Abstract
Obesity represents an important risk factor for prostate cancer, driving more aggressive disease, chemoresistance, and increased mortality. White adipose tissue (WAT) overgrowth in obesity is central to the mechanisms that lead to these clinical observations. Adipose stromal cells (ASCs), the progenitors to mature adipocytes and other cell types in WAT, play a vital role in driving PCa aggressiveness. ASCs produce numerous factors, especially chemokines, including the chemokine CXCL12, which is involved in driving EMT and chemoresistance in PCa. A greater understanding of the impact of WAT in obesity-induced progression of PCa and the underlying mechanisms has begun to provide opportunities for developing interventional strategies for preventing or offsetting these critical events. These include weight loss regimens, therapeutic targeting of ASCs, use of calorie restriction mimetic compounds, and combinations of compounds as well as specific receptor targeting strategies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Prostate cancer (PCa) is the most frequently diagnosed non-skin cancer and is the second most common cause of cancer death in men in the United States (US). Approximately 1 in 8 men will be diagnosed with PCa during their lifetime and about 1 in 41 will die of PCa. It is estimated that ~ 268,490 new cases of PCa will be diagnosed and ~ 34,500 PCa-related deaths will occur in the US in 2022. Concurrently, obesity is a major public health crisis, with 42–46% of men over age 40 with obesity [1]. While there is a high prevalence of both obesity and PCa among men, data associating obesity and PCa risk are inconclusive [2, 3]. Although most men with localized PCa are cancer-free for 5 years after radical prostatectomy (RP), 20–30% of these men show signs of biochemical recurrence (BCR) after the 5-year mark [4, 5], and recently reported studies have shown obese men, particularly with androgen-responsive PCa and who rapidly gained weight after RP, have increased risk of metastatic progression, development of BCR, and decreased overall survival [6]. Men with obesity and PCa are at increased risk of lymphedema, positive surgical margins, BCR, and fatal PCa [7,8,9]. Being overweight at the time of diagnosis also increases the likelihood of dying from PCa by 50% [10]. Weight gain after RP is associated with higher risk of BCR [11, 12] and PCa-specific mortality [13, 14]. The role of weight loss after RP is less clear with observational data suggesting a lower risk of BCR [11] while other data show increased risk of death, likely due to reverse causality [15]. Detriments associated with being overweight/obese at RP include longer operative times, higher blood loss during RP, and slower recovery to continence and potency [9]. Also, RP is more difficult to perform in men with higher BMIs [9]. In contrast, several studies have shown that men with a smaller waist-to-hip ratio, lower BMI, and low body fat have low-volume PCa in comparison to men with high-volume PCa [16, 17]. Since the prevalence of obesity is increasing continuously [18], a clear knowledge of the mechanism(s) underlying disease aggressiveness as well as interventions designed to disrupt the obesity-PCa link are urgently needed to reduce morbidity and mortality associated with PCa.
2 Mechanisms associated with obesity and PCa progression
A number of mechanisms have been suggested for the effects of obesity on PCa progression. These include hyperinsulinemia, higher levels of growth factors in the circulation, inflammation and increased inflammatory mediators such as cytokines and chemokines, as well as changes in levels of steroid hormones and adiponectin as well as other factors (e.g., changes in microbiome, angiogenesis) [19, 20]. Obesity and particularly abdominal obesity are associated with abnormal and excessive expansion of white adipose tissue (WAT). Multiple studies have confirmed the link between PCa progression and WAT overgrowth [21,22,23]. In addition, excessive WAT expansion leads to a pathological state of chronic inflammation thought to play a leading role in the obesity-cancer link [24,25,26]. Although WAT was considered previously as an inactive organ for fat storage, numerous recent studies now confirm its secretory and endocrine function to serve an essential physiological role and maintenance of various signaling events [27, 28]. Adipocytes are by far the major component of WAT along with vascular endothelial cells; immune cells including monocytes, macrophages, fibroblasts, pericytes, and stem; and importantly adipocyte progenitor cells. The mesenchymal adipocyte progenitor cells with stem cell-like properties are known as adipose stromal cells (ASCs) and together with other cells in the WAT, produce numerous adipokines [26, 29] such as various cytokines (IL-1α, IL-1β, IL-6, TNFα), chemokines (CCL2, CCL3, CCL5, CCL7, CXCL1, CXCL8, CXCL12, CXCL16), growth factors (IGF-1), leptin, steroid hormones, and angiogenesis-related factors (VEGFA, VEGFB, VEGFC) [30,31,32]. These factors are associated with various essential cell signaling pathways for cancer development and progression and their higher secretion by WAT during obesity leads to progression of various cancers including PCa.
As noted above, in addition to ASCs, other cells of the WAT may also play essential roles in driving PCa progression. These include adipocytes, leukocytes (including various immune cells such as macrophages), and lymphocytes that are expanded in obesity [33]. For example, obesity is associated with increased accumulation of macrophages in WAT and these cells can further transform to cancer-associated macrophages and are recruited in tumor stroma [34, 35]. In addition, macrophages in obese conditions acquire a more proinflammatory phenotype and secrete various proinflammatory mediators leading to chronic low-grade inflammation, growth promotion, and angiogenesis [35]. The inflammatory states of WAT also contribute to activation of T-lymphocytes leading to secretion of various proinflammatory chemokines and cytokines such as TNF-α, IL-6, IL-1α, CXCL2, and various angiogenesis factors including VEGF and MMPs [33]. Finally, adipocytes in the WAT are also involved in cancer progression [36]. Adipocytes in peri prostatic WAT (ppWAT) come in direct contact with cancer cells and upon lipolysis, they serve as the energy source for cancer cells [36,37,38].
ppWAT is the fat layer that surrounds the prostate surface and plays an essential role in cancer progression [39,40,41,42,43]. During the cancer progression, invasion of cancer cells into the ppWAT of the tumor microenvironment leads to tumor-adipocyte crosstalk and facilitates PCa aggressiveness and leads to poor prognosis [43,44,45]. It was reported that the number of pre-adipocytes is higher in ppWAT of PCa patients compared to BPH patients [46]. These pre-adipocytes are also present in larger quantities in ppWAT compared to visceral WAT [47] and their interaction with cancer cells leads to formation of cancer-associated adipocytes that contribute to development of aggressive PCa [48, 49]. Taken together, the various cell types in the WAT/ppWAT that are increased in number and activated during obesity and that interact either directly or by secreting various mediators ultimately drive aggressive cancer.
3 ASCs as important drivers of PCa progression in obesity
ASCs constitute an important cellular component of periprostatic WAT (ppWAT), increase in number in obesity, and are mobilized from their perivascular niche. The recruitment of ASCs into the tumor microenvironment in obesity leads to increased cancer cell proliferation and tumor angiogenesis [50]. The migrated ASCs in the tumor microenvironment increase tumor viability by enhancing the tumor vasculature which provides oxygen and essential nutrients while removing toxic metabolites [51]. ppWAT proliferation during obesity is linked with a gene expression profile that stimulates proliferation, inhibits apoptosis and lipolysis, and increases matrix remodeling, lipid metabolism, and metastasis leading to increased PCa mortality [48, 52, 53]. Numerous animal studies have shown that ASCs, which are expanded in obesity, migrate from WAT to adjacent tumors [51, 54,55,56,57,58,59]. Once there, they become part of the cell population in the tumor stroma [51]. Trafficking of WAT-derived ASCs into tumors of obese PCa patients has been linked with decreased survival [60]. Recent studies have also shown that human PCa cell–derived CXCL1 serves as a ligand for CXCR1 and CXCR2 receptors on ASC [60] and that activation of this CXCL1-CXCR1 signaling axis in obesity drives recruitment of ASC to the tumor microenvironment [60].
Recent studies have also shown that ASCs play several roles in PCa progression stages. In this regard, paracrine signals for ASCs lead to anti-apoptotic, mitogenic, angiogenic, and immunosuppressive signals that together promote cancer progression [54, 61, 62]. ASCs can acquire perivascular localization which promotes tumor vascularization [51]. MCP-1 secreted by ASCs [63] recruit macrophages [64] that also contribute to PCa progression [65] as well as effects on T cells that can inhibit anti-tumor immune responses [35, 63, 66]. ASCs can facilitate remodeling of ECM leading to stimulation of cancer cell invasiveness through alterations in mechanotransduction and desmoplasia [67]. Studies in mice with grafted PCa tumors showed differentiation of ASCs into adipocytes further stimulating growth of nearby cancer cells [68]. The reported effects of ASCs on cancer progression [51, 57, 58] have been reproduced by several research groups [69,70,71]. In addition, ASCs have the ability to stimulate metastasis [70,71,72,73,74] and therapy resistance of multiple cancer types [75,76,77,78]. Recent studies using co-culture model systems have also revealed that ASCs induce EMT in PCa cells [79]. Obesity has also been shown to induce EMT in a mouse PCa allograft tumor model [64]. Notably, in this study, WAT invasion into the tumor microenvironment was observed. These latter studies also provided evidence for involvement of ASC in oxidative stress signaling in cancer cells [79] and have further clarified the induction of cancer cell invasiveness and chemoresistance mediated by ASCs.
4 WAT and ASC-derived factors in obesity-driven PCa progression
A number of adipokines and cytokines and their role in obesity-driven PCa have been previously reviewed by us and others [27, 30, 31, 80,81,82,83,84]. Here, we will focus the discussion mainly on chemokines (both CXC and CC chemokines) and on lipids implicated in obesity-driven PCa progression.
4.1 CXC chemokines
CXCL12 and its receptors CXCR4 and CXCR7
Mouse models have proven invaluable in understanding the role of obesity on cancer progression, including PCa [85,86,87,88,89]. We have shown, using the HiMyc mouse PCa model, that obesity drives disease progression while continuous calorie restriction (cCR) inhibits disease progression [85]. Obese HiMyc mice exhibited a dramatic infiltration of inflammatory cells and elevated inflammatory gene expression compared to mice on control and cCR diets [85, 86]. Further analyses of the prostate tumor microenvironment in obese HiMyc mice support the hypothesis that CXCL12 derived from ASCs stimulates invasiveness of PCa cells and plays an important role in obesity-induced PCa progression [79, 86]. CXCL12, an extensively studied chemokine, acts as a potent chemotactic factor for hematopoietic cells [90, 91] and is involved in metastatic progression of several cancer types including PCa [reviewed in 92]. CXCL12 binds with its receptors CXCR4 and CXCR7 to modulate various downstream cell signaling events [reviewed in 92]. For example, when CXCL12 binds with CXCR4, it induces trimeric G protein signaling and stimulates numerous downstream signaling events including PI3K/Akt, MAPK (ERK and JNK), and Src/STAT3, leading to increased invasiveness and migration of cancer cells [92,93,94,95,96].
Although CXCR7 was initially considered a decoy receptor due to lack of a specific G protein recruiting motif, more recent evidence suggests that binding of CXCL12 with CXCR7 stimulates downstream signaling, including MAP-kinase signaling pathway, by recruiting β-arrestin and subsequent ligand-receptor internalization [97]. CXCR4 and CXCR7 are expressed in cells either individually or together [92]. It is important to note that when co-expressed, they can form both homo and heterodimers; the heterodimers appear to play an essential role in modulating downstream signaling [97,98,99]. CXCL12 signaling is associated with progression of several cancers, including prostate, breast, pancreatic, and lung cancers [reviewed in 92]. Recent studies have also shown that CXCL12/CXCR4 signaling activates epidermal growth factor receptor family members [93, 100, 101]. Wang et al. [102] further reported that higher CXCR7 expression in human PCa cells was associated with more aggressive tumors. In addition, higher CXCR7 levels led to increased expression of pro-angiogenic factors (IL-8 and VEGF), activation of Akt, and were associated with increased xenograft tumor growth.
CXCL12 and its receptors have also been linked to metastasis of PCa to bone and are considered a poor prognostic marker [103,104,105,106]. CXCL12/CXCR4 has been shown to participate in localizing PCa to the bone marrow which can be blocked by neutralizing antibody to CXCR4 [107]. Bone marrow adipose tissue (BMAT) is present in the bone marrow space and bone tissue and constitutes approximately 10% of total adipose tissue in humans and also plays an important role in cancer progression including PCa [108]. Although BMAT shares some phenotypic characteristics with other WAT depots and serves as an energy storage in addition to secreting various adipokines and fatty acids, it is actually increased in young calorie restricted mice and in lean anorexia nervosa patients [109,110,111]. Several recent studies have confirmed the importance of BMAT in driving cancer progression by promoting survival, proliferation, migration, and invasion of various cancer cells [112, 113]. In particular, Herroon et al. showed that BMAT stimulated PCa growth in bone marrow by induction of fatty acid binding protein 4 [112]. Furthermore, CXCL1 and CXCL2 derived from BMAT are also involved in osteolysis in metastatic PCa [114]. Finally, a population of CXCL12+ pre-adipocyte like stromal cells has been identified in bone marrow that expresses CXCL12 and regulates marrow adipogenesis [115]. These CXCL12-producing stromal cells in bone marrow may also participate in driving PCa aggressiveness and metastatic disease.
We recently reported that treatment of cultured mouse PCa cells (HMVP2 cells isolated from HiMyc mice) with CXCL12 significantly increased migration, invasion, and activated several signaling events associated with development and progression of PCa [86]. We further showed that inhibition of both CXCL12 receptors (CXCR4 and CXCR7) attenuated the effect of CXCL12 on PCa progression [86]. Collectively, these studies suggest that CXCL12 is highly expressed and secreted from ppWAT and ASCs in obesity and plays an important role in PCa progression.
CXCL1 and CXCL8 and their receptors CXCR1 and CXCR2
CXCL1 and CXCL8 are two additional chemotactic chemokines secreted by a variety of cells in the WAT and involved in progression of cancers including PCa [116,117,118]. Both chemokines have also been shown to induce migration, angiogenesis, metastatic progression and PCa aggressiveness [119, 120]. CXCL1 and CXCL8 share the common receptors CXCR1 and CXCR2 and Zhang et al. showed that both chemokines facilitated the migration of ASCs that express CXCR1 and CXCR2 into the tumor microenvironment [60]. Multiple animal and human studies also have confirmed higher CXCL1 and CXCL8 expression in obesity and high-grade PCa [120, 121]. Numerous cytokines and chemokines (CXCL12, TNF-α, IL-1, and IL-6) that are highly upregulated in obesity further stimulate the expression of CXCL8 [122, 123]. Binding of ligand with CXCR1/CXCR2 causes activation of numerous cell signaling events mediated via pertussis toxin-sensitive G protein signaling [124]. This includes dissociation of GTP-bound Gα subunit from Gβγ subunit leading to activation of phospholipase C (PLC) and protein kinase C (PKC) and subsequent cytosolic mobilization of Ca2+ from endoplasmic reticulum [124, 125]. In addition, activation of CXCR1/2 leads to induction of PI3K/AKT, RhoA, Src kinases, FAK, and MAPKs including ERK1/2, JNK, and p38 ultimately playing important roles in regulation of various cellular processes including migration and chemotaxis [126,127,128,129]. In addition to binding with CXCL1 and CXCL8, CXCR2 also binds with the chemokine CXCL5 that is released from various cell types in the SVF of adipose tissue [130, 131]. Expression of CXCL5 is also elevated in obese patients as well in prostate tumor samples and is associated with activation of JAK/STAT, PI3K, and MAPK signaling pathways that ultimately leads to higher migration, invasion, and cell survival [131, 132]. In summary, CXCL1, CXCL5, and CXCL8 acting through their receptors CXCR1 and CXCR2 play important roles in obesity-induced PCa progression.
CXCL13 and its receptor CXCR5
The chemokine CXCL13 secreted by adipocytes and other cells of the TME plays important role in cancer progression including induction of cell survival, migration, invasion, angiogenesis, and metastasis [133,134,135]. CXCL13 triggers its effect by binding with the G protein-coupled receptor CXCR5. Like other G protein-coupled chemokine receptors, CXCL13/CXCR5 interaction leads to G protein subunit dissociation and subsequent induction of different downstream signaling events including activation of ERK1/2, JNK, Src, FAK, and PI3K/AKT signaling [136, 137]. CXCL13 is also involved in the induction of PCa cell survival, migration, invasion, and metastasis [136,137,138]. Hypoxia increases the expression of both CXCL13 and CXCR5 in adipocytes and WAT isolated from mice on obesity inducing diet showed increased expression of CXCL13 [134]. Taken together, CXCL13 along with its receptor CXCR5 also play an important role in obesity-induced PCa progression.
CXCL16 and its receptor CXCR6
CXCL16 is another chemokine that has been shown to be involved in obesity and cancer and is expressed in various cells including adipocytes and tumor cells. The effect of CXCL16 is mediated through its interaction with the GPCR, CXCR6 which when bound with CXCL16 leads to activation of multiple downstream cell signaling pathways such as PI3K/AKT, NFκB, ERK1/2, and GSK3β. In addition to binding with CXCR6, recent studies have also shown an alternative transmembrane-mediated signaling called “Inverse signaling” [139]. In this type of signaling, membrane-bound CXCL16 acts like a receptor and CXCL16 in soluble form binds with the transmembrane CXCL16 and induces activation of AKT and ERK and stimulates various biological effects including induction of proliferation and reduction of apoptosis [139]. WAT of obese mice showed increased CXCL16 levels compared to non-obese mice and mice undergoing calorie restriction appear to show lower level of CXCL16 [140]. CXCL16 also involved in PCa metastasis and development of chemoresistance [141, 142].
4.2 CC chemokines
In addition to CXC chemokines, various CC chemokines derived from WAT are also involved in obesity-related PCa cancer progression. Several of these are summarized in this section.
CCL2
CCL2 is expressed in various tissues including adipose tissue and preadipocytes [143,144,145] and is associated with macrophage mobilization in adipose tissue, chronic inflammation particularly inflammation of the adipose tissue, and is considered a promising biomarker of human PCa [146]. Various proinflammatory mediators such as TNFα, interferon γ (IFNγ), IL-1, IL-6, transforming growth factor β (TGFβ), vascular endothelial growth factor (VEGF), lipopolysaccharide (LPS), and platelet-derived growth factor (PDGF) increase the expression of CCL2 [147]. The expression of CCL2 also increases with higher BMI and weight loss decreses circulatory levels of CCL2 [148]. Like many CXC chemokines, CCL2 also mediates its effect by binding with a GPCR, CCR2. CCR2 serves as the receptor for CCL2 and this receptor ligand interaction leads to activation of different cell signaling pathways including STAT3, PI3K, and MAPK leading to increased survival, anti-apotosis, therapy resistance, and tumor metastasis [149, 150].
CCL3, CCL4, and CCL5
The chemokines CCL3, CCL4, and CCL5 share common receptor, namely CCR5, in addition to their other binding receptors and all three ligands are highly expressed in WAT and serum of obese mice compared to their non-obese counterparts [86, 151, 152]. WAT of obese mice also showed higher CCL3 level compared to lean mice. We also found higher CCL5 level in ppWAT of obese HiMyc mice compared to control mice [86]. CLL4 and CCL5 are also associated with promoting PCa progression through activation of the STAT3 signaling pathway [153, 154].
CCL7
CCL7 or monocyte chemoattractant protein-3 (MCP-3) is a chemokine produced by various cells including adipocytes and cancer cells and functions as chemoattractant for immune cells [36, 155,156,157,158]. There are several receptors for CCL7, for example, CCR1, CCR2, CCR3, CCR5, and CCR10 [159, 160]. During obesity, adipocytes including periprostatic adipocytes produce higher amount of CCL7 [155, 161] leading to increased migration of CCR3 expressing cancer cells including PCa cells into the ppWAT of tumor microenvironments [36]. The expression of CCL7 is higher in the adipose tissue of patients with severe obesity compared to their lean counterparts and it is also associated with increased migratory potential of PCa cells by induction of ERK and high MMP3 level mediated through CCR3 [162]. The level of CCR3 also shown to be higher in adipose tissue of obese subjects and in PCa patients and involved in poor prognosis [36].
4.3 Fatty acids
Long chain fatty acid (LCFA) is mainly stored in WAT [163] and undergoes lipolysis due to activation of catecholamines and β3 adrenergic signaling [164, 165]. Hydrolysis of triglycerides via various lipases such as adipose triglyceride lipase, monoacylglycerol lipase, and hormone sensitive lipase is responsible for the release of free LCFAs [166]. It was reported that monoacylglycerol lipase which serves in the final step of lipolysis is highly expressed in androgen-independent PCa cells (DU145 and PC3) compared to androgen-dependent cells (LNCaP) [167]. Monoacylglycerol lipase is also associated with a gene signature that involves EMT and cancer stem cell-like properties and inhibition of this enzyme reduces PCa aggressiveness [167]. Animal studies demonstrated that loss of body fat during aggressive cancers and growth of tumors stimulates lipolysis in adipocytes [168,169,170]. Lower free LCFA levels reduce cancer pathogenicity [166] due to decreased lipolysis. Several cancers including pancreatic, breast, and ovarian cancers show increased invasive potential due to alteration in adipose tissue lipolysis in tumor microenvironment [171,172,173]. Cancer cells acquire LCFAs derived from lipolysis of adipocytes [38] that leads to accelerated proliferation and invasion of breast cancer cells [74, 174, 175]. LCFAs from adipocyte also cause AMPK-mediated induction of mitochondrial biogenesis [168, 176]. Recent studies in animal models have shown the potential importance of adipocyte-derived LCFAs for PCa progression [177, 178].
5 Brown adipose tissue (BAT) in PCa
BAT, named based on the color, was initially thought to be present only in infants. However, recent advancement of imaging techniques has led to accurate identification and distribution of BAT in human adults [179]. BAT plays an essential role in non-shivering thermogenesis. Recent evidence suggests that formation of beige/brown adipose tissue from WAT may play a role in PCa progression [35, 180]. Browning of WAT is associated with higher expression of PPARγ or PGC-1 as well as activation of the sympathetic nervous system, leading to stimulation of β-adrenergic signaling [181, 182]. Although the role of WAT in PCa progression has been extensively studied and is well recognized, the impact of BAT and the browning of WAT in PCa progression are not clearly understood at the present time. Further studies are clearly warranted to understand the role of BAT in PCa progression.
6 Strategies to offset obesity effects on PCa progression
6.1 Calorie restriction
Calorie restriction (CR) is a very potent and broad acting dietary intervention for weight loss and cancer inhibition in experimental studies, including studies in non-human primates [183]. Using the HiMyc mouse model of PCa, a 30% continuous CR (cCR) diet significantly decreased in situ adenocarcinoma (AC) formation and completely inhibited the progression of AC to locally invasive PCa compared to both overweight control and obese mice [85]. This was associated with a marked reduction in mRNA expression of various inflammatory and angiogenesis-related genes. cCR further decreased various important cell signaling pathways in prostate lobes such as AKT/mTORC1, STAT3, NFκB, and CXCL12/CXCR4/CXCR7 signaling involved in cancer progression and metastasis [85, 86]. Galet et al. also showed that cCR decreased growth of 22Rv1 xenograft tumors by modulating IGF-1 signaling [184]. Similarly, cCR with a no carbohydrate ketogenic diet prolonged survival and reduced LAPC-4 PCa tumor growth in SCID mice by modulating insulin and IGF-1 axis compared to mice on a Western diet [185].
Translation of CR studies to human cancer prevention strategies is of great importance because of the high prevalence of overweight/obesity and the essential role of obesity on metabolic changes and progression of multiple cancers, including PCa [186]. Several funded clinical trials have addressed whether the potential benefits of cCR reported in preclinical studies translate to humans. The Comprehensive Assessment of Long-Term Effects of Reducing Intake of Energy (CALERIE) Study evaluated the impacts of a 2-year cCR regimen (25% less energy than controls) in nonobese, healthy individuals [187]. Results from this study indicated that humans showed many similar cCR-induced endocrine and metabolic changes observed in rodents and monkeys. The results are consistent with studies in patients at high risk for breast cancer showing better reduction in insulin, metabolic, and growth factor signaling pathways by cCR or 2 days/week of calorie restriction (hereafter referred to as intermittent CR or iCR) [188,189,190]. The observed metabolic effects of iCR are particularly important, since it is much easier, attainable, and more feasible to restrict calories for 2 days/week, than to chronically restrict calories. While iCR data show better insulin and body fat/weight reduction in the short-term in women [188], effects in older men and long-term sustainability of the weight/fat loss and metabolic improvements are unknown.
In contrast, clinical trials show that intentional weight loss of greater than 5% by cCR with exercise in men prior to RP is feasible. In an RCT, 25–30% cCR with exercise led to 5.5% of weight loss (95% CI, 4–7%) from baseline to RP (mean = 5 weeks) [191]. Furthermore, 26 weeks post-RP, the cCR group maintained or kept losing to a net loss of 11% initial body weight (95% CI, 8–14%). The cCR regimen decreased insulin, leptin:adiponectin ratio (LAR), and visceral adipose tissue (VAT) associated with inflammation (Hamilton-Reeves, unpublished data). In an earlier study, a novel finding of lower level of CXCL12, CXCR7, and CXCR4 comparing biopsy to RP tissue with cCR was observed [192, 193], which agrees with other evidence of altered gene expression of CXCR4, CCL2, and IGF-2 receptor in prostate tissue with 5% weight loss by cCR diet in four men prior to RP [194]. VAT, LAR, and chronic inflammation are parameters to which cCR weight loss studies report inconsistencies with a higher degree of variability. Henning et al. showed significant decrease in insulin and body fat, without alteration in LAR or trunk fat mass in a similar study design with combined cCR (1200–1500 kcal/day) and 60 min/day exercise [195]. Alternatively, Demark-Wahnefried et al. showed no change in insulin or body fat but a significant decrease in leptin, with intentional weight loss by cCR of a 1000 kcal/day deficit and additional 250–500 kcal expenditure through aerobic exercise in men prior to RP compared to the control group [196]; participants’ microbiota changed in response to types of foods but not due to cCR [197]. Wilson et al. showed a significant decrease in VAT and fat mass in a retrospective analysis of patients following cCR of 750–1000 kcal/day and 90 min/day of aerobic exercise for up to 12 weeks before RP [198]. Although cCR improves health overall, it is possible that iCR may have as great or greater impact on molecular pathways and may be easier for adherence; thus, efficacy in men with obesity and PCa needs to be tested.
Data from some human studies suggest that compared to cCR, iCR shows greater increases in insulin sensitivity [188, 189], faster weight loss [188], greater reductions in fat mass [188], better retention of lean mass [199], alterations in gut microbiota [200, 201], inflammation reduction [188, 202], and better LAR [188], while systematic reviews deem iCR and cCR equivalent for weight/fat loss and metabolic changes [203, 204] although primarily only women were studied [205]. While it seems plausible that iCR may allow more flexibility for adherence and for avoiding weight regain, long-term effects in humans are unknown. Currently, there are no published human iCR intervention studies in patients with cancer. It is also unknown if greater metabolic benefits occur when the two fasting days are consecutive or if the carry-over effects of spontaneous CR observed on normal eating days persist long term [188, 189]. Restricting calories mostly from carbohydrates for 2 days/week is appealing for simplicity and also to mitigate risk by allowing sufficient protein intake on restricted days to prevent muscle/function loss [206]. Some benefits may be independent from weight loss, with one study in healthy men showing improved insulin sensitivity from iCR without weight/fat loss [207], which is particularly interesting since men and women have different acute effects to fasting [208]. iCR may make greater improvements than cCR on the cancer biomarkers that contribute to PCa progression and BCR.
Since one of the major impacts of obesity is the extensive alteration of WAT and its secretory status, it is important to look at the role of CR on WAT. Several studies reported that cCR provides beneficial effects by modulating the WAT. This includes a significant decrease in adiposity leading to lower level of various secretory components, biosynthesis of fatty acid, mitochondrial biogenesis, and alteration in adipose tissue metabolism. In this regard, using the HiMyc mouse model of PCa, 30% cCR significantly decreases ppWAT mass as well as complete inhibition of the infiltration of adipocytes into the prostate gland compared to control or obese mice [86]. Miller et al. recently showed that cCR in mice significantly decreased adiposity and high molecular weight adiponectin levels. CR also activated downstream signaling of adiponectin receptor such as increased expression of peroxisome proliferator-activated receptor gamma coactivator 1 alpha (PGC-1α), SIRT1, and nicotinamide phosphoribosyl transferase (NAMPT) in adipose tissue of cCR mice [209]. These authors also found a greater oxidative metabolic state of the adipose tissue microenvironment [209]. In addition, adipose tissue lipid metabolism was altered with cCR in mice with differences in free fatty acid (FFA), monounsaturated fatty acid (MUFA), and polyunsaturated fatty acid (PUFA) as well as decreases in desaturation, elongation, and omega-3 metabolism in cCR mice [209]. cCR also increased mitochondrial biogenesis, mitochondrial DNA, and oxidative phosphorylation in WAT [210,211,212]. Decreases in IL-15 signaling and TNF-α expression were also observed in WAT of cCR mice [213]. AMPK and SIRT1 are modulated by cCR and both of them act as positive regulators of PGC-1α. In addition, recent studies showed that induction of PGC-1α in WAT during cCR is mediated by sterol regulatory element-binding protein 1c (SREBP1c) and fibroblast growth factor 21 (FGF21) [214, 215]. Taken together, cCR produces an array of changes in WAT including metabolic reprogramming leading to reversal of the harmful effect of obesity.
6.2 Physical activity
Exercise training is a potential strategy to offset PCa progression. Data from several studies have demonstrated that a higher level of physical activity is associated with a lower risk of PCa progression [216] and cancer-specific mortality [217,218,219]. There is growing evidence linking the molecular pathways underlying these associations. Postulated mechanisms of exercise training include indirect effects by modulating cytokines, improving glucose regulation, reducing insulin resistance, reducing adiposity, and modulating hormones [220]. Physical activity may also directly suppress tumors and the tumor microenvironment by lowering chronic inflammation [221], altering tumor vascularization, modulating growth factors, and improving immune function [220, 222].
Physical activity is prescribed with recommendations for the frequency, intensity, time/duration, and type of activity; however, the understanding of how these different variables inhibit carcinogenesis is not fully understood. Exercise physiology elicits acute effects after a single bout of exercise and different effects after weeks of consistent exercise training. These differences are summarized in a review by Kim et al. comparing the changes in PCa cell lines treated with serum obtained after a single exercise session showing reductions in AKT, mTOR, ERK, and mitogenic activity to the changes in PCa cell lines treated with serum obtained after consistent long-term exercise training showing increased p53, reduced PCNA, and increased apoptosis [223]. Interestingly, after single bouts of exercise, several groups have reported increased circulating CXCL12 directly after moderate to vigorous exercise [224,225,226,227,228,229,230] while no changes in plasma CXCL12 were found after a single bout of strength training [231, 232] suggesting intensity and type may impact this outcome in the plasma. Effects of acute or prolonged exercise on the prostate tissue expression of CXCL12 or related pathways are unknown. Wedell-Neergaard et al. demonstrated that IL-6 signaling is required for exercise to reduce visceral adipose tissue mass; IL-6 increases acutely after a bout of exercise [233]. In general, prolonged exercise interventions have been purported to lower inflammation and improve immune editing, and although these outcomes are known to be highly variable, a meta-analysis by Khosravi et al. of 27 exercise intervention trials in cancer (prostate cancer = 4) reported that exercise training decreased circulating pro-inflammatory markers CRP and TNF [234]. As proposed by Rocha-Rodriques et al., exercise training may also alter the tumor microenvironment by the effects on the secretions of adipokines from WAT/ppWAT and on secretions of myokines by skeletal muscle and the crosstalk between the two [235]; however, these effects have not yet been tested in patients with PCa or in animal models of PCa. Idorn et al. proposed the idea of using exercise to help hone T cells for immunotherapy through its effects of promoting infiltration of immune cells into the tumor [236]. These data open several future exciting areas of research, including adjuvant exercise with cancer therapy.
A meta-analysis by Bourke et al. found that exercise interventions across all stages of PCa improved cancer-specific quality of life and cancer-specific fatigue in men with up to 6 months of follow-up [237]. A more recent meta-analysis by Anderson et al. confirmed that exercise improved cancer-specific quality of life with a greater magnitude than previously reported and significantly improved cardiovascular fitness as well [238]. An interesting sub-study of INTERVAL-GAP4 trial [239] which uses moderate to high intensity exercise training for 6 months showed tumor suppressive effects in the supervised exercise arm compared to the control arm when serum obtained from participants with metastatic PCa was added to the human PCa cell line, DU145 [240]. In summary, exercise training is a potential strategy to offset the progression of PCa with or without obesity and there are several potential areas of research opportunities to further evaluate the impact of frequency, intensity, duration, and type of exercise training to benefit patients with PCa at various stages of disease.
6.3 Calorie restriction mimetics (CRM)
Although numerous studies have established the potential benefit of cCR from animal studies to humans for a large number of diseases including obesity and cancer, it is extremely difficult to maintain a certain level of cCR for a long period of time to get a desired clinical outcome. In addition, since there are no prolonged cCR studies in human, it is still unclear whether long-term dietary restrictions might cause any adverse side effects. An alternative approach is the use of agents that can provide the similar benefits of CR without altering the dietary manipulations. This has led to recently developed concept of CRM to describe the agents or molecules that mimic the beneficial effects of CR. In order to be an ideal candidate, CRM agents are proposed [241] to have several key characteristics including (i) exert the similar physiological, metabolic, and hormonal effects produced by CR; (ii) not significantly decrease long-term food intake; (iii) produce similar levels of stress response pathways and show protection against a variety of stressors seen in CR; and (iv) reduce age-related diseases as well as inflammation and autoimmunity. Based on these criteria, several agents have been identified as CRM agents including 2- Deoxy-D-Glucose, metformin, rapamycin, peroxisome proliferator-activated receptor (PPAR) activators, and certain natural phytochemicals such as resveratrol, curcumin, EGCG, and quercetin. Here we will briefly discuss the potential benefits of some of the CRM agents in reversing the effects of obesity and PCa progression.
Resveratrol
The plant-derived polyphenol resveratrol is found in grapes, berries, peanuts, and red wine. Numerous studies reported the potential health benefits of resveratrol including its effects as anti-cancer, anti-aging, anti-diabetes, and as an anti-obesity agent [242]. Recently, resveratrol has been considered as an ideal CRM agent for its role in metabolic effects and effects on adipose tissue. In this regard, Lagouge et al. showed that dietary administration of resveratrol significantly decreased the body weight and WAT deposits in mice maintained on a high-fat diet [243]. Several other reported studies also confirmed that resveratrol decreases body weight and WAT mass in mice on HFD [244,245,246]. Although there are only a few human trials to evaluate the effect of resveratrol in comparison to vast majority animal studies, the beneficial effect of resveratrol was still evident. For example, administration of resveratrol at a dose of 500 mg 3 × /day for 3 months in patients with metabolic syndrome [247] showed significant decrease in body weight, BMI, fat content, and waist circumference (WC) compared to patient with placebo group. Similarly, several other clinical trials found positive outcomes of resveratrol intake and body weight reduction as well as decrease in adipose tissue mass [248,249,250]. In addition to its effect on obesity and WAT mass, resveratrol has shown potential benefit as an anti-cancer agent for several cancers including PCa [251,252,253,254,255]. Preclinical studies using both cultured cells and transgenic animal models of PCa showed that resveratrol decreases PCa cell growth and metastasis by modulating multiple cell signaling pathways [251, 252, 256, 257]. For example, Seth et al. reported that oral administration of resveratrol decreases xenografted PCa tumor growth and lung metastasis by modulating AKT/miRNA-21 pathway [251].
Curcumin
Curcumin is another plant-derived polyphenolic compound with CRM activity. It is present in the rhizome of the plant Curcuma longa, widely consumed as a food additive and several studies showed potential anti-obesity effect of curcumin. Dietary administration of curcumin showed significant weight loss in both DIO and ob/ob mice [258]. Curcumin also showed reduction of macrophage infiltration in WAT, reduced NFκB activity, and increased adiponectin production [258]. Shao et al. similarly reported that curcumin feeding reversed the HFD-induced weight gain in mice [259]. In addition, curcumin administration increased insulin sensitivity, decreased oxidative and inflammatory signaling in adipocytes, and decreased lipogenic gene expression in the liver [259]. Additionally, Ejaz et al. reported that supplementation of 500 mg/kg of curcumin per day in 22 kcal% HFD significantly reduced body weight, adipose tissue mass, and microvessel density in adipose tissue along with decreased expression of VEGF and VEGFR2 compared to mice fed in HFD [260]. Numerous cell culture and preclinical animal model studies also showed the ability of curcumin to inhibit growth and progression of various cancers including PCa [261]. In addition, several completed and ongoing clinical studies with curcumin show promise for its effect on cancer treatment [261].
Quercetin
Quercetin is a widely studied polyphenolic compound present in a diverse range of fruits and vegetables including apple, berries, onion, broccoli, cherries, and red grapes [262]. Quercetin is considered as a CRM because of its potential to modulate many cellular changes that are similar to changes observed with cCR. For example, several studies have reported that administration of quercetin causes activation of AMPK, reduction in inflammation, induction of autophagy, and lifespan increase [263,264,265,266,267]. In mice maintained in 40 kCal% HFD, dietary supplementation of quercetin (0.33%) produced significant decrease in body weight gain, lipid level in serum, and liver without modulation of energy intake [268]. In another study, oral administration of 100 μg/day quercetin produced a 29% decrease in body weight of C57BL6 mice. Additionally, quercetin together with exercise showed further decrease in body weight with 43.5% reduction compared to control mice [269]. Recently, Pei et al. reported that dietary administration of 1% quercetin decreased inflammation in brown adipose tissue (BAT) in HFD-induced obese mice by modulation of NFκB and SIRT1 signaling [270]. Furthermore, Dong et al. showed that 0.1% quercetin decreased body weight gain in C57BL/6 mice fed a HFD and improved glucose intolerance and insulin sensitivity. Quercetin administration also decreased expression of proinflammatory cytokines and activated AMPK and SIRT1 in epididymis adipose tissue of HFD-fed mice [265]. In comparison to a vast majority of animal studies, there are only a few human studies [271, 272] to evaluate the potential CRM benefits of quercetin. For example, in a randomized, double-blind, placebo-controlled study, Lee et al. reported that daily intake of onion peel extract capsules containing 100 mg of quercetin significantly decreased the body weight, BMI, and percentage of body fat in subjects in the treated group compared to the subjects in the placebo group [271]. Thus, in order to fully elucidate the beneficial effect of quercetin as a CRM agent, further properly designed human studies are necessary.
Metformin
Worldwide, metformin is a highly prescribed agent used for the treatment of type II diabetes and several studies identified its potential as a CRM agent [88, 241, 273, 274]. In this regard, several animal studies showed anti-cancer, anti-aging, and lifespan extension after treatment with metformin. For example, Martin-Montalvo et al. reported that long-term administration of 0.1% metformin in drinking water increased survival of mice compared to control and was associated with better physical performance, higher insulin sensitivity, lowered LDL and cholesterol levels, and activation of AMPK [273]. In another study, Anisimov et al. showed that female outbred SHR mice treated with 100 mg/kg of metformin in drinking water produced a decrease in body weight and extension of the mean lifespan by 37.8% compared to control mice [274]. Numerous published data in preclinical models including our own studies suggested the potential anti-cancer effect of metformin in part due to modulation of multiple cell signaling pathways as well as its effect on body weight reduction [88, 275, 276]. Although there is a growing interest for the potential anti-cancer and anti-obesity effects of metformin, the results in human studies have been inconsistent. A review by Lev M Berstein nicely pointed out some of the factors related to the inconsistency [277]. However, future, more carefully designed long-term human clinical studies will be needed to provide definitive information related to the use of metformin as a CRM agent in treating cancer and obesity. In this regard, multiple clinical studies are currently ongoing in several countries with metformin to clarify its role in cancer and obesity.
Rapamycin
Rapamycin is a potent bacterial derived macrolide compound used widely for its role as immunosuppressant treatment for organ transplant recipients as well as in autoimmune disorders. Rapamycin possesses many characteristics of cCR and numerous reports including our own studies showed the potential anti-aging, anti-obesity, anti-cancer, and lifespan extension effect of rapamycin in animal models [275, 278,279,280]. Chang et al. showed that rapamycin treatment decreased body weight and epidydimal fat pad in obese C57BL/6 mice maintained on HFD compared to control treatment [279]. We also showed that rapamycin treatment reduced TPA-induced skin tumor promotion as well as reduction of PCa progression in HiMyc mouse model with complete inhibition of adenocarcinoma formation in this mouse model [275, 280]. Similar to cCR, rapamycin exerts it effect by inhibiting mTORC1 signaling pathways with subsequent decrease in activation of downstream signaling pathways, including p70S6K and ribosomal S6 kinase [85, 275], and might serve as a potential CRM agent for treating obesity and cancer including PCa.
Phytochemical combinations as CRM
The use of combinations of agents including phytochemical combinations for various diseases such as cancer has received considerable recent interest due to the potential to (i) provide synergistic effects, (ii) target multiple cell signaling pathways leading to better beneficial effects, and (iii) produce less toxic effects due to smaller dose requirements. Several recent studies including our own studies with combinations of agents have shown effects on obesity as well as modulation of key signaling such as AMPK [275, 281]. In this regard, one recent study by Lee et al. showed that treatment with a combination of phytochemicals containing p-octopamine hydrochloride, p-synephrine, and hispidulin significantly lowered the body weight of HFD-induced obese mice compared to control mice with concomitant decrease in the expression of various adipogenic markers such as CCAAT/enhancer-binding protein alpha (C/EBPα), CCAAT/enhancer-binding protein beta (C/EBPβ), and peroxisome proliferator-activated receptor gamma (PPARγ) [281]. Furthermore, combinations of several phytochemicals (resveratrol, genistein, and quercetin) together with vitamin D showed decreased body weight and adiposity compared to control diet in aged ovariectomized rats. We also showed that combinations of curcumin + ursolic acid, resveratrol + ursolic acid, curcumin + resveratrol, and metformin + rapamycin produced significant decreases in tumor growth in a PCa allograft tumor model as well as in the HiMyc mouse model of PCa [275, 282]. In addition, these combinations of agents have shown induction of AMPK, a signaling pathway previously shown to be activated by cCR and considered as a marker of CRM [275, 282]. Taken together, the concept of using combinations of agents including phytochemical combinations has shown promise and should be considered as potential CRM approach for treating obesity-related cancer, including PCa.
6.4 Targeting ASC in WAT and the tumor microenvironment
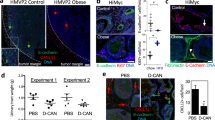
As noted above, ASCs are believed to play a major role in obesity-driven PCa progression. Targeting ASC in WAT and the tumor stroma could be a strategy for offsetting the effects of obesity on PCa progression. By screening a combinatorial library in vivo, Kolonin and colleagues isolated a cyclic peptide called WAT7 with a sequence of CSWKYWFGEC that attacks ASCs that are found in both WAT and tumor stroma [283, 284]. This peptide was attached to another peptide (KLAKKLAK) to generate the “Hunter-Killer” peptide and ultimately targeting and killing ASCs. This hybrid peptide, called D-CAN, has been used to selectively deplete ASCs in vivo [284, 285]. In this regard, D-CAN has been tested in vivo for suppression of EMT and tumor growth in obese mice [79]. D-CAN injected into mice harboring HMVP2 allograft tumors led to significant inhibition of tumor growth, reduced periprostatic WAT, and a reversion of EMT-related changes in the tumors [79]. In addition, targeting ASC has been shown to reverse chemoresistance in both mouse allograft and human xenograft PCa models [79]. Further studies have revealed that targeting ASCs with D-CAN in vivo in obese HiMyc mice significantly reduced the number of CXCL12 producing cells in tumor stroma and reversed EMT changes associated with obesity [178]. These and other data have provided strong evidence that ASCs are the source of CXCL12 involved in driving PCa progression in obesity and that targeting ASCs could be a novel strategy for offsetting the effects of obesity on PCa.
6.5 Targeting FAs
In PCa, higher lipogenesis is a characteristic feature of more advanced PCa [286]. As noted above, reduced levels of free LCFA have been shown to reduce cancer pathogenicity [166] due to decreased lipolysis. These observations have prompted investigation into whether lipogenesis enzymes might represent therapeutic targets in obesity and PCa [166]. FA synthase (FASN) inhibitors have been shown to reduce cancer cell viability and have demonstrated promise in studies using animal models of PCa [287]. Unfortunately, systemic toxicity has been a problem with lipogenesis inhibitors as well as the observation that cancer cells can switch to extracellular LCFA utilization thereby resisting the effects of these types of inhibitors [288]. Further development of agents and research in this area is warranted to determine whether targeting FAs is a viable approach for treating obesity-driven PCa progression.
6.6 Targeting chemokine receptors
As noted above, various chemokines have been shown to play a role in obesity-driven PCa progression. ASCs produce CXCL12 which has been linked, at least in part, to PCa aggressiveness in obesity [79, 86, 178]. CXCL12 binds to both CXCR4 and CXCR7 [92]. Both CXCR4 and CXCR7 are highly expressed in various tumors [103, 104, 289, 290] and have been proposed as important players in cancer progression [291, 292]. CXCL12 binding to CXCR4 activates several signaling pathways related to cell survival, angiogenesis, and cell proliferation, such as JAK-STAT, PI3K-AKT-NFκB, and MEK-ERK signaling [293,294,295]. CXCR7 is an atypical chemokine receptor, and its role in cancer progression has been somewhat controversial [95, 296]. Previously, we reported a correlation of CXCR4 and CXCR7 expression in obesity with increased HiMyc tumor growth and also demonstrated the importance of CXCL12 signaling for cancer cell invasiveness as noted above [86]. Notably, blockade of either CXCR4 (either genetically or pharmacologically) or CXCR7 (pharmacologically) suppresses obesity-driven EMT and PCa progression in mouse models [297].
A summary of the various strategies discussed above, to offset the effects of obesity on PCa, is presented in Table 1.
7 Conclusions and future perspectives
While not completely defined, it is clear from current research that ppWAT and cells within this compartment, especially ASCs, are critical drivers of PCa progression in obesity. Multiple mechanisms likely play a role in obesity-driven PCa progression but secreted factors, especially chemokines appear to play a significant role. In addition to chemokines, other molecules such as cytokines, angiogenesis factors, growth factors, and lipids [27, 30, 84] also likely contribute to the effects of obesity with the possibility that multiple pathways that are activated may synergize to drive PCa aggressiveness and chemoresistance. The accumulating knowledge of mechanisms has led to research into strategies to offset the effects of obesity on PCa and other cancers and include weight loss interventions, CRMs, and more targeted approaches to block specific signaling pathways. Figure 1 summarizes some of the relevant mechanisms associated with obesity-driven PCa progression and potential prevention/treatment strategies discussed in this review. Further understanding of the cellular and molecular mechanisms underlying the activity of ppWAT and in particular ASCs, as well as other components of the ppWAT, will allow further development of strategies for their inhibition and will facilitate the development of strategies to reduce obesity-associated PCa progression.
Obesity-driven PCa progression and intervention strategies. Obesity is associated with adipocyte hypertrophy and hyperplasia along with the presence of higher number of other cells such as ASCs, immune cells, and endothelial cells. These increased number of cells produce and secrete excessive amount of various signaling molecules and factors including various inflammatory chemokines (CXCL1, CXCL8, CXCL12 etc.), cytokines (IL-1, IL-6, TNFα), and growth factors (IGF-1, VEGF). These factors modulate multiple cell signaling pathways such as PI3K/AKT/mTOR, JAK/STAT3, ERK/JNK/p38 MAPK, NFκB, insulin/IGF-1, and MAPK signaling leading to higher EMT, proliferation, survival, angiogenesis, inflammation, migration, invasion, and tumor progression that ultimately results in development and progression of aggressive PCa. There are several interventional strategies such as use of CR and CRM, physical activity, and approaches that target ASCs, FAs, and chemokine receptors that could be utilized to reverse the effect of obesity on aggressive PCa
References
Hales, C. M., Carroll, M. D., Fryar, C. D., & Ogden, C. L. (2020). Prevalence of obesity and severe obesity among adults: United States, 2017–2018. NCHS Data Brief, (360), 1–8.
Nomura, A. M. (2001). Body size and prostate cancer. Epidemiologic Reviews, 23(1), 126–131.
Porter, M. P., & Stanford, J. L. (2005). Obesity and the risk of prostate cancer. Prostate, 62(4), 316–321.
Roehl, K. A., Han, M., Ramos, C. G., Antenor, J. A., & Catalona, W. J. (2004). Cancer progression and survival rates following anatomical radical retropubic prostatectomy in 3,478 consecutive patients: Long-term results. Journal of Urology, 172(3), 910–914. https://doi.org/10.1097/01.ju.0000134888.22332.bb
Pound, C. R., Partin, A. W., Eisenberger, M. A., Chan, D. W., Pearson, J. D., & Walsh, P. C. (1999). Natural history of progression after PSA elevation following radical prostatectomy. JAMA, 281(17), 1591–1597.
Flavin, R., Zadra, G., & Loda, M. (2011). Metabolic alterations and targeted therapies in prostate cancer. The Journal of Pathology, 223(2), 283–294. https://doi.org/10.1002/path.2809
Cao, Y., & Ma, J. (2011). Body mass index, prostate cancer–specific mortality, and biochemical recurrence: A systematic review and meta-analysis. Cancer Prevention Research, 4(4), 486–501. https://doi.org/10.1158/1940-6207.capr-10-0229
Freedland, S. J., Terris, M. K., Presti, J. C., Jr., Amling, C. L., Kane, C. J., Trock, B., et al. (2004). Obesity and biochemical outcome following radical prostatectomy for organ confined disease with negative surgical margins. Journal of Urology, 172(2), 520–524. https://doi.org/10.1097/01.ju.0000135302.58378.ae
Campeggi, A., Xylinas, E., Ploussard, G., Ouzaid, I., Fabre, A., Allory, Y., et al. (2012). Impact of body mass index on perioperative morbidity, oncological, and functional outcomes after extraperitoneal laparoscopic radical prostatectomy. Urology, 80(3), 576–584. https://doi.org/10.1016/j.urology.2012.04.066
Haque, R., Van Den Eeden, S. K., Wallner, L. P., Richert-Boe, K., Kallakury, B., Wang, R., et al. (2014). Association of body mass index and prostate cancer mortality. Obesity Research & Clinical Practice, 8(4), e374-381. https://doi.org/10.1016/j.orcp.2013.06.002
Joshu, C. E., Mondul, A. M., Menke, A., Meinhold, C., Han, M., Humphreys, E. B., et al. (2011). Weight gain is associated with an increased risk of prostate cancer recurrence after prostatectomy in the PSA era. Cancer Prevention Research (Philadelphia, Pa.), 4(4), 544–551. https://doi.org/10.1158/1940-6207.capr-10-0257
Whitley, B. M., Moreira, D. M., Thomas, J. A., Aronson, W. J., Terris, M. K., Presti, J. C., Jr., et al. (2011). Preoperative weight change and risk of adverse outcome following radical prostatectomy: Results from the Shared Equal Access Regional Cancer Hospital database. Prostate Cancer and Prostatic Diseases, 14(4), 361–366. https://doi.org/10.1038/pcan.2011.42
Chen, Q., Chen, T., Shi, W., Zhang, T., Zhang, W., Jin, Z., et al. (2016). Adult weight gain and risk of prostate cancer: A dose-response meta-analysis of observational studies. International Journal of Cancer, 138(4), 866–874. https://doi.org/10.1002/ijc.29846
Bonn, S. E., Wiklund, F., Sjolander, A., Szulkin, R., Stattin, P., Holmberg, E., et al. (2014). Body mass index and weight change in men with prostate cancer: Progression and mortality. Cancer Causes and Control, 25(8), 933–943. https://doi.org/10.1007/s10552-014-0393-3
Troeschel, A. N., Hartman, T. J., Jacobs, E. J., Stevens, V. L., Gansler, T., Flanders, W. D., et al. (2020). Postdiagnosis body mass index, weight change, and mortality from prostate cancer, cardiovascular disease, and all causes among survivors of nonmetastatic prostate cancer. Journal of Clinical Oncology, 38(18), 2018–2027. https://doi.org/10.1200/JCO.19.02185
Allott, E. H., Masko, E. M., & Freedland, S. J. (2013). Obesity and prostate cancer: Weighing the evidence. European Urology, 63(5), 800–809. https://doi.org/10.1016/j.eururo.2012.11.013
Spitz, M. R., Strom, S. S., Yamamura, Y., Troncoso, P., Babaian, R. J., Scardino, P. T., et al. (2000). Epidemiologic determinants of clinically relevant prostate cancer. International Journal of Cancer, 89(3), 259–264.
Eheman, C., Henley, S. J., Ballard-Barbash, R., Jacobs, E. J., Schymura, M. J., Noone, A. M., et al. (2012). Annual Report to the Nation on the status of cancer, 1975–2008, featuring cancers associated with excess weight and lack of sufficient physical activity. Cancer, 118(9), 2338–2366. https://doi.org/10.1002/cncr.27514
Zadra, G., Photopoulos, C., & Loda, M. (2013). The fat side of prostate cancer. Biochimica Et Biophysica Acta-Molecular and Cell Biology of Lipids, 1831(10), 1518–1532. https://doi.org/10.1016/J.Bbalip.2013.03.010
Blando, J., Saha, A., Kiguchi, K., & DiGiovanni, J. (2013). Obesity, inflammation and prostate cancer. In A. J. Dannenberg, & N. A. Berger (Eds.), Obesity, inflammation and cancer (Vol. 7, pp. 235–256, Energy Balance and Cancer, Vol. 7). Springer.
De Nunzio, C., Albisinni, S., Freedland, S. J., Miano, L., Cindolo, L., Finazzi Agro, E., et al. (2013). Abdominal obesity as risk factor for prostate cancer diagnosis and high grade disease: A prospective multicenter Italian cohort study. Urologic Oncology, 31(7), 997–1002. https://doi.org/10.1016/j.urolonc.2011.08.007
van Kruijsdijk, R. C., van der Wall, E., & Visseren, F. L. (2009). Obesity and cancer: The role of dysfunctional adipose tissue. Cancer Epidemiology, Biomarkers & Prevention, 18(10), 2569–2578. https://doi.org/10.1158/1055-9965.EPI-09-0372
Lysaght, J., van der Stok, E. P., Allott, E. H., Casey, R., Donohoe, C. L., Howard, J. M., et al. (2011). Pro-inflammatory and tumour proliferative properties of excess visceral adipose tissue. Cancer Letters, 312(1), 62–72. https://doi.org/10.1016/j.canlet.2011.07.034
Coussens, L. M., & Werb, Z. (2002). Inflammation and cancer. Nature, 420(6917), 860–867. https://doi.org/10.1038/nature01322
Cowey, S., & Hardy, R. W. (2006). The metabolic syndrome: A high-risk state for cancer? American Journal of Pathology, 169(5), 1505–1522. https://doi.org/10.2353/ajpath.2006.051090
Park, J., Euhus, D. M., & Scherer, P. E. (2011). Paracrine and endocrine effects of adipose tissue on cancer development and progression. Endocrine Reviews, 32(4), 550–570. https://doi.org/10.1210/er.2010-0030
Baillargeon, J., & Rose, D. P. (2006). Obesity, adipokines, and prostate cancer (review). International Journal of Oncology, 28(3), 737–745.
Trayhurn, P., & Wood, I. S. (2004). Adipokines: Inflammation and the pleiotropic role of white adipose tissue. British Journal of Nutrition, 92(3), 347–355.
Ouchi, N., Parker, J. L., Lugus, J. J., & Walsh, K. (2011). Adipokines in inflammation and metabolic disease. Nature Reviews Immunology, 11(2), 85–97. https://doi.org/10.1038/nri2921
Khandekar, M. J., Cohen, P., & Spiegelman, B. M. (2011). Molecular mechanisms of cancer development in obesity. Nature Reviews Cancer, 11(12), 886–895. https://doi.org/10.1038/nrc3174
Roberts, D. L., Dive, C., & Renehan, A. G. (2010). Biological mechanisms linking obesity and cancer risk: New perspectives. Annual Review of Medicine, 61, 301–316. https://doi.org/10.1146/annurev.med.080708.082713
Grossmann, M. E., Ray, A., Nkhata, K. J., Malakhov, D. A., Rogozina, O. P., Dogan, S., et al. (2010). Obesity and breast cancer: Status of leptin and adiponectin in pathological processes. Cancer and Metastasis Reviews, 29(4), 641–653. https://doi.org/10.1007/s10555-010-9252-1
Johnson, A. R., Milner, J. J., & Makowski, L. (2012). The inflammation highway: Metabolism accelerates inflammatory traffic in obesity. Immunological Reviews, 249(1), 218–238. https://doi.org/10.1111/j.1600-065X.2012.01151.x
Weisberg, S. P., McCann, D., Desai, M., Rosenbaum, M., Leibel, R. L., & Ferrante, A. W., Jr. (2003). Obesity is associated with macrophage accumulation in adipose tissue. The Journal of Clinical Investigation, 112(12), 1796–1808. https://doi.org/10.1172/JCI19246
Lengyel, E., Makowski, L., DiGiovanni, J., & Kolonin, M. G. (2018). Cancer as a matter of fat: The crosstalk between adipose tissue and tumors. Trends in Cancer, 4(5), 374–384. https://doi.org/10.1016/j.trecan.2018.03.004
Laurent, V., Guerard, A., Mazerolles, C., Le Gonidec, S., Toulet, A., Nieto, L., et al. (2016). Periprostatic adipocytes act as a driving force for prostate cancer progression in obesity. [Research Support, Non-U.S. Gov’t]. Nature Communications, 7, 10230. https://doi.org/10.1038/ncomms10230
Palm, W., & Thompson, C. B. (2017). Nutrient acquisition strategies of mammalian cells. Nature, 546(7657), 234–242. https://doi.org/10.1038/nature22379
Ye, H., Adane, B., Khan, N., Sullivan, T., Minhajuddin, M., Gasparetto, M., et al. (2016). Leukemic stem cells evade chemotherapy by metabolic adaptation to an adipose tissue niche. Cell Stem Cell, 19(1), 23–37. https://doi.org/10.1016/j.stem.2016.06.001
Ribeiro, R., Monteiro, C., Silvestre, R., Castela, A., Coutinho, H., Fraga, A., et al. (2012). Human periprostatic white adipose tissue is rich in stromal progenitor cells and a potential source of prostate tumor stroma. [Research Support, Non-U.S. Gov’t]. Experimental Biology and Medicine, 237(10), 1155–1162. https://doi.org/10.1258/ebm.2012.012131
van Roermund, J. G., Hinnen, K. A., Tolman, C. J., Bol, G. H., Witjes, J. A., Bosch, J. L., et al. (2011). Periprostatic fat correlates with tumour aggressiveness in prostate cancer patients. BJU International, 107(11), 1775–1779. https://doi.org/10.1111/j.1464-410X.2010.09811.x
Toren, P., & Venkateswaran, V. (2014). Periprostatic adipose tissue and prostate cancer progression: New insights into the tumor microenvironment. Clinical Genitourinary Cancer, 12(1), 21–26. https://doi.org/10.1016/j.clgc.2013.07.013
Finley, D. S., Calvert, V. S., Inokuchi, J., Lau, A., Narula, N., Petricoin, E. F., et al. (2009). Periprostatic adipose tissue as a modulator of prostate cancer aggressiveness. Journal of Urology, 182(4), 1621–1627. https://doi.org/10.1016/j.juro.2009.06.015
Nassar, Z. D., Aref, A. T., Miladinovic, D., Mah, C. Y., Raj, G. V., Hoy, A. J., et al. (2018). Peri-prostatic adipose tissue: The metabolic microenvironment of prostate cancer. BJU International, 121(Suppl 3), 9–21. https://doi.org/10.1111/bju.14173
Walz, J., Burnett, A. L., Costello, A. J., Eastham, J. A., Graefen, M., Guillonneau, B., et al. (2010). A critical analysis of the current knowledge of surgical anatomy related to optimization of cancer control and preservation of continence and erection in candidates for radical prostatectomy. European Urology, 57(2), 179–192. https://doi.org/10.1016/j.eururo.2009.11.009
Sung, M. T., Eble, J. N., & Cheng, L. (2006). Invasion of fat justifies assignment of stage pT3a in prostatic adenocarcinoma. Pathology, 38(4), 309–311. https://doi.org/10.1080/00313020600820914
Xie, H., Li, L., Zhu, G., Dang, Q., Ma, Z., He, D., et al. (2016). Correction: Infiltrated pre-adipocytes increase prostate cancer metastasis via modulation of the miR-301a/androgen receptor (AR)/TGF-beta1/Smad/MMP9 signals. Oncotarget, 7(50), 83829–83830. https://doi.org/10.18632/oncotarget.13913
Ribeiro, R., Monteiro, C., Silvestre, R., Castela, A., Coutinho, H., Fraga, A., et al. (2012). Human periprostatic white adipose tissue is rich in stromal progenitor cells and a potential source of prostate tumor stroma. Experimental Biology and Medicine (Maywood, N.J.), 237(10), 1155–1162. https://doi.org/10.1258/ebm.2012.012131
Ribeiro, R., Monteiro, C., Cunha, V., Oliveira, M. J., Freitas, M., Fraga, A., et al. (2012). Human periprostatic adipose tissue promotes prostate cancer aggressiveness in vitro. Journal of Experimental & Clinical Cancer Research, 31, 32. https://doi.org/10.1186/1756-9966-31-32
Ribeiro, R. J., Monteiro, C. P., Cunha, V. F., Azevedo, A. S., Oliveira, M. J., Monteiro, R., et al. (2012). Tumor cell-educated periprostatic adipose tissue acquires an aggressive cancer-promoting secretory profile. Cellular Physiology and Biochemistry, 29(1–2), 233–240. https://doi.org/10.1159/000337604
Takeda, K., Sowa, Y., Nishino, K., Itoh, K., & Fushiki, S. (2015). Adipose-derived stem cells promote proliferation, migration, and tube formation of lymphatic endothelial cells in vitro by secreting lymphangiogenic factors. Annals of Plastic Surgery, 74(6), 728–736. https://doi.org/10.1097/SAP.0000000000000084
Zhang, Y., Daquinag, A. C., Amaya-Manzanares, F., Sirin, O., Tseng, C., & Kolonin, M. G. (2012). Stromal progenitor cells from endogenous adipose tissue contribute to pericytes and adipocytes that populate the tumor microenvironment. Cancer Research, 72(20), 5198–5208. https://doi.org/10.1158/0008-5472.CAN-12-0294
Himbert, C., Delphan, M., Scherer, D., Bowers, L. W., Hursting, S., & Ulrich, C. M. (2017). Signals from the adipose microenvironment and the obesity-cancer link-A systematic review. Cancer Prevention Research (Philadelphia, Pa.), 10(9), 494–506. https://doi.org/10.1158/1940-6207.CAPR-16-0322
Ribeiro, R., Monteiro, C., Catalan, V., Hu, P., Cunha, V., Rodriguez, A., et al. (2012). Obesity and prostate cancer: Gene expression signature of human periprostatic adipose tissue. BMC Medicine, 10, 108. https://doi.org/10.1186/1741-7015-10-108
Kolonin, M. G., Evans, K. W., Mani, S. A., & Gomer, R. H. (2012). Alternative origins of stroma in normal organs and disease. Stem Cell Res., 8(2), 312–323. https://doi.org/10.1016/j.scr.2011.11.005
Bellows, C. F., Zhang, Y., Chen, J., Frazier, M. L., & Kolonin, M. G. (2011). Circulation of progenitor cells in obese and lean colorectal cancer patients. Cancer Epidemiology, Biomarkers & Prevention, 20(11), 2461–2468.
Bellows, C. F., Zhang, Y., Simmons, P. J., Khalsa, A. S., & Kolonin, M. G. (2011). Influence of BMI on level of circulating progenitor cells. Obesity, 19(8), 1722–1726. https://doi.org/10.1038/oby.2010.347
Zhang, Y., Daquinag, A., Traktuev, D. O., Amaya, F., Simmons, P. J., March, K. L., et al. (2009). White adipose tissue cells are recruited by experimental tumors and promote cancer progression in mouse models. Cancer Research, 69(12), 5259–5266.
Klopp, A. H., Zhang, Y., Solley, T., Amaya-Manzanares, F., Marini, F., Andreeff, M., et al. (2012). Omental adipose tissue-derived stromal cells promote vascularization and growth of endometrial tumors. Clinical Cancer Research, 18(3), 771–782. https://doi.org/10.1158/1078-0432.CCR-11-1916
Sirin, O., & Kolonin, M. G. (2013). Treatment of obesity as a potential complementary approach to cancer therapy. Drug Discovery Today, 11(12), 567–573.
Zhang, T., Tseng, C., Zhang, Y., Sirin, O., Corn, P. G., Li-Ning-Tapia, E. M., et al. (2016). CXCL1 mediates obesity-associated adipose stromal cell trafficking and function in the tumour microenvironment. Nature Communications, 7, 11674. https://doi.org/10.1038/ncomms11674
Peng, Y. C., Levine, C. M., Zahid, S., Wilson, E. L., & Joyner, A. L. (2013). Sonic hedgehog signals to multiple prostate stromal stem cells that replenish distinct stromal subtypes during regeneration. [Research Support, N.I.H., Extramural Research Support, Non-U.S. Gov’t]. Proceedings of the National Academy of Sciences, 110(51), 20611–20616. https://doi.org/10.1073/pnas.1315729110
Zhang, Y., & Kolonin, M. G. (2016). Cytokine signaling regulating adipose stromal cell trafficking. Adipocyte, 5(4), 369–374. https://doi.org/10.1080/21623945.2016.1220452
Kaplan, J. L., Marshall, M. A., McSkimming, C. C., Harmon, D. B., Garmey, J. C., Oldham, S. N., et al. (2015). Adipocyte progenitor cells initiate monocyte chemoattractant protein-1-mediated macrophage accumulation in visceral adipose tissue. Molecular Metabolism, 4(11), 779–794. https://doi.org/10.1016/j.molmet.2015.07.010
Murdoch, C., Muthana, M., Coffelt, S. B., & Lewis, C. E. (2008). The role of myeloid cells in the promotion of tumour angiogenesis. Nature Reviews Cancer, 8(8), 618–631. https://doi.org/10.1038/nrc2444
Iyengar, N. M., Brown, K. A., Zhou, X. K., Gucalp, A., Subbaramaiah, K., Giri, D. D., et al. (2017). Metabolic obesity, adipose inflammation and elevated breast aromatase in women with normal body mass index. Cancer Prevention Research, 10(4), 235–243. https://doi.org/10.1158/1940-6207.CAPR-16-0314
Hanahan, D., & Coussens, L. M. (2012). Accessories to the crime: Functions of cells recruited to the tumor microenvironment. Cancer Cell, 21(3), 309–322. https://doi.org/10.1016/j.ccr.2012.02.022
Lu, P., Weaver, V. M., & Werb, Z. (2012). The extracellular matrix: A dynamic niche in cancer progression. Journal of Cell Biology, 196(4), 395–406. https://doi.org/10.1083/jcb.201102147
Park, J., Morley, T. S., Kim, M., Clegg, D. J., & Scherer, P. E. (2014). Obesity and cancer--mechanisms underlying tumour progression and recurrence. [Research Support, N.I.H., Extramural Research Support, Non-U.S. Gov’t Research Support, U.S. Gov’t, Non-P.H.S. Review]. Nature Reviews Endocrinology, 10(8), 455–465. https://doi.org/10.1038/nrendo.2014.94
Kidd, S., Spaeth, E., Watson, K., Burks, J., Lu, H., Klopp, A., et al. (2012). Origins of the tumor microenvironment: Quantitative assessment of adipose-derived and bone marrow-derived stroma. PLoS ONE, 7(2), e30563. https://doi.org/10.1371/journal.pone.0030563
Orecchioni, S., Gregato, G., Martin-Padura, I., Reggiani, F., Braidotti, P., Mancuso, P., et al. (2013). Complementary populations of human adipose CD34+ progenitor cells promote growth, angiogenesis, and metastasis of breast cancer. [Research Support, Non-U.S. Gov’t]. Cancer Research, 73(19), 5880–5891. https://doi.org/10.1158/0008-5472.CAN-13-0821
Rowan, B. G., Gimble, J. M., Sheng, M., Anbalagan, M., Jones, R. K., Frazier, T. P., et al. (2014). Human adipose tissue-derived stromal/stem cells promote migration and early metastasis of triple negative breast cancer xenografts. PLoS ONE, 9(2), e89595. https://doi.org/10.1371/journal.pone.0089595
Martin-Padura, I., Gregato, G., Marighetti, P., Mancuso, P., Calleri, A., Corsini, C., et al. (2012). The white adipose tissue used in lipotransfer procedures is a rich reservoir of CD34+ progenitors able to promote cancer progression. Cancer Research, 72(1), 325–334. https://doi.org/10.1158/0008-5472.CAN-11-1739
Zhao, M., Dumur, C. I., Holt, S. E., Beckman, M. J., & Elmore, L. W. (2010). Multipotent adipose stromal cells and breast cancer development: Think globally, act locally. Molecular Carcinogenesis, 49(11), 923–927. https://doi.org/10.1002/mc.20675
Picon-Ruiz, M., Pan, C., Drews-Elger, K., Jang, K., Besser, A. H., Zhao, D., et al. (2016). Interactions between adipocytes and breast cancer cells stimulate cytokine production and drive Src/Sox2/miR-302b-mediated malignant progression. Cancer Research, 76(2), 491–504. https://doi.org/10.1158/0008-5472.CAN-15-0927
Nowicka, A., Marini, F. C., Solley, T. N., Elizondo, P. B., Zhang, Y., Sharp, H. J., et al. (2013). Human omental-derived adipose stem cells increase ovarian cancer proliferation, migration, and chemoresistance. PLoS ONE, 8(12), e81859. https://doi.org/10.1371/journal.pone.0081859
Salimian Rizi, B., Caneba, C., Nowicka, A., Nabiyar, A. W., Liu, X., Chen, K., et al. (2015). Nitric oxide mediates metabolic coupling of omentum-derived adipose stroma to ovarian and endometrial cancer cells. [Research Support, Non-U.S. Gov’t]. Cancer Research, 75(2), 456–4571. https://doi.org/10.1158/0008-5472.CAN-14-1337
Duong, M. N., Cleret, A., Matera, E. L., Chettab, K., Mathe, D., Valsesia-Wittmann, S., et al. (2015). Adipose cells promote resistance of breast cancer cells to trastuzumab-mediated antibody-dependent cellular cytotoxicity. Breast Cancer Research, 17, 57–63. https://doi.org/10.1186/s13058-015-0569-0
Houthuijzen, J. M., Daenen, L. G., Roodhart, J. M., & Voest, E. E. (2012). The role of mesenchymal stem cells in anti-cancer drug resistance and tumour progression. [Review]. British Journal of Cancer, 106(12), 1901–1906. https://doi.org/10.1038/bjc.2012.201
Su, F., Ahn, S., Saha, A., DiGiovanni, J., & Kolonin, M. G. (2019). Adipose stromal cell targeting suppresses prostate cancer epithelial-mesenchymal transition and chemoresistance. Oncogene, 38(11), 1979–1988. https://doi.org/10.1038/s41388-018-0558-8
Giovannucci, E., & Michaud, D. (2007). The role of obesity and related metabolic disturbances in cancers of the colon, prostate, and pancreas. Gastroenterology, 132(6), 2208–2225. https://doi.org/10.1053/j.gastro.2007.03.050
Giovannucci, E., Rimm, E. B., Colditz, G. A., Stampfer, M. J., Ascherio, A., Chute, C. C., et al. (1993). A prospective study of dietary fat and risk of prostate cancer. Journal of the National Cancer Institute, 85(19), 1571–1579.
Freedland, S. J., & Aronson, W. J. (2004). Examining the relationship between obesity and prostate cancer. Revista de Urología, 6(2), 73–81.
Strom, S. S., Wang, X., Pettaway, C. A., Logothetis, C. J., Yamamura, Y., Do, K. A., et al. (2005). Obesity, weight gain, and risk of biochemical failure among prostate cancer patients following prostatectomy. Clinical Cancer Research, 11(19 Pt 1), 6889–6894.
Frasca, F., Pandini, G., Sciacca, L., Pezzino, V., Squatrito, S., Belfiore, A., et al. (2008). The role of insulin receptors and IGF-I receptors in cancer and other diseases. Archives of Physiology and Biochemistry, 114(1), 23–37. https://doi.org/10.1080/13813450801969715
Blando, J., Moore, T., Hursting, S., Jiang, G., Saha, A., Beltran, L., et al. (2011). Dietary energy balance modulates prostate cancer progression in Hi-Myc mice. [Research Support, N.I.H., Extramural Research Support, Non-U.S. Gov’t]. Cancer Prevention Research, 4(12), 2002–2014. https://doi.org/10.1158/1940-6207.CAPR-11-0182
Saha, A., Ahn, S., Blando, J., Su, F., Kolonin, M. G., & DiGiovanni, J. (2017). Proinflammatory CXCL12-CXCR4/CXCR7 signaling axis drives Myc-induced prostate cancer in obese mice. Cancer Research. https://doi.org/10.1158/0008-5472.CAN-17-0284
Rossi, E. L., Khatib, S. A., Doerstling, S. S., Bowers, L. W., Pruski, M., Ford, N. A., et al. (2018). Resveratrol inhibits obesity-associated adipose tissue dysfunction and tumor growth in a mouse model of postmenopausal claudin-low breast cancer. Molecular Carcinogenesis, 57(3), 393–407. https://doi.org/10.1002/mc.22763
Checkley, L. A., Rho, O., Angel, J. M., Cho, J., Blando, J., Beltran, L., et al. (2014). Metformin inhibits skin tumor promotion in overweight and obese mice. Cancer Prevention Research (Philadelphia, Pa.), 7(1), 54–64. https://doi.org/10.1158/1940-6207.CAPR-13-0110
Moore, T., Beltran, L., Carbajal, S., Hursting, S. D., & DiGiovanni, J. (2012). Energy balance modulates mouse skin tumor promotion through altered IGF-1R and EGFR crosstalk. Cancer Prevention Research (Philadelphia, Pa.), 5(10), 1236–1246. https://doi.org/10.1158/1940-6207.CAPR-12-0234
Aiuti, A., Webb, I. J., Bleul, C., Springer, T., & Gutierrez-Ramos, J. C. (1997). The chemokine SDF-1 is a chemoattractant for human CD34+ hematopoietic progenitor cells and provides a new mechanism to explain the mobilization of CD34+ progenitors to peripheral blood. Journal of Experimental Medicine, 185(1), 111–120.
Secchiero, P., Celeghini, C., Cutroneo, G., Di Baldassarre, A., Rana, R., & Zauli, G. (2000). Differential effects of stromal derived factor-1 alpha (SDF-1 alpha) on early and late stages of human megakaryocytic development. Anatomical Record, 260(2), 141–147.
Hattermann, K., & Mentlein, R. (2013). An infernal trio: The chemokine CXCL12 and its receptors CXCR4 and CXCR7 in tumor biology. [Review]. Annals of Anatomy, 195(2), 103–110. https://doi.org/10.1016/j.aanat.2012.10.013
Conley-LaComb, M. K., Saliganan, A., Kandagatla, P., Chen, Y. Q., Cher, M. L., & Chinni, S. R. (2013). PTEN loss mediated Akt activation promotes prostate tumor growth and metastasis via CXCL12/CXCR4 signaling. Molecular Cancer, 12(1), 85. https://doi.org/10.1186/1476-4598-12-85
Rivat, C., Sebaihi, S., Van Steenwinckel, J., Fouquet, S., Kitabgi, P., Pohl, M., et al. (2014). Src family kinases involved in CXCL12-induced loss of acute morphine analgesia. Brain, Behavior, and Immunity, 38, 38–52. https://doi.org/10.1016/j.bbi.2013.11.010
Sun, X., Cheng, G., Hao, M., Zheng, J., Zhou, X., Zhang, J., et al. (2010). CXCL12 / CXCR4 / CXCR7 chemokine axis and cancer progression. [Research Support, N.I.H., Extramural Research Support, Non-U.S. Gov’t Review]. Cancer and Metastasis Reviews, 29(4), 709–722. https://doi.org/10.1007/s10555-010-9256-x
Duda, D. G., Kozin, S. V., Kirkpatrick, N. D., Xu, L., Fukumura, D., & Jain, R. K. (2011). CXCL12 (SDF1alpha)-CXCR4/CXCR7 pathway inhibition: An emerging sensitizer for anticancer therapies? Clinical Cancer Research, 17(8), 2074–2080. https://doi.org/10.1158/1078-0432.CCR-10-2636
Decaillot, F. M., Kazmi, M. A., Lin, Y., Ray-Saha, S., Sakmar, T. P., & Sachdev, P. (2011). CXCR7/CXCR4 heterodimer constitutively recruits beta-arrestin to enhance cell migration. Journal of Biological Chemistry, 286(37), 32188–32197. https://doi.org/10.1074/jbc.M111.277038
Levoye, A., Balabanian, K., Baleux, F., Bachelerie, F., & Lagane, B. (2009). CXCR7 heterodimerizes with CXCR4 and regulates CXCL12-mediated G protein signaling. [Research Support, Non-U.S. Gov’t]. Blood, 113(24), 6085–6093. https://doi.org/10.1182/blood-2008-12-196618
Luker, K. E., Gupta, M., & Luker, G. D. (2009). Imaging chemokine receptor dimerization with firefly luciferase complementation. [Research Support, N.I.H., Extramural Research Support, Non-U.S. Gov’t]. The FASEB Journal, 23(3), 823–834. https://doi.org/10.1096/fj.08-116749
Zhao, H. B., Tang, C. L., Hou, Y. L., Xue, L. R., Li, M. Q., Du, M. R., et al. (2012). CXCL12/CXCR4 axis triggers the activation of EGF receptor and ERK signaling pathway in CsA-induced proliferation of human trophoblast cells. [Clinical Trial Research Support, Non-U.S. Gov’t]. PLoS One, 7(7), e38375. https://doi.org/10.1371/journal.pone.0038375
McGinn, O. J., Marinov, G., Sawan, S., & Stern, P. L. (2012). CXCL12 receptor preference, signal transduction, biological response and the expression of 5T4 oncofoetal glycoprotein. [Research Support, Non-U.S. Gov’t]. Journal of Cell Science, 125(Pt 22), 5467–5478. https://doi.org/10.1242/jcs.109488
Wang, J., Shiozawa, Y., Wang, J., Wang, Y., Jung, Y., Pienta, K. J., et al. (2008). The role of CXCR7/RDC1 as a chemokine receptor for CXCL12/SDF-1 in prostate cancer. [Research Support, N.I.H., Extramural Research Support, U.S. Gov’t, Non-P.H.S.]. Journal of Biological Chemistry, 283(7), 4283–4294. https://doi.org/10.1074/jbc.M707465200
Akashi, T., Koizumi, K., Tsuneyama, K., Saiki, I., Takano, Y., & Fuse, H. (2008). Chemokine receptor CXCR4 expression and prognosis in patients with metastatic prostate cancer. Cancer Science, 99(3), 539–542. https://doi.org/10.1111/j.1349-7006.2007.00712.x
Sun, Y. X., Wang, J., Shelburne, C. E., Lopatin, D. E., Chinnaiyan, A. M., Rubin, M. A., et al. (2003). Expression of CXCR4 and CXCL12 (SDF-1) in human prostate cancers (PCa) in vivo. Journal of Cellular Biochemistry, 89(3), 462–473. https://doi.org/10.1002/jcb.10522
Mochizuki, H., Matsubara, A., Teishima, J., Mutaguchi, K., Yasumoto, H., Dahiya, R., et al. (2004). Interaction of ligand-receptor system between stromal-cell-derived factor-1 and CXC chemokine receptor 4 in human prostate cancer: A possible predictor of metastasis. Biochemical and Biophysical Research Communications, 320(3), 656–663. https://doi.org/10.1016/j.bbrc.2004.06.013
Jung, S. J., Kim, C. I., Park, C. H., Chang, H. S., Kim, B. H., Choi, M. S., et al. (2011). Correlation between chemokine receptor CXCR4 expression and prognostic factors in patients with prostate cancer. Korean Journal of Urology, 52(9), 607–611. https://doi.org/10.4111/kju.2011.52.9.607
Sun, Y. X., Schneider, A., Jung, Y., Wang, J., Dai, J., Wang, J., et al. (2005). Skeletal localization and neutralization of the SDF-1(CXCL12)/CXCR4 axis blocks prostate cancer metastasis and growth in osseous sites in vivo. Journal of Bone and Mineral Research, 20(2), 318–329. https://doi.org/10.1359/JBMR.041109
Cawthorn, W. P., Scheller, E. L., Learman, B. S., Parlee, S. D., Simon, B. R., Mori, H., et al. (2014). Bone marrow adipose tissue is an endocrine organ that contributes to increased circulating adiponectin during caloric restriction. Cell Metabolism, 20(2), 368–375. https://doi.org/10.1016/j.cmet.2014.06.003
Devlin, M. J., Cloutier, A. M., Thomas, N. A., Panus, D. A., Lotinun, S., Pinz, I., et al. (2010). Caloric restriction leads to high marrow adiposity and low bone mass in growing mice. Journal of Bone and Mineral Research, 25(9), 2078–2088. https://doi.org/10.1002/jbmr.82
Bredella, M. A., Fazeli, P. K., Miller, K. K., Misra, M., Torriani, M., Thomas, B. J., et al. (2009). Increased bone marrow fat in anorexia nervosa. Journal of Clinical Endocrinology and Metabolism, 94(6), 2129–2136. https://doi.org/10.1210/jc.2008-2532
Morris, E. V., & Edwards, C. M. (2016). Bone marrow adipose tissue: A new player in cancer metastasis to bone. Frontiers in Endocrinology (Lausanne), 7, 90. https://doi.org/10.3389/fendo.2016.00090
Herroon, M. K., Rajagurubandara, E., Hardaway, A. L., Powell, K., Turchick, A., Feldmann, D., et al. (2013). Bone marrow adipocytes promote tumor growth in bone via FABP4-dependent mechanisms. Oncotarget, 4(11), 2108–2123. https://doi.org/10.18632/oncotarget.1482
Templeton, Z. S., Lie, W. R., Wang, W., Rosenberg-Hasson, Y., Alluri, R. V., Tamaresis, J. S., et al. (2015). Breast cancer cell colonization of the human bone marrow adipose tissue niche. Neoplasia, 17(12), 849–861. https://doi.org/10.1016/j.neo.2015.11.005
Hardaway, A. L., Herroon, M. K., Rajagurubandara, E., & Podgorski, I. (2015). Marrow adipocyte-derived CXCL1 and CXCL2 contribute to osteolysis in metastatic prostate cancer. Clinical & Experimental Metastasis, 32(4), 353–368. https://doi.org/10.1007/s10585-015-9714-5
Matsushita, Y., Chu, A. K. Y., Ono, W., Welch, J. D., & Ono, N. (2021). Intercellular interactions of an adipogenic CXCL12-expressing stromal cell subset in murine bone marrow. Journal of Bone and Mineral Research, 36(6), 1145–1158. https://doi.org/10.1002/jbmr.4282
Hebert, C. A., & Baker, J. B. (1993). Interleukin-8: A review. Cancer Investigation, 11(6), 743–750. https://doi.org/10.3109/07357909309046949
Holmes, W. E., Lee, J., Kuang, W. J., Rice, G. C., & Wood, W. I. (2009). Structure and functional expression of a human interleukin-8 receptor. Science. 1991. 253: 1278–1280. Journal of Immunology, 183(5), 2895–2897
Morohashi, H., Miyawaki, T., Nomura, H., Kuno, K., Murakami, S., Matsushima, K., et al. (1995). Expression of both types of human interleukin-8 receptors on mature neutrophils, monocytes, and natural killer cells. Journal of Leukocyte Biology, 57(1), 180–187. https://doi.org/10.1002/jlb.57.1.180
Kim, S. J., Uehara, H., Karashima, T., McCarty, M., Shih, N., & Fidler, I. J. (2001). Expression of interleukin-8 correlates with angiogenesis, tumorigenicity, and metastasis of human prostate cancer cells implanted orthotopically in nude mice. Neoplasia, 3(1), 33–42. https://doi.org/10.1038/sj.neo.7900124
Miyake, M., Lawton, A., Goodison, S., Urquidi, V., & Rosser, C. J. (2014). Chemokine (C-X-C motif) ligand 1 (CXCL1) protein expression is increased in high-grade prostate cancer. Pathology, Research and Practice, 210(2), 74–78. https://doi.org/10.1016/j.prp.2013.08.013
Straczkowski, M., Dzienis-Straczkowska, S., Stepien, A., Kowalska, I., Szelachowska, M., & Kinalska, I. (2002). Plasma interleukin-8 concentrations are increased in obese subjects and related to fat mass and tumor necrosis factor-alpha system. Journal of Clinical Endocrinology and Metabolism, 87(10), 4602–4606. https://doi.org/10.1210/jc.2002-020135
Wald, O., Shapira, O. M., & Izhar, U. (2013). CXCR4/CXCL12 axis in non small cell lung cancer (NSCLC) pathologic roles and therapeutic potential. Theranostics, 3(1), 26–33. https://doi.org/10.7150/thno.4922
Brat, D. J., Bellail, A. C., & Van Meir, E. G. (2005). The role of interleukin-8 and its receptors in gliomagenesis and tumoral angiogenesis. Neuro-Oncology, 7(2), 122–133. https://doi.org/10.1215/S1152851704001061
Wu, D., LaRosa, G. J., & Simon, M. I. (1993). G protein-coupled signal transduction pathways for interleukin-8. Science, 261(5117), 101–103. https://doi.org/10.1126/science.8316840
Wu, Y., Wang, S., Farooq, S. M., Castelvetere, M. P., Hou, Y., Gao, J. L., et al. (2012). A chemokine receptor CXCR2 macromolecular complex regulates neutrophil functions in inflammatory diseases. Journal of Biological Chemistry, 287(8), 5744–5755. https://doi.org/10.1074/jbc.M111.315762
Fuhler, G. M., Knol, G. J., Drayer, A. L., & Vellenga, E. (2005). Impaired interleukin-8- and GROalpha-induced phosphorylation of extracellular signal-regulated kinase result in decreased migration of neutrophils from patients with myelodysplasia. Journal of Leukocyte Biology, 77(2), 257–266. https://doi.org/10.1189/jlb.0504306
Knall, C., Worthen, G. S., & Johnson, G. L. (1997). Interleukin 8-stimulated phosphatidylinositol-3-kinase activity regulates the migration of human neutrophils independent of extracellular signal-regulated kinase and p38 mitogen-activated protein kinases. Proceedings of the National Academy of Sciences, 94(7), 3052–3057. https://doi.org/10.1073/pnas.94.7.3052
Thelen, M., Uguccioni, M., & Bosiger, J. (1995). PI 3-kinase-dependent and independent chemotaxis of human neutrophil leukocytes. Biochemical and Biophysical Research Communications, 217(3), 1255–1262. https://doi.org/10.1006/bbrc.1995.2903
Waugh, D. J., & Wilson, C. (2008). The interleukin-8 pathway in cancer. Clinical Cancer Research, 14(21), 6735–6741. https://doi.org/10.1158/1078-0432.CCR-07-4843
Chavey, C., Lazennec, G., Lagarrigue, S., Clape, C., Iankova, I., Teyssier, J., et al. (2009). CXC ligand 5 is an adipose-tissue derived factor that links obesity to insulin resistance. Cell Metabolism, 9(4), 339–349. https://doi.org/10.1016/j.cmet.2009.03.002
Begley, L. A., Kasina, S., Mehra, R., Adsule, S., Admon, A. J., Lonigro, R. J., et al. (2008). CXCL5 promotes prostate cancer progression. Neoplasia, 10(3), 244–254. https://doi.org/10.1593/neo.07976
Qi, Y., Zhao, W., Li, M., Shao, M., Wang, J., Sui, H., et al. (2018). High C-X-C motif chemokine 5 expression is associated with malignant phenotypes of prostate cancer cells via autocrine and paracrine pathways. International Journal of Oncology, 53(1), 358–370. https://doi.org/10.3892/ijo.2018.4388
Ammirante, M., Shalapour, S., Kang, Y., Jamieson, C. A., & Karin, M. (2014). Tissue injury and hypoxia promote malignant progression of prostate cancer by inducing CXCL13 expression in tumor myofibroblasts. Proceedings of the National Academy of Sciences, 111(41), 14776–14781. https://doi.org/10.1073/pnas.1416498111
Kusuyama, J., Bandow, K., Ohnishi, T., Amir, M. S., Shima, K., Semba, I., et al. (2019). CXCL13 is a differentiation- and hypoxia-induced adipocytokine that exacerbates the inflammatory phenotype of adipocytes through PHLPP1 induction. The Biochemical Journal, 476(22), 3533–3548. https://doi.org/10.1042/BCJ20190709
Kabir, S. M., Lee, E. S., & Son, D. S. (2014). Chemokine network during adipogenesis in 3T3-L1 cells: Differential response between growth and proinflammatory factor in preadipocytes vs. adipocytes. Adipocyte, 3(2), 97–106. https://doi.org/10.4161/adip.28110
El Haibi, C. P., Sharma, P. K., Singh, R., Johnson, P. R., Suttles, J., Singh, S., et al. (2010). PI3Kp110-, Src-, FAK-dependent and DOCK2-independent migration and invasion of CXCL13-stimulated prostate cancer cells. Molecular Cancer, 9, 85. https://doi.org/10.1186/1476-4598-9-85
El-Haibi, C. P., Singh, R., Sharma, P. K., Singh, S., & Lillard, J. W., Jr. (2011). CXCL13 mediates prostate cancer cell proliferation through JNK signalling and invasion through ERK activation. Cell Proliferation, 44(4), 311–319. https://doi.org/10.1111/j.1365-2184.2011.00757.x
El-Haibi, C. P., Sharma, P., Singh, R., Gupta, P., Taub, D. D., Singh, S., et al. (2013). Differential G protein subunit expression by prostate cancer cells and their interaction with CXCR5. Molecular Cancer, 12, 64. https://doi.org/10.1186/1476-4598-12-64
Hattermann, K., Bartsch, K., Gebhardt, H. H., Mehdorn, H. M., Synowitz, M., Schmitt, A. D., et al. (2016). “Inverse signaling” of the transmembrane chemokine CXCL16 contributes to proliferative and anti-apoptotic effects in cultured human meningioma cells. Cell Communication and Signaling: CCS, 14(1), 26. https://doi.org/10.1186/s12964-016-0149-7
Kurki, E., Shi, J., Martonen, E., Finckenberg, P., & Mervaala, E. (2012). Distinct effects of calorie restriction on adipose tissue cytokine and angiogenesis profiles in obese and lean mice. Nutrition & Metabolism (London), 9(1), 64. https://doi.org/10.1186/1743-7075-9-64
Jung, Y., Kim, J. K., Shiozawa, Y., Wang, J., Mishra, A., Joseph, J., et al. (2013). Recruitment of mesenchymal stem cells into prostate tumours promotes metastasis. Nature Communications, 4, 1795. https://doi.org/10.1038/ncomms2766
Kapur, N., Mir, H., Sonpavde, G. P., Jain, S., Bae, S., Lillard, J. W., Jr., et al. (2019). Prostate cancer cells hyper-activate CXCR6 signaling by cleaving CXCL16 to overcome effect of docetaxel. Cancer Letters, 454, 1–13. https://doi.org/10.1016/j.canlet.2019.04.001
Kanda, H., Tateya, S., Tamori, Y., Kotani, K., Hiasa, K., Kitazawa, R., et al. (2006). MCP-1 contributes to macrophage infiltration into adipose tissue, insulin resistance, and hepatic steatosis in obesity. The Journal of Clinical Investigation, 116(6), 1494–1505. https://doi.org/10.1172/JCI26498
Sartipy, P., & Loskutoff, D. J. (2003). Monocyte chemoattractant protein 1 in obesity and insulin resistance. Proceedings of the National Academy of Sciences, 100(12), 7265–7270. https://doi.org/10.1073/pnas.1133870100
Gerhardt, C. C., Romero, I. A., Cancello, R., Camoin, L., & Strosberg, A. D. (2001). Chemokines control fat accumulation and leptin secretion by cultured human adipocytes. Molecular and Cellular Endocrinology, 175(1–2), 81–92. https://doi.org/10.1016/s0303-7207(01)00394-x
Tsaur, I., Noack, A., Makarevic, J., Oppermann, E., Waaga-Gasser, A. M., Gasser, M., et al. (2015). CCL2 chemokine as a potential biomarker for prostate cancer: A pilot study. Cancer Research and Treatment, 47(2), 306–312. https://doi.org/10.4143/crt.2014.015
Gschwandtner, M., Derler, R., & Midwood, K. S. (2019). More than just attractive: How CCL2 influences myeloid cell behavior beyond chemotaxis. Frontiers in Immunology, 10, 2759. https://doi.org/10.3389/fimmu.2019.02759
Christiansen, T., Richelsen, B., & Bruun, J. M. (2005). Monocyte chemoattractant protein-1 is produced in isolated adipocytes, associated with adiposity and reduced after weight loss in morbid obese subjects. International Journal of Obesity, 29(1), 146–150. https://doi.org/10.1038/sj.ijo.0802839
Huang, M., Narita, S., Numakura, K., Tsuruta, H., Saito, M., Inoue, T., et al. (2012). A high-fat diet enhances proliferation of prostate cancer cells and activates MCP-1/CCR2 signaling. Prostate, 72(16), 1779–1788. https://doi.org/10.1002/pros.22531
Loberg, R. D., Ying, C., Craig, M., Yan, L., Snyder, L. A., & Pienta, K. J. (2007). CCL2 as an important mediator of prostate cancer growth in vivo through the regulation of macrophage infiltration. Neoplasia, 9(7), 556–562. https://doi.org/10.1593/neo.07307
Mathews, J. A., Wurmbrand, A. P., Ribeiro, L., Neto, F. L., & Shore, S. A. (2014). Induction of IL-17A precedes development of airway hyperresponsiveness during diet-induced obesity and correlates with complement factor D. Frontiers in Immunology, 5, 440. https://doi.org/10.3389/fimmu.2014.00440
Xu, H., Barnes, G. T., Yang, Q., Tan, G., Yang, D., Chou, C. J., et al. (2003). Chronic inflammation in fat plays a crucial role in the development of obesity-related insulin resistance. The Journal of Clinical Investigation, 112(12), 1821–1830. https://doi.org/10.1172/JCI19451
Fang, L. Y., Izumi, K., Lai, K. P., Liang, L., Li, L., Miyamoto, H., et al. (2013). Infiltrating macrophages promote prostate tumorigenesis via modulating androgen receptor-mediated CCL4-STAT3 signaling. Cancer Research, 73(18), 5633–5646. https://doi.org/10.1158/0008-5472.CAN-12-3228
Huang, R., Wang, S., Wang, N., Zheng, Y., Zhou, J., Yang, B., et al. (2020). CCL5 derived from tumor-associated macrophages promotes prostate cancer stem cells and metastasis via activating beta-catenin/STAT3 signaling. Cell Death & Disease, 11(4), 234. https://doi.org/10.1038/s41419-020-2435-y
Liu, Y., Cai, Y., Liu, L., Wu, Y., & Xiong, X. (2018). Crucial biological functions of CCL7 in cancer. PeerJ, 6, e4928. https://doi.org/10.7717/peerj.4928
Van Damme, J., Proost, P., Lenaerts, J. P., & Opdenakker, G. (1992). Structural and functional identification of two human, tumor-derived monocyte chemotactic proteins (MCP-2 and MCP-3) belonging to the chemokine family. Journal of Experimental Medicine, 176(1), 59–65. https://doi.org/10.1084/jem.176.1.59
Tourniaire, F., Romier-Crouzet, B., Lee, J. H., Marcotorchino, J., Gouranton, E., Salles, J., et al. (2013). Chemokine expression in inflamed adipose tissue is mainly mediated by NF-kappaB. PLoS ONE, 8(6), e66515. https://doi.org/10.1371/journal.pone.0066515
Cuesta-Gomez, N., Graham, G. J., & Campbell, J. D. M. (2021). Chemokines and their receptors: Predictors of the therapeutic potential of mesenchymal stromal cells. Journal of Translational Medicine, 19(1), 156. https://doi.org/10.1186/s12967-021-02822-5
Nagarsheth, N., Wicha, M. S., & Zou, W. (2017). Chemokines in the cancer microenvironment and their relevance in cancer immunotherapy. Nature Reviews Immunology, 17(9), 559–572. https://doi.org/10.1038/nri.2017.49
Ford, J., Hughson, A., Lim, K., Bardina, S. V., Lu, W., Charo, I. F., et al. (2018). CCL7 is a negative regulator of cutaneous inflammation following leishmania major infection. Frontiers in Immunology, 9, 3063. https://doi.org/10.3389/fimmu.2018.03063
Palomino, D. C., & Marti, L. C. (2015). Chemokines and immunity. Einstein (Sao Paulo), 13(3), 469–473. https://doi.org/10.1590/S1679-45082015RB3438
Zhu, F., Liu, P., Li, J., & Zhang, Y. (2014). Eotaxin-1 promotes prostate cancer cell invasion via activation of the CCR3-ERK pathway and upregulation of MMP-3 expression. Oncology Reports, 31(5), 2049–2054. https://doi.org/10.3892/or.2014.3060
Rosen, E. D., & Spiegelman, B. M. (2014). What we talk about when we talk about fat. Cell, 156(1–2), 20–44. https://doi.org/10.1016/j.cell.2013.12.012
Ducharme, N. A., & Bickel, P. E. (2008). Lipid droplets in lipogenesis and lipolysis. Endocrinology, 149(3), 942–949.
Granneman, J. G., & Moore, H. P. (2008). Location, location: protein trafficking and lipolysis in adipocytes. [Research Support, N.I.H., Extramural Research Support, U.S. Gov’t, Non-P.H.S. Review]. Trends in Endocrinology & Metabolism, 19(1), 3–9. https://doi.org/10.1016/j.tem.2007.10.006
Currie, E., Schulze, A., Zechner, R., Walther, T. C., & Farese, R. V., Jr. (2013). Cellular fatty acid metabolism and cancer. Cell Metabolism, 18(2), 153–161. https://doi.org/10.1016/j.cmet.2013.05.017
Nomura, D. K., Lombardi, D. P., Chang, J. W., Niessen, S., Ward, A. M., Long, J. Z., et al. (2011). Monoacylglycerol lipase exerts dual control over endocannabinoid and fatty acid pathways to support prostate cancer. Chemistry & Biology, 18(7), 846–856. https://doi.org/10.1016/j.chembiol.2011.05.009
Wang, Y. Y., Attane, C., Milhas, D., Dirat, B., Dauvillier, S., Guerard, A., et al. (2017). Mammary adipocytes stimulate breast cancer invasion through metabolic remodeling of tumor cells. JCI Insight, 2(4), e87489. https://doi.org/10.1172/jci.insight.87489
Arner, P., & Langin, D. (2014). Lipolysis in lipid turnover, cancer cachexia, and obesity-induced insulin resistance. Trends in Endocrinology and Metabolism, 25(5), 255–262. https://doi.org/10.1016/j.tem.2014.03.002
Rohm, M., Schafer, M., Laurent, V., Ustunel, B. E., Niopek, K., Algire, C., et al. (2016). An AMP-activated protein kinase-stabilizing peptide ameliorates adipose tissue wasting in cancer cachexia in mice. Nature Medicine, 22(10), 1120–1130. https://doi.org/10.1038/nm.4171
Okumura, T., Ohuchida, K., Sada, M., Abe, T., Endo, S., Koikawa, K., et al. (2017). Extra-pancreatic invasion induces lipolytic and fibrotic changes in the adipose microenvironment, with released fatty acids enhancing the invasiveness of pancreatic cancer cells. Oncotarget, 8(11), 18280–18295. https://doi.org/10.18632/oncotarget.15430
Yamaguchi, J., Ohtani, H., Nakamura, K., Shimokawa, I., & Kanematsu, T. (2008). Prognostic impact of marginal adipose tissue invasion in ductal carcinoma of the breast. American Journal of Clinical Pathology, 130(3), 382–388. https://doi.org/10.1309/MX6KKA1UNJ1YG8VN
Nieman, K. M., Kenny, H. A., Penicka, C. V., Ladanyi, A., Buell-Gutbrod, R., Zillhardt, M. R., et al. (2011). Adipocytes promote ovarian cancer metastasis and provide energy for rapid tumor growth. Nature Medicine, 17(11), 1498–1503. https://doi.org/10.1038/nm.2492
Dirat, B., Bochet, L., Dabek, M., Daviaud, D., Dauvillier, S., Majed, B., et al. (2011). Cancer-associated adipocytes exhibit an activated phenotype and contribute to breast cancer invasion. Cancer Research, 71(7), 2455–2465. https://doi.org/10.1158/0008-5472.CAN-10-3323
Balaban, S., Shearer, R. F., Lee, L. S., van Geldermalsen, M., Schreuder, M., Shtein, H. C., et al. (2017). Adipocyte lipolysis links obesity to breast cancer growth: Adipocyte-derived fatty acids drive breast cancer cell proliferation and migration. Cancer & Metabolism, 5, 1. https://doi.org/10.1186/s40170-016-0163-7
Wen, Y. A., Xing, X., Harris, J. W., Zaytseva, Y. Y., Mitov, M. I., Napier, D. L., et al. (2017). Adipocytes activate mitochondrial fatty acid oxidation and autophagy to promote tumor growth in colon cancer. Cell Death & Disease, 8(2), e2593. https://doi.org/10.1038/cddis.2017.21
Daquinag, A. C., Gao, Z., Fussell, C., Immaraj, L., Pasqualini, R., Arap, W., et al. (2021). Fatty acid mobilization from adipose tissue is mediated by CD36 posttranslational modifications and intracellular trafficking. JCI Insight, 6(17). https://doi.org/10.1172/jci.insight.147057
Su, F., Daquinag, A. C., Ahn, S., Saha, A., Dai, Y., Zhao, Z., et al. (2021). Progression of prostate carcinoma is promoted by adipose stromal cell-secreted CXCL12 signaling in prostate epithelium. NPJ Precision Oncology, 5(1), 26. https://doi.org/10.1038/s41698-021-00160-9
Leitner, B. P., Huang, S., Brychta, R. J., Duckworth, C. J., Baskin, A. S., McGehee, S., et al. (2017). Mapping of human brown adipose tissue in lean and obese young men. Proceedings of the National Academy of Sciences, 114(32), 8649–8654. https://doi.org/10.1073/pnas.1705287114
Alvarez-Artime, A., Garcia-Soler, B., Sainz, R. M., & Mayo, J. C. (2021). Emerging roles for browning of white adipose tissue in prostate cancer malignant behaviour. International Journal of Molecular Sciences, 22(11). https://doi.org/10.3390/ijms22115560
Collins, S. (2011). beta-Adrenoceptor signaling networks in adipocytes for recruiting stored fat and energy expenditure. Frontiers in Endocrinology (Lausanne), 2, 102. https://doi.org/10.3389/fendo.2011.00102
Puigserver, P., Wu, Z., Park, C. W., Graves, R., Wright, M., & Spiegelman, B. M. (1998). A cold-inducible coactivator of nuclear receptors linked to adaptive thermogenesis. Cell, 92(6), 829–839. https://doi.org/10.1016/s0092-8674(00)81410-5
Hursting, S. D., Dunlap, S. M., Ford, N. A., Hursting, M. J., & Lashinger, L. M. (2013). Calorie restriction and cancer prevention: A mechanistic perspective. Cancer & Metabolism, 1(1), 10. https://doi.org/10.1186/2049-3002-1-10
Galet, C., Gray, A., Said, J. W., Castor, B., Wan, J., Beltran, P. J., et al. (2013). Effects of calorie restriction and IGF-1 receptor blockade on the progression of 22Rv1 prostate cancer xenografts. International Journal of Molecular Sciences, 14(7), 13782–13795. https://doi.org/10.3390/ijms140713782
Freedland, S. J., Mavropoulos, J., Wang, A., Darshan, M., Demark-Wahnefried, W., Aronson, W. J., et al. (2008). Carbohydrate restriction, prostate cancer growth, and the insulin-like growth factor axis. Prostate, 68(1), 11–19. https://doi.org/10.1002/pros.20683
Allott, E. H., & Hursting, S. D. (2015). Obesity and cancer: Mechanistic insights from transdisciplinary studies. Endocrine-Related Cancer, 22(6), R365-386. https://doi.org/10.1530/ERC-15-0400
Dorling, J. L., van Vliet, S., Huffman, K. M., Kraus, W. E., Bhapkar, M., Pieper, C. F., et al. (2020). Effects of caloric restriction on human physiological, psychological, and behavioral outcomes: Highlights from CALERIE phase 2. Nutrition Reviews. https://doi.org/10.1093/nutrit/nuaa085
Harvie, M., Wright, C., Pegington, M., McMullan, D., Mitchell, E., Martin, B., et al. (2013). The effect of intermittent energy and carbohydrate restriction v. daily energy restriction on weight loss and metabolic disease risk markers in overweight women. British Journal of Nutrition, 110(8), 1534–1547. https://doi.org/10.1017/S0007114513000792
Harvie, M. N., Pegington, M., Mattson, M. P., Frystyk, J., Dillon, B., Evans, G., et al. (2011). The effects of intermittent or continuous energy restriction on weight loss and metabolic disease risk markers: A randomized trial in young overweight women. International Journal of Obesity, 35(5), 714–727. https://doi.org/10.1038/ijo.2010.171
Harvie, M. N., Sims, A. H., Pegington, M., Spence, K., Mitchell, A., Vaughan, A. A., et al. (2016). Intermittent energy restriction induces changes in breast gene expression and systemic metabolism. Breast Cancer Research, 18(1), 57. https://doi.org/10.1186/s13058-016-0714-4
Hamilton-Reeves, J. M. (2017-2021). Weight management aimed to reduce risk and improve outcomes from radical prostatectomy (WARRIOR). (ed.). NCT03261271
Dimachkie, M. D., Bechtel, M. D., Robertson, H. L., Michel, C., Lee, E. K., Sullivan, D. K., et al. (2021). Exploration of biomarkers from a pilot weight management study for men undergoing radical prostatectomy. Urologic Oncology. https://doi.org/10.1016/j.urolonc.2021.01.010
Hamilton-Reeves, J. M., Johnson, C. N., Hand, L. K., Bechtel, M. D., Robertson, H. L., Michel, C., et al. (2020). Feasibility of a weight management program tailored for overweight men with localized prostate cancer - A pilot study. Nutrition and Cancer, 1–16.https://doi.org/10.1080/01635581.2020.1856890
Lin, D. W., Neuhouser, M. L., Schenk, J. M., Coleman, I. M., Hawley, S., Gifford, D., et al. (2007). Low-fat, low-glycemic load diet and gene expression in human prostate epithelium: A feasibility study of using cDNA microarrays to assess the response to dietary intervention in target tissues. Cancer Epidemiology, Biomarkers & Prevention, 16(10), 2150–2154.
Henning, S. M., Galet, C., Gollapudi, K., Byrd, J. B., Liang, P., Li, Z., et al. (2018). Phase II prospective randomized trial of weight loss prior to radical prostatectomy. Prostate Cancer and Prostatic Diseases, 21(2), 212–220. https://doi.org/10.1038/s41391-017-0001-1
Demark-Wahnefried, W., Rais-Bahrami, S., Desmond, R. A., Gordetsky, J. B., Hunter, G. R., Yang, E. S., et al. (2017). Presurgical weight loss affects tumour traits and circulating biomarkers in men with prostate cancer. [Clinical Study]. British Journal of Cancer, 117, 1303. https://doi.org/10.1038/bjc.2017.303. https://www.nature.com/articles/bjc2017303#supplementary-information
Fruge, A. D., Ptacek, T., Tsuruta, Y., Morrow, C. D., Azrad, M., Desmond, R. A., et al. (2018). Dietary changes impact the gut microbe composition in overweight and obese men with prostate cancer undergoing radical prostatectomy. Journal of the Academy of Nutrition and Dietetics, 118(4), 714–723 e711. https://doi.org/10.1016/j.jand.2016.10.017
Wilson, R. L., Shannon, T., Calton, E., Galvao, D. A., Taaffe, D. R., Hart, N. H., et al. (2020). Efficacy of a weight loss program prior to robot assisted radical prostatectomy in overweight and obese men with prostate cancer. Surgical Oncology, 35, 182–188. https://doi.org/10.1016/j.suronc.2020.08.006
Varady, K. A. (2011). Intermittent versus daily calorie restriction: Which diet regimen is more effective for weight loss? Obesity Reviews, 12(7), e593-601. https://doi.org/10.1111/j.1467-789X.2011.00873.x
Ozkul, C., Yalinay, M., & Karakan, T. (2019). Islamic fasting leads to an increased abundance of Akkermansia muciniphila and Bacteroides fragilis group: A preliminary study on intermittent fasting. The Turkish Journal of Gastroenterology, 30(12), 1030–1035. https://doi.org/10.5152/tjg.2019.19185
Guo, Y., Luo, S., Ye, Y., Yin, S., Fan, J., & Xia, M. (2020). Intermittent fasting improves cardiometabolic risk factors and alters gut microbiota in metabolic syndrome patients. Journal of Clinical Endocrinology and Metabolism. https://doi.org/10.1210/clinem/dgaa644
Johnson, J. B., Summer, W., Cutler, R. G., Martin, B., Hyun, D. H., Dixit, V. D., et al. (2007). Alternate day calorie restriction improves clinical findings and reduces markers of oxidative stress and inflammation in overweight adults with moderate asthma. Free Radical Biology & Medicine, 42(5), 665–674. https://doi.org/10.1016/j.freeradbiomed.2006.12.005
Schwingshackl, L., Zahringer, J., Nitschke, K., Torbahn, G., Lohner, S., Kuhn, T., et al. (2020). Impact of intermittent energy restriction on anthropometric outcomes and intermediate disease markers in patients with overweight and obesity: Systematic review and meta-analyses. Critical Reviews in Food Science and Nutrition, 1–12.https://doi.org/10.1080/10408398.2020.1757616
Cioffi, I., Evangelista, A., Ponzo, V., Ciccone, G., Soldati, L., Santarpia, L., et al. (2018). Intermittent versus continuous energy restriction on weight loss and cardiometabolic outcomes: A systematic review and meta-analysis of randomized controlled trials. Journal of Translational Medicine, 16(1), 371. https://doi.org/10.1186/s12967-018-1748-4
Harris, L., Hamilton, S., Azevedo, L. B., Olajide, J., De Brun, C., Waller, G., et al. (2018). Intermittent fasting interventions for treatment of overweight and obesity in adults: A systematic review and meta-analysis. JBI Database of Systematic Reviews and Implementation Reports, 16(2), 507–547. https://doi.org/10.11124/JBISRIR-2016-003248
Farsijani, S., Payette, H., Morais, J. A., Shatenstein, B., Gaudreau, P., & Chevalier, S. (2017). Even mealtime distribution of protein intake is associated with greater muscle strength, but not with 3-y physical function decline, in free-living older adults: The Quebec longitudinal study on Nutrition as a Determinant of Successful Aging (NuAge study). American Journal of Clinical Nutrition, 106(1), 113–124. https://doi.org/10.3945/ajcn.116.146555
Halberg, N., Henriksen, M., Soderhamn, N., Stallknecht, B., Ploug, T., Schjerling, P., et al. (2005). Effect of intermittent fasting and refeeding on insulin action in healthy men. Journal of Applied Physiology (1985), 99(6), 2128–2136. https://doi.org/10.1152/japplphysiol.00683.2005
Soeters, M. R., Sauerwein, H. P., Groener, J. E., Aerts, J. M., Ackermans, M. T., Glatz, J. F., et al. (2007). Gender-related differences in the metabolic response to fasting. Journal of Clinical Endocrinology and Metabolism, 92(9), 3646–3652. https://doi.org/10.1210/jc.2007-0552
Miller, K. N., Burhans, M. S., Clark, J. P., Howell, P. R., Polewski, M. A., DeMuth, T. M., et al. (2017). Aging and caloric restriction impact adipose tissue, adiponectin, and circulating lipids. Aging Cell, 16(3), 497–507. https://doi.org/10.1111/acel.12575
Linford, N. J., Beyer, R. P., Gollahon, K., Krajcik, R. A., Malloy, V. L., Demas, V., et al. (2007). Transcriptional response to aging and caloric restriction in heart and adipose tissue. Aging Cell, 6(5), 673–688. https://doi.org/10.1111/j.1474-9726.2007.00319.x
Nisoli, E., Tonello, C., Cardile, A., Cozzi, V., Bracale, R., Tedesco, L., et al. (2005). Calorie restriction promotes mitochondrial biogenesis by inducing the expression of eNOS. Science, 310(5746), 314–317. https://doi.org/10.1126/science.1117728
Okita, N., Hayashida, Y., Kojima, Y., Fukushima, M., Yuguchi, K., Mikami, K., et al. (2012). Differential responses of white adipose tissue and brown adipose tissue to caloric restriction in rats. Mechanisms of Ageing and Development, 133(5), 255–266. https://doi.org/10.1016/j.mad.2012.02.003
Giovannini, S., Carter, C. S., Leeuwenburgh, C., Flex, A., Biscetti, F., Morgan, D., et al. (2020). Effects of aging and life-long moderate calorie restriction on IL-15 signaling in the rat white adipose tissue. European Review for Medical and Pharmacological Sciences, 24(5), 2738–2749. https://doi.org/10.26355/eurrev_202003_20547
Kobayashi, M., Fujii, N., Narita, T., & Higami, Y. (2018). SREBP-1c-dependent metabolic remodeling of white adipose tissue by caloric restriction. International Journal of Molecular Sciences, 19(11). https://doi.org/10.3390/ijms19113335
Fujii, N., Uta, S., Kobayashi, M., Sato, T., Okita, N., & Higami, Y. (2019). Impact of aging and caloric restriction on fibroblast growth factor 21 signaling in rat white adipose tissue. Experimental Gerontology, 118, 55–64. https://doi.org/10.1016/j.exger.2019.01.001
Richman, E. L., Kenfield, S. A., Stampfer, M. J., Paciorek, A., Carroll, P. R., & Chan, J. M. (2011). Physical activity after diagnosis and risk of prostate cancer progression: Data from the cancer of the prostate strategic urologic research endeavor. Cancer Research, 71(11), 3889–3895. https://doi.org/10.1158/0008-5472.CAN-10-3932
Kenfield, S. A., Stampfer, M. J., Giovannucci, E., & Chan, J. M. (2011). Physical activity and survival after prostate cancer diagnosis in the health professionals follow-up study. Journal of Clinical Oncology, 29(6), 726–732. https://doi.org/10.1200/JCO.2010.31.5226
Bonn, S. E., Sjolander, A., Lagerros, Y. T., Wiklund, F., Stattin, P., Holmberg, E., et al. (2015). Physical activity and survival among men diagnosed with prostate cancer. Cancer Epidemiology, Biomarkers & Prevention, 24(1), 57–64. https://doi.org/10.1158/1055-9965.EPI-14-0707
Gunnell, A. S., Joyce, S., Tomlin, S., Taaffe, D. R., Cormie, P., Newton, R. U., et al. (2017). Physical activity and survival among long-term cancer survivor and non-cancer cohorts. Frontiers in Public Health, 5, 19. https://doi.org/10.3389/fpubh.2017.00019
McTiernan, A. (2008). Mechanisms linking physical activity with cancer. Nature Reviews Cancer, 8(3), 205–211. https://doi.org/10.1038/nrc2325
Winters-Stone, K. M., Wood, L. J., Stoyles, S., & Dieckmann, N. F. (2018). The effects of resistance exercise on biomarkers of breast cancer prognosis: A pooled analysis of three randomized trials. Cancer Epidemiology, Biomarkers & Prevention, 27(2), 146–153. https://doi.org/10.1158/1055-9965.Epi-17-0766
Dai, J. Y., Wang, B., Wang, X., Cheng, A., Kolb, S., Stanford, J. L., et al. (2019). Vigorous physical activity is associated with lower risk of metastatic-lethal progression in prostate cancer and hypomethylation in the CRACR2A gene. Cancer Epidemiology, Biomarkers & Prevention, 28(2), 258–264. https://doi.org/10.1158/1055-9965.EPI-18-0622
Kim, J. S., Galvao, D. A., Newton, R. U., Gray, E., & Taaffe, D. R. (2021). Exercise-induced myokines and their effect on prostate cancer. Nature Reviews. Urology, 18(9), 519–542. https://doi.org/10.1038/s41585-021-00476-y
Schmid, M., Martins, H. C., Schratt, G., Kropfl, J. M., & Spengler, C. M. (2021). MiRNA126 - RGS16 - CXCL12 cascade as a potential mechanism of acute exercise-induced precursor cell mobilization. Frontiers in Physiology, 12, 780666. https://doi.org/10.3389/fphys.2021.780666
Zaldivar, F., Eliakim, A., Radom-Aizik, S., Leu, S. Y., & Cooper, D. M. (2007). The effect of brief exercise on circulating CD34+ stem cells in early and late pubertal boys. Pediatric Research, 61(4), 491–495. https://doi.org/10.1203/pdr.0b013e3180332d36
Chang, E., Paterno, J., Duscher, D., Maan, Z. N., Chen, J. S., Januszyk, M., et al. (2015). Exercise induces stromal cell-derived factor-1alpha-mediated release of endothelial progenitor cells with increased vasculogenic function. Plastic and Reconstructive Surgery, 135(2), 340e–350e. https://doi.org/10.1097/PRS.0000000000000917
Niemiro, G. M., Parel, J., Beals, J., van Vliet, S., Paluska, S. A., Moore, D. R., et al. (2017). Kinetics of circulating progenitor cell mobilization during submaximal exercise. Journal of Applied Physiology (1985), 122(3), 675–682. https://doi.org/10.1152/japplphysiol.00936.2016
Niemiro, G. M., Allen, J. M., Mailing, L. J., Khan, N. A., Holscher, H. D., Woods, J. A., et al. (2018). Effects of endurance exercise training on inflammatory circulating progenitor cell content in lean and obese adults. Journal of Physiology, 596(14), 2811–2822. https://doi.org/10.1113/JP276023
Van Craenenbroeck, E. M., Beckers, P. J., Possemiers, N. M., Wuyts, K., Frederix, G., Hoymans, V. Y., et al. (2010). Exercise acutely reverses dysfunction of circulating angiogenic cells in chronic heart failure. European Heart Journal, 31(15), 1924–1934. https://doi.org/10.1093/eurheartj/ehq058
Van Craenenbroeck, E. M., Hoymans, V. Y., Beckers, P. J., Possemiers, N. M., Wuyts, K., Paelinck, B. P., et al. (2010). Exercise training improves function of circulating angiogenic cells in patients with chronic heart failure. Basic Research in Cardiology, 105(5), 665–676. https://doi.org/10.1007/s00395-010-0105-4
Ribeiro, F., Ribeiro, I. P., Goncalves, A. C., Alves, A. J., Melo, E., Fernandes, R., et al. (2017). Effects of resistance exercise on endothelial progenitor cell mobilization in women. Science and Reports, 7(1), 17880. https://doi.org/10.1038/s41598-017-18156-6
Ross, M. D., Malone, E. M., Simpson, R., Cranston, I., Ingram, L., Wright, G. P., et al. (2018). Lower resting and exercise-induced circulating angiogenic progenitors and angiogenic T cells in older men. American Journal of Physiology. Heart and Circulatory Physiology, 314(3), H392–H402. https://doi.org/10.1152/ajpheart.00592.2017
Wedell-Neergaard, A. S., Lang Lehrskov, L., Christensen, R. H., Legaard, G. E., Dorph, E., Larsen, M. K., et al. (2019). Exercise-induced changes in visceral adipose tissue mass are regulated by IL-6 signaling: A randomized controlled trial. Cell Metabolism, 29(4), 844–855 e843. https://doi.org/10.1016/j.cmet.2018.12.007
Khosravi, N., Stoner, L., Farajivafa, V., & Hanson, E. D. (2019). Exercise training, circulating cytokine levels and immune function in cancer survivors: A meta-analysis. Brain, Behavior, and Immunity, 81, 92–104. https://doi.org/10.1016/j.bbi.2019.08.187
Rocha-Rodrigues, S., Matos, A., Afonso, J., Mendes-Ferreira, M., Abade, E., Teixeira, E., et al. (2021). Skeletal muscle-adipose tissue-tumor axis: Molecular mechanisms linking exercise training in prostate cancer. International Journal of Molecular Sciences, 22(9). https://doi.org/10.3390/ijms22094469
Idorn, M., & Thor Straten, P. (2018). Chemokine receptors and exercise to tackle the inadequacy of T cell homing to the tumor site. Cells, 7(8). https://doi.org/10.3390/cells7080108
Bourke, L., Smith, D., Steed, L., Hooper, R., Carter, A., Catto, J., et al. (2016). Exercise for men with prostate cancer: A systematic review and meta-analysis. European Urology, 69(4), 693–703. https://doi.org/10.1016/j.eururo.2015.10.047
Andersen, M. F., Midtgaard, J., & Bjerre, E. D. (2022). Do patients with prostate cancer benefit from exercise interventions? A systematic review and meta-analysis. International Journal of Environmental Research and Public Health, 19(2). https://doi.org/10.3390/ijerph19020972
Newton, R. U., Kenfield, S. A., Hart, N. H., Chan, J. M., Courneya, K. S., Catto, J., et al. (2018). Intense Exercise for Survival among Men with Metastatic Castrate-Resistant Prostate Cancer (INTERVAL-GAP4): A multicentre, randomised, controlled phase III study protocol. British Medical Journal Open, 8(5), e022899. https://doi.org/10.1136/bmjopen-2018-022899
Kim, J. S., Taaffe, D. R., Galvao, D. A., Hart, N. H., Gray, E., Ryan, C. J., et al. (2022). Exercise in advanced prostate cancer elevates myokine levels and suppresses in-vitro cell growth. Prostate Cancer and Prostatic Diseases, 25(1), 86–92. https://doi.org/10.1038/s41391-022-00504-x
Ingram, D. K., Zhu, M., Mamczarz, J., Zou, S., Lane, M. A., Roth, G. S., et al. (2006). Calorie restriction mimetics: An emerging research field. Aging Cell, 5(2), 97–108. https://doi.org/10.1111/j.1474-9726.2006.00202.x
Salehi, B., Mishra, A. P., Nigam, M., Sener, B., Kilic, M., Sharifi-Rad, M., et al. (2018). Resveratrol: A double-edged sword in health benefits. Biomedicines, 6(3). https://doi.org/10.3390/biomedicines6030091
Lagouge, M., Argmann, C., Gerhart-Hines, Z., Meziane, H., Lerin, C., Daussin, F., et al. (2006). Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1alpha. Cell, 127(6), 1109–1122. https://doi.org/10.1016/j.cell.2006.11.013
Alberdi, G., Rodriguez, V. M., Miranda, J., Macarulla, M. T., Arias, N., Andres-Lacueva, C., et al. (2011). Changes in white adipose tissue metabolism induced by resveratrol in rats. Nutrition & Metabolism (London), 8(1), 29. https://doi.org/10.1186/1743-7075-8-29
Cho, S. J., Jung, U. J., & Choi, M. S. (2012). Differential effects of low-dose resveratrol on adiposity and hepatic steatosis in diet-induced obese mice. British Journal of Nutrition, 108(12), 2166–2175. https://doi.org/10.1017/S0007114512000347
Qiao, Y., Sun, J., Xia, S., Tang, X., Shi, Y., & Le, G. (2014). Effects of resveratrol on gut microbiota and fat storage in a mouse model with high-fat-induced obesity. Food & Function, 5(6), 1241–1249. https://doi.org/10.1039/c3fo60630a
Mendez-del Villar, M., Gonzalez-Ortiz, M., Martinez-Abundis, E., Perez-Rubio, K. G., & Lizarraga-Valdez, R. (2014). Effect of resveratrol administration on metabolic syndrome, insulin sensitivity, and insulin secretion. Metabolic Syndrome and Related Disorders, 12(10), 497–501. https://doi.org/10.1089/met.2014.0082
Arzola-Paniagua, M. A., Garcia-Salgado Lopez, E. R., Calvo-Vargas, C. G., & Guevara-Cruz, M. (2016). Efficacy of an orlistat-resveratrol combination for weight loss in subjects with obesity: A randomized controlled trial. Obesity (Silver Spring), 24(7), 1454–1463. https://doi.org/10.1002/oby.21523
Faghihzadeh, F., Adibi, P., Rafiei, R., & Hekmatdoost, A. (2014). Resveratrol supplementation improves inflammatory biomarkers in patients with nonalcoholic fatty liver disease. Nutrition Research, 34(10), 837–843. https://doi.org/10.1016/j.nutres.2014.09.005
Most, J., Timmers, S., Warnke, I., Jocken, J. W., van Boekschoten, M., de Groot, P., et al. (2016). Combined epigallocatechin-3-gallate and resveratrol supplementation for 12 wk increases mitochondrial capacity and fat oxidation, but not insulin sensitivity, in obese humans: A randomized controlled trial. American Journal of Clinical Nutrition, 104(1), 215–227. https://doi.org/10.3945/ajcn.115.122937
Sheth, S., Jajoo, S., Kaur, T., Mukherjea, D., Sheehan, K., Rybak, L. P., et al. (2012). Resveratrol reduces prostate cancer growth and metastasis by inhibiting the Akt/MicroRNA-21 pathway. PLoS ONE, 7(12), e51655. https://doi.org/10.1371/journal.pone.0051655
Gill, C., Walsh, S. E., Morrissey, C., Fitzpatrick, J. M., & Watson, R. W. (2007). Resveratrol sensitizes androgen independent prostate cancer cells to death-receptor mediated apoptosis through multiple mechanisms. Prostate, 67(15), 1641–1653. https://doi.org/10.1002/pros.20653
Tan, L., Wang, W., He, G., Kuick, R. D., Gossner, G., Kueck, A. S., et al. (2016). Resveratrol inhibits ovarian tumor growth in an in vivo mouse model. Cancer, 122(5), 722–729. https://doi.org/10.1002/cncr.29793
Li, W., Ma, J., Ma, Q., Li, B., Han, L., Liu, J., et al. (2013). Resveratrol inhibits the epithelial-mesenchymal transition of pancreatic cancer cells via suppression of the PI-3K/Akt/NF-kappaB pathway. Current Medicinal Chemistry, 20(33), 4185–4194. https://doi.org/10.2174/09298673113209990251
Liang, Z. J., Wan, Y., Zhu, D. D., Wang, M. X., Jiang, H. M., Huang, D. L., et al. (2021). Resveratrol mediates the apoptosis of triple negative breast cancer cells by reducing POLD1 expression. Frontiers in Oncology, 11, 569295. https://doi.org/10.3389/fonc.2021.569295
Harper, C. E., Patel, B. B., Wang, J., Arabshahi, A., Eltoum, I. A., & Lamartiniere, C. A. (2007). Resveratrol suppresses prostate cancer progression in transgenic mice. Carcinogenesis, 28(9), 1946–1953. https://doi.org/10.1093/carcin/bgm144
Wang, K., Chen, Z., Shi, J., Feng, Y., Yu, M., Sun, Y., et al. (2020). Resveratrol inhibits the tumor migration and invasion by upregulating TET1 and reducing TIMP2/3 methylation in prostate carcinoma cells. Prostate, 80(12), 977–985. https://doi.org/10.1002/pros.24029
Weisberg, S. P., Leibel, R., & Tortoriello, D. V. (2008). Dietary curcumin significantly improves obesity-associated inflammation and diabetes in mouse models of diabesity. Endocrinology, 149(7), 3549–3558. https://doi.org/10.1210/en.2008-0262
Shao, W., Yu, Z., Chiang, Y., Yang, Y., Chai, T., Foltz, W., et al. (2012). Curcumin prevents high fat diet induced insulin resistance and obesity via attenuating lipogenesis in liver and inflammatory pathway in adipocytes. PLoS ONE, 7(1), e28784. https://doi.org/10.1371/journal.pone.0028784
Ejaz, A., Wu, D., Kwan, P., & Meydani, M. (2009). Curcumin inhibits adipogenesis in 3T3-L1 adipocytes and angiogenesis and obesity in C57/BL mice. Journal of Nutrition, 139(5), 919–925. https://doi.org/10.3945/jn.108.100966
Aggarwal, B. B., Kumar, A., & Bharti, A. C. (2003). Anticancer potential of curcumin: Preclinical and clinical studies. Anticancer Research, 23(1A), 363–398.
Vafadar, A., Shabaninejad, Z., Movahedpour, A., Fallahi, F., Taghavipour, M., Ghasemi, Y., et al. (2020). Quercetin and cancer: New insights into its therapeutic effects on ovarian cancer cells. Cell & Bioscience, 10, 32. https://doi.org/10.1186/s13578-020-00397-0
Rivera, L., Moron, R., Sanchez, M., Zarzuelo, A., & Galisteo, M. (2008). Quercetin ameliorates metabolic syndrome and improves the inflammatory status in obese Zucker rats. Obesity (Silver Spring), 16(9), 2081–2087. https://doi.org/10.1038/oby.2008.315
Stewart, L. K., Soileau, J. L., Ribnicky, D., Wang, Z. Q., Raskin, I., Poulev, A., et al. (2008). Quercetin transiently increases energy expenditure but persistently decreases circulating markers of inflammation in C57BL/6J mice fed a high-fat diet. Metabolism, 57(7 Suppl 1), S39-46. https://doi.org/10.1016/j.metabol.2008.03.003
Dong, J., Zhang, X., Zhang, L., Bian, H. X., Xu, N., Bao, B., et al. (2014). Quercetin reduces obesity-associated ATM infiltration and inflammation in mice: A mechanism including AMPKalpha1/SIRT1. Journal of Lipid Research, 55(3), 363–374. https://doi.org/10.1194/jlr.M038786
He, Y., Cao, X., Guo, P., Li, X., Shang, H., Liu, J., et al. (2017). Quercetin induces autophagy via FOXO1-dependent pathways and autophagy suppression enhances quercetin-induced apoptosis in PASMCs in hypoxia. Free Radical Biology & Medicine, 103, 165–176. https://doi.org/10.1016/j.freeradbiomed.2016.12.016
Chondrogianni, N., Kapeta, S., Chinou, I., Vassilatou, K., Papassideri, I., & Gonos, E. S. (2010). Anti-ageing and rejuvenating effects of quercetin. Experimental Gerontology, 45(10), 763–771. https://doi.org/10.1016/j.exger.2010.07.001
Hoek-van den Hil, E. F., van Schothorst, E. M., van der Stelt, I., Swarts, H. J., Venema, D., Sailer, M., et al. (2014). Quercetin decreases high-fat diet induced body weight gain and accumulation of hepatic and circulating lipids in mice. Genes & Nutrition, 9(5), 418. https://doi.org/10.1007/s12263-014-0418-2
Mahini, H., Ainsworth, G., & Garelnabi, M. (2014). Exercise and quercetin intake reduces body weight in c57bl6 mice. Atherosclerosis, 235(2), e109–e110.
Pei, Y., Parks, J. S., & Kang, H. W. (2021). Quercetin alleviates high-fat diet-induced inflammation in brown adipose tissue. Journal of Functional Foods, 85.https://doi.org/10.1016/j.jff.2021.104614
Lee, J. S., Cha, Y. J., Lee, K. H., & Yim, J. E. (2016). Onion peel extract reduces the percentage of body fat in overweight and obese subjects: A 12-week, randomized, double-blind, placebo-controlled study. Nursing Research & Practice, 10(2), 175–181. https://doi.org/10.4162/nrp.2016.10.2.175
Pfeuffer, M., Auinger, A., Bley, U., Kraus-Stojanowic, I., Laue, C., Winkler, P., et al. (2013). Effect of quercetin on traits of the metabolic syndrome, endothelial function and inflammation in men with different APOE isoforms. Nutrition, Metabolism, and Cardiovascular Diseases, 23(5), 403–409. https://doi.org/10.1016/j.numecd.2011.08.010
Martin-Montalvo, A., Mercken, E. M., Mitchell, S. J., Palacios, H. H., Mote, P. L., Scheibye-Knudsen, M., et al. (2013). Metformin improves healthspan and lifespan in mice. Nature Communications, 4, 2192. https://doi.org/10.1038/ncomms3192
Anisimov, V. N., Berstein, L. M., Egormin, P. A., Piskunova, T. S., Popovich, I. G., Zabezhinski, M. A., et al. (2008). Metformin slows down aging and extends life span of female SHR mice. Cell Cycle, 7(17), 2769–2773. https://doi.org/10.4161/cc.7.17.6625
Saha, A., Blando, J., Tremmel, L., & DiGiovanni, J. (2015). Effect of metformin, rapamycin and their combination on growth and progression of prostate tumors in himyc mice. Cancer Prevention Research (Philadelphia, Pa.). https://doi.org/10.1158/1940-6207.CAPR-15-0014
Goodwin, P. J., & Stambolic, V. (2011). Obesity and insulin resistance in breast cancer–chemoprevention strategies with a focus on metformin. Breast, 20(Suppl 3), S31-35. https://doi.org/10.1016/S0960-9776(11)70291-0
Berstein, L. M. (2012). Metformin in obesity, cancer and aging: Addressing controversies. Aging (Albany NY), 4(5), 320–329. https://doi.org/10.18632/aging.100455
Harrison, D. E., Strong, R., Sharp, Z. D., Nelson, J. F., Astle, C. M., Flurkey, K., et al. (2009). Rapamycin fed late in life extends lifespan in genetically heterogeneous mice. Nature, 460(7253), 392–395. https://doi.org/10.1038/nature08221
Chang, G. R., Chiu, Y. S., Wu, Y. Y., Chen, W. Y., Liao, J. W., Chao, T. H., et al. (2009). Rapamycin protects against high fat diet-induced obesity in C57BL/6J mice. Journal of Pharmacological Sciences, 109(4), 496–503. https://doi.org/10.1254/jphs.08215FP
Checkley, L. A., Rho, O., Moore, T., Hursting, S., & DiGiovanni, J. (2011). Rapamycin is a potent inhibitor of skin tumor promotion by 12-O-tetradecanoylphorbol-13-acetate. Cancer Prevention Research (Philadelphia, Pa.), 4(7), 1011–1020. https://doi.org/10.1158/1940-6207.CAPR-10-0375
Lee, D., Lee, J. H., Kim, B. H., Lee, S., Kim, D. W., & Kang, K. S. (2022). Phytochemical combination (p-synephrine, p-octopamine hydrochloride, and hispidulin) for improving obesity in obese mice induced by high-fat diet. Nutrients, 14(10). https://doi.org/10.3390/nu14102164
Lodi, A., Saha, A., Lu, X., Wang, B., Sentandreu, E., Collins, M., et al. (2017). Combinatorial treatment with natural compounds in prostate cancer inhibits prostate tumor growth and leads to key modulations of cancer cell metabolism. NPJ Precision Oncology, 1.https://doi.org/10.1038/s41698-017-0024-z
Daquinag, A. C., Dadbin, A., Snyder, B., Wang, X., Sahin, A., Ueno, N. T., et al. (2017). Non-glycanated decorin is a drug target on human adipose stromal cells. Molecular Therapy - Oncolytics, 6, 1–9.
Daquinag, A. C., Tseng, C., Zhang, Y., Amaya-Manzanares, F., Florez, F., Dadbin, A., et al. (2016). Targeted pro-apoptotic peptides depleting adipose stromal cells inhibit tumor growth. Molecular Therapy, 1, 34–40. https://doi.org/10.1038/mt.2015.155
Daquinag, A. C., Salameh, A., Zhang, Y., Tong, Q., & Kolonin, M. G. (2015). Depletion of white adipocyte progenitors induces beige adipocyte differentiation and suppresses obesity development. Cell Death & Diff., 22, 351–363. https://doi.org/10.1038/cdd.2014.148
Zaidi, N., Lupien, L., Kuemmerle, N. B., Kinlaw, W. B., Swinnen, J. V., & Smans, K. (2013). Lipogenesis and lipolysis: The pathways exploited by the cancer cells to acquire fatty acids. Progress in Lipid Research, 52(4), 585–589. https://doi.org/10.1016/j.plipres.2013.08.005
Ros, S., Santos, C. R., Moco, S., Baenke, F., Kelly, G., Howell, M., et al. (2012). Functional metabolic screen identifies 6-phosphofructo-2-kinase/fructose-2,6-biphosphatase 4 as an important regulator of prostate cancer cell survival. Cancer Discovery, 2(4), 328–343. https://doi.org/10.1158/2159-8290.CD-11-0234
Kuemmerle, N. B., Rysman, E., Lombardo, P. S., Flanagan, A. J., Lipe, B. C., Wells, W. A., et al. (2011). Lipoprotein lipase links dietary fat to solid tumor cell proliferation. Molecular Cancer Therapeutics, 10(3), 427–436. https://doi.org/10.1158/1535-7163.MCT-10-0802
Domanska, U. M., Kruizinga, R. C., Nagengast, W. B., Timmer-Bosscha, H., Huls, G., de Vries, E. G., et al. (2013). A review on CXCR4/CXCL12 axis in oncology: No place to hide. European Journal of Cancer, 49(1), 219–230. https://doi.org/10.1016/j.ejca.2012.05.005
Zhao, H., Guo, L., Zhao, H., Zhao, J., Weng, H., & Zhao, B. (2015). CXCR4 over-expression and survival in cancer: A system review and meta-analysis. Oncotarget, 6(7), 5022–5040. https://doi.org/10.18632/oncotarget.3217
Darash-Yahana, M., Pikarsky, E., Abramovitch, R., Zeira, E., Pal, B., Karplus, R., et al. (2004). Role of high expression levels of CXCR4 in tumor growth, vascularization, and metastasis. The FASEB Journal, 18(11), 1240–1242. https://doi.org/10.1096/fj.03-0935fje
Cui, K., Zhao, W., Wang, C., Wang, A., Zhang, B., Zhou, W., et al. (2011). The CXCR4-CXCL12 pathway facilitates the progression of pancreatic cancer via induction of angiogenesis and lymphangiogenesis. [Research Support, Non-U.S. Gov’t]. Journal of Surgical Research, 171(1), 143–150. https://doi.org/10.1016/j.jss.2010.03.001
Teicher, B. A., & Fricker, S. P. (2010). CXCL12 (SDF-1)/CXCR4 pathway in cancer. Clinical Cancer Research, 16(11), 2927–2931. https://doi.org/10.1158/1078-0432.CCR-09-2329
Scala, S. (2015). Molecular pathways: Targeting the CXCR4-CXCL12 axis–Untapped potential in the tumor microenvironment. Clinical Cancer Research, 21(19), 4278–4285. https://doi.org/10.1158/1078-0432.CCR-14-0914
Bleul, C. C., Fuhlbrigge, R. C., Casasnovas, J. M., Aiuti, A., & Springer, T. A. (1996). A highly efficacious lymphocyte chemoattractant, stromal cell-derived factor 1 (SDF-1). Journal of Experimental Medicine, 184(3), 1101–1109. https://doi.org/10.1084/jem.184.3.1101
Hernandez, L., Magalhaes, M. A., Coniglio, S. J., Condeelis, J. S., & Segall, J. E. (2011). Opposing roles of CXCR4 and CXCR7 in breast cancer metastasis. Breast Cancer Research, 13(6), R128. https://doi.org/10.1186/bcr3074
Ahn, S., Saha, A., Kolonin, M. G., J. DiGiovanni, J. (2021). Signaling via both CXCR4 and CXCR7 in prostate cancer cells promotes tumor progression and underlies obesity-associated epithelial-mesenchymal transition. In revision
Funding
This work was supported in part by NIH grants R01 CA196259 and R01 CA228404 (to JD). Jill Hamilton-Reeves was supported by a Research Scholar Grant, RSG-17–050-01—NEC, from the American Cancer Society.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Saha, A., Hamilton-Reeves, J. & DiGiovanni, J. White adipose tissue-derived factors and prostate cancer progression: mechanisms and targets for interventions. Cancer Metastasis Rev 41, 649–671 (2022). https://doi.org/10.1007/s10555-022-10056-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10555-022-10056-0