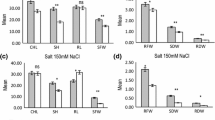
Abstract
A segregating F4 population from the cross between drought sensitive (Yecora Rojo) and drought tolerant (Pavon 76) genotypes was made to identify molecular markers linked to a wheat (Triticum aestivum L.) abscisic acid (ABA) content at two water regimes. The parents and 150 F4 lines were evaluated phenotypically for drought tolerance using two irrigation treatments [0.25 and 0.75 m3(H2O) m−2(soil)]. Forty different target region amplification polymorphism (TRAP) primer combinations, 98 different sequence-related amplified polymorphism (SRAP) primer combinations, and 400 simple sequence repeat (SSR) primers were tested for polymorphism among the parental genotypes and the F4 lines. Seven loci in the F4 lines treated with the drought stress were identified. Single quantitative trait loci (QTLs) were located on chromosomes 1B, 2A, 3A, 5D, and 7B and each of them explained from 15 to 31 % of phenotypic variance with a LOD value of 7.2 to 15.7. Five QTLs were located on chromosome 4A and six QTLs on chromosome 5A. In control (well-watered) F4 lines, two QTLs were mapped on chromosome 3B and one QTL on each chromosome 5B and 5D. Statistically the most significant groups of QTLs for the ABA content were identified in the regions of chromosomes 3B, 4A, and 5A mostly near to Barc164, Wmc96, and Trap9 markers. Therefore, these markers linked to QTLs for the drought-induced ABA content can be further used in breeding for drought tolerance in wheat.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- BSA:
-
bulk segregant analysis
- CIM:
-
composite interval mapping
- LOD:
-
likelihood ratio
- QTL:
-
quantitative trait locus
- RIL:
-
recombinant inbred line
- SSR:
-
simple sequence repeat
- SRAP:
-
sequence-related amplified polymorphism
- TRAP:
-
target region amplification polymorphism
References
Barakat, M.N., Al-Doss, A.A., Moustafa, K.A., Ahmed, E.I., Elshafei, A.A.: Morphological and molecular characterization of Saudi wheat genotypes under drought stress. — J. Food Agr. Environ. 8: 220–228, 2010.
Barakat, M.N., Wahba, L.E., Milad, S.I.: Molecular mapping of QTLs for flag leaf senescence under water stressed conditions in wheat (Triticum aestivum L). — Biol. Plant. 57: 79–84, 2013.
Chandler, P.M., Robertson, M.: Gene expression regulated by abscisic acid and its relation to stress tolerance. — Annu. Rev. Plant Physiol. Plant mol. Biol. 45: 113–141, 1994.
Davies, W.J., Zhang, J.: Root signals and the regulation of growth and development of plants in drying soil. — Annu. Rev. Plant Physiol. Plant mol. Biol. 42: 55–76, 1991.
Dubcovsky, J., Luo, M.C., Dvorák, J.: Linkage relationships among stress-induced genes in wheat. — Theor. appl. Genet. 91: 795–801, 1995.
Elshafei, A.A., Saleh, M.S., Al-Doss, A.A., Moustafa, K.A., Al-Qurainy, F.H., Barakat, M.N.: Identification of new SRAP markers linked to leaf chlorophyll content, flag leaf senescence and cell membrane stability traits in wheat under water-stress condition. — Aust. J. Crop Sci. 7: 887–893, 2013.
Golabadi, M., Arzani, A., Maibody, S.M., Tabatabaei, B.S., Mohammadi, S.A.: Identification of microsatellite markers linked with yield components under drought stress at terminal growth stages in durum wheat. — Euphytica 177: 207–221, 2011.
Gupta, P., Balyan, H., Edwards, K., Isaac, P., Korzun, V., Röder, M. Leroy, P.: Genetic mapping of 66 new microsatellite (SSR) loci in bread wheat. — Theor. appl. Genet. 105: 413–422, 2002.
Hu, J., Vick, B.A.: Target region amplification polymorphism: a novel marker technique for plant genotyping. — Plant mol. Biol. Rep. 21: 289–294, 2003.
Iuchi, S., Kobayashi, M., Taji, T., Naramoto, M., Seki, M., Kato, T., Tabata, S., Kakubari, Y., Qin, X.Q., Zeevaart, J.A.D.: Overexpression of a 9-cis-epoxycarotenoiddioxygenase gene in Nicotiana plumbaginifolia increases abscisic acid and phaseic acid levels and enhances drought tolerance. — Plant Physiol. 128: 544–551, 2002.
Kobayashi, F., Takumi, S., Handa, H.: Identification of quantitative trait loci for ABA responsiveness at seedling stage associated with ABA-regulated gene expression in common wheat. — Theor. appl. Genet. 121: 629–641, 2010.
Krochko, J.E., Abrams, G.D., Loewen, M.K., Abrams, S.R., Cutler, A.: (+)-abscisic acid 80-hydroxylase is a cytochrome P450 monooxygenase. — Plant Physiol. 118: 849–860, 1998.
Lebreton, C., Lazic-Jancic, V., Steed, A., Pekic, S., Quarrie, S.A.: Identification of QTLs for drought responses in maize and their use in testing causal relationships between traits. — J. exp. Bot. 46: 853–865, 1995.
Li, G., Quiros, C.F.: Sequence-related amplified polymorphism (SRAP), a new marker system based on a simple PCR reaction: its application to mapping and gene tagging in Brassica. — Theor. appl. Genet. 103: 455–461, 2001.
Li, S.S., Jia, J.Z., Wei, X.Y., Zhang, X.C., Li, L.Z., Chen, H.M., Fan, Y.D., Sun, H.Y., Zhao, X.H., Lei, T.D., Xu, Y.F., Jiang, F.S., Wang, H.G., Li, L.H.: A intervarietal genetic map and QTL analysis for yield traits in wheat. — Mol. Breed. 20: 167–178, 2007.
McWilliam, J.: The dimensions of drought. — In: Baker, F.W.G. (ed.): Drought Resistance in Cereals. Pp. 1–11. CAB International, Wallingford 1989.
Muthurajan, R., Shobbar, Z.S., Jagadish, S.V.K., Bruskiewich, R., Ismail, A., Leung, H., Bennett, J.: Physiological and proteomic responses of rice peduncles to drought stress. — Mol. Biotechnol. 48: 173–182, 2011.
Nelson, J.C.: QGENE: software for marker-based genomic analysis and breeding. — Mol. Breed. 3: 239–245, 1997.
Quarrie, S.A., Gulli, M., Calestani, C., Steed, A., Marmiroli, N.: Localization of drought-induced abscisic acid production on the long arm of chromosome 5 A of wheat. — Theor. appl. Genet. 89: 794–800, 1994.
Quarrie, S.A., Laurie, D.A., Zhu, J., Lebreton, C., Semikhodskii, A., Steed, A., Witsenboer, H., Calestani, C.: QTL analysis to study the association between leaf size and abscisic acid accumulation in droughted rice leaves and comparisons across cereals. — Plant mol. Biol. 35: 155–165, 1997.
Röder, M.S., Korzun, V., Gill, B.S., Ganal, M.W.: The physical mapping of microsatellite markers in wheat. — Genome 41: 278–283, 1998.
Saeedipour, S., Moradi, F.: Relationship of endogenous ABA and IAA to accumulation of grain protein and starch in two winter wheat cultivars under post-anthesis water deficit. — J. agr. Sci. 4: 147–156, 2012.
Saghai-Maroof, M.A., Soliman, K.M., Jorgensen, R.A., Allard, R.W.: Ribosomal DNA spacer-length polymorphism in barley: Mendelian inheritance, chromosomal location, and population dynamics. — Proc. nat. Acad. Sci. USA 81: 8014–8018, 1984.
Saleh, M.S., Al-Doss, A.A., Elshafei, A.A., Moustafa, K.A., Al-Qurainy, F.H., Barakat, M.N.: Identification of new TRAP markers linked to leaf chlorophyll content, flag leaf senescence and cell membrane stability traits in wheat under water-stress conditions. — Biol. Plant. 58: 64–70, 2014.
Sanguineti, M.C., Tuberosa, R., Landi, P., Salvi, S., Maccaferri, M., Casarini, E., Conti, S.: QTL analysis of drought-related traits and grain yield in relation to genetic variation for leaf abscisic acid concentration in field-grown maize. — J. exp. Bot. 50: 1289–1297, 1999.
Song, Q., Shi, J., Singh, S., Fickus, E., Costa, J., Lewis, J., Cregan, P.: Development and mapping of microsatellite (SSR) markers in wheat. — Theor. appl. Genet. 110: 550–560, 2005.
Steel, R.G., Torrie, J.H.: Principles and Procedures of Statistics, a Biometrical Approach. — McGraw-Hill, Kogakusha 1980.
Thompson, D.S., Wilkinson, S., Bacon, M.A., Davies, W.J.: Multiple signals and mechanisms that regulate leaf growth and stomatal behavior during water deficit. — Physiol. Plant. 100: 303–313, 1997.
Tuberosa, R., Sanguineti, M.C., Landi, P., Salvi, S., Casarini, E., Conti, S.: RFLP mapping of quantitative trait loci controlling abscisic acid concentration in leaves of droughtstressed maize (Zea mays L.). — Theor. appl. Genet. 97: 744–755, 1998.
You, J., Li, Q., Yue, B., Xue, W.-Y., Luo, L.-J., Xiong, L.-Z.: Identification of quantitative trait loci for ABA sensitivity at seed germination and seedling stages in rice. — Acta genet. sin. 33: 532–541, 2006.
Ünyayar, S., Keles, Y., Ünal, E.: Proline and ABA levels in two sunflower genotypes subjected to water stress. — Bulg. J. Plant Physiol. 30: 34–47, 2004.
Zeevaart, J.A.D., Creelman, R.A.: Metabolism and physiology of abscisic acid. — Annu. Rev. Plant Physiol. Plant mol. Biol. 39: 439–473, 1988.
Zhu, J.K.: Salt and drougtht stress signal transduction in plants. — Annu. Rev. Plant Biol. 53: 247–273, 2002.
Żur, I., Krzewska, M., Dubas, E., Gołębiowska-Pikania, G., Janowiak, F., Stojałowski, S.: Molecular mapping of loci associated with abscisic acid accumulation in triticale (×Triticosecale Wittm.) anthers in response to low temperature stress inducing androgenic development. — Plant Growth Regul. 68: 483–492, 2012.
Author information
Authors and Affiliations
Corresponding author
Additional information
Acknowledgements: The authors extend their appreciation to the Deanship of Scientific Research at King Saud University for funding the work through the research group project no. RGPVPP-161.
Rights and permissions
About this article
Cite this article
Barakat, M.N., Saleh, M.S., Al-Doss, A.A. et al. Mapping of QTLs associated with abscisic acid and water stress in wheat. Biol Plant 59, 291–297 (2015). https://doi.org/10.1007/s10535-015-0499-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10535-015-0499-9