Abstract
We performed field measurements on the spatial and temporal variability in CH4 emissions from stem surfaces of mature Fraxinus mandshurica Rupr. trees in a floodplain forest of northern Japan. Stem CH4 fluxes were measured by a static closed-chamber method at ca. 15 cm above ground on ten selected trees to test among-individual variability, and the diurnal and seasonal changes in three representative trees. Daytime stem CH4 emission rates varied between 81 and 1,305 µg CH4 m−2 h−1 among individual trees, and showed a spatial gradient apparently corresponding to the difference in water table depth at the experimental site. Stem CH4 fluxes were quite stable throughout a 24 h period for foliated trees in August and were similar for defoliated trees in November. Large differences were observed in the magnitude of seasonal changes in stem CH4 flux among individual trees; one sampled tree showed no clear seasonal changes in stem CH4 flux, while another tree exhibited drastic seasonal changes ranging larger than one order of magnitude. Results demonstrated the high variability in stem CH4 emissions in space and time, and suggested the importance of soil temperature, water table depth and porewater CH4 concentration as possible environmental factors controlling stem CH4 emissions from temperate forested wetlands.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Methane (CH4) has a strong greenhouse effect, and its global warming potential is 25 times greater than carbon dioxide for a 100 year time horizon (WMO 2013a). Global abundance of CH4 as a mole fraction was 1,819 ± 1 ppb in 2012, which is 260 % of its preindustrial level of 700 ppb; an annual increase averaging 3.7 ppb year−1 has been observed during the last 10 years (WMO 2013b).
Anaerobic soils in natural wetlands and rice paddies are the greatest producers of atmospheric CH4; bottom-up global estimates of CH4 emissions from those two sources for the decade of 2000–2009 were 177–284 Tg year−1 and 33–40 Tg year−1, respectively; these constitute substantial parts of the total global emission of 542–852 Tg year−1 (Ciais et al. 2013). In these ecosystems, methanogens produce CH4 in anoxic soils. The CH4 is subsequently released into the atmosphere by three different pathways: molecular diffusion at soil/water–atmosphere interfaces; ebullition of gas bubbles; and plant-mediated transport (Carmichael et al. 2014; Joabsson et al. 1999; Schütz et al. 1991). Among these three pathways, plant-mediated gas transport through the internal air space of plant bodies makes the greatest contribution to total CH4 flux from soil to the atmosphere, causing more than 90 % of the total flux in various ecosystems (Schütz et al. 1989; Shannon et al. 1996; Whiting and Chanton 1992).
Studies have documented plant-mediated CH4 transport in rice plants (Cicerone and Shetter 1981; Holzapfel-Pschorn et al. 1986; Nouchi et al. 1990; Schütz et al. 1989; Seiler et al. 1984), aquatic plants (Chanton et al. 1992; Dacey and Klug 1979; Sebacher et al. 1985), and wetland herbaceous plants (King et al. 1998; Shannon et al. 1996; Whiting and Chanton 1992). These plants develop aerenchyma tissue, i.e., internal lacunae formed by cell separation or cell breakdown, in their roots and culms through which atmospheric oxygen can be transported downward to the anoxic rhizosphere (Armstrong 1979). Plant-mediated CH4 emissions, thus, are the upward transportation of soil-borne CH4 through the aerenchyma tissue of plants that have adapted well to anoxic soil environments (Schütz et al. 1991).
As a response to soil inundation, several flood-tolerant woody tree species, such as Fraxinus or Alnus spp. in temperate forests and some species in tropical regions, can develop various adaptive changes in the anatomy and morphology of roots and stems including aerenchyma formation that allows their roots to survive anoxic belowground environments (De Simone et al. 2002; Grosse and Schröder 1984; Hook 1984; Yamamoto et al. 1995a; Yamamoto et al. 1995b). Therefore, woody species could also most likely mediate the transport of CH4 from the soil to the atmosphere (Carmichael et al. 2014; Schütz et al. 1991). In fact, some experimental studies in laboratories or mesocosms have demonstrated that CH4 is transported through bodies of tree seedlings or saplings from the submerged soil layer or methane-enriched root zone to the atmosphere (Garnet et al. 2005; Machacova et al. 2013; Pangala et al. 2014; Rice et al. 2010; Rusch and Rennenberg 1998; Vann and Megonigal 2003). Furthermore, in recent field studies, emissions of CH4, which were presumably produced anaerobically in the submerged soil layer, from the stem surfaces of mature trees have been measured in temperate wetland forests (Gauci et al. 2010; Terazawa et al. 2007) and a tropical forested peatland (Pangala et al. 2013), as have CH4 emissions from pneumatophores of mangroves (Kreuzwieser et al. 2003; Purvaja et al. 2004) and cypress knees (Pulliam 1992). The field study by Pangala et al. (2013) noted CH4 emissions from tree stems accounted for at least 62 % of the total CH4 emissions from the tropical forested peatland in Southeast Asia (Pangala et al. 2013). To date, however, details related to the nature of CH4 emissions from tree stems in natural wetlands remain unclear. To our knowledge, a very few field studies have addressed the variability of CH4 emission rates, factors controlling those rates and the underlying mechanisms of CH4 emissions from tree stems (Pangala et al. 2013), although some experimental studies have attempted to tackle these issues (Pangala et al. 2014).
In this paper, we present the results of a field study on the spatial and temporal variability of CH4 emissions from stem surfaces of mature Fraxinus trees in a temperate floodplain forest. The specific aims of this paper are to show: (1) the variations of stem CH4 emission rates among individual trees in a site with a spatial gradient of belowground water regime; (2) diurnal changes in stem CH4 emission rates in both foliate and defoliate periods of deciduous Fraxinus trees; (3) seasonal changes in stem CH4 emission rates and possible related changes in the belowground environment, such as soil temperature, water table depth and porewater CH4 concentration.
Materials and methods
Study site
This study was conducted in a mature stand of F. mandshurica Rupr., a deciduous tall tree species, in central Hokkaido, northern Japan. The research site was located on a floodplain along a small mountain stream (43°22′N, 141°36′E; 60 m a.s.l.). The canopy was dominated by F. mandshurica trees planted in 1931, and the subcanopy was composed of Salix udensis Trautv. et C. A. Mey. Mean height, mean diameter at breast height (DBH), tree density, and relative dominance in the basal area of F. mandshurica were 28.1 ± 1.7 m (mean ± SD), 30.7 ± 4.0 cm, 183 trees ha−1, and 74 %, respectively. From June through August, the forest floor was mostly covered by large perennial herbs such as Filipendula camtschatica (Pall.) Maxim., Fallopia sachalinensis (F. Schmidt) Ronse Decr., and Phragmites australis (Cav.) Trin. ex Steud. The maximum height of forest floor vegetation reached ca. 4 m above ground in August.
The annual mean air temperature at the study site is 6.0 °C, with the highest monthly mean temperature in August (19.5 °C) and the lowest in January (−6.8 °C) (Japan Meteorological Agency 2002). Total annual precipitation is 1,460 mm, of which approximately 470 mm falls in the form of snow from December to March (Japan Meteorological Agency 2002).
The soil at the study site was composed of alluvial deposits with depths exceeding 1.5 m and classified as a Gleysol (IUSS Working Group WRB 2006). The mostly loam and clay-loam soil contained no gravel and had a pH of 5.4 at the surface.
Variation in stem CH4 flux among individual trees
A 20 × 60 m experimental plot was set up in the stand (Fig. 1). This plot was established by extending the former experimental quadrat from our previous work (Terazawa et al. 2007). Ten dominant F. mandshurica trees within the plot were selected for the measurements of CH4 flux from the stem surfaces (referred as “stem CH4 flux”, hereafter). Sampled trees ranged from 26 to 31 m (mean 28.0 ± 1.8 m) in height and 26–39 cm (mean 30.7 ± 4.4 cm) in DBH. Each sampled tree had a single trunk without branches and apparent scars from injury or stem disease up to at least 3 m above ground level.
Locations of trees and measuring points for belowground environment in the experimental plot for the study of stem CH4 flux in a floodplain forest of northern Japan. The plot is 20 × 60 m. A dashed line indicates the boundary between two vegetation types of the forest floor, i.e., Filipendula and Phragmites populations. Larger closed circles with Arabic numbers indicate the trees for the CH4 flux measurement at the stem surface
Stem CH4 flux was measured using a static closed-chamber method in the daytime (between 10:00 and 16:00) on July 20–22, 2011. A custom-made stainless steel chamber (70 mm wide × 180 mm long × 80 mm high; Alfa Kikai Co. Ltd., Ibaraki, Japan) with an acrylic lid was used for the flux measurements. A chamber was attached to a stem surface at ca. 15 cm above ground using a urethane frame placed between the chamber and bark. Gaps between the chamber, frame, and tree bark were sealed with pulp clay (Kutsuwa Co., Ltd., Osaka, Japan). The chamber body had a fringe (10 mm in width) on the edge of its open end, and an acrylic lid was fixed firmly on the fringe with silicone rubber packing and spring clips when gas flux measurement was started. Air inside the chamber was sampled at 0, 10, and 20 min after chamber closure through a silicone rubber septum attached on the lid using a 50 ml gas-tight plastic syringe. Samples of gases (40 ml each) were injected into pre-evacuated 30 ml glass vials with butyl rubber stoppers. A Tedlar® bag containing 120 ml air was attached to each chamber to allow the inside pressure to equilibrate with atmospheric pressure during gas sampling. Chambers were shaded by aluminum foil during measurement to prevent overheating from direct sunlight.
Diurnal change in stem CH4 flux
Diurnal changes in stem CH4 flux from the stem surfaces were measured for the three representative individuals of the ten dominant F. mandshurica trees that were also used for the measurement of among-tree variability in stem CH4 flux described above. Among these three individuals, two trees (Trees 2 and 5) were located within the site where a Filipendula population dominated the forest floor, while another individual (Tree 8) was located within a Phragmites population (Fig. 1). The three individuals (Trees 2, 5, and 8) were located within several meters (≤6 m) from each corresponding point that was used for measurement of the belowground environment, i.e., 10, 30 and 50 m points (Fig. 1). Heights of the sampled trees (Trees 2, 5, and 8) were 28, 26 and 30 m, and DBHs were 29, 26 and 34 cm, respectively.
Stem CH4 fluxes of the three sampled trees were concurrently measured seven times at 4-h intervals during each 24 h on August 9–10 and November 1–2, 2011. The August and November measurements represented the foliate and defoliate periods of Fraxinus trees, respectively. Procedures for CH4 flux measurement were the same as described above.
Seasonal change in stem CH4 flux
Seasonal changes in the stem CH4 emission rate were measured for the same three representative trees of F. mandshurica (Trees 2, 5, and 8) on which the diurnal changes in stem CH4 flux were measured. The flux measurements were conducted from May 25 to October 30 in 2012, and again from August 2 to November 1 in 2013 with an almost 1-month interval between measurements. Measurements of stem CH4 flux in May, October and November were conducted in the leafless season of canopy trees, and the sampled trees had no intact leaves on their crowns on these measurement days. Procedures for CH4 flux measurement were the same as described above.
Stem CH4 fluxes were measured regularly at the fixed position on a stem in each sampled tree throughout the study. To test the effect of position of CH4 flux measurement in a circumferential direction on a stem surface, CH4 fluxes were measured at two different positions on the same height (ca. 15 cm above ground) for each sampled tree on August 30, 2012. The two positions (Position A and B) were separated at least a quarter of a circle on a stem; the Position A was the regular position for stem CH4 measurements in this study.
Environmental variables
Water table depths and soil temperatures at 112.5 cm below the soil surface were monitored at five points with 10 m-intervals along the center line of the experimental plot (Fig. 1) using water height data loggers (TruTrack WT-HR 1500; Intech Instruments Ltd., Christchurch, New Zealand), which were set in perforated PVC pipes, in 2011 and 2012. In 2013, the number of measuring points for water table depth and soil temperature was reduced to three, with measurements taken at 10, 30 and 50 m points within the plot (Fig. 1). Soil temperatures were also monitored at 25 and 50 cm below the soil surface by thermistor sensors and data loggers (UIZ3633, Uizin, Tokyo, Japan) at the center of the experimental plot. Water table depth and soil temperature at 112.5 cm were recorded at 6 h intervals, and those for soil temperature at 25 and 50 cm were 1 h intervals.
On each day of the stem CH4 flux measurements, air temperature and relative humidity at 1 m above ground were recorded by a temperature/relative humidity logger (HOBO U23-001; Onset Computer Corporation, Bourne, MA., USA) with 15 min intervals at the center of the experimental plot. For the measurements of diurnal change in stem CH4 flux, in particular, vapor pressure deficit (VPD) was calculated from the temperature and relative humidity data, and the photosynthetic photon flux density (PPFD) was monitored at the height of 5 m above ground at an open site close to the experimental plot by a quantum sensor (PAR-001; Prede Co., Ltd., Tokyo, Japan) and a portable recorder (Model 3057; Yokogawa Electric Corporation, Tokyo, Japan).
Daily precipitation and daily mean temperature data from May to November in 2012 and 2013 were obtained from the Tsukigata Regional Meteorological Station, which was located 6 km southeast of the study site.
Porewater CH4 concentration
On the day or following day of the stem CH4 flux measurement in 2012 and 2013, porewater CH4 concentration in the soil was measured by the following procedures based on in situ sampling method for dissolved components in sediment porewater (Hesslein 1976). A ceramic porous cup (55 mm long, 18 mm in external diameter, 1.5 mm thick; Sekiya Rika Co., Ltd., Tokyo, Japan) attached to a short acrylic pipe (60 mm long, 22 mm in external diameter, 3 mm thick) was filled with 17 ml distilled-deionized water. The open end of the pipe was plugged by a silicone stopper with a careful attention to purge any bubbles inside the pipe as much as possible through an injection needle inserted in the stopper. After the plugging was completed, the injection needle was removed from the stopper. This water-filled porous cup was attached at the end of a 150-cm-long PVC pipe, buried at a given depth in the soil and allowed to equilibrate with porewater for at least 13 days. The porous cup was collected from the soil on the day the porewater CH4 concentration was measured. Immediately after the porous cup collection, 10 ml water was sampled from the cup by a 50 ml gas-tight plastic syringe without any exposure of the sampled water to ambient air. Then, 40 ml of N2 gas was added to the syringe, and the syringe was shaken by hand for 3 min to equilibrate the gas concentration between the gaseous and liquid phases in the syringe. A 15 ml sample of gas was taken from the headspace and injected into a pre-evacuated 10 ml glass vial thorough a butyl rubber stopper.
Porewater CH4 concentrations were measured at 80 and 120 cm depths in 2012 and at 120 cm depth in 2013 at three points in the experimental plot (Fig. 1). These points for measurement of the porewater CH4 concentration were set at 2 m apart from each corresponding point for water table measurement (10, 30 and 50 m point) to avoid possible interference from the perforated pipes for water depth measurement.
Soil profile and carbon content
Soil cores were taken by a 1.5 m long soil auger within a distance of 2 m from each measurement point for the belowground environment (10, 30, and 50 m point; Fig. 1). On each soil core, the soil profile was carefully observed with special attention to noting the depths of the gley horizon with or without oxidized iron mottles. Soil samples taken from each 10 cm depth of the soil cores were used for carbon content determination. Carbon content of the air-dried soil samples was determined by an NC analyzer (Sumigraph NC-220F, Sumika Chemical Analysis Service, Ltd., Tokyo, Japan).
Laboratory gas analysis and calculation of stem CH4 flux and porewater CH4 concentration
For the determination of stem CH4 flux and porewater CH4 concentration, the CH4 concentration of each gas sample was determined using a gas chromatograph (GC-14BPTF; Shimadzu Co., Kyoto, Japan) equipped with a flame ionization detector and a Unibeads C column (2 m × 1.5 mm diameter; GL Science, Tokyo, Japan).
Stem CH4 flux was calculated using a linear regression for the time course of gas concentration in the chamber. Of the regression coefficients for linear regressions, 93 % of the individual measurements were higher than 0.999, and the minimum regression coefficient value was 0.993 except for the lowest value of 0.890 which was deleted from further analysis. The porewater CH4 concentration was calculated using the Henry’s Law from the headspace CH4 concentration inside the syringe and the ambient temperature at the time when the syringe was shaken.
Statistical analysis
A generalized linear model (GLM) was used to examine the effects of environmental variables on the variation in stem CH4 flux for the three representative trees (Trees 2, 5, and 8). The explanatory variables included to the models were soil temperature at 50 cm below soil surface, water table depth, porewater CH4 concentration, tree individuals, and interaction terms between tree individuals and each environmental variable. Soil temperature at 25 and 112.5 cm below soil surface were not included to the model because of the multicollinearity with soil temperature at 50 cm below soil surface. Selection of explanatory variables was conducted based on Akaike’s information criterion (AIC). The response variable was assumed to follow a gamma distribution and a reciprocal function was adopted as a link function in the GLM analysis. The statistical software R version 3.1.1 (R Core Team 2014) was used for the analysis.
Results
Variation in stem CH4 flux among individual trees
Substantial stem CH4 emissions were detected for all ten trees measured. Daytime stem CH4 emission rates at the height of ca. 15 cm above ground varied between 81 and 1,305 µg CH4 m−2 h−1 among the trees (Fig. 2a).
Stem CH4 emission rates seemed to have a spatial gradient within the experimental plot, increasing with the distance on the Y-axis of the plot (Fig. 2a). The difference in stem CH4 flux corresponded well to both the difference in belowground water table depth and forest floor vegetation within the experimental plot (Fig. 2a, b). Eight individual trees in the Filipendula population, where water table depths were around 100 cm below the soil surface, showed relatively low stem CH4 emission rates ranging between 81 and 512 µg CH4 m−2 h−1. In contrast, two individual trees growing in a Phragmites population, where the water table laid just a few centimeters below soil surface, emitted CH4 at a rate exceeding 800 µg CH4 m−2 h−1. No clear correlation was found between stem CH4 emission rates and DBH of the trees.
Diurnal change in stem CH4 flux
In both sets of measurements in August and November, stem CH4 emissions were observed throughout a 24 h period (Fig. 3a, e). Diurnal variations of stem CH4 emission rates were relatively small for all of the measurement series. For Trees 2, 5 and 8, average stem CH4 emission rates were 119, 215 and 189 µg CH4 m−2 h−1 on August 9–10, and 84, 154, 494 µg CH4 m−2 h−1 on November 1–2, respectively. The coefficient of variance of stem CH4 emission rates of each measurement series fell between 0.02 and 0.08. No distinct pattern of variation was found in the diurnal change of stem CH4 emission rates, except for Tree 8 in the November measurement which showed apparent slight decrease in stem CH4 flux at night (from 18:00 to 6:00).
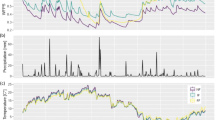
Diurnal changes in stem CH4 flux of F. mandshurica trees (a, e), PPFD (photosynthetic photon flux density) (b, f), air temperature and VPD (vapor pressure deficit) (c, g), and soil temperature (d, h) in a floodplain forest of northern Japan. Stem CH4 fluxes were measured on three canopy trees (Trees 2, 5, and 8; Fig. 1) at 4-h intervals for 24 h on August 9–10 (a), and November 1–2 (e), 2011. Measurement intervals of soil temperatures were 1 h for 25 and 50 cm, and 6 h for 112.5 cm depths. Night is shown by the gray belt in each graph
Air temperature varied gently between 20.8 and 27.1 °C during the stem CH4 measurements in August, corresponding to the diurnal change in PPFD (Fig. 3b, c). In November, air temperature changed more drastically during the stem CH4 measurements, ranging between −0.9 °C and 15.3 °C with a consistent decline throughout the night (Fig. 3g). Changes in VPD were similar to those of PPFD in both August and November (Fig. 3c, g). Soil temperatures were stable during the stem CH4 measurements even at the upper most measuring depth, i.e., 25 cm (Fig. 3d, h).
Water table depths on the day of stem CH4 flux measurements on August 9–10 were 115, 89 and 11 cm below soil surface, at the 10, 30 and 50 m points, respectively. Water table depths on November 1–2 were 69, 46, and 20 cm, at the 10, 30, and 50 m points, respectively.
Seasonal change in stem CH4 flux
Large differences were observed in the range of seasonal variation of stem CH4 emission rates among the three sampled trees. Stem CH4 emission rates of Tree 2 were rather stable throughout the entire period of measurement, ranging between 59 and 124 µg CH4 m−2 h−1 (Fig. 4a, e). In contrast, Tree 8 showed a drastic change in stem CH4 emission rates during the measuring period. In 2012, the stem CH4 emission rate of Tree 8 peaked in late August at 1,514 µg CH4 m−2 h−1 (Fig. 4a); similarly in 2013, it peaked in late September at 1,492 µg CH4 m−2 h−1 (Fig. 4e). The magnitude of seasonal change in stem CH4 emission rates of Tree 5 was intermediate between the other two individuals (Trees 2 and 8), ranging between 139 and 331 µg CH4 m−2 h−1 (Fig. 4a, e). A late-summer to early-autumn increase in the stem CH4 emission rate observed in Tree 5 was less drastic than that observed in Tree 8.
Seasonal changes in stem CH4 flux of F. mandshurica trees in a floodplain forest of northern Japan in 2012 (a) and 2013 (e). Water table depth and precipitation (b, f), soil temperature (c, g), and air temperature (d, h) during the corresponding period are also shown. Stem CH4 fluxes were measured at three canopy trees (Trees 2, 5, and 8). Each measuring point of water table depth (10, 30 and 50 m points) corresponded to each tree for stem CH4 flux measurement, Trees 2, 5, and 8, respectively. Precipitation and air temperature data were obtained from the Tsukigata Regional Meteorological Station located 6 km southeast of the study site
On August 30, 2012, differences in stem CH4 emission rates measured at two positions at the same height in Trees 2, 5, and 8 were 106, 32 and 162 µg CH4 m−2 h−1, accounting for 60, 9 and 10 % of the mean stem CH4 emission rate of each respective tree on that day (Fig. 5).
The ranges of fluctuation in the water table depth differed among the three measuring points (Fig. 4b, f); the fluctuation range during the period between May 25 and November 1 in each year was smallest at the 10 m point (38–70 cm below soil surface in 2012, and 55–76 cm in 2013). It was the largest at the 50 m point (18–72 cm below soil surface in 2012, and 16–85 cm in 2013). Intermediate measurements were observed at the 30 m point (28–65 cm below soil surface in 2012, and 24–54 cm in 2013). Mean depths of the water table during the study period at the 10, 30 and 50 m point were 60, 48 and 40 cm below the soil surface in 2012, respectively, and 68, 37 and 39 cm below the soil surface in 2013, respectively.
Soil temperatures at 112.5 cm in depth peaked in late-September in 2012 and in early-September in 2013 (Fig. 4c, g).
Porewater CH4 concentration
Large differences were observed in porewater CH4 concentration among the measuring points at both 80 and 120 cm below the soil surface (Fig. 6). Porewater CH4 concentrations were consistently highest at the 50 m point, exceeding 600 µmol l−1 throughout the study period at both depths. In contrast, at the 10 m point, porewater CH4 concentrations were 10−4 to 10−3-fold lower than those of the 50 m point during the initial stage of the experiment period. Porewater CH4 concentrations at the 30 m point were intermediate between 50 and 10 m points at both depths.
Seasonal changes in porewater CH4 concentrations at 80 cm (a) and 120 cm (b) below the soil surface in 2012, and 120 cm below the soil surface in 2013 (c) in a floodplain forest of northern Japan. Each measuring point for porewater CH4 concentration (10, 30, and 50 m points) corresponded to each tree used for stem CH4 flux measurement, Trees 2, 5, and 8, respectively
Porewater CH4 concentrations increased consistently from late May to late September in 2012 at both depths of the 10 and 30 m points (Fig. 6a, b). A consistent increase in porewater CH4 concentration was also observed at the 120 cm deep of the 10 m point in 2013 (Fig. 6c).
Effects of environmental variables on stem CH4 flux
As a result of the GLM analysis, soil temperature at 50 cm deep, water table depth, porewater CH4 concentration at 120 cm below the soil surface, tree individuals, and an interaction between porewater CH4 and tree individuals were selected as important factors in the best model for stem CH4 flux (Table 1). Since a reciprocal function was adopted as a link function in the GLM analysis, a negative value of the estimated parameter represents a positive effect of the variable on the stem CH4 flux, and vice versa. Soil temperature and water table depth tended to have a positive and a negative effect on stem CH4 flux, respectively (Table 1, Fig. 7). Porewater CH4 concentration tended to have positive effects on stem CH4 flux (Table 1, Fig. 7). However, the effect of porewater CH4 concentration on stem CH4 flux differed between tree individuals, because an interaction term (porewater CH4 × tree) was in the model (Table 1). Stem CH4 flux also differed between tree individuals (Table 1).
Relationships between stem CH4 flux of F. mandshurica trees and environmental variables in a floodplain forest of northern Japan: porewater CH4 concentration at 120 cm below the soil surface (a), water table depth (b), and soil temperature at 50 cm below the soil surface (c). Data collected during the daytime measurements of diurnal changes (August and November, 2011) and seasonal changes (May, 2012–November, 2013) in stem CH4 flux were plotted
Soil profile and carbon content
The gley horizon and the layer with mottles of oxidized iron were observed in each soil core taken from the 10, 30 and 50 m points. Depths of these reduced layers in the soil cores differed among sampling points. A gley horizon (5Y 4/1–7.5GY 4/1 in Munsell color notation) without mottles was observed from a depth deeper than 100 cm at the 10 m point, while they were observed from shallower depths at the 30 and 50 m points, appearing at 75 and 70 cm below the soil surface, respectively. The pseudogley horizon (2.5Y 4/2–2.5Y 4/3) containing mottles of oxidized iron was observed below 60 cm deep at the 10 m point, while it appeared at the much shallower depth of 20 cm below the soil surface at both the 30 and 50 m points.
Total carbon content of the surface soil was 3.8–4.3 %, and decreased exponentially with soil depth. Carbon content was higher at the 50 m point when compared with the 10 and 30 m points to a depth of 60 cm.
Discussion
In the present study, large variations were observed in stem CH4 emission rates among individual trees within a site; the CH4 emission rates differed greater than one order of magnitude between individual trees in July, 2011 (Fig. 2a) and in late-summer to early-autumn in 2012 and 2013 (Fig. 4a, e). This result simply suggests that we should pay attention to the possibility of inter-individual or spatial variability in stem CH4 emissions, when we attempt to precisely evaluate the impact of stem CH4 emissions on the CH4 budget at an ecosystem or global scale by scaling up the CH4 flux measured on individual trees in a small area. When compared with these large variations among individual trees, differences in CH4 emission rates measured at two different horizontal positions on an individual tree stem were relatively small (Fig. 5); this allowed us to discuss the inter-individual and spatial variability in stem CH4 flux in a site in more detail.
The variation in stem CH4 emission rates among ten sampled trees in the present study showed a spatial gradient, which apparently corresponded to the difference in water table depth (Fig. 2). This result implies that one of the possible rate-controlling environmental factors of stem CH4 emissions may be the depth of the water table, which alters the magnitude of anaerobic/aerobic conditions in the soil, resulting in an alteration in CH4 production/oxidation rates and CH4 concentration in the root zone (e.g., Christensen 2010). Water table depth was also selected as an important explanatory variable in the GLM analysis which was performed to examine the effects of environmental variables on the stem CH4 flux for the three representative trees (Trees 2, 5, and 8; Table 1; Fig. 7). Pangala et al. (2014) recently reported an experimental study showing water table depth significantly influenced CH4 emissions from tree stems of pot-grown saplings of Alnus glutinosa.
In the present study, stem CH4 fluxes were quite stable throughout a 24-h period, and no distinct patterns of change in stem CH4 flux were found in both August and November measurements on foliated and defoliated trees, respectively, except for one case (Tree 8 in November, 2011), (Fig. 3a, e). Many of the previous studies on CH4 emissions from natural wetlands with herbaceous plants or rice paddies reported considerable diurnal changes in CH4 emission rates (e.g., Holzapfel-Pschorn and Seiler 1986; Shannon et al. 1996; van der Nat et al. 1998; Whiting and Chanton 1996). These changes were mainly associated with soil or peat temperature, which is one of the factors responsible for the belowground CH4 production rate (Holzapfel-Pschorn and Seiler 1986; Shannon et al. 1996). Changes in CH4 emission rates have also been associated with light regime for some plant species in which convective CH4 transport occurs during the day (van der Nat et al. 1998; Whiting and Chanton 1996). Why, then, were stem CH4 emission rates so stable during a day in the present study? One possible reason may be the diel stability of soil temperature in the deeper layer where CH4 was produced under water-saturated conditions. Water table depths on the days of the August and November measurements in the present study were 11 and 20 cm at highest, and 115 and 69 cm at lowest, respectively, and the soil temperatures at 25 cm and deeper were quite stable during each day of stem CH4 flux measurement (Fig. 3d, h). The results of the GLM analysis for the stem CH4 flux also suggests soil temperature is an important factor responsible for controlling stem CH4 emission rates (Table 1; Fig. 7). No marked diel variation of stem CH4 emissions was similarly reported from the experimental study using Alnus glutinosa saplings under flooded soil conditions (Pangala et al. 2014).
In the present study, almost identical rates of CH4 emissions were observed at both night and day (Fig. 3a, e). In addition, comparable or even higher CH4 emission rates were measured in November when all sampled trees had no intact leaves on their crowns as compared with August measurements for foliated trees (Fig. 3a, e). These results suggest that the transpirational stream in tree stems may have no or only a very small contribution to the CH4 emissions from tree stems. CH4 probably could not be transported in a dissolved form in the upward transpirational stream. Instead, CH4 was most likely transported in a gaseous form through internal air spaces in the tree body, such as aerenchyma tissues or intercellular spaces associated with a flooded soil condition (De Simone et al. 2002; Yamamoto et al. 1995a, b). As for anatomical evidence for a possible gas conduit in a mature tree body of F. mandshurica at the present experimental site, development of the aerenchyma tissues was observed in the cortex of fine roots, which were sampled from 10–60 cm deep in the soil at the north edge of the site (Yamamoto et al. unpublished data). Similarly, in their experimental study of CH4 and N2O emissions from seedling stems of Fagus sylvatica and Alnus glutinosa, Machacova et al. (2013) assumed that CH4 was transported through the aerenchyma system rather than via the xylem sap because of the low solubility of CH4 in water. Pangala et al. (2013) also reported a significant positive relationship between stem CH4 flux and wood specific density, which is an indicator of wood properties including porosity and anatomical composition, for seven tree species coexisting in a tropical forested peatland in Southeast Asia. Their studies suggested the possibility of the existence of a gaseous transport of CH4 through tree bodies in these species.
Large differences were found in the magnitude and pattern of seasonal changes in stem CH4 flux among three sampled trees (Fig. 4a, e). Tree 2, which showed the lowest stem CH4 emission rate with no clear seasonal changes, was located near the 10 m point where the water table depth remained below 50 cm deep during most of the study period (Fig. 4b, f). In contrast, Tree 8, which showed drastic seasonal changes in the stem CH4 emission rate, was located at the 50 m point where water table depth and its range of variation were much higher than those of the 10 m point (Fig. 4b, f). The contrasting water regimes between the 10 and 50 m points were clearly reflected to their soil profiles, i.e., depth of the gley horizon and thickness of the layer with mottles of oxidized iron. The water table depth and its seasonal fluctuation have been frequently reported as predominant factors relating to the plant-mediated CH4 emission rates at various wetland ecosystems (Shannon and White 1994; Treat et al. 2007; Turetsky et al. 2008; Waddington et al. 1996). Therefore, the difference in magnitude and patterns of seasonal changes in stem CH4 emissions among the sampled trees in the present study may be, in part, the result of the different water regimes within the experimental plot. The GLM analysis for the stem CH4 flux also supported this hypothesis (Table 1; Fig. 7).
Comparable large seasonal changes in stem CH4 emission rates to those in the present study were reported in Alnus glutinosa trees in a wetland in the United Kingdom (Gauci et al. 2010). In that study, one of the sampled trees showed an increase of the stem CH4 emission rate of over 40 times during almost 1 month from the early May to early June (Gauci et al. 2010). Relatively small seasonal changes in the average stem CH4 flux measured in 2005 at the same stand of the present study (Terazawa et al. 2007) may be attributed to the location of the measurement. The measurements in 2005 were conducted on five Fraxinus trees including Trees 2 and 5 in the southern half of the present experimental plot where water table was relatively low and fluctuated less.
Large differences in the magnitude and pattern of seasonal changes in porewater CH4 concentrations were also found (Fig. 6). The porewater in the 50 m point, in the immediate vicinity of Tree 8, was apparently supersaturated with CH4 throughout the study period, given the solubility of CH4 in distilled water (ca. 1,850 µmol l−1 at 12 °C: Yamamoto et al. 1976). The consistent increases of porewater CH4 concentrations at the 10 and 30 m points during the study period may have been caused by the accumulation of CH4 produced in the saturated soil layer over time (Schütz et al. 1989). Seasonal increases in the porewater CH4 concentration were reported from other temperate wetlands (Shannon et al. 1996; Sun et al. 2012), although the magnitudes of increase were much smaller than observed in the present study.
Results of the GLM analysis related to the stem CH4 flux showed that porewater CH4 concentration had a positive effect on the stem CH4 emission rates with an interaction with tree individuals (Table 1; Fig. 7). The results are in agreement with other recent studies related to CH4 emissions from tree stems in which positive relationships were detected between the stem CH4 flux and the porewater CH4 concentration in mesocosms (Machacova et al. 2013; Pangala et al. 2014) as well as in a natural tropical wetland (Pangala et al. 2013). The interaction between porewater CH4 concentration and tree individuals in the present study implies that structural or/and physiological properties of a tree might have an effect on stem CH4 flux along with porewater CH4 concentration. However, no evidence was obtained in the present study on the parameters that were responsible for the difference in tree properties related to stem CH4 flux.
Results of the present field study suggest that the CH4 emitted from stem surfaces of wetland trees may be produced in the submerged soil layer, and it may be transported in a gaseous form through internal air spaces in tree bodies, as has also been observed for various wetland herbaceous plants and rice (e.g., Cicerone and Shetter 1981; Nouchi et al. 1990; Shannon et al. 1996; Whiting and Chanton 1992). Forested wetlands represent up to 60 % of the total wetland area worldwide (Matthews and Fung 1987). According to recent studies of upscaled CH4 emissions from wetland trees, the estimated annual amount of tree-mediated CH4 flux on a global scale ranged from 2 Tg yr−1 (Carmichael et al. 2014) to 60 ± 20 Tg yr−1 (Rice et al. 2010), which could account for 0.2 and 10 % of the global CH4 source, respectively. To gain a better understanding and more accurate estimation of CH4 emission rates from woody plants in forested wetlands, further field and experimental studies will be needed. Such studies should particularly focus on (i) biogeochemical processes and factors involved in controlling stem CH4 emissions, (ii) vertical variations in CH4 flux on a tree stem up to the canopy level, which mostly showed a decreasing tendency as far as it was measured below the breast height level (Pangala et al. 2013; Terazawa et al. 2007), for more accurate evaluation of the contribution of CH4 emissions from entire trees to the total CH4 flux of a forested wetland, and (iii) the possibility and magnitude of CH4 production in living trees and its release to the atmosphere (Covey et al. 2012; Zeikus and Ward 1974).
References
Armstrong W (1979) Aeration in higher plants. In: Woolhouse HW (ed) Advances in Botanical Research, vol 7. Academic Press, London, pp 226–332
Carmichael MJ, Bernhardt ES, Bräuer SL, Smith WK (2014) The role of vegetation in methane flux to the atmosphere: should vegetation be included as a distinct category in the global methane budget? Biogeochemistry 119:1–24
Chanton JP, Martens CS, Kelley CA, Crill PM, Showers WJ (1992) Methane transport mechanisms and isotopic fractionation in emergent macrophytes of an Alaskan tundra lake. J Geophys Res 97(D15):16681–16688
Christensen TR (2010) Wetlands. In: Reay D, Smith P, van Amstel A (eds) Methane and climate change. Earthscan, London, pp 27–41
Ciais P, Sabine C, Bala G, Bopp L, Brovkin V, Canadell J, Chhabra A, DeFries R, Galloway J, Heimann M, Jones C, Le Quéré C, Myneni RB, Piao S, Thornton P. (2013) Carbon and other biogeochemical cycles. In: Stocker TF, Qin D, Plattner G-K, Tignor M, Allen SK, Boschung J, Nauels A, Xia Y, Bex V, Midgley PM (eds) Climate change: the physical science basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change, Cambridge University Press, Cambridge, pp 465–570
Cicerone RJ, Shetter JD (1981) Sources of atmospheric methane: measurements in rice paddies and a discussion. J Geophys Res 86(C8):7203–7209
Covey KR, Wood SA, Warren RJ II, Lee X, Bradford MA (2012) Elevated methane concentrations in trees of an upland forest. Geophys Res Lett 39:L15705. doi:10.1029/2012GL052361
Dacey JWH, Klug MJ (1979) Methane efflux from lake sediments through water lilies. Science 203:1253–1255
De Simone O, Müller E, Junk WJ, Schmidt W (2002) Adaptations of Central Amazon tree species to prolonged flooding: root morphology and leaf longevity. Plant Biol 4:515–522
Garnet KN, Megonigal JP, Litchfield C, Taylor GE Jr (2005) Physiological control of leaf methane emission from wetland plants. Aquat Bot 81:141–155
Gauci V, Gowing DJG, Hornibrook ERC, Davis JM, Dise NB (2010) Woody stem methane emission in mature wetland alder trees. Atmos Environ 44:2157–2160
Grosse W, Schröder P (1984) Oxygen supply of roots by gas transport in alder-trees. Z Naturforsch 39c:1186–1188
Hesslein RH (1976) An in situ sampler for close interval pore water studies. Limnol Oceanogr 21:912–914. doi:10.4319/lo.1976.21.6.0912
Holzapfel-Pschorn A, Seiler W (1986) Methane emission during a cultivation period from an Italian rice paddy. J Geophys Res 91(D11):11803–11814
Holzapfel-Pschorn A, Conrad R, Seiler W (1986) Effects of vegetation on the emission of methane from submerged paddy soil. Plant Soil 92:223–233
Hook DD (1984) Adaptations to flooding with fresh water. In: Kozlowski TT (ed) Flooding and plant growth. Academic Press, Orlando, pp 265–294
IUSS Working Group WRB (2006) World reference base for soil resources, 2nd edn. World Soil References Reports No. 103, FAO, Rome, p 128
Japan Meteorological Agency (2002) Mesh climatic data of Japan. Meteorological Business Support Center, Tokyo, CD-ROM
Joabsson A, Christensen TR, Wallén B (1999) Vascular plant controls on methane emissions from northern peatforming wetlands. Trends Ecol Evol 14:385–388
King JY, Reeburgh WS, Regli SK (1998) Methane emission and transport by arctic sedges in Alaska: results of a vegetation removal experiment. J Geophys Res 103(D22):29083–29092
Kreuzwieser J, Buchholz J, Rennenberg H (2003) Emission of methane and nitrous oxide by Australian mangrove ecosystems. Plant Biol 5:423–431
Machacova K, Papen H, Kreuzwieser J, Rennenberg H (2013) Inundation strongly stimulates nitrous oxide emissions from stems of the upland tree Fagus sylvatica and the riparian tree Alnus glutinosa. Plant Soil 364:287–301
Matthews E, Fung I (1987) Methane emission from natural wetlands: global distribution, area, and environmental characteristics of sources. Glob Biogeochem Cycles 1:61–86
Nouchi I, Mariko S, Aoki K (1990) Mechanism of methane transport from the rhizosphere to the atmosphere through rice plants. Plant Physiol 94:59–66
Pangala SR, Moore S, Hornibrook ERC, Gauci V (2013) Trees are major conduits for methane egress from tropical forested wetlands. New Phytol 197:524–531
Pangala SR, Gowing DJ, Hornibrook ERC, Gauci V (2014) Controls on methane emissions from Alnus glutinosa saplings. New Phytol 201:887–896
Pulliam WM (1992) Methane emissions from cypress knees in a southeastern floodplain swamp. Oecologia 91:126–128
Purvaja R, Ramesh R, Frenzel P (2004) Plant-mediated methane emission from an Indian mangrove. Glob Change Biol 10:1825–1834
R Core Team (2014) R: a language and environment for statistical computing. R Foundation for statistical Computing, Vienna. http://www.R-project.org/
Rice AL, Butenhoff CL, Shearer MJ, Teama D (2010) Emissions of anaerobically produced methane by trees. Geophys Res Lett 37:L03807. doi:10.1029/2009GL041565
Rusch H, Rennenberg H (1998) Black alder (Alnus glutinosa (L.) Gaertn.) trees mediate methane and nitrous oxide emission from the soil to the atmosphere. Plant Soil 201:1–7
Schütz H, Seiler W, Conrad R (1989) Processes involved in formation and emission of methane in rice paddies. Biogeochemistry 7:33–53
Schütz H, Schröder P, Rennenberg H (1991) Role of plants in regulating the methane flux to the atmosphere. In: Sharkey TD, Holland EA, Mooney HA (eds) Trace gas emissions by plants. Academic Press, San Diego, pp 29–63
Sebacher DI, Harriss RC, Bartlett KB (1985) Methane emissions to the atmosphere through aquatic plants. J Environ Qual 14:40–46
Seiler W, Holzapfel-Pschorn A, Conrad R, Scharffe D (1984) Methane emission from rice paddies. J Atmos Chem 1:241–268
Shannon RD, White JR (1994) A three-year study of controls on methane emissions from two Michigan peatlands. Biogeochemistry 27:35–60
Shannon RD, White JR, Lawson JE, Gilmour BS (1996) Methane efflux from emergent vegetation in peatlands. J Ecol 84:239–246
Sun X, Song C, Guo Y, Wang X, Yang G, Li Y, Mao R, Lu Y (2012) Effect of plants on methane emissions from a temperate marsh in different seasons. Atmos Environ 60:277–282
Terazawa K, Ishizuka S, Sakata T, Yamada K, Takahashi M (2007) Methane emissions from stems of Fraxinus mandshurica var. japonica trees in a floodplain forest. Soil Biol Biochem 39:2689–2692
Treat CC, Bubier JL, Varner RK, Crill PM (2007) Timescale dependence of environmental and plant-mediated controls on CH4 flux in a temperate fen. J Geophys Res 112:G01014. doi:10.1029/2006JG000210
Turetsky MR, Treat CC, Waldrop MP, Waddington JM, Harden JW, McGuire AD (2008) Short-term response of methane fluxes and methanogen activity to water table and soil warming manipulations in an Alaskan peatland. J Geophys Res 113:G00A10. doi:10.1029/2007JG000496
van der Nat F-JWA, Middelburg JJ, Van Meteren D, Wielemakers A (1998) Diel methane emission patterns from Scirpus lacustris and Phragmites australis. Biogeochemistry 41:1–22
Vann CD, Megonigal JP (2003) Elevated CO2 and water depth regulation of methane emissions: comparison of woody and non-woody wetland plant species. Biogeochemistry 63:117–134
Waddington JM, Roulet NT, Swanson RV (1996) Water table control of CH4 emission enhancement by vascular plants in boreal peatlands. J Geophys Res 101(D17):22775–22785
Whiting GJ, Chanton JP (1992) Plant-dependent CH4 emission in a subarctic Canadian fen. Glob Biogeochem Cycles 6:225–231
Whiting GJ, Chanton JP (1996) Control of the diurnal pattern of methane emission from emergent aquatic macrophytes by gas transport mechanisms. Aquat Bot 54:237–253
WMO (2013a) WMO WDCGG data summary, WDCGG no.37, GAW data Volume IV—greenhouse gases and other atmospheric gases. Japan Meteorological Agency, Tokyo
WMO (2013b) WMO greenhouse gas bulletin, the state of greenhouse gases in the atmosphere based on global observations through 2012. World Meteorological Organization, Geneva
Yamamoto S, Alcauskas JB, Crozier TE (1976) Solubility of methane in distilled water and seawater. J Chem Eng Data 21:78–80
Yamamoto F, Sakata T, Terazawa K (1995a) Physiological, morphological and anatomical responses of Fraxinus mandshurica seedlings to flooding. Tree Physiol 15:713–719
Yamamoto F, Sakata T, Terazawa K (1995b) Growth, morphology, stem anatomy, and ethylene production in flooded Alnus japonica seedlings. IAWA J 16:47–59
Zeikus JG, Ward JC (1974) Methane formation in living trees, a microbial origin. Science 184:1181–1183
Acknowledgments
This work was supported by the Japan Society for the Promotion of Science (JSPS) Grant-in-Aid for Scientific Research (B) (Grant No. 23380090). We thank the management office of “Domin-no-mori (people’s forest in Hokkaido)” and its local management staff of the Tsukigata Zone for their valuable support for our field measurement efforts. We also thank two anonymous reviewers for their constructive comments designed to improve the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Christopher Williams.
Rights and permissions
About this article
Cite this article
Terazawa, K., Yamada, K., Ohno, Y. et al. Spatial and temporal variability in methane emissions from tree stems of Fraxinus mandshurica in a cool-temperate floodplain forest. Biogeochemistry 123, 349–362 (2015). https://doi.org/10.1007/s10533-015-0070-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10533-015-0070-y