Abstract
Though biosecurity and best management practices (BMPs), vaccines, immunostimulants, probiotics and prebiotics are often used for health management in aquaculture, the use of chemotherapeutics/antibiotics often becomes the preferred method of choice once the infectious disease outbreak occurs. Phage-based control of bacterial pathogens (phage therapy) has recently re-emerged as an attractive therapeutic alternative due to the global emergence of antimicrobial-resistant bacterial pathogens. Target specificity with minimal disruption of natural microbiota, auto-dosing, safety, no production of toxic metabolites/residues and relatively inexpensive production are some of the distinct advantages of phages over antibiotics. In vivo, experimental studies have demonstrated the efficacy of phage therapy through immersion, oral, injection, topical application, and anal intubation routes against Aeromonas hydrophila, A. salmonicida, Edwardsiella tarda, Flavobacterium columnare, Pseudomonas aeruginosa, Streptococcus iniae, Vibrio anguillarum, V. harveyi, V. parahaemolyticus, etc. in aquaculture. Several factors such as phage selection, therapeutic dose, age of fish, specific targeting of the pathogen, disease condition of fish, environmental conditions, and administration route influence the efficacy of phage therapy in aquaculture. The application of a mixture of phages (phage cocktail) has also been suggested to overcome the narrow host range of phages and the development of phage resistance in bacteria. At present, a few commercial phage cocktail-based products are also available for control of Yersinia ruckeri, Aeromonas spp., Pseudomonas spp., and shrimp pathogenic vibrios in aquaculture. To successfully implement phage therapy, there is a need to develop region-specific/localized phage repositories with associated safety and efficacy data which could be used to quickly formulate effective phage cocktails after the identification of pathogenic bacterial strains in specific aquaculture areas.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Access to safe, nutritious, and affordable food for all people contributes immensely to the economic and political stability of the world. Food security is the most important step toward improving a country’s socio-economic condition and combating hunger. The global human population is increasing at the rate of 75 million people per year, and it is expected to reach 9.2 billion by 2050 (Bongaarts 2009). As underdeveloped countries are still dealing with food and nutritional security issues, human nutrition inadequacies place a premium on the inclusion of animal protein in one’s daily diet. The fisheries sector provides a large proportion of animal protein to people’s diets all over the world. The aquatic animals are highly nutritious, and relatively cheaper protein sources, which supplement the human diet by providing essential amino acids, lipids, vitamins, minerals, and micronutrients. Total fisheries and aquaculture production reached a record high of 214 million tons worth US$ 424 billion in 2020, comprising 178 million tons of aquatic animals and 36 million tons of algae (FAO 2022). Out of 178 million tons aquatic animals, 89% were for human consumption, whereas the remaining were for non-food uses. Though still a major contributor to overall fish production, global capture fisheries production has been almost stagnant over the last decade. As the wild capture fisheries stocks are overexploited and declining, the only option is to produce more and more food from aquaculture to supplement the nutritional deficiencies of the masses in developing countries. With 87.5 million tons of aquatic animals worth about US$ 264.8 billion, the contribution of aquaculture to the total global fish production has rapidly increased from 7% in 1974 to 49.2% in 2020 (FAO 2022).
The increase in aquaculture production has not been without the challenges of serious disease outbreaks threatening the growth and sustainability of the fisheries sector. Frequent disease outbreaks are the major constraints for the growth of the aquaculture industry, which in turn impact the socio-economic development of many countries around the globe. Infectious diseases in aquaculture are often caused by bacteria, viruses, protists, helminths, fungi, and oomycetes. A big concern in aquaculture is disease outbreaks due to indigenous and non-indigenous pathogenic bacteria, particularly the multi-drug resistant ones. Globally, bacterial diseases account for > 20% of total disease outbreaks in the aquaculture sector (Karunasagar et al. 2003 ; Karunasagar and Ababouch 2012 ; Rodger 2016 ; Mishra et al. 2017). Frequent outbreaks of bacterial diseases not only lead to economic losses for the farmer but may also result in serious health consequences for the consumers, as many of these aquatic pathogens may also cause food-borne infections (Igbinosa 2016).
The need to develop strategies for the successful control of disease in aquaculture is constantly increasing, not only to improve animal welfare and increase production but also to benefit local, national, and global economies. Antibiotics are often the first port of call for the treatment of infectious diseases. However, continued use/misuse of antibiotics has resulted in the occurrence of antibiotic residues in fishery products and antimicrobial resistance in bacterial pathogens (Okocha et al. 2018). Therefore, we must be able to develop alternative therapeutic strategies for combating bacterial infections causing significant mortalities in aquaculture. The development of such techniques would benefit aquaculture, and improve food security across the globe. One such promising alternative is the use of bacterial viruses, known as bacteriophages (phages), which can infect and kill the host bacteria. Though control of bacterial pathogens by lytic phages (phage therapy) has been around for almost a century, the recent emergence of antibiotic-resistant bacteria has led to renewed interest in phage research (Lin et al. 2017). In this review, we have summarized the currently available disease prophylactic and therapeutic strategies while extensively focusing on the use of bacteriophages (phages) for the control of bacterial pathogens (phage therapy) in aquaculture. Besides highlighting the advantages of phage therapy, we have covered the current status of phage therapy in aquaculture from laboratory research, field trials and product commercialization perspectives. Finally, we have outlined the present challenges of phage therapy in aquaculture and its future prospects.
Health Management in Aquaculture
Several disease prophylactic and therapeutic strategies are used in aquaculture.
Biosecurity and Best Management Practices
As “prevention is better than cure” is the basic underlying concept of any health management strategy, the role of biosecurity measures in aquaculture becomes very important. Farm-level best management practices (BMPs) and biosecurity measures include maintenance of ideal culture conditions, regular use of disinfectants, strict quarantine protocols, usage of certified/disease-free seed, traffic control, sanitation of hands and equipment before and after use and in between the uses, usage of clean feed and appropriate disposal of carcasses (Assefa and Abunna 2018). These practices reduce the stress on animals, thereby making them less susceptible to diseases (Scarfe and Palić 2020).
Vaccines
Vaccination is used to boost the host’s immunity against a particular disease or a set of diseases. Vaccines operate by exposing an animal’s immune system to an “antigen” (a portion of a pathogen or the full pathogen) and giving it time to establish a defense and a “memory/booster” that will speed up this defense during subsequent infection by the same pathogen. Normally, vaccines are given to healthy animals before disease epidemics (Sommerset et al. 2005). In 1976, the first vaccine in fish was used against enteric red mouth disease. Several types of vaccines such as inactivated/killed vaccines, attenuated vaccines, recombinant vaccines, synthetic peptide vaccines, DNA vaccines, and mucosal vaccines have been tested against various fish pathogens (Mondal and Thomas 2022). At present, 26 licensed vaccines are commercially available against fish pathogens for use in aquaculture. Most of these vaccines are formalin-inactivated vaccines made from the whole cell of pathogenic strains (Ma et al. 2019). The development of vaccines necessitates a thorough knowledge of the host, pathogen, and their interactions. Additionally, extensive financial and scientific efforts are needed for vaccine development (Sommerset et al. 2005 ; Collins et al. 2019). As reviewed previously, several factors like stress, age, sex, pre-existing diseases, degree of immune response, types of adjuvants, routes of administration, and dose can also influence the vaccine efficacy (Khati and Anita 2015 ; Shirajum Monir et al. 2020).
Immunostimulants
Immunostimulants are also regularly used in fish and shellfish farming. Immunostimulants could be prepared from a variety of biological and synthetic compounds. Immunostimulants activate and amplify the innate and/or specific immune response by directly interacting with and stimulating the immune system cells directly, thereby boosting the body’s natural defensive system and resistance to disease. They primarily enhance the activity of phagocytes. There is no production of memory components, and the immunological reaction lasts for a short time only (Wang et al. 2017a, 2017b). Compounds like peptidoglycan, chitin, chitosan, levamisole, lentinan, schizophyllan, muramyl dipeptide, oligosaccharides and glucan, vitamin, and yeast combinations as well as various animal and plant-derived products have been used as immunostimulants for the disease prevention. In fish and shrimp farming, immunostimulants have been used to successfully control pathogenic bacteria such as Aeromonas salmonicida, A. hydrophila, Vibrio anguillarum, V. vulnificus, V. salmonicida, Yersinia ruckeri, and Streptococcus spp., as well as the viruses that cause infectious diseases like hematopoietic necrosis, yellow head virus, and viral hemorrhage. Immunostimulants typically improve certain aspects of the non-specific immune response; although, this does not always result in a higher survival rate. Additionally, immunostimulants, administered in excessive doses or for an extended period, may have immunosuppressive effects (Mohan et al. 2019).
Probiotics and Prebiotics
Probiotics are live microbial feed supplements, which are used as biological disease control agents in aquaculture. After administration, these non-pathogenic microbes multiply, support the growth of natural microbiota and maintain the microbial equilibrium in the hosts (Martínez Cruz et al. 2012). Besides, probiotics also compete with pathogens for space and nutrients, resulting in the competitive exclusion of pathogens. Recognition of conserved microbe-associated molecular patterns (MAMPs) of probiotic bacteria by the host cells also stimulates the immune response in terms of respiratory burst, phagocytic activity, complement activity, cytokine production, serum lysozyme activity, serum protein content, and antibody production. Besides, probiotics also produce several beneficial compounds which either have antimicrobial properties or help in maintaining intestinal and immune homeostasis (Hoseinifar et al. 2018). Besides the commonly used genera Lactobacillus and Bacillus, bacteria belonging to genera Enterococcus, Bifidobacterium, Alteromonas, Flavobacterium, Pseudomonas, Clostridium, Streptomyces, and Cytophaga are also used as probiotics in the aquaculture (Sahu et al. 2008). The immunomodulatory effect of probiotics depends on various factors like source, type, dose, and duration of supplementation of probiotics (Nayak 2010).
Prebiotics are non-digestible food ingredients that beneficially affect the host by selectively stimulating the growth and/or activity of one or a limited number of bacteria (Gibson and Roberfroid 1995). Prebiotics are selectively fermented by Bifidobacteria, Lactobacillus, and Bacteroides. However, there is no universal prebiotic which can be fermented by all the members of gut microbiota. The ability of a bacterium to ferment specific prebiotic depends on the presence of required enzymes (Wee et al. 2022). Some of the prebiotics widely used in animal feeds include inulin and oligofructose, trans-galactooligosaccharides (TOS), lactulose, isomaltose oligosaccharides (IMO), lactosucrose, xylo-oligosaccharides (XOS), soyabean oligosaccharides, and gluco-oligosaccharides (GOS). Prebiotics have been reported to have numerous beneficial effects in fish, such as increased disease resistance (Mohammadi et al. 2022) and improved nutrient availability (Lu et al. 2019). Often probiotics and prebiotics are administered together as synbiotics to enhance their potential benefits for the host (Huynh et al. 2017).
Antibiotics
Antibiotics have been important medicines for the safety and well-being of humans and animals. Antibiotics are often used in food animals and aquaculture, and their application can be categorized as therapeutic, prophylactic, or metaphylactic. Besides, antibiotics are often used as growth-promoting agents during the production of food animals. Due to a lack of proper surveillance and monitoring systems, the exact amount of antimicrobials used worldwide is very difficult to estimate. However, estimates of antimicrobial usage in aquaculture are available. In the year 2017, 10,259 t (3163–44,727 t at 95% uncertainty interval) of antimicrobials were estimated to be used in the aquaculture sector across the globe. Majority (93.8%) of these antimicrobials were used in Asia-Pacific region with China (57.9%), India (11.3%), Indonesia (8.6%), and Vietnam (5%) accounting for the largest share. Besides, global antimicrobial consumption was estimated to increase by 33% to 13,600 tons (4193–59,295 t at 95% uncertainty interval) by the year 2030 with China (55.9%), India (11.3%), Indonesia (10.1%), and Vietnam (5.2%) again expected to emerge the largest consumers (Schar et al. 2020).
Indiscriminate use of antibiotics, without following the recommended dosage regimes and withdrawal periods, has accelerated the global emergence of antimicrobial resistance (AMR) in bacterial pathogens. The presence of AMR pathogens in various aquaculture systems has been reported across the globe (Tyagi et al. 2019 ; Reverter et al. 2020 ; Schar et al. 2021). A total of 511 research studies from 61 countries have investigated the antibiotics usage and AMR bacteria in various aquaculture environments from 1996 to 2021(Caputo et al. 2023). A systematic review of research studies conducted between the years 2000 and 2018 in Asia revealed that 33% of tested antimicrobial compounds had >50% resistance rates. Almost half of these studies were conducted on Vibrio spp. and Aeromonas spp. (Schar et al. 2021). These studies indicate the rising trends of AMR in aquaculture.
Bacteriophages
Bacteriophages, also known as phages, are viruses that infect bacteria. About 1031 numbers of phages, belonging to 106 different species, are the most prevalent biological organisms in the biosphere. By eliminating nearly half of the bacterial population once every 48 hours, phages play a significant part in bacterial control in nature (Brussow and Hendrix 2002). The high selectivity/specificity of phages permits the targeting of specific bacteria, without affecting the desirable bacterial flora. Depending upon the life cycle, phages can be categorized as either lytic or lysogenic. After entry into the host cell, the genome of lytic phage quickly takes over the bacterial machinery to rapidly synthesize phage components. This is followed by the assembly of mature phage progenies, lysis of host bacterial cell and subsequent release of phage particles into the environment, which can go on to infect other bacterial cells. On the other hand, the genome of lysogenic or temperate phages either integrates into a host cellular replicon, such as the chromosome, a plasmid, or another phage genome or exists as a self-replicating plasmid. At this stage, the phage genome is termed a prophage. It replicates along with the host and may remain in the lysogenic life cycle for thousands of generations (Williamson et al. 2001; Clokie et al. 2011). Though discovered almost a century back, lytic phages have now emerged as attractive therapeutic alternatives due to better understanding of phage molecular biology, and due to the rapid emergence of antibiotic resistance bacteria.
Several excellent articles and reviews cover general phage properties, biology, life cycle, morphology, and taxonomy (Brussow and Hendrix 2002 ; Nakai and Park 2002 ; Novik et al. 2017 ; Ofir and Sorek 2018 ; Kortright et al. 2019 ; Ramos-Vivas et al. 2021). To avoid redundancy, these aspects have not been covered in the present review.
Advantages of Phages over Antibiotics
Application of phage therapy has several distinct advantages over antibiotics (Fig. 1).
Specificity and Minimum Disruption of Natural Microbiota
As phages are host specific with activity against particular bacterial species, phage therapy can be applied directly to control the targeted pathogenic bacteria. Rarely, does the phage have the capacity to infect more than one closely related bacterial genus; whereas, antibiotic treatment creates collateral damage as it disrupts the microbiome (Yoon and Yoon 2018). Phage therapy is free from the short-term and long-term consequences of antibiotic-associated modification of the microbiota.
Auto-dosing and Self-limitation
As lytic phages use the host for their propagation, they increase in number when host bacteria are present. During therapeutic phage application, the presence of pathogenic host bacterium creates a unique auto-dosing effect. Due to auto-dosing, low-phage doses can be applied. Reduction of host bacterium during treatment also limits the phage propagation resulting in self-limitation of the phage population (Loc-Carrillo and Abedon 2011).
Safety
Phage are significantly safer and better tolerated, as they replicate only in the target bacterium, and cannot infect mammalian cells (Kaźmierczak and Dąbrowska 2018). However, phages may also sometimes carry potentially virulence-related or antibiotic resistance genes resulting in the phage-mediated transformation of non-pathogenic bacteria to potentially pathogenic ones. Besides, some lysogenic phages also contain genes for the integration of the phage genome into the host bacterial genome. Thus, genomes of the candidate phages should be thoroughly investigated for the presence of any harmful gene before therapeutic applications (Goodridge and Bisha 2011). With the arrival of next-generation sequencing and computational tools, it has now become possible to sequence, assemble and annotate large numbers of phage genomes at rapidly reducing costs.
No Production of Toxic Metabolites or Environmental Residues
As the phages are made up of only proteins and nucleic acids, their eventual breakdown products consist exclusively of amino acids and nucleic acids. On the other hand, toxic or carcinogenic metabolites might be produced from antibiotics. Thus, the presence of antibiotics and their metabolites in food animals and the environment is a big concern (Hassan et al. 2013).
Antibiofilm Activity
Compared to antibiotics, phages have a greater ability to destroy biofilms (Gutierrez et al. 2016). Biofilms have open structures with water-filled channels that facilitate phage access inside the biofilm. Besides, certain phages are capable of producing polysaccharide depolymerases that disrupt the biofilms (Vukotic et al. 2020)..
Reduced Potential for the Development of Resistance
Compared to antibiotics, the development of phage resistance by bacteria is less recurrent. Resistance against phages develops about ten times slower than antibiotic resistance (Schmelcher and Loessner 2014). Moreover, phages can evolve and thereby overcome bacterial resistance. Selection will favor mutant phages that are capable of infecting resistant bacteria. Besides, resistance could also cost bacteria their fitness, thereby weakening their ability to compete with their phage-sensitive counterparts (Bohannan and Lenski 2000 ; Koskella and Brockhurst 2014). Phage resistance can also impact virulence, as the receptors used by phages to attack bacteria might be virulence determinants as well (Levin and Bull 2004).
Inexpensive to Produce with Rapid Commercialization Potential
In contrast to antibiotics, phages can be easily produced on a large scale with a low cost of commercialization (Loc-Carrillo and Abedon 2011). There is a huge diversity of phages in nature. Wherever there is a host bacterium, a phage can be expected. In contrast to synthetic antibiotics, which are screened and discovered from the library of chemically altered compounds, the natural phage diversity in nature allows quick selection and characterization of pathogen-specific candidate phages. As antibiotics and their metabolites may have potentially harmful side effects, strict safety and regulatory guidelines are in place for their development and commercialization. It takes an average of 13–14 years with an investment of > 100–500 million euros from antibiotics discovery to its commercialization (Miethke et al. 2021).
Application and Formulation Versatility
Similar to antibiotics, versatile phage formulations in the forms of creams, liquids, and microencapsulated dry powders can be developed. Once developed, these formulations can be administered topically, intravenously, orally or through nebulization/inhalation. Phages targeting different species or strains of bacteria can be combined in the form of a cocktail to broaden their antibacterial activity spectrum. Besides, phages can also be combined with antibiotics to enhance their activity spectrum (Loc-Carrillo and Abedon 2011).
Phage Therapy in Aquaculture
The high incidences of disease in aquaculture, combined with concerns of antibiotic resistance and antibiotic residues, has led to a large amount of research into the potential of using phage therapy to control bacterial pathogens in aquaculture. Aquaculture farms are essentially artificial microcosms of the aquatic environment, which is naturally teaming with phages. Though conditions that are conducive to/for the growth of aquaculture species and their bacterial pathogens also appear to be conducive to the survival of phages, environmental variables may affect the therapeutic efficacy of phages in aquaculture farms. In a controlled experimental study, the variation of physicochemical parameters (pH, temperature, salinity, and organic matter) within the naturally expected range in aquaculture farms did not significantly affect phage survival. However, salinity and organic matter significantly affected the therapeutic efficacy of phage (Silva et al. 2014a, 2014b). Besides, outdoor aquaculture facilities are exposed to solar radiation which can cause decay in phage viability (Wommack et al. 1996 ; Gomes et al. 2022).
The first report of phage-based biocontrol of fish pathogens, dating back to 1981, demonstrated the efficacy of bacteriophage AH1 against A. hydrophila infection in Japanese weather loach (Misgurnus anguillicaudatus) under laboratory conditions (Wu et al. 1981). Another landmark study, leading to the development of field-level experiments on phage therapy, demonstrated the protective efficiency of phages against Lactococcus garvieae infection in yellowtail (Seriola quinqueradiata) (Nakai et al. 1999). These researchers reported that the survival rate was much higher in the yellowtail that received intraperitoneal injection of the phage after intraperitoneal challenge with L. garvieae, compared with that of control infected fish without phage injection (Nakai et al. 1999). Richards (2014) reviewed the the success of phage therapy against other fish pathogenic bacteria such as A. salmonicida, Edwardsiella tarda, Flavobacterium columnare, Pseudomonas aeruginosa, Streptococcus iniae, V. harveyi, V. parahaemolyticus, etc. (Richards 2014).
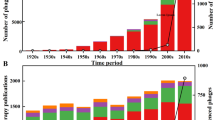
Even after proper characterization and selection of therapeutic phages, the route of administration remains a major challenge for phage therapy in aquaculture. In the scientific literature, various phage administration methods in aquaculture have been reported: immersion, feed incorporation, intramuscular or intraperitoneal injection administration, anal intubation, topical application and direct release of phages in the culture system. Whatever method is used, the phage particles must make contact with the bacterial host, whether in water, on the surface, or inside fish (Culot et al. 2019 ; Ramos-Vivas et al. 2021). Furthermore, the combined application of two or more phages (phage cocktails) is opening new therapeutic avenues. Besides, applications of phages with other therapeutics such as antibiotics and lysozymes have also been reported (Ryan et al. 2012 ; Choudhury et al. 2019). Table 1 provides the comprehensive status of phage therapy in aquaculture from the initial landmark studies to the most recent ones. Details of the pathogen, farmed species, phages, mode of applications and significant outcomes of field trials have also been covered.
Challenges of Phage Therapy
Presence of Lysogeny, Virulence-Related, and Antibiotic Resistance-Related Genes in Phage Genomes
Sometimes, phage may also carry potentially virulent or antibiotic resistance genes resulting in the phage-mediated transformation of non-pathogenic bacteria to potentially pathogenic ones. Besides, some lysogenic phages also contain genes for the integration of their phage genome into the host bacterial genome. Thus, phage genomes should be thoroughly investigated for the presence of any harmful gene before their applications for phage therapy (Goodridge and Bisha 2011).
Development of Phage Resistance in Bacteria
As in the case of antibiotics, bacteria can develop resistance to phage, which may hamper the effectiveness of phage therapy. The first step of phage infection to bacteria is the adhesion of phage on the bacterial surface by binding to specific receptors. If the bacteria loose these specific receptors, they become resistant to phage. Bacteria may also horizontally acquire a restriction-modification system that degrades the nucleic acid of an injected phage. In addition, phage resistance may also be caused by a mutation in a gene, the product of which is essential for phage replication or assembly. The development of phage cocktails (a mixture of two or more phages) has been suggested to overcome these challenges (Carlton 1999).
Specificity and Narrow Host Range
Phage specifically infects the host bacterium. Though specificity is an important characteristic of phage therapy as it allows the targeting of specific bacterial pathogens without affecting the normal beneficial microbiota of the host, the highly specific nature of phage with a very narrow host range could be an issue during field applications. Before the application of phage therapy, bacteria from the infection site should be tested for their sensitivity against the phage under simulated experimental conditions. In nature, polyvalent phages with activity against several strains of the same or different species do occur, and these can be used to increase the host range spectrum during phage therapy applications. Moreover, the phage cocktail approach can also be used to overcome the host range limitations of phages. However, the desire to increase coverage by adding more members to a phage cocktail must be balanced with the challenge of producing and testing well-defined multi-component mixtures for regulatory approval (Lu and Koeris 2011).
Effect of Environmental Conditions
Several physicochemical factors (pH, temperature, ionic, and bile salt concentrations) may have a significant effect on the survival and persistence of the phages. Some phages can be stored for a long period at neutral pH (6 to 8) in solution or dried form (Jończyk et al. 2011). Phage titers generally decrease slowly with pH. The proliferation of several phages is limited when the pH is lower than 4.5. One of the solutions to protect the bacteriophage at the site of infection and during its journey to the site is microencapsulation. Microencapsulation has been applied to enhance the viability of phage bacteria during processing and passage through the gastrointestinal tract (Ma et al. 2008 ; Colom et al. 2017). When applied directly in water, UV radiations can also have a detrimental effect on the phage viability (Sinton et al. 2002).
Ability of Phage to Reach the Target Bacteria
Depending upon the environmental conditions, route of administration, as well as the structure and composition of the target matrix, the ability of phage to reach the pathogenic bacteria in various tissues during systematic infection might be significantly affected. Oral feed-based administration of phage is a preferred approach in aquaculture as other routes such as immersion, intubation, injection, and topical application are almost impractical to implement in large-scale aquaculture settings (Dang et al. 2021). Though one may think that orally administered phages cannot tolerate the harsh acidic conditions of the fish gut, recent studies have shown that orally administered phages not only survive in the fish gut but also cross the intestinal barrier to reach the kidney, spleen, and other organs of fish. In juvenile rainbow trout, orally administered Flavobacterium psychrophilum phage FpV-9 could be continuously detected in the intestine. Besides, phages were also found in 40–50% of the sampled spleen and kidney tissues (Christiansen et al. 2014). In another study, continuous administration of Edwarsiella tarda phage ETP-1, bioencapsulated in Artemia nauplii, over 10 days led to constant detection of phage in the gut, kidney, liver, and spleen of zebrafish from the first day onwards (Nikapitiya et al. 2020). Donati and colleagues reported the presence of orally administered F. psychrophilum phages in all the intestine and kidney samples of rainbow trout. Besides, phages could also be detected in 20% of spleen and 40% of brain samples. In contrast to intraperitoneally injected phages, which significantly protected fish against F. psychrophilum infection, orally administered phages did not offer significant protection. The authors suggested that a higher phage dose during oral administration might be necessary to protect the fish against bacterial infection (Donati et al. 2021). To develop effective phage formulations for aquaculture, each candidate phages must be specifically evaluated for its survival and distribution in the aquaculture environment/fish organs.
Rapid Clearance of Phages from Circulation and Development of Antiphage Antibodies
One of the biggest drawbacks of phage studies is the lack of any supporting pharmacokinetic data. Being a foreign object, the phage can be rapidly cleared from the circulatory system of the animal being treated (Carlton 1999). One approach to compensate for this rapid clearance is repeated administration of phages during the treatment period. Besides, the administration of phage may result in immune system activation and the development of antiphage antibodies. The production of antiphage antibodies might not be an issue during active infection. However, during chronic infection, these antiphage antibodies may neutralize a proportion of administered phages resulting in a decreased efficiency of treatment. Though not much information regarding these antiphage antibodies is available in fish, recent studies in humans have suggested that the effect of neutralizing antiphage antibodies is not significant during phage therapy (Zaczek et al. 2016). Moreover, it has also been suggested that a higher dose of phages could be administered to compensate for the activity loss due to neutralizing antiphage antibodies (Carlton 1999).
Commercial Phage Products for Use in Aquaculture
The development of commercial phage products for aquaculture use is an active area of research. Phage products for aquaculture typically involve isolating and selecting phages that are effective against specific bacterial pathogens. These phages undergo testing to assess their efficacy, safety, and stability before being formulated into commercial products. These commercial products provide a targeted and environmentally friendly approach to disease management by specifically targeting harmful bacteria while leaving beneficial bacteria unharmed. The regulatory landscape for phage products in aquaculture varies across different countries, and it can influence the availability and commercialization of phage products in particular regions. Table 2 provides an overview of phage products currently available or under development for use in aquaculture.
Conclusions
Alternative approaches to fight infectious diseases are gaining ground in the context of antibiotic resistance in bacteria and problems with drug residues in aquatic food animals. Lytic phages are the potential alternatives to antibiotics for the control of bacterial infections in aquaculture systems. Most of the studies reviewed in this paper demonstrated the efficacy of phages for controlling bacterial pathogens in various aquatic organisms. These studies also provide a positive outlook on the future benefits of phage therapy to treat aquaculture diseases. However, almost all of these studies have been carried out under controlled laboratory conditions. To better understand the effectiveness of phage therapies, research trials must be carried out under conditions that mimic the actual situations in field aquaculture settings. Before applying phage therapy against pathogenic bacteria, one must confirm the lytic activity of phages against prevalent strains of a pathogen in a particular aquaculture area. Rather than developing a universal phage formulation/cocktail to control a particular bacterium across the globe, the focus should be on developing region-specific/localized phage repositories with associated safety data. These phage repositories could be used to quickly formulate effective phage cocktails after the identification of pathogenic bacterial strains in specific aquaculture areas. The combined efficacy of phage therapy with probiotics could also be evaluated. In this approach, probiotics can promote healthy microbiota, whereas phages can selectively target specific pathogens. There is also a need to establish appropriate regulatory standards to ensure the safety and performance of commercial phage products for use in aquaculture. Additionally, it is also necessary to generate awareness about the advantages of phage-based products to increase their consumer acceptance and demand resulting in further research and development in this area.
Data Availability
This study does not have any associated data.
References
Assefa A, Abunna F (2018) Maintenance of fish health in aquaculture: review of epidemiological approaches for prevention and control of infectious disease of fish. Vet Med Int 2018:5432497. https://doi.org/10.1155/2018/5432497
Benala M, Vaiyapuri M, Sivam V, Raveendran K, Mothadaka MP, Badireddy MR (2023) Genome characterization and infectivity potential of vibriophage LV6 with lytic activity against luminescent vibrios of Penaeus vannamei shrimp aquaculture. Viruses 15:868
Bohannan BJM, Lenski RE (2000) Linking genetic change to community evolution: insights from studies of bacteria and bacteriophage. Ecol Lett 3:362–377. https://doi.org/10.1046/j.1461-0248.2000.00161.x
Bongaarts J (2009) Human population growth and the demographic transition. Philos Trans R Soc Lond B Biol Sci 364:2985–2990. https://doi.org/10.1098/rstb.2009.0137
Brussow H, Hendrix RW (2002) Phage genomics: small is beautiful. Cell 108:13–16. https://doi.org/10.1016/s0092-8674(01)00637-7
Cao Y, Li S, Han S, Wang D, Zhao J, Xu L, Liu H, Lu T (2020) Characterization and application of a novel Aeromonas bacteriophage as treatment for pathogenic Aeromonas hydrophila infection in rainbow trout. Aquaculture 523:735193. https://doi.org/10.1016/j.aquaculture.2020.735193
Caputo A, Bondad-Reantaso MG, Karunasagar I, Hao B, Gaunt P, Verner-Jeffreys D, Fridman S, Dorado-Garcia A (2023) Antimicrobial resistance in aquaculture: A global analysis of literature and national action plans. Rev Aquac 15:568–578. https://doi.org/10.1111/raq.12741
Carlton RM (1999) Phage therapy: past history and future prospects. Arch Immunol Ther Exp (Warsz) 47:267–274
Castillo D, Higuera G, Villa M, Middelboe M, Dalsgaard I, Madsen L, Espejo RT (2012) Diversity of Flavobacterium psychrophilum and the potential use of its phages for protection against bacterial cold water disease in salmonids. J Fish Dis 35:193–201. https://doi.org/10.1111/j.1365-2761.2011.01336.x
Chen L, Fan J, Yan T, Liu Q, Yuan S, Zhang H, Yang J, Deng D, Huang S, Ma Y (2019) Isolation and characterization of specific phages to prepare a cocktail preventing Vibrio sp. Va-F3 infections in shrimp (Litopenaeus vannamei). Front Microbiol 10:2337. https://doi.org/10.3389/fmicb.2019.02337
Choudhury TG, Maiti B, Venugopal MN, Karunasagar I (2019) Influence of some environmental variables and addition of r-lysozyme on efficacy of Vibrio harveyi phage for therapy. J Biosci 44:8. https://doi.org/10.1007/s12038-018-9830-x
Christiansen RH, Dalsgaard I, Middelboe M, Lauritsen AH, Madsen L (2014) Detection and quantification of Flavobacterium psychrophilum-specific bacteriophages in vivo in rainbow trout upon oral administration: implications for disease control in aquaculture. Appl Environ Microbiol 80:7683–7693. https://doi.org/10.1128/AEM.02386-14
Clokie MRJ, Millard AD, Letarov AV, Heaphy S (2011) Phages in nature. Bacteriophage 1:31–45. https://doi.org/10.4161/bact.1.1.14942
Collins C, Lorenzen N, Collet B (2019) DNA vaccination for finfish aquaculture. Fish Shellfish Immunol 85:106–125. https://doi.org/10.1016/j.fsi.2018.07.012
Colom J, Cano-Sarabia M, Otero J, Arinez-Soriano J, Cortes P, Maspoch D, Llagostera M (2017) Microencapsulation with alginate/CaCO3: a strategy for improved phage therapy. Sci Rep 7:41441. https://doi.org/10.1038/srep41441
Culot A, Grosset N, Gautier M (2019) Overcoming the challenges of phage therapy for industrial aquaculture: a review. Aquaculture 513:734423. https://doi.org/10.1016/j.aquaculture.2019.734423
Dang THO, Xuan TTT, Duyen LTM, Le NP, Hoang HA (2021) Protective efficacy of phage PVN02 against haemorrhagic septicaemia in striped catfish Pangasianodon hypophthalmus via oral administration. J Fish Dis 44:1255–1263. https://doi.org/10.1111/jfd.13387
Dien LT, Ky LB, Huy BT, Mursalim MF, Kayansamruaj P, Senapin S, Rodkhum C, Dong HT (2022) Characterization and protective effects of lytic bacteriophage pAh6.2TG against a pathogenic multidrug-resistant Aeromonas hydrophila in Nile tilapia (Oreochromis niloticus). Transbound Emerg Dis 69:e435–e450. https://doi.org/10.1111/tbed.14321
Donati VL, Dalsgaard I, Sundell K, Castillo D, Er-Rafik M, Clark J, Wiklund T, Middelboe M, Madsen L (2021) Phage-mediated control of Flavobacterium psychrophilum in aquaculture: in vivo experiments to compare delivery methods. Front Microbiol 12:628309. https://doi.org/10.3389/fmicb.2021.628309
Droubogiannis S, Katharios P (2022) Genomic and biological profile of a novel bacteriophage, Vibrio phage virtus, which improves survival of sparus aurata larvae challenged with Vibrio harveyi. Pathogens 11:630. https://doi.org/10.3390/pathogens11060630
Easwaran M, Dananjaya SHS, Park SC, Lee J, Shin H-J, De Zoysa M (2017) Characterization of bacteriophage pAh-1 and its protective effects on experimental infection of Aeromonas hydrophila in Zebrafish (Danio rerio). J Fish Dis 40:841–846. https://doi.org/10.1111/jfd.12536
El-Araby DA, El-Didamony G, Megahed M (2016) New approach to use phage therapy against Aeromonas hydrophila induced motile Aeromonas septicemia in Nile tilapia. J Mar Sci Res Dev 6:2. https://doi.org/10.4172/2155-9910.1000194
FAO (2022) The State of World Fisheries and Aquaculture 2022: Towards Blue Transformation. FAO, Rome. https://doi.org/10.4060/cc0461en
Gibson GR, Roberfroid MB (1995) Dietary modulation of the human colonic microbiota: introducing the concept of prebiotics. J Nutr 125:1401–1412. https://doi.org/10.1093/jn/125.6.1401
Gomes M, Bartolomeu M, Vieira C, Gomes ATPC, Faustino MAF, Neves MGPMS, Almeida A (2022) Photoinactivation of phage phi6 as a sars-cov-2 model in wastewater: Evidence of efficacy and safety. Microorganisms 10:659. https://doi.org/10.3390/microorganisms10030659
Goodridge LD, Bisha B (2011) Phage-based biocontrol strategies to reduce foodborne pathogens in foods. Bacteriophage 1:130–137. https://doi.org/10.4161/bact.1.3.17629
Gutierrez D, Rodriguez-Rubio L, Martinez B, Rodriguez A, Garcia P (2016) Bacteriophages as weapons against bacterial biofilms in the food industry. Front Microbiol 7:825. https://doi.org/10.3389/fmicb.2016.00825
Hassan MN, Rahman M, Hossain MB, Hossain MM, Mendes R, Nowsad AAKM (2013) Monitoring the presence of chloramphenicol and nitrofuran metabolites in cultured prawn, shrimp and feed in the Southwest coastal region of Bangladesh. Egypt J Aquat Res 39:51–58. https://doi.org/10.1016/j.ejar.2013.04.004
Higuera G, Bastías R, Tsertsvadze G, Romero J, Espejo RT (2013) Recently discovered Vibrio anguillarum phages can protect against experimentally induced vibriosis in Atlantic salmon, Salmo salar. Aquaculture 392-395:128–133. https://doi.org/10.1016/j.aquaculture.2013.02.013
Hoseinifar SH, Sun Y-Z, Wang A, Zhou Z (2018) Probiotics as means of diseases control in aquaculture, a review of current knowledge and future perspectives. Front Microbiol 9:2429. https://doi.org/10.3389/fmicb.2018.02429
Huynh T-G, Shiu Y-L, Nguyen T-P, Truong Q-P, Chen J-C, Liu C-H (2017) Current applications, selection, and possible mechanisms of actions of synbiotics in improving the growth and health status in aquaculture: a review. Fish Shellfish Immunol 64:367–382. https://doi.org/10.1016/j.fsi.2017.03.035
Igbinosa EO (2016) Detection and antimicrobial resistance of Vibrio isolates in aquaculture environments: Implications for public health. Microb Drug Resist 22:238–245. https://doi.org/10.1089/mdr.2015.0169
Imbeault S, Parent S, Lagacé M, Uhland CF, Blais J-F (2006) Using bacteriophages to prevent furunculosis caused by aeromonas salmonicida in farmed brook trout. J Aquat Anim Health 18:203–214. https://doi.org/10.1577/H06-019.1
Jończyk E, Kłak M, Międzybrodzki R, Górski A (2011) The influence of external factors on bacteriophages—review. Folia Microbiol 56:191–200. https://doi.org/10.1007/s12223-011-0039-8
Jun JW, Han JE, Giri SS, Tang KFJ, Zhou X, Aranguren LF, Kim HJ, Yun S, Chi C, Kim SG, Park SC (2018) Phage application for the protection from acute hepatopancreatic necrosis disease (AHPND) in Penaeus vannamei. Indian J Microbiol 58:114–117. https://doi.org/10.1007/s12088-017-0694-9
Jun JW, Kim JH, Shin SP, Han JE, Chai JY, Park SC (2013) Protective effects of the Aeromonas phages pAh1-C and pAh6-C against mass mortality of the cyprinid loach (Misgurnus anguillicaudatus) caused by Aeromonas hydrophila. Aquaculture 416-417:289–295. https://doi.org/10.1016/j.aquaculture.2013.09.045
Kalatzis PG, Bastías R, Kokkari C, Katharios P (2016) Isolation and characterization of two lytic bacteriophages, φSt2 and φGrn1; phage therapy application for biological control of Vibrio alginolyticus in aquaculture live feeds. PloS One 11:e0151101. https://doi.org/10.1371/journal.pone.0151101
Karunasagar I, Ababouch L (2012) Shrimp viral diseases, import risk assessment and international trade. Indian J Virol 23:141–148. https://doi.org/10.1007/s13337-012-0081-4
Karunasagar I, Karunasagar I, Otta SK (2003) Disease problems affecting fish in tropical environments. J Appl Aquacult 13:231–249. https://doi.org/10.1300/J028v13n03_03
Karunasagar I, Shivu MM, Girisha SK, Krohne G, Karunasagar I (2007) Biocontrol of pathogens in shrimp hatcheries using bacteriophages. Aquaculture 268:288–292. https://doi.org/10.1016/j.aquaculture.2007.04.049
Kaźmierczak Z, Dąbrowska K (2018) Interaction of bacteriophages with mammalian cells. In: Azeredo J, Sillankorva S (eds) Bacteriophage therapy: from lab to clinical practice. Springer New York, New York, NY, pp 113–122
Khairnar K, Raut MP, Chandekar RH, Sanmukh SG, Paunikar WN (2013) Novel bacteriophage therapy for controlling metallo-beta-lactamase producing Pseudomonas aeruginosa infection in Catfish. BMC Vet Res 9:264. https://doi.org/10.1186/1746-6148-9-264
Khati A, Anita RRN (2015) Vaccines and their role in fish disease management-a review. Biochem Cell Arch 15:39–46
Kim JH, Choresca CH, Shin SP, Han JE, Jun JW, Park SC (2015) Biological control of Aeromonas salmonicida subsp. salmonicida infection in rainbow trout (Oncorhynchus mykiss) using Aeromonas phage Pas-1. Transbound Emerg Dis 62:81–86. https://doi.org/10.1111/tbed.12088
Kortright KE, Chan BK, Koff JL, Turner PE (2019) Phage therapy: a renewed approach to combat antibiotic-resistant bacteria. Cell Host Microbe 25:219–232. https://doi.org/10.1016/j.chom.2019.01.014
Koskella B, Brockhurst MA (2014) Bacteria–phage coevolution as a driver of ecological and evolutionary processes in microbial communities. FEMS Microbiol Rev 38:916–931. https://doi.org/10.1111/1574-6976.12072
Le TS, Nguyen TH, Vo HP, Doan VC, Nguyen HL, Tran MT, Tran TT, Southgate PC, Kurtböke Dİ (2018) Protective effects of bacteriophages against Aeromonas hydrophila causing Motile Aeromonas Septicemia (MAS) in striped catfish. Antibiotics 7:16. https://doi.org/10.3390/antibiotics7010016
Levin BR, Bull JJ (2004) Population and evolutionary dynamics of phage therapy. Nat Rev Microbiol 2:166–173. https://doi.org/10.1038/nrmicro822
Li Z, Li X, Zhang J, Wang X, Wang L, Cao Z, Xu Y (2016) Use of phages to control Vibrio splendidus infection in the juvenile sea cucumber Apostichopus japonicus. Fish Shellfish Immunol 54:302–311. https://doi.org/10.1016/j.fsi.2016.04.026
Lin DM, Koskella B, Lin HC (2017) Phage therapy: an alternative to antibiotics in the age of multi-drug resistance. World J Gastrointest Pharmacol Ther 8:162–173. https://doi.org/10.4292/wjgpt.v8.i3.162
Loc-Carrillo C, Abedon ST (2011) Pros and cons of phage therapy. Bacteriophage 1:111–114. https://doi.org/10.4161/bact.1.2.14590
Lomelí-Ortega CO, Martínez-Díaz SF (2014) Phage therapy against Vibrio parahaemolyticus infection in the whiteleg shrimp (Litopenaeus vannamei) larvae. Aquaculture 434:208–211. https://doi.org/10.1016/j.aquaculture.2014.08.018
Lu J, Bu X, Xiao S, Lin Z, Wang X, Jia Y, Wang X, Qin JG, Chen L (2019) Effect of single and combined immunostimulants on growth, anti-oxidation activity, non-specific immunity and resistance to Aeromonas hydrophila in Chinese mitten crab (Eriocheir sinensis). Fish Shellfish Immunol 93:732–742. https://doi.org/10.1016/j.fsi.2019.08.027
Lu TK, Koeris MS (2011) The next generation of bacteriophage therapy. Curr Opin Microbiol 14:524–531. https://doi.org/10.1016/j.mib.2011.07.028
Luo X, Liao G, Liu C, Jiang X, Lin M, Zhao C, Tao J, Huang Z (2018) Characterization of bacteriophage HN48 and its protective effects in Nile tilapia Oreochromis niloticus against Streptococcus agalactiae infections. J Fish Dis 41:1477–1484. https://doi.org/10.1111/jfd.12838
Ma J, Bruce TJ, Jones EM, Cain KD (2019) A review of fish vaccine development strategies: Conventional methods and modern biotechnological approaches. Microorganisms 7:569. https://doi.org/10.3390/microorganisms7110569
Ma Y, Pacan JC, Wang Q, Xu Y, Huang X, Korenevsky A, Sabour PM (2008) Microencapsulation of bacteriophage Felix O1 into chitosan-alginate microspheres for oral delivery. Appl Environ Microbiol 74:4799–4805. https://doi.org/10.1128/aem.00246-08
Madsen L, Bertelsen SK, Dalsgaard I, Middelboe M (2013) Dispersal and survival of Flavobacterium psychrophilum phages in vivo in rainbow trout and in vitro under laboratory conditions: Implications for their use in phage therapy. Appl Environ Microbiol 79:4853–4861. https://doi.org/10.1128/AEM.00509-13
Martínez Cruz P, Ibáñez AL, Monroy Hermosillo OA, Ramírez Saad HC (2012) Use of probiotics in aquaculture. ISRN Microbiol 2012:916845. https://doi.org/10.5402/2012/916845
Martínez-Díaz SF, Hipólito-Morales A (2013) Efficacy of phage therapy to prevent mortality during the vibriosis of brine shrimp. Aquaculture 400-401:120–124. https://doi.org/10.1016/j.aquaculture.2013.03.007
Matsuoka S, Hashizume T, Kanzaki H, Iwamoto E, Se Chang P, Yoshida T, Nakai T (2007) phage therapy against β-hemolytic streptococcicosis of Japanese flounder Paralichthys olivaceus. Fish Pathol 42:181–189. https://doi.org/10.3147/jsfp.42.181
Miethke M, Pieroni M, Weber T, Brönstrup M, Hammann P, Halby L, Arimondo PB, Glaser P, Aigle B, Bode HB, Moreira R, Li Y, Luzhetskyy A, Medema MH, Pernodet J-L, Stadler M, Tormo JR, Genilloud O, Truman AW et al (2021) Towards the sustainable discovery and development of new antibiotics. Nat Rev Chem 5:726–749. https://doi.org/10.1038/s41570-021-00313-1
Mishra SS, Rakesh D, Dhiman M, Choudhary P, Debbarma J, Sahoo SN, Barua A, Giri BS, Ramesh R, Ananda K, Mishra CK, Swain P (2017) Present status of fish disease management in freshwater aquaculture in India: state-of-the-art-review. HSOA J Aquac Fish 1:1–9. https://doi.org/10.24966/AAF-5523/100003
Mohammadi G, Hafezieh M, Karimi AA, Azra MN, Van Doan H, Tapingkae W, Abdelrahman HA, Dawood MAO (2022) The synergistic effects of plant polysaccharide and Pediococcus acidilactici as a synbiotic additive on growth, antioxidant status, immune response, and resistance of Nile tilapia (Oreochromis niloticus) against Aeromonas hydrophila. Fish Shellfish Immunol 120:304–313. https://doi.org/10.1016/j.fsi.2021.11.028
Mohan K, Ravichandran S, Muralisankar T, Uthayakumar V, Chandirasekar R, Seedevi P, Abirami RG, Rajan DK (2019) Application of marine-derived polysaccharides as immunostimulants in aquaculture: a review of current knowledge and further perspectives. Fish Shellfish Immunol 86:1177–1193. https://doi.org/10.1016/j.fsi.2018.12.072
Mondal H, Thomas J (2022) A review on the recent advances and application of vaccines against fish pathogens in aquaculture. Aquacult Int 30:1971–2000. https://doi.org/10.1007/s10499-022-00884-w
Nakai T, Park SC (2002) Bacteriophage therapy of infectious diseases in aquaculture. Res Microbiol 153:13–18. https://doi.org/10.1016/S0923-2508(01)01280-3
Nakai T, Sugimoto R, Park KH, Matsuoka S, Mori K, Nishioka T, Maruyama K (1999) Protective effects of bacteriophage on experimental Lactococcus garvieae infection in yellowtail. Dis Aquat Org 37:33–41. https://doi.org/10.3354/dao037033
Nayak SK (2010) Probiotics and immunity: A fish perspective. Fish Shellfish Immunol 29:2–14. https://doi.org/10.1016/j.fsi.2010.02.017
Nikapitiya C, Dananjaya SHS, Edirisinghe SL, Chandrarathna HPSU, Lee J, De Zoysa M (2020) Development of phage delivery by bioencapsulation of artemia nauplii with Edwardsiella tarda phage (ETP-1). Braz J Microbiol 51:2153–2162. https://doi.org/10.1007/s42770-020-00324-y
Novik GI, Ladutska AI, Rakhuba D (2017) Bacteriophage taxonomy and classification. In: Méndez-Vilas A (ed) Antimicrobial research: novel bioknowledge and educational programs. Formatex Research Center, Badajoz, pp 251–259
Ofir G, Sorek R (2018) Contemporary phage biology: from classic models to new insights. Cell 172:1260–1270. https://doi.org/10.1016/j.cell.2017.10.045
Okocha RC, Olatoye IO, Adedeji OB (2018) Food safety impacts of antimicrobial use and their residues in aquaculture. Public Health Rev 39:21. https://doi.org/10.1186/s40985-018-0099-2
Park SC, Nakai T (2003) Bacteriophage control of Pseudomonas plecoglossicida infection in ayu Plecoglossus altivelis. Dis Aquat Org 53:33–39. https://doi.org/10.3354/dao053033
Park SC, Shimamura I, Fukunaga M, Mori K-I, Nakai T (2000) Isolation of bacteriophages specific to a fish pathogen, Pseudomonas plecoglossicida, as a candidate for disease control. Appl Environ Microbiol 66:1416–1422. https://doi.org/10.1128/AEM.66.4.1416-1422.2000
Prasad Y, Arpana KD, Sharma AK (2011) Lytic bacteriophages specific to Flavobacterium columnare rescue catfish, Clarias batrachus (Linn.) from columnaris disease. J Environ Biol 32:161–168
Raghu Patil J, Desai SN, Roy P, Durgaiah M, Saravanan RS, Vipra A (2014) Simulated hatchery system to assess bacteriophage efficacy against Vibrio harveyi. Dis Aquat Organ 112:113–119. https://doi.org/10.3354/dao02806
Ramos-Vivas J, Superio J, Galindo-Villegas J, Acosta F (2021) Phage therapy as a focused management strategy in aquaculture. Int J Mol Sci 22:10436. https://doi.org/10.3390/ijms221910436
Ren H, Li Z, Xu Y, Wang L, Li X (2019) Protective effectiveness of feeding phage cocktails in controlling Vibrio parahaemolyticus infection of sea cucumber Apostichopus japonicus. Aquaculture 503:322–329. https://doi.org/10.1016/j.aquaculture.2019.01.006
Reverter M, Sarter S, Caruso D, Avarre J-C, Combe M, Pepey E, Pouyaud L, Vega-Heredía S, de Verdal H, Gozlan RE (2020) Aquaculture at the crossroads of global warming and antimicrobial resistance. Nat Commun 11:1870. https://doi.org/10.1038/s41467-020-15735-6
Richards GP (2014) Bacteriophage remediation of bacterial pathogens in aquaculture: a review of the technology. Bacteriophage 4:e975540. https://doi.org/10.4161/21597081.2014.975540
Rodger HD (2016) Fish disease causing economic impact in global aquaculture. In: Adams A (ed) Fish Vaccines. Springer Basel, Basel, pp 1–34
Rong R, Lin H, Wang J, Khan MN, Li M (2014) Reductions of Vibrio parahaemolyticus in oysters after bacteriophage application during depuration. Aquaculture 418:171–176. https://doi.org/10.1016/j.aquaculture.2013.09.028
Ryan EM, Alkawareek MY, Donnelly RF, Gilmore BF (2012) Synergistic phage-antibiotic combinations for the control of Escherichia coli biofilms in vitro. FEMS Immunol Med Microbiol 65:395–398. https://doi.org/10.1111/j.1574-695X.2012.00977.x
Sahu MK, Swarnakumar NS, Sivakumar K, Thangaradjou T, Kannan L (2008) Probiotics in aquaculture: Importance and future perspectives. Indian J Microbiol 48:299–308. https://doi.org/10.1007/s12088-008-0024-3
Scarfe AD, Palić D (2020) Aquaculture biosecurity: Practical approach to prevent, control, and eradicate diseases. In: Powell MD (ed) Kibenge FSB. Aquaculture Health Management, Academic Press, pp 75–116
Schar D, Klein EY, Laxminarayan R, Gilbert M, Van Boeckel TP (2020) Global trends in antimicrobial use in aquaculture. Sci Rep 10:21878. https://doi.org/10.1038/s41598-020-78849-3
Schar D, Zhao C, Wang Y, Larsson DGJ, Gilbert M, Van Boeckel TP (2021) Twenty-year trends in antimicrobial resistance from aquaculture and fisheries in Asia. Nat Commun 12:5384. https://doi.org/10.1038/s41467-021-25655-8
Schmelcher M, Loessner MJ (2014) Application of bacteriophages for detection of foodborne pathogens. Bacteriophage 4:e28137. https://doi.org/10.4161/bact.28137
Shirajum Monir M, Yusoff SM, Mohamad A, Ina-Salwany MY (2020) Vaccination of tilapia against motile aeromonas septicemia: a review. J Aquat Anim Health 32:65–76. https://doi.org/10.1002/aah.10099
Silva YJ, Costa L, Pereira C, Cunha Â, Calado R, Gomes NCM, Almeida A (2014a) Influence of environmental variables in the efficiency of phage therapy in aquaculture. Microb Biotechnol 7:401–413. https://doi.org/10.1111/1751-7915.12090
Silva YJ, Costa L, Pereira C, Mateus C, Cunha Â, Calado R, Gomes NCM, Pardo MA, Hernandez I, Almeida A (2014b) Phage therapy as an approach to prevent Vibrio anguillarum infections in fish larvae production. PloS One 9:e114197. https://doi.org/10.1371/journal.pone.0114197
Silva YJ, Moreirinha C, Pereira C, Costa L, Rocha RJM, Cunha Â, Gomes NCM, Calado R, Almeida A (2016) Biological control of Aeromonas salmonicida infection in juvenile Senegalese sole (Solea senegalensis) with Phage AS-A. Aquaculture 450:225–233. https://doi.org/10.1016/j.aquaculture.2015.07.025
Sinton LW, Hall CH, Lynch PA, Davies-Colley RJ (2002) Sunlight inactivation of fecal indicator bacteria and bacteriophages from waste stabilization pond effluent in fresh and saline waters. Appl Environ Microbiol 68:1122–1131. https://doi.org/10.1128/AEM.68.3.1122-1131.2002
Sommerset I, Krossøy B, Biering E, Frost P (2005) Vaccines for fish in aquaculture. Expert Rev Vaccines 4:89–101. https://doi.org/10.1586/14760584.4.1.89
Tyagi A, Singh B, Billekallu Thammegowda NK, Singh NK (2019) Shotgun metagenomics offers novel insights into taxonomic compositions, metabolic pathways and antibiotic resistance genes in fish gut microbiome. Arch Microbiol 201:295–303. https://doi.org/10.1007/s00203-018-1615-y
Vinod MG, Shivu MM, Umesha KR, Rajeeva BC, Krohne G, Karunasagar I, Karunasagar I (2006) Isolation of Vibrio harveyi bacteriophage with a potential for biocontrol of luminous vibriosis in hatchery environments. Aquaculture 255:117–124. https://doi.org/10.1016/j.aquaculture.2005.12.003
Vukotic G, Obradovic M, Novovic K, Di Luca M, Jovcic B, Fira D, Neve H, Kojic M, McAuliffe O (2020) Characterization, antibiofilm, and depolymerizing activity of two phages active on carbapenem-resistant Acinetobacter baumannii. Front Med (Lausanne) 7:426. https://doi.org/10.3389/fmed.2020.00426
Wang W, Sun J, Liu C, Xue Z (2017a) Application of immunostimulants in aquaculture: Current knowledge and future perspectives. Aquacult Res 48:1–23. https://doi.org/10.1111/are.13161
Wang Y, Barton M, Elliott L, Li X, Abraham S, O'Dea M, Munro J (2017b) Bacteriophage therapy for the control of Vibrio harveyi in greenlip abalone (Haliotis laevigata). Aquaculture 473:251–258. https://doi.org/10.1016/j.aquaculture.2017.01.003
Wee W, Abdul Hamid NK, Mat K, Khalif RIAR, Rusli ND, Rahman MM, Kabir MA, Wei LS (2022) The effects of mixed prebiotics in aquaculture: a review. Aquac Fisheries. https://doi.org/10.1016/j.aaf.2022.02.005
Williamson SJ, McLaughlin MR, Paul JH (2001) Interaction of the φHSIC virus with its host: lysogeny or pseudolysogeny? Appl Environ Microbiol 67:1682–1688. https://doi.org/10.1128/AEM.67.4.1682-1688.2001
Wommack KE, Hill RT, Muller TA, Colwell RR (1996) Effects of sunlight on bacteriophage viability and structure. Appl Environ Microbiol 62:1336–1341. https://doi.org/10.1128/aem.62.4.1336-1341.1996
Wu JL, Chao WJ (1982) Isolation and application of a new bacteriophage, ET-1, which infect Edwardsiella tarda, the pathogen of edwardsiellosis. Rep Fish Dis Res (Taiwan) 4:8–17
Wu J-L, Lin H-M, Jan L, Hsu Y-L, Chang L-H (1981) Biological control of fish bacterial pathogen, Aeromonas hydrophila, by bacteriophage AH 1. Fish Pathol 15:271–276. https://doi.org/10.3147/jsfp.15.271
Yoon MY, Yoon SS (2018) Disruption of the gut ecosystem by antibiotics. Yonsei Med J 59:4–12. https://doi.org/10.3349/ymj.2018.59.1.4
Zaczek M, Lusiak-Szelachowska M, Jonczyk-Matysiak E, Weber-Dabrowska B, Miedzybrodzki R, Owczarek B, Kopciuch A, Fortuna W, Rogoz P, Gorski A (2016) antibody production in response to staphylococcal MS-1 phage cocktail in patients undergoing phage therapy. Front Microbiol 7:1681. https://doi.org/10.3389/fmicb.2016.01681
Acknowledgements
The authors are grateful to the Dean (College of Fisheries, Guru Angad Dev Veterinary & Animal Sciences University, Ludhiana, India) for all the necessary support during this study.
Funding
This work is a part of the Board of Research in Nuclear Sciences (BRNS) Grant 55/14/02/2021-BRNS “Development of phage-based strategies for biocontrol of antibiotic resistant Aeromonas species in fishery products” to Anuj Tyagi.
Author information
Authors and Affiliations
Contributions
Sumeet Rai: investigation, data analysis, data curation, writing—original draft, visualization. Basmeet Kaur: investigation, data analysis, data curation, writing—original draft, visualization. Prabjeet Singh: methodology, writing—original draft, writing—review and editing. Avtar Singh: methodology, writing—original draft, writing—review and editing. Soottawat Benjakul: data curation, writing—review and editing, supervision. Vijay Kumar Reddy S.: writing—review and editing, funding acquisition. Vandan Nagar: writing—review and editing, funding acquisition. Anuj Tyagi: conceptualization, writing—original draft, writing—review and editing, funding acquisition, project administration.
Corresponding author
Ethics declarations
Ethics Approval
No ethical permission was required for the study.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Handling Editor: Brian Austin
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rai, S., Kaur, B., Singh, P. et al. Perspectives on phage therapy for health management in aquaculture. Aquacult Int 32, 1349–1393 (2024). https://doi.org/10.1007/s10499-023-01220-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-023-01220-6