Abstract
Triatomines (Hemiptera: Heteroptera: Reduviidae), commonly known as conenose or kissing bugs, includes species of outstanding medical importance given their role as vectors of the protozoan parasite Trypanosoma cruzi Chagas (Kinetoplastida: Trypanosomatidae), the aetiological agent of Chagas disease. Herein, we present an updated review of the chemical ecology of triatomines, with special reference to molecules involved in the communication of these insects. Infochemicals, including hormones and semiochemicals (pheromones and allelochemicals), have a pivotal role in mediating information and shaping interactions within and between triatomines, during distinct behavioural contexts. We analyse the new findings about the chemical composition of alarm, aggregation and sex pheromones, as well as on cuticular hydrocarbons, host attractants and repellents. We also discuss how triatomine hormones may serve as communication signals and highlight the importance of the reverse chemical ecology approach in studying the complex chemoecology of these arthropod vectors. In the final section, we outline how knowledge about triatomine chemical ecology can be used for implementing real-world control operations with proven epidemiological impact.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
key message
-
Triatomines are insects of medical importance as they are vectors of the parasite Trypanosoma cruzi, the aetiological agent of Chagas disease.
-
Knowledge on semiochemicals and hormones affecting triatomine behavior is reviewed; these compounds have a key role in shaping the pathogen-vector-host interaction.
-
Triatomine chemical ecology can be exploited for developing and implementing real-world vector management programs with proven epidemiological impact, reducing the trasmission of Chagas disease. In this framework, novel research approaches in chemoecology and promising control tools are discussed.
Introduction
Triatomines, also known as kissing bugs, are hematophagous insects from the subfamily Reduviidae (Hemiptera: Heteroptera) (Schofield and Galvão 2009; di Giovanni et al. 2021). Around more than 150 species have been described which are currently distributed in five tribes, i.e. Alberproseniini, Bolboderini, Cavernicolini, Rhodniini and Triatomini. The last two are the most diverse and of medical importance (Justi and Galvão 2017; Ceccarelli et al. 2020), and act as vectors of Trypanosoma cruzi, the etiological agent of the Chagas disease. This illness is of major concern as it affects more than 8 million people, mostly in the Americas for which there is not an effective post-infection treatment (Ceccarelli et al. 2020). The triatomine life cycle is composed by pre-nymph, five nymph stages, and imagos (adults). Nymphs generally occupy the same habitat and feed on the same hosts of the adults (Noireau and Dujardin 2010).
Kissing bug natural history and ecology make them prominent vectors of T. cruzi. These insects inhabit a wide variety of environments, i.e. domestic, peridomestic, and wild habitats, in tropical, subtropical, and temperate areas (Ceccarelli et al. 2020). During the day, kissing bugs remain hidden inside shelters (e.g., cracks and crevices), where they find conspecific presence and key conditions of humidity and temperature (Lazzari 2021). These shelters protect them against predators and extreme abiotic conditions. At night, the insects come out in search of blood sources which comprise a variety of vertebrate hosts, including humans (Noireau and Dujardin 2010). Kissing bug olfaction is fundamental for their day-to-day activities (Latorre-Estivalis et al. 2015). The aptitude of organisms to perceive and transmit information through chemicals is a remarkable aspect of animal biology and ecology (Tirindelli et al. 2009). Chemical ecology examines the role of molecules in the interactions between living organisms and their environment, and it also deals with the consequences of those chemicals on the behaviour and evolution of involved organisms.
Conventional chemical ecology is based on extracting secretions from chemical signal senders, separating extracts into fractions following bioassay-guided protocols (using receivers to assist in the identification of active molecules) (Leal et al. 2008), concluding with the elucidation of chemical structures and in some cases with the chemical synthesis (Leal 2017). Techniques as gas chromatography coupled to mass spectrometry (GC–MS), gas chromatography coupled to infrared spectroscopy and electrophysiology studies [electroantennography (EAG) (May-Concha et al. 2018b), gas chromatography-electroantennography (GC-EAD) (Vitta et al. 2009), and single sensillum recording (SSR) (Campetella et al. 2021)] are routinary used for elucidation of insect infochemicals. A crucial part of the development of infochemicals is shedding light on the potential biological function, which is usually investigated through behavioural assays under laboratory and field conditions (Syed 2015).
Basic knowledge on triatomine chemical ecology has been reviewed earlier (Cruz-López et al. 2001). Recently, other contributions on this topic have been published (Latorre-Estivalis 2022; Lorenzo 2022), mostly focused on Rhodnius prolixus Stål physiology (Barrozo et al. 2017). The aim of the present review is to summarize and discuss current information and recent advances about triatomine chemical ecology, and the development of chemoecology-based monitoring and control tools. Although information in this regard is still fragmented, we believe there is room for both a literature analysis and a future research agenda, the latter highlighting the tight connection between basic chemical ecology research and the implementation of effective and sustainable methods for managing kissing bugs in the Integrated Vector Management (IVM) framework.
Infochemicals and triatomine behaviour
Traditionally, infochemicals have been seen as synonymous with semiochemicals. Notwithstanding, we followed the most recent classification proposed to use the term infochemicals for both hormones and semiochemicals because of their unifying function in information transfer (Supplementary Fig. 1) (Wehrenfennig et al. 2013; Müller et al. 2020). Recently, it has become clear that chemical ecology must deal with the influence of hormones and semiochemicals in the causes and consequences of species interactions, distribution, abundance, and diversity, providing mechanisms to understand ecological interactions and allowing novel vector management options. (Raguso et al. 2015).
Infochemicals, which include hormones and semiochemicals (pheromones and allelochemicals), play a key role in intra- and inter-specific interactions mainly during search for shelter and food, mating, and situations of alarm and threat (Cruz-López et al. 2001). These compounds are released by specialized glands that develop at the adult stage only (Guerenstein and Guerin 2004). The most studied glands are the Brindley (BGs) and metasternal glands (MGs) (Crespo and Manrique 2007; Manrique and Lorenzo 2012). However, it is of remarkable importance to investigate other glands (Santiago et al. 2020). Even though some studies have described the gland morphology for some species, the large variability as well as the lack of descriptions for most species do not allow to reach generalizations in this regard (Lopes et al. 2020). Since the first works on chemical ecology of triatomines, importance was attributed to the glandular system of the bugs (see Cruz-López et al. 2001 and references therein). A confusion emerged about the function of the compounds produced by both MGs and BGs. Moreover, as more data were available, it was clear that while MGs emit compounds that mediate sexual behaviour (Lopes et al. 2020), BGs emit compounds that underlie alarm/defence behaviour (Crespo and Manrique 2007). Apparently both glands are involved during sexual and defensive behaviours. However, detailed studies on different species are still needed (Palottini and Manrique 2016). Notice that these glands are also present in other insects, e.g., wheel bugs (Heteroptera: Reduviidae: Harpactorinae: Arilus), which produce isobutyric acid and other short chain carboxylic acids from BGs (Aldrich et al. 2013). The volatile components of MGs of Megacyllene robiniae (Forster) (Coleoptera: Cerambycidae) males and females contain 2-(1,3-hexadien-1-yl)-5-methyltetrahydrofuran and some esters (Mitchell et al. 2017). Intra-and inter-specific recognition is key for gregarious insects, and kissing bugs make use of cuticular hydrocarbons to achieve such function (Juárez and Fernández 2007; Figueiras et al. 2009). In the same fashion, by using olfaction they can distinguish the sites that serve as refuge (Lazzari 2021), hosts, and food sources (Castillo-Neyra et al. 2015).
Pheromones and cuticular compounds
Sex pheromones
Compared to other insect groups, our knowledge of chemical-mediated sexual behaviour in triatomines is still limited. As a general trend, before mating, triatomine females emit a sexual pheromone from the MGs, and males use these cues to locate the females (Manrique and Lorenzo 2012). Even though there has been evidence of sexual pheromones in triatomines, until recently, a compound or a synthetic mixture that works as a pheromone has been uncovered (Bohman et al. 2018). Most studies have focused on determining the compounds coming from the MGs for some species; Triatoma brasiliensis Neiva (Vitta et al. 2009), Triatoma dimidiata Latreille (May-Concha et al. 2013), Triatoma infestans (Klug) (Manrique et al. 2006), R. prolixus (Bohman et al. 2018), and three species of the Triatoma phyllosoma group (May-Concha et al. 2018a). Volatile oxygenated compounds, such as ketones and alcohols, are present (Table 1). Opposite to what occurs with triatomines, MGs in other insects (e.g., long horned beetles, Coleoptera: Cerambycidae) are involved in terpene and terpenoid production and/or storage and are used for defence (Ohmura et al. 2009).
Once copulation starts, a volatile-mediated response takes place by which conspecific males aggregate around the mating couple (Fontan et al. 2002b; Rocha Pires et al. 2004; Pontes et al. 2008; May-Concha et al. 2013). Although little is known about the effect of chiral compounds on kissing bug behaviour, the existence of such molecules in MGs should be highlighted (Unelius et al. 2010), as the bioactivity of pheromones depends on their chirality (Fig. 1a) (Mori 2007). For example, T. infestans and T. brasiliensis produce the (4R, 5R) and (4S, 5S) isomers of 2,2,4-triethyl-5-methyl-1,3-dioxolane as the most abundant compounds (Fig. 1b) (Bohman et al. 2011). Moreover, not all compounds, both chiral and achiral, are antennal active. For example, while T. brasiliensis produces 16 compounds in the MGs, only 5 elicit antennal activity (Vitta et al. 2009). A mixture of 10 compounds from R. prolixus MGs were recently proposed as a synthetic pheromone (Bohman et al. 2018) (Fig. 2). Yet results with other species seem to be contradictory or preliminary. For example, an absence of a sexual pheromone was reported for Triatoma mazzotti Usinger, although these studies have not been conclusive and have not determined the glands’ chemical composition (Rojas et al. 1991). In preliminary studies, it has been showed that terpenoids, as the rose oxides, could affect the sexual communication of triatomines (May-Concha et al. 2018a). However, the chirality of compounds was not elucidated, and only two of the four possible compounds were reported (Fig. 3). Pheromone research has limitations, especially in the identification of compounds, due to the amounts produced by insects. However, with the recent creation of libraries of chiral retention indices of compounds would greatly increase the accuracy and speed of chemical identification for these runs (Alavez-Rosas et al. 2022). More studies are needed to perform a correct chiral identification of triatomine semiochemicals. Further experimental research should be also directed to the influence of pheromone chirality on the sexual behaviour of kissing bugs.
Compounds from Rhodnius prolixus females, formulated as a synthetic pheromone (Bohman et al. 2018). (1) 2-Butanone, (2) (S)-2-butanol, (3) 2-Methyl-3-buten-2-ol, (4) 3-Methyl-2-butanol, (5) 2-Methyl-1-butanol, (6) 2-Pentanone, (7) 3-Pentanol, (8) (S)-2-pentanol, (9) (E)-2-Methyl-3-penten-2-ol and (10) 4-Methyl-3-penten-2-ol
Cis- and trans-Rose oxides (isomers of 4-methyl-2-(2-methylprop-1-en-1-yl)tetrahydro-2H-pyran). Compounds found in the metasternal glands of triatomines of the Triatoma phyllosoma group (May-Concha et al. 2018a)
Alarm pheromones
When disturbed, adult triatomines (both males and females) release an alarm pheromone emitted by the BGs (Palottini et al. 2014). This leads to a locomotion and escape response by conspecifics (Manrique et al. 2006). Elucidation of BGs components has been carried out for some species of the Phyllosoma complex (May-Concha et al. 2018a), T. infestans (Crespo and Manrique 2007; González et al. 2007), three haplogroups of T. dimidiata (May-Concha et al. 2015), and R. prolixus (Rojas et al. 2002). The pheromone includes volatiles whose combination varies intra-specifically and includes carboxylic acids: isobutyric acid, acetic acid, propanoic acid, butanoic acid, pentanoic acid, hexanoic acid, heptanoic acid, octanoic acid, nonanoic acid, and isovaleric acid (González et al. 2007). Esters and alcohols derived from the above-mentioned acids (González et al. 2007; May-Concha et al. 2015), some ketones (3-methyl-2-hexanone, 2-butanone, 3-pentanone), and other ramified pentanones and hexanones have been also detected (González et al. 2007; May-Concha et al. 2015, 2018a) (Fig. 4). The main component of the pheromone is isobutyric acid, which was the first compound identified and catalogued as an alarm pheromone in species such as T. brasiliensis, T. maculata (Ericsson), T. infestans, T. phyllosoma, Triatoma vitticeps (Stål), Triatoma lecticularia (Stål), R. prolixus and Panstrongylus megistus (Burmeister) (Cruz-López et al. 2001). Interestingly, isobutyric acid bears implications on cognitive roles (i.e. acquiring knowledge through experience) (Minoli et al. 2013b) as well as innate responses (Minoli et al. 2018). Other species possess different compounds that are used in the alarm/defence behaviour, such as in the case of Dipetalogaster maxima (Uhler), which produces 3-methyl-2-hexanone via the MGs (Guerenstein and Guerin 2004).
Aggregation pheromones
Aggregation pheromones play a key role during mate choice, group foraging, and collective gathering near the pheromone source, either by attracting them from a distance (attractant activity) or inducing passing conspecifics to remain at the pheromone source (arrestant activity) (Gries et al. 2014; Mitaka et al. 2020). Related to this, triatomine bugs use different physical and chemical cues to orient towards refuges (Galvez-Marroquin et al. 2018; May-Concha et al. 2020). Triatoma species are attracted to and aggregate around conspecific as well as on heterospecific faeces (Cruz-López et al. 1993; Lorenzo-Figueiras and Lazzari 1998; Pires et al. 2002), footprints (Vitta et al. 2002, 2007; May-Concha et al. 2018c), and cuticular lipids (Figueiras et al. 2009; Cocchiararo-Bastias et al. 2011). However, this was not the case for R. prolixus, which deposit faeces inside the refuges thereby releasing a weak aggregation response by conspecifics (Falvo et al. 2016). Also, other Rhodnius species did not respond to these aggregation signals (Mosquera and Lorenzo 2020). Despite important efforts aimed to look for aggregation signals on triatomine bugs (Cruz-López et al. 1993, 2001; Cruz-López and Morgan 1995; Lorenzo-Figueiras and Lazzari 1998), there is no report of an aggregation pheromone.
Compounds responsible for attraction have also been reported, which are used during control programs (Rojas de Arias et al. 2012). Short chain aldehydes (C6–C10) have been found in faeces or volatiles from aggregated insects that work as attractants of triatomines (Cruz-López et al. 2001; Fontan et al. 2002a; Galvez-Marroquin et al. 2018). Alkanes (C18–C25) have also been found in faeces of T. dimidiata and T. infestans (Galvez-Marroquin et al. 2018), while two quinazolines occurred in faeces of T. infestans, to which nymphs were attracted to (Cruz-López and Morgan 1995; Alzogaray et al. 2005). Other faecal compounds (3-methylbutyric acid, hexanoic acid, 2,3-butanediol, acetic acid and acetamide) function to attract T. infestans, P. megistus, and T. brasiliensis (Fig. 5) (Mota et al. 2014). Lipids (hexadecanoic acid, octadecanoic acid, and octacosanoic acid) and fatty alcohols (eicosanol and docosanol) from the cuticle mediate aggregation in T. infestans (Figueiras et al. 2009; Cocchiararo-Bastias et al. 2011). Different classes of polar compounds are deposited in the faeces of triatomines, yet their function is unclear (Antunes et al. 2013), it is possible that these compounds act as arrestants. Other related aspects also remain unexplored, including (a) the possible origin of the pheromone components, (b) the components associated with the arrestant behaviour, and (c) the mixture of compounds that act as an aggregation pheromone.
Compounds found in the faeces of triatomines that were attractive to conspecifics, usually short-chain aldehydes, long-chain carboxylic acids, alcohols and some aromatics. In some species compounds as 2,3-butanediol (1), acetamide (2), acetic acid (3), hexanoic acid (4), and 3-methylbutyric acid (5) could be found. Polar compounds are usually amino acids or derived products, histamine (6), histidine (7) and proline (8) (Antunes et al. 2013)
Cuticular compounds (CCs), as signature mixtures in triatomines
As in most insects, CCs have the function of protecting against desiccation (Dulbecco et al. 2020), serve as a barrier to prevent the entry of microorganisms, and participate during chemical communication (Juárez and Fernández 2007). Although a defined role for CCs in triatomine chemical communication is not clear, the profile of compounds varies inter-specifically (Juárez and Blomquist 1993). There are some further differences such as ontogenetic (nymphs vs. adults) and adult sex variation in CCs (Juarez et al. 2001). In T. infestans, CCs promote aggregation and locomotion arrest (Figueiras et al. 2009), and propensity to mate by males. The most abundant CCs are hydrocarbons, fatty alcohols, and free and esterified fatty acids, eicosanol and docosanol are responsible of the attraction of males to females (Cocchiararo-Bastias et al. 2011). It is likely that CCs function as cues or signature mixtures in kissing bugs. Interestingly, CCs are also useful for species identification (e.g., a species complex in T. dimidiata) (Calderon-Fernández et al. 2011).
Allelochemicals
Host location
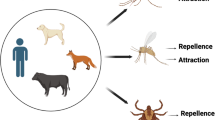
In triatomines,chemical mechanisms during host search had been less studied than other behaviours (Cruz-López et al. 2001). However, in recent years, robust data have been obtained leading the better understanding of chemical aspects of this process (Guerenstein and Lazzari 2009, 2010; Lazzari 2022). Triatomines use volatiles from hosts (Fig. 6) as these induce a strong antennal response (Campetella et al. 2021) and promote locomotion and exit from roosts (Ferreira et al. 2019). Compounds such as ammonia, isobutylamine, isobutyric acid, heptanal, octanal, nonanal, α-pinene, furane, pyridine, terpinen-4-ol, and other aromatic amines evoke electrophysiological responses in nymphs and adults of T. infestans, R. prolixus and D. maxima (Guerenstein and Lazzari 2009). Odours such as CO2 and ammonia (ubiquitous in vertebrates) are attractive to R. prolixus and have a synergistic combination effect (Otálora-Luna et al. 2004). Some compounds that have been found attractive to T. infestans are heptanal, octanal, nonanal and isobutyric acid, isolated from sheep wool, chicken feathers and rabbit hair (Guerenstein and Guerin 2001). Some volatile amines present as vertebrate waste products are attractive to R. prolixus, T. infestans, and Panstrongylus geniculatus Latreille (Otálora-Luna and Guerin 2014). l-lactic acid synergizes the attractive effect of CO2, while the d-isomer has no biological activity, highlighting the importance of chiral compounds in triatomine attraction (Barrozo and Lazzari 2004). Further, host-borne odours attract T. dimidiata and R. prolixus over short and long distances (Milne et al. 2009); the role of chemical signals from the host stimulates and guides kissing bugs to oviposit (Guidobaldi and Guerenstein 2015).
Antennal active volatiles found in different triatomine hosts. Ammonia (1), carbon dioxide (2), 2-methylpropanamine (3), isobutyric acid (4), terpineol (5), furan (6), pyridine (7), pinene (8) and nonanal (9) (Guerenstein and Lazzari 2009)
Hormones and their role in triatomines
Hormones are important for species interactions. These molecules mediate the production of allelochemicals, and their expression is influenced by many interspecific interactions (Raguso et al. 2015). The most studied hormone in triatomines, and in many insects, is the juvenile hormone (JH), a sesquiterpenoid hormone (Fig. 7) which regulates both insect metamorphosis and reproduction (Riddiford 2020). Among other functions in triatomines, JH manages lipid storage thereby affecting growth and reproduction (Ramos et al. 2021). JH also regulates production of sex pheromones (Kotaki et al. 2009). It has been speculated that hormones promote a signalling system so that triatomines can start copulation yet the exact mechanisms for this are unknown (Kotaki et al. 2009; Ramos et al. 2021). Another hormone that has a critical role in signalling the way in which triatomines adapt to nutritional conditions is the adipokinetic hormone (AKH) (Gäde and Marco 2022). In insects, the AKHs are 8–10 amino acids long (Fig. 8) (Marco et al. 2022). Recently, using bioinformatics on genes of R. prolixus, a novel mature octapeptide AKH was deduced (Marco et al. 2013). This hormone regulates lipid metabolism and carbohydrate mobilization, allowing them to survive during long fasting periods (Leyria et al. 2021). Understanding how AKH works could led to a strategy to mitigate the dispersal flights of triatomines, thus these insects will not be able to reach new areas and colonize them.
Structures of some natural juvenile hormones, JH III is found in triatomines (Riddiford, 2020)
pGlu-Leu-Thr-Phe-Ser-Thr-Asp-Trp amide, the sequence of a novel mature octapeptide AKH in some species of triatomines (Marco et al. 2013)
Reverse chemical ecology of triatomines: a leap forward
Coupled techniques (GC–MS, GC-EAD, and GC-SSR) have reduced analysis to even single individuals in many cases, yet these technniques imply killing the animal (Syed 2015). In the last two decades, with the understanding of the molecular basis of insect olfaction (Liu et al. 2021), an emerging approach has been used to the study of bloodsucking insects semiochemicals, the “reverse chemical ecology” (Supplementary Fig. 2) (Leal et al. 2008; Choo et al. 2018). This is a new term for screening of molecules based on the binding ability of odour binding proteins (OBPs) to test compounds, rather than performing series of behavioural bioassays (Rihani et al. 2021). This approach comprises the in silico study of binding potential between a OBP and a molecule (Kamala et al. 2014). Additional to OBPs, the odorant receptors (ORs) are proteins used to the screening process (Franco et al. 2016). OBPs are involved in the transport of molecules through the sensillum lymph to the olfactory receptor neurons (ORNs) where ORs are embedded (Campetella et al. 2021; Liu et al. 2021). ORs form ion channels with the obligatory odorant receptor coreceptor (ORCO) (Faro de Brito et al. 2016), and infochemicals activating OR-ORCO complexes generate electrical signals that are transmitted to the brain and lead to a behavioural response (Latorre-Estivalis et al. 2015, 2017; Franco et al. 2018) (Supplementary Fig. 3). However, evidence of binding to an OBP or activation of an OR is not sufficient to infer behavioural activity, and it must be confirmed by laboratory and field bioassays (Leal et al. 2008; Choo et al. 2018; Mweresa et al. 2020). Reverse chemical ecology has been applied to triatomines seeking infochemicals of possible applications for monitoring or controlling populations, e.g., silencing the host searching behaviour (Franco et al. 2016) and developing effective repellents. Using a reverse chemical ecology approach, Franco et al. (2018) accidentally found four good repellents for R. prolixus; the initial goal of the authors was to identify odorant receptor sensitive to putative pheromones receptors. Using an exposure device, the time spent by the insect near to the host in the presence or absence of the compounds tested was quantified. This highlights the importance of behavioural assays in reverse chemical ecology studies. In this scenario, reverse chemical ecology provides molecules that repel triatomines, potentially increasing the number of available repellents for these insects (Sainz et al. 2012; Ramírez et al. 2020). However, this approach is scarcely studied in kissing bugs, most of the studies are centred in basic aspects (characterization of OBPs (Oliveira et al. 2018), proteomic analysis of antennas (Oliveira et al. 2017), and antennal gene expression (Latorre-Estivalis et al. 2022)) and more substantial and robust knowledge of triatomine OBPs, ORs and ORCO are needed for future research (Sparks et al. 2015).
Practical applications
Traps baited with compounds identified in triatomine faces (mostly short-chain aldehydes) have been used to capture triatomines with good results (Lazzari and Lorenzo 2009). In this regard, CO2 has been used successfully (Ryelandt et al. 2011). However, CO2-free baits have also been used in traps with good results in the laboratory (Guidobaldi and Guerenstein 2013, 2016). For post-control surveillance of T. sordida and T. infestans, hexanal, octanal and nonanal have been used in South America (Fig. 9) (Rojas de Arias et al. 2012). Somehow related to this, a key problem to control kissing bugs using insecticides is its aggregation behaviour as this impedes penetration to the insect shelters. To overcome this issue, one may combine insecticide application with a substance that causes bugs to leave their shelters. For this, compounds produced by BGs have been used as “dislodging agents” in triatomine control (Fig. 9) (Minoli et al. 2013a). Reverse chemical ecology-based approach has led to the discovery of repellents for R. prolixus (Franco et al. 2018), and it is leading the new era of “rational infochemicals design”.
Semiochemicals applied in triatomine post-control surveillance along South America. Benzaldehyde (1), hexanal (2), octanal (3) and nonanal (4) (Rojas de Arias et al. 2012). 3-Methylbutanol (5), 3-pentanol (6) and isobutyric acid (7) are effective dislodging agents for triatomines (Minoli et al. 2013a)
Conclusions and research challenges
Having a robust database of chemical compounds that kissing bugs use to communicate could improve our knowledge and practical application in the control and monitoring of these insects (Tobias and Latorre-Estivalis 2021). Insect chemical ecology research has its limitations, especially in the identification of compounds, due to the amounts produced by insects (Yew and Chung 2015). Techniques as Nuclear Magnetic Resonance (NMR) or X-Ray diffraction are limited in pheromone research yet their integration will facilitate the purification and chemical identification of triatomine pheromones and other infochemicals (Nojima et al. 2011). The emergent techniques used in metabolomics could provide new opportunities to study such ecological relevant signalling molecules (Prince and Pohnert 2010). These techniques facilitate the identification of infochemicals and possibly reducing the need of bioassay-guided fractionation (Kuhlisch and Pohnert 2015). Transcriptomics and proteomics could contribute to the identification of molecules involved in vector–host interactions (Santiago et al. 2020). An unexplored part is the possible influence of T. cruzi parasites on the chemical communication of triatomines. In this regard, the possible influence of the parasite on the production and perception of the compounds has been hypothesized (Córdoba-Aguilar 2020). In support of this, a difference has been shown in the patterns of antennal sensilla between infected and uninfected insects (May-Concha et al. 2021), and circadian cycles (Chacón et al. 2022). Also, fifth instar parasitized nymphs were able to better detect a potential host (Botto-Mahan et al. 2006). These differences may be influenced by the parasite where morphological/behavioural changes in the infected bug may facilitate host detection and, thereby, parasite transmission from the vector to the host (Córdoba-Aguilar 2020). Further studies should test whether communication at the chemical level differs between parasitized and non-parasitized triatomines. In the near future, structures of triatomines will be solved, and we will understand the molecular basis of kissing bug olfaction. Such types of research will open up promising perspectives in triatomine regulation based on the manipulation in chemosensory behaviour (Marchant et al. 2021). With this information, we could design infochemicals for controlling populations of these insects, saving human lives by reducing transmission of the parasite (Leal 2014). The next step in studies of triatomine chemical ecology should be focused on the implementation of Integrated Vector Management (IVM) approaches (Lizzi et al. 2014; Chanda et al. 2017). For example, sex pheromones could be used as baits for traps, in an attraction and kill system (Barrera 2022; Lopes et al. 2022). In this regard, aggregation pheromones could be useful to attract nymphs and both imagoes (female and male) (Bell et al. 2018). Alarm pheromone could be evaluated as dislodgement agents (Minoli et al. 2013a). Knowledge about host-borne compounds (both attractants and repellents) could be used in push–pull strategies (Lorenzo et al. 2014; Wooding et al. 2020). Lastly, the reverse chemical ecology could represent the basis of a new management strategy; indeed, following a rational infochemical design it is possible to develop a species- or instar-selective attractant/repellent products (Olsson et al. 2015).
Author contributions
All authors have made substantial contributions to the following: (i) conception of the study or data collection (DAR, RVA, CAF, MMA, AGC, GB, LC, ACA), (ii) drafting the article and/or revising it critically for important intellectual content (DAR, RVA, CAF, MMA, AGC, GB, LC, ACA), (iii&iv) final approval of the submitted version (DAR, GB, LC, ACA).
References
Alavez-Rosas D, Nguyen LMN, Keefover-Ring K (2022) Retention indices for naturally-occurring chiral and achiral compounds on common gas chromatography chiral stationary phases. Results Chem 4:1–10. https://doi.org/10.1016/j.rechem.2022.100659
Aldrich J, Chauhan K, Zhang A, Zarbin P (2013) Exocrine secretions of wheel bugs (Heteroptera: Reduviidae: Arilus spp.): clarification and chemistry. Zeitschrift Fur Naturforschung Section C Journal of Biosciences 68:522–526. https://doi.org/10.1515/znc-2013-11-1211
Alzogaray R, Camps F, Masuh H et al (2005) Behavioural response of Triatoma infestans (Klug) (Hemiptera: Reduviidae) to quinazolines. Molecules 10:1190–1196. https://doi.org/10.3390/molecules10091190
Antunes L, Han J, Pan J et al (2013) Metabolic signatures of triatomine vectors of Trypanosoma cruzi unveiled by metabolomics. PLoS ONE 8:1–12. https://doi.org/10.1371/journal.pone.0077283
Barrera R (2022) New tools for Aedes control: mass trapping. Curr Opin Insect Sci 52. https://doi.org/10.1016/j.cois.2022.100942
Barrozo R, Lazzari C (2004) The response of the blood-sucking bug Triatoma infestans to carbon dioxide and other host odours. Chem Senses 29:319–329. https://doi.org/10.1093/chemse/bjh035
Barrozo R, Reisenman C, Guerenstein P et al (2017) An inside look at the sensory biology of triatomines. J Insect Physiol 97:3–19. https://doi.org/10.1016/j.jinsphys.2016.11.003
Bell MJ, Sedda L, Gonzalez MA et al (2018) Attraction of Lutzomyia longipalpis to synthetic sex-aggregation pheromone: Effect of release rate and proximity of adjacent pheromone sources. PLoS Negl Trop Dis 12:1–19. https://doi.org/10.1371/journal.pntd.0007007
Bohman B, Weinstein A, Unelius C, Lorenzo M (2018) Attraction of Rhodnius prolixus males to a synthetic female-pheromone blend. Parasit Vectors 11:1–7. https://doi.org/10.1186/s13071-018-2997-z
Bohman B, Franke S, Lorenzo M et al (2011) Structure elucidation and synthesis of dioxolanes emitted by two Triatoma species (Hemiptera: Reduviidae). J Nat Prod 74:690–694. https://doi.org/10.1021/np100748r |
Botto-Mahan C, Cattan P, Medel R (2006) Chagas disease parasite induces behavioural changes in the kissing bug Mepraia spinolai. Acta Trop 98:219–223. https://doi.org/10.1016/j.actatropica.2006.05.005
Calderon-Fernández G, Girotti J, Juárez P (2011) Cuticular hydrocarbons of Triatoma dimidiata (Hemiptera: Reduviidae): intraspecific variation and chemotaxonomy. J Med Entomol 48:262–271. https://doi.org/10.1603/ME10141
Campetella F, Ignell R, Beutel R et al (2021) Comparative dissection of the peripheral olfactory system of the Chagas disease vectors Rhodnius prolixus and Rhodnius brethesi. PLoS Negl Trop Dis 15:1–23. https://doi.org/10.1371/journal.pntd.0009098
Castillo-Neyra R, Barbu C, Salazar R et al (2015) Host-seeking behavior and dispersal of Triatoma infestans, a vector of Chagas disease, under semi-field conditions. PLoS Negl Trop Dis 9:1–12. https://doi.org/10.1371/journal.pntd.0003433
Ceccarelli S, Justi S, Rabinovich J et al (2020) Phylogenetic structure of geographical co-occurrence among New World Triatominae species, vectors of Chagas disease. J Biogeogr 47:1218–1231. https://doi.org/10.1111/jbi.13810
Chacón F, Muñoz-San Martín C, Bacigalupo A et al (2022) Trypanosoma cruzi parasite load modulates the circadian activity pattern of Triatoma infestans. Insects 13:1–12. https://doi.org/10.3390/insects13010076
Chanda E, Ameneshewa B, Bagayoko M et al (2017) Harnessing integrated vector management for enhanced disease prevention. Trends Parasitol 33:30–41. https://doi.org/10.1016/j.pt.2016.09.006
Choo Y-M, Xu P, Hwang J et al (2018) Reverse chemical ecology approach for the identification of an oviposition attractant for Culex quinquefasciatus. PNAS 115:714–719. https://doi.org/10.1073/pnas.1718284115
Cocchiararo-Bastias L, Mijailovsky S, Calderon-Fernández G et al (2011) Epicuticle lipids mediate mate recognition in Triatoma infestans. J Chem Ecol 37:246–252. https://doi.org/10.1007/s10886-011-9927-2
Córdoba-Aguilar A (2020) Chagas bugs and Trypanosoma cruzi: Puppets and puppeteer? Acta Trop 211. https://doi.org/10.1016/j.actatropica.2020.105600
Crespo J, Manrique G (2007) Mating behavior of the hematophagous bug Triatoma infestans: Role of Brindley’s and metasternal glands. J Insect Physiol 53:708–714. https://doi.org/10.1016/j.jinsphys.2007.03.014
Cruz-López L, Morgan E (1995) Chemical investigation of aggregation behavior of Triatoma bugs (Hemiptera: Reduviidae). J Chem Ecol 21:2069–2078. https://doi.org/10.1007/BF02033863
Cruz-López L, Malo E, Rojas J (1993) Aggregation pheromone in five species of Triatominae (Hemiptera: Reduvidae). Mem Inst Oswaldo Cruz 88:535–539. https://doi.org/10.1590/S0074-02761993000400006
Cruz-López L, Malo E, Rojas J, Morgan E (2001) Chemical ecology of triatomine bugs: vectors of Chagas disease. Med Vet Entomol 15:351–357. https://doi.org/10.1046/j.0269-283x.2001.00340.x
di Giovanni F, Wilke ABB, Beier JC et al (2021) Parasitic strategies of arthropods of medical and veterinary importance. Entomologia Generalis 41:511–522. https://doi.org/10.1127/entomologia/2021/1155
Dulbecco A, Moriconi D, Lynn S et al (2020) Deciphering the role of Rhodnius prolixus CYP4G genes in straight and methyl-branched hydrocarbon formation and in desiccation tolerance. Insect Mol Biol 29:431–443. https://doi.org/10.1111/imb.12653
Falvo M, Lorenzo-Figueiras A, Manrique G (2016) Spatio-temporal analysis of the role of faecal depositions in aggregation behaviour of the triatomine Rhodnius prolixus. Physiol Entomol 41:24–30. https://doi.org/10.1111/phen.12120
Faro de Brito N, Moreira M, Melo A (2016) A look inside odorant-binding proteins in insect chemoreception. J Insect Physiol 95:51–65. https://doi.org/10.1016/j.jinsphys.2016.09.008
Ferreira R, Guarneri A, Lorenzo M (2019) Activity and shelter-related behavior in Rhodnius prolixus: The role of host odours. Acta Trop 196:150–154. https://doi.org/10.1016/j.actatropica.2019.05.022
Figueiras A, Girotti J, Mijailovsky S, Juárez P (2009) Epicuticular lipids induce aggregation in Chagas disease vectors. Parasit Vectors 2:1–7. https://doi.org/10.1186/1756-3305-2-8
Fontan A, Audino P, Martinez A et al (2002a) Attractant volatiles released by female and male Triatoma infestans (Hemiptera: Reduviidae), a vector of chagas disease: chemical analysis and behavioral bioassay. J Med Entomol 39:191–197. https://doi.org/10.1603/0022-2585-39.1.191
Fontan A, Gonzalez P, Martinez A et al (2002b) Attractant volatiles released by female and male Triatoma infestans (Hemiptera: Reduviidae), a vector of Chagas disease: chemical analysis and behavioral bioassay. J Med Entomol 39:191–197. https://doi.org/10.1603/0022-2585-39.1.191
Franco T, Oliveira D, Moreira M et al (2016) Silencing the odorant receptor co-receptor RproOrco affects the physiology and behavior of the Chagas disease vector Rhodnius. Insect Biochem Mol Biol 69:82–90. https://doi.org/10.1016/j.ibmb.2015.02.012
Franco TA, Xu P, Brito N et al (2018) Reverse chemical ecology-based approach leading to the accidental discovery of repellents for Rhodnius prolixus, a vector of Chagas diseases refractory to DEET. Insect Biochem Mol Biol 103:46–52. https://doi.org/10.1016/j.ibmb.2018.10.004
Gäde G, Marco H (2022) The adipokinetic peptides of Hemiptera: structure, function, and evolutionary trends. Frontiers in Insect Science 2:1–24. https://doi.org/10.3389/finsc.2022.891615The
Galvez-Marroquin Z, Cruz-López L, Malo E et al (2018) Behavioural and electrophysiological responses of Triatoma dimidiata nymphs to conspecific faecal volatiles. Med Vet Entomol 32:102–110. https://doi.org/10.1111/mve.12271
González A, Alzogaray R, Vassena C et al (2007) Volatile compounds secreted by Brindley’s glands of adult Triatoma infestans: identification and biological activity of previously unidentified compounds. J Vector Ecol 32:75–82. https://doi.org/10.3376/1081-1710(2007)32[75:VCSBBO]2.0.CO;2
Gries R, Britton R, Holmes M et al (2014) Bed bug aggregation pheromone finally identified. Angewandte Chemie International Edition 54:1–5. https://doi.org/10.1002/anie.201409890
Guerenstein P, Guerin P (2001) Olfactory and behavioural responses of the blood-sucking bug Triatoma infestans to odours of vertebrate hosts. J Exp Biol 204:585–597
Guerenstein PG, Guerin PM (2004) A comparison of volatiles emitted by adults of three triatomine species. Entomol Exp Appl 111:151–155. https://doi.org/10.1111/j.0013-8703.2004.00160.x
Guerenstein P, Lazzari C (2009) Host-seeking: How triatomines acquire and make use of information to find blood. Acta Trop 110:148–158. https://doi.org/10.1016/j.actatropica.2008.09.019
Guerenstein P, Lazzari C (2010) The role of olfaction in host seeking of triatomine bugs. In: Knols B, Takken W (eds) Olfaction in vector-host interactions. Wageningen Academic Publishers, Wageningen, pp 309–325
Guidobaldi F, Guerenstein P (2013) Evaluation of a CO2-free commercial mosquito attractant to capture triatomines in the laboratory. J Vector Ecol 38:245–250. https://doi.org/10.1111/j.1948-7134.2013.12037.x
Guidobaldi F, Guerenstein P (2015) Oviposition in the blood-sucking insect Rhodnius prolixus is modulated by host odors. Parasit Vectors 8:1–7. https://doi.org/10.1186/s13071-015-0867-5
Guidobaldi F, Guerenstein P (2016) A CO2-free synthetic host–odor mixture that attracts and captures triatomines: Effect of emitted odorant ratios. J Med Entomol 53:770–775. https://doi.org/10.1093/jme/tjw057
Juarez P, Blomquist G, Schofield C (2001) Hydrocarbons of Rhodnius prolixus, a Chagas disease vector. Comp Biochem Physiol 129B:733–746
Juárez P, Blomquist G (1993) Cuticular hydrocarbons of Triatoma infestans and T. mazzotti. Comp Biochem Physiol 106B:667–674
Juárez MP, Fernández GC (2007) Cuticular hydrocarbons of triatomines. Comp Biochem Physiol 147A:711–730. https://doi.org/10.1016/j.cbpa.2006.08.031
Justi S, Galvão C (2017) The evolutionary origin of diversity in Chagas disease vectors. Trends Parasitol 33:42–52. https://doi.org/10.1016/j.pt.2016.11.002
Kamala J, Kempraj V, Aurade R et al (2014) Computational reverse chemical ecology: virtual screening and predicting behaviorally active semiochemicals for Bactrocera dorsalis. BMC Genomics 15:1–7. https://doi.org/10.1186/1471-2164-15-209
Kotaki T, Shinada T, Numata H (2009) Structure determination of a natural juvenile hormone isolated from a heteropteran insect. Org Lett 11:5234–5237. https://doi.org/10.1155/2012/924256
Kuhlisch C, Pohnert G (2015) Metabolomics in chemical ecology. Nat Prod Rep 32:937–955. https://doi.org/10.1039/c5np00003c
Latorre-Estivalis J (2022) Molecular basis of olfaction in kissing bugs. In: Ignell R, Lazzari C, Lorenzo M, Hill S (eds) Sensory ecology of disease vectors. Wageningen Academic Publishers, Wageningen, pp 153–170
Latorre-Estivalis J, Omondi B, Desouza O et al (2015) Molecular basis of peripheral olfactory plasticity in Rhodnius prolixus, a Chagas disease vector. Front Ecol Evol 3:1–9. https://doi.org/10.3389/fevo.2015.00074
Latorre-Estivalis J, Robertson H, Walden K et al (2017) The molecular sensory machinery of a Chagas disease vector: expression changes through imaginal moult and sexually dimorphic features. Sci Rep 7:1–16. https://doi.org/10.1038/srep40049
Latorre-Estivalis J, Grobe-Wilde E, da Rocha G et al (2022) Changes in antennal gene expression underlying sensory system maturation in Rhodnius prolixus. Insect Biochem Mol Biol 140:1–15. https://www.sciencedirect.com/science/article/abs/pii/S0965174821001879?via%3Dihub
Lazzari C (2022) Modulation of host-seeking behaviour in kissing bugs. In: Ignell R, Lazzari C, Lorenzo M, Hill S (eds) Sensory ecology of disease vectors. Wageningen Academic Publishers, Wageningen, pp 801–813
Lazzari CR, Lorenzo MG (2009) Exploiting triatomine behaviour: alternative perspectives for their control. Mem Inst Oswaldo Cruz 104:65–70. https://doi.org/10.1590/s0074-02762009000900011
Lazzari C (2021) The behaviour of kissing bugs. In: Guarneri A, Lorenzo M (eds) The biology of Chagas disease vectors. Springer, Cham, pp 215–238
Leal W (2014) Deciphering the Rosetta stone of insect chemical communication. Am Entomol 60:223–230. https://doi.org/10.1093/ae/60.4.223
Leal W (2017) Reverse chemical ecology at the service of conservation biology. PNAS 114:12094–12096. https://doi.org/10.1073/pnas.1717375114
Leal W, Barbosa M, Xu W et al (2008) Reverse and conventional chemical ecology approaches for the development of oviposition attractants for Culex mosquitoes. PLoS ONE 3:1–11. https://doi.org/10.1371/journal.pone.0003045
Leyria J, El-Mawed H, Orchard I, Lange A (2021) Regulation of a trehalose-specific facilitated transporter (TRET) by insulin and adipokinetic hormone in Rhodnius prolixus, a vector of chagas disease. Front Physiol 12:1–21. https://doi.org/10.3389/fphys.2021.624165
Liu F, Chen Z, Ye Z, Liu N (2021) The olfactory chemosensation of hematophagous Hemipteran insects. Front Physiol 12:1–15. https://doi.org/10.3389/fphys.2021.703768
Lizzi KM, Qualls WA, Brown SC, Beier JC (2014) Expanding integrated vector management to promote healthy environments. Trends Parasitol 30:394–400. https://doi.org/10.1016/j.pt.2014.06.001
Lopes R, Santos-Mallet J, Barbosa CF et al (2020) Morphological and ultrastructural analysis of an important place of sexual communication of Rhodnius prolixus (Heteroptera: Reduviidae): The metasternal glands. Tissue Cell 67:1–7. https://doi.org/10.1016/j.tice.2020.101416
Lopes RL, Gomes SAO, Spiegel CN (2022) An overview of studies on sex pheromones of insect vectors of pathogenic trypanosomatids of medical and veterinary importance. Res Soc Dev 11:1–26. https://doi.org/10.33448/rsd-v11i11.32864
Lorenzo MG (2022) Olfactory-driven behaviours in kissing bugs. In: Ignell R, Lazzari C, Lorenzo M, Hill S (eds) Sensory ecology of disease vectors. Wageningen Academic Publishers, Wageningen, pp 277–296
Lorenzo MG, Vidal DM, Zarbin PHG (2014) Control of neglected disease insect vectors: future prospects for the use of tools based on behavior manipulation-interference. J Braz Chem Soc 25:1799–1809. https://doi.org/10.5935/0103-5053.20140202
Lorenzo-Figueiras A, Lazzari C (1998) Aggregation behaviour and interspecific responses in three species of Triatominae. Mem Inst Oswaldo Cruz 93:133–137. https://doi.org/10.1590/S0074-02761998000100025
Manrique G, Lorenzo M (2012) The sexual behaviour of Chagas’ disease vectors: chemical signals mediating communication between male and female triatomine bugs. Psyche (London) 862891:1–8. https://doi.org/10.1155/2012/862891
Manrique G, Vitta A, Ferreira R et al (2006) Chemical communication in Chagas disease vectors. Source, identity, and potential function of volatiles released by the metasternal and Brindley’s glands of Triatoma infestans adults. J Chem Ecol 32:2035–2052. https://doi.org/10.1007/s10886-006-9127-7
Marchant A, Mougel F, Jacquin-Joly E et al (2021) Chemosensory gene expression for two closely relative species Rhodnius robustus and R. prolixus (Hemiptera, Reduviidae, Triatominae) vectors of Chagas disease. Front Ecol Evol 9:1–15. https://doi.org/10.3389/fevo.2021.725504
Marco H, Simek P, Clark K, Gäde G (2013) Novel adipokinetic hormones in the kissing bugs Rhodnius prolixus, Triatoma infestans, Dipetalogaster maxima and Panstrongylus megistus. Peptides (NY) 41:21–30. https://doi.org/10.1016/j.peptides.2012.09.032
Marco H, König S, Gäde G (2022) Mass spectrometric proof of predicted peptides: novel adipokinetic hormones in insects. Molecules 27:1–17. https://doi.org/10.3390/molecules27196469
May-Concha I, Rojas J, Cruz-López L et al (2013) Volatile compounds emitted by Triatoma dimidiata, a vector of Chagas disease: chemical analysis and behavioural evaluation. Med Vet Entomol 27:165–174. https://doi.org/10.1111/j.1365-2915.2012.01056.x
May-Concha I, Rojas J, Cruz-lópez L et al (2015) Volatile compound diversity and conserved alarm behaviour in Triatoma dimidiata. Parasit Vectors 8:1–14. https://doi.org/10.1186/s13071-015-0678-8
May-Concha I, Cruz-lópez L, Rojas J, Ramsey J (2018a) “Sweeter than a rose”, at least to Triatoma phyllosoma complex males (Triatominae: Reduviidae). Parasit Vectors 11:1–12. https://doi.org/10.1186/s13071-018-2677-z
May-Concha I, Guerenstein P, Malo E et al (2018b) Electroantennogram responses of the Triatoma dimidiata complex to volatiles produced by its exocrine glands. Acta Trop 185:336–343. https://doi.org/10.1016/j.actatropica.2018.06.018
May-Concha I, Lobbia P, Mougabure-Cueto G (2018c) Interaction between two aggregation chemical signals in Triatoma infestans (Hemiptera: Reduviidae). J Insect Physiol 109:79–84. https://doi.org/10.1016/j.jinsphys.2018.06.007
May-Concha I, Remón C, Mougabure-Cueto G (2020) Behavioral response mediated by feces in Triatoma infestans (Hemiptera: Reduviidae: triatominae) susceptible and resistant to deltamethrin. Acta Trop 206:105442. https://doi.org/10.1016/j.actatropica.2020.105442
May-Concha I, Escalante-Talavera M, Dujardin J, Waleckx E (2021) Trypanosoma cruzi affects the sensory biology of Triatoma dimidiata (Hemiptera: Reduviidae). Res Sq,pp 1–25. https://doi.org/10.21203/rs.3.rs-589244/v1
Milne M, Ross E, Sonenshine D (2009) Attraction of Triatoma dimidiata and Rhodnius prolixus (Hemiptera: Reduviidae) to combinations of host cues tested at two distances. J Med Entomol 46:1062–1073. https://doi.org/10.1603/033.046.0513
Minoli S, Palottini F, Crespo J, Manrique G (2013a) Dislodgement effect of natural semiochemicals released by disturbed triatomines: a possible alternative monitoring tool. J Vector Ecol 38:353–360. https://doi.org/10.1111/j.1948-7134.2013.12051.x
Minoli S, Palottini F, Manrique G (2013b) The main component of an alarm pheromone of kissing bugs plays multiple roles in the cognitive modulation of the escape response. Front Behav Neurosci 7:1–10. https://doi.org/10.3389/fnbeh.2013.00077
Minoli S, Cano A, Pontes G et al (2018) Learning spatial aversion is sensory-specific in the hematophagous insect Rhodnius prolixus. Front Psychol 9:1–11. https://doi.org/10.3389/fpsyg.2018.00989
Mitaka Y, Matsuyama S, Mizumoto N et al (2020) Chemical identification of an aggregation pheromone in the termite Reticulitermes speratus. Sci Rep 10:1–10. https://doi.org/10.1038/s41598-020-64388-4
Mitchell RF, Curkovic T, Mongold-diers J et al (2017) Evidence that Cerambycid beetles mimic Vespid wasps in odor as well as appearance. J Chem Ecol 43:75–83. https://doi.org/10.1007/s10886-016-0800-1.EVIDENCE
Mori K (2007) Significance of chirality in pheromone science. Bioorg Med Chem 15:7505–7523. https://doi.org/10.1016/j.bmc.2007.08.040
Mosquera K, Lorenzo M (2020) Triatomines of the genus Rhodnius do not mark shelters with feces. J Chem Ecol 46:865–870. https://doi.org/10.1007/s10886-020-01199-x
Mota T, Vitta A, Lorenzo-Figueiras A et al (2014) A multi-species bait for Chagas disease vectors. PLoS Negl Trop Dis 8:1–8. https://doi.org/10.1371/journal.pntd.0002677
Müller C, Caspers B, Gadau J, Kaiser S (2020) The power of infochemicals in mediating individualized niches. Trends Ecol Evol 35:981–989. https://doi.org/10.1016/j.tree.2020.07.001
Mweresa C, Mukabana W, van Loon J et al (2020) Use of semiochemicals for surveillance and control of hematophagous insects. Chemoecology 30:277–286. https://doi.org/10.1007/s00049-020-00317-1
Noireau F, Dujardin J-P (2010) Biology of Triatominae. In: Telleria J, Tibayrec M (eds) American Trypanosomiasis Chagas Disease One Hundred Years of Research, First. Elsevier, London, pp 149–168
Nojima S, Kiemle D, Webster F et al (2011) Nanogram-scale preparation and NMR analysis for mass-limited small volatile compounds. PLoS ONE 6:1–7. https://doi.org/10.1371/journal.pone.0018178
Ohmura W, Hishiyama S, Nakashima T et al (2009) Chemical composition of the defensive secretion of the longhorned beetle, Chloridolum loochooanum. J Chem Ecol 35:250–255. https://doi.org/10.1007/s10886-009-9591-y
Oliveira D, Brito N, Nogueira F et al (2017) Proteomic analysis of the kissing bug Rhodnius prolixus antenna. J Insect Physiol 100:108–118. https://doi.org/10.1016/j.jinsphys.2017.06.004
Oliveira D, Brito N, Franco T et al (2018) Functional characterization of odorant binding protein 27 (RproOBP27) from Rhodnius prolixus antennae. Front Physiol 9:1–11. https://doi.org/10.3389/fphys.2018.01175
Olsson SB, Challiss RAJ, Cole M et al (2015) Biosynthetic infochemical communication. Bioinspir Biomim 10:1–12. https://doi.org/10.1088/1748-3190/10/4/043001
Otálora-Luna F, Guerin P (2014) Amines from vertebrates guide triatomine bugs to resources. J Insect Physiol 71:52–60. https://doi.org/10.1016/j.jinsphys.2014.09.007
Otálora-Luna F, Perret J, Guerin P (2004) Appetence behaviours of the triatomine bug Rhodnius prolixus on a servosphere in response to the host metabolites carbon dioxide and ammonia. J Comp Physiol A 190:847–854. https://doi.org/10.1007/s00359-004-0540-5
Palottini F, Manrique G (2016) Compounds released by disturbed adults of the haematophagous bug Triatoma infestans (Hemiptera: Reduviidae): behavioural effects of single compounds. Physiol Entomol 41:234–240. https://doi.org/10.1111/phen.12147
Palottini F, González A, Manrique G (2014) Filling dynamics of the Brindley’s glands in the blood-sucking bug Triatoma infestans (Hemiptera: Reduviidae). J Insect Physiol 71:122–127. https://doi.org/10.1016/j.jinsphys.2014.10.012
Pires H, Lorenzo M, Diotaiuti L et al (2002) Aggregation behaviour in Panstrongylus megistus and Triatoma infestans: Inter and intraspecific responses. Acta Trop 81:47–52. https://doi.org/10.1016/S0001-706X(01)00185-1
Pontes G, Bohman B, Unelius R, Lorenzo M (2008) Metasternal gland volatiles and sexual communication in the triatomine bug, Rhodnius prolixus. J Chem Ecol 34:450–457. https://doi.org/10.1007/s10886-008-9431-5
Prince EK, Pohnert G (2010) Searching for signals in the noise: metabolomics in chemical ecology. Anal Bioanal Chem 396:193–197. https://doi.org/10.1007/s00216-009-3162-5
Raguso RA, Agrawal AA, Douglas AE et al (2015) The raison d’être of chemical ecology. Ecology 96:617–630. https://doi.org/10.1890/14-1474.1
Ramírez M, Ortiz M, Guerenstein P, Molina J (2020) Novel repellents for the blood-sucking insects Rhodnius prolixus and Triatoma infestans, vectors of Chagas disease. Parasit Vectors 13:1–9. https://doi.org/10.1186/s13071-020-04013-5
Ramos F, Leyria J, Nouzova M et al (2021) Juvenile hormone mediates lipid storage in the oocytes of Dipetalogaster maxima. Insect Biochem Mol Biol 133:103499. https://doi.org/10.1016/j.ibmb.2020.103499
Riddiford L (2020) Rhodnius, golden oil, and Met: a history of juvenile hormone. Front Cell Dev Biol 8:1–18. https://doi.org/10.3389/fcell.2020.00679
Rihani K, Ferveur J, Briand L (2021) The 40-year mystery of insect odorant-binding proteins. Biomolecules 11:2–27
Rocha Pires H, Lorenzo M, Lazzari C et al (2004) The sexual behaviour of Panstrongylus megistus (Hemiptera: Reduviidae): an experimental study. Mem Inst Oswaldo Cruz 99:295–300. https://doi.org/10.1590/S0074-02762004000300010
Rojas J, Ramirez-Rovelo A, Cruz-López L (1991) Search for a sex pheromone in Triatoma mazzottii (Hemiptera: Reduviidae). J Med Entomol 28:469–470
Rojas J, Rios-Candelaria E, Cruz-López L et al (2002) A reinvestigation of Brindley’s gland exocrine compounds of Rhodnius prolixus (Hemiptera: Reduviidae). J Med Entomol 39:256–265. https://doi.org/10.1603/0022-2585-39.2.256
Rojas de Arias A, Abad-Franch F, Acosta N et al (2012) Post-control surveillance of Triatoma infestans and Triatoma sordida with chemically-baited sticky traps. PLoS Negl Trop Dis 6:1–10. https://doi.org/10.1371/journal.pntd.0001822
Ryelandt J, Noireau F, Lazzari C (2011) A multimodal bait for trapping blood-sucking arthropods. Acta Trop 117:131–136. https://doi.org/10.1016/j.actatropica.2010.11.005
Sainz P, Sanz J, Burillo J et al (2012) Essential oils for the control of reduviid insects. Phytochem Rev 11:361–369. https://doi.org/10.1007/s11101-012-9261-5
Santiago PB, de Araújo CN, Charneau S et al (2020) The pharmacopea within Triatomine salivary glands. Trends Parasitol 36:250–265
Schofield C, Galvão C (2009) Classification, evolution, and species groups within the Triatominae. Acta Trop 110:88–100. https://doi.org/10.1016/j.actatropica.2009.01.010
Sparks JT, Bohbot JD, Dickens JC (2015) Olfactory disruption: Toward controlling important insect vectors of disease, 1st edn. Elsevier Inc., Amsterdam.
Syed Z (2015) Chemical ecology and olfaction in arthropod vectors of diseases. Curr Opin Insect Sci 10:83–89. https://doi.org/10.1016/j.cois.2015.04.011
Tirindelli R, Dibattista M, Pifferi S, Menini A (2009) From pheromones to behavior. Physiol Rev 89:921–956. https://doi.org/10.1152/physrev.00037.2008.-In
Tobias N, Latorre-Estivalis J (2021) Perspectives in triatomine biology studies: “OMICS” -based approaches. In: Guarneri A, Lorenzo M (eds) Triatominae—the biology of Chagas disease vectors. Springer, Cham, pp 557–592
Unelius CR, Bohman B, Lorenzo MG et al (2010) A new volatile released by a triatomine bug. Org Lett 12:5601–5603. https://doi.org/10.1021/ol102165q
Vitta A, Lorenzo Figueiras A, Lazzari C et al (2002) Aggregation mediated by faeces and footprints in Triatoma pseudomaculata (Heteroptera: Reduviidae), a Chagas disease vector. Mem Inst Oswaldo Cruz 97:865–867. https://doi.org/10.1590/S0074-02762002000600020
Vitta A, Mota T, Diotaiuti L, Lorenzo M (2007) The use of aggregation signals by Triatoma brasiliensis (Heteroptera: Reduviidae). Acta Trop 101:147–152. https://doi.org/10.1016/j.actatropica.2006.10.013
Vitta A, Bohman B, Unelius C, Lorenzo M (2009) Behavioral and electrophysiological responses of Triatoma brasiliensis males to volatiles produced in the metasternal glands of females. J Chem Ecol 35:1212–1221. https://doi.org/10.1007/s10886-009-9709-2
Wehrenfennig C, Schott M, Gasch T et al (2013) On-site airborne pheromone sensing. Anal Bioanal Chem 405:6389–6403. https://doi.org/10.1007/s00216-013-7113-9
Wooding M, Naudé Y, Rohwer E, Bouwer M (2020) Controlling mosquitoes with semiochemicals: a review. Parasit Vectors 13. https://doi.org/10.1186/s13071-020-3960-3
Yew J, Chung H (2015) Insect pheromones: an overview of function, form, and discovery. Prog Lipid Res 59:88–105. https://doi.org/10.1016/j.plipres.2015.06.001
Acknowledgements
The authors are grateful to the Editor Dr. Nicolas Desneux for inviting this Review on Journal of Pest Science. Two anonymous reviewers kindly improved an earlier version of the manuscript.
Funding
Part of this work was supported by The National Council of Science and Technology (CONACyT), with the project “Ciencia de Frontera 2019, clave 376136, número 292”, and CONTEX “Unifying Texan and Mexican efforts towards controlling Chagas disease by deducing parasite-vector dynamics”, ECO-IE-503.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing financial interest.
Additional information
Communicated by Andre Wilke.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10340_2023_1678_MOESM2_ESM.pptx
The reverse chemical ecology approach to elucidate the role of infochemicals in triatomines. 4-Methylciclohexanol (1), γ-octalactone (2), acetophenone (3), and 2-heptanone (4), effective repellents of Rhodnius prolixus, discovered by serendipity using a reversechemical ecology protocol (Franco et al. 2018) (PPTX 3195 KB)
10340_2023_1678_MOESM3_ESM.pptx
Schematic representation of odour perception in triatomines. OBP; Odour Binding Protein, OR; Odour Receptor, ORCO; Odour Receptor Coreceptor, ORN; Odour Receptor Neuron (PPTX 2136 KB)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Alavez-Rosas, D., Vargas-Abasolo, R., Albores-Flores, C.I. et al. Chemical ecology of triatomines: current knowledge and implications for Chagas disease vector management. J Pest Sci 97, 507–520 (2024). https://doi.org/10.1007/s10340-023-01678-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-023-01678-6