Abstract
Microplastics are emerging contaminants that have been detected recently in most environmental and biological systems, yet their health risk for humans has not been clearly summarized. Here we review human health risk associated with exposure to microplastics with focus on methods of exposure assessment, hazard identification, dose–response assessment, exposure assessment, and risk characterization. Hazards include direct hazards, hazards from contaminants released by microplastics, and hazards from microplastic interactions with surrounding contaminants. Microplastics trigger oxidative stress, disrupt metabolism, interfere with gut microflora and gastrointestinal functions, disrupt hepatic, cardiopulmonary and immune systems, and degrade reproductive health. Some additives leached from microplastics such as phthalates are endocrine disruptors and thus impact reproductive health. The interaction of microplastics with other pollutants in the environment induces varied hazards following synergistic or antagonistic effects.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
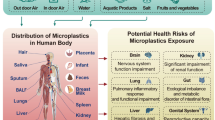
The ubiquitous presence of microplastics in the environment is raising global attention. Microplastics can be termed either primary microplastics or secondary microplastics, with the former originating from microbeads in personal care products, microfibers from synthetic fabrics and wearing of vehicle tires for instance, while the latter deriving from the breakdown of large plastic items entering the environment. Microplastics can enter water and soil directly or be distributed by wind and rain into different spheres of the environment (Fig. 1) (Tang 2022). In the atmospheric environment, microplastics have been detected in both indoor and outdoor air. Microplastics concentrations of 1 particle/m3 to 1583 particles/m3 have been reported in the indoor environment (Zhao et al. 2023). Microplastics deposition rates between 22 fibers/m2/day and 9900 particles/m2/day were observed indoor (Soltani et al. 2021; Zhang et al. 2020). In the outdoor environment, the concentration of suspended microplastics could be as high as 2.84 particles/m3. Microplastics deposition was even higher with 154,000 particles/L of snow reported in some European cities and up to 14,400 particles/L retrieved from the Arctic snow samples, indicating that microplastics have permeated the remote regions of the world (Bergmann et al. 2023; Tang 2023a). In some instances, microplastics in the indoor environment are higher than those outdoor due to indoor sources such as synthetic clothing as well as the surface finishes of household items and furniture which release microplastics upon wear and tear. Besides, outdoor microplastics could drift indoor and are deposited due to the slower airflow of the indoor environment and the presence of items acting as interceptors of airborne MP (Dris et al. 2017; Kacprzak and Tijing 2022).
Aquatic and terrestrial plastic pollution has resulted in the entry of microplastics into the food chain. Microplastics have polluted various food items consumed by humans. Humans are also exposed to airborne microplastics. The entry of microplastics to human body is evident through their detection in human feces. The exposure to microplastics could adversely affect multiple organs
Microplastics are widely present in various waterbodies (Fig. 1). A study on the microplastics in the sediment and water samples of the North Sea revealed the presence of microplastics in all the samples with concentrations ranging from 0.1 to 245.4 particles/m3 and 2.8–1188.8 particles/kg in the water and sediment samples, respectively. Majority of the microplastics in the samples were of sizes < 100 µm (Lorenz et al. 2019). Microplastics were also detected in remote marine environment such as the water of the Arctic Central Basin with their concentrations varying at different depths from the surface. The Polar Mixed Layer contained the highest abundance of microplastics of 0–375 particles/m3 followed by the deep and bottom water (0–104 particles/m3) (Kanhai et al. 2020). Unsurprisingly, the presence of microplastics has been reported for freshwater environments at widely varying concentrations. Sediment and surface water samples of the Wei River in China were found to contain 1020 particles/L (0.918 g/L) of microplastics in comparison with those of the Taihu Lake in China with 123 particles/L (Su et al. 2016). In Australia, surface water samples taken from the Maribyrnong and Yarra Rivers contained an average of 2868 particles/L or 2.5803 g/L of microplastics, while sediment samples from the Bloukrans River contained an average of 240 particles/L (0.216 g/L) of microplastics (Kowalczyk et al. 2017; Nel et al. 2019). Contamination of the freshwater environment by microplastics is partly attributed to surface runoffs containing microplastics (Tang 2023b; Helcoski et al. 2020; Weber and Opp 2020).
The environmental prevalence of microplastics has led to their entry into food chains (Fig. 1). Jabeen et al. (2017) revealed the presence of microplastics and mesoplastics in various seawater fish sold in fish markets in Shanghai. Another study by Markic et al. (2018) also highlighted the presence of marine plastics in 33 of the 34 species of commercial fish sampled at an abundance of 2.4 particles per fish. In addition, analysis of single-use polyethylene terephthalate (PET)-bottled water samples in Bangkok, Thailand, showed microplastics of ≥ 50 µm amounting to 140 particles/L, while samples of glass bottled water in Germany contained 6292 particles/L of microplastics sized > 5 µm (Kankanige and Babel 2020; Oßmann et al. 2018). Microplastics concentrations in salt samples varied regionally with 12 particles/kg reported for sea salt samples in Bulgaria, 550–680 particles/kg for those in China and 1400 particles/kg for samples in Indonesia (Kim et al. 2018). With microplastics in human food, human exposure to microplastics is inevitable (Fig. 1). Evidence has pointed to the detection of microplastics in human specimens. A study on the abundance of microplastics in human feces revealed 20 particles of microplastics per 10 g of stool samples collected in Vienna, Australia (Schwabl et al. 2019). Human exposure to airborne microplastics was indicated through a study on the lung tissue samples taken from Sao Paulo, Brazil, unveiling an average microplastic abundance of 0.59 particles per g of lung tissues or 470 particles per lung (Amato-Lourenço et al. 2021) (Fig. 1). However, few studies are devoted to assessing the risk associated with human exposure to microplastics. Most current studies on microplastics risk assessment focus on the ecological aspect (Redondo-Hasselerharm et al. 2023; Chau et al. 2023). There was a preliminary attempt to assess human exposure to microplastics through intake of microplastics-contaminated food (Lin et al. 2022). However, the health risk was not characterized. To fill in the gap, this review aims to present a preliminary assessment of health risk related to human exposure to microplastics with published exposure and chemical data using a conservative approach. It has the novelty of integrating various published data on the concentrations of microplastics in air, drinking water and food items, their exposure through ingestion and inhalation primarily, and the toxicity of microplastics to conduct a conservative risk assessment, which has rarely been included in previous studies on human health effects of microplastics.
Human risk assessment
Human health risk assessment typically comprises four steps, namely hazard identification, dose–response assessment, exposure assessment, and risk characterization. Hazard identification involves the identification of whether exposure to microplastics and the common chemicals leached from microplastics could lead to adverse health effects (Zhang et al. 2023). This was accomplished through a review of the relevant literature related to the hazards of microplastics, as well as the hazards resulted from plastic additives leached from microplastics and the interactions of microplastics with pollutants mainly in the aqueous environment. Dose–response assessment revolves around an evaluation of the likelihood of developing a response or adverse effect as a function of the dose administered (Zhang et al. 2023). This paper presents the toxicological or safe doses of microplastics as well as the common plastic additives in microplastics through a literature review.
During exposure assessment, human exposure to microplastics is typically calculated with respect to the extent, frequency and duration of such exposure (Senathirajah et al. 2021). A typical equation used for this calculation is E = C × R × T, where E is the exposure (in mg or µg), C is the concentration of the chemical in the medium of exposure (in mg/L or µg/L for water, mg/m3 or µg/m3 for air, mg/kg or µg/kg for soil or food), R is the intake rate or contact rate of the medium of exposure (in L/day for water, m3/day for air, kg/day for soil or food, cm2/day for skin) and T is the duration of exposure (in days) (US EPA 2019). In this section, the sources of microplastics and the major exposure pathways were described. A literature review was conducted to obtain the exposure or intake rates of microplastics through the major pathways. Finally, during risk characterization, the nature of risk was evaluated with reference to the toxicological/safe doses of microplastics and the exposure rates (Zhang et al. 2023). A conservative approach using global averages and higher exposure rates to capture the worst-case scenarios, as well as an uncertainty factor of 100, was used in assessing the risk. A conservative approach is akin to Tier 0 of risk assessment of the Organization for Economic Cooperation and Development where all types of microplastics were considered as a group of substance without making distinction of the toxicities of different types, shapes and sizes of microplastics. In fact, it is shown in subsequent hazard identification that different types of microplastics might have similar toxic effects, particularly the effects on reproductive health and the gastrointestinal system. An uncertainty factor of 100 is typically used to generate safe values or reference doses for human exposure from studies conducted on animals (Walton et al. 2001). This is according to the Reference Dose: Description and Use in Health Risk Assessments published by the US Environmental Protection Agency, which provides guidance on the use of uncertainty factors in deriving reference doses. An uncertainty factor of 100 has been assumed to account for both the differences in species and the variability in human sensitivity to the toxic effects of a chemical. The uncertainty factor of 100 is usually the product of two tenfold factors: one for interspecies differences and one for human variability (US EPA 2023). The risk was qualitatively described.
In essence, a literature review was conducted for the first three parts of the risk assessment with these inclusion criteria for the articles reviewed: 1) The articles must be peer-reviewed, scholarly in nature and written in English; 2) The articles must be published in the past 10 years; 3) The articles on hazard identification address the hazards of microplastics alone, the hazards of common additives in microplastics or the hazards from the interactions of microplastics with other pollutants in the environment; 4) The articles on dose–response assessment must present or suggest the toxicological doses of microplastics or the common plastic additives explicitly; 5) The articles on exposure assessment must present the exposure rates through the major exposure pathways. First, literature search was conducted using scholarly journal databases comprising Web of Science, PubMed, Scopus, and ScienceDirect, with key phrases consisting of health impacts, microplastics, cell lines, animal models, in vitro, in vivo, health risk assessment, and a combination of the key phrases such as health impacts of microplastics on animal models. An initial search yielded 434 articles. After screening with inclusion criteria 1 and 2 above, 257 articles remained. Further article selection by skimming of the abstracts and texts using criteria 3, 4 and 5 yielded 121 articles. These articles were included in this review.
Hazard identification
The hazard of microplastics on human has not been well-characterized, and the current studies on microplastic hazards rely predominantly on those conducted on animal models. While multiple studies have been conducted on aquatic organisms to examine the potential impacts of microplastics on their physiological functions and morphology, few have been conducted on higher mammals, resulting in limited information available to enable reliable assessment of microplastics impacts on human health. More studies on human cell lines and epidemiological studies based on human exposure are necessary to provide human-specific impacts of microplastics. Though data from experimental studies on human subjects would be most representative, these studies face significant ethical issues and challenges in the recruitment of subjects. Therefore, human health impacts of microplastics are frequently extrapolated from studies on animal models. Such extrapolation could be limited by the significant differences in the metabolism and toxicokinetics of a chemical between animal species and humans, which may affect the relevance of the observed effects (Domenech and Marcos 2021). The hazards associated with microplastics are frequently related to those of the microplastics themselves, those of the chemicals leached from the microplastics and those resulted from the interactions of microplastics with other pollutants in the environment. To better understand the deleterious effects of microplastics on humans, only studies conducted on mammals and human cell lines were examined.
Direct hazards
The extant studies on the hazards of microplastics employed predominantly the in vivo and in vitro model systems with the former conducted mostly on mice and rats, and the latter conducted on human cell lines. Polystyrene is the most extensively studied microplastic type in terms of its hazards on higher mammals, particularly those in the size range of 0.5–50 µm (Table 1). Polystyrene microplastics of other sizes including those in the nanoscale were also studied. For instance, Wei et al. (2021) tested the cardiotoxicity of polystyrene microplastics sized 500 µm (0.5 mm) on Wistar rats, while some studies examined the toxicity of polystyrene nanoparticles of sizes ranging from 20 to 100 nm (Fournier et al. 2020; Huang et al. 2022). Polyethylene microplastics have also been studied though less frequently than polystyrene microplastics, and in few instances, modified polystyrene microplastics were tested of their toxicity on human cell lines (Li et al. 2020a; Hesler et al. 2019). In vivo studies often involved longer durations of exposure to microplastics, typically 14 days to 90 days than in vitro studies usually lasting for 24 to 48 h (Lim et al. 2021; Hou et al. 2021a; An et al. 2021; Stock et al. 2019). Nonetheless, short-term in vivo exposure to microplastics and plastic nanoparticles have been conducted to examine their accumulation and effects on certain organs such as testes, as well as their effects on behaviors (Estrela et al. 2021; Jin et al. 2021).
In vivo studies revealed that exposure to polystyrene microplastics could disrupt energy and lipid metabolism and trigger oxidative stress in mice (Deng et al. 2017). They demonstrated a size-dependent accumulation in the livers, kidneys, and guts of mice. Polystyrene nanoparticles were also detected in the brains, lungs, and spleens of pregnant Sprague–Dawley rats as well as the brains, hearts, kidneys, livers, and lungs of the fetus, but a correlation with the size of the nanoparticles could not be established since only particles of 20 nm were used in the study (Fournier et al. 2020). However, this pointed to potential translocation of nano-polystyrene from pregnant mice to fetus. Similar to Deng et al. (2017), disrupted liver metabolism leading to lipid disorder was reported by Zheng et al. (2021) (Fig. 2 and Table 1). Lu et al. (2018) observed a disruption of lipid metabolism which was linked to dysbiosis in gut. Alteration of gut flora has been disclosed in multiple studies. Such alteration has been associated with compromised gut and intestinal functions such as reduced intestinal and gut secretion (Jin et al. 2019; Lu et al. 2018), disrupted intestinal barrier in normal mice and dams (Luo et al. 2019), and colitis (Zheng et al. 2021) (Fig. 2 and Table 1). Inflammation of the colon and duodenum was also observed in mice exposed to polyethylene microplastics for 5 weeks and in an instance, damaged organelles in stomachs and spleens were more prevalent among female dams (Li et al. 2020a; Park et al. 2020).
Hazards of microplastics on various human systems and the typical exposure routes of humans to microplastics. Ingestion and inhalation are the major exposure routes. Microplastics can potentially affect multiple systems in human body ranging from the cardiovascular, hepatic, and pulmonary systems to the reproductive and digestive systems
In addition to the hazards on the hepatic and gastrointestinal systems, microplastics exposure was found to have adverse effects on the cardiopulmonary system, evidenced by impaired heart structure and function upon 90-day exposure of Wistar rats to polystyrene microplastics which triggered oxidative stress, apoptosis, pyroptosis and inflammatory responses (Wei et al. 2021). Inflammatory responses in the lungs were observed among Sprague–Dawley rats exposed to polystyrene microplastics for 14 days via inhalation in a dose-dependent manner, as well as mice subjected to intratracheal polystyrene microplastics exposure leading to dose-dependent pulmonary fibrosis (Lim et al. 2021; Li et al. 2022) (Fig. 2 and Table 1). There are relatively more studies on the hazards of microplastics on reproductive system than other bodily systems. An in vitro study revealed polystyrene microplastics exposure caused significant decline in fetal and placental weights (Fournier et al. 2020), which aligns with the findings of Park et al. (2020) reporting a reduction in the body weight of mice offspring, in addition to altered number of live births, and sex ratio upon exposure to polyethylene microplastics. Similarly, pregnant and lactating mice exposed to nano-polystyrene gave birth to offspring with reduced body weights at and after birth, confirming the intergenerational effect of microplastics observed in other studies (Huang et al. 2022; Park et al. 2020; Luo et al. 2019; Fournier et al. 2020). Male mice exposed to polystyrene microplastics were observed to have lower sperm count as well as deteriorated sperm health and qualities in terms of motility, deformity, shedding, apoptosis, and atrophy (Xie et al. 2020; Hou et al. 2021a) (Fig. 2 and Table 1). This was resonated by Wei et al. (2022), reporting the decline of sperm and spermatogenic cell counts and higher sperm deformity among male C57BL/6 mice exposed to polystyrene microplastics for 30 or 44 days. A decrease in testosterone level has been linked to microplastics exposure (Xie et al. 2020; Jin et al. 2021).
Microplastics exposure not only affects the male reproductive health. In female mice, microplastics resulted in a lower count of growing follicles, smaller ovaries, lower pregnancy rate, and fewer embryos (An et al. 2021; Wei et al. 2022) (Fig. 2 and Table 1). The damages on male and female reproductive systems caused by microplastics were frequently associated with oxidative stress, which triggered deleterious responses such as inflammation, apoptosis, and fibrosis (An et al. 2021; Huang et al. 2022; Wei et al. 2022; Jin et al. 2021). This concurs with the findings of Hou et al. (2021b) that exposure to polystyrene microplastics caused apoptosis and pyroptosis of ovarian granulosa cells. The inflammation responses were likely initiated by Wingless/Integrated (Wnt)/β-catenin signaling pathway (An et al. 2021) or p38 mitogen-activated protein kinase signaling pathway (Xie et al. 2020). Microplastics were also shown to trigger immune responses in mice with polyethylene microplastics raising neutrophil and IgA levels in blood serum (Park et al. 2020) (Fig. 2 and Table 1).
Furthermore, microplastics exposure might have cognitive and behavioral implications. da Costa Araújo and Malafaia (2021) found Swiss mice exposed to polyethylene microplastics to aggregate less upon encountering predatory threat probably due to a decline in their manifestation of risk-evaluating behavior. Another study revealed altered cognitive function of Swiss mice exposed to nano-polystyrene for 3 days due to oxidative stress and reduced acetylcholinesterase activity, which did not escalate into locomotor and behavioral modifications (Estrela et al. 2021). However, Rafiee et al. (2018) did not observe significant differences in neurobehavioral effects among adult Wistar male rats exposed to nano-polystyrene for 5 weeks. Cell line studies confirmed cellular accumulation of microplastics but revealed COOH-modified polystyrene microplastics to be weakly embryotoxic and non-genotoxic (Hesler et al. 2019). Stock et al. (2019) revealed higher cellular uptake of smaller polystyrene microplastics and the entry of small microplastics into intestinal wall, but they did not trigger oxidative, inflammatory and immune responses. Despite contradictory findings between in vivo and in vitro studies, it can generally be concluded that polystyrene and polyethylene microplastics pose hazards on multiple systems comprising the hepatic, gastrointestinal, cardiopulmonary, reproductive, and immune systems, often through triggering oxidative stress (Fig. 2). They could also affect cognition, which may or may not translate into behavioral changes. It is worth noting that the dosages of microplastics used in animal exposure studies are often higher than those of real-world human exposure to induce observable effects in a short period of time. This contrasts with the chronic and low-level human exposure to microplastics. Nonetheless, human exposure to microplastics may vary significantly depending on demographic and environmental factors. In severe instances, the exposure could come close to the exposure levels of animal models (Cox et al. 2019).
Hazards of chemicals released by microplastics
Plastics are known to contain a myriad of chemicals. A common group of chemicals added is the functional additives consisting of antistatic agents, antimicrobials, flame retardants, plasticizers, and stabilizers among others (Fig. 3) (Li and Tang 2023). Colorants are also added to impart different colors to plastic products. Besides, fillers such as kaolin, clay, and mica and reinforcing materials such as glass and carbon fibers are in the list of plastic additives (Bridson et al. 2021). The hazards related to leaching of plasticizers from microplastics, particularly phthalates, have been more extensively studied compared to other additives (Li et al. 2024). A study on historical exposure of pregnant women to Di-(2-ethylhexyl) phthalate (DEHP) due to a scandal found an association of the exposure with changes in their thyroid hormones (Huang et al. 2016). Intraperitoneal injection of DEHP on prepuberal mice resulted in hormonal changes probably due to structural changes in theca cells, which could negatively affect the onset of puberty (Lai et al. 2017) (Table 2).
The common chemicals leached from microplastics comprise phthalates, bisphenols, polybrominated diphenyl ethers, and polychlorinated biphenyls. Each of the chemicals poses distinct health hazard. Microplastics can interact with a wide array of pollutants in the environment and the most common pollutants included in co-exposure studies are antimicrobials, leached plastic additives and heavy metals
Another study exposing prepuberal female rats to DEHP shortened the duration of their vaginal opening and lengthened the duration of estrous cycles. The exposure disrupted serum levels of growth hormone and progesterone in addition to hypothalamic functions, leading to precocious puberty (Liu et al. 2018), in parallel to the findings of Lai et al. (2017). Mono-(2-ethylhexyl) phthalate was reported to hinder rat granulosa cell proliferation and upregulate the secretion of steroid hormone (Li et al. 2018). Upon exposing pregnant Wistar rats to diisopentyl phthalate, da Silva et al. (2019) observed downregulated expression of Esr 1 in the pituitary gland and hypothalamus of the offspring. This affected the mating behaviors and sexual motivation of male offspring. Similarly, exposure of human granulosa cell line to dibutyl phthalate (DBP) increased estradiol secretion, which potentially affected steroidogenesis in ovaries (Ma et al. 2019). These studies point to potential hazards of phthalates on reproductive system and development as well as their intergenerational deleterious effects (Table 2).
Bisphenols are another group of plasticizers usually added to polycarbonate plastics to impart rigidity (Fig. 3). Like phthalates, bisphenols were found to disrupt hormonal balance by reducing testosterone level of male rate, while causing testicular oxidative stress. In vivo exposure to bisphenol-A (BPA) showed its potential toxicity on spermatogenesis (Ullah et al. 2018). BPA exposure during fetal-perinatal period adversely affected spermatozoa viability, motility and health (Chioccarelli et al. 2020). Eker et al. (2021) revealed a positive correlation between serum BPA levels and nonfunctional adrenal incidentaloma (NFAI), while Ma et al. (2021) found exposure to BPA and a high-fat diet worsened prediabetic symptoms by impairing glucose tolerance. The study showed possible interference of neuron functions by BPA in mice after 12-week exposure (Ma et al. 2021). Exposing human luteinized granulosa cells to bisphenol-S (BPS) lowered progesterone and estradiol secretion though environmental BPS concentrations were expected to have insignificant effect on steroidogenesis (Amar et al. 2020). The hazards of bisphenols are mostly reproductive, adrenal, diabetic, and neuronal in nature (Table 2).
Polybrominated diphenyl ethers (PBDEs) used as a flame retardant in plastics were found to be carcinogenic to rats and mice through triggering oxidative stress, disrupting hormones and altering molecular reactions in tissues (Dunnick et al. 2018). Human exposure to PBDEs in indoor dust adversely affected reproductive health, particularly semen volume, sperm count and motility, while interfering the balance of reproductive and thyroid hormones (Yu et al. 2019). In female humans, PBDEs could lengthen menstrual cycle and duration (Shi et al. 2022). 4-bromodiphenyl ether (BDE-3), another type of flame retardant, was reported to stimulate adrenal cells, causing elevated serum aldosterone and corticosterone levels (Chen et al. 2019). Plastics may contain metal-based additives such as lead (Pb) and chromium (Cr). Karaulov et al. (2022) found mice exposed to chromium (Cr) and benzene to develop immune system disorders. These were manifested as lower thymocytes count, plasma cell-macrophage transformation, as well as the apoptosis of lymphocytes and thymocytes in spleen. The co-exposure did not demonstrate additive effect. Polychlorinated biphenyls (PCBs) may be added as a lubricant in plastic manufacturing (Fig. 3). A study revealed historical exposure of porpoises to PCBs resulted in reduced testes weights and hence affected male fertility (Williams et al. 2021). Overall, the chemicals leached from plastics show reproductive hazard attributed to endocrine disrupting effect, certain carcinogenic effect linked to PBDEs, and they might have negative implications on adrenal cells, immune system, mating behaviors, glucose tolerance, neuron and development. However, these additives vary between different types of microplastics, and their leaching behaviors are not well understood (Table 2).
Hazards from the interactions of microplastics with other pollutants
Microplastics could interact with other pollutants in the environment through sorption resulting in altered ecotoxicities. As with studies on the direct hazards of microplastics, most of the studies in this area used polystyrene (Table 3). Nano-polystyrene was reported to work synergistically with parabens in promoting the proliferation of estrogen-sensitive breast cancer cells as nano-polystyrene could facilitate the translocation and adsorption of parabens (Roje et al. 2019). Microplastics led to higher accumulation of pollutants in certain organs (Deng et al. 2021), for instance, higher bioaccumulation of sulfamethoxazole in the heart, kidney, liver, lung and spleen tissues of mice (Liu et al. 2022a). Greater hazards on reproductive system have been observed as microplastics interacted with other pollutants. Microplastics contaminated with phthalate esters (PAEs) resulted in higher alteration of sperm physiology and spermatogenesis than the respective substances (Deng et al. 2021), while the tendency of microplastics to accumulate in ovaries and uterus increased the toxicity of lead (Pb) to organs (Feng et al. 2022). Mice exposed to microplastics and cadmium (Cd) for 30 days had reduced sperm count as well as impaired testicular structure and function. The toxic effects of microplastics were aggravated but those of cadmium (Cd) were reduced (Hassine et al. 2023).
Hazards on gastrointestinal system were manifested as dysbiosis of gut microbiome and disrupted bile acids, but the effects are not always synergistic, as reported in the study of Liu et al. (2022a) that microplastics increased the effect of sulfamethoxazole on gut microbiome and antibiotic resistance. Jiang et al. (2021), contrarily, observed a decline in toxicity when polystyrene microplastics combined with tributyltin. Synergistic effect was reported by Li et al. (2023) when mice were exposed to polycarbonate microplastics and imidacloprid for 4 weeks, resulting in more severe disruption of gut microbiome and lipid metabolism. Hepatoxicity and affected liver functions are the other hazards identified. Co-exposure of mice to polystyrene microplastics and epoxiconazole caused greater damage to liver tissue and slower removal of epoxiconazole by liver, while Arsenic (As) and polystyrene microplastics resulted in excessive autophagy, apoptosis and pyroptosis in the liver (Sun et al. 2022; Zhong et al. 2022). Menéndez-Pedriza et al., (2022) revealed more pronounced lipidomic changes and triglyceride enhancement upon co-exposing human hepatoma cell line to microplastics and PCBs for 48 h. Mice co-exposed to microplastics and imidacloprid for 4 weeks suffered from liver tissue damage and disrupted lipid metabolism (Li et al. 2023). Furthermore, liver organoid from human-pluripotent stem cells exposed to both polystyrene microplastics and BPA experienced interference of lipid metabolism and synergistic hepatoxicity (Cheng et al. 2023).
Microplastics and iron co-exposure led to impaired cognitive function of C57BL/6 mice due to disrupted iron homeostasis in the brain. The co-exposure also caused ferroptosis in brain (Liu et al. 2022b). This implies the potential hazard of microplastics–pollutant mix on brain and neurons depending on the pollutants combined. In addition, the in vivo and in vitro studies conducted by Zhang et al. (2022) showed combined neurotoxic effects resulted from microplastics and DEHP co-exposure. In an instance of co-exposing mice and their red blood cells to polystyrene microplastics and cadmium (Cd), an antagonistic effect was actually observed, indicating a reduced hazard on hypochromic anemia and polycythemia vera caused by exposure to cadmium chloride (CdCl2) and polystyrene microplastics, respectively (Wang et al. 2022). Shi et al. (2023) found co-exposure of mice to polystyrene microplastics and DEHP slowed skin healing and triggered more severe oxidative stress and inflammatory response. Damage to kidney tissue was reported by Sun et al. (2022) among mice co-exposed to polystyrene microplastics and epoxiconazole. Therefore, microplastics interactions with other contaminants will more likely increase their hazards on reproductive system, gut, liver, nervous system, and kidney through additive or synergistic effects. In few instances, antagonistic effects were observed, indicating microplastics might decrease the bioavailability of certain pollutants through sorption.
Dose–response assessment
The dose–response assessment of microplastics is highly complex as it not only needs to consider the direct effect of microplastics but also the additives leached from microplastics, and the chemicals released by microplastics due to sorption. Microplastics could contain different additives to confer the desirable properties to the plastics manufactured and they interact with a diverse array of contaminants in the environment (Tang 2023c). These contaminants might differ by localities. In addition, there are more studies examining the effects of polystyrene microplastics on higher mammals than other microplastic types, thus leading to a significant gap in the information available for dose–response assessment (Table 1). Microplastics in the environment have various sizes below 5 mm but only a narrow size range of microplastics has been subjected to extensive study, particularly those in the range of 0.5 – 50 µm. Besides, it is convenient to use commercial microplastics of a certain size (monodisperse) rather than environmental microplastics with more diverse sizes, resulting in constraints in the sizes of microplastics examined for their toxicity (Jin et al. 2019; Luo et al. 2019; Hesler et al. 2019). The doses applied varied with some studies only testing a particular dose (Lu et al. 2018; Fournier et al. 2020), while other studies used two or three different doses (Wei et al. 2021; Lim et al. 2021; Huang et al. 2022). The limited doses tested make it challenging to establish dose–response curves for microplastics. For instance, Lee et al. (2023) examined the toxicity of polyethylene microplastics sized 10–50 µm administered orally at 500, 1000 and 2000 mg/kg/day to ICR mice but it was challenging to establish a dose–response assessment from the limited doses tested. Additionally, the histopathological nature of the study also makes dose–response assessment challenging in the sense that only mice subjected to 28-day repeated dose showed inflammatory response in the lung tissue and no changes were observed in mice given single oral dose (Lee et al. 2023). The changes observed were too minute to constitute a statistically significant dose–response curve. Similarly, in other studies (Table 2), the microplastic doses employed often led to only minor changes such as interference with gut microbiome and gut barrier, hormonal alteration and disrupted metabolism without clear signs of toxicity among the experimental animals. These changes might not be statistically significant or sufficiently noticeable to enable the determination of toxicological doses.
The nature of microplastics could limit dose–response assessment. Establishing a threshold for microplastics is complicated considering the diverse sizes, constituents and types of microplastics. Besides, some studies showed microplastics are well tolerated by experimental animals and human cell line due partly to low translocation and absorption (Hesler et al. 2019; Stock et al. 2019; Rafiee et al. 2018). Carr et al. (2012) reported that the absorption rate of microplastics ranged from 0.04 to 0.3%, while Walczak et al. (2015) estimated a rate of 0.2–1.7% depending on the charge. Many of the deleterious effects observed are linked to oxidative stress and inflammatory response, and these responses are often not observable to a certain extent. There are currently very few studies that establish the different safe doses of microplastics and there is currently no evidence indicating microplastics themselves are carcinogenic. In some instances, microplastics triggered positive or negative effects at a very low dose at cellular level and such effects are poorly understood at individual level since a positive effect at low dose does not always translate to a positive effect on the organism (Chen et al. 2020). The supplementary information summarizes the toxicological doses of microplastics and common plastic additives. It can be seen that there is much uncertainty in the toxicological doses of microplastics. The no-observed adverse effect level (NOAEL) of polyethylene microplastics was deduced to be < 1000 mg/kg (< 35.7 mg/kg/day) for male mice and < 500 mg/kg (< 17.9 mg/kg/day) (Fig. 4) for female mice, indicating that the NOAELs could occur at any values below those stipulated and that many existing studies did not have sufficient dosages tested to pinpoint the NOAEL (Lee et al. 2022a). Similarly, the NOAEL of weathered polypropylene sized 85.2 µm was deduced to be above 25 mg/kg/day (Fig. 4) (Kim et al. 2021) and that of polyethylene below 60 mg/kg/day (Park et al. 2020). The determination of toxicological doses for microplastics is complicated by their diverse sizes, shapes, types, contents, and interactions. The current toxicological doses for microplastics are, therefore, constrained in terms of the use of monodisperse microplastics, limited dosage and dose intervals, and commercial microplastics mostly of polystyrene and polyethylene with uniform shapes. These may not accurately reflect how environmental microplastics, which humans are most frequently exposed to, behave toxicologically.
Estimated worst-case exposure of humans to microplastics through inhalation and ingestion. The worst-case ingestion rate of an adult is estimated to be 11.5–23.7 mg/kg/day. The worst-case global inhalation rate is estimated to be 0.27–0.49 mg/kg/day while that for an adult in the United States is 20.2 mg/kg/day. Note: MP = Microplastics; NOAELs = No observed adverse effect levels; PE = Polyethylene; PP = Polypropylene
Besides, plastics are known to contain a myriad of plastic additives to confer them desirable properties based on their uses. While toxicological doses of plastic additives have been determined in multiple studies, there are still many additives, which have not been subjected to extensive dose–response studies. Phthalates are a popular subject of toxicological studies, but the toxicological doses of certain phthalates such as di-iso-decyl phthalate and di-iso-nonyl phthalate are inconclusive due to the progressive tightening of their NOAELs over time as more evidence pointing to statistically observable changes on experimental animals at lower doses emerged (Ambe et al. 2019). More common phthalates such as DEHP have more consistent NOAEL reported (Conley et al. 2021). The NOAELs of bisphenols, particularly BPA, bisphenol-F and bisphenol-S have been proposed in addition to those of different PBDEs such as BDE-47, BDE-99 and BDE-209 (Genuis et al. 2012; Lee et al. 2022b; Lee et al. 2022c; Ermler and Kortenkamp 2022) (see Supplementary Information). PBDEs are used as flame retardants and they are not incorporated chemically into plastics, making their leaching from plastics into the environment a concern. The Stockholm Convention on Persistent Organic Pollutants has included the three PBDEs in Annex A bound for elimination, together with PCBs (Stockholm Convention 2019). As probable human carcinogens, safe exposure thresholds for PCBs are often not available as exposure at any amounts could be genotoxic (US EPA 2016). Nonetheless, a mixture of PCBs with 60% chlorine called Aroclor 1260 has a NOAEL and lowest-observed-adverse-effect level (LOAEL) of 0.625 mg/kg/day and 1.25 mg/kg/day, respectively. LOAEL is defined as the lowest dose or exposure level that causes any observable harmful effect (Aloysius et al. 2015). Generally, the toxicity levels of microplastics are not as definitive as their additives due probably to their relatively milder toxicity and less comprehensive dose–response studies.
Exposure assessment
Humans are exposed to microplastics via inhalation, ingestion, and dermal contact. Inhalation and ingestion are the most prevailing microplastics exposure pathways that most studies focus on as microplastics have been widely detected in food, water, and air (Table 4). Currently, there is limited evidence showing the entry of microplastics through skin unless the dermal barrier is damaged. Studies have revealed the presence of microplastics in a wide array of food items, resulting in substantial exposure of humans to microplastics through ingestion. EFSA CONTAM Panel (2016) estimated that mussel ingestion would expose a person to 900 microplastic particles or 7 µg microplastics per mussel. Meanwhile, salt ingestion may expose a person to a maximum of 37 microplastics per year (Karami et al. 2017). A study conducted on a comprehensive list of processed food items in the USA constituting 15% of the national caloric intake revealed an approximated microplastics consumption rate of 39,000–52,000 particles/person/year. Taking into account inhalation and water intake, microplastics consumption was estimated at 117,000 to 263,000 particles/person/year (Cox et al. 2019). Oliveri Conti et al. (2020) estimated the microplastics intake from fruits and vegetable, and revealed an average adult to have a microplastics intake rate of 2.96 × 104–4.62 × 105 particles/kg-bw/day from apples and pears consumption, which is significantly higher than the American estimation (Table 4). Drinking water is also a major source of microplastics with exposure rates ranging from 28 particles/day to 3,350,208 polyethylene terephthalate microplastics/kg-bw/day depending on the types of drinking water, the consumption rate used and the regions the studies were conducted (Eerkes-Medrano et al. 2019; Zuccarello et al. 2019).
Exposure to microplastics in indoor air results in both ingestion and inhalation of microplastics. An Australian estimation revealed age-dependent ingestion rates with children aged 1 to 6 years having the highest rate of 6.1 mg/kg-bw/year in comparison with 0.5 mg/kg-bw/year for those aged 20 years and above. As for inhalation, the mean intake rate is 0.2 mg/kg-bw/year (Soltani et al. 2021). An Iranian study, however, reported the rates as microplastics ingested and inhaled, and these rates are also age-dependent with infants having the highest exposure rates of 0.329–0.908 microplastics/kg-bw/day by ingestion, and 3.033–8.372 microplastics/kg-bw/day by inhalation (Kashfi et al. 2022). Using a breathing thermal manikin, Vianello et al. (2019) approximated the microplastics inhalation rate to be 272 microplastics/day/capita, while performing light activity. It is on the high side when compared to the microplastics inhalation of 0.640–1.767 microplastics/kg-bw/day of an adult approximated by Kashfi et al. (2022). Infants are also exposed to microplastics through ingestion of infant formula in polypropylene feeding bottles and the global exposure of a 12-year-old was estimated to be 14,000–4,550,000 microplastics/day/capita (Li et al. 2020b). Senathirajah et al. (2021) provided a global ingestion rate of 0.1–5 g microplastics/week (sized 0–1 mm) based on the review of 59 publications, whereas Domenech and Marcos (2021) estimated the global daily inhalation rate to be 5.9 microplastics/day/capita. The latter seems to be lower than the rates reported in other studies.
Risk characterization and implications
Assessing the risk of microplastics on human health is constrained by the limited toxicological data available for higher mammals and cell line models, which make allometric scaling and extrapolation challenging, unlike many other chemicals with well-established dose–response assessments. In addition, microplastics contain a myriad of chemicals whose leaching is a concern though studies have shown that leaching might be minimal or insignificant for certain chemicals. Nonetheless, the diverse chemicals in microplastics make assessment of leaching risk extremely difficult, especially when the leaching itself has not been comprehensively studied and leaching models are limited in multiple ways (Do et al. 2022). More studies may need to be conducted on how plastic additives distribute between microplastics and the aqueous environment at equilibrium. Another important complexity is the interactions of microplastics with other chemicals in the environment, which alter the toxicity of microplastics. There are innumerable possibilities for such interactions, for instance, microplastics could interact with one single chemical or more than one chemical concurrently. The formation of biofilms on microplastics also alters the properties of microplastics, leading to variation in the leaching of additives from microplastics and their interactions with chemicals.
In view of much uncertainty and a lack of data on the leaching of chemicals from microplastics as well as multiple interactions of microplastics with diverse chemicals in the environment, the risk assessment in this paper is limited only to the direct toxicological effects of microplastics on human, without interactions with other environmental pollutants or emphasis on a particular chemical leached from them. The ubiquity of microplastics means they have permeated most if not all food items and the atmospheric compartment. Using the upper limit of the global average ingestion rate of 0.1–5 g microplastics/week suggested by Senathirajah et al. (2021) (Fig. 4), and 62 kg as the average weight of the world population (Walpole et al. 2012), the microplastics exposure is 11.5 mg/kg/day per capita. Using the available NOAELs in Table 1, an uncertainty factor of 100 is applied for interspecies conversion to generate the safe values for human (Fig. 4), known as reference doses, thus yielding reference doses of < 0.358 mg/kg/day (male) and < 0.179 mg/kg/day (female) for polyethylene, as well as > 0.25 mg/kg/day for weathered polypropylene (Lee et al. 2022a; Kim et al. 2021). Assuming that the microplastics exposed consist of polyethylene exclusively, the exposure is much higher than the reference dose. If the microplastics exposed to were exclusively polypropylene, it remains uncertain whether the exposure is safe since the reference dose was deduced to be > 0.25 mg/kg/day (lower limit). Therefore, microplastics exposure due to ingestion alone is likely to exceed the reference doses. Taking the worst-case annual microplastics ingestion rate (food = 52,000 particles/person, and drinks = 90,000 particles/person) of the US which is 142,000 particles/person (Table 4), and assuming a minimum and maximum particle weights of 2.8 × 10−3 g/particle and 5.12 × 10−3 g/particle, respectively (Senathirajah et al. 2021), as well as an average US adult’s weight of 84 kg, the microplastics ingestion rate ranges from 13 mg/kg/day to 23.7 mg/kg/day (Cox et al. 2019) (Fig. 4). The values are higher than the global average (Table 4). As for infants aged 12 months, a study provided a very high global exposure rate at 14,600–4,550,000 microplastics/day/capita (Li et al. 2020b). With such high exposure rate, the ingested microplastics per kg body weight is deemed to be extremely high. This aligns with the generally higher exposure of infants and children to microplastics (Soltani et al. 2021; Kashfi et al. 2022).
For inhalation, adopting the global daily inhalation rate of 5.9 microplastics/day/capita suggested by Domenech and Marcos (2021), as well as the same microplastics weights and global average human weight as above, individual microplastics exposure ranges from 0.27 mg/kg/day to 0.49 mg/kg/day (Fig. 4). Adding to the ingestion rate, the reference doses of polyethylene are exceeded, while the lower limit of the reference dose of polypropylene is also significantly exceeded (Fig. 4); the upper limit of no-observed adverse effect level (NOAEL) for polypropylene has not been defined as in the Supplementary Information. As for the case of an adult in the US (Cox et al. 2019), with a worst-case annual exposure of 121,000 particles/person through inhalation, which is translated to a maximum weight of 1697.3 mg/day (weight = 5.12 × 10−3 g/particle), the exposure rate is 20.2 mg/kg/day which is higher than the global daily inhalation rate of Domenech & Marcos, as well as the regional rates in Australia and Iran reported (Soltani et al. 2021; Kashfi et al. 2022). In view of this, the risk associated with human microplastics exposure through inhalation and ingestion is not negligible. In fact, it is highly probable that an individual is exposed to microplastics though the extent of exposure varies. The severity of microplastics exposure is often low (see Table 1), resulting in low-to-medium risk based on a conservative and preliminary assessment, assuming that the exposure, hence the manifestation of effects, spans across a wide range of frequency (see Fig. 5 for a typical risk assessment matrix).
As microplastics exposure varies spatially, temporally, and demographically, this conservative approach is crucial to identify the upper bound of risk. It accounts for the uncertainty and variability in the data and the methods used for exposure assessment, while providing a precautionary approach to protect human health. This risk assessment contributes to the preliminary understanding of the risk ubiquitous microplastics in the environment poses on human health, a topic which has not been subjected to extensive study due to the limited information and looming uncertainties. With increasing microplastics in the environment, hence increasing exposure, it highlights the need to reduce or limit the environmental prevalence of microplastics to control the risk of microplastics on human health. It permits policymakers to design evidence-based and targeted policies to address the most relevant sources, pathways, and hazards of microplastics exposure, for instance, through restriction of adding microplastics to consumer products and setting standards to regulate the contents of microplastics in food and drinking water. Future studies can focus on dose–response assessment using polydisperse, environmentally representative microplastics to fill in the large gap present currently. More comprehensive studies on exposure assessment or those with regional significance could also be conducted to facilitate risk characterization. Furthermore, understanding the combined risk of microplastics, leaching of additives from microplastics and interactions of microplastics with other pollutants is crucial. Specifically, there is a need for adequate particle characterization and selection for toxicity testing. Future studies should use standardized methods and criteria to measure and report the physical and chemical properties of microplastics and select relevant and realistic microplastic types and concentrations for exposure scenarios in different environmental matrices. Future studies of microplastics dose–response should account for confounding factors and sources of variability that may influence the toxicity of microplastics such as particle aggregation, bioavailability, bioaccumulation, biodegradation, and interactions with other stressors.
Conclusion
With the limited no-observed adverse effect levels (NOAELs) available and an uncertainty factor of 100, this preliminary risk assessment revealed that human exposure to microplastics is significantly above the NOAELs or the lower limits of the NOAELs. Therefore, the risk could range from low to medium, considering the low consequences reported for animal models and cell lines, ranging from no significant changes of body and organ weights to altered markers for energy and lipid metabolisms, oxidative stress, loss of cell viability, and reduced reproductive health. However, the risk assessment has certain limitations. The current information related to toxicity of microplastics is limited to enable a detailed human health risk assessment. While it is certain that human exposure to microplastics mainly occurs through inhalation and ingestion, current studies related to such exposure are fragmented focusing on exposure to particular food items, drinking water, air or objects. Where available, these attempts are complicated by the wide variabilities of microplastics abundance across different regions, in different items or in the same items produced in different regions, conferring much uncertainty to exposure assessment. Additionally, the diverse properties of microplastics, particularly their sizes, shapes, and compositions, also contributed to the uncertainty. As for dose–response assessment, an obvious limitation is the use of specific commercial monodisperse microplastics, particularly polystyrene, polyethylene, and polypropylene, which may not sufficiently represent the diverse, polydisperse, aged microplastics in the environment. Environmental microplastics are present in diverse sizes and smaller microplastics are commonly associated with more harm due to their larger surface area to volume ratio, which may increase their ability to adsorb and release other chemicals, as well as to penetrate and damage cells and tissues. The limited doses tested also constrain identification of toxicological doses. Risk assessment of microplastics is complicated by the various additives present therein to impart certain desirable properties and these additives have different leaching behaviors in different environments. Besides, microplastics could interact with other pollutants and microorganisms in the environment through adsorption and subsequent leaching as well as biofilm formation, resulting in changes in their toxicological and physicochemical properties. Such interactions could result in co-exposure of human to the environmental pollutants adsorbed on microplastics or pathogenic microorganisms harbored by microplastics. Microplastics may interact synergistically, antagonistically, or additively with these chemicals and microorganisms, enhancing or reducing their bioavailability, toxicity, and growth. Thus, further risk assessments should attempt to address these limitations.
References
Aloysius H, Desaulniers D, Bandiera SM (2015) Evaluating toxic endpoints of exposure to a commercial PCB mixture: an in vivo laboratory study. AIMS Environ Sci 2:314–332. https://doi.org/10.3934/environsci.2015.2.314
Amar S, Binet A, Téteau O, Desmarchais A, Papillier P, Lacroix MZ, Maillard V, Guérif F, Elis S (2020) Bisphenol S impaired human granulosa cell steroidogenesis in vitro. Int J Mol Sci. https://doi.org/10.3390/ijms21051821
Amato-Lourenço LF, Carvalho-Oliveira R, Júnior GR, dos Santos Galvão L, Ando RA, Mauad T (2021) Presence of airborne microplastics in human lung tissue. J Hazard Mater 416:126124. https://doi.org/10.1016/j.jhazmat.2021.126124
Ambe K, Sakakibara Y, Sakabe A, Makino H, Ochibe T, Tohkin M (2019) Comparison of the developmental/reproductive toxicity and hepatotoxicity of phthalate esters in rats using an open toxicity data source. J Toxicol Sci 44:245–255. https://doi.org/10.2131/jts.44.245
An R, Wang X, Yang L, Zhang J, Wang N, Xu F, Hou Y, Zhang H, Zhang L (2021) Polystyrene microplastics cause granulosa cells apoptosis and fibrosis in ovary through oxidative stress in rats. Toxicology 449:152665. https://doi.org/10.1016/j.tox.2020.152665
Bergmann M, Mützel S, Primpke S, Tekman MB, Trachsel J, Gerdts G (2023) White and wonderful? Microplast Prevail Snow Alps Arctic Sci Adv 5:eaax1157. https://doi.org/10.1126/sciadv.aax1157
Bridson JH, Gaugler EC, Smith DA, Northcott GL, Gaw S (2021) Leaching and extraction of additives from plastic pollution to inform environmental risk: a multidisciplinary review of analytical approaches. J Hazard Mater 414:125571. https://doi.org/10.1016/j.jhazmat.2021.125571
Carr KE, Smyth SH, McCullough MT, Morris JF, Moyes SM (2012) Morphological aspects of interactions between microparticles and mammalian cells: intestinal uptake and onward movement. Prog Histochem Cytochem 46:185–252. https://doi.org/10.1016/j.proghi.2011.11.001
Catarino AI, Macchia V, Sanderson WG, Thompson RC, Henry TB (2018) Low levels of microplastics (MP) in wild mussels indicate that MP ingestion by humans is minimal compared to exposure via household fibres fallout during a meal. Environ Pollut 237:675–684. https://doi.org/10.1016/j.envpol.2018.02.069
Chau HS, Xu S, Ma Y, Wang Q, Cao Y, Huang G, Ruan Y, Yan M, Liu M, Zhang K, Lam PKS (2023) Microplastic occurrence and ecological risk assessment in the eight outlets of the Pearl River Estuary, a new insight into the riverine microplastic input to the northern South China Sea. Mar Pollut Bull 189:114719. https://doi.org/10.1016/j.marpolbul.2023.114719
Chen X, Mo J, Zhang S, Li X, Huang T, Zhu Q, Wang S, Chen X, Ge RS (2019) 4-Bromodiphenyl ether causes adrenal gland dysfunction in rats during puberty. Chem Res Toxicol 32:1772–1779. https://doi.org/10.1021/acs.chemrestox.9b00123
Chen Q, Lackmann C, Wang W, Seiler TB, Hollert H, Shi H (2020) Microplastics lead to hyperactive swimming behaviour in adult zebrafish. Aquat Toxicol 224:105521. https://doi.org/10.1016/j.aquatox.2020.105521
Cheng W, Zhou Y, Xie Y, Li Y, Zhou R, Wang H, Feng Y, Wang Y (2023) Combined effect of polystyrene microplastics and bisphenol A on the human embryonic stem cells-derived liver organoids: the hepatotoxicity and lipid accumulation. Sci Total Environ 854:158585. https://doi.org/10.1016/j.scitotenv.2022.158585
Chioccarelli T, Manfrevola F, Migliaccio M, Altucci L, Porreca V, Fasano S, Cobellis G (2020) Fetal-perinatal exposure to bisphenol-A affects quality of spermatozoa in adulthood mouse. Int J Endocrinol 2020:2750501. https://doi.org/10.1155/2020/2750501
Conley JM, Lambright CS, Evans N, Cardon M, Medlock-Kakaley E, Wilson VS, Gray LE (2021) A mixture of 15 phthalates and pesticides below individual chemical no observed adverse effect levels (NOAELs) produces reproductive tract malformations in the male rat. Environ Int 156:106615–106698. https://doi.org/10.1016/j.envint.2021.106615
Stockholm Convention (2019) All POPs listed in the Stockholm Convention. https://www.pops.int/TheConvention/ThePOPs/AllPOPs/tabid/2509/Default.aspx. Accessed 28 July 2023.
Cox KD, Covernton GA, Davies HL, Dower JF, Juanes F, Dudas SE (2019) Human consumption of microplastics. Environ Sci Technol 53:7068–7074. https://doi.org/10.1021/acs.est.9b01517
da Costa Araújo AP, Malafaia G (2021) Microplastic ingestion induces behavioral disorders in mice: a preliminary study on the trophic transfer effects via tadpoles and fish. J Hazard Mater 401:123263. https://doi.org/10.1016/j.jhazmat.2020.123263
da Silva GN, Zauer Curi T, Lima Tolouei SE, Tapias Passoni M, Sari Hey GB, Marino Romano R, Martino-Andrade AJ, Dalsenter PR (2019) Effects of diisopentyl phthalate exposure during gestation and lactation on hormone-dependent behaviours and hormone receptor expression in rats. J Neuroendocrinol 31:e12816. https://doi.org/10.1111/jne.12816
Deng Y, Zhang Y, Lemos B, Ren H (2017) Tissue accumulation of microplastics in mice and biomarker responses suggest widespread health risks of exposure. Sci Rep. https://doi.org/10.1038/srep46687
Deng Y, Yan Z, Shen R, Huang Y, Ren H, Zhang Y (2021) Enhanced reproductive toxicities induced by phthalates contaminated microplastics in male mice (Mus musculus). J Hazard Mater 406:124644. https://doi.org/10.1016/j.jhazmat.2020.124644
Do ATN, Ha Y, Kwon JH (2022) Leaching of microplastic-associated additives in aquatic environments: a critical review. Environ Pollut 305:119258–119298. https://doi.org/10.1016/j.envpol.2022.119258
Domenech J, Marcos R (2021) Pathways of human exposure to microplastics, and estimation of the total burden. Curr Opin Food Sci 39:144–151. https://doi.org/10.1016/j.cofs.2021.01.004
Dris R, Gasperi J, Mirande C, Mandin C, Guerrouache M, Langlois V, Tassin B (2017) A first overview of textile fibers, including microplastics, in indoor and outdoor environments. Environ Pollut 221:453–458. https://doi.org/10.1016/j.envpol.2016.12.013
Dunnick JK, Pandiri AR, Merrick BA, Kissling GE, Cunny H, Mutlu E, Waidyanatha S, Sills R, Hong HL, Ton TV, Maynor T, Recio L, Phillips SL, Devito MJ, Brix A (2018) Carcinogenic activity of pentabrominated diphenyl ether mixture (DE-71) in rats and mice. Toxicol Reports 5:615–624. https://doi.org/10.1016/j.toxrep.2018.05.010
Eerkes-Medrano D, Leslie HA, Quinn B (2019) Microplastics in drinking water: a review and assessment. Curr Opin Environ Sci Heal 7:69–75. https://doi.org/10.1016/j.coesh.2018.12.001
EFSA Contam Panel (2016) Statement on the presence of microplastics and nanoplastics in food, with particular focus on seafood. EFSA J 14:4501. https://doi.org/10.2903/j.efsa.2016.4501
Eker F, Gungunes A, Durmaz S, Kisa U, Celik ZR (2021) Nonfunctional adrenal incidentalomas may be related to bisphenol-A. Endocrine 71:459–466. https://doi.org/10.1007/s12020-020-02502-2
Ermler S, Kortenkamp A (2022) Declining semen quality and polybrominated diphenyl ethers (PBDEs): review of the literature to support the derivation of a reference dose for a mixture risk assessment. Int J Hyg Environ Health 242:113953. https://doi.org/10.1016/j.ijheh.2022.113953
Estrela FN, Guimarães ATB, da Costa Araújo AP, Silva FG, da Luz TM, Silva AM, Pereira PS, Malafaia G (2021) Toxicity of polystyrene nanoplastics and zinc oxide to mice. Chemosphere 271:129476. https://doi.org/10.1016/j.chemosphere.2020.129476
Eun-Jung P, Han JS, Eun-Jun P, Seong E, Lee GH, Kim DW, Son HY, Han HY, Lee BS (2020) Repeated-oral dose toxicity of polyethylene microplastics and the possible implications on reproduction and development of the next generation. Toxicol Lett 324:75–85. https://doi.org/10.1016/j.toxlet.2020.01.008
Feng Y, Yuan H, Wang W, Xu Y, Zhang J, Xu H, Fu F (2022) Co-exposure to polystyrene microplastics and lead aggravated ovarian toxicity in female mice via the PERK/eIF2α signaling pathway. Ecotoxicol Environ Saf 243:113966. https://doi.org/10.1016/j.ecoenv.2022.113966
Fournier SB, D’Errico JN, Adler DS, Kollontzi S, Goedken MJ, Fabris L, Yurkow EJ, Stapleton PA (2020) Nanopolystyrene translocation and fetal deposition after acute lung exposure during late-stage pregnancy. Part Fibre Toxicol 17:55. https://doi.org/10.1186/s12989-020-00385-9
Genuis SJ, Beesoon S, Lobo RA, Birkholz D (2012) Human Elimination of phthalate compounds: blood, urine, and sweat (BUS). Study Sci World J 2012:615068. https://doi.org/10.1100/2012/615068
Hassine MBH, Venditti M, Rhouma MB, Minucci S, Messaoudi I (2023) Combined effect of polystyrene microplastics and cadmium on rat blood-testis barrier integrity and sperm quality. Environ Sci Pollut Res 30:56700–56712. https://doi.org/10.1007/s11356-023-26429-z
Helcoski R, Yonkos LT, Sanchez A, Baldwin AH (2020) Wetland soil microplastics are negatively related to vegetation cover and stem density. Environ Pollut 256:113391. https://doi.org/10.1016/j.envpol.2019.113391
Hesler M, Aengenheister L, Ellinger B, Drexel R, Straskraba S, Jost C, Wagner S, Meier F, von Briesen H, Büchel C, Wick P, Buerki-Thurnherr T, Kohl Y (2019) Multi-endpoint toxicological assessment of polystyrene nano- and microparticles in different biological models in vitro. Toxicol Vitr 61:104610. https://doi.org/10.1016/j.tiv.2019.104610
Hou B, Wang F, Liu T, Wang Z (2021a) Reproductive toxicity of polystyrene microplastics: in vivo experimental study on testicular toxicity in mice. J Hazard Mater 405:124028. https://doi.org/10.1016/j.jhazmat.2020.124028
Hou J, Lei Z, Cui L, Hou Y, Yang L, An R, Wang Q, Li S, Zhang H, Zhang L (2021b) Polystyrene microplastics lead to pyroptosis and apoptosis of ovarian granulosa cells via NLRP3/Caspase-1 signaling pathway in rats. Ecotoxicol Environ Saf 212:112012. https://doi.org/10.1016/j.ecoenv.2021.112012
Huang PC, Tsai CH, Liang WY, Li SS, Huang HB, Kuo PL (2016) Early phthalates exposure in pregnant women is associated with alteration of thyroid hormones. PLoS ONE 11:e0159398
Huang T, Zhang W, Lin T, Liu S, Sun Z, Liu F, Yuan Y, Xiang X, Kuang H, Yang B, Zhang D (2022) Maternal exposure to polystyrene nanoplastics during gestation and lactation induces hepatic and testicular toxicity in male mouse offspring. Food Chem Toxicol 160:112803. https://doi.org/10.1016/j.fct.2021.112803
Jabeen K, Su L, Li J, Yang D, Tong C, Mu J, Shi H (2017) Microplastics and mesoplastics in fish from coastal and fresh waters of China. Environ Pollut 221:141–149. https://doi.org/10.1016/j.envpol.2016.11.055
Jiang P, Yuan G, Jiang B, Zhang J, Wang Y, Lv H, Zhang Z, Wu J, Wu Q, Li L (2021) Effects of microplastics (MPs) and tributyltin (TBT) alone and in combination on bile acids and gut microbiota crosstalk in mice. Ecotoxicol Environ Saf 220:112345. https://doi.org/10.1016/j.ecoenv.2021.112345
Jiao Li, Jie Li, Zhai L, Lu K (2023) Co-exposure of polycarbonate microplastics aggravated the toxic effects of imidacloprid on the liver and gut microbiota in mice. Environ Toxicol Pharmacol 101:104194. https://doi.org/10.1016/j.etap.2023.104194
Jin Y, Lu L, Tu W, Luo T, Fu Z (2019) Impacts of polystyrene microplastic on the gut barrier, microbiota and metabolism of mice. Sci Total Environ 649:308–317. https://doi.org/10.1016/j.scitotenv.2018.08.353
Jin H, Ma T, Sha X, Liu Z, Zhou Y, Meng X, Chen Y, Han X, Ding J (2021) Polystyrene microplastics induced male reproductive toxicity in mice. J Hazard Mater 401:123430. https://doi.org/10.1016/j.jhazmat.2020.123430
Kacprzak S, Tijing LD (2022) Microplastics in indoor environment: sources, mitigation and fate. J Environ Chem Eng. https://doi.org/10.1016/j.jece.2022.107359
Kanhai LDK, Gardfeldt K, Krumpen T, Thompson RC, O’Connor I (2020) Microplastics in sea ice and seawater beneath ice floes from the Arctic Ocean. Sci Rep 10:5004. https://doi.org/10.1038/s41598-020-61948-6
Kankanige D, Babel S (2020) Smaller-sized micro-plastics (MPs) contamination in single-use PET-bottled water in Thailand. Sci Total Environ 717:137232. https://doi.org/10.1016/j.scitotenv.2020.137232
Karami A, Golieskardi A, Keong Choo C, Larat V, Galloway TS, Salamatinia B (2017) The presence of microplastics in commercial salts from different countries. Sci Rep 7:46173. https://doi.org/10.1038/srep46173
Karaulov AV, Smolyagin AI, Mikhailova IV, Stadnikov AA, Ermolina EV, Filippova YV, Kuzmicheva NA, Vlata Z, Djordjevic AB, Tsitsimpikou C, Hartung T, Hernandez AF, Tsatsakis A (2022) Assessment of the combined effects of chromium and benzene on the rat neuroendocrine and immune systems. Environ Res 207:112096. https://doi.org/10.1016/j.envres.2021.112096
Kashfi FS, Ramavandi B, Arfaeinia H, Mohammadi A, Saeedi R, De-la-Torre GE, Dobaradaran S (2022) Occurrence and exposure assessment of microplastics in indoor dusts of buildings with different applications in Bushehr and Shiraz cities. Iran. Sci Total Environ 829:154651. https://doi.org/10.1016/j.scitotenv.2022.154651
Kim JS, Lee HJ, Kim SK, Kim HJ (2018) Global pattern of microplastics (MPs) in commercial food-grade salts: sea salt as an indicator of seawater MP pollution. Environ Sci Technol 52:12819–12828. https://doi.org/10.1021/acs.est.8b04180
Kim J, Maruthupandy M, An KS, Lee KH, Jeon S, Kim JS, Cho WS (2021) Acute and subacute repeated oral toxicity study of fragmented microplastics in Sprague-Dawley rats. Ecotoxicol Environ Saf 228:112964. https://doi.org/10.1016/j.ecoenv.2021.112964
Kosuth M, Mason SA, Wattenberg EV (2018) Anthropogenic contamination of tap water, beer, and sea salt. PLoS ONE 13:e0194970
Kowalczyk N, Blake N, Charko F, Quek Y (2017). Microplastics in the maribyrnong and yarra rivers, melbourne, Australia. https://www.ecocentre.com/wp-content/uploads/2021/07/Microplastics-in-the-Maribyrnong-and-Yarra-Rivers_2017-1.pdf. Accessed 28 July 2023
Lai FN, Liu JC, Li L, Ma JY, Liu XL, Liu YP, Zhang XF, Chen H, De Felici M, Dyce PW, Shen W (2017) Di (2-ethylhexyl) phthalate impairs steroidogenesis in ovarian follicular cells of prepuberal mice. Arch Toxicol 91:1279–1292. https://doi.org/10.1007/s00204-016-1790-z
Lee SJ, Baek SK, Kim W, Quah Y, Kim SY, Jeong JS, Lee J, Yu WJ (2022) Reproductive and developmental toxicity screening of bisphenol F by oral gavage in rats. Regul Toxicol Pharmacol 136:105286. https://doi.org/10.1016/j.yrtph.2022.105286
Li C, Tang KHD (2023) Effects of pH and temperature on the leaching of Di (2-Ethylhexyl) phthalate and Di-n-butyl phthalate from microplastics in simulated marine environment. Biointerface Res Appl Chem 13:269
Li N, Liu T, Guo K, Zhu J, Yu G, Wang S, Ye L (2018) Effect of mono-(2-ethylhexyl) phthalate (MEHP) on proliferation of and steroid hormone synthesis in rat ovarian granulosa cells in vitro. J Cell Physiol 233:3629–3637. https://doi.org/10.1002/jcp.26224
Li B, Ding Y, Cheng X, Sheng D, Xu Z, Rong Q, Wu Y, Zhao H, Ji X, Zhang Y (2020a) Polyethylene microplastics affect the distribution of gut microbiota and inflammation development in mice. Chemosphere 244:125492. https://doi.org/10.1016/j.chemosphere.2019.125492
Li D, Shi Y, Yang L, Xiao L, Kehoe DK, Gun’ko YK, Boland JJ, Wang JJ, (2020b) Microplastic release from the degradation of polypropylene feeding bottles during infant formula preparation. Nat Food 1:746–754. https://doi.org/10.1038/s43016-020-00171-y
Li X, Zhang T, Lv W, Wang H, Chen H, Xu Q, Cai H, Dai J (2022) Intratracheal administration of polystyrene microplastics induces pulmonary fibrosis by activating oxidative stress and Wnt/β-catenin signaling pathway in mice. Ecotoxicol Environ Saf 232:113238. https://doi.org/10.1016/j.ecoenv.2022.113238
Li Y, Liu C, Yang H, He W, Li B, Zhu X, Liu S, Jia S, Li R, Tang KH (2024) Leaching of chemicals from microplastics: a review of chemical types, leaching mechanisms and influencing factors. Sci Total Environ 906:167666. https://doi.org/10.1016/j.scitotenv.2023.167666
Lim D, Jeong J, Song KS, Sung JH, Oh SM, Choi J (2021) Inhalation toxicity of polystyrene micro(nano)plastics using modified OECD TG 412. Chemosphere 262:128330. https://doi.org/10.1016/j.chemosphere.2020.128330
Lin Q, Zhao S, Pang L, Sun C, Chen L, Li F (2022) Potential risk of microplastics in processed foods: Preliminary risk assessment concerning polymer types, abundance, and human exposure of microplastics. Ecotoxicol Environ Saf 247:114260. https://doi.org/10.1016/j.ecoenv.2022.114260
Liu T, Wang Y, Yang M, Shao P, Duan L, Li M, Zhu M, Yang J, Jiang J (2018) Di-(2-ethylhexyl) phthalate induces precocious puberty in adolescent female rats. Iran J Basic Med Sci 21:848
Liu J, Lv M, Sun A, Ding J, Wang Y, Chang X, Chen L (2022a) Exposure to microplastics reduces the bioaccumulation of sulfamethoxazole but enhances its effects on gut microbiota and the antibiotic resistome of mice. Chemosphere 294:133810. https://doi.org/10.1016/j.chemosphere.2022.133810
Liu X, Yang H, Yan X, Xu S, Fan Y, Xu H, Ma Y, Hou W, Javed R, Zhang Y (2022b) Co-exposure of polystyrene microplastics and iron aggravates cognitive decline in aging mice via ferroptosis induction. Ecotoxicol Environ Saf 233:113342–113398. https://doi.org/10.1016/j.ecoenv.2022.113342
Lorenz C, Roscher L, Meyer MS, Hildebrandt L, Prume J, Löder MGJ, Primpke S, Gerdts G (2019) Spatial distribution of microplastics in sediments and surface waters of the southern North Sea. Environ Pollut 252:1719–1729. https://doi.org/10.1016/j.envpol.2019.06.093
Lu L, Wan Z, Luo T, Fu Z, Jin Y (2018) Polystyrene microplastics induce gut microbiota dysbiosis and hepatic lipid metabolism disorder in mice. Sci Total Environ 631–632:449–458. https://doi.org/10.1016/j.scitotenv.2018.03.051
Lu C, Liang Y, Cheng Y, Peng C, Sun Y, Liu K, Li Y, Lou Y, Jiang X, Zhang A, Liu J, Cao J, Han F (2023) Microplastics cause reproductive toxicity in male mice through inducing apoptosis of spermatogenic cells via p53 signaling. Food Chem Toxicol 179:113970. https://doi.org/10.1016/j.fct.2023.113970
Luo T, Wang C, Pan Z, Jin C, Fu Z, Jin Y (2019) Maternal polystyrene microplastic exposure during gestation and lactation altered metabolic homeostasis in the dams and their F1 and F2 offspring. Environ Sci Technol 53:10978–10992. https://doi.org/10.1021/acs.est.9b03191
Ma Y, Jun Z, Zeng R, Qiao X, Cheng R, Nie Y, Luo Y, Li S, Zhang Jing XuW (2019) Effects of the dibutyl phthalate (DBP) on the expression and activity of aromatase in human granulosa cell line KGN. Ann Clin Lab Sci 49:175–182
Ma Q, Deng P, Lin M, Yang L, Li L, Guo L, Zhang L, He M, Lu Y, Pi H, Zhang Y, Yu Z, Chen C, Zhou Z (2021) Long-term bisphenol A exposure exacerbates diet-induced prediabetes via TLR4-dependent hypothalamic inflammation. J Hazard Mater 402:123926. https://doi.org/10.1016/j.jhazmat.2020.123926
Markic A, Niemand C, Bridson JH, Mazouni-Gaertner N, Gaertner JC, Eriksen M, Bowen M (2018) Double trouble in the South Pacific subtropical gyre: Increased plastic ingestion by fish in the oceanic accumulation zone. Mar Pollut Bull 136:547–564. https://doi.org/10.1016/j.marpolbul.2018.09.031
Menéndez-Pedriza A, Jaumot J, Bedia C (2022) Lipidomic analysis of single and combined effects of polyethylene microplastics and polychlorinated biphenyls on human hepatoma cells. J Hazard Mater 421:126777. https://doi.org/10.1016/j.jhazmat.2021.126777
Nehring I, Staniszewska M (2023) Comparison of prenatal and postnatal exposure to endocrine active phenol derivatives in mammals—Humans and Baltic grey seals. Mar Pollut Bull 196:115567. https://doi.org/10.1016/j.marpolbul.2023.115567
Nel HA, Dalu T, Wasserman RJ, Hean JW (2019) Colour and size influences plastic microbead underestimation, regardless of sediment grain size. Sci Total Environ 655:567–570. https://doi.org/10.1016/j.scitotenv.2018.11.261
Oliveri Conti G, Ferrante M, Banni M, Favara C, Nicolosi I, Cristaldi A, Fiore M, Zuccarello P (2020) Micro- and nano-plastics in edible fruit and vegetables. the first diet risks assessment for the general population. Environ Res 187:109677. https://doi.org/10.1016/j.envres.2020.109677
Oßmann BE, Sarau G, Holtmannspötter H, Pischetsrieder M, Christiansen SH, Dicke W (2018) Small-sized microplastics and pigmented particles in bottled mineral water. Water Res 141:307–316. https://doi.org/10.1016/j.watres.2018.05.027
Pham DT, Jinwoo K, Lee SH, Juyang K, Kim D, Hong S, Jung J, Kwon JH (2023) Analysis of microplastics in various foods and assessment of aggregate human exposure via food consumption in korea. Environ Pollut 322:121153. https://doi.org/10.1016/j.envpol.2023.121153
Rafiee M, Dargahi L, Eslami A, Beirami E, Jahangiri-rad M, Sabour S, Amereh F (2018) Neurobehavioral assessment of rats exposed to pristine polystyrene nanoplastics upon oral exposure. Chemosphere 193:745–753. https://doi.org/10.1016/j.chemosphere.2017.11.076
Redondo-Hasselerharm PE, Rico A, Koelmans AA (2023) Risk assessment of microplastics in freshwater sediments guided by strict quality criteria and data alignment methods. J Hazard Mater 441:129814. https://doi.org/10.1016/j.jhazmat.2022.129814
Roje Ž, Ilić K, Galić E, Pavičić I, Turčić P, Stanec Z, Vinković Vrček I (2019) Synergistic effects of parabens and plastic nanoparticles on proliferation of human breast cancer cells. Arh Hig Rada Toksikol 70:310–314
Saeed A, Akhtar MF, Saleem A, Akhtar B, Sharif A (2023) Reproductive and metabolic toxic effects of polystyrene microplastics in adult female Wistar rats: a mechanistic study. Environ Sci Pollut Res 30:63185–63199. https://doi.org/10.1007/s11356-023-26565-6
Schwabl P, Köppel S, Königshofer P, Bucsics T, Trauner M, Reiberger T, Liebmann B (2019) Detection of various microplastics in human stool. Ann Intern Med 171:453–457. https://doi.org/10.7326/M19-0618
Senathirajah K, Attwood S, Bhagwat G, Carbery M, Wilson S, Palanisami T (2021) Estimation of the mass of microplastics ingested – A pivotal first step towards human health risk assessment. J Hazard Mater 404:124004. https://doi.org/10.1016/j.jhazmat.2020.124004
Shi X, Wang X, Peng L, Chen Y, Liu C, Yang Q, Wu K (2022) Associations between polybrominated diphenyl ethers (PBDEs) levels in adipose tissues and female menstrual cycle and menstrual bleeding duration in Shantou, China. Environ Pollut 301:119025. https://doi.org/10.1016/j.envpol.2022.119025
Shi X, Xu T, Cui W, Qi X, Xu S (2023) Combined negative effects of microplastics and plasticizer DEHP: The increased release of Nets delays wound healing in mice. Sci Total Environ 862:160861. https://doi.org/10.1016/j.scitotenv.2022.160861
Sijoon L, Kang KK, Sung SE, Choi JH, Sung M, Seong KY, Sunjong L, Yang SY, Seo MS, Kim K (2022) Toxicity study and quantitative evaluation of polyethylene microplastics in ICR mice. Polymers 14:402. https://doi.org/10.3390/polym14030402
Sijoon L, Dongseon K, Kang KK, Sung SE, Choi JH, Sung M, Shin CH, Jeon E, Dongkyu K, Dongmin K, Sunjong L, Kim HK, Kim K (2023) Toxicity and biodistribution of fragmented polypropylene microplastics in ICR mice. Int J Mol Sci 24:8463. https://doi.org/10.3390/ijms24108463
Soltani NS, Taylor MP, Wilson SP (2021) Quantification and exposure assessment of microplastics in Australian indoor house dust. Environ Pollut 283:117064. https://doi.org/10.1016/j.envpol.2021.117064
Somin L, An KS, Kim HJ, Noh HJ, Lee JW, Lee J, Song KS, Chae C, Ryu HY (2022) Pharmacokinetics and toxicity evaluation following oral exposure to bisphenol F. Arch Toxicol 96:1711–1728. https://doi.org/10.1007/s00204-022-03246-w
Stock V, Böhmert L, Lisicki E, Block R, Cara-Carmona J, Pack LK, Selb R, Lichtenstein D, Voss L, Henderson CJ, Zabinsky E, Sieg H, Braeuning A, Lampen A (2019) Uptake and effects of orally ingested polystyrene microplastic particles in vitro and in vivo. Arch Toxicol 93:1817–1833. https://doi.org/10.1007/s00204-019-02478-7
Su L, Xue Y, Li L, Yang D, Kolandhasamy P, Li D, Shi H (2016) Microplastics in Taihu Lake, China. Environ Pollut 216:711–719. https://doi.org/10.1016/j.envpol.2016.06.036
Sun W, Yan S, Meng Z, Tian S, Jia M, Huang S, Wang Y, Zhou Z, Diao J, Zhu W (2022) Combined ingestion of polystyrene microplastics and epoxiconazole increases health risk to mice: based on their synergistic bioaccumulation in vivo. Environ Int 166:107391. https://doi.org/10.1016/j.envint.2022.107391
Tang KHD (2022) Abundance of microplastics in wastewater treatment sludge. J Human Earth Futur 3:138–146
Tang KHD (2023a) Microplastics in and near landlocked countries of central and East Asia: a review of occurrence and characteristics. Trop Aquat Soil Pollut 3:120–130
Tang KHD (2023b) Microplastics in agricultural soils in China: sources, impacts and solutions. Environ Pollut 322:121235. https://doi.org/10.1016/j.envpol.2023.121235
Tang KHD (2023c) Environmental co-existence of microplastics and perfluorochemicals: a review of their interactions. Biointerface Res Appl Chem 13:587
Ullah A, Pirzada M, Jahan S, Ullah H, Shaheen G, Rehman H, Butt MA (2018) Bisphenol A and its analogs bisphenol B, bisphenol F, and bisphenol S: comparative in vitro and in vivo studies on the sperms and testicular tissues of rats. Chemosphere 209:508–516. https://doi.org/10.1016/j.chemosphere.2018.06.089
US EPA (2016) Polychlorinated biphenyls (PCBs) (Arochlors). https://www.epa.gov/sites/default/files/2016-09/documents/polychlorinated-biphenyls.pdf. Accessed 30 July 2023.
US EPA (2019) Guidelines for Human Exposure Assessment. https://www.epa.gov/risk/guidelines-human-exposure-assessment. Accessed 30 July 2023.
US EPA (2023) Reference Dose (RfD): Description and Use in Health Risk Assessments. https://www.epa.gov/iris/reference-dose-rfd-description-and-use-health-risk-assessments. Accessed 30 July 2023.
Vianello A, Jensen RL, Liu L, Vollertsen J (2019) Simulating human exposure to indoor airborne microplastics using a Breathing Thermal Manikin. Sci Rep. https://doi.org/10.1038/s41598-019-45054-w
Walczak AP, Hendriksen PJM, Woutersen RA, van der Zande M, Undas AK, Helsdingen R, van den Berg HHJ, Rietjens IMCM, Bouwmeester H (2015) Bioavailability and biodistribution of differently charged polystyrene nanoparticles upon oral exposure in rats. J Nanoparticle Res 17:231. https://doi.org/10.1007/s11051-015-3029-y
Walpole SC, Prieto-Merino D, Edwards P, Cleland J, Stevens G, Roberts I (2012) The weight of nations: an estimation of adult human biomass. BMC Public Health 12:439. https://doi.org/10.1186/1471-2458-12-439
Walton K, Dorne JL, Renwick AG (2001) Uncertainty factors for chemical risk assessment: interspecies differences in the in vivo pharmacokinetics and metabolism of human CYP1A2 substrates. Food Chem Toxicol 39:667–680. https://doi.org/10.1016/S0278-6915(01)00006-0
Wang L, Xu M, Chen J, Zhang X, Wang Q, Wang Y, Cui J, Zhang S (2022) Distinct adverse outcomes and lipid profiles of erythrocytes upon single and combined exposure to cadmium and microplastics. Chemosphere 307:135942. https://doi.org/10.1016/j.chemosphere.2022.135942
Weber CJ, Opp C (2020) Spatial patterns of mesoplastics and coarse microplastics in floodplain soils as resulting from land use and fluvial processes. Environ Pollut 267:115390. https://doi.org/10.1016/j.envpol.2020.115390
Wei J, Wang X, Liu Q, Zhou N, Zhu S, Li Z, Li X, Yao J, Zhang L (2021) The impact of polystyrene microplastics on cardiomyocytes pyroptosis through NLRP3/Caspase-1 signaling pathway and oxidative stress in Wistar rats. Environ Toxicol 36:935–944. https://doi.org/10.1002/tox.23095
Wei Z, Wang Y, Wang S, Xie J, Han Q, Chen M (2022) Comparing the effects of polystyrene microplastics exposure on reproduction and fertility in male and female mice. Toxicology 465:153059. https://doi.org/10.1016/j.tox.2021.153059
Williams RS, Curnick DJ, Brownlow A, Barber JL, Barnett J, Davison NJ, Deaville R, ten Doeschate M, Perkins M, Jepson PD, Jobling S (2021) Polychlorinated biphenyls are associated with reduced testes weights in harbour porpoises (Phocoena phocoena). Environ Int 150:106303. https://doi.org/10.1016/j.envint.2020.106303
Xie X, Deng T, Duan J, Xie J, Yuan J, Chen M (2020) Exposure to polystyrene microplastics causes reproductive toxicity through oxidative stress and activation of the p38 MAPK signaling pathway. Ecotoxicol Environ Saf 190:110133. https://doi.org/10.1016/j.ecoenv.2019.110133
Yu Y, Lin B, Chen X, Qiao J, Li L, Liang Y, Zhang G, Jia Y, Zhou X, Chen C, Kan H (2019) Polybrominated diphenyl ethers in human serum, semen and indoor dust: effects on hormones balance and semen quality. Sci Total Environ 671:1017–1025. https://doi.org/10.1016/j.scitotenv.2019.03.319
Zhang Q, Zhao Y, Du F, Cai H, Wang G, Shi H (2020) Microplastic fallout in different indoor environments. Environ Sci Technol 54:6530–6539. https://doi.org/10.1021/acs.est.0c00087
Zhang W, Sun X, Qi X, Liu X, Zhang Y, Qiao S, Lin H (2022) Di-(2-Ethylhexyl) phthalate and microplastics induced neuronal apoptosis through the PI3K/AKT pathway and mitochondrial dysfunction. J Agric Food Chem 70:10771–10781. https://doi.org/10.1021/acs.jafc.2c05474
Zhang S, Han Y, Peng J, Chen Y, Zhan L, Li J (2023) Human health risk assessment for contaminated sites: a retrospective review. Environ Int 171:107700. https://doi.org/10.1016/j.envint.2022.107700
Zhao X, Zhou Y, Liang C, Song J, Yu S, Liao G, Zou P, Tang KHD, Wu C (2023) Airborne microplastics: occurrence, sources, fate, risks and mitigation. Sci Total Environ 858:159943. https://doi.org/10.1016/j.scitotenv.2022.159943
Zheng H, Wang J, Wei X, Chang L, Liu S (2021) Proinflammatory properties and lipid disturbance of polystyrene microplastics in the livers of mice with acute colitis. Sci Total Environ 750:143085. https://doi.org/10.1016/j.scitotenv.2020.143085
Zhong G, Rao G, Tang L, Wu S, Tang Z, Huang R, Ruan Z, Hu L (2022) Combined effect of arsenic and polystyrene-nanoplastics at environmentally relevant concentrations in mice liver: activation of apoptosis, pyroptosis and excessive autophagy. Chemosphere 300:134566. https://doi.org/10.1016/j.chemosphere.2022.134566
Zuccarello P, Ferrante M, Cristaldi A, Copat C, Grasso A, Sangregorio D, Fiore M, Oliveri Conti G (2019) Exposure to microplastics (<10 μm) associated to plastic bottles mineral water consumption the first quantitative study. Water Res 157:365–371
Funding
The authors declare that no funds, grants, or other support was received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
KHDT contributed to the conceptualization of the review, the writing and editing of original draft, and project administration; RL contributed to the writing of original draft; ZL and DW contributed to the review and editing of the original draft. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tang, K.H.D., Li, R., Li, Z. et al. Health risk of human exposure to microplastics: a review. Environ Chem Lett 22, 1155–1183 (2024). https://doi.org/10.1007/s10311-024-01727-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10311-024-01727-1