also vary among stocks to facilitate differences in spawning phenology observed across the species’ range. As noted previously, several telemetry studies have reported long-distance migrations that were not always apparent from the samples we obtained across the coast. In addition, as the relative abundance of stocks changes through time, we expect that stock compositions in coastal environments will change accordingly. Consequently, there remains a need for continued genetic monitoring (Dunton et al. 2012; ASMFC 2017), as well as additional work to explore spatiotemporal variation within specific geographic areas.
Nonetheless, it is clear that many populations are vulnerable to threats far from their spawning and nursery habitats (Collins et al. 2000; Stein et al. 2004; Wirgin et al. 2015a), and that these threats occur throughout an individual’s life cycle. Throughout their U.S. range, Atlantic sturgeon encountered in inshore and offshore waters reflected a mixture of local and distant populations, and so it is inaccurate to assume that individuals encountered in these habitats reflect only local populations. Given the observed spatial patterns, local genetic assignment data are necessary to accurately characterize stock composition in a specific geographic area. Moreover, given the large spatial distribution of stocks, this research highlights the ability of human activities to negatively impact populations which spawn hundreds of kilometers or more away. However, this also suggests that populations may benefit from mitigation activities at local and regional levels, as discrete demographic units wander over broad areas and conservation measures in other locations could still benefit the overall population.
In the future, expanded genetic baselines that include additional populations that are not currently represented will improve inferences from mixed-stock analyses. These uncharacterized populations are generally thought to be small, which limited our ability to obtain DNA samples. Characterizing these populations is not likely to change the broad patterns observed in this study, as individuals from uncharacterized populations would be assigned to the most similar population in the baseline, which is likely in close proximity and part of the same DPS. However, it may provide insight into the status and threats to populations that are at the greatest risk of extirpation. In addition, there is a great opportunity to combine genetic assignment testing with the large body of acoustic telemetry data that are now available for Atlantic sturgeon. Combining these data sets would allow stock composition to be modeled over vast areas of the coast, including locations where sturgeon are known to occur but often difficult to physically capture, as well as offshore habitats where wind development and sand mining are planned. Continued collection of tissue samples of Atlantic sturgeon will allow us to further understand threats to Atlantic sturgeon stocks and track recovery progress.
References
Anderson EC, Waples RS, Kalinowski ST (2008) An improved method for predicting the accuracy of genetic stock identification. Can J Fish Aquat Sci 65:1475–1486
Arendt M, Post W, Frazier B, Talierco M, Farrae D, Darden T, Geer P, Kalinowsky C (2017) Temporal and spatial distribution of Atlantic sturgeon (Acipenser oxyrinchus) in U.S. Territorial waters off South Carolina and Georgia. Final Report for Grant Number NA13NMF4720045
ASMFC (Atlantic States Marine Fisheries Commission) (1998) Amendment 1 to the interstate fishery management plan for Atlantic sturgeon. Fishery Management Report 31. Washington DC
ASMFC (Atlantic States Marine Fisheries Commission) (2017) Atlantic Sturgeon Benchmark Stock Assessment and Peer Review Report, Arlington, VA. http://www.asmfc.org/files/Meetings/AtlMenhadenBoardNov2017/AtlSturgonBenchmarkStockAssmt_PeerReviewReport_2017.pdf
ASSRT (Atlantic Sturgeon Status Review Team) (2007) Status review of Atlantic sturgeon (Acipenser oxyrinchus oxyrinchus). Report to National Marine Fisheries Service. Northeast Regional Office, Gloucester, MA
Breece MW, Fox DA, Dunton KJ, Frisk MG, Jordaan A, Oliver MJ (2016) Dynamic seascapes predict the marine occurrence of an endangered species: Atlantic Sturgeon Acipenser oxyrinchus oxyrinchus. Methods Ecol Evol 7:725–733
Collins MR, Rogers SG, Smith TIJ, Moser ML (2000) Primary factors affecting sturgeon populations in the southeastern United States: fishing mortality and degradation of essential habitats. Bull Mar Sci 66:917–928
COSEWIC (Committee on the Status of Endangered Wildlife in Canada) (2011) COSEWIC assessment and status report on the Atlantic sturgeon Acipenser oxyrinchus in Canada. Committee on the Status of Endangered Wildlife in Canada, Ottawa
Di Santo V (2016) Intraspecific variation in physiological performance of a benthic elasmobranch challenged by ocean acidification and warming. J Exp Biol 219:1725–1733
Dunton KJ, Jordaan A, Conover DO, McKown KA, Bonacci LA, Frisk MG (2015) Marine distribution and habitat use of Atlantic Sturgeon in New York lead to fisheries interactions and bycatch. Marine and Coastal Fisheries 7(1):18–32
Dunton KJ, Jordaan A, McKown KA, Conover DO, Frisk MG (2010) Abundance and distribution of Atlantic Sturgeon (Acipenser oxyrhincus) within the Northwest Atlantic Ocean, determined from five fishery-independent surveys. Fish Bull 108:450–465
Dunton KJ, Chapman D, Jordaan A, Feldheim K, O’Leary SJ, McKown KA, Frisk MG (2012) Genetic mixed-stock analysis of Atlantic sturgeon Acipenser oxyrinchus oxyrinchus in a heavily exploited marine habitat indicates the need for routine genetic monitoring. J Fish Biol 80(1):207–217
Erickson DL, Kahnle A, Millard MJ, Mora EA, Bryja M, Higgs A, Mohler J, Dufour M, Kenney G, Sweka J, Pikitch EK (2011) Use of pop-up satellite archival tags to identify oceanic-migratory patterns for adult Atlantic Sturgeon, Acipenser oxyrinchus oxyrinchus Mitchell, 1815. J Appl Ichthyol 27:356–365
Farrae DJ, Post WC, Darden TL (2017) Genetic characterization of Atlantic sturgeon, Acipenser oxyrinchus oxyrinchus, in the Edisto River, South Carolina and identification of genetically discrete fall and spring spawning. Conserv Genet 18:813–823
Fritts MW, Grunwald C, Wirgin I, King TL, Peterson DL (2016) Status and genetic character of Atlantic Sturgeon in the Satilla River, Georgia. Trans Am Fish Soc 145:69–82
Fox AG, Peterson DL (2019) Movement and out-migration of juvenile Atlantic sturgeon in Georgia, USA. Trans Am Fish Soc 148:952–962
Grunwald C, Maceda L, Waldman J, Stabile J, Wirgin I (2008) Conservation of Atlantic sturgeon Acipenser oxyrinchus oxyrinchus: delineation of stock structure and distinct population segments. Conserv Genet 9(5):1111–1124
Henderson-Arzapalo A, King TL (2002) Novel microsatellite markers for Atlantic Sturgeon (Acipenser oxyrhinchus) population delineation and broodstock management. Mol Ecol Notes 2:437–439
Hijmans RJ (2020) raster: geographic data analysis and modeling. R Package Version 3.4–20. https://CRAN.R-project.org/package=raster
Hilton EJ, Kynard B, Balazik MT, Horodysky AZ (2016) Dillman CB (2016) Review of the biology, fisheries, and conservation status of the Atlantic sturgeon, (Acipenser oxyrinchus oxyrinchus Mitchill, 1815). J Appl Ichthy 32(Suppl. 1):30–66
Kalinowski ST, Manlove KR, Taper ML (2007) ONCOR: a computer program for genetic stock identification. Department of Biology, Montana State University, Bozeman, Montana
Kazyak DC, Flowers AM, Hostetter NJ, Madsen JA, Breece M, Higgs A, Brown LM, Royle JA, Fox DA (2020) Integrating side-scan sonar and acoustic telemetry to estimate the annual spawning run size of Atlantic Sturgeon in the Hudson River. Can J Fish Aquat Sci. https://doi.org/10.1139/cjfas-2019-0398
King TL, Lubinski BA, Spidle AP (2001) Microsatellite DNA variation in Atlantic Sturgeon (Acipenser oxyrinchus oxyrinchus) and cross-species amplification in the Acipenseridae. Conserv Genet 2:103–119
May B, Krueger CC, Kincaid HL (1997) Genetic variation at microsatellite loci in sturgeon: primer sequence homology in Acipenser and Scaphirhynchus. Can J Fish Aquat Sci 54:1542–1547
Melnychuk MC, Dunton KJ, Jordaan A, McKown KA, Frisk MG (2017) Informing conservation strategies for the endangered Atlantic sturgeon using acoustic telemetry and multi-state mark–recapture models. J Appl Ecol 54:914–925
Miralles L, Mrugala A, Sanchez-Jerez P, Juanes F, Garcia-Vazquez E (2016) Potential impact of Mediterranean aquaculture on the wild predatory bluefish. Marine Coast Fish 8:92–99
National Marine Fisheries Service (NMFS) (2012a) Endangered and threatened wildlife and plants; threatened and endangered status for distinct population segments of Atlantic sturgeon in the northeast region. Fed Reg 77:5880–5912
National Marine Fisheries Service (NMFS) (2012b) Endangered and threatened wildlife and plants; final listing determinations for two distinct population segments of Atlantic sturgeon (Acipenser oxyrinchus oxyrinchus) in the southeast. Fed Reg 77:5914–5982
Neuheimer AB, MacKenzie BR, Payne MR (2018) Temperature-dependent adaptation allows fish to meet their food across their species’ range. Sci Adv 4:e4349
O’Leary SJ, Dunton KJ, King TL, Frisk MG, Chapman DD (2014) Genetic diversity and effective size of Atlantic sturgeon, Acipenser oxyrhinchus oxyrhinchus river spawning populations estimated from the microsatellite genotypes of marine-captured juveniles. Conserv Genet 15(5):1173–1181
Pappalardo P, Pringle JM, Wares JP, Byers JE (2015) The location, strength, and mechanisms behind biogeographic boundaries of the east coast of North America. Ecography 38:722–731
Piry S, Alapetite A, Cornuet JM, Paetkau D, Baudouin L, Estoup A (2004) GeneClass2: a software for genetic assignment and first-generation migrant detection. J Hered 95:536–539
Peterson DL, Schueller P, Devries R, Fleming J, Grunwald C, Wirgin I (2008) Annual run size and genetic characteristics of Atlantic Sturgeon in the Altamaha River, Georgia. Trans Am Fish Soc 137:393–401
R Core Team (2019) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Rannala B, Mountain JL (1997) Detecting immigration by using multilocus genotypes. Proc Natl Acad Sci 94:9197–9201
Rulifson RA, Bangley CW, Cudney JL, Dell’Apa A, Dunton KJ, Frisk MG, Loeffler MS, Balazik MT, Hager C, Savoy T, Brundage HM III (2020) Seasonal presence of Atlantic Sturgeon and sharks at Cape Hatteras, a large continental shelf constriction to coastal migration. Marine Coast Fish 12:308–321
Secor DH (2002) Atlantic Sturgeon fisheries and stock abundances during the late nineteenth century. In Van Winkle W, Anders PJ, Secor DH, Dixon DA (eds) Biology, management, and protection of North American sturgeon. American Fisheries Society, Symposium 28, Bethesda, MA, pp 89–97
Stein AB, Friedland KD, Sutherland M (2004) Atlantic Sturgeon marine bycatch and mortality on the continental shelf of the northeast United States. North Am J Fish Manag 24:171–183
Taylor AD, Ohashi K, Sheng J, Litvak MK (2016) Oceanic distribution, behaviour, and a winter aggregation area of adult Atlantic Sturgeon, Acipenser oxyrinchus oxyrinchus, in the Bay of Fundy, Canada. PLoS ONE 11(4):e0152470. https://doi.org/10.1371/journal.pone.0152470
USFWS (U.S. Fish and Wildlife Service) and NMFS (National Marine Fisheries Service) (1996) Policy regarding the recognition of distinct vertebrate population segments under the Endangered Species Act. Fed Reg 61:4722–4725
Vine JR, Kanno Y, Holbrook SC, Post WC, Peoples BK (2019) Using side-scan sonar and N-mixture modeling to estimate Atlantic Sturgeon spawning migration abundance. North Am J Fish Manag 39:939–950
Waldman JR, Grunwald C, Stabile J, Wirgin I (2002) Impacts of life history and biogeography on the genetic stock structure of Atlantic sturgeon Acipenser oxyrinchus oxyrinchus, Gulf sturgeon A. oxyrinchus desotoi, and shortnose sturgeon A. brevirostrum. J Appl Ichthyol 18:509–518
Waldman JR, King T, Savoy T, Maceda L, Grunwald C, Wirgin I (2013) Stock origins of subadult and adult Atlantic Sturgeon, Acipenser oxyrinchus, in a non-natal estuary, Long Island Sound. Estuar Coasts 36(2):257–267
Waldman J, Alter SE, Peterson D, Maceda L, Roy N, Wirgin I (2019) Contemporary and historical effective population sizes of Atlantic sturgeon Acipenser oxyrinchus oxyrinchus. Conserv Genet 20:167–184
Wickham H (2016) ggplot2: elegant graphics for data analysis. Springer-Verlag, New York. https://doi.org/10.1007/978-3-319-24277-4
Wilkinson T, Wiken E, Bezaury-Creel J, Hourigan T, Agardy T, Herrmann H, Janishevski L, Madden C, Morgan L, Padilla M (2009) Marine ecoregions of North America. Commission for Environmental Cooperation, Montreal, QC
Wirgin I, Breece MW, Fox DA, Maceda L, Wark KW, King T (2015a) Origin of Atlantic Sturgeon collected off the Delaware coast during spring months. North Am J Fish Manag 35(1):20–30
Wirgin I, Maceda L, Grunwald C, King TL (2015b) Population origin of Atlantic sturgeon Acipenser oxyrinchus oxyrinchus by-catch in US Atlantic coast fisheries. J Fish Biol 86(4):1251–1270
Wirgin I, Maceda L, Waldman JR, Wehrell S, Dadswell M, King T (2012) Stock origin of migratory Atlantic Sturgeon in Minas Basin, Inner Bay of Fundy, Canada, determined by microsatellite and mitochondrial DNA analyses. Trans Am Fish Soc 141(5):1389–1398
Wirgin I, Roy NK, Maceda L, Mattson MT (2018) DPS and population origin of subadult Atlantic Sturgeon in the Hudson River. Fish Res 207:165–170
Acknowledgements
We extend a special thank you to the many individuals and agencies who have provided tissue samples from Atlantic sturgeon encountered in the field. We specifically thank the Northeast Fisheries Observer Program, Delaware Department of Natural Resources and Environmental Control, New York State Department of Environmental Conservation, Bill Post, Ramsey Noble, Carter Griggs, Gabriel Irigaray, Kirk Moore, Noelle Mathies, Jay Russo, Tracy Massey, Craig Marcusson, Maddie Speirs, April Deacy, Desiree Nuckols, Matthew Fisher, John O’Herron, Normandeau Associates, Guy Verreault, Gail Wippelhauser, Jerre Mohler, Hal Brundage, Jason Kahn, Christian Hager, Albert Spells, Matt Balazik, Michael Loeffler, Darren Wray, Corbet Norwood, Doug Peterson, Adam Fox, Mark Fritts, and Carter Watterson. Without their contributions, it would be intractable to draw inferences over such a large spatial scale. The genetic baseline and microsatellite panel used in this study reflects substantial contributions by the late Dr. Tim King, who helped advance our understanding of stock structure in Atlantic sturgeon and was a mentor to several coauthors. Ike Wirgin (New York University) and Tanya Darden (South Carolina Department of Natural Resources) shared tissue samples and genotypic data which substantially enhanced our analysis. We apologize to anyone we may have inadvertently missed who contributed to this effort over the many years of collections and laboratory research. The Greater Atlantic Regional Fisheries Office and Southeast Regional Office of the National Marine Fisheries Service provided funding to USGS conduct this research. Use of trade, product, or firm names does not imply endorsement by the U.S. Government.
Funding
This study was funded by the National Marine Fisheries Service through an interagency agreement to the U.S. Geological Survey.
Author information
Authors and Affiliations
Contributions
DCK designed the study, performed statistical analyses, and drafted the manuscript. BAL, RJ, and ME located samples, performed laboratory analyses, and contributed to manuscript preparation, SLW distilled an agency report and turned it into a viable manuscript and performed GIS and ggplot wizardry.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kazyak, D.C., White, S.L., Lubinski, B.A. et al. Stock composition of Atlantic sturgeon (Acipenser oxyrinchus oxyrinchus) encountered in marine and estuarine environments on the U.S. Atlantic Coast. Conserv Genet 22, 767–781 (2021). https://doi.org/10.1007/s10592-021-01361-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-021-01361-2