Abstract
We have previously shown that the antifungal activity of human lactoferrin (hLf) against Candida albicans relies on its ability to induce cell death associated with apoptotic markers. To gain a deeper understanding of the mechanisms underlying hLf-induced apoptosis, we characterized this cell death process in the well-established Saccharomyces cerevisiae model. Our results indicate that hLf induces cell death in S. cerevisiae in a manner that requires energy and de novo protein synthesis. Cell death is associated with nuclear chromatin condensation, preservation of plasma membrane integrity, and is Yca1p metacaspase-dependent. Lactoferrin also caused mitochondrial dysfunction associated with ROS accumulation and release of cytochrome c. Pre-incubation with oligomycin, an oxidative phosphorylation inhibitor, increased resistance to hLf and, accordingly, mutants deficient in the F1F0-ATP synthase complex were more resistant to death induced by hLf. This indicates that mitochondrial energetic metabolism plays a key role in the killing effect of hLf, though a direct role of F1F0-ATP synthase cannot be precluded. Overexpression of the anti-apoptotic protein Bcl-xL or pre-incubation with N-acetyl cysteine reduced the intracellular level of ROS and increased resistance to hLf, confirming a ROS-mediated mitochondrial cell death process. Mitochondrial involvement was further reinforced by the higher resistance of cells lacking mitochondrial DNA, or other known yeast mitochondrial apoptosis regulators, such as, Aif1p, Cyc3p and Aac1/2/3p. This study provides new insights into a detailed understanding at the molecular level of hLf-induced apoptosis, which may allow the design of new strategies to overcome the emergence of resistance of clinically relevant fungi to conventional antifungals.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Lactoferrin (Lf) is an iron-binding glycoprotein produced by mucosal epithelial cells or neutrophils in many mammalian species. It is found in several biological fluids, with particular abundance in milk and colostrum. Many biological activities have been attributed to this protein since its discovery, from which its high antimicrobial activity stands out [1]. In fact, it has been demonstrated that Lf exhibits antimicrobial activity against a broad spectrum of filamentous fungi, yeasts, bacteria, protozoa, viruses and parasites [2]. Regarding its antifungal activity, it inhibits the growth of different fungal plant pathogens such as Aspergillus niger and Trichoderma viride [3], of dermatophytic fungi like Trichophyton mentagrophytes [4], and inhibits spore germination and mycelial growth of Botrytis cinerea in a time- and dose-dependent manner [5]. However, most studies on Lf antifungal activity have been performed with species of the genus Candida, probably due to the fact that most species are human pathogens [6]. Lactoferrin candidacidal activity was reported to be species-dependent, with Candida tropicalis and Candida krusei being the most susceptible species [7]. In a previous study, we have demonstrated that the candidacidal activity of Lf is modulated by the metabolic state of the cell and by the extracelular cation concentration, being optimal at aerobic and low-strength conditions, respectively [8]. More recently, we found that Lf induces apoptosis-like cell death in Candida albicans, which depends on the K+-channel-mediated K+ efflux [9]. However, few information about the mechanisms underlying Lf-induced cell death is available. To gain further insight into this process, we decided to use the yeast Saccharomyces cerevisiae, which has been extensively used to elucidate cell death processes [10, 11]. In fact, it has been reported that S. cerevisiae cells can undergo an apoptotic-like cell death in response to different stimuli exhibiting the typical hallmarks of the mammalian apoptosis, including phosphatidylserine externalization, chromatin condensation and DNA fragmentation. Conservation of mitochondria and lysosomal-like apoptotic pathways was also demonstrated [10, 12, 13]. This, together with the molecular tools available, makes S. cerevisiae an ideal model organism for the elucidation of the molecular events occurring during cell death processes [11, 14–16].
Here, we found that hLf triggers a mitochondrial- and caspase-dependent regulated cell death in S. cerevisiae characterized by nuclear chromatin condensation, preservation of the plasma membrane integrity, caspase activation, ROS accumulation and cytochrome c release. The lethal effect of hLf is inhibited by cycloheximide, 2-deoxyglucose and oligomycin, indicating that the cell death process depends on de novo protein synthesis, and mitochondrial energetic metabolism. Mutants deficient in the yeast metacaspase Yca1p and in other known yeast apoptotic regulators, mainly mitochondrial proteins, were more resistant to death induced by hLf. This evidence together with the mitochondrial dysfunctions induced by hLf reinforce the relevance of mitochondria in the cell death process. This study also provides a deeper understanding of hLf-induced apoptosis at the molecular level, which may allow the design of new strategies to overcome the emergence of resistance of clinically relevant fungi to conventional antifungals.
Materials and methods
Strains, media and growth conditions
All S. cerevisiae strains used in this study are listed in Table 1. S. cerevisiae BY4741 and W303-1A and -1B were used as the wild-type strains. The W303-1B strain was transformed with pCM184-Bcl-xL by the LiAc/SS Carrier DNA/PEG method [17]. S. cerevisiae BY4741, W303-1A and -1B, as well as respective mutants were all grown and maintained in YPD (yeast extract-peptone-dextrose) medium with 5 % glucose at 30 °C. S. cerevisiae W303-1B strain transformed with the plasmid pCM184-Bcl-xL was grown at 30 °C in 0.175 % yeast nitrogen-base medium, with 0.5 % ammonium sulphate, 0.1 % potassium di-hydrogen phosphate, 0.01 % adenine, 0.01 % histidine, 0.01 % leucine, 0.01 % uracil and 2 % glucose as the carbon source. The expression of Bcl-xL was repressed by the presence of 1 µg/mL doxycycline (Sigma-Aldrich, St. Louis, MO, USA).
Cell death assays
The activity of hLf against the different strains used throughout this study was assessed by standard dilution plate counts [9]. Briefly, yeast cells were grown in YPD supplemented with 5 % or 2 % glucose, washed twice with Tris–HCl (10 mM pH 7.4), resuspended (105 cells/mL), and incubated with hLf (1.56–6.25 µM) for 90 or 120 min. Cells were diluted, plated onto YPD-agar, and incubated for 24–48 h at 30 °C. The percentage of cell survival was calculated as the number of colonies recovered from hLf-treated cells divided by the number of colonies recovered from control cells. The effects of cycloheximide (10 µg/mL), 2-deoxyglucose (2-DG, 20 nM), oligomycin (4 µg/mL) and N-acetyl-l-cysteine (NAC, 5 mM) were evaluated by pre-incubation with these compounds for 30 min. These three compounds were purchased from Sigma-Aldrich (St. Louis, MO, USA).
Assessment of plasma membrane integrity
The integrity of the plasma membrane was assessed by staining with propidium iodide (PI, Sigma-Aldrich, St. Louis, MO, USA). Briefly, cells were harvested by centrifugation, ressuspended in phosphate buffered saline solution (PBS), and incubated with 2 µg/mL PI at room temperature for 5 min, in the dark, and then analysed by flow cytometry. Cells with red fluorescence [FL-3 channel (488/620 nm)] were considered to have lost their plasma membrane integrity.
Monitoring chromatin condensation
In order to evaluate chromatin condensation, cells were fixed in ethanol-PBS (1:1, v/v), stained with 2 μg/mL of 4,6-diamido-2-phenyl-indole (DAPI, Sigma-Aldrich, St. Louis, MO, USA) for 10 min in the dark and observed by fluorescence microscopy (Leica Microsystems DM-5000B; Leica DCF350FX digital camera). At least 300 cells per experiment were counted.
Caspase activation assays
Caspase activation was assessed by staining with the CaspSCREEN Flow Cytometric Apoptosis Detection Kit (BioVision Inc, Milpitas, CA, USA). Cells were incubated with the non-fluorescent caspase substrate D2R [(Asp)2-rhodamine 110] at 37 °C for 90 min and then analysed by flow cytometry. Cells with green fluorescence [FL-1 channel (488/525 nm)] were considered to display caspase activation. Representative histograms of each condition are available on Online Resource 1.
Determination of reactive oxygen species (ROS) accumulation
Intracellular ROS accumulation was determined by flow cytometry by staining with the dihydroethidium (DHE) probe (Molecular Probes, Eugene, OR, USA). S. cerevisiae suspensions were harvested by centrifugation, ressuspended in PBS, and incubated 30 min with 5 µg/mL DHE in the dark, and then analysed by flow cytometry. Cells with red fluorescence [FL-3 channel (488/620 nm)] were considered to accumulate superoxide anion. Representative histograms of each experiment are available on Online Resource 2 and 3.
Cytochrome c detection
For cytochrome c detection, cells were grown and treated under the conditions above described, harvested, and mitochondrial and cytosolic fractions were prepared as previously described [18]. Mitochondrial and cytosolic fractions were separated electrophoretically on a 15 % SDS–polyacrylamide gel and transferred to a Hybond-Polyvinylidene difluoride membrane (PVDF, GE Healthcare, Pittsburgh, PA, USA) at 60 mA during 1 h 30 m. Membranes were incubated with the primary antibodies: mouse monoclonal anti-yeast phosphoglycerate kinase (PGK1) antibody (1:5000, Molecular Probes, Eugene, OR, USA), mouse monoclonal anti-yeast porin (POR1) antibody (1:5000, Molecular Probes, Eugene, OR, USA) and rabbit polyclonal anti-yeast cytochrome c (CYC1) antibody (1:2000, custom-made by Millegen, Labège, France), followed by incubation with secondary antibodies against mouse or rabbit IgG-peroxidase (1:5000; Sigma Aldrich, St. Louis, MO, USA). Immunodetection of bands was revealed by chemiluminescence (ECL, GE Healthcare, Pittsburgh, PA, USA).
Flow cytometry analysis
Analysis by flow cytometry were performed in an Epics® XL™ (Beckman Coulter) flow cytometer equipped with an argon-ion laser emitting a 488 nm beam at 15 mW. Twenty thousand cells were analysed per sample at low flow rate. Data were analysed by WinMDI 2.8 software (The Scripps Research Institute, La Jolla, CA, USA).
Statistical analysis
All experiments were performed at least three times and data are expressed as the mean values with the corresponding standard deviation (SD). Statistical analysis were performed using GraphPad Prism 4.0 software (GraphPad Software, CA, USA) and p values <0.05 were considered statistically significant in all assays.
Results
hLf-induced cell death in Saccharomyces cerevisiae is active and energy-dependent
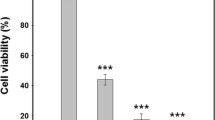
In order to ascertain if hLf triggers cell death in S. cerevisiae BY4741, we exposed exponentially grown cells to increasing concentrations of hLf (1.56, 3.12 and 6.25 µM) in Tris–HCl buffer (pH 7.4) for 90 min, and evaluated cell survival by standard dilution plate counts. Our results indicate that hLf induces a concentration-dependent loss of cell viability that reached approximately 65 % for the highest concentration tested (Fig. 1a). Then, to further characterize the cell death process induced by hLf, the same experiment was carried out in cells pre-incubated with either cycloheximide or 2-deoxyglucose (2-DG) for 30 min. Cycloheximide is an inhibitor of protein synthesis [19], and 2-DG is a non-metabolizable analogue of glucose that inhibits glycolysis and leads to ATP depletion [20]. We observed that both compounds attenuated the toxic effect of hLf and enhanced the percentage of cell survival. Indeed, for the highest dose of hLf (6.25 µM), cycloheximide completely inhibited the hLf lethal effect (grey bars). As for 2-DG, it increased cell survival approximately 30 % in comparison with cells incubated only with hLf (white bars) (Fig. 1b). Altogether, these results revealed that hLf-induced cell death requires active participation of the cellular machinery, depending on de novo protein synthesis and on the energy generated by glucose metabolism.
Susceptibility of S. cerevisiae to hLf. a Cell survival of S. cerevisiae BY4741 to hLf (1.56–6.25 μM) in 10 mM Tris–HCl buffer pH 7.4 for 90 min, evaluated by cfu counting. b Cell survival of S. cerevisiae BY4741 to hLf (3.12 and 6.25 μM) without or after pre-incubation with 10 μg/ml cycloheximide, a protein synthesis inhibitor (grey bars) or 20 nM 2-deoxyglucose, an inhibitor of glycolysis (white bars), evaluated by cfu counting. Values are mean ± SD of three independent experiments. Values significantly different: **P < 0.01, ***P < 0.001 in comparison with untreated cells and ###P < 0.001 in comparison with the indicated conditions
hLf induces a caspase-dependent regulated cell death in Saccharomyces cerevisiae with typical apoptotic markers
To further characterize cell death induced by hLf, we assessed whether it is accompanied by typical apoptotic markers. The ability of hLf to permeabilize the plasma membrane of S. cerevisiae BY4741 cells was investigated by measuring the number of cells stained with the fluorescent probe PI by flow cytometry. PI is a membrane impermeable DNA stain that can diffuse into cells only when the plasma membrane is disrupted. We observed that yeast cells treated with increasing doses of hLf for 90 min were mainly PI-negative, indicating that cell death induced by hLf is a controlled process that, unlike necrosis, does not compromise plasma membrane integrity (Fig. 2a).
Apoptotic markers induced by hLf in S. cerevisiae . a Assessment of plasma membrane integrity in BY4741 cells treated with hLf (1.56–6.25 μM) for 90 min in 10 mM Tris–HCl buffer (pH 7.4) by flow cytometry using PI staining. b Determination of the number of cells with nuclear chromatin condensation (nuclei that change from a round to a kidney or ring shape) in BY4741 cells treated with hLf in the same conditions. (i) and (ii)—aspect of nuclei in control or hLf-treated cells, respectively. Bar 5 µm (c) Cell survival of S. cerevisiae BY4741 wild-type (black bars) and a mutant lacking the yeast metacaspase Yca1p (yca1Δ) (grey bars), evaluated by cfu counting. d Caspase activation monitored by flow cytometry using the caspase substrate D2R. Values are mean ± SD of at least three independent experiments. Values significantly different: *P < 0.05, **P < 0.01, ***P < 0.001 in comparison with untreated cells and ###P < 0.001 in comparison with the indicated conditions
Another hallmark of apoptosis is the appearance of nuclei with chromatin condensation. In order to appraise if hLf induces this nuclear alteration, nuclei of hLf-treated cells were observed after staining with DAPI. In fact, we found that hLf induces an increase in kidney-shaped condensed chromatin characteristic of apoptotic cells (Fig. 2b-ii), in contrast with control cells that exhibited only bright, round-shaped nuclei (Fig. 2b-i). Preservation of plasma membrane integrity and chromatin condensation were also observed in C. albicans cells exposed to lethal concentrations of Lf [9].
Caspases play an important role in the apoptotic signalling network, and are usually activated in the early stages of apoptosis. In 2002, Madeo et al. found a yeast ortholog of the mammalian caspases, the metacaspase Yca1p [21]. To examine whether this protein is involved in hLf-induced cell death, cell survival in response to hLf was monitored in cells lacking Yca1p (yca1Δ). Results show that the absence of Yca1p renders cells significantly more resistant to the toxic effect of hLf than the wild-type strain (Fig. 2c), suggesting that hLf-induced cell death is caspase-dependent. To further investigate the role of Yca1p in this process, cells were incubated with D2R, enabling the determination of the percentage of cells with caspase activation by flow cytometry [21]. Data revealed that, in fact, hLf induces a significant dose-dependent increase in the number of cells with active caspase in the wild-type strain, with almost 50 % of cells exposed to the highest dose of bLf exhibiting caspase activation (Fig. 2d).
Overall, these results demonstrate that hLf induces a Yca1p-dependent regulated cell death in S. cerevisiae associated with preservation of plasma membrane integrity, nuclear chromatin condensation and metacaspase activation.
Mitochondria participate in hLf-induced apoptosis
ROS accumulation has been implicated in the induction and regulation of apoptosis in yeast [10, 19]. These include the superoxide anion, hydroxyl radicals and hydrogen peroxide. When the levels of ROS exceed the antioxidant capacity of the cell, homeostasis is disrupted and yeast survival is compromised. Therefore, to determine the production and accumulation of intracellular ROS in response to hLf treatment, we used the ROS sensitive probe DHE, a non-fluorescent probe that intercalates into nucleic acids after oxidation by superoxide anion to ethidium.
Cells exposed to hLf (3.12 and 6.25 µM) displayed an accumulation of superoxide anion, in contrast to control cells. Superoxide anion was detected in 36.1 ± 1.8 and 45.4 ± 2.3 % of cells treated with 3.12 and 6.25 µM hLf, respectively (Fig. 3a). These results suggest that hLf-induced cell death is associated with accumulation of superoxide anion.
Involvement of ROS production and cytochrome c release in cell death induced by hLf. a Levels of superoxide anion in S. cerevisiae strain BY4741 exposed to different concentrations of hLf for 90 min using the DHE probe by flow cytometry. H2O2 (3 mM) was used as a positive control. b Western blot analysis of cytochrome c release in S. cerevisiae strain BY4741, after exposure to Tris–HCl buffer (control, C) and hLf for 60 and 120 min, in both mitochondrial and cytosolic fractions. Cytosolic phosphoglycerate kinase (Pgk1p) and mitochondrial porin (Por1p) levels were used as loading controls of cytosolic and mitochondrial fractions, respectively. c Survival of S. cerevisiae BY4741 for 90 min to hLf after pre-incubation with the antioxidant NAC. d Levels of superoxide anion after pre-incubation with NAC, using the DHE probe by flow cytometry. e Survival of S. cerevisiae BY4741 and mutant strains aif1Δ and cyc3Δ exposed to hLf for 90 min. Values are mean ± SD of at least three independent experiments. Values significantly different: **P < 0.01, ***P < 0.001 in comparison with untreated cells and #P < 0.05, ###P < 0.001 in comparison with the indicated conditions
A crucial event in yeast apoptosis is the translocation of cytochrome c from mitochondria to the cytosol. Therefore, we next determined whether hLf triggers the release of cytochrome c. Cytosolic and mitochondrial fractions were isolated by differential centrifugation and the levels of cytochrome c in the mitochondrial fraction and postmitochondrial supernatant, containing soluble cytosolic proteins, were analysed by western blot. We found that incubation of cells with hLf induces a time-dependent release of cytochrome c to the cytosol (Fig. 3b), indicating that hLf induces permeabilization of the mitochondrial membrane, and suggesting a critical role of mitochondria in this cell death process.
NAC is an aminothiol and a synthetic precursor of intracellular cysteine and of glutathione S transferase (GSH), and thus considered an important antioxidant. The action of NAC results from its antioxidant or free radical scavenging properties by increasing the intracellular levels of GSH [22]. Since hLf led to the accumulation of superoxide anion, we next investigated the effect of NAC in the viability (Fig. 3c) and accumulation of this ROS species (Fig. 3d) of cells treated with hLf.
We confirmed that a cell suspension pre-incubated with 5 mM NAC exhibited a drastic reduction in the percentage of cells with superoxide anion accumulation, and were significantly more resistant to hLf than cells exposed to hLf without the antioxidant. Indeed, cell survival was close to 100 % even for the higher concentration of hLf after 90 min of treatment (Fig. 3c). Therefore, pre-incubation of cells with NAC reduced the levels of superoxide anion in cells treated with hLf to the levels observed in control cells (Fig. 3d).
Cyc3p is required for the covalent binding of the heme group to isoforms 1 and 2 of apocytochrome c to form mature cytochrome c, which is released from mitochondria to trigger apoptosis [23]. Mutant cells deficient in this enzyme exhibited increased survival in response to hLf (Fig. 3e—white bars). Furthermore, several stimuli were shown to trigger a mitochondrial-dependent apoptotic pathway in yeast, associated with different mitochondrial dysfunctions including permeabilization of the mitochondrial outer membrane (MOMP) and subsequent release of pro-apoptotic regulators other than cytochrome c, such as Aif1p. Aif1p is a flavoprotein with oxidoreductase activity localized in the mitochondrial intermembrane space that, upon apoptosis induction, translocates to the nucleus where it leads to chromatin condensation and DNA degradation [24]. The involvement of this mitochondrial apoptotic regulator in the cell death process induced by hLf was also studied. We found that absence of Aif1p enhanced cell survival in response to hLf (Fig. 3e—grey bars).
Altogether, the results indicate that both mature cytochrome c and Aif1p are required for the execution of apoptosis by hLf, reinforcing the pivotal role of mitochondria in hLf-induced apoptosis.
Several studies suggest that the connection between mitochondria and apoptosis is intricate, and abolishment of mitochondrial function can be either deleterious or beneficial for the cell depending on the death scenario. The ability of yeast cells to grow on non-fermentable carbon sources and to regulate metabolism, switching from aerobic fermentation at high glucose concentration to respiratory metabolism, or to grow in the absence of mitochondrial DNA (ρ0) or of components of ATPase synthase, have made yeast an excellent tool to address the need for mitochondria in the execution of cell death [25]. In order to better understand the involvement of mitochondria in hLf-inducing apoptosis, we assessed the survival of ρ0, atp10∆, atp2∆ and aac1/2/3∆ mutants in response to hLf. Atp10p was identified as a mitochondrial inner membrane component required for the assembly of the mitochondrial F1F0 complex [26], and Atp2p as the beta subunit of the F1 sector of mitochondrial F1F0 ATP synthase [26]. Aac1/2/3p are the three isoforms of the mitochondrial inner membrane ADP/ATP translocator, required for MOMP and cytochrome c release in yeast apoptosis [27]. The ρ0 mutant lacks mitochondrial DNA and all the respiratory complexes, and has only poorly developed mitochondria [28].
As can be observed in Fig. 4a and b, the wild-type strains W303-1B and -1A behaved like the BY4741 strain in response to hLf, exhibiting similar cell survival in the presence of increasing doses of hLf. In contrast, ρ0, atp10∆ and atp2∆ and aac1/2/3∆ mutant strains were more resistant to death induced by hLf, comparatively to the wild-type strain.
Mitochondria contribution to cell death induced by hLf. a Survival of S. cerevisiae strain W303-1B, atp2Δ and aac1/2/3Δ and b W303-1A, ρ0 and atp10Δ mutant strains, in response to hLf for 90 min. c Effect of the mitochondrial ATPase inhibitor oligomycin (4 μg/ml) on cell survival in response to hLf. d Survival of S. cerevisiae W303-1B overexpressing the anti-apoptotic protein Bcl-xL to hLf. e Effect of overexpressing the anti-apoptotic protein Bcl-xL in the levels of superoxide anion in cells treated with hLf, monitored by flow cytometry using the DHE probe. Values are mean ± SD of at least three independent experiments. Values significantly different: **P < 0.01, ***P < 0.001 in comparison with untreated cells and #P < 0.05, ##P < 0.01, ###P < 0.001 in comparison with the indicated conditions
Oligomycin inhibits the mitochondrial ATP synthase by blocking its proton channel (F0 subunit), which is necessary for oxidative phosphorylation and for energy production by conversion of ADP to ATP. Wild-type cells were co-treated with hLf and oligomycin and, in accordance with the higher resistance of atp10∆ and atp2∆ mutants to hLf, co-incubation of the wild-type strain with oligomycin enhanced cell survival in response to hLf (Fig. 4c). This result reinforces the dependence of the lethal effect of hLf on mitochondrial energetic metabolism.
To confirm that mitochondria have a pivotal function in hLf-induced apoptosis, we analysed whether expression of Bcl-xL affected this process. Bcl-xL is a member of the Bcl-2 family of proteins, and acts as a pro-survival protein by preventing the release of mitochondrial contents such as cytochrome c, which could trigger caspase activation in mammalian cells [29]. For that purpose, the wild-type strain was transformed with the pCM184 vector (with a tetracycline-regulatable promoter system for regulated gene expression) containing the gene encoding Bcl-xL protein, and its effect in cell viability and ROS accumulation was determined. Expression of Bcl-xL protects cells against death induced by hLf in comparison with cells grown in the presence of doxycycline, where Bcl-xL is not expressed (Fig. 4d). We also found that the higher survival of cells expressing Bcl-xL in response to hLf is linked to a decrease in the accumulation of superoxide anion (Fig. 4e).
Discussion
Lf is a natural protein of paramount interest in clinical applications due to its multiple biological activities, including in the treatment of fungal infections. Lf was shown to exhibit lethal effects against clinically important fungi such as Candida spp. Our group has demonstrated that human Lf induces an apoptosis-like cell death in C. albicans, which may underlie its antifungal activity [9]. In spite of these and other studies, very little is known regarding the hLf antifungal mechanism of action. Due to the potential applications of hLf to treat infections caused by yeast pathogens, a deeper understanding of the molecular basis of its antifungal activity and ability to induce apoptosis is of prime importance. Therefore, to dissect the apoptotic cascade induced by hLf we used the well-established yeast apoptosis model in S. cerevisiae.
Our results show that, like in C. albicans [9], hLf triggers cell death in S. cerevisiae. The cell death process is active and under genetic control, and shares features typical of mammalian apoptotic cells. Indeed, this process is delayed by de novo protein synthesis and metabolic inhibitors, and characterised by chromatin condensation associated with preservation of plasma membrane integrity. Pre-treatment with the antioxidant NAC caused a significant reduction in superoxide anion accumulation associated with higher cell survival, indicating that this reactive oxygen species plays an executor role in the death process. Similar observations were found in our previous studies with C. albicans, which commits into an apoptotic cell death mediated by ROS in response to hLf [9]. However, in that study the involvement of known regulators of the core yeast apoptotic machinery was not addressed. We therefore investigated their role in hLf-induced apoptosis.
The yeast metacaspase Yca1p has been implicated in apoptosis scenarios in response to different stimuli [21, 30, 31]. Here, we show that cells lacking Yca1p display higher cell survival upon exposure to hLf. In accordance, hLf-induced apoptosis requires caspase activation. The percentage of cells with active metacaspase is not strictly correlated with the percentage of dead cells assessed by cfus and PI staining (e.g., treatment with 6.25 μm of hLf resulted in 50 % wild-type strain cells with caspase activation while associated with 70 % loss of cell proliferation and 15 % of PI positive cells). These results suggest that caspase activation mainly occurs in non-proliferating cells that preserve their plasma membrane integrity, which further supports the involvement of Yca1p in hLf-induced apoptosis.
Numerous studies demonstrated the involvement of mitochondria in yeast apoptosis [10, 11, 13]. Here, we present several evidence indicating that hLf triggers a mitochondrial-mediated apoptotic pathway which involves cytochrome c release from the mitochondria to the cytosol. Additionally, we found that absence of holocytochrome c and Aif1p in cyc3Δ and aif1Δ mutant strains enhances cell survival. The release of cytochrome c in cells undergoing apoptosis induced by hLf, together with the higher resistance of the mutant lacking the CYC3 gene, which encodes a heme-lyase essential for heme binding to apocytochrome c (isoform 1 and 2) [32], supports the interpretation that mature cytochrome c and its release are important for the execution of the death process. Moreover, the resistant phenotype of cells lacking Aif1p, suggests that, besides cytochrome c, and as reported in other yeast apoptotic scenarios [11, 24], the mitochondrial pro-apoptotic factor Aif1p is involved in hLf-induced apoptosis.
S. cerevisiae strains W303-1A and 1B display cell survival percentages and levels of superoxide anion accumulation in response to hLf identical to S. cerevisiae strain BY4741. We therefore characterised the phenotype of other mitochondrial deficient mutants, namely a respiratory deficient mutant (ρ0) and a mutant deficient in subunit 10 of the ATPase complex (atp10Δ) in the W303-1A background, as well as mutants deficient in subunit 2 of the ATPase complex (atp2Δ) and in the ADP/ATP carrier (AAC) proteins (aac1/2/3Δ) in the W303-1B background. The ρ0 mutant, which is devoided of both mitochondrial DNA and respiration, particularly exhibits high cell survival. The repression of the CYC1 gene, encoding for iso-1-CytC [33], further supports the role of cytochrome c in apoptosis induced by hLf.
Different molecular mechanisms/components underlying MOMP and subsequent release of cytochrome c and other pro-apoptotic factors have been proposed [34–37]. These include the permeability transition pore complex (PTPC), the protein pores formed by Bax/Bak oligomers, and the ceramide channels, acting alone or in combination in MOMP. Though extensive studies suggested different cytosolic and mitochondrial proteins as components/regulators of PTPC, its precise molecular composition and regulation remains elusive. Some of the proteins identified include, among others, isoforms of ANT, VDAC and HK, CYPD, PiC, GSK3β, p53, and several members of the Bcl-2 protein family. Though yeast does not possess obvious orthologs of p53 and of the Bcl-2 protein family, the same does not apply to other putative components of the PTPC. Therefore, the role of several of the yeast orthologs of the putative components of the PTPC has been studied [27]. Absence of ADP/ATP carrier proteins, yeast orthologs of the mammalian adenine nucleotide translocator (ANT), protects cells exposed to various apoptotic stimuli such as acetic acid and diamide, while hindering MOMP and cytochrome c release [27]. The hypothesis that AAC proteins also play a critical role in hLf-induced apoptosis is supported by the higher survival of cells lacking the three AAC isoforms (aac1/2/3Δ). Mitochondrial ATPase complex subunit 10 and 2, encoding for the F1F0- ATPase assembly factor and the β subunit of ATP synthase, respectively, were implicated in acetic acid-induced apoptosis, though with opposite roles [13, 27]. Here, we show that cells lacking Atp10p or Atp2p are less susceptible to hLf-induced apoptosis. This indicates that a fully assembled and fuctional FoF1-ATPase is required for the killing effect of hLf. However, failure in mitochondrial ATP synthesis could underlie the protective effect of both mutants deficient in oxidative phosphorylation. This appears to be the case since cells of the wild-type strain pre-treated with oligomycin are unable to commit to cell death in response to hLf. Recent data propose that mitochondrial ATP synthase plays a key role in mitochondria permeability transition (MPT) [38, 39]. Though not fully confirmed, it is hypothesised that the c subunit in particular constitutes the pore-forming unit of the PTPC. Taking into account that oligomycin specifically inhibits the c subunit of ATP synthase, and at the same time protects cells from hLf-induced apoptosis, the implication of ATP synthase in MPT warrants further studies.
Expression of the mammalian anti-apoptotic protein Bcl-xL in yeast was shown to protect cells from death induced by both exogenous and endogenous stimuli, namely from acetic-acid induced apoptosis and from heterologous expression of Bax [40]. Similarly, we found that hLf-induced cell death is rescued by heterologous expression of Bcl-xL, which also causes a reduction in superoxide anion accumulation. This result further supports the involvement of a ROS-dependent mitochondria-mediated pathway in the apoptotic death induced by hLf.
In summary, we found that hLf induces a mitochondrial- and caspase-dependent regulated cell death in S. cerevisiae, and implicated different known yeast apoptosis regulators. S. cerevisiae has been increasingly reported to act as an opportunist agent and promote invasive infection, especially in immunocompromised or critically ill patients [41–44]. Our study therefore provides new clues that may have relevant clinical applications. The results obtained may also be useful in the aplication of hLf alone or in combination with classical antifungals such as azoles [45], and for development of hLf-derived peptides with interesting antifungal activities [46, 47]. Finally, the high prevalence of invasive fungal diseases in hospitalized and immunocompromised patients, in particular caused by Candida spp, and the resistance of these pathogens to conventional antifungal drugs emphasize the need for treatments with novel antifungals [48], among which hLf appears very promising.
References
Garcia-Montoya I, Gonzalez-Chavez SA, Salazar-Martinez J, Arevalo-Gallegos S, Sinagawa-Garcia S, Rascon-Cruz Q (2013) Expression and characterization of recombinant bovine lactoferrin in E. coli. Biometals 26(1):113–122. doi:10.1007/s10534-012-9598-7
Vogel HJ (2012) Lactoferrin, a bird’s eye view. Biochem Cell Biol 90(3):233–244. doi:10.1139/o2012-016
Lahoz E, Pisacane A, Iannaccone M, Palumbo D, Capparelli R (2008) Fungistatic activity of iron-free bovin lactoferrin against several fungal plant pathogens and antagonists. Nat Prod Res 22(11):955–961. doi:10.1080/14786410701650253
Wakabayashi H, Uchida K, Yamauchi K, Teraguchi S, Hayasawa H, Yamaguchi H (2000) Lactoferrin given in food facilitates dermatophytosis cure in guinea pig models. J Antimicrob Chemother 46(4):595–602
Wang J, Xia XM, Wang HY, Li PP, Wang KY (2013) Inhibitory effect of lactoferrin against gray mould on tomato plants caused by Botrytis cinerea and possible mechanisms of action. Int J Food Microbiol 161(3):151–157. doi:10.1016/j.ijfoodmicro.2012.11.025
Al-Sheikh H (2009) Effect of lactoferrin and iron on the growth of human pathogenic Candida species. Pak J Biol Sci 12(1):91–94
Xu YY, Samaranayake YH, Samaranayake LP, Nikawa H (1999) In vitro susceptibility of Candida species to lactoferrin. Med Mycol 37(1):35–41
Viejo-Diaz M, Andres MT, Fierro JF (2004) Modulation of in vitro fungicidal activity of human lactoferrin against Candida albicans by extracellular cation concentration and target cell metabolic activity. Antimicrob Agents Chemother 48(4):1242–1248
Andres MT, Viejo-Diaz M, Fierro JF (2008) Human lactoferrin induces apoptosis-like cell death in Candida albicans: critical role of K+ -channel-mediated K+ efflux. Antimicrob Agents Chemother 52(11):4081–4088. doi:10.1128/AAC.01597-07
Pereira C, Silva RD, Saraiva L, Johansson B, Sousa MJ, Corte-Real M (2008) Mitochondria-dependent apoptosis in yeast. Biochim Biophys Acta 1783(7):1286–1302. doi:10.1016/j.bbamcr.2008.03.010
Carmona-Gutierrez D, Eisenberg T, Buttner S, Meisinger C, Kroemer G, Madeo F (2010) Apoptosis in yeast: triggers, pathways, subroutines. Cell Death Differ 17(5):763–773. doi:10.1038/cdd.2009.219
Sousa M, Duarte AM, Fernandes TR, Chaves SR, Pacheco A, Leao C, Corte-Real M, Sousa MJ (2013) Genome-wide identification of genes involved in the positive and negative regulation of acetic acid-induced programmed cell death in Saccharomyces cerevisiae. BMC Genom 14:838. doi:10.1186/1471-2164-14-838
Ludovico P, Rodrigues F, Almeida A, Silva MT, Barrientos A, Corte-Real M (2002) Cytochrome c release and mitochondria involvement in programmed cell death induced by acetic acid in Saccharomyces cerevisiae. Mol Biol Cell 13(8):2598–2606. doi:10.1091/mbc.E01-12-0161
Madeo F, Engelhardt S, Herker E, Lehmann N, Maldener C, Proksch A, Wissing S, Frohlich KU (2002) Apoptosis in yeast: a new model system with applications in cell biology and medicine. Curr Genet 41(4):208–216. doi:10.1007/s00294-002-0310-2
Ludovico P, Madeo F, Silva M (2005) Yeast programmed cell death: an intricate puzzle. IUBMB Life 57(3):129–135. doi:10.1080/15216540500090553
Frohlich KU, Fussi H, Ruckenstuhl C (2007) Yeast apoptosis-from genes to pathways. Semin Cancer Biol 17(2):112–121. doi:10.1016/j.semcancer.2006.11.006
Gietz RD, Woods RA (2006) Yeast transformation by the LiAc/SS Carrier DNA/PEG method. Methods Mol Biol 313:107–120. doi:10.1385/1-59259-958-3:107
Rego A, Costa M, Chaves SR, Matmati N, Pereira H, Sousa MJ, Moradas-Ferreira P, Hannun YA, Costa V, Corte-Real M (2012) Modulation of mitochondrial outer membrane permeabilization and apoptosis by ceramide metabolism. PLoS One 7(11):e48571. doi:10.1371/journal.pone.0048571
Madeo F, Frohlich E, Ligr M, Grey M, Sigrist SJ, Wolf DH, Frohlich KU (1999) Oxygen stress: a regulator of apoptosis in yeast. J Cell Biol 145(4):757–767
O’Donnell AF, McCartney RR, Chandrashekarappa DG, Zhang BB, Thorner J, Schmidt MC (2015) 2-Deoxyglucose impairs Saccharomyces cerevisiae growth by stimulating Snf1-regulated and alpha-arrestin-mediated trafficking of hexose transporters 1 and 3. Mol Cell Biol 35(6):939–955. doi:10.1128/MCB.01183-14
Madeo F, Herker E, Maldener C, Wissing S, Lachelt S, Herlan M, Fehr M, Lauber K, Sigrist SJ, Wesselborg S, Frohlich KU (2002) A caspase-related protease regulates apoptosis in yeast. Mol Cell 9(4):911–917
Sun SY (2010) N-acetylcysteine, reactive oxygen species and beyond. Cancer Biol Ther 9(2):109–110
Dumont ME, Cardillo TS, Hayes MK, Sherman F (1991) Role of cytochrome c heme lyase in mitochondrial import and accumulation of cytochrome c in Saccharomyces cerevisiae. Mol Cell Biol 11(11):5487–5496
Wissing S, Ludovico P, Herker E, Buttner S, Engelhardt SM, Decker T, Link A, Proksch A, Rodrigues F, Corte-Real M, Frohlich KU, Manns J, Cande C, Sigrist SJ, Kroemer G, Madeo F (2004) An AIF orthologue regulates apoptosis in yeast. J Cell Biol 166(7):969–974. doi:10.1083/jcb.200404138
Eisenberg T, Buttner S, Kroemer G, Madeo F (2007) The mitochondrial pathway in yeast apoptosis. Apoptosis 12(5):1011–1023. doi:10.1007/s10495-007-0758-0
Ackerman SH, Tzagoloff A (1990) ATP10, a yeast nuclear gene required for the assembly of the mitochondrial F1-F0 complex. J Biol Chem 265(17):9952–9959
Pereira C, Camougrand N, Manon S, Sousa MJ, Corte-Real M (2007) ADP/ATP carrier is required for mitochondrial outer membrane permeabilization and cytochrome c release in yeast apoptosis. Mol Microbiol 66(3):571–582. doi:10.1111/j.1365-2958.2007.05926.x
Slonimski PP, Perrodin G, Croft JH (1968) Ethidium bromide induced mutation of yeast mitochondria: complete transformation of cells into respiratory deficient non-chromosomal “petites”. Biochem Biophys Res Commun 30(3):232–239
Kuwana T, Newmeyer DD (2003) Bcl-2-family proteins and the role of mitochondria in apoptosis. Curr Opin Cell Biol 15(6):691–699
Mazzoni C, Falcone C (2008) Caspase-dependent apoptosis in yeast. Biochim Biophys Acta 1783(7):1320–1327. doi:10.1016/j.bbamcr.2008.02.015
Vachova L, Palkova Z (2007) Caspases in yeast apoptosis-like death: facts and artefacts. FEMS Yeast Res 7(1):12–21. doi:10.1111/j.1567-1364.2006.00137.x
Pearce DA, Sherman F (1995) Degradation of cytochrome oxidase subunits in mutants of yeast lacking cytochrome c and suppression of the degradation by mutation of yme1. J Biol Chem 270(36):20879–20882
Epstein CB, Waddle JA, Wt Hale, Dave V, Thornton J, Macatee TL, Garner HR, Butow RA (2001) Genome-wide responses to mitochondrial dysfunction. Mol Biol Cell 12(2):297–308
Rasola A, Bernardi P (2007) The mitochondrial permeability transition pore and its involvement in cell death and in disease pathogenesis. Apoptosis 12(5):815–833. doi:10.1007/s10495-007-0723-y
Scharstuhl A, Mutsaers HA, Pennings SW, Russel FG, Wagener FA (2009) Involvement of VDAC, Bax and ceramides in the efflux of AIF from mitochondria during curcumin-induced apoptosis. PLoS One 4(8):e6688. doi:10.1371/journal.pone.0006688
Tsujimoto Y, Shimizu S (2007) Role of the mitochondrial membrane permeability transition in cell death. Apoptosis 12(5):835–840. doi:10.1007/s10495-006-0525-7
Zhivotovsky B, Galluzzi L, Kepp O, Kroemer G (2009) Adenine nucleotide translocase: a component of the phylogenetically conserved cell death machinery. Cell Death Differ 16(11):1419–1425. doi:10.1038/cdd.2009.118
Bonora M, Bononi A, De Marchi E, Giorgi C, Lebiedzinska M, Marchi S, Patergnani S, Rimessi A, Suski JM, Wojtala A, Wieckowski MR, Kroemer G, Galluzzi L, Pinton P (2013) Role of the c subunit of the F0 ATP synthase in mitochondrial permeability transition. Cell Cycle 12(4):674–683. doi:10.4161/cc.23599
Bonora M, Wieckowski MR, Chinopoulos C, Kepp O, Kroemer G, Galluzzi L, Pinton P (2015) Molecular mechanisms of cell death: central implication of ATP synthase in mitochondrial permeability transition. Oncogene 34(12):1475–1486. doi:10.1038/onc.2014.96
Kissova I, Plamondon LT, Brisson L, Priault M, Renouf V, Schaeffer J, Camougrand N, Manon S (2006) Evaluation of the roles of apoptosis, autophagy, and mitophagy in the loss of plating efficiency induced by Bax expression in yeast. J Biol Chem 281(47):36187–36197. doi:10.1074/jbc.M607444200
Aucott JN, Fayen J, Grossnicklas H, Morrissey A, Lederman MM, Salata RA (1990) Invasive infection with Saccharomyces cerevisiae: report of three cases and review. Rev Infect Dis 12(3):406–411
Cimolai N, Gill MJ, Church D (1987) Saccharomyces cerevisiae fungemia: case report and review of the literature. Diagn Microbiol Infect Dis 8(2):113–117
Enache-Angoulvant A, Hennequin C (2005) Invasive Saccharomyces infection: a comprehensive review. Clin Infect Dis 41(11):1559–1568. doi:10.1086/497832
Strope PK, Skelly DA, Kozmin SG, Mahadevan G, Stone EA, Magwene PM, Dietrich FS, McCusker JH (2015) The 100-genomes strains, an S. cerevisiae resource that illuminates its natural phenotypic and genotypic variation and emergence as an opportunistic pathogen. Genome Res 25(5):762–774. doi:10.1101/gr.185538.114
Kobayashi T, Kakeya H, Miyazaki T, Izumikawa K, Yanagihara K, Ohno H, Yamamoto Y, Tashiro T, Kohno S (2011) Synergistic antifungal effect of lactoferrin with azole antifungals against Candida albicans and a proposal for a new treatment method for invasive candidiasis. Jpn J Infect Dis 64(4):292–296
Lupetti A, Brouwer CP, Bogaards SJ, Welling MM, de Heer E, Campa M, van Dissel JT, Friesen RH, Nibbering PH (2007) Human lactoferrin-derived peptide’s antifungal activities against disseminated Candida albicans infection. J Infect Dis 196(9):1416–1424. doi:10.1086/522427
Mishra B, Leishangthem GD, Gill K, Singh AK, Das S, Singh K, Xess I, Dinda A, Kapil A, Patro IK, Dey S (1828) A novel antimicrobial peptide derived from modified N-terminal domain of bovine lactoferrin: design, synthesis, activity against multidrug-resistant bacteria and Candida. Biochim Biophys Acta 2:677–686. doi:10.1016/j.bbamem.2012.09.021
Cerqueira DF, Portela MB, Pomarico L, de Araujo Soares RM, de Souza IP, Castro GF (2010) Oral Candida colonization and its relation with predisposing factors in HIV-infected children and their uninfected siblings in Brazil: the era of highly active antiretroviral therapy. J Oral Pathol Med 39(2):188–194. doi:10.1111/j.1600-0714.2009.00857
Acknowledgments
This work was supported by the Ministerio de Educación, Programa Campus de Excelencia Internacional-Subprograma de Fortalecimiento (Convocatoria 2010) and the grant UNOV-10-BECDOC given to Maikel Acosta Zaldívar by University of Oviedo. This work was supported by Fundação para a Ciência e Tecnologia (FCT) through the strategic funding UID/BIA/04050/2013, and through the project Pest- FCT-ANR/BEX-BCM/0175/2012. A. Rego was the recipient of a FCT fellowship (SFRH/BD/79523/2011). C.S.Pereira was the recipient of a fellowship from the project FCT-ANR/BEX-BCM/0175/2012.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Acosta-Zaldívar, M., Andrés, M.T., Rego, A. et al. Human lactoferrin triggers a mitochondrial- and caspase-dependent regulated cell death in Saccharomyces cerevisiae . Apoptosis 21, 163–173 (2016). https://doi.org/10.1007/s10495-015-1199-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-015-1199-9