Abstract
Research into the human placenta’s complex functioning is complicated by a lack of suitable physiological in vivo models. Two complementary approaches have emerged recently to address these gaps in understanding, computational in silico techniques, including multi-scale modeling of placental blood flow and oxygen transport, and cellular in vitro approaches, including organoids, tissue engineering, and organ-on-a-chip models. Following a brief introduction to the placenta’s structure and function and its influence on the substantial clinical problem of preterm birth, these different bioengineering approaches are reviewed. The cellular techniques allow for investigation of early first-trimester implantation and placental development, including critical biological processes such as trophoblast invasion and trophoblast fusion, that are otherwise very difficult to study. Similarly, computational models of the placenta and the pregnant pelvis at later-term gestation allow for investigations relevant to complications that occur when the placenta has fully developed. To fully understand clinical conditions associated with the placenta, including those with roots in early processes but that only manifest clinically at full-term, a holistic approach to the study of this fascinating, temporary but critical organ is required.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Human reproduction is a critical element affecting women’s health but is a relatively understudied phenomenon within the biomedical sciences and engineering. Pregnancy involves dramatic physiologic changes within a relatively short timeframe, from embryo implantation, through gestation, to delivery and parturition. There are apparent bioengineering factors in many aspects of pregnancy, including the physical properties of uterine tissues, blood flow and oxygen diffusion in and around the placenta, and the considerations of how and when the fetus exits the womb. These topics have been the subject of research since the mid-1800s.33,35 There have also been significant advances in the development of medical device technologies to save preterm babies’ lives, such as the incubator and infant ventilator. As a result, the limit of viability for premature infants has decreased by several weeks gestation, and the associated morbidity has also declined.31
Human pregnancies are considered full term at approximately 40 weeks gestation, and preterm birth is defined as occurring prior to 37 weeks gestation. Approximately one in ten births worldwide is preterm,95 although the preterm birth rate can vary substantially from country to country,95 or even more locally, such as from state to state within the US17 or from country to country within sub-Saharan Africa.95 Local variations are due to race, poverty, healthcare infrastructure, maternal education, and access to health insurance, but preterm birth rates are currently rising in most countries.95 Racial disparities are particularly stark in the US,28 where maternal stress has been implicated as a significant factor.38
Preterm birth is categorized by the completed weeks of gestation (Table 1) with more severe consequences for pregnancies that deliver much earlier than the milestone of 37 weeks gestational age (GA). The survival rate for preterm babies varies geographically, with a lower survival rate at more advanced GA in low-income countries compared to high-income countries.95 Preterm birth can have many different contributing causes, but these are categorized roughly as (1) maternal; (2) fetal; (3) placental; (4) associated with early labor; and (5) medically indicated.94 An intriguing feature of many placenta-related preterm births is that early placental development problems do not manifest fully until much later in the gestational period. Other forms of placenta-associated preterm birth, such as those due to injury, may occur later in an otherwise normally progressing pregnancy.
In this review, bioengineering approaches for studying the placenta and placenta-related preterm birth are considered within a holistic framework from cell to organ. To better understand preterm birth related to the placenta, we must first briefly examine the placenta’s origin, development, and structure. Next, we examine specific pregnancy complications related to the placenta. Bioengineering techniques for placental investigation are divided into three categories: (1) tissue engineering and organoids; (2) microfluidic devices; (3) computational modeling studies across length-scales. Finally, the current state of the art and outlook for future research needs are considered.
The Placenta
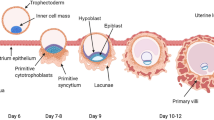
The placenta is a temporary organ that exists only during pregnancy to nourish and support the developing fetus. A fertilized egg after several cell division cycles becomes an early blastocyst, with cells of two primary types, the inner cell mass and the trophectoderm. At implantation, trophoblast cells derived from the blastocyst invade the uterine lining (endometrium) (Fig. 1a). The trophoblast cells remodel the maternal spiral arteries and convert them into highly dilated vessels capable of providing sufficient nutrients and oxygen to the fetus.65 The trophectoderm layer (Fig. 1a) develops into the chorion layer of the fetal membranes and the fetal components of the placenta (Figs. 1b and 1c). The maternal decidua—the transformed endometrial layer of the uterus—is categorized by its geometrical relationship with the implanting blastocyst. The decidua basalis (or placentalis) is at the site of implantation and the decidua parietalis is distant from the implantation site. The placental disc traverses the fetal chorion and maternal decidua basalis (Fig. 1c). The highly specialized cytotrophoblast cells and syncytiotrophoblast derive from the trophectoderm layer, and these trophoblast cells are responsible for the critical functions of the placenta.90 The “blueprint” for the eventual placental structure is established by the end of the first trimester.90
(a) The implanting blastocyst (trophectoderm, inner cell mass) at implantation into the maternal (endometrium) uterine tissue contrasted with (b, c) the late pregnancy fetus within the uterus including extra-embryonic tissues: the fetal membranes and placenta. The color scheme in part (a) is maintained, to show the origin of amnion, chorion and decidual tissues in parts (b) and (c). The placenta is fetal trophoblast-derived but traverses the maternal decidua.
The normal third-trimester placenta has a fully formed and sophisticated interface between the open maternal circulation and the closed fetal circulation at the placental terminal villi (Fig. 2), the tree-like structures that form the exchange surface between maternal and fetal blood. In these villi, oxygen and nutrients are transported from the maternal open circulation, which pools blood in the intervillous space (Fig. 2), to the fetus across the trophoblastic tissue layer. Placental oxygen transport occurs passively and can be described as a six-step process,63 in which the oxygen (1) dissociates from maternal red blood cells, (2) diffuses through maternal plasma, (3) diffuses across the trophoblastic epithelium, (4) diffuses across the basal lamina, villus stroma and endothelium, (5) diffuses through fetal plasma, and finally (6) binds to the hemoglobin (Hb) of fetal erythrocytes. Steps (3) and (4) in this sequence are often combined and the composite tissue classed as the “villous membrane”63 (Fig. 2). Therefore, oxygen transport across the placenta depends on the diffusing capacity of oxygen in each medium, on the placental surface area, placental oxygen consumption, maternal-placental blood flow, the oxygen content of maternal arterial blood, oxygen affinity of maternal blood, fetoplacental blood flow, oxygen affinity of fetal blood, and the directions of both bloodstreams.19 In contrast to oxygen transport, which is mostly passive and dominated by blood flow, transport of glucose and amino acids from mother to fetus relies on transporter proteins and is significantly affected by placental uptake of the nutrients.15 Recent works have included computational models for more detailed placental transport kinetic studies of oxygen and solutes37 and for quantifying the balance between transport and tissue metabolism.56
Highly schematized illustration of the critical exchange unit in the placenta. The enclosed feto-placental capillary ‘trees’ (placental terminal villi) meet the open maternal circulation, via blood pooled in the intervillous space. Diffusion of oxygen occurs across the composite tissue known as the “villous membrane”.63
Trophoblast cells have a complex early task in the first trimester of pregnancy, to help remodel the maternal uterine decidua to form the placenta. Although invasion of the uterus by trophoblast cells plays a critical role in reproductive outcome, limited understanding of the mechanisms controlling trophoblast invasion remains a significant obstacle to progress in pregnancy research.50 Learning more about what factors influence trophoblast invasion and the cellular and molecular basis of the invasion pathway is essential for understanding placental development. A significant challenge in pregnancy research arises in that late pregnancy placenta-related complications in the third trimester of gestation are rooted in developmental issues that occur in the first trimester,16 preventing the normal development of the maternal-fetal interface (Fig. 2). The placenta’s unusual status as a temporary organ, and one that changes continuously throughout the pregnancy, further complicates its study. Researchers have direct access to study the placenta only at the end of pregnancy, whether that is term or preterm gestation. This limits the information available to researchers to elucidate the essential functions of placental development earlier in gestation.70 However, before we consider how bioengineering approaches are starting to allow for better study of the placenta across all stages of development, but particularly in the early stages, we next consider the clinical complications of pregnancy that motivate this further study.
Placental Complications of Pregnancy
A significant fraction of preterm births are associated with pathologies of the placenta, including those medically indicated deliveries required to remove the placenta to preserve the mother’s life. Insufficient trophoblast invasion in the first trimester leads to deficient spiral artery remodeling. This is a significant factor associated with severe pregnancy disorders, including subsequent failure to form the deep placental bed and the placental-uterine interface. These failures are associated with the “great obstetrical syndromes” of pre-eclampsia (PE), fetal growth restriction (FGR), placental abruption, preterm labor, and stillbirth.16 This failure of trophoblast invasion has led to an interest in studying the process from a cellular biomechanics perspective.2 The opposite case, of trophoblast invasion that is too deep instead of too shallow, is related to the trophoblastic cancer choriocarcinoma,48 which spreads easily from the uterus to the lungs. Although invasion of the uterus by trophoblast cells plays a critical role in reproductive outcome, limited understanding of the mechanisms controlling trophoblast invasion remains an obstacle to progress in pregnancy research.50,101 Learning more about what factors influence trophoblast invasion and the cellular and molecular basis of the invasion pathway is essential for understanding the sequelae of poor placental development later in the pregnancy.
Pre-eclampsia (PE), which affects 5-7% of all pregnancies, is a leading cause of maternal and fetal morbidity and mortality.77 Eclampsia is a complication of severe PE and is associated with seizures and even coma. PE is unique because it has such profound implications for women’s health that last even beyond pregnancy.77 The hallmarks of PE are elevated blood pressure (hypertension) in a previously normotensive patient, along with kidney- and liver-related symptoms. The condition is thus systemic in the pregnant person, even though its origin is in the placenta. In many cases, the only way to protect the mother’s health from PE and eclampsia is to deliver the fetus and placenta by C-section no matter the gestational age. Removing the placenta leads to rapid improvement in many maternal symptoms, although there are still risks of postpartum hypertension and severe cardiovascular complications. Prior PE is also a risk factor in future pregnancies.77
Fetal growth restriction (FGR), also known as intrauterine growth restriction (IUGR), affects 5-10% of all pregnancies.66 It is a leading cause of perinatal mortality and is responsible for 30% of stillbirths. FGR has a distinct placental pathogenesis, which differentiates it from other small-for-gestational-age fetuses. In FGR, the idiopathic placental insufficiency can appear either early in pregnancy, with a greater risk of a wide range of fetal morbidities including preterm birth and fetal mortality, or in late pregnancy (> 32 weeks GA), with a high risk of stillbirth. The sequelae can be due to placental insufficiency alone or coupled with maternal malnutrition in developing countries.86 There is no obvious maternal presentation for FGR without knowledge of the pregnancy’s GA; both the fetus and placenta are small for gestational age and thus appear associated with a less advanced pregnancy.86 The FGR placenta has poorly developed vascular branching structures and an overall smaller diffusion surface at the maternal-fetal interface, likely due to deficiencies in trophoblast function.86 The lasting effects of FGR for the fetus can present as cardiovascular complications in childhood and even in adulthood.60
Placenta accreta syndrome (PAS) results when the placental trophoblast develops abnormal, invasive adherence to the uterine myometrium, particularly into scar tissue from a previous C-section, such that the placenta is not restricted to the uterine decidua. The incidence of this condition has increased with the increase in surgical deliveries, although it is still relatively rare at less than 1% of births.12 It is frequently associated with placenta previa, when the placenta is “low” in the uterus to the point of partially or fully covering the cervix. PAS is subdivided into three categories depending on the extent of invasiveness, from accreta (placental invasion through the endometrium) to increta (placenta invasion into the muscular myometrium layer of the uterus) and finally percreta (placenta traverses through the uterine wall and affects other tissues such as the bladder).12 The danger of PAS is failure of the placental detachment at delivery, resulting in hemorrhage and the potential need for hysterectomy to prevent blood loss and further damage. PAS is therefore often associated with medically indicated delivery several weeks before full-term gestation.13
Placental abruption (PA) is the detachment of the placenta from the uterine wall prior to delivery. PA occurs in <1% of pregnancies but is responsible for poor maternal and fetal outcomes due to hemorrhage. This is due in part to the human maternal circulation’s unique organization, as it is open at the placental interface. It is not always clear that significant blood loss has occurred if the blood is trapped at the myometrial interface.87 Although many risk factors for abruption have been identified, such as maternal hypertension, smoking, and alcohol use, the mechanisms underlying the primary condition are mostly unknown. Trauma87 and pre-eclampsia77 are both associated with PA due to physical loading of the uterine-placental interface. PA is a frequent outcome of motor vehicle accidents, where the uteroplacental interface may be directly loaded by the seatbelt, airbag, or steering wheel.82
Despite the broad spectrum of clinical complications of pregnancy as presented above, the human placenta’s development remains something of an enigma. There are ethical challenges in performing experimental research with pregnant women. There are severe limitations to the use of animal studies due to substantial inter-species differences in placental anatomy and function.70 The investigation of early-pregnancy development of the placenta can be limited by local governmental restrictions regarding the research use of embryonic stem cells. That said, with very recent developments in human induced pluripotent stem cells (iPSCs) and trophoblast stem cells, significant advances have been made just in the last year or two.
Two broad categories of research approaches have seen recent growth for the study of human pregnancy, with a particular focus on preterm birth prevention. These include in vitro techniques3,4,7,8,10,27,39,69 based on tissue engineering, organoids, and microfluidic devices, and in silico computational modeling techniques.20,23,47,71,73,83,97 The in vitro techniques take advantage of two recent developments, those in the cellular milieu noted above and those to do with biomaterials and small-scale fabrication technologies. A separate category is for works that can be performed virtually, using computational models for the exploration of larger-scale systems far too complex for small-scale in vitro experimental approaches. Each of these three categories of placental bioengineering research techniques will next be considered in turn.
Organoids and Biomimetic Tissue Engineering
Tissues consist of cells plus extracellular matrix (ECM), with structural connective tissues dominated by the latter. Tissue engineering and organoid approaches exist on a continuum (Fig. 3), from seeding cells onto biomaterial scaffolds at one end, typical of connective tissue engineering, to coaxing the self-organization of cells into tissue-like structures on the other end, the organoid-type approach now favored for cell-dominated tissues. In the intermediate region (Fig. 3) are approaches that use exogenous biomaterials such as hydrogels to a greater or lesser extent to try to encourage cell differentiation and organization in three dimensions. While a full examination of placental and uterine cell biology is well beyond the scope of the current review, a few recent approaches are summarized in the following paragraphs as a ‘taster’ of current and future research directions in ex vivo placenta research, from implantation10 to mature placental barrier functions.18 It should be emphasized at this point that much of this work represents novel approaches and quite recent scientific advances, and thus is largely “proof-of-concept” research. This is therefore in stark contrast to many more well-developed fields of bioengineering inquiry, such as Orthopaedic tissue engineering, and highlights the challenges and opportunities available in placental bioengineering research.
Organoids
Organoids are distinguished by their spontaneous—but uncontrolled—morphogenesis, mimicking early embryogenesis.62 However, this process is clouded by many protocols’ reliance on Matrigel,44,80 a commercial hydrogel matrix derived from mouse tumor cells and recognized for its batch variability80 and the presence of thousands of poorly characterized proteins and growth factors.46 With developments in hydrogel technology, some researchers are starting to use more controlled hydrogel matrices in organoid culture,62 which can only improve repeatability and reliability of results.80 This is an area of growing and much-needed collaboration between cell biologists and biomaterials engineers, for tuning both chemical and mechanical cues to cells in a spatio-temporally controlled manner.44 In addition to improved hydrogels for 3D culture support, added complexity in organoids has been achieved through co-culture with different cell lineages.44
A range of organoids designed to recapitulate tissues of the female reproductive system have been derived, including ovaries, cervix, fallopian tubes, and endometrium.7 These approaches to organoid development utilize Matrigel hydrogel matrices.7,27 The organoids have been developed for various applications, including fundamental biological science, models of human disease and therapeutics, and pharmacological screening.7,27 Recent publications have detailed the development of cytotrophoblast organoids41 and organoids with villous-tree-like structures and cells that differentiate to both syncytiotrophoblast and extravillous trophoblast.92 In both cases, these organoid cultures were derived using cells obtained from first trimester placental explants and grown using Matrigel.41,92 These novel approaches to developing trophoblast organoids have generated significant excitement for their potential in exploring early developmental phases of this difficult-to-study organ.
Tissue Engineering
Tissue engineering (TE) approaches have historically focused predominantly on the generation of replacement tissue for failed body parts, although artificial tissues have been considered as model systems for basic science and studies such as drug toxicology. The difference between TE approaches and organoids is the presence of an artificial ECM biomaterial. Early work on placental tissue models used a synthetic polymer microfiber-based bioreactor culture system to study trophoblast cell lines,58 work that now appears as a clear precursor to current microfluidic and nanofiber matrix-based approaches.8
Gargus et al.39 presented a thorough review of tissue engineering approaches for both male and female reproductive tissues, including uterus and placenta. The primary focus was on tissue transplantation, with some discussion of drug delivery and even the fetus’s futuristic ex vivo development. Biomimetic tissue engineering of the human uterine endometrium has recently been reviewed in the context of comparing decellularized native tissue with synthetic scaffold materials and structures for potential clinical treatment of uterine defects and infertility.42 Most studies related to tissue engineering of the placenta use decellularization of the tissue to produce an ECM for use in other applications, such as osteochondral defects.76 The recapitulation of functional placental tissue has been more in the domain of microfluidics approaches, and will be discussed below.
Hybrid Approaches
Some recent approaches have been a hybrid, intermediate between organoid and traditional tissue engineering approaches (Fig. 3). Endometrial organoids, as developed previously,91 were seeded onto porous collagen tissue engineering scaffolds to allow for cell co-culture and improved endometrial tissue mimicry, including hormone responsiveness.4 Engineered surfaces based on hydrogel micropatterning have been used to study biophysical control of the fusion of placental trophoblast cells into the syncytiotrophoblast membrane that forms at the maternal-fetal interface.59 Cell culture inserts featuring permeable membranes have been used for both trophoblast migration studies2,3 and for co-culture mimicking the placental barrier.18 Three-dimensional bioprinting has recently been used to generate biomimetic (methacrylated gelatin, gelMA) membranes designed to improve this technique with a more cell-friendly environment for creating placental-type barriers.51 Similar gelMA materials and 3D printing techniques have been used to develop a trophoblast invasion assay based on concentric rings of cell- and growth-factor chemoattractant loaded hydrogel in a truly 3D system without a membrane barrier.32 Other three-dimensional trophoblast invasion assays have been developed with gelatin hydrogels.52,100
The repeatability of studies based on organoid, tissue engineering or hybrid approaches using biomimicking hydrogels is an ongoing question that requires further research if these approaches are to substantially impact our understanding of the placenta. A consideration with hydrogel-based studies is the biomaterial’s mechanical stiffness and how it compares with the native tissue whose ECM it is mimicking.1 As mentioned previously, many of the human reproductive tissue organoid studies have used Matrigel as the artificial ECM.7,27 Abbas et al..1 examined Matrigel and quantified its batch-to-batch stiffness variability, consistent with its having a non-reproducible composition. Some clues as to the necessary ECM proteins for trophoblast survival and function can be found in works that have examined the placental ECM.53,67 Ideally a trophoblast hydrogel biomaterial growth environment would include both the necessary chemical and biomechanical cues for cell differentiation and villus formation. Overall, the biomechanical properties of the uterus and placenta have been studied very little compared with other human tissues, and this presents a research opportunity that will enhance the use of in vitro approaches for placental bioengineering research.
Placental Microfluidic Devices
Recently, several approaches have been considered for examining the placenta in the context of an “organ-on-a-chip” framework.2,55,57,69,70,78,79,98,102 Currently, a distinguishing feature between organoid research and organ-on-a-chip approaches26 has been the distinction between cell biology and bioengineering as the primary field of publication.96 Chip-based approaches are already commonly used in pregnancy-related applications, as glucometer and pregnancy tests are currently the most common tests completed on microfluidic devices.81 While polydimethylsiloxane (PDMS) is the most commonly used material in microfluidic devices,81 other polymers or hydrogels14 could also serve as functional materials. The excitement about a potential placenta-on-a-chip is related not just to the placenta’s research importance and to the lack of existing in vitro models for studying its functions. Because of the complexities of obtaining and culturing relevant cell types, microfluidic devices are favored as they allow for experiments using small cell numbers. Three different types of placental microfluidics experiments are shown in Fig. 4, and these will be described in the remainder of this section.
(a) A microfluidic device used to examine trophoblast invasion, with three channels: a central hydrogel seeded with cells and two peripheral channels filled with media with different fluid chemical compositions to establish a chemical gradient across the hydrogel. Time-lapse confocal z-stack images can be taken at various locations in the hydrogel channel to track individual cell motion in response to the chemical gradient. Figure adapted from Ref. 2. (b) A microfluidic device representing maternal cells and the fetal membranes separated by the placental barrier. Solutions can be inserted into the maternal side of the device to analyze how the placental barrier protects the fetus from harmful substances; figure adapted from Ref. 61. (c) A stacked placental barrier microfluidics device, after.88 This figure shows a microfluidic device with channels A and B stacked on top of each other with the central membrane in between. This figure depicts an out-of-plane layout of a microfluidic device, rather than an in-plane version as depicted in (a, b).
Trophoblast Invasion Microfluidics Assay
The mechanics of trophoblast migration have been investigated2 in a hydrogel microfluidics platform.85The assay was originally developed to study cancer metastasis, a common microfluidics application.45,99 Cells are seeded in a central hydrogel-filled channel and exposed to a chemical gradient established across the hydrogel (Fig. 4a2). In this assay, trophoblast migration using primary cells was assessed in response to a gradient of the uterine chemoattractant GM-CSF.2 This is important as there are questions surrounding whether the invasive nature of choriocarcinoma trophoblast-like cell lines is diminished relative to primary cells. A similar approach, but utilizing a flow-free microfluidic device, has been used for chemotaxis investigations of sperm in response to a progesterone gradient.14 This more straightforward device design may have potential for future investigation of chemotaxis in placental trophoblasts.
Placental Barrier Microfluidics Studies
Microfluidic approaches have also shown potential in devices that consider the placenta’s barrier function between mother and fetus. Different configurations of artificial lab-on-a-chip barriers have been considered, including in-plane (Fig. 4b61) and out-of-plane relative to the microfluidic device (Fig. 4c88). Recent studies using microfluidic devices have been focused on drug transport across the placental barrier. Such approaches have been reviewed8,69 in the context of the substantial advantage in vitro strategies have for testing pharmaceutical agents. The potential for drugs to penetrate the placental barrier and to impact the fetus has long been a concern with the otherwise routine treatment of pregnant women, following the disastrous thalidomide tragedy of the mid-20th century.93 Thus, microfluidics approaches present a new screening opportunity that could help indirectly advance the NIH’s goals of including more women—including pregnant women—in clinical trials including drug trials by providing a pre-test of possible fetal exposure due to placental barrier penetration. Placenta-on-a-chip models have been used to examine transport of glucose, heparin, caffeine, and nanoparticles across the placental barrier.69 In addition to drug models, these approaches can consider pathogenic exposures, such as bacteria102 or the Zika virus.8 There is also developing interest in delivery of drugs to the placenta itself, in the development of potential therapeutics for placenta-based conditions such as pre-eclampsia.8
All of the in vitro models considered here have advanced placental research dramatically in just the last five years. However, in parallel, there has been significant growth in the study of computational models of the placenta in silico, taking research out of the wet lab and into the bioengineering modeling domain. These computational approaches, which will be reviewed next, have ranged from the smallest-scale transport in fetal capillaries to macro-scale traumatic loading of the pregnant patient.
Computational Placenta Models
The placenta has been the focus of recent biomechanical models across a wide range of length scales, demonstrating the potential for computational modeling in understanding placental pathology and placenta-related clinical conditions. The development of new computational approaches for investigations of the placenta’s fundamental functions, such as blood flow, shear stress, and oxygen diffusion, have been the subject of recent scientific meetings5 and review papers.23,73,84
Placental Function Models
As the last decade has passed, there has been a progression from analytical models,64 continuum porous media models based on representative or idealized geometries20 to computational models based on the extraordinarily complex real geometries found in the placental vasculature (Fig. 5a71). Recent approaches to collecting geometrical representations of placentas for modeling purposes have included 2D placental cross-section scans54, 3D confocal laser scanning image stacks,36,72,74 and 3D micro-computed tomography (microCT) images utilizing polymer casts.11 Some approaches have generated multi-scale vascular networks in silico based on known “rules” governing the formation of vascular networks in vivo.22,49 In addition to baseline models of normal placental function, recent works have used computer-generated models to examine altered placental structure and oxygen transport in clinical conditions such as FGR86 (Fig. 5b89). This exciting development explores the real potential for addressing complex clinical problems with computational in silico methodologies: The limitation of scarce availability of pathological tissue is removed when pathologies are simulated based on known structural changes in the placenta.86,89
(a) 2D confocal (CLSM) image stack view of a terminal placental villus, and (b) corresponding three-dimensional reconstruction of the same tissue region, illustrating the internal fetal capillary loops and the external diffusion surface of the tissue in contact with maternal blood in the intervillous space. Image adapted from Ref. 71. (b) Computer-generated models of the vascular networks in (left) normal and (right) FGR placentae, illustrating how overall structural differences in the placental vascular networks could be used to simulate pathology; figure portions reproduced from Ref. 89.
A significant challenge associated with all of these computational models is the question of experimental validation. Different approaches have been used to address this challenge at different length-scales relative to the placental function. In converting image data to computational model domains, objects of known geometry have been used to validate the imaging and conversion of micrometer-scale image data into quantitative computational geometric domains.71 Three-dimensional printing of scaled-up models, along with particle flow velocimetry of real physical flows, have been used to confirm the validity of the microvasculature flow simulations.72 At macroscopic scales, medical imaging data is available via magnetic resonance imaging.30 Multi-scale modeling approaches are increasingly favored22 and provide promise for improving the validation of models with clinically-obtainable data. A clear direction for future research in this area is bridging the length-scales from millimeter-scale medical imaging data to micrometer-scale computational models at the scale of the functional placental exchange units, the terminal villi.
Placental Trauma Models
At a more macroscopic level, there has been a recent increase in blunt trauma studies in pregnant women, particularly in the context of motor vehicle accidents and automotive safety. Trauma is an unfortunately common complication of pregnancy, and most commonly associated with motor vehicle accidents, domestic violence, and falls.25 The most common cause of fetal death, aside from maternal death, has been placental abruption.43 Direct loading of the pregnant uterus, as could occur in trauma cases, has been simulated using finite element (FE) modeling of rigid bar impacts; the model included a placenta, fetus, amniotic fluid, and is based on third-trimester computed tomography (CT) scans.68 Rigid bar loading and seatbelt loading have both been considered in a more complex model in which a full FE representation of the fetal skull is presented.6 The strain level at the uterine-placental interface (UPI) of 0.60 was taken as representative of placental abruption; this strain level was routinely exceeded in simulations of unrestrained or airbag-only restraint but rarely exceeded for seatbelt restraint and never exceeded for combined seatbelt and airbag restraint.6
A whole-body FE model of a 26-week gestation simulated several different accident scenarios and predictors of adverse fetal outcomes in addition to the UPI strain, and including head injury criteria for both mother and fetus and intrauterine pressure.9 This study included detailed investigations of the kinematics of different loading phases due to an MVA, including maternal forward bending and direct abdominal compression by the lap seatbelt. These authors suggested the 0.6 strain threshold for the UPI was too great and that placental abruption was likely to occur at smaller strains.9 In related work, the UPI strain in a simulated frontal MVA was surprisingly independent of the placenta’s precise location within the uterus.29 The same challenge of experimental validation is associated with these macro-scale FE models as with the micro-scale placental transport models, in that comparisons with any empirical data are limited to modified crash test dummies and non-pregnant cadavers. Neither of these perfectly represents the traumatic loading response of a living pregnant driver.
Outlook
Bioengineering approaches to the examination of pregnancy have a long history. Some consider Leonardo da Vinci the first “bioengineer” due to his parallel interests in both the human body and in technology; his drawings of the dissected pregnant uterus were the first to capture the anatomy correctly.34 Da Vinci first observed the fetal membranes and realized that the maternal and fetoplacental circulations were independent.34 Biomechanical studies of the fetal membranes appeared throughout the 19th and 20th century scientific literature (reviewed in Ref. 21). Bioengineering studies of the placenta have been far more common in the 21st century, especially given the attention provided by the US National Institutes of Health and their Human Placenta Project.40
The current diversity of placental bioengineering research is reflected by the word cloud shown in Fig. 6a for “bioengineering” and “pregnancy”, which includes many but not all of the topics considered in this review; here, the focus has been on recent advances made through in vitro and in silico approaches. The growth in the field of placental bioengineering in particular is reflected in the recent rapid expansion of the literature, particularly in the last five years (Fig. 6b). The bioengineering study modalities considered herein are particularly critical in human pregnancy research, in which in vivo studies are (rightly) ethically limited, and biofidelic animal models simply do not exist due to anatomical and embryological differences between species. Complementary to fast-moving developments in bioengineering are those in placental biophysics, particularly as concerns the gathering and interpretation of quantitative medical imaging data.
(a) Word cloud for Scopus database search of “placenta” and “bioengineering”. Top word results within the 132 documents found in this search were (number of times in document titles): human (13); cells (10); model (9); fetal (7); health (7); engineering (6); pregnant (6); display (5); future (5); tissue (5). (b) Pubmed documents per year for a review search on “placenta” and “engineering” showing significant growth in the last decade.
Examination of the recent literature also highlights the significant challenges that are still in need of further research. In particular, bridges are needed across length- and time-scales to connect clinical diseases late in pregnancy, such as FGR and PE, to their early timepoint origins. Although there have been some recent advances in computational modeling of placental function, a multi-scale “virtual placenta” model is needed. Such a model would join together small-scale but detailed oxygen exchange models at the individual placental villus scale to the macroscopic circulation, which is accessible to medical imaging technologies (MRI, ultrasound) for clinical diagnosis. Microfluidics, tissue engineering, and organoid approaches further open new avenues in biomaterials and cellular research, with clear needs in developing controlled hydrogel biomaterials for improving studies’ reliability and tissue-specificity. There are other biomaterials needs and opportunities within pregnancy research as have been highlighted recently in a review on contraception.24 With the biomedical engineering field evolving, and engineers increasingly collaborating with medical professionals in obstetrics, midwifery, neonatology, and other relevant clinical areas, the future of placental bioengineering looks bright.
References
Abbas, Y., A. Carnicer-Lombarte, L. Gardner, J. Thomas, J. J. Brosens, A. Moffett, A. M. Sharkey, K. Franze, G. J. Burton, and M. L. Oyen. Tissue stiffness at the human maternal-fetal interface. Hum. Reprod. 34:1999–2008, 2019.
Abbas, Y., C. M. Oefner, W. J. Polacheck, L. Gardner, L. Farrell, A. Sharkey, R. Kamm, A. Moffett, and M. L. Oyen. A microfluidic assay to study invasion of human placental trophoblast cells. J. R. Soc. Interface 14:20170131, 2017.
Abbas, Y., M. Y. Turco, G. J. Burton, and A. Moffett. Investigation of human trophoblast invasion in vitro. Hum. Reprod. Update. 26:501–513, 2020.
Abbas, Y., L. G. Brunel, M. S. Hollinshead, R. C. Fernando, L. Gardner, I. Duncan, A. Moffett, S. Best, M. Y. Turco, G. J. Burton, and R. E. Cameron. Generation of a three-dimensional collagen scaffold-based model of the human endometrium. Interface Focus. 10:20190079, 2020.
Acharya, G., J. Aplin, P. Brownbill, J. Bulmer, G. Burton, L. Chamley, I. Chernyavsky, A. Clark, S. Collins, E. Cottrell, M. Dilworth, D. Elad, M. Filoche, N. Hannan, A. E. P. Heazell, O. Jensen, E. D. Johnstone, L. Leach, R. Lewis, T. Morgan, J. Myers, G. Nye, M. Oyen, C. Salafia, H. Schneider, and P. O’Tierney-Ginn. IFPA meeting 2017 workshop report: Clinical placentology, 3D structure-based modeling of placental function, placental bed, and treating placental dysfunction. Placenta 64(Supp. 1):S4–S8, 2018.
Acar, B. S., M. Meric, and V. Esat. Hybrid foetus with an FE head for a pregnant occupant model for vehicle safety investigations. Int. J. Crashworthiness 23:540–548, 2018.
Alzamil, L., K. Nikolakopoulou, and M. Y. Turco. Organoid systems to study the human female reproductive tract and pregnancy. Cell Death Differ. 2020. https://doi.org/10.1038/s41418-020-0565-5.
Arumugasaamy, N., K. D. Rock, C.-Y. Kuo, T. L. Bale, and J. P. Fisher. Microphysiological systems of the placental barrier. Adv. Drug Deliv. Rev. 2020. https://doi.org/10.1016/j.addr.2020.08.010.
Auriault, F., L. Thollon, J. Peres, and M. Behr. Adverse fetal outcome in road accidents: Injury mechanism study and injury criteria development in a pregnant woman finite element model. Accid. Anal. Prev. 97:96–102, 2016.
Ban, Z., F. Knoespel, and M. R. Schneider. Shedding light into the black box: advances in in vitro systems for studying implantation. Dev. Biol. 463:1–10, 2020.
Bappoo, N., L. J. Kelsey, L. Parker, T. Crough, C. M. Moran, A. Thomson, M. C. Holmes, C. S. Wyrwoll, and B. J. Doyle. Viscosity and haemodynamics in a late gestation rat feta-placental arterial network. Biomech. Model. Mechanobiol. 16:1361–1372, 2017.
Bartels, H.C., J. D. Postle, P. Downey, and D. J. Brennan. Placenta accrete spectrum: A review of pathology, molecular biology, and biomarkers. Disease Markers. 1507674, 2018. https://doi.org/10.1155/2018/1507674.
Belfort, M. A. Indicated preterm birth for placenta accrete. Semin. Perinatol. 35:252–256, 2011.
Berendsen, J. T. W., S. A. Kruit, N. Atak, E. Willink, and L. I. Segerink. Flow-free microfluidic device for quantifying chemotaxis in spermatozoa. Anal. Chem. 92:3302–3306, 2020.
Bell, AW, W. W. Hay, and R. A. Ehrhardt. Placental transport of nutrients and its implications for fetal growth. J Reprod Fertil Suppl. 54:401-410, 1999.
Brosens, I., R. Pijnenborg, L. Vercruysse, and R. Romero. The “Great Obstetrical Syndromes” are associated with disorders of deep placentation. Am. J. Obstet. Gynecol. 204:193–201, 2011.
Bublitz, M. H., M. Carpenter, and G. Bourjeily. Preterm birth disparities between states in the United States: an opportunity for public health interventions. J. Psychosom. Obstet. Gynecol. 41:38–46, 2020.
Campagnolo, L., V. Lacconi, M. Massimiani, A. Magrini, and A. Pietroiusti. In vitro experimental models to study the efficiency of the placental barrier for environmental toxicants: tumor cell lines versus trophoblast primary cells. Biomed. Prev. Issues. 1:157, 2018.
Carter, A. M. Evolution of factors affecting placental oxygen transfer. Placenta 30:19–25, 2009.
Chernyavsky, I. L., O. E. Jensen, and L. Leach. A mathematical model of intervillous blood flow in the human placentome. Placenta 31:44–52, 2010.
Chua, W., and M. L. Oyen. Do we know the strength of the chorioamnion? A critical review and analysis. Eur. J. Obstet. Gynecol. Reprod. Biol. 144(Supp. 1):128–133, 2009.
Clark, A. R., M. Lin, M. Tawhai, R. Saghian, and J. L. James. Multi-scale modelling of the feto-placental vasculature. Interface Focus. 5:20140078, 2015.
Clark, A. R., T. C. Lee, and J. L. James. Computational modeling of the interactions between the maternal and fetal circulations in human pregnancy. WIREs Syst. Biol. Med. 2020. https://doi.org/10.1002/wsbm.1502.
Claure, I., D. Anderson, C. M. Klapperich, W. Kuohung, and J. Y. Wong. Biomaterials and contraception: promises and pitfalls. Ann. Biomed. Eng. 48:2113–2131, 2020.
Connolly, A. M., V. L. Katz, K. L. Bash, M. J. McMahon, and W. F. Hansen. Trauma and pregnancy. Am. J. Perinatology. 14:331–336, 1997.
Convery, N., and N. Gadegaard. 30 years of microfluidics. Micro Nano Eng. 2:76–91, 2019.
Cui, Y., H. Zhao, S. We, and X. Li. Human female reproductive system organoids: applications in developmental biology, disease modeling, and drug discovery. Stem Cell. Rev. Rep. 2020. https://doi.org/10.1007/s12015-020-10039-0.
Culhane, J. F., and R. L. Goldenberg. Racial disparities in preterm birth. Semin. Perinatol. 35:234–239, 2011.
Delotte, J., M. Behr, L. Thollon, A. Bongain, and C. Brunet. Does placenta position modify the risk of placental abruption in car crashes? Comp. Methods Biomech. Biomed. Eng. 12:399–405, 2009.
DellSchaft, N. S., G. Hutchinson, S. Shah, N. W. Jones, C. Bradley, L. Leach, C. Platt, R. Bowtell, and P. A. Gowland. The haemodynamics of the human placenta in utero. PLoS Biol. 18:e3000676, 2020.
DiGregorio, S. Early: An intimate history of premature birth and what it teaches us about being human. New York: HarperCollins Publishers, 2020.
Ding, H., N. P. Illsley, and R. C. Chang. 3D bioprinted GelMA based models for the study of trophoblast cell invasion. Sci. Rep. 9:18854, 2019.
Duncan, J. M. On a lower limit to the power exerted in the function of parturition, trans. R. Soc. Edinb. 24:639–651, 1867.
Dunn, P. M. Leonardo Da Vinci (1452–1519) and reproductive anatomy. Arch. Dis. Child. 77:F249–F251, 1997.
Dunn, P. M. John Whitridge Williams MD (1866-1931) of Baltimore: pioneer of academic obstetrics. Arch. Dis. Child. Fetal Neonatal Ed. 92:F74–F77, 2007.
Erlich, A., P. Pearce, R. P. Mayo, O. E. Jensen, and I. L. Chernyavsky. Physical and geometric determinants of transport in fetoplacental microvascular networks. Sci. Adv. 5:eaav6326, 2019.
Erlich A, G.A. Nye, P. Brownbill, O.E. Jensen, I.L. Chernyavsky, Quantifying the impact of tissue metabolism on solute transport in feto-placental microvascular networks, Interface Focus 9: 20190021, 2019, https://doi.org/10.1098/rsfs.2019.0021
Frey, H. A., and M. A. Klebanoff. The epidemiology, etiology, and costs of preterm birth. Semin. Fetal Neonatal Med. 21:68–73, 2016.
Gargus, E. S., H. B. Rogers, K. E. McKinnon, M. E. Edmonds, and T. K. Woodruff. Engineered reproductive tissues. Nature. Biomed. Eng. 4:381–393, 2020.
Guttmacher, A. E., Y. T. Maddox, and C. Y. Spong. The Human Placenta Project: Placental structure, development, and function in real time. Placenta 35:303–304, 2014.
Haider, S., G. Meinhardt, L. Saleh, V. Kunihs, M. Gamperl, U. Kaindl, A. Ellinger, T. R. Burkard, C. Fiala, J. Pollheimer, S. Mendjan, P. A. Latos, and M. Knöfler. Self-renewing trophoblast organoids recapitulate the developmental program of the Early Human Placenta. Stem Cell Rep. 11:537–551, 2018.
Han, Q., and Y. Du. Advances in the application of biomimetic endometrium interfaces for uterine bioengineering in female infertility. Front. Bioeng. Biotechnol. 8:153, 2020.
Hill, C. C., and J. Pickinpaugh. Trauma and surgical emergencies in the obstetric patient. Surg. Clin. N Am. 88:421–440, 2008.
Holloway, E. M., M. M. Capeling, and J. R. Spence. Biologically inspired approaches to enhance human organoid complexity. Development 146:dev166173, 2019. https://doi.org/10.1242/dev.166173.
Huang, Y., B. Agrawal, D. Sun, J. S. Kuo, and J. C. Williams. Microfluidics-based devices: New tools for studying cancer and cancer stem cell migration. Biomicrofluidics 5:13412, 2011.
Hughes, C. S., L. M. Postovit, and G. A. Lajoie. Matrigel: a complex protein mixture required for optimal growth of cell culture. Proteomics 10:1886–1890, 2010.
Jensen, O. E., and I. L. Chernyavsky. Blood flow and transport in the human placenta. Ann Rev Fluid Mech. 51:25–47, 2019.
Jiao, L., E. Ghorani, N. J. Sebire, and M. J. Seckl. Intraplacental choriocarcinoma: systematic review and management guidance. Gynecol. Oncol. 141:624–631, 2016.
Kato, Y., G. J. Burton, and M. L. Oyen. Villous tree model with active contractions for estimating blood flow conditions in the human placenta. Open Biomed. Eng. J. 11:36–48, 2017.
Kaufmann, P., S. Black, and B. Huppertz. Endovascular trophoblast invasion: implications for the pathogenesis of intrauterine growth retardation and pre-eclampsia. Biol. Reprod. 69:1–7, 2003.
Kreuder, A.-E., A. Bolanos-Rosales, C. Palmer, A. Thomas, M.-A. Geiger, T. Lam, A.-K. Amler, U. R. Markert, R. Lauster, and L. Kloke. Inspired by the human placenta: a novel 3D bioprinted membrane system to create barrier models. Scientific Reports. 10:15606, 2020.
Kuo, C.-Y., A. Eranki, J. K. Placone, K. R. Rhodes, H. Aranda-Espinoza, R. Fernandes, J. P. Fisher, and P. C. W. Kim. Development of a 3D printed, bioengineered placenta model to evaluate the role of trophoblast migration in pre-eclampsia. ACS Biomaterials Sci. Eng. 2:1817–1826, 2016.
Kuo, C.-Y., T. Guo, J. Cabrera-Luque, N. Aramugasaamy, L. Bracaglia, A. Garcia-Vivas, M. Santoro, H. Baker, J. Fisher, and P. Kim. Placental basement membrane proteins are required for effective cytotrophoblast invasion in a 3D bioprinted placenta model. J. Biomed. Mater. Res. A. 106:1476–1487, 2018.
Lecarpentier, E., M. Bhatt, G. I. Bertin, B. Deloison, L. J. Salomon, P. Deloron, T. Fournier, A. I. Barakat, and V. Tsatsaris. Computational fluid dynamics simulations of maternal circulation: wall shear stress in the human placenta and its biological implications. PLoS ONE 11:e0147262, 2016.
Lee, J. S., R. Romero, Y. M. Han, H. C. Kim, C. J. Kim, J.-S. Hong, and D. Huh. Placenta-on-a-chip: a novel platform to study the biology of the human placenta. J. Matern Fetal Neonatal Med. 29:1046–1054, 2016.
Lewis, R. M., J. K. Cleal, and B. G. Sengers. Placental Perfusion and mathematical modelling. Placenta 93:43–48, 2020.
Liu, J., B. Mosavati, A. V. Oleinikov, and E. Du. Biosensors for detection of human placental pathologies: a review of emerging technologies and current trends. Translat. Res. 213:23–49, 2019.
Ma, T., S.-T. Yang, and D. A. Kniss. Development of an in vitro human placenta model by the cultivation of human trophoblasts in a fiber-based bioreactor system. Tissue Eng. 5:91–102, 1999.
Ma, Z., L. Sagrillo-Fagundes, R. Tran, P. K. Parameshwar, N. Kalashnikov, C. Vaillancourt, and C. Moraes. Biomimetic micropatterned adhesive surfaces to mechanobiologically regulate placental trophoblast fusion. ACS Appl. Mater. Interfaces 2020. https://doi.org/10.1021/acsami.9b.19906.
Malhotra, A., B. J. Allison, M. Castillo-Melendez, G. Jenkin, G. R. Polglase, and S. I. Miller. Neonatal morbidities of fetal growth restriction: pathophysiology and impact. Front. Endocrinol. 10:00055, 2019.
Mandt, D., P. Gruber, M. Markovic, M. Tromayer, M. Rothbauer, S. R. A. Krayz, F. Ali, J. V. Hoorick, W. Holnthoner, S. Mühleder, P. Dubruel, S. V. Vlierberghe, P. Ertl, R. Liska, and A. Ovsianikov. Fabrication of biomimetic placental barrier structures within a microfluidic device utilizing two-photon polymerization. Int. J. Bioprint. 4:144, 2018.
Marti-Figueroa, C. R., and R. S. Ashton. The case for applying tissue engineering methodologies to instruct human organoid morphogenesis. Acta Biomater. 54:35–44, 2017.
Mayhew, T. M. Estimating oxygen diffusive conductances of gas-exchange systems: a stereological approach illustrated with the human placenta. Ann. Anatomy 196:34–40, 2014.
Mirbod, P. Analytical model of the feta-placental vascular system: consideration of placental oxygen transport. R. Soc. Open Sci. 5:180219, 2018.
Moffett, A., and C. Loke. Immunology of placentation in eutherian mammals. Nat. Rev. Immunol. 6:584–594, 2006.
Nardozza, L. M., A. C. Caetano, A. C. Zamarian, J. B. Mazzola, C. P. Silva, V. M. Marcal, T. F. Lobo, A. B. Peixoto, and E. Araujo Junior. Fetal growth restriction: current knowledge. Arch Gynecol Obstet. 295:1061–1077, 2017.
Oefner, C. M., A. Sharkey, L. Gardner, H. Critchley, M. L. Oyen, and A. Moffett. Collagen type IV at the fetal-maternal interface. Placenta 36:59–68, 2015.
Parizi, M. I., M. T. Ahmadian, and H. Mohammadi. Rigid-bar loading on pregnant uterus and development of pregnant abdominal response corridor based on finite element biomechanical model. Int. J Num. Methods Biomed. Eng. 36:e3284, 2020.
Pemathilaka, R. L., D. E. Reynolds, and N. N. Hashemi. Drug transport across the human placenta: review of placenta-on-a-chip and previous approaches. Interface Focus. 9:20190031, 2019.
Pemathilaka, R. L., J. D. Caplin, S. S. Aykar, R. Montazami, and N. N. Hashemi. Placenta-on-a-chip: in vitro study of caffeine transport across placental barrier using liquid chromatography mass spectrometry. Glob. Chall. 3:1800112, 2019.
Plitman Mayo, R., D. S. Charnock-Jones, G. J. Burton, and M. L. Oyen. Three-dimensional modeling of human placental villi. Placenta. 43:54–60, 2016.
Plitman Mayo, R., J. Olsthoorn, D. S. Charnock-Jones, G. J. Burton, and M. L. Oyen. Computational modeling of the structure-function relationship in human placental terminal villi. J. Biomech. 49:3780–3787, 2016.
Plitman Mayo, R. Advances in human placental biomechanics. Comput. Struct. Biol. J. 16:298–306, 2018.
Plitman Mayo, R., Y. Abbas, D. S. Chanock-Jones, G. J. Burton, and G. Marom. Three-dimensional morphological analysis of placental terminal villi. Interface Focus. 9:20190037, 2019.
Quinn, J.-A., F. M. Munoz, B. Gonik, L. Frau, C. Cutland, T. Mallett-Moore, A. Kissou, F. Wittke, M. Das, T. Nunes, S. Pye, W. Watson, A.-M. A. Ramos, J. F. Cordero, W.-T. Huang, S. Kochhar, J. Buttery, Brighton Collaboration Preterm Birth Working Group. Preterm birth: case definition & guidelines for data collection, analysis, and presentation of immunisation safety data. Vaccine. 34:6047–6056, 2016.
Rameshbabu, A. P., P. Ghosh, E. Subramani, K. Bankoti, K. Kapat, S. Datta, P. P. Maity, B. Subramanian, S. Roy, K. Chaudhury, and S. Dhara. Investigating the potential of human placenta-derived extracellular matrix sponges coupled with amniotic membrane-derived stem cells for osteochondral tissue engineering. J. Mater. Chem. B 4:613–625, 2016.
Rana, S., E. Lemoine, J. P. Granger, and S. A. Karumanchi. Pre-eclampsia: pathophysiology, challenges, and perspectives. Circ. Res. 124:1094–1112, 2019.
Richardson, L., S. Jeong, S. Kim, A. Han, and R. Menon. Amnion membrane organ-on-chip: an innovative approach to study cellular interactions. FASEB J. 33:8945–8960, 2019.
Richardson, L., S. Kim, R. Menon, and A. Han. Organ-on-chip technology: the future of feto-maternal interface research? Front. Physiol. 11:715, 2020.
Rossi, G., A. Manfrin, and M. P. Lutolf. Progress and potential in organoid research. Nat. Rev. Genet. 19:671–687, 2018.
Sackmann, E. K., A. L. Fulton, and D. J. Beebe. The present and future role of microfluidics in biomedical research. Nature 507:181–189, 2014.
Sakamoto, J., C. Michels, B. Eisfelder, and N. Joshi. Trauma in pregnancy. Emerg. Med. Clin. North Am. 37:317–338, 2019.
Serov, A. S., C. Salafia, P. Brownbill, D. S. Grebenkov, and M. Filoche. Optimal villi density for maximal oxygen uptake in the human placenta. J. Theor. Biol. 368:133–144, 2015.
Serov, A. S., C. Salafia, D. S. Grebenkov, and M. Filoche. The role of morphology in mathematical models of placental gas exchange. J. Appl. Physiol. 120:17–28, 2016.
Shin, Y., S. Han, J. S. Jeon, K. Yamamoto, I. K. Zervantonakis, R. Sudo, R. D. Kamm, and S. Chung. Microfluidic assay for simultaneous culture of multiple cell types on surfaces or within hydrogels. Nat. Protoc. 7:1247–1259, 2012.
Sun, C., K. M. Groom, C. Oyston, L. W. Charley, A. R. Clark, and J. L. James. The placenta in fetal growth restriction: what is going wrong? Placenta 96:10–18, 2020.
Tikkanen, M. Placental abruption: epidemiology, risk factors, and consequences. Acta Obstet. Gynecol. Scand. 90:140–149, 2011.
Truskey, G. A. Human microphysiological systems and organoids as in vitro models for toxicological studies. Front Public Health. 6:185, 2018.
Tun, W. M., C. H. Yap, S. N. Saw, J. L. James, and A. R. Clark. Differences in placental capillary shear stress in fetal growth restriction may affect endothelial cell function and vascular network formation. Sci. Rep. 9:9876, 2019.
Turco, M. Y., and A. Moffett. Development of the human placenta. Development 146:163428, 2019.
Turco, M. Y., L. Gardner, J. Hughes, T. Cindrova-Davies, M. J. Gomez, L. Farrell, M. Hollinshead, S. G. E. Marsh, J. J. Brosens, H. O. Critchley, B. D. Simons, M. Hemberger, B.-Y. Koo, A. Moffett, and G. J. Burton. Long-term, hormone-responsive organoid cultures of human endometrium in a chemically defined medium. Nat. Cell Biol. 19:568–577, 2017.
Turco, M. Y., L. Gardner, R. G. Kay, R. S. Hamilton, M. Prater, M. S. Hollinshead, A. McWhinnie, L. Esposito, R. Fernando, H. Skelton, F. Reimann, F. M. Gribble, A. Sharkey, S. G. E. Marsh, S. O’Rahilly, M. Hemberger, G. J. Burton, and A. Moffett. Trophoblast organoids as a model for maternal–fetal interactions during human placentation. Nature 564:263–267, 2018.
Vargesson, N. Thalidomide-induced teratogenesis: history and mechanisms. Birth Defects Res. Part C 105:140–156, 2015.
Villar, J., A. T. Papageorghiou, H. E. Knight, M. G. Gravett, J. Iams, S. A. Waller, M. Kramer, J. F. Culhane, F. C. Barros, A. Conde-Agudelo, Z. A. Bhutta, and R. L. Goldenberg. The preterm birth syndrome: a prototype phenotypic classification. Am. J. Obstet. Gynecol. 206:119–123, 2012.
Walani, S. R. Global Burden of Preterm Birth. Int. J. Gynecol Obstet 150:31–33, 2020.
Wang, Z., X. He, H. Qiao, and P. Chen. Global trends of organoid and organ-on-a-chip in the past decade: a bibliometric and comparative study. Tissue Eng. Part A 26:656–671, 2020.
Westerveldt, A. R., and K. M. Myers. Computer modeling tools to understand the causes of preterm birth. Semin. Perinatol. 41:485–492, 2017.
Xiao, S., J. R. Coppeta, H. B. Rogers, B. C. Isenberg, J. Zhu, S. A. Olalekan, K. E. McKinnon, D. Dokic, A. S. Rashedi, D. J. Haisenleder, S. S. Malpani, C. A. Arnold-Murray, K. Chen, M. Jiang, L. Bai, C. T. Nguyen, J. Zhang, M. M. Laronda, T. J. Hope, K. P. Maniar, M. E. Pavone, M. J. Avram, E. C. Sefton, S. Getsios, J. E. Burdette, J. J. Kim, J. T, Borenstein, and T. K. Woodruff. A microfluidic culture model of the human reproductive tract and 28-day menstrual cycle. Nat. Commun. 8:14584, 2017.
Xu, H., X. Liu, and W. Le. Recent advances in microfluidic models for cancer metastasis research. Trends Anal. Chem. 105:1–6, 2018.
Zambuto, S. G., K. B. H. Clancy, and B. A. C. Harley. A gelatin hydrogel to study endometrial angiogenesis and trophoblast invasion. Interface Focus. 9:20190016, 2019.
Zhu, J.-Y., Z.-J. Pang, and Y.-H. Yu. Regulation of trophoblast invasion: the role of matrix metalloproteinases. Rev. Obstet. Gynecol. 5:e137–e143, 2012.
Zhu, Y., F. Yin, H. Wang, L. Wang, J. Yuan, and J. Qin. Placental barrier-on-a-chip: modeling placental inflammatory responses to bacterial infection. ACS Biomaterials Sci Eng. 4:3356–3363, 2018.
Funding
Funding was provided by the East Carolina University Division of Research, Economic Development and Engagement (REDE) via start-up funds to MLO.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interest to declare.
Additional information
Associate Editor Stefan M. Duma oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wheeler, M.L., Oyen, M.L. Bioengineering Approaches for Placental Research. Ann Biomed Eng 49, 1805–1818 (2021). https://doi.org/10.1007/s10439-020-02714-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-020-02714-7