Abstract
Arnica montana L. is a medical plant of the Asteraceae family and grows preferably on nutrient poor soils in mountainous environments. Such surroundings are known to make plants dependent on symbiosis with other organisms. Up to now only arbuscular mycorrhizal fungi were found to act as endophytic symbiosis partners for A. montana. Here we identified five Streptomyces strains, microorganisms also known to occur as endophytes in plants and to produce a huge variety of active secondary metabolites, as inhabitants of A. montana. The secondary metabolite spectrum of these strains does not contain sesquiterpene lactones, but consists of the glutarimide antibiotics cycloheximide and actiphenol as well as the diketopiperazines cyclo-prolyl-valyl, cyclo-prolyl-isoleucyl, cyclo-prolyl-leucyl and cyclo-prolyl-phenylalanyl. Notably, genome analysis of one strain was performed and indicated a huge genome size with a high number of natural products gene clusters among which genes for cycloheximide production were detected. Only weak activity against the Gram-positive bacterium Staphylococcus aureus was revealed, but the extracts showed a marked cytotoxic activity as well as an antifungal activity against Candida parapsilosis and Fusarium verticillioides. Altogether, our results provide evidence that A. montana and its endophytic Streptomyces benefit from each other by completing their protection against competitors and pathogens and by exchanging plant growth promoting signals with nutrients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Arnica montana L. is a herbaceous plant of the Asteraceae family. It is endemic in Europe and predominantly found in nutrient-poor grasslands, shrublands, and in alpine mountain environments (Sugier et al. 2013). Preparations, especially from its flowerheads have a long tradition for the external treatment of inflammatory diseases. They have had a positive judgment from the European Scientific Cooperative on Phytotherapy (ESCOP) and the European Pharmacopoeia for the treatment of several injuries such as bruises, distortions, sprains, hematoma, inflamed insect stings as well as rheumatic complaints (ESCOP 2003; Ph.Eur 2012). Sesquiterpene lactones such as helenalin, 11α,13-dihydrohelenalin and their esters are the most effective compounds responsible for the utilised anti-inflammatory activity (Teuscher et al. 2009; Merfort 2010). These compounds were proven to inhibit the transcription factor NF-κB thus targeting the inflammatory processes at a central point (Lyss et al. 1997; García-Piñeres et al. 2001). Additionally, those molecules exert an antibacterial activity against Gram-positive bacteria (Lee et al. 1977).
Even though A. montana grows on nutrient-poor acidic soils, which makes plants often more dependent on symbiosis with other organisms, only few details about symbiotic interactions with A. montana are known. There is evidence for symbiosis with arbuscular mycorrhizal fungi, but no report exists on a symbiosis with endophytic Streptomyces (Ryszka et al. 2010). Up to now only a few species of the large plant family of Asteraceae were reported to interact with endophytic Streptomyces, among them Artemisia annua, which is also a plant with known medicinal properties (Sardi et al. 1992; Jiménez-Esquilín and Roane 2005; Castillo et al. 2007; Lin et al. 2012; Tanvir et al. 2014).
Streptomyces and other endophytes are known to produce various bioactive metabolites which target e.g. bacteria, fungi, and cancer cells (Porras-Alfaro and Bayman 2011; Bérdy 2005). Regarding the secondary metabolites of plants, it is known, that in several cases the real producer of the compounds is not the plant itself but its endophytes (Bérdy 2005). There are also cases in which both plant and endophyte produce the same metabolites. One prominent example is the anticancer drug taxol which is isolated from the bark of Taxus brevifolia. It has been shown that the inhabiting endophyte Taxomyces andreanae is also able to synthesize this substance (Stierle et al. 1993). This raises the question if endophytes of A. montana are also able to produce the medically active sesquiterpene lactone derivatives synthesised by this plant.
The aims of this study were to identify species of Streptomyces inhabiting A. montana, as well as to determine active secondary metabolites in the Streptomyces extracts. From the possible huge variety of pharmacological effects, we focused on the cytotoxic, antibacterial and antifungal properties of these extracts. Altogether these studies will further increase the knowledge about this medicinal plant and its interactions with microorganisms.
Materials and methods
Plant material and isolation of bacterial strains
Arnica montana L. was collected in Vosges mountains, Breitfirstkopf, in France 2009. The isolation of actinobacteria was performed as described in literature (Coombs and Franco 2003) with minor modifications. A. montana L. roots from three plants were dried for 48 h at room temperature, and were afterwards thoroughly washed to remove all soil residues. The roots were excised into pieces with a diameter of approximately 0.3–0.7 cm and a length of 2–3 cm. This material was subjected to a three-step surface sterilisation procedure: a 60 s wash in 99 % ethanol, followed by a 6-min wash in 3.125 % NaOCl, a 30-s wash in 99 % ethanol, and a final rinse with sterile water. The surface-sterilised roots were then smashed into small fragments under sterile conditions and plated onto the isolation media at 28 °C for up to 2 weeks. TSB and MS agar media were used in the isolation experiment. Finally, the strains were isolated on the MS agar, because on TSB agar medium Actinomycetes were overgrown by other bacteria. The efficacy of the surface sterilisation procedure was assessed by rolling surface-sterilised roots in the isolation medium which was then incubated at 28 °C. Endophytic bacterial strains, in total 5, were stored in 20 % saccharose solution at −80 °C.
Cultivation of bacterial strains and preparation of the extracts
The bacterial strains were pre-cultivated in 300 mL Erlenmeyer flasks (2 days, 28 °C, 180 rpm) filled with 50 mL TSB medium and 100 µL of sucrose-culture (1 L TSB consists of tryptic soy broth 30.0 g). The main cultivation was made in 500 mL Erlenmeyer flasks containing 100 mL DNPM-medium (1 L DNPM medium consists of dextrin 40.0 g, soy tone 7.50 g, fresh yeast 5.00 g, 4-morpholinepropanesulfonic acid (MOPS) 21.0 g, pH 6.8). The cultures were inoculated with 5 mL pre-culture and cultivated for 7 days (30 °C, 180 rpm). Altogether five extracts were obtained. For confirmation of the presence of diketopiperazines NL-5 medium was used instead of DNPM medium (1 L NL-5 medium consists of NaCl 1.00 g, K2HPO4 1.00 g, MgSO4 × 7 H2O 0.50 g, glycerine 25.0 g, l-glutamine 5.84 g, trace-element-solution 2.00 mL, pH 7.3; trace-element-solution consists of ZnCl 40.0 mg, FeCl3 × 6 H2O 200 mg, CuCl2 × 2 H2O 10.0 mg, MnCl2 × 4 H2O, Na2B4O7 × 10 H2O 10.0 mg, (NH4)Mo7O24 × 4 H2O). For extraction the whole 100 mL cultures were stirred with 100 mL ethyl acetate for 30 min and were subsequently centrifuged (4000 rpm, 9 min) to separate biomass and aqueous phase from organic layer. The organic layer was concentrated under reduced pressure, transferred into 10 mL glass vials and evaporated using a sample concentrator with a light nitrogen flow and 40 °C. The samples were re-suspended in 500 µL (200-fold concentration) methanol.
Analysis of bacterial metabolites
Methanolic extracts were analysed by GC–MS (GC-system: Agilent 6890 Series; detector: Agilent 5973 Network Mass Selective Detector, Agilent Technologies; ionization: electron ionization with 70 eV ionization energy) using a fused silica capillary column (30 m × 0.25 mm) coated with 0.25 µm dimethyl polysiloxane (RTX®-1 MS, Restek) and helium as carrier gas with a flow rate of 1.0 mL per minute. The temperature profile started at 120 °C followed by a heating rate of 10 °C per minute to 270 °C, which was maintained for 15 min. The injector and detector temperatures were 280 °C, the injection volume was 1.0 μL and was split 10:1. Obtained mass spectra were compared with National Institute of Standards and Technology (NIST) 11 reference database and a self-assembled database of sesquiterpene lactones and their esters found in A. montana. This comparison led to propositions for molecules with the best matching fragmentation pattern. Further confirmation was performed by comparing the mass spectra with those available in literature (for actiphenol and diketopiperazines) or obtained with purchased reference substance (for cycloheximide, Sigma-Aldrich). Dereplication of results was conducted by UPLC-UV-HRESI-MS-system at LTQ Orbitrap (Thermo Scientific) using 18 min gradient starting with solvent B from 5 % up to 95 % (A: water + 0.1 % formic acid, B: CH3CN + 0.1 % formic acid) on Waters BEH C18 column (100 × 2.1, 1.7 µm). The flow was set to 0.6 mL per minute, column temperature to 45 °C and injection to 1 µL per sample). MS and UV data were analysed using Dictionary of Natural Products (DNP, CRD Press, Frances and Taylor).
Cell culture
Jurkat T cells obtained from DSMZ (Deutsche Sammlung von Mikroorganismen und Zellkulturen GmbH) were cultivated in Roswell Park Memorial Institute (RPMI) 1640 medium supplemented by 10 % heat-inactivated foetal bovine serum (both Gibco) and 1 mL penicillin/streptomycin solution (Roche Diagnostics). Cells were grown in a humidified atmosphere at 37 °C with 5 % CO2. Medium was renewed regularly.
MTT assay
For this colorimetric cell viability assay from Mosmann (1983) Jurkat T cells were seeded in 96 well plates (15,000 cells per well in 150 µL RPMI 1640 medium). Subsequently, the cells were incubated overnight in a humidified atmosphere at 37 °C with 5 % CO2. On the following day, the cells were incubated for 24 h with 10 or 50 µg of the Streptomyces extracts mL−1 dissolved in DMSO. For each condition three technical replicates were conducted. In the last 2 h of the incubation time the cells were additionally treated with 0.5 mg 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide (MTT, Roth) mL−1 in phosphate buffered saline (PBS). The obtained formazan crystals were dissolved in DMSO and the absorption of the solution was measured by a microplate reader (Bio-Rad) at 595 nm.
The values of the samples were referred to untreated cells as control. Viability was calculated as percentage of surviving cells compared with the mentioned untreated cells. 0.4 µg Act D mL−1 (Axxora) was used as positive control and 0.25 % DMSO as negative control. This viability test was performed twice.
Analysis of antibacterial activity
Antimicrobial susceptibility testing was performed by the disc diffusion test using the reproducible method of EUCAST (Matuschek et al. 2014). Three Gram-positive strains (Staphylococcus aureus (ATCC 29213), Enterococcus faecalis (ATCC 29212), Streptococcus pneumoniae (ATCC 49619) and three Gram-negative bacterial strains (Escherichia coli (ATCC 25922), Pseudomonas aeruginosa (ATCC 27583), Haemophilus influenzae (NCTC 8468) were tested. According to EUCAST guidelines the inoculum suspensions were adjusted to the density of a 0.5 McFarland standard. The inoculum suspension was then regularly spread over the agar plate using an automatic plate rotator. For S. aureus, E. faecalis, E. coli, and P. aeruginosa and Mueller–Hinton agar without supplements was utilised. Those strains were incubated at 35 °C for 16–20 h after adding the paper disks. Aberrantly S. pneumoniae and H. influenzae were grown on Mueller–Hinton agar with 5 % mechanically defibrinated horse blood and 20 mg β-nicotinamide adenine dinucleotide L−1 and kept at 35 °C with 5 % CO2 for 16–20 h after adding the testing disks.
The Streptomyces extracts were tested in concentrations of 10, 50, and 100 µg per disk. As negative control the solvent of the extracts, methanol, was applied in the highest used volume of 20 µL per disc. Different positive controls were used for the different strains, E. coli susceptibility with 10 µg of ampicillin, S. aureus and S. pneumoniae with one unit of penicillin, P. aeruginosa with 30 µg piperacillin, E. faecalis and H. influenzae with 2 µg ampicillin (all from BD Sensi-Disc™). In the case of extract 10 tested against S. aureus nalidixic acid (Sigma-Aldrich) was also analysed using concentrations of 15 and 30 µg. After incubation the zone diameter of the inhibition zones was measured with a calliper. This disc diffusion test was performed at least two times.
Analysis of antifungal properties
Extracts 2, 3, and 4 were tested for their effects on the fungi Candida parapsilosis (DSM 5784) and Fusarium verticillioides (DSM 62264) using the disc diffusion method. These fungal strains were grown in yeast medium (1 L of medium consists of 3 g yeast extract, 3 g malt extract, 5 g peptone from soybean and 10 g glucose). To prepare agar plates 20 g agar was used for 1 L of yeast medium. Methanol was used as negative control and 75 µg of nystatin as positive control. The inoculated plates were kept at 28 °C for 16–20 (C. parapsilosis) or 24–48 (F. verticillioides) hours before evaluation. The disc diffusion test was performed two times.
Genome sequencing and analysis
Genome sequencing of the cultivated strain was performed using Illumina MiSEQ technology. High molecular mass DNA was extracted from the selected strain, and two libraries were created: 8 kb mate-pair library, as well as a shorter-insert (~600 bp) shotgun paired-end library. Reads of an approximate length 2 × 250 (before trimming) were obtained for both libraries. Newbler software version 2.8 was used for genome assembly.
In the first step, gene finding was done using GISMO (Krause et al. 2007) followed by GenDB 2.0 automatic annotation (Meyer et al. 2003). In the second annotation step, all predicted ORFs were manually re-inspected to correct start codon and function assignments. For the identification of secondary metabolite clusters, the genome of Streptomyces sp. Arnica3 was scanned for homologues to known secondary metabolite synthases via BLAST search. These manual investigations were supported by antiSMASH (Medema et al. 2011).
16sDNA sequencing and phylogenetic analysis
Isolated strains 3 and 5 were grown in 10 ml of TSB media at 28 °C for 3 days and total DNA was isolated as described (Kieser et al. 2000). Amplification of the 16S rRNA gene was carried out with primers: 8F (AGAGTTTGATYMTGGCTCAG) and 1510R (TACGGYTACCTTGTTACGACTT). Obtained fragments were gel purified using QIAquick Gel Extraction Kit (Qiagen, Venlo, Netherlands) and sequenced with the amplification primers 8F and 1510R generating almost the entire gene sequence (1365 ~ 1401 bp). 31 rDNA sequences were aligned using MAFFT v7.017 (gap open penalty 1.53, offset value 0.123, scoring matrix 200PAM/k = 2, algorithm: auto) (Katoh et al. 2002). The dendrogram was built using Geneious (Kearse et al. 2012). The following settings were chosen: Tamura–Nei genetic distance model, neighbor-joining method, E. coli as an outgroup, bootstrap value 1000, consensus tree with 50 % support threshold. Tree branches were then proportionally transformed for display. Ribosomal RNA sequences for strains were extracted from genome annotations in GenBank.
Statistical analysis
Statistical analysis was performed with GraphPad Prism 5 software (GraphPad Software Inc.). Values are shown as mean ± standard deviation (SD). For comparison between groups analysis of variance (ANOVA) followed by Bonferroni’s post-test was used. p-values < 0.05 were considered as significant.
Results
Identification of secondary metabolites
The five extracts from the isolated actinobacteria were analysed by GC–MS and LC–MS. The diketopiperazines cyclo-prolyl-valyl, cyclo-leucyl-prolyl, and cyclo-isoleucyl-prolyl could be identified in all five extracts, whereas cycloheximide, actiphenol, and cyclo-prolyl-phenylalanyl only occurred in some of the extracts (see Table 1, for structures see Fig. 1). Cyclo-isoleucyl-prolyl and cyclo-prolyl-valyl were not present in the NIST11 database. Identification was done by comparison with published fragmentation patterns (Bitzer 2005; Wang et al. 2010; Ortiz-Castro et al. 2011). Only extract 3 contained all the mentioned molecules and the respective strain was therefore chosen for genome sequencing.
All found compounds were reported in the literature as metabolites produced by Streptomyces (Highet and Prelog 1959; Macherla et al. 2005; Pettit et al. 2006). Nevertheless, diketopiperazines are also known to exist in autoclaved peptone containing medium used for cultivation of Streptomyces (Tamura et al. 1964). This can be explained by non-enzymatic cyclisation of linear dipeptides at extreme temperatures (Skwierczynski and Connors 1993). Due to the usage of DNPM medium with soytone as amino acid source it was necessary to conduct further experiments to confirm the diketopiperazines as metabolites from Streptomyces. On the one hand a mock cultivation of DNPM medium without adding Streptomyces was performed, on the other hand the Streptomyces strains were cultivated in NL-5 medium, whose only amino acid is glutamine. The yielded extracts were analysed and revealed the presence of diketopiperazines after cultivation in NL-5 as well as in the mock extracts of DNPM medium. Therefore, it can be concluded that the identified diketopiperazines are present as artefact from DNPM medium but also as secondary metabolites produced by Streptomyces.
Viability reducing properties of the Streptomyces extracts
Due to their known cytotoxic properties (Otani et al. 1989; Lakhvich et al. 1993), the identification of cycloheximide and actiphenol in some of the Streptomyces extracts lead to the assumption that those extracts could have an impact on the viability of cancer cells. Therefore, we performed MTT assays using Jurkat T cells, an immortalised human T lymphocyte cell line. The cells were treated with either 0.4 µg Act D mL−1 as positive control, 0.25 % DMSO as negative control or with the Streptomyces extracts at 10 or 50 µg mL−1 concentrations. Extracts 2, 3, 4, and 5 exhibited a significant and concentration dependent reduction of cell viability (see Fig. 2). In contrast, no reduction of viability was observed with extract 1. In summary, all extracts containing either cycloheximide or actiphenol or both molecules showed significant viability reducing effects against Jurkat T cells.
Cell viability of Jurkat T cells after 24 h of treatment with Streptomyces extracts. Act D represents the positive control and DMSO the negative control. Streptomyces extracts were tested in two concentrations, 10 and 50 µg mL−1. Numbers 1 to 5 refer to the respective extracts. Data represent mean ± standard deviation (SD) of two independent experiments. ** p < 0.01 and *** p < 0.001 versus DMSO
Furthermore a caspase-3/-7 assay was performed for the most cytotoxic extracts 2 and 3 to determine whether the occurring cell death is caused by apoptosis (data not shown). The test showed no marked increase in caspase-3/-7 activity, indicating that this mechanism of cell death does not play any role.
Antibacterial activity of the Streptomyces extracts
Antibacterial properties are often found for metabolites from Streptomyces. Therefore, we investigated the effect of the extracts on six common bacterial test strains. Because three Gram-positive and also three Gram-negative strains with varying characteristics were used, this would provide a reasonable overview on the antibacterial activity of the examined extracts. Only extract 4 showed a slight concentration dependent activity against S. aureus, one of the Gram-positive bacterial strains. This effect manifested itself in a zone around the test disks with a diffuse decline in bacterial population compared to the rest of the inoculated and incubated agar plate. The same situation could be shown using 15 µg of nalidixic acid, to which S. aureus is resistant (Ball 2000). In contrast, one unit of penicillin leads to a more definite inhibition zone (Fig. 3). Hence it can be stated that there is a slight activity of extract 4 against S. aureus, but that the presence of colonies of S. aureus in the inhibition zone may imply a development of resistance as it is the case for nalidixic acid.
Antibacterial activity against S. aureus measured by disc diffusion test. Disk number 1 contained penicillin one unit, 2 the solvent control methanol, 3–5 extract 4 in the concentrations of 50 (3), 100 (4) and 150 µg (5), and 6–7 nalidixic acid in the concentrations of 15 (6) and 30 µg (7). A representative susceptibility test is shown
Additionally to the Streptomyces extracts, 100 µg of the mock extract containing DNPM medium without Streptomyces, which also comprised diketopiperazines, was tested against S. aureus. No zone of inhibition could be detected.
Antifungal activity of the Streptomyces extracts
Cycloheximide is known for its antifungal effect (Sharma et al. 2010). Therefore, the cycloheximide containing extracts 2 and 3 were tested for their antifungal properties in comparison with cycloheximide-lacking extract 4. As test strains the pathogenic yeast C. parapsilosis, which can be found in soil, and the mold fungus Fusarium verticillioides, a fungus known to inhabit soil and plants, were used (Brown et al. 2006; Nosek et al. 2009). As presumed extracts 2 and 3 but not 4 showed growth-inhibitory activity against C. parapsilosis and F. verticillioides (Table 2).
Genome analysis
One of the isolated Streptomyces strains producing extract 3 was sequenced as described in “Materials and Methods”, resulting in 7 contigs, the longest of which (9206603 bp) contains 96 % of all the sequence data and is one of the largest sequenced Streptomyces genomes. Judging by the genome size, this Streptomyces sp. is probably well-fit for highly-variable nutrient and environmental conditions. This genome contains 8319 predicted protein coding sequences (yielding genome coding density of 85.7 %), 87 tRNAs, 1 tmRNA, 23 other RNAs, and 39 putative secondary metabolite clusters. GC content of this genome is 70.6 %, which is common for Streptomyces.
Of the 39 putative secondary metabolite clusters, 1 was identified for siderophore, 1 for ectoine, 7 for terpene, 8 (2 per type) of bacteriocin, butyrolactone, lantipeptide and melanin, nine NRPS, four PKS, and nine clusters of other and hybrid types.
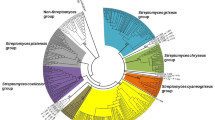
Finally, we decided to check if the sequenced strain has genes for cycloheximide production. We have used the cycloheximide biosynthesis cluster sequence from GenBank, accession number JX014302.1 (Streptomyces sp. YIM 56141). We succeeded in finding all ten ChxA-ChxJ genes. Average gene-level nucleotide identity to JX014302.1 is 79 % with sequence coverage in 92–100 % range (average 97 %). In addition, we have performed the phylogenetic analysis of two isolated strains using 16S ribosomal RNA encoding genes. Based on the 16S phylogeny, the two isolated Streptomyces sp. are evolutionary closed to each other (Fig. 4), and they are the most similar to Streptomyces avidinii and Streptomyces spororaveus, followed by Streptomyces fulvissimus and Streptomyces griseus subsp. griseus.
Discussion
Streptomyces produce a huge variety of bioactive molecules, including approximately 55 % of all clinically useful antibiotic drugs (Demain 1999). Biosynthesis of secondary metabolites in Streptomyces is dependent on many environmental criteria. Those criteria include temperature, light, pH, concentration of phosphate and oxygen and the amount and kind of carbon and nitrogen source (Ruiz et al. 2010). Therefore it is attractive to examine those bacterial species inhabiting environments with unusual ecological niches. One of those ecological niches is occupied by A. montana, a plant which survives in a nutrient-poor hostile environment. Soil is known to be a harsh, competitive environment for microorganisms displaying extreme temperatures, moisture, and poor nutrient (Loria et al. 2006). This pertains especially in the mountainous environment without the massive usage of fertilizers, where A. montana is growing preferably (Sugier et al. 2013). It is not surprising that Streptomyces inhabiting this area search for an ecological niche which is able to provide protection as well as a carbon and energy source. Former studies already revealed the coexistence of A. montana with arbuscular mycorrhizal fungi (Ryszka et al. 2010), but no reports exist about the discovery of a further inhabitant, Streptomyces, in the medicinally used plant of A. montana.
As nutrient source the roots of A. montana contain carbohydrates such as inulin, glucose, fructose and sucrose. Free amino acids are available as synthesis precursors. Beyond that, a large variety of protective molecules are produced in the roots to keep competitor microorganisms away. Compounds with antibacterial activity are polyacetylenic compounds, essential oils, phenolic ethers and esters. The polyacetylenic molecules additionally provide an antifungal effect. Phenolic acids add an antioxidative facet (Rossetti et al. 1984; Teuscher et al. 2009; Jurkiewicz et al. 2010; Pljevljakušić et al. 2012). Consequently, the benefits for endophytes inhabiting of A. montana are apparent.
We found that Streptomyces detected in A. montana produce several metabolites which can be beneficial for the plant itself. Cycloheximide can be useful to the host plant as an antifungal and antiviral compound (Ji et al. 2010). The antifungal activity could be confirmed by our studies as only cycloheximide-containing extracts 2 and 3, but not cycloheximide-lacking extract 4, showed a growth-inhibitory effect against C. parapsilosis and F. verticillioides. Antifungal effects of cycloheximide against other species of the genera Candida and Fusarium have already been described (Sharma et al. 2010). Both fungi are present in soil and Fusarium is additionally known to be an important plant pathogen (Brown et al. 2006; Nosek et al. 2009). This emphasizes the possible importance of Streptomyces producing cycloheximide to protect the host plant against pathogenic fungi. Actiphenol, which has a similar chemical structure, is also known to exhibit an antifungal and antiviral activity as well. Moreover it is proposed to act as a herbicidal agent (Fukuda et al. 2005; Ji et al. 2010). The identified diketopiperazines and the glutarimide antibiotics have been found before in Streptomyces (Highet and Prelog 1959; Macherla et al. 2005; Pettit et al. 2006). The role of the different diketopiperazines is multi-layered. The antibacterial activity, which is frequently found in secondary metabolites of Streptomyces in order to inhibit competitors (Diminic et al. 2014), is heterogeneously reported. Cyclo-prolyl-valyl exerted no activity against S. aureus, B. subtilis and E. coli (Effendi 2004), but all four diketopiperazines found in our extracts are reported to be active against Vibrio anguillarum (Fdhila et al. 2003). Cyclo-leucyl-prolyl is reported to inhibit the growth of Mycobacterium marinum, E. faecalis, B. subtilis and S. aureus (Rhee 2002; Effendi 2004; Martins and Carvalho 2007). Notwithstanding, another group could not show activity of this molecule against B. subtilis and E. coli (Cabrera et al. 2006). The most active among our four diketopiperazines seems to be cyclo-prolyl-phenylalanyl. A moderate activity against six bacterial strains was already shown for this molecule, among them E. coli, P. aeruginosa, S. aureus and S. pneumoniae (Milne et al. 1998). However, we could not find a pronounced antibacterial effect in our Streptomyces extracts containing diketopiperazines except the weak growth reduction of S. aureus. This small effect could be explained due to the reported activity of cyclo-prolyl-phenylalanyl (Milne et al. 1998). It is conceivable that the concentration of these moderately antibacterial acting molecules in our extracts is too low to exert more inhibitory activity. Perhaps the low concentration used could also explain why we did not find an activity against S. aureus by the diketopiperazine-containing DNPM medium, which was also tested. Beyond that, it is probably not necessary for the Streptomyces strains inhabiting A. montana to produce more active antibacterial compounds due to the presence of above-mentioned molecules, who accomplish this function in the A. montana root (Jurkiewicz et al. 2010). Other reported activities of the diketopiperazines comprise an antifungal effect of cyclo-leucyl-prolyl and cyclo-prolyl-phenylalanyl, and a phytotoxic effect of cyclo-prolyl-phenylalanyl against certain plants (Holden et al. 1999; Wang et al. 1999; Klose 2006). The most interesting diketopiperazines regarding the interaction with A. montana are cyclo-prolyl-valyl and cyclo-prolyl-phenylalanyl. In addition to the mentioned antibacterial, antifungal and phytotoxic properties, those molecules seem to promote plant growth. Especially cyclo-prolyl-phenylalanyl is discussed recently to act as a signalling mimic of the plant hormone auxin (Ortiz-Castro et al. 2011; Venturi and Fuqua 2013).
Interestingly, our presumption that these Streptomyces could possibly produce sesquiterpene lactones could not be confirmed. Thus, A. montana, but not the endophytic Streptomyces strains, seems to be the producer of those medicinally effective compounds.
Complementary to the structure identification and examination of the antibacterial and antifungal properties, we tested the impact of the Streptomyces extracts on the viability of Jurkat T cells, a certain sort of T lymphocyte-like cancer cell line. Only those Streptomyces extracts which contain cycloheximide and or actiphenol (2, 3, 4 and 5) revealed a concentration dependent reduction in cell viability. Therefore, these extracts can be described as cytotoxic. Both identified glutarimide antibiotics are known to act as cytotoxic substances (Otani et al. 1989; Lakhvich et al. 1993). This reinforces the presumption that those two compounds could be responsible for this activity. Actinobacteria isolated from other members of the Asteraceae family (Parthenium hysterophorus, Ageratum conyzoides, Sonchus oleraceus, S. asper and Hieracium canadense) were also shown to produce highly cytotoxic substances, but their structures were not elucidated (Tanvir et al. 2014).
In summary, it seems conceivable that these two organisms interact on the one hand to complete their protection against competitors and pathogens and on the other hand to exchange plant growth promoting signals with a carbon and energy source for the microorganism. In our case A. montana is highly likely to contribute to this community by offering carbon and synthesis precursors to the Streptomyces and protective substances for the defense against competing bacteria and fungi. The Streptomyces strains in return could augment the protection against pathogenic fungi by producing cycloheximide and possibly support the growth of A. montana by producing cyclo-prolyl-phenylalanyl. These observations suggest this symbiosis facilitates life in this ecological niche of a nutrient-poor and adverse environment. The genomic data of the sequenced strain 3 suggest the huge biosynthetic potential of endophytic bacteria. The number of natural products gene clusters is higher than those of model Streptomyces strains such as Streptomyces coelicolor, Streptomyces lividans or Streptomyces albus. Therefore, the exploitation of endophytic actinobacteria might have a great impact on drug discovery.
Abbreviations
- Act D:
-
Actinomycin D
- ANOVA:
-
Analysis of variance
- EUCAST:
-
European Committee on Antimicrobial Susceptibility Testing
- ESCOP:
-
European Scientific Cooperative on Phytotherapy
- HRESI:
-
High-resolution electrospray ionization
- NIST:
-
National Institute of Standards and Technology
- UPLC:
-
Ultra performance liquid chromatography
References
Ball P (2000) Quinolone generations: natural history or natural selection? J Antimicrob Chemother 46:17–24
Bérdy J (2005) Bioactive microbial metabolites. J Antibiot (Tokyo) 58:1–26
Bitzer J (2005) Isolierung und Strukturaufklärung neuer Naturstoffe aus Bakterien und endophytischen Pilzen durch chemisches Screening. Disseration, University of Göttingen
Brown DW, Butchko RAE, Proctor RH (2006) Fusarium genomic resources: tools to limit crop diseases and mycotoxin contamination. Mycopathologia 162:191–199
Cabrera GM, Butler M, Rodriguez MA, Godeas A, Haddad R, Eberlin MN (2006) A sorbicillinoid urea from an intertidal Paecilomyces marquandii. J Nat Prod 69:1806–1808. doi:10.1021/np060315d
Castillo UF, Browne L, Strobel G, Hess WM, Ezra S, Pacheco G, Ezra D (2007) Biologically active endophytic streptomycetes from Nothofagus spp. and other plants in Patagonia. Microb Ecol 53:12–19. doi:10.1007/s00248-006-9129-6
Coombs JT, Franco CMM (2003) Isolation and identification of actinobacteria from surface-sterilized wheat roots. Appl Environ Microbiol 69:5603–5608. doi:10.1128/AEM.69.9.5603-5608.2003
Demain AL (1999) Pharmaceutically active secondary metabolites of microorganisms. Appl Microbiol Biotechnol 52:455–463
Diminic J, Starcevic A, Lisfi M, Baranasic D, Gacesa R, Hranueli D, Long PF, Cullum J, Zucko J (2014) Evolutionary concepts in natural products discovery: what Actinomycetes have taught us. J Ind Microbiol Biotechnol 41:211–217. doi:10.1007/s10295-013-1337-8
Effendi H (2004) Isolation and structure elucidation of bioactive secondary metabolites of sponge-derived fungi collected from the Mediterranean sea (Italy) and Bali sea (Indonesia). Dissertation, University of Düsseldorf
ESCOP (2003) ESCOP monographs: the scientific foundation for herbal medicinal products, 2nd edn. Thieme, Stuttgart
Fdhila F, Vázquez V, Sánchez JL, Riguera R (2003) dd-diketopiperazines: antibiotics active against Vibrio anguillarum isolated from marine bacteria associated with cultures of Pecten maximus. J Nat Prod 66:1299–1301. doi:10.1021/np030233e
Fukuda T, Matsumoto A, Takahashi Y, Tomoda H, Omura S (2005) Phenatic acids A and B, new potentiators of antifungal miconazole activity produced by Streptomyces sp. K03-0132. J Antibiot (Tokyo) 58:252–259. doi:10.1038/ja.2005.29
García-Piñeres AJ, Castro V, Mora G, Schmidt TJ, Strunck E, Pahl HL, Merfort I (2001) Cysteine 38 in p65/NF-kappaB plays a crucial role in DNA binding inhibition by sesquiterpene lactones. J Biol Chem 276:39713–39720. doi:10.1074/jbc.M101985200
Highet RJ, Prelog V (1959) Stoffwechselprodukte von Actinomyceten. 18. Mitteilung. Actiphenol. Helv Chim Acta 42:1523–1526. doi:10.1002/hlca.19590420514
Holden MT, Ram Chhabra S, de Nys R et al (1999) Quorum-sensing cross talk: isolation and chemical characterization of cyclic dipeptides from Pseudomonas aeruginosa and other gram-negative bacteria. Mol Microbiol 33:1254–1266
Ji X, Zhong Z, Xue S, Meng S, He W, Gao R, Li Y, Li Z (2010) Synthesis and antiviral activities of synthetic glutarimide derivatives. Chem Pharm Bull (Tokyo) 58:1436–1441
Jiménez-Esquilín AE, Roane TM (2005) Antifungal activities of actinomycete strains associated with high-altitude sagebrush rhizosphere. J Ind Microbiol Biotechnol 32:378–381. doi:10.1007/s10295-005-0007-x
Jurkiewicz A, Ryszka P, Anielska T, Waligórski P, Białońska D, Góralska K, Tsimilli-Michael M, Turnau K (2010) Optimization of culture conditions of Arnica montana L.: effects of mycorrhizal fungi and competing plants. Mycorrhiza 20:293–306. doi:10.1007/s00572-009-0280-z
Katoh K, Misawa K, Kuma K, Miyata T (2002) MAFFT: a novel method for rapid multiple sequence alignment based on fast Fourier transform. Nucleic Acids Res 30:3059–3066. doi:10.1093/nar/gkf436
Kearse M, Moir R, Wilson A et al (2012) Geneious basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 28:1647–1649. doi:10.1093/bioinformatics/bts199
Kieser T, Bibb MJ, Buttner MJ, Chater KF, Hopwood DA (2000) Practical Streptomyces genetics. The John Innes Foundation, Norwich
Klose KE (2006) Increased chatter: cyclic dipeptides as molecules of chemical communication in Vibrio spp. J Bacteriol 188:2025–2026. doi:10.1128/JB.188.6.2025-2026.2006
Krause L, McHardy AC, Nattkemper TW, Pühler A, Stoye J, Meyer F (2007) GISMO-gene identification using a support vector machine for ORF classification. Nucleic Acids Res 35:540–549. doi:10.1093/nar/gkl1083
Lakhvich FA, Buravskaya TN, Akhrem AA (1993) Synthesis of actiphenol and its normethyl analogues and 6-hydroxy derivatives. Chem Nat Compd 29:526–529
Lee K-H, Ibuka T, Wu R-Y, Geissman TA (1977) Structure-antimicrobial activity relationships among the sesquiterpene lactones and related compounds. Phytochemistry 16:1177–1181
Lin L, Ge HM, Yan T, Qin YH, Tan RX (2012) Thaxtomin A-deficient endophytic Streptomyces sp. enhances plant disease resistance to pathogenic Streptomyces scabies. Planta 236:1849–1861. doi:10.1007/s00425-012-1741-8
Loria R, Kers J, Joshi M (2006) Evolution of plant pathogenicity in Streptomyces. Annu Rev Phytopathol 44:469–487. doi:10.1146/annurev.phyto.44.032905.091147
Lyss G, Schmidt T, Merfort I, Pahl H (1997) Helenalin, an anti-inflammatory sesquiterpene lactone from Arnica, selectively inhibits transcription factor NF-kappaB. Biol Chem 378:951–961
Macherla VR, Liu J, Bellows C, Teisan S, Nicholson B, Lam KS, Potts BCM (2005) Glaciapyrroles A, B, and C, pyrrolosesquiterpenes from a Streptomyces sp. isolated from an Alaskan marine sediment. J Nat Prod 68:780–783. doi:10.1021/np049597c
Martins MB, Carvalho I (2007) Diketopiperazines: biological activity and synthesis. Tetrahedron 63:9923–9932. doi:10.1016/j.tet.2007.04.105
Matuschek E, Brown DFJ, Kahlmeter G (2014) Development of the EUCAST disk diffusion antimicrobial susceptibility testing method and its implementation in routine microbiology laboratories. Clin Microbiol Infect 20:O255–O266. doi:10.1111/1469-0691.12373
Medema MH, Blin K, Cimermancic P, De Jager V, Zakrzewski P, Fischbach MA, Weber T, Takano E, Breitling R (2011) AntiSMASH: rapid identification, annotation and analysis of secondary metabolite biosynthesis gene clusters in bacterial and fungal genome sequences. Nucleic Acids Res 39:339–346. doi:10.1093/nar/gkr466
Merfort I (2010) Arnika—aktueller Stand hinsichtlich Wirksamkeit, Pharmakokinetik und Nebenwirkungen. Z Phyther 31:188–192. doi:10.1055/s-0030-1262391
Meyer F, Goesmann A, McHardy AC et al (2003) GenDB—an open source genome annotation system for prokaryote genomes. Nucleic Acids Res 31:2187–2195. doi:10.1093/nar/gkg312
Milne PJ, Hunt AL, Rostoll K, Van Der Walt JJ, Graz CJ (1998) The biological activity of selected cyclic dipeptides. J Pharm Pharmacol 50:1331–1337
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63
Nosek J, Holesova Z, Kosa P, Gacser A, Tomaska L (2009) Biology and genetics of the pathogenic yeast Candida parapsilosis. Curr Genet 55:497–509. doi:10.1007/s00294-009-0268-4
Ortiz-Castro R, Díaz-Pérez C, Martínez-Trujillo M, del Río RE, Campos-García J, López-Bucio J (2011) Transkingdom signaling based on bacterial cyclodipeptides with auxin activity in plants. Proc Natl Acad Sci USA 108:7253–7258. doi:10.1073/pnas.1006740108
Otani T, Sasaki T, Minami Y, Marunaka T, Yu QW (1989) New glutarimide antibiotics, S-632-B1 and B2. I. Taxonomy of producing strain, fermentation and biological properties. J Antibiot (Tokyo) 42:647–653
Pettit GR, Du J, Pettit RK, Richert LA, Hogan F, Mukku VJRV, Hoard MS (2006) Antineoplastic agents. 554. The manitoba bacterium Streptomyces sp. J Nat Prod 69:804–806. doi:10.1021/np058087v
Ph.Eur. (2012) Arnikablüten. In: Eur. Arzneib., 7.3 edn. DAV, Stuttgart, pp 5602–5604
Pljevljakušić D, Rančić D, Ristić M, Vujisić L, Radanović D, Dajić-Stevanović Z (2012) Rhizome and root yield of the cultivated Arnica montana L., chemical composition and histochemical localization of essential oil. Ind Crops Prod 39:177–189. doi:10.1016/j.indcrop.2012.02.030
Porras-Alfaro A, Bayman P (2011) Hidden fungi, emergent properties: endophytes and microbiomes. Annu Rev Phytopathol 49:291–315. doi:10.1146/annurev-phyto-080508-081831
Rhee K-H (2002) Isolation and characterization of Streptomyces sp KH-614 producing anti-VRE (vancomycin-resistant enterococci) antibiotics. J Gen Appl Microbiol 48:321–327
Rossetti V, Lombard A, Sancin P, Buffa M, Di Stefano R (1984) Characterization of Arnica montana L. dried roots. Int J Crude Drug Res 22:53–60
Ruiz B, Chávez A, Forero A et al (2010) Production of microbial secondary metabolites: regulation by the carbon source. Crit Rev Microbiol 36:146–167. doi:10.3109/10408410903489576
Ryszka P, Błaszkowski J, Jurkiewicz A, Turnau K (2010) Arbuscular mycorrhiza of Arnica montana under field conditions-conventional and molecular studies. Mycorrhiza 20:551–557. doi:10.1007/s00572-010-0302-x
Sardi P, Saracchi M, Quaroni S, Petrolini B, Borgonovi GE, Merli S (1992) Isolation of endophytic Streptomyces strains from surface-sterilized roots. Appl Environ Microbiol 58:2691–2693
Sharma N, Thakur M, Chaudhry SC (2010) Monooxovanadium(V) 2-phenylphenoxides: synthesis, characterization, and antimicrobial potential. J Coord Chem 63:1071–1079. doi:10.1080/00958971003710377
Skwierczynski R, Connors K (1993) Demethylation kinetics of aspartame and l-phenylalanine methyl ester in aqueous solution. Pharm Res 10:1174–1180
Stierle A, Strobel G, Stierle D (1993) Taxol and taxane production by Taxomyces andreanae, an endophytic fungus of Pacific yew. Science 260:214–216. doi:10.1126/science.8097061
Sugier D, Sugier P, Gawlik-Dziki U (2013) Propagation and introduction of Arnica montana L. into cultivation: a step to reduce the pressure on endangered and high-valued medicinal plant species. Sci World J. doi:10.1155/2013/414363
Tamura S, Suzuki A, Aoki Y, Otake N (1964) Isolation of several diketopiperazines from peptone. Agric Biol Chem 28:650–652. doi:10.1271/bbb1961.28.650
Tanvir R, Sajid I, Hasnain S (2014) Biotechnological potential of endophytic actinomycetes associated with Asteraceae plants: isolation, biodiversity and bioactivities. Biotechnol Lett 36:767–773. doi:10.1007/s10529-013-1430-0
Teuscher E, Willuhn G, Loew D (2009) Arnicae flos. In: Wichtl M (ed) Teedrogen und Phytopharmaka, 5th edn. WVG, Stuttgart, pp 91–95
Venturi V, Fuqua C (2013) Chemical signaling between plants and plant-pathogenic bacteria. Annu Rev Phytopathol 51:17–37. doi:10.1146/annurev-phyto-082712-102239
Wang Y, Mueller UG, Clardy J (1999) Antifungal diketopiperazines from symbiotic fungus of fungus-growing ant Cyphomyrmex minutus. J Chem Ecol 25:935–941
Wang J-H, Quan C-S, Qi X-H et al (2010) Determination of diketopiperazines of Burkholderia cepacia CF-66 by gas chromatography-mass spectrometry. Anal Bioanal Chem 396:1773–1779. doi:10.1007/s00216-009-3379-3
Acknowledgments
The authors thank Barbara Schuler for her help regarding GC–MS implementation.
Compliance with ethical standards
This article does not contain any studies with human participants and animals performed by any of the authors.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wardecki, T., Brötz, E., De Ford, C. et al. Endophytic Streptomyces in the traditional medicinal plant Arnica montana L.: secondary metabolites and biological activity. Antonie van Leeuwenhoek 108, 391–402 (2015). https://doi.org/10.1007/s10482-015-0492-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-015-0492-5