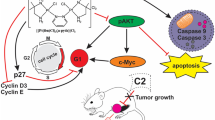
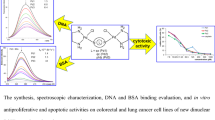
Abstract
Four new platinum(II) complexes: PtII L1·H2O (C1, H2 L1 = C20H16N2O2), PtII L2Cl2 (C2, L2 = C22H16N2O2), PtII L3Cl2·H2O (C3, L3 = C20H16N2), PtII L4Cl2·0.4H2O (C4, L4 = C18H14N4) have been synthesized and characterized by using various physico-chemical techniques. The binding interaction of the four platinum(II) complexes C1–C4 with calf thymus (CT)-DNA has been investigated by UV–Vis and fluorescence emission spectrometry. The apparent binding constant (K app) values follow the order: C3 > C1 > C2 > C4. In addition, fluorescence spectrometry of bovine serum albumin (BSA) with the four platinum(II) complexes C1–C4 showed that the quenching mechanism might be a static quenching procedure. For C1–C4, the number of binding sites was about one for BSA and the binding constants follow the order: C3 (7.08 × 105M−1) > C1 (2.82 × 105M−1) > C2 (0.85 × 105M−1) > C4 (0.15 × 105M−1). With the single condition change such as absence of an external agent, the DNA cleavage abilities of C3 exhibit remarkable changes. In addition, the cytotoxicity of C3 in vitro on tumor cells lines (MCF-7, HepG2 and HT29) were examined by MTT and showed better antitumor effects on the tested cells.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Discovery of platinum (Pt)-based chemotherapy has been the front-line treatment for a variety of malignancies including testicular, lung, and ovarian cancer (Kelland 2007; Brabec et al. 2012; Schneider et al. 2010; Roos and Kaina 2013). It is reported that the activity of platinum (Pt)-based chemotherapy is related to its binding to DNA and protein. For example, the interaction of cisplatin results in the formation of covalent cross-links, and the major adduct is 1,2 intrastrand crosslink formed between the [Pt(NH3)2]2+ unit and the two adjacent guanine bases of DNA (Icsel et al. 2015). It is well known that cis-platin drugs could be utilized to remedy 70–80% of cancer patients. However, the resistance to Pt-based regimens remains a major limitation in the successful treatment for many cancers including epithelial ovarian cancer (EOC) and non-small cell lung cancer (NSCLC) (Ozols et al. 2004; Einhorn 2008). Besides, the severe toxic and side effects, such as nephrotoxicity, ototoxicity and neurotoxicity have limited the platinum (Pt)-based chemotherapy for a wide range of uses (Kelland 2007; Ta 2006; Jamieson and Lippard 1999; O’Dwyer 1999; Hanigan and Devarajan 2003). At last, the poor water-solubility is another disadvantage for the use of platinum (Pt)-based chemotherapy. Consequently, to design and synthesize new platinum (Pt)-based complexes exhibiting strong DNA and protein binding propensities are highly desirable for the development of potential chemotherapeutic agents for cancer.
DNA is one of the main targets for anticancer agents. The anticancer activity of cisplatin arises from its ability to damage DNA, with the major Pt-DNA adducts formed being intrastrand d(GpG) and d(ApG) crosslinks (Todd and Lippard 2010; Jung and Lippard 2007; Wang and Lippard 2005; Zhen et al. 1992; Lepre et al. 1990). Coordinated ligands also play an important role towards the binding of platinum complexes to DNA. Multiple modes of DNA binding can be achieved by changing different ligands. Thus, the new drug design was based on new binding modes between DNA and platinum (Pt)-based chemotherapy (Ruhayel et al. 2012; Pereira-Maia and Garnier-Suillerot 2003; Jamieson and Lippard 1999; Wheate et al. 2003; Francisco et al. 2011).
Binding to plasma proteins is regarded as a reason for the platinum (Pt)-based low general toxicity. Serum albumin is the most abundant plasma protein distributed in the blood, and more than 90% plasma protein can be combined with metal drugs in form of co-valent bonds (Huang et al. 2004; Su et al. 2008; Marzano et al. 2002). Such interactions between platinum and proteins have attracted considerable attention, consequently affecting the therapeutic dose of the drugs transferred to their target positions (Marzano et al. 2002; Matesanz et al. 2011).
So in this paper, we investigated the interaction of four novel mononuclear platinum(II) complexes with CT-DNA and bovine serum albumin (BSA) protein, supported by UV–vis, fluorescence. The complex C3 was tested for DNA nuclease activity against pBR322 plasmid DNA. The in vitro anticancer activity of the complex C3 was assessed by the MMT assay in MCF-7, HT-29 and HepG-2 cancer cells and compared to that of cisplatin.
Experimental
Materials and methods
All chemical solvents were dehydrated and distilled by standard methods before use. Bovine serum albumin, ethidium bromide (EB), pBR322 DNA and calf thymus DNA (CT-DNA) were purchased from Sigma.
Physical measurements
IR spectra were recorded on a Nicolet 380 spectrometer as KBr pellets in the range 4000–400 cm−1 with the OMNIC software. Electrospray ionization mass spectrometry (ESI-MS) was analyzed by using a Triple TOF™ 5600+ system with an ion spray source in the positive-ion mode. Electronic spectra were measured by employing a Hewlett Packard HP-8453 spectrophotometer. Fluorescence spectral data were obtained by means of a RF-5301 fluorescence spectrophotometer at the approved temperature.
DNA binding experiments
UV absorption and fluorescence spectrometry relative binding of the complex to calf thymus (CT-DNA) were studied in 5 mM Tris–HCl-50 mM NaCl buffer (pH 7.20). For CT-DNA free of protein, the ratio of UV absorbance at 260 nm to that at 280 nm should be 1.8–1.9. The CT-DNA concentration per nucleotide was determined by absorption spectrometry with a molar extinction coefficient value of 6600 M−1 cm−1 at 260 nm. With varying amounts of CT-DNA to complex, the absorption spectra of complex binding to DNA was changed. Through the McGhee–von Hippel (MvH) method (Marmur 1961), the change in the absorption intensity of the spectral bands, the intrinsic equilibrium binding constant (K b ) and the binding site size (s) of C3 to CT-DNA was obtained by regression analysis using the equation (Wolf et al. 1987):
where ε a is the absorb intensity observed at a given DNA concentration, ε f is the absorb intensity of free complex in solution, ε b is the absorb intensity of complex when fully bound to DNA (it is assumed that further addition of DNA does not change the absorb intensity), K b is the equilibrium binding constant, C t is the total DNA concentration, (DNA) is the DNA concentration, and s is the binding site size. Both K b and s are obtained from the best fit line. The non-linear least-squares analyses were conducted through the use of Origin Lab, version 7.5. By the fluorescence spectral method, the apparent DNA binding constant (K a ) values of complex were obtained by using an EB-bound CT-DNA solution in 5 mM Tris–HCl–50 mM NaCl buffer (pH 7.20) (Lakowicz et al. 1973). The sample was excited at 510 nm, and the emission range was recorded at 500–700 nm. The fluorescence intensities were recorded with increasing amounts of the added complex concentrations. Such an experiment was carried out by titrating the Pt(II) complex into EB-DNA solution at 580 nm. Due to fluorescence quenching of the free EB by the solvent molecules, EB showed no apparent emission in Tris-buffer. The non-linear least-squares analyses were also done with Origin Lab, version 7.5.
Bovine serum albumin binding
A protein binding study was performed by tryptophan fluorescence quenching experiments with BSA stock solution (BSA, 10−4 M, based on its molecular mass of 66 kDa) in 10 mM Tris–HCl buffer (pH 7.20) to keep the physiological conditions. Fluorescence emission spectra were recorded in the wavelength range 300–500 nm by an excitation wavelength of 285 nm. With the complex concentration increasing, the quenching of the emission intensity of tryptophan residues of BSA at 344 nm was monitored with the Pt(II) complex as a quencher. Fluorescence measurements were carried out by means of a RF-5301 spectrofluorimeter by keeping the concentration of BSA constant (1.0 μM). A linear fit of the data was done by using the Stern–Volmer equation: \({\text{F}}_{0} /{\text{F}} = 1 + {\text{k}}_{\text{q}} \tau_{0} \left[ {\text{Q}} \right] = 1 + {\text{K}}_{\text{sv}} \left[ {\text{Q}} \right]\) (Han et al. 2012).
MTT assay
MTT assay was used to assess the influence of the Pt(II) complexes on the growth of HepG2, MCF7 and HT29 cells. Briefly, cells (HepG2, MCF7 and HT29) at a density 4 × 104 were plated into 96-well microplates and incubated at 37 °C in an atmosphere of 5% CO2 in air overnight and three replica wells were used for controls. Those compounds with the range of concentrations (0, 12.5, 25, 50 and 100 μM) were added and incubated for 48 h. Then, 20 μL of MTT (5 mg mL−1) in phosphate buffered saline (PBS, pH 7.4) was added to each well and incubated for further 4 h. Then medium was discarded and the formazan dissolved with DMSO (100 μL per well). The absorbance was determined at 590 nm with an enzyme-linked immunosorbent assay (ELISA) reader.
DNA cleavage experiments
The DNA cleavage experiments were done by agarose gel electrophoresis (Gao et al. 2015). It was performed by incubation at 37 °C as follows: pBR322 DNA (0.1 μg μL−1) in 50 mM Tris–HCl buffer, pH 7.2, containing 20 mM NaCl treated with C3. After incubation for about 3 h, the loading buffer (0.25% bromophenol-blue, 45% glycerol and 2 mM EDTA) was added. Then the sample was electrophoresed for 2 h at 120 V on 0.9% agarose gel using Tris–boric acid–EDTA buffer and stained with 1 mg·mL−1 EB. In the end, bands were visualized by UV light and photographed.
Preparation of Pt(II) complexes
Synthesis of H2 L1 (C20H16N2O2) and PtII L1·H2O (C20H14N2O2Pt·H2O, C1 )
H2 L1 was synthesized according to the procedure described in previous literature (Ding et al. 2009). Yield: 90.6%. Electrospray ionization mass spectrometry: m/z 316.12 [H2 L1 + H]+, 317.1233. Selected IR data [KBr, max (cm−1)]: 3053 w, 2986 w, 1615 s, 1484 s, 1275 s, 1188 s (s, strong; m, medium; w, weak). Elemental analysis calcd for C20H16N2O2: C%: 75.93; N%: 8.86; H%: 5.10, found: C%: 75.90; N%: 8.87; H%: 5.14.
One millimole of ethylenediamine solution was dropped to solution of 1 mmol H2 L1 in absolute methyl alcohol (30 mL) to make H2 L1 completely dissolved, and then 1 mmol Potassium platinochloride in 3 mL water was dropwise to the resulting solution. Leave the reaction stirring at room temperature for 6 h. The crude product of Pt(II) metal complex C1 was obtained and washed by distilled water, ethanol for three times respectively. Yield: 65.4%. Electrospray ionization mass spectrometry: m/z 509.07, [C1 + H]+, 510.0679. Selected IR data [KBr, max (cm−1)]: 3053 w, 2986 w, 1615 s, 1480 s, 1277 s, 1190 s (s, strong; m, medium; w, weak). Elemental analysis calcd for C20H14N2O2Pt·H2O: C%: 45.54; N%: 5.31; H%: 3.06, found: C%: 45.58; N%: 5.28; H%: 3.19.
Synthesis of L2 (C22H16N2O2) and PtIIL2Cl2(C22H16Cl2N2O2Pt, C2)
L2 was prepared by following the same general procedure as described for H2 L1, except that 1,4-phthalaldehyde (10 mmol) was reacted instead of Salicylaldehyde. Yield: 39.2%. Electrospray ionization mass spectrometry: m/z 340.37 [L 2 + H]+, 341.1225. Selected IR data [KBr, max (cm−1)]: 2876 w, 1691 s, 1611 s, 1300 s (s, strong; m, medium; w, weak). Elemental analysis calcd for C22H16N2O2: C%: 77.63; N%: 8.23; H%: 4.74, found: C%: 77.60; N%: 8.26; H%: 4.76. C2 was prepared by following the same general procedure as described for C1, except that Cis-diamminedichloro-platinum(II) (0.5 mmol) was reacted instead of Potassium platinochloride. Yield: 59.8%. Electrospray ionization mass spectrometry: m/z 352.25 [C2 + 3CH3OH + 2H]2+ 352.0230. Selected IR data [KBr, max (cm−1)]: 2876 w, 1691 s, 1611 s, 1300 s (s, strong; m, medium; w, weak). Elemental analysis calcd for C22H16Cl2N2O2Pt: C%: 43.58; N%: 4.62; H%: 2.66, found: C%: 43.54; N%: 4.67; H%: 2.64.
Synthesis of L3 (C20H16N2) and PtIIL3Cl2·H2O (C20H16Cl2N2Pt·H2O, C3)
L3 was prepared by following the same general procedure as described for H2 L1, except that benzaldehyde was reacted instead of salicylaldehyde. Yield: 30.1%. Electrospray ionization mass spectrometry: m/z 284.35 [L3 + H]+, 285.1335. Selected IR data [KBr, max (cm−1)]: 2980 w, 1638 s, 1296 s (s, strong; m, medium; w, weak). Elemental analysis calcd for C20H16N2: C%: 84.48; N%: 9.85; H%: 5.67, found: C%: 84.51; N%: 9.87; H%: 5.64. C3 was prepared by following the same general procedure as described for C2. Yield: 48.4%. Electrospray ionization mass spectrometry: m/z 549.03 [C3 + 2H]2+, 548.5346. Selected IR data [KBr, max (cm−1)]: 2917 s, 1631 m, 1311 s (s, strong; m, medium; w, weak). Elemental analysis calcd for C20H16Cl2N2Pt·H2O: C%: 42.26; N%: 4.93; H%: 3.19, found: C%: 42.19; N%: 4.91; H%: 3.39.
Synthesis of L4 (C18H14N) and PtIIL4Cl2·0.4H2O (C18H14Cl2N4Pt·H2O, C4)
L4 was prepared by following the same general procedure as described for H2 L1, except that 2-pyridinecarboxaldehyde was reacted instead of Salicylaldehyde. Yield: 35.7%. Electrospray ionization mass spectrometry: m/z 286.33 [L4 + H]+, 287.1219. Selected IR data [KBr, max (cm−1)]: 1648 m, 1308 m (s, strong; m, medium; w, weak). Elemental analysis calcd for C18H14N4: C%: 75.50; N%: 19.57; H%: 4.93, found: C%: 75.49; N%: 19.56; H%: 4.95. C4 was prepared by following the same general procedure as described for C4. Yield: 25.9%. Electrospray ionization mass spectrometry: m/z 157.10 [C4 + 4H2O + 4H]4+, 157.0349. Selected IR data [KBr, mmax (cm−1)]: 1632 m, 1293 s (s, strong; m, medium; w, weak). Elemental analysis calcd for C18H14Cl2N4Pt·0.4H2O: C%: 38.64; N%: 10.01; H%: 2.67, found: C%: 38.68; N%: 9.97; H%: 2.87.
Results and discussion
Synthesis and structural characterization
As shown in Fig. 1, four Pt(II) complexes were obtained and their chemical structures were determined by elemental analyses (EAs), Electrospray ionization mass spectra (ESI–MS) (Figs. S1, S2, S3, S4, S5, S6, S7, S8) and FT-IR spectra (IR) analysis.
DNA-binding studies
DNA binding ability is critical for DNA cleavage of effective metal based chemotherapeutic drugs (Bloodgood et al. 2013; Dhar et al. 2006; Sha et al. 2012). The potential binding ability of the complexes C1 – C4 to CT-DNA was studied by UV spectroscopy. The typical titration curve for complex C3 is shown in Fig. 2a (similar spectra of C1 and C2 and C4 are presented as Figs. S9, S10, S11), and a plot of (εa−εf)/(εb−εf) versus [DNA] for the titration of DNA to C1 – C4 is shown in Fig. 2b. The absorption peaks at 276–280 nm as these complexes are attributed to intraligand π–π* transition. With the increasing of CT-DNA, an increase in molar absorptivity (hyperchromism, 2–7 fold) for C1 – C4 was found. It is suggested that C1 – C4 binding to DNA is the intercalative mode involving a strong stacking interaction between an aromatic chromophore and the base pairs of DNA (Baldini et al. 2004; Silvestri et al. 2007; Navarro et al. 2003).
a Absorption spectra of C3 (10 μM) in the absence (dash line) and presence (solid line) of increasing amounts of CT-DNA (2.5, 5.0, 7.5, 10.0, 12.5, 15.0, 17.5, 20.0, 22.5 μM) in 5 mM Tris–HCl/50 mM NaCl buffer (pH 7.2). The arrow shows the absorbance changes on increasing DNA concentration. Inset Plot of (εa−εf)/(εb−εf) versus [DNA] for the titration of DNA to C3. b Plot of (εa−εf)/(εb−εf) versus [DNA] for the titration of DNA to C1 – C4
In order to determine the interaction between C1 and C4 and CT-DNA, intrinsic binding constants Kb was calculated according to the Eqs. (1) and (2). As shown in Table 1, the Kb values follow the order: C3 (8.2 × 105 M−1) > C1 (6.4 × 105 M−1) > C2 (5.1 × 105 M−1) > C4 (3.6 × 105 M−1), which shows the similar binding propensity with the previously reported Pt complexes (Bielawska et al. 2010; Shao et al. 2014a, b; Ferreira et al. 2016). In short, the binding constants indicate medium binding strength of the four complexes with CT-DNA.
Besides, to further clarify the binding of C1 – C4, fluorescence spectrum was used to analyze and verify the form of interaction between C1 and C4 and DNA. The apparent binding constant (Kapp) of the complexes was determined by fluorescence spectral technique using EB. Ethidium bromide emits intense fluorescent light in the presence of DNA due to its strong intercalation between the adjacent DNA base pairs, which could be quenched by the addition of another molecule (Gohel et al. 1999). The relative binding propensity of four complexes to CT-DNA studied with an EB bound CT-DNA solution in 5 mM Tris–HCl/50 mM NaCl buffer (pH 7.20) is shown in Fig. 3 (similar spectra of C1 and C2 and C4 are presented as Figs. S12, S13, S14, respectively). Fluorescence intensities at 580 nm (510 nm excitation) were measured at different complex concentrations. From Fig. 3, the fluorescence spectra show a significant reduction in fluorescence intensity, indicating that the EB are displaced from their DNA binding sites and are replaced by C3 under investigation (Barton et al. 1986).
According to the classical Stern–Volmer formula: \({\text{F}}_{0} {\text{/ F = 1 + K}}_{\text{sv}} [ {\text{Q]}}\), F0 and F of the formula are fluorescence intensity in absence and presence of DNA respectively; KSV is the Stern–Volmer binding constant; [Q] is the concentration of the complex C1 – C4. As shown in Table 1, the calculated apparent binding constant values (Kapp) follows the order: C3 (7.22 × 105 M−1) > C1 (3.52 × 105 M−1) > C2 (2.19 × 105 M−1) > C4 (1.05 × 105 M−1), which is consistent with the results obtained Kb values by UV spectroscopy. The calculated binding constant is less than 107 M−1 (Wang et al. 2006; Laitinen et al. 2006), indicating that the binding effect between C1 and C4 and DNA is medium.
BSA-binding ability
The interactions between serum albumin and metal complexes have attracted increasing research interest in recent years (Annaraj et al. 2016; Li et al. 2016; Anjomshoa et al. 2015; Lu et al. 2012; Anjomshoa et al. 2016; Mohamadi et al. 2016; Raghav et al. 2016; Easmon et al. 2001; Loganathan et al. 2014; Shao et al. 2014a, b; Galisteo-González and Molina-Bolívar 2014), since serum albumin constitutes nearly 55% of the total protein in blood plasma and it plays a pivotal role in drug transport and drug metabolism (Galisteo-González and Molina-Bolívar 2014). Bovine serum albumin is the most extensively studied serum albumin, due to its about 78% structural homology with human serum albumin (HSA) (Shi et al. 2010; Sevilla et al. 2007). As shown in Fig. 4, the strong fluorescence emission peak at 342 nm corresponds for BSA. The fluorescence intensity of BSA at 342 nm was decreased regularly with the increasing concentration of complexes C3, which confirmed that the interaction between C3 and BSA have occurred. According to the Stern–Volmer formula, \({\text{F}}_{0} /{\text{F}} = 1+ {\text{K}}_{\text{q}} \tau_{0} \left[ {\text{Q}} \right] = 1+ {\text{K}}_{\text{sv}} \left[ {\text{Q}} \right]\), F0 and F of the formula are fluorescence intensity in absence and presence of quencher respectively; Kq is rate constants; Ksv is the quenching constants; τ0 is average life expectancy without quencher (τ0 = 10−8 s) (Grzegorz 2006), [Q] is the concentration of quencher. The calculated values of KSV and kq for the interaction of the complexes with BSA are given in Table 2.
As shown in Table 2, the K q of C3 was 1.61 × 1012 M−1 s−1, greater than the maximum K q numerically, showing that the quenching is not caused by dynamic quenching, but due to the static quenching of the formation of complex (Guo et al. 2009; Jiang et al. 2004). The equation, \({ \log }[({\text{F}}_{0} - {\text{F}})/{\text{F}}] = { \log }K_{a} + {\text{nlog}}\left[ {\text{Q}} \right]\), could be used to calculate the binding constants and binding sites. K a and n are the apparent binding constants and binding sites respectively (as shown in Fig. 5). The binding constants and binding sites of complexes C1–C4 and BSA could be obtained and are listed in Table 2. It is indicated that C3 exhibits higher binding constants for BSA than other three Pt(II) complexes and the K a and n values follow the order: C3 (7.08 × 105M−1, 1.21) > C1 (2.82 × 105M−1, 1.14) > C2 (0.85 × 105M−1, 0.97) > C4 (0.15 × 105M−1, 0.86). The values of n are associated with binding constants K a , which verify the conclusion that there is a direct relation between the binding constant and number of binding sites (Sathyadevi et al. 2011).
MTT assay
MTT assay is a colorimetric assay based on the conversion of the yellow tetrazolium salt to purple formazan crystals by metabolically active cells. The cytotoxic effects of C3 on the viability of several human cancer cell lines (MCF-7, HepG2 and HT29) were examined by MTT assay. A variety of tumor cells were treated with C3 and incubated for 48 h at increasing concentration, respectively. As shown in Fig. 6, C3 shows better antitumor effects on the tested cancer cells.
DNA cleavage studies
To explore the DNA cleavage abilities of C3, the extent of DNA cleavage was monitored by agarose gel electrophoresis. The supercoiled pBR322 plasmid DNA as a substrate was incubated with C3 in a medium of 50 mM Tris–HCl buffer containing 20 mM NaCl (pH 7.2) for 3 h. The concentration-dependent DNA cleavage activities by C3 were observed without any external agents. As shown in Fig. 7, the results carried out in the concentration range 20–80 μM for C3 and could cause DNA cleavage at 20 μM, which suggests that C3 partially degraded SC DNA into undetectable minor fragments.
Conclusions
A series of mononuclear platinum(II) complexes have been synthesized and characterized. The partial intercalation between the complexes and CT-DNA has been confirmed by using absorption and emission spectral methods and medium binding strength follows the order: C3 > C1 > C2 > C4. With the single condition change such as absence of an external agent, the DNA cleavage abilities of C3 exhibit remarkable changes. Further, the ability to bind to BSA also has been explored, and the quenching mechanisms of BSA by the complexes are static procedures. The vitro cytotoxicity of C3 on cancer cell lines (MCF-7, HepG2 and HT29) has been assessed by MTT and the results indicate that C3 shows better antitumor effects on the tested cells.
Abbreviations
- DNA:
-
Deoxyribonucleic acid
- BSA:
-
Bovine serum albumin
- EB:
-
Ethidium bromide
- MTT:
-
3-(4,5-Dimethyl-2-thiazolyl)-2,5-diphenyl-2-H-tetrazolium bromide
- H 2 L1 :
-
C20H16N2O2
- L2 :
-
C22H16N2O2
- L3 :
-
C20H16N2
- L4 :
-
C18H14N4
- C1 :
-
C20H14N2O2Pt·H2O
- C2 :
-
C22H16Cl2N2O2Pt
- C3 :
-
C20H16Cl2N2Pt·H2O
- C4 :
-
C18H14Cl2N4Pt·0.4H2O
References
Anjomshoa M, Hadadzadeh H, Torkzadeh-Mahani M, Fatemi SJ, Adeli-Sardou M, Rudbari HA, Nardo VM (2015) A mononuclear Cu(II) complex with 5,6-diphenyl-3-(2-pyridyl)-1,2,4-triazine: synthesis, crystal structure, DNA- and BSA-binding, molecular modeling, and anticancer activity against MCF-7, A-549, and HT-29 cell lines. Eur J Med Chem 96:66–82
Anjomshoa M, Torkzadeh-Mahani M, Shakeri M, Adeli-Sardou M (2016) The Zn(II) nano-complex: sonochemical synthesis, characterization, DNA- and BSA-binding, cell imaging, and cytotoxicity against the human carcinoma cell lines. J Fluoresc 26:1007–1020
Annaraj B, Balakrishnan C, Neelakantan MA (2016) Synthesis, structure information, DNA/BSA binding affinity and in vitro cytotoxic studies of mixed ligand copper(II) complexes containing a phenylalanine derivative and diimine co-ligands. J Photochem Photobiol B 160:278–291
Baldini P, Belicchi-Ferrari M, Bisceglie F, Dall’Aglio PP, Pelosi G, Pinelli S, Tarasconi P (2004) Copper(II) complexes with substituted thiosemicarbazones of alpha-ketoglutaric acid: synthesis, X-ray structures, DNA binding studies, and nuclease and biological activity. Inorg Chem 43:7170–7179
Barton JK, Goldberg JM, Kumar CV, Turro NJ (1986) Bindingmodes and base specificity of tris (phenanthroline) ruthenium (II) enantiomers with nucleic acids: tuning the stereo selectivity. J Am Chem Soc 108:2081–2088
Bielawska A, Poplawska B, Surazynski A, Czarnomysy R, Bielawski K (2010) Cytotoxic efficacy of a novel dinuclear platinum(II) complex in human breast cancer cells. Eur J Pharmacol 643:34–41
Bloodgood BL, Sharma N, Browne HA, Trepman AZ, Greenberg ME (2013) The activity- dependent transcription factor NPAS4 regulates domain-specific inhibition. Nature 503:121–125
Brabec V, Griffth DM, Kisova A, Kostrhunova H, Zerzankova L, Marmion CJ, Kasparkova J (2012) Valuable insight into the anticancer activity of the platinum–histone deacetylase inhibitor conjugate, cis-[Pt(NH3)2malSAHA–2H)]. Mol Pharm 9:1990–1999
Dhar S, Nethaji M, Chakravarty AR (2006) DNA cleavage on photoexposure at the d–d band in ternary copper(II) complexes using red-light laser. Inorg Chem 45:11043–11050
Ding F, Zhao GY, Huang JL, Sun Y, Zhang L (2009) Fluorescence spectroscopic investigation of the interaction between chloramphenicol and lysozyme. Eur J Med Chem 44:4083–4089
Easmon J, Pürstinger G, Heinisch G, Roth T, Fiebig HH, Holzer W, Jager W, Jenny M, Hofmann J (2001) Synthesis, cytotoxicity, and antitumor activity of copper(II) and iron(II) complexes of 4 N-azabicyclo[3.2.2]nonane thiosemicarbazones derived from acyl diazines. J Med Chem 44:2164–2171
Einhorn LH (2008) First-line chemotherapy for non-small-cell lung cancer: is there a superior regimen based on histology. J Clin Oncol 26:3485–3486
Francisco C, Gama S, Mendes F, Marques F, Santos IC, Paulo A, Santos I, Coimbra J, Gabano E, Ravera M (2011) Pt(II) complexes with bidentate and tridentate pyrazolyl-containing chelators: synthesis, structural characterization and biological studies. Dalton Trans 40:5781–5792
Ferreira BJ, Brandão P, Meireles M, Martel F, Correia-Branco A, Fernandes DM, Santos TM, Félix V (2016) Synthesis, structural characterization, cytotoxic properties and DNA binding of a dinuclear copper(II) complex. J Inorg Biochem 161:9–17
Galisteo-González F, Molina-Bolívar JA (2014) Systematic study on the preparation of BSA nanoparticles. Colloids Surf B 123:286–292
Gao CY, Ma ZY, Zhang YP, Li ST, Gu W, Liu X, Tian JL, Xu JY, Zhao JZ, Yan SP (2015) Four related mixed-ligand nickel(II) complexes: effect of steric encumbrance on the structure, DNA/BSA binding, DNA cleavage and cytotoxicity. RSC Adv 5:30768–30779
Gohel A, McCarthy MB, Gronowicz G (1999) Estrogen prevents glucocorticoid-induced apoptosis in osteoblasts in vivo and in vitro. Endocrinology 140:5339–5347
Grzegorz P (2006) Luminescent metal–ligand complexes as probes of macromolecular interactions and biopolymer dynamics. Arch Biochem Biophys 453:54–62
Guo XJ, Zhang L, Sun XD, Han XW, Guo C, Kang PL (2009) Spectroscopic studies on the interaction between sodium ozagrel and bovine serum albumin. J Mol Struct 928:114–120
Han H, Lu LP, Wang QM, Zhu ML, Yuan CX, Xing S, Fu XQ (2012) Synthesis and evaluation of oxovanadium(IV) complexes of Schiff-base condensates from 5-substituted-2-hydroxy- benzaldehyde and 2-substituted-benzenamine as selective inhibitors of protein tyrosine phosphatase 1B. Dalton Trans 41:11116–11124
Hanigan MH, Devarajan P (2003) Cisplatin nephrotoxicity: molecular mechanisms. Cancer Ther 1:47–61
Huang BX, Kim HY, Dass C (2004) Probing three-dimensional structure of bovine serum albumin by chemical cross-linking and mass spectrometry. J Am Soc Mass Spectrom 15:1237–1247
Icsel C, Yilmaz VT, Kaya Y, Durmus S, Sarimahmut M, Buyukgungor O, Ulukaya E (2015) Cationic Pd(II)/Pt(II) 5,5-diethylbarbiturate complexes with bis(2-pyridylmethyl)amine and terpyridine: synthesis, structures, DNA/BSA interactions, intracellular distribution, cytotoxic activity and induction of apoptosis. J Inorg Biochem 152:38–52
Jamieson ER, Lippard SJ (1999) Structure, recognition, and processing of cisplatin–DNA adducts. Chem Rev 99:2467–2498
Jiang M, Xie XM, Zheng D, Liu Y, Li YX, Chen XY (2004) Spectroscopic studies on the interaction of cinnamic acid and its hydroxyl derivatives with human serum albumin. J Mol Struct 692:71–80
Jung Y, Lippard SJ (2007) Direct cellular responses to platinum-induced DNA damage. Chem Rev 107:1387–1407
Kelland L (2007) The resurgence of platinum-based cancer chemotherapy. Nat Rev Cancer 7:573–584
Laitinen H, Hytonen VP, Nordlund VR, Kuloma MS (2006) Genetically engineered avidins and streptavidins. Cell Mol Life Sci 63:2992–3017
Lakowicz JR, Webber G (1973) Quenching of fluorescence by oxygen. A probe for structural fluctuations in macromolecules. Biochemistry 12:4161–4170
Lepre CA, Lippard SJ, Eckstein F, Lilley DMJ (1990) Nucleic acids and molecular biology. Nucleic Acids Mol Biol 4:9–38
Li C, Qi W, Cao H, Qi Y, Zhang S, Xu S, Sun J, Guo S (2016) BSA-binding properties and anti-proliferative effects of amino acids functionalized polyoxomolybdates. Biomed Pharmacother 79:78–86
Loganathan R, Ramakrishnan S, Suresh E, Palaniandavar M, Riyasdeen A, Akbarsha MA (2014) Mixed ligand mu-phenoxo-bridged dinuclear copper(II) complexes with diimine co-ligands: efficient chemical nuclease and protease activities and cytotoxicity. Dalton Trans 43:6177–6194
Lu S, Yu S, Yang Y, Li X (2012) Spectroscopic investigation on the intermolecular interaction between N-confused porphyrins-(3-methylisoxazole) diad and bovine serum albumin. Spectrochim Acta A 99:116–121
Marmur J (1961) Procedure for isolation of deoxyribonucleic acid from micro-organisms. J Mol Biol 3:208–218
Marzano C, Trevisan A, Giovagnini L, Fregona D (2002) Synthesis of a new platinum(II) complex: anticancer activity and nephrotoxicity in vitro. Toxicol In Vitro 16:413–419
Matesanz AI, Hernández C, Rodríguez A, Souza P (2011) Novel bis(thiosemicarbazones) of the 3,5-diacetyl-1,2,4-triazol series and their platinum(II) complexes: chemistry, antiproliferative activity and preliminary nephrotoxicity studies. Dalton Trans 40:5738–5745
Mohamadi M, Ebrahimipour SY, Castro J, Torkzadeh-Mahani M (2016) Synthesis, characterization, crystal structure, DNA and BSA binding, molecular docking and in vitro anticancer activities of a mononuclear dioxido-uranium(VI) complex derived from a tridentate ONO aroylhydrazone. J Photochem Photobiol B 158:219–227
Navarro M, Cisneros-Fajardo EJ, Sierralta A, Fernández-Mestre M, Silva P, Arrieche D, Marchán E (2003) Design of copper DNA intercalators with leishmanicidal activity. J Biol Inorg Chem 8:401–408
O’Dwyer PJ, Stevenson JP, Johnson SW (1999) Clinical status of cisplatin, carboplatin and other platinum-based antitumor drugs. In: Lippert B (ed) Cisplatin: chemistry and biochemistry of a leading anticancer drug. Wiley, Weinheim, pp 29–70
Ozols RF, Bookman MA, Connolly DC, Daly MB, Godwin AK, Schilder RJ, Xu X, Hamilton TC (2004) Focus on epithelial ovarian cancer. Cancer Cell 5:19–24
Pereira-Maia E, Garnier-Suillerot A (2003) Impaired hydrolysis of cisplatin derivatives to aquated species prevents energy-dependent uptake in GLC4 cells resistant to cisplatin. J Biol Inorg Chem 8:626–634
Raghav N, Garg S, Ravish I (2016) Conversion of 2′-substituted chalcones in the presence of BSA as evidenced by (1)H NMR studies. Int J Biol Macromol 85:23–28
Roos WP, Kaina B (2013) DNA damage-induced cell death: from specific DNA lesions to the DNA damage response and apoptosis. Cancer Lett 332:237–248
Ruhayel RA, Langner JS, Oke MJ, Berners-Price SJ (2012) Chimeric platinum-polyamines and DNA binding. Kinetics of DNA interstrand cross-link formation by dinuclear platinum complexes with polyamine linkers. J Am Chem Soc 134:7135–7146
Sathyadevi P, Krishnamoorthy P, Butorac RR, Cowley AH, Bhuvanesh NSH, Dharmaraj N (2011) Effect of substitution and planarity of the ligand on DNA/BSA interaction, free radical scavenging and cytotoxicity of diamagnetic Ni(II) complexes: a systematic investigation. Dalton Trans 40:9690–9702
Schneider S, Reißner T, Ziv O, Livneh Z, Carell T (2010) Translesion synthesis of 1,3-GTG cisplatin DNA lesions. Chem Biol Chem 11:1521–1524
Sevilla P, Rivas JM, GarcÍa-Blanco F (2007) Identification of the antitumoral drug emodin binding sites in bovine serum albumin by spectroscopic methods. Biochim Biophys Acta 1774:1359–1369
Sha JQ, Li X, Qiu HB, Zhang YH, Yan H (2012) Nickel complexes of the different quinolone antibacterial drugs: synthesis, structure and interaction with DNA. Inorg Chim Acta 383:178–184
Shao J, Bao WG, Tian H, Li B, Zhao XF, Qiao X, Xu JY (2014a) Nuclease activity and protein-binding properties of a novel tetranuclear thiosemicarbazide Pt(II) complex. Dalton Trans 43:1663–1671
Shao J, Ma ZY, Li A, Liu YH, Xie CZ, Qiang ZY, Xu JY (2014b) Thiosemicarbazone Cu(II) and Zn(II) complexes as potential anticancer agents: syntheses, crystal structure, DNA cleavage, cytotoxicity and apoptosis induction activity. J Inorg Biochem 13:13–23
Shi XL, Li XW, Gui MY, Zhou HY, Yang RJ, Zhang HQ, Jin JR (2010) Systematic study on the preparation of BSA nanoparticles. J Lumin 130:637–644
Silvestri A, Barone G, Ruisi G, Anselmo D, Riela S, Liveri VT (2007) The interaction of native DNA with Zn(II) and Cu(II) complexes of 5-triethyl ammonium methyl salicylidene orto-phenylendiimine. J Inorg Biochem 101:841–848
Su R, Qi W, He Z, Zhang Y, Jin F (2008) Multilevel structural nature and interactions of bovine serum albumin during heat-induced aggregation process. Food Hydrocoll 22:995–1005
Ta L (2006) Neurotoxicity of oxaliplatin and cisplatin for dorsal root ganglion neurons correlates with platinum–DNA binding. Neurotoxicology 27:992–1002
Todd RC, Lippard SJ (2010) Structure of duplex DNA containing the cisplatin 1,2-{Pt(NH3)2}2+-d(GpG) cross-link at 1.77 Å resolution. J Inorg Biochem 104:902–908
Wang D, Lippard S (2005) Cellular processing of platinum anticancer drugs. Nat Rev Drug Discov 4:307–320
Wang BD, Yang ZY, Wang Q, Cai TK, Crewdson P (2006) Synthesis, characterization, cytotoxic activities, and DNA-binding properties of the La(III) complex with naringenin Schiff-base. Bioorg Med Chem 14:1880–1888
Wheate NJ, Evison BJ, Herlt AJ, Phillips DR, Collins JG (2003) DNA binding of the anti-cancer platinum complex trans-[{Pt(NH3)2Cl}2-μ-dpzm]2+. Dalton Trans 18:3486–3492
Wolf A, Shimer GHJ, Meehan T (1987) Polycyclic aromatic hydrocarbons physically intercalate into duplex regions of denatured DNA. Biochemistry 26:6392–6396
Zhen W, Link CJ, O’Connor PM, Reed E, Parker R, Howell SB, Bohr VA (1992) Increased gene-specific repair of cisplatin interstrand cross-links in cisplatin-resistant human ovarian cancer cell lines. Mol Cell Biol 12:3689–3698
Acknowledgements
This work were financially supported by the National Natural Science Foundation for Young Scientists of China (21301150, 21571154), the Natural Science Foundation of the Jiangsu Higher Education Institutions of China (13KJB150037, 14KJB150027), the Post-Doctoral Foundation of Jiangsu Provincial (1501032B), the six Taleng Peak Project in Jiangsu Province (SWYY-063), the practice inovation trainng program projects for the Jiangsu College students (201310324014Z, 201410324053), the Natural Science Foundation of Yancheng Teachers University (10YCKL017,14YCKL006) and sponsored by Qing Lan Project of Jiangsu Provices.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, Q., Yang, L., Wu, J. et al. Four mononuclear platinum(II) complexes: synthesis, DNA/BSA binding, DNA cleavage and cytotoxicity. Biometals 30, 17–26 (2017). https://doi.org/10.1007/s10534-016-9984-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-016-9984-7