Abstract
Isolated left ventricular non-compaction (LVNC) is a rare cardiomyopathy that leads to severe clinical complications. This study is to investigate whether or not prolonged QTc is a good indicator for evaluating the severity of fibrosis and predicting the prognosis of LVNC, and if native T1 can be used to quantify the fibrosis. 32 LVNC patients and 14 healthy controls with matched age and sex were examined by CMR and ECG to acquire native T1, QTc interval, and ECG abnormalities. 18 LVNC patients had normal QTc and 14 LVNC patients had prolonged QTc. The mean native T1 value of the normal controls, normal QTc and prolonged QTc patients was 1096.0 ± 41.5, 1141.98 ± 45.46, and 1182.67 ± 42.02 ms, respectively. One-way ANVOA showed significant differences in native T1 among three groups (F = 14.9, p < 0.001). In LVNC patients, the QTc interval significantly correlated with LVEF (p = 0.003, r = 0.51) and native T1 values (p = 0.015, R = −0.47). This suggests that prolonged QTc is associated with more severe compacted myocardial fibrosis, more cardiac dysfunction, and a poorer prognosis in LVNC patients. Follow-up data showed significant differences in adverse events between patients with normal QTc and patients with prolonged QTc (p = 0.036). Prolonged QTc interval leads to more severe compacted myocardial fibrosis, poorer cardiac dysfunction, and poorer prognosis in LVNC patients.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Isolated left ventricular non-compaction (LVNC) is a rare cardiomyopathy caused by the arrest of normal embryogenesis of myocardium during the embryonic period [1, 2]. The pathology of LVNC is characterized by prominent trabeculations and deep intertrabecular recesses within the left ventricular wall [3, 4]. Clinical complications of LVNC patients include heart failure, arrhythmias, and sudden cardiac death [4, 5]. Currently there is no clinical marker for evaluating the severity of fibrosis of compacted myocardium and predicting prognosis of LVNC patients.
Recently, the native T1 mapping of cardiac magnetic resonance imaging (CMR) has been used, without any contrast agent, for quantitative measurement of myocardial tissue fibrosis and evaluation of the prognosis [6, 7]. Bull et al. [6] reported that native T1 values were correlated with histological percentage of fibrosis by biopsy. Claridge et al. [7] reported that in non-ischemic patients, native T1 was the sole predictor of the primary endpoint. Corrected QT interval between the start of the Q wave and the end of the T wave (QTc) in the heart’s electrical cycle is significantly associated with heart failure, arrhythmias, and sudden cardiac death [8,9,10,11]. Studies have shown that patients with LVNC have prolonged QTc [12,13,14]; for example, Towbin et al. reported that some patients with LVNC had QTc interval prolongation [12], and Steffel et al. [15] found that 52% of the patients with LVNC had QTc interval prolongation. However, the correlation between QTc and native T1 value in LVNC patients has not been demonstrated. In this study, we hypothesized that prolonged QTc is a good indicator for evaluating the severity of fibrosis and predicting the prognosis of LVNC, and that native T1 can be used to quantify the fibrosis.
Methods
Study population
We recruited 32 patients with LVNC between December 2008 and June 2014 in the Department of Cardiology of Peking Union Medical College Hospital. LVNC was diagnosed according to the Jenni echocardiography criteria [3] and Petersen CMR criteria [16] by two senior cardiologists. Patients were excluded from the study if they were pregnant, had contraindications to CMR, or had a history of other cardiomyopathies. The CMR scanning was performed for all recruited LVNC patients. One patient was excluded from the study as he was unable to hold his breath during the CMR scan. As a control, we enrolled 14 healthy volunteers with matching age and gender, and without history of cardiac diseases or known cardiac risk factors (diabetes mellitus, hypertension, and smoking). The study was approved by the local Institutional Review Board and written informed consent was obtained from every subject.
QTc interval measurement
The 12-lead resting electrocardiograms (ECG) were recorded at a paper speed of 25 mm/s on a Marquette Resting ECG recorder. The intervals between the start of the Q wave and the end of the T wave in the heart’s electrical cycle were automatically analyzed (GE Medical Systems, Information Technologies, Freiburg, Germany). The determined heart rate was corrected using the Bazett formula [17]. The patients were divided into a prolonged QTc group (QTc interval >440 ms) and a normal QTc group (QTc interval ≤440 ms) [18, 19].
CMR imaging protocol
All CMR imaging was performed on a 3.0 T MR system (Achieva TX, Philips Healthcare, Best, Netherlands) using a 32-channel phased array (InVivo, Gainesville, Florida, USA). ECG-gated balanced steady-state free precession (bSSFP) cine images were acquired from the long and short axis (SAX) images covering the entire left ventricular wall (LV). Typical scan parameters included TR/TE at 2.7/1.3 ms with 30 heart phases, imaging voxel size at 1.8 × 1.5 × 8.0 mm3, 8 mm slice thickness, and 2 mm in-between gap. The native T1 data were acquired using modified look-locker inversion recovery (MOLLI) imaging protocol [20] at 8 inversion times over an 11-heart-beat breath-hold at end-expiration with 2 inversions using a 5(3)3 scheme [21]. Native T1 values were measured on three SAX slices with 20 mm in-between gap, covering 50 mm of the mid-ventricle cavity. Typical imaging parameters for MOLLI were: bSSFP single shot read out with flip angle of 35°, typical field of view of 300 × 150 mm2, slice thickness of 10 mm, voxel size at 1.5 × 1.5 × 10.0 mm3, TR/TE at 2.5/1.1 ms, 0.85 partial echo factor, and parallel imaging factor of 2.5.
CMR image analysis
The images for the compacted wall of the left ventricular (LV) were manually segmented on both SAX SSFP cine images and T1 maps. Papillary muscles were carefully excluded from the LV to avoid ambiguities. The ventricular volumes, left ventricular ejection fraction (LVEF), and LV compacted myocardium mass were quantified using SAX cine stack. The native T1 was averaged over three slices per case before the statistical analysis. All images were processed using MATLAB (MathWorks, Natick, Massachusetts, USA). The maximum ratio of non-compacted and compacted myocardium thicknesses (NC:C ratio) from long axis views (LAX) in the end of diastole was measured. Corresponding wall thickness of compacted myocardium was recorded. The myocardium was divided into 17 segments using the standard AHA model [22].
Follow-up data acquisition
Clinical assessment included a detailed medical history with standardized questionnaires. Medical records were reviewed. Adverse events were defined as sudden death, deteriorating heart failure requiring hospital admission, cardiogenic shock, syncope, and severe arrhythmia (sick sinus syndrome).
Statistical analysis
Data for continuous variables were presented as mean ± SD. Statistical analysis was performed using SPSS Version 20.0 (SPSS, Chicago, IL, USA). Comparison between groups was performed using one-way ANOVA with a post-hoc Student–Newman–Keuls method. The Chi square test or Fisher’s exact test was performed to compare discrete data when appropriate. The correlation between continuous variables was assessed with the Spearman test. Statistical significance was defined as p < 0.05.
Result
Baseline clinical data and imaging data
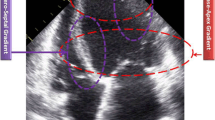
The LVNC patients were divided into two groups based on QTc interval: normal QTc group (QTc ≤ 440 ms, n = 18), and prolonged QTc group (QTc > 440 ms, n = 14). The typical LVNC imaging was shown in the Fig. 1. The healthy volunteers showed normal electrocardiogram. Table 1 shows the baseline characteristics of patients and healthy controls. There were no significant differences in age, or body mass index among patients with normal QTc, patients with prolonged QTc, and the healthy controls. Family history of LVNC was found in seven patients in the normal QTc group and two patients in the prolonged QTc group.
Comparison of electrocardiogram data
All patients in the prolonged QTc group showed abnormalities in the resting electrocardiogram; three patients had left bundle branch block, five had ventricular premature beat, six had higher left ventricular voltage, seven had intraventricular conduction delay, and nine had T wave changes. In contrast, ECG abnormalities in the normal QTc were only seen in one patient with left bundle branch block, one with ventricular premature beat, seven with higher left ventricular voltage, four with T wave changes, and four with intraventricular conduction delay (Table 2).
Comparison of CMR data
The CMR imaging data are presented in Table 2. The mean T1 values in the healthy controls and LVNC patients (patients with prolonged QTc and patients with normal QTc) were 1096.0 ± 41.5 and 1159.1 ± 47.4 ms, respectively. The mean T1 values in patients with normal QTc and patients with prolonged QTc were 1141.98 ± 45.46 and 1182.67 ± 42.02 ms, respectively (Fig. 2). One-way ANOVA showed significant differences in native T1 among three groups (F = 14.9, p < 0.001) while Post Hoc Student–Newman–Keuls analysis showed significant differences in native T1 values between any two groups. No significant differences were observed in the thickness of compacted myocardium and the NC: C ratio between normal QTc and prolonged QTc group.
The mean Left Venticular ejection fraction (LVEF) of the healthy controls,normal QTc patients, and prolonged QTc patients were 60.3 ± 6.2, 49.35 ± 10.39 and 31.84 ± 15.87%. One-way ANOVA showed significant differences in LVEF among three groups (F = 23.36, p < 0.001). In addition, significant differences were found between any two groups by Post Hoc Student–Newman–Keuls analysis.
After adjusting for body surface area, one-way ANOVA showed significant differences in LV mass, LV end-systolic volume (LVESV), and LV end-diastolic volume (LVEDV) among the three groups (F = 10.8, p < 0.001; F = 17.5, p < 0.001; F = 13.2, p < 0.001; respectively). The post-hoc with Student–Newman–Keuls analysis showed significant differences in LV mass, LVESV, and LVEDV among three groups, but no significant differences were observed between all patients and healthy controls (Table 3). There was no difference in non-compaction segments between normal QTc and prolonged QTc group (p = 0.79). The distribution of non-compaction segments of the two groups are shown in the Fig. 3.
Distribution of non-compaction (NC) segments. a Normal QTc LVNC patient. b Prolonged QTc LVNC patient. The heart was divided into 17 segments according to AHA standard segments. The number in each segment shows the total NC segments. For example, at the apical, a and b pictures have seven and ten subjects with NC segments, respectively
The relationship between QTc, native T 1 values, and LVEF
In LVNC patients, the native T1 relaxation times positively correlated with QTc interval (r = 0.51, p = 0.003) (Fig. 4a). The QTc interval was significantly increased and negatively correlated with LVEF (r = −0.47, p = 0.015) (Fig. 4b) in LVNC patients. In addition, the Multiple Regression Line Analysis have shown that that QTc interval is significantly correlated with LVEF (p = 0.006) and native T1 values (p = 0.025).
Follow-up
The mean follow-up duration was 30.4 ± 3.4 months. Ten adverse events were found in five patients (Fig. 5). A significant difference (p = 0.028) was observed in QTc interval between the patients with (466.3 ± 40.4 ms) and without adverse events (427.3 ± 31.3 ms). Follow-up data showed significant differences in adverse events between patients with normal QTc and patients with prolonged QTc (p = 0.036).
The ROC analysis showed that the QTc interval yielded an area under the ROC curve of 0.87 ± 0.07. At a cutoff value of 443.5 ms, QTc interval has an 80% diagnostic sensitivity and 77% diagnostic specificity for identifying LVNC patients with adverse events (Fig. 6).
Discussion
This study investigated the relationships among the QTc interval and the native T1 values of CMR, cardiac function, and the prognosis of LVNC patients. We found that the mean native T1 value was highest in patients with prolonged QTc, followed by patients with normal QT, and then normal controls. Furthermore, the QTc interval positively correlated with the native T1 values. These suggest that patients with prolonged QTc interval suffer more serious fibrosis. The QTc interval was significantly negatively correlated with LVEF, suggesting that the QTc interval may reflect the cardiac systolic function. In addition, follow-up results showed that the patients with prolonged QTc had a poor prognosis.
Previous studies have observed the QTc interval prolongation in LVNC patients [12, 15, 17]. Our previous study revealed that native T1 is an independent indicator of myocardial late gadolinium enhancement [23] and can detect early myocardial fibrosis. One major finding in this study is that prolonged QTc interval positively correlates with increased native T1 values in LVNC patients. In a histological study, the QTc interval prolongation correlates with the progression of myocardial fibrosis in Alström syndrome patients [24]. Inoue et al. reported that clinically unrecognized myocardial fibrosis was significantly associated with a longer QT interval [25]. It may suggest that the QTc prolongation is associated with cardiac conduction system abnormality caused by myocardial fibrosis. Therefore, the combined evaluation of the CMR and QTc interval might result in a more accurate understanding of the severity of myocardial fibrosis in LVNC patients.
In this study, we found that the QTc interval negatively correlates with LVEF, which is consistent with findings in a previous study in heart failure models [26]. Previous studies have also demonstrated that low LVEF is related to fibrosis [27, 28], which leads to the cardiac conduction system abnormality. Thus, the QTc interval prolongation may correlate with cardiac systolic dysfunction. We also observed that more adverse events occurred in patients with prolonged QTc interval than patients with normal QTc interval. This may indicate that patients with prolonged QTc are more susceptible to adverse events than the patients with normal QTc. Our previous study demonstrated that the adverse events are associated with the decrease in cardiac function [23]. The follow-up results in this study are consistent with the study by Pickham et al., which demonstrated that prolongation of QTc interval correlates with the severity of cardiac disease and poor prognosis [29]. Other studies have also found that the QTc interval is significantly correlated with ventricular arrhythmias and sudden death, which are the main clinical characteristics of LVNC [15, 30].
This study has several limitations. First, this study was conducted in a single center. Second, this study used a relatively small sample size due to LVNC being a rare disease. Thirdly, the diagnosis was based on CMR cine pictures, and not all patients with coronary artery disease can be ruled out by invasive coronary angiography. Fourthly, the clinical follow-up time was reduced from what could be optimal. Finally, we only analyzed the T1 value of the compacted wall dense layer of myocardium. A report showed fibrosis occurred in the non-compact myocardium in the inner wall based on the autopsy of a patient’s heart [31]. Prior study already showed that non-compacted and compacted myocardium exhibited different signal intensity on T2-weighted image [32]. Thus, the future study should compare the T2 value between non-compact and compacted myocardium.
In conclusion, prolonged QTc interval may suggest more severe compacted myocardial fibrosis, more cardiac dysfunction, and poorer prognosis in LVNC patients.
References
Sen-Chowdhry S, McKenna WJ (2008) Left ventricular noncompaction and cardiomyopathy: cause, contributor, or epiphenomenon? Curr Opin Cardiol 23:171–175
Luxán G, Casanova JC, Martínez-Poveda B, Prados B, D’Amato G, MacGrogan D, Gonzalez-Rajal A, Dobarro D, Torroja C, Martinez F, Izquierdo-García JL, Fernández-Friera L, Sabater-Molina M, Kong YY, Pizarro G, Ibañez B, Medrano C, García-Pavía P, Gimeno JR, Monserrat L, Jiménez-Borreguero LJ, de la Pompa JL (2013) Mutations in the NOTCH pathway regulator MIB1 cause left ventricular noncompaction cardiomyopathy. Nat Med 19:193–201
Jenni R, Oechslin E, Schneider J, Attenhofer Jost C, Kaufmann PA (2001) Echocardiographic and pathoanatomical characteristics of isolated left ventricular non-compaction: a step towards classification as a distinct cardiomyopathy. Heart 86:666–671
Chin TK, Perloff JK, Williams RG, Jue K, Mohrmann R (1990) Isolated noncompaction of left ventricular myocardium. A study of eight cases. Circulation 82:507–513
Paterick TE, Tajik AJ (2012) Left ventricular noncompaction. Circ J 76:1556–1562
Bull S, White SK, Piechnik SK, Flett AS, Ferreira VM, Loudon M, Francis JM, Karamitsos TD, Prendergast BD, Robson MD, Neubauer S, Moon JC, Myerson SG (2013) Human non-contrast T1 values and correlation with histology in diffuse fibrosis. Heart 99:932–937
Claridge S, Mennuni S, Jackson T, Behar JM, Porter B, Sieniewicz B, Bostock J, O’Neill M, Murgatroyd F, Gill J, Carr-White G, Chiribiri A, Razavi R, Chen Z, Rinaldi CA (2017) Substrate dependent risk stratification for implantable cardioverter defibrillator therapies using cardiac magnetic resonance imaging: The importance of T1 mapping in non-ischemic patients. J Cardiovasc Electrophysiol. doi:10.1111/jce.13226
Beinart R, Zhang Y, Lima JA, Bluemke DA, Soliman EZ, Heckbert SR, Post WS, Guallar E, Nazarian S (2014) The QT interval is associated with incident cardiovascular events: the MESA study. J Am Coll Cardiol 64:2111–2119
McMurray JJ, Adamopoulos S, Anker SD, Auricchio A, Böhm M, Dickstein K, Falk V, Filippatos G, Fonseca C, Gomez-Sanchez MA, Jaarsma T, Køber L, Lip GY, Maggioni AP, Parkhomenko A, Pieske BM, Popescu BA, Rønnevik PK, Rutten FH, Schwitter J, Seferovic P, Stepinska J, Trindade PT, Voors AA, Zannad F, Zeiher A (2012) ESCCommittee for Practice Guidelines. ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: the task force for the diagnosis and treatment of acute and chronic heart failure 2012 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association (HFA) of the ESC. Eur Heart J 33:1787–1847
Shawl FA, Velasco CE, Goldbaum TS, Forman MB (1990) Effect of coronary angioplasty on electrocardiographic changes in patients with unstable angina secondary to left anterior descending coronary artery disease. J Am Coll Cardiol 16:325–331
Castro-Torres Y, Carmona-Puerta R, Katholi RE (2015) Ventricular repolarization markers for predicting malignant arrhythmias in clinical practice. World J Clin Cases 3:705–720
Towbin JA, Lorts A, Jefferies JL (2015) Left ventricular non-compaction cardiomyopathy. The Lancet 386:813–825
Nakashima K, Kusakawa I, Yamamoto T, Hirabayashi S, Hosoya R, Shimizu W, Sumitomo N (2013) A left ventricular noncompaction in a patient with long QT syndrome caused by a KCNQ1 mutation: a case report. Heart Vessel 28:126–129
Onay OS, Yildirim I, Beken B, Erdem S, Karagoz T, Yilmaz M, Yigit S (2013) Successful implantation of an intracardiac defibrillator in an infant with long QT syndrome and isolated noncompaction of the ventricular myocardium. Pediatr Cardiol 34:189–193
Steffel J, Kobza R, Oechslin E, Jenni R, Duru F (2009) Electrocardiographic characteristics at initial diagnosis in patients with isolated left ventricular noncompaction. Am J Cardiol 104:984–989
Petersen SE, Selvanayagam JB, Wiesmann F, Robson MD, Francis JM, Anderson RH, Watkins H, Neubauer S (2005) Left ventricular non-compaction: insights from cardiovascular magnetic resonance imaging. J Am Coll Cardiol 46:101–105
Bazett HC (1920) An analysis of the time-relations of electrocardiograms. Heart 7:353–367
Vrtovec B, Delgado R, Zewail A, Thomas CD, Richartz BM, Radovancevic B (2003) Prolonged QTc interval and high B-type natriuretic peptide levels together predict mortality in patients with advanced heart failure. Circulation 107:1764–1769
Hombach V, Merkle N, Torzewski J, Kraus JM, Kunze M, Zimmermann O, Kestler HA, Wöhrle J (2009) Electrocardiographic and cardiac magnetic resonance imaging parameters as predictors of a worse outcome in patients with idiopathic dilated cardiomyopathy. Eur Heart J 30:2011–2018
Messroghli DR, Radjenovic A, Kozerke S, Higgins DM, Sivananthan MU, Ridgway JP (2004) Modified Look-Locker inversion recovery (MOLLI) for high-resolution T1 mapping of the heart. Magn Reson Med 52:141–146
Kellman P, Hansen MS (2014) T1-mapping in the heart: accuracy and precision. J Cardiovasc Magn Reson 16:2
Cerqueira MD, Weissman NJ, Dilsizian V, Jacobs AK, Kaul S, Laskey WK, Pennell DJ, Rumberger JA, Ryan T, Verani MS, American Heart Association Writing Group on Myocardial Segmentation and Registration for Cardiac Imaging (2002) Standardized myocardial segmentation and nomenclature for tomographic imaging of the heart. A statement for healthcare professionals from the Cardiac Imaging Committee of the Council on Clinical Cardiology of the American Heart Association. Circulation 105:539–542
Zhou H, Lin X, Fang L, Zhao X, Ding H, Chen W, Xu R, Bai X, Wang Y, Fang Q (2016) Characterization of compacted myocardial abnormalities by cardiac magnetic resonance with native T1 mapping in left ventricular non-compaction patients. Circ J 80:1210–1216
Edwards NC, Moody WE, Yuan M, Warfield AT, Cramb R, Paisey RB, Geberhiwot T, Steeds RP (2015) Diffuse left ventricular interstitial fibrosis is associated with sub-clinical myocardial dysfunction in Alstrom Syndrome: an observational study. Orphanet J Rare Dis 10:83
Inoue YY, Ambale-Venkatesh B, Mewton N, Volpe GJ, Ohyama Y, Sharma RK, Wu CO, Liu CY, Bluemke DA, Soliman EZ, Lima JA, Ashikaga H (2017) Electrocardiographic impact of myocardial diffuse fibrosis and scar: MESA (multi-ethnic study of atherosclerosis). Radiology 282:690–698
Long VP, Bonilla IM, Vargas-Pinto P, Nishijima Y, Sridhar A, Li C, Mowrey K, Wright P, Velayutham M, Kumar S, Lee NY, Zweier JL, Mohler PJ, Györke S, Carnes CA (2015) Heart failure duration progressively modulates the arrhythmia substrate through structural and electrical remodeling. Life Sci 123:61–71
Chimura M, Kiuchi K, Okajima K, Shimane A, Sawada T, Onishi T, Yamada S, Taniguchi Y, Yasaka Y, Kawai H (2015) Distribution of ventricular fibrosis associated with life threatening ventricular tachyarrhythmias in patients with nonishcemic dilated cardiomyopathy. J Cardiovasc Electrophysiol. doi:10.1111/jce.12767
Florian A, Ludwig A, Engelen M, Waltenberger J, Rösch S, Sechtem U, Yilmaz A (2014) Left ventricular systolic function and the pattern of late-gadolinium-enhancement independently and additively predict adverse cardiac events in muscular dystrophy patients. J Cardiovasc Magn Reson 16:81
Pickham D, Helfenbein E, Shinn JA, Chan G, Funk M, Weinacker A, Liu JN, Drew BJ (2012) High prevalence of corrected QT interval prolongation in acutely ill patients is associated with mortality: results of the QT in Practice (QTIP) Study. Crit Care Med 40:394–399
Ichida F, Hamamichi Y, Miyawaki T, Ono Y, Kamiya T, Akagi T, Hamada H, Hirose O, Isobe T, Yamada K, Kurotobi S, Mito H, Miyake T, Murakami Y, Nishi T, Shinohara M, Seguchi M, Tashiro S, Tomimatsu H (1999) Clinical features of isolated noncompaction of the ventricular myocardium: long-term clinical course, hemodynamic properties, and genetic background. J Am Coll Cardiol 34:233–240
Finsterer J, Stöllberger C, Feichtinger H (2002) Histological appearance of left ventricular hypertrabeculation/noncompaction. Cardiology 98:162–164
Daimon Y, Watanabe S, Takeda S, Hijikata Y, Komuro I (2002) Two-layered appearance of noncompaction of the ventricular myocardium on magnetic resonance imaging. Circ J 66:619–621
Funding
This study was supported by Key Projects in Chinese National Science & References Technology Pillar Program during the Twelfth Five-year Plan Period (2011BAI11B11) and National Natural Science Foundation of China (81271536, 81670349).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Rights and permissions
About this article
Cite this article
Zhou, H., Lin, X., Fang, L. et al. Prolonged QTc indicates the clinical severity and poor prognosis in patients with isolated left ventricular non-compaction. Int J Cardiovasc Imaging 33, 2013–2020 (2017). https://doi.org/10.1007/s10554-017-1209-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-017-1209-9