Abstract
Purpose of Review
Microplastics in the environment enter the human body through diet, drinking water, and air inhalation. The widespread detection of microplastics in several human tissues was conducted. However, limited knowledge exists on the number of microplastics that can be ingested by humans and the potential adverse effects on various organs. To address these issues, we reviewed the types and abundance of microplastics through different pathways and summarized the average annual intake in humans.
Recent Findings
An adult can ingest about (4.88–5.77) × 105 microplastics/year through the dietary route [including salt (5.00–7.00) × 103, fish (0.50–1.20)×104, fruits (4.48–4.62) × 105, and vegetables (2.96–9.55)×104]. The amount of microplastics ingested via drinking water route was approximately (0.22–1.2)×106 microplastics/year. Inhalation of microplastics via atmospheric environment was nearly (0.21–2.51) × 106 microplastics/year [including indoor (0.16–2.30) × 106 and outdoor (0.46–2.10)×105].
Summary
In conclusion, we found that the human body ingests microplastics most through inhalation, followed by drinking water and diet. We also summarized the types and abundance of microplastics that were enriched in different organs after microplastics entered the human body. Microplastics entering the body would cross the barrier into the target effector organs and cause adverse health effects, mainly including induction of intracellular oxidative stress, genotoxicity, reproductive toxicity, and inflammatory responses. In conclusion, exposure to microplastics can cause many adverse effects on the health of the organism. Thus, an increased awareness of the crisis, urgent discussion, and practical actions are needed to mitigate microplastics contaminants in the environment.
Graphical Abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Microplastics are nearly ubiquitous in environments. The presence of microplastics can be detected from the Tibetan Plateau to the Marianas Trench [1] and from the houses we live in every day to the untouched skies over the Pacific Ocean [2, 3]. Since the concept of microplastics was first introduced in the marine environment in 2004 [4], a large number of studies have emerged to investigate microplastics pollution in the ocean. Microplastics in the ocean are mainly from direct emissions of plastic waste, laundry, marine activities, floating plastics, and industrial emissions. Indoor air microplastics are mainly from home decoration, synthetic textiles [5], air conditioning filters [6, 7], and microplastics from the wear and tear of household items and electronics. Outdoor microplastics are mainly from vehicle emissions and tire wear [8], road marking paint, asphalt on roads [9], and street dust [10]. The main sources of microplastics in soil include the casual disposal of discarded plastic products [11,12,13], agricultural plastic films, plastic components added to agricultural supplies such as fertilizers and pesticides [14, 15], artificial fillers in municipal greening and landscape construction projects, and pollution sources such as industrial wastewater, municipal sewage, and garbage microplastics that enter the soil through water flow. The widespread presence of microplastics in the environment inevitably results in people taking in microplastics from the environment through various means (e.g., diet, drinking water, breathing, and skin contact). Therefore, the calculation of microplastics intake are essential for the assessment of human health risks.
Several studies have reported the presence of microplastics in the human skin, lung [16, 17], liver, spleen, kidney [18•], colon [19], feces [20, 21, 22•, 23], blood [24], saliva [25], placenta [26,27,28,29], and breast milk [30]. This suggests that microplastics enter the body through different pathways, reach everywhere through the circulatory system, and are enriched in the target effector organs. A recent study reported the presence of microplastics in human blood clots [31, 32], which proves that the impact of exogenous microplastics on human health cannot be ignored. Therefore, a comprehensive understanding of the absorption, distribution, metabolism, and excretion characteristics of microplastics in humans is essential to investigate the potential toxicological effects of microplastics exposure.
Numerous in vivo cellular and in vitro animal studies have demonstrated that microplastics exposure cause liver fibrosis and metabolic disorders, significant impairment of kidney function, inflammatory response and functional impairment of lung, ecological imbalance and metabolic disorders of intestinal flora, and impairment of neurological function and affects reproduction. However, the adverse effects and main mechanisms of microplastics on different target effector organs and systems have not been systematically elucidated. We analyzed the literature on the potential toxic effects of microplastics exposure on health in recent years to analyze the health hazards of microplastics comprehensively. The effects of microplastics on organism health at different organ levels were also summarized. Then, new ideas and improved methods for environmental management and medical health fields could be provided.
This study aims to estimate the annual intake of microplastics in humans through different routes and review the types and abundance of microplastics in different tissues and organs in the human body. By summarizing the results of available in vitro cellular and in vivo animal experiments, the hazard of microplastics exposure to different organ tissues are explored, and the potential health effects of microplastics exposure are outlined. This review will contribute to the understanding of the health hazards of airborne microplastics and provide a strong basis for the development of laws and regulations related to plastic products in the environment.
Results
Human Microplastics Exposure Through Different Routes
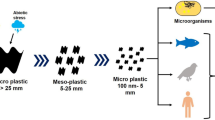
Plastic products bring great convenience to our life, and they also lead to the emergence of plastic waste and pollution. Not properly treated plastic waste will gradually decompose into plastic particles under environmental or human factors. These microplastics will enter into the human body through different ways and cause exposure risks to human health (Fig. 1).
Human Microplastics Exposure Through Dietary Route
Microplastics in Salt and Human Intake
Numerous studies have detected microplastics in table salt extracted from oceans, lake, well, and rock salts in various countries around the world. Salt is an essential food for humans every day. In 2018, global salt consumption reached 300 Mt, of which 11.6% was used for salt and food processing. In sea salt, PET was the most common microplastics, followed by PE and ST. Fragments smaller than 200 µm and fibrous microplastics accounted for 55% of the total microplastics. In lake and well salts, ST is the most common microplastics. ST is an organic cellulose-based polymer that is often used as a release agent in food and cigarette packaging and utilized in the manufacture of fiberglass and rubber products. In general, lake, rock, and well salts have low contamination levels of microplastics compared with sea salt. This condition may be closely related to the local population density and economic development level.
The World Health Organization (WHO) estimates that adults consume approximately 9–12 g of salt per day. Water evaporates during salt crystallization, but microplastics are not removed and remain enriched and retained in the salt body. Salt serves as an important carrier of microplastics intake in humans, and microplastics can be internal and cause health risks during people’s daily salt intake. Based on the data collected from 11 representative papers (Table 1), an adult can ingest about (5.00–7.00) × 103 microplastics from salt in a year.
Microplastics in Aquatic Products and Human Intake
Microplastics in the aquatic environment can be transferred from the environment to the organism and subsequently enter the food web and eventually enrich in the human body. Microplastics have been found in many edible shellfish (mussels, oysters, clams, field snails, and scallops [2, 3, 52, 81,82,83], commercial fish [84], mollusks, and even mammals. PET, PP, PA, PE, and nylon are the main components of microplastics detected in commercial fish, with fibers being the predominant form. By contrast, shellfish (bivalves) are mainly PP, PE, PS, and PET [81]. The reason for the higher percentage of microplastics in the fibrous form in commercial fish species may be that fishing nets are mainly made of synthetic or natural fibers. On the contrary, shellfish culture often uses filter feeding, which leads to its microplastic content in the form of films and fragments. In general, the abundance of microplastics is higher in commercial fish than in shellfish. The reason may be the influence of wider dietary choices and higher nutritional levels in fish, which leads to a higher intake of microplastics in carnivorous and omnivorous fish.
Some research statistics claim that each person consumes about 2.4–4.8 kg of mollusks and 7.3–13.7 kg of lean meat or fish aquatic products per year. Based on the data collected in 18 representative papers (Table 1), an adult can consume approximately (0.50–1.20) × 104 microplastics a year from aquatic products.
Microplastics in Crops and Human Intake
At least 473,000 t of plastic waste is estimated to be released into the soil each year in European Union countries [12]. The presence of microplastics was detected in terrestrial ecosystems such as agricultural fields and river floodplains. Microplastics in soil can be heavily absorbed and enriched by plant roots through processes such as adhesion and uptake. They can migrate upward from the roots and accumulate in the stems and leaves that can be directly consumed. Microplastics can accumulate in crops such as arabidopsis, lettuce, wheat [85], and rice [86]. Studies comparing the number and size of microplastics in fruits and vegetables (carrots, lettuce, broccoli, potatoes, apples, and pears) found that apples were the most contaminated fruit samples, while carrots were the most contaminated vegetables [80••].
Standardized procedures for collecting, fractionating, characterizing, and quantifying polymer particles need to be established due to the inadequacy of current detection methods and instruments. Relatively few studies are available on measuring microplastics content in agricultural crops (fruits and vegetables); currently, only one article reports the daily intake of microplastics from fruits of approximately 4.48–4.62 × 105 and from vegetables of 2.96–9.55 × 104 for adults [80••] (Table 1).
Human Microplastics Exposure Through Drinking Water Route
Surface freshwater (including rivers, lakes, and reservoir water) and groundwater are the main raw materials for drinking water. Microplastics in surface water mainly originate from washing and bathing wastewater produced in human production and life [87, 88•], followed by direct degradation of plastic products in the environment, wear and tear of plastic products, and deposition of microplastics in the atmosphere. Microplastics in groundwater mainly come from contaminated soil leachate and direct injection from surface loss streams. Microplastics contamination in drinking water is more dangerous than other pathways (e.g., fish and seafood) because the amount of water consumed daily by humans far exceeds the amount consumed by fish and seafood.
Since the first detection of microplastics in tap water by Kosuth et al. in 2018 [69, 89], a large number of studies have successively reported the presence of microplastics in bottled water and beverages, beer [68, 90], tea [68, 90], and functional drinks [68]. The main reason for the higher levels of microplastics in bottled water and beverages than in tap water is the presence of plastic products in the production, processing, and packaging of bottled water and the wear and tear of production equipment, which makes the sources of microplastics contaminant in bottled water more widespread. Fragments are the most common particulate form in bottled water (65%). PP and PET are the most common types of polymers in bottled water, both of which may come from common plastics used in the manufacture of caps and bottles. Different packaging materials have different microplastics contamination in bottled water [89]. Schymanski’s team used micro-Raman spectroscopy to compare drinking water in plastic bottles, glass bottles, and beverage cartons. They found that the microplastics content in glass bottles was lower than that in plastic bottles, and disposable plastic bottles and cartons of beverages had lower microplastics content than recyclable plastic bottles. The repeated use of these plastic products increases the wear and tear of the packaging material and produces more particles of microplastics.
The amount of water consumed by an adult varies according to gender, climate, diet, and physical activity and the WHO guideline value of 2 L per day for an adult (default weight of 60 kg). Based on the data collected from 8 representative papers (Table 1), an adult can consume approximately (0.22–1.20) × 106 microplastics through drinking water routes in a year.
Human Microplastics Exposure Through Air Inhalation Route
Microplastics, as a new type of atmospheric environmental pollutant, are nearly everywhere. The sources and distribution of microplastics in the air have their own characteristics indoors and outdoors. Among them, indoor microplastics are mainly derived from synthetic textiles [5], air conditioning filter [6, 7], and indoor dust, and their main components are PP, PE, and PET [70, 71, 78]. Outdoor microplastics are mainly derived from tire wear [8], road marking coatings, asphalt on the road [9], and street dust [10], and their main components are PET and PE [2, 3, 70, 75••, 76, 78]. The main forms of indoor and outdoor microplastics are not only fibers (95%) [5] but also debris (4%) and foam and film (less than 1%). The reason why fibers account for such a large proportion may be due to the good mechanical properties, wear resistance, and chemical stability of fibers, which are widely used in various industries around the world.
The concentration of microplastics in indoor air is much higher than that in the outdoors [71]. The reason is that outdoor air has more circulation than indoor air, which is conducive to the diffusion of pollutants. Meanwhile, microplastics in indoor air are more widely available than in outdoor sources. Under different indoor environmental conditions, the abundance of microplastics in the air is also different. For example, nail salons have a concentration of microplastics in indoor air (46 ± 55 microplastics/m3) 1.6 times that in outdoor air (28 ± 24 microplastics/m3) [6, 7]. The reason is that the materials used in nail art involve more microplastics. Another example is that the concentration of microplastics in the air of rooms with clothes dryers in the home (1.6 ± 1.8 blue fibers/m3) is higher than that in rooms without dryers (0.17 ± 0.27 blue fibers/m3). This condition may be due to the fact that synthetic textiles themselves are the source of microplastics on the one hand, and the heating and ventilation function of the dryer accelerates the spread of microplastics on the other hand.
The main ways for microplastics in the atmosphere to enter the human body are respiratory inhalation. According to statistics, adults consume around 10–20 m3 of air per day. According to relevant literature, people spend nearly 87% of the day indoors and only 13% outdoors. Based on the data collected in 9 representative papers, the amount of microplastics exposed by inhalation per person per year was about (0.21–2.51) × 106 microplastics/year, including about (0.16–2.30) × 106 microplastics inhaled indoors and (0.46–2.10) × 105 microplastics inhaled outdoors (Table 1).
Accumulation of Microplastics in the Human Body
The human body ingests microplastics from the environment on a daily basis through atmospheric inhalation, diet, drinking water, and skin contact. Microplastics smaller than 10 µm can cross cell membranes, enter the human circulatory system, and diffuse and transfer through various organs and tissues in the human body by adsorption, migration, and transformation. Table 2 summarizes the current studies related to the size, category, content, and abundance of microplastics in various tissues that have been detected in different countries, regions, and populations. In two recent studies, PP, PET, and PS were found to be the top three most abundant microplastics detected in feces, and their detected sizes were also broadly similar, which were mainly in the range of 20–800 µm [21, 22•, 23]. PP, which is the most abundant microplastics in food, is mainly used for food packaging. PET is commonly used for the packaging of daily necessities and containers for drinking water. PS is commonly used for manufacturing building materials, toys, and stationery. Since people are exposed to more of these three plastics on a daily basis, it is not surprising that they are found in the highest amounts in human feces. However, a study by the team of Wibowo in 2021 found the highest levels of HDPE microplastics in stool samples [20], and the main reason for this difference was the high concentration of HDPE in the toothpaste used by the subjects. Therefore, human intake of microplastics from toothpaste may also be an important source of microplastics contamination in the body.
Zhang’s team found significantly higher concentrations of PET in infants’ feces than in adults, and PET is often made into polyester, which is one of the main raw materials for clothing and carpets [22•, 23]. Therefore, the team believed that the main reason for the difference may be that infants crawl on carpets, chew, and suck on fabrics frequently,thus, they are more likely to be exposed to PET microplastics than adults. Moreover, the detection of microplastics in breast milk suggests that breast milk may be another important source of microplastics in newborns [30]. Previously, Ragusa et al. first detected PP microplastics of 5–10 µm size in the human placenta [28], while Braun et al. detected microplastics of larger particle size (50–500 µm) and higher diversity (PP, PET, and PS) in the placenta [27]. In fact, Grafmueller et al. in 2015 already demonstrated the ability of polystyrene nanoparticles to cross the placental barrier through an ex vivo human placenta perfusion model. Therefore, microplastics in infants and children may be transferred from the mother to her body through the placenta in addition to their uptake in the external environment and breast milk after birth. By contrast, an Iranian study examined and compared microplastics in the placenta of normal and intrauterine growth retardation (IUGR) pregnancies and found significantly higher levels of microplastics in the placenta of IUGR pregnancies than in the normal group. The team also measured and calculated the relationship between various indicators of the newborn fetus (weight, length, head circumference, and 1 min Apgar score) and the abundance of microplastics in the placenta [26]. They found a significant negative correlation between all indicators and the abundance of microplastics. The results of this study suggest that maternal microplastics are likely to pass through the placenta to the fetus and have toxic effects on the fetus.
Air inhalation is another important route of environmental microplastics entering the human body. Thus, the lungs are an important site for airborne microplastics accumulation. In 1998, Pauly et al. first detected the presence of microplastics in human lung tissues. Subsequent studies further detected microplastics of different sizes (1.60–5.56 µm) and categories (PP, PET, and PS) with different contents (31 and 39 microplastics) in human lung tissues [2, 3]. Given that the lungs are an organ for gas exchange with the outside, they are easily affected by the surrounding environment and people’s lifestyles [91, 92•]. Jiang et al. showed a higher abundance of microplastics in sputum and nasal lavage fluid of office staff than that of couriers. Moreover, sputum and nasal lavage fluid of office staff measured more PVC (used in the production of building materials and furniture), while couriers were detected with higher concentrations of PE (used in the production of masks) and PA (courier packaging) in the case of the COVID-19 pandemic.
A recent study first confirmed the presence of microplastics, including PP, PET, and PS, in the human blood [24]. This study provides strong evidence that microplastics can be transferred to the human body through blood circulation. The discovery of microplastics in human blood clots indicates that the impact of microplastics exposure on human health should not be underestimated. The study showed that microplastics detected in blood clots were larger, which may be caused by the aggregation of small particles in the body and the formation of large particles [31, 32]. Abbasi et al. also found the presence of microplastics in human saliva, skin, and hair. PP and PET were the most abundant, and hair had the most presence and saliva had the least [25]. Thomas et al. also found the presence of microplastics in the liver, spleen, and kidney. This study provides evidence for the aggregation of microplastics in peripheral organs through blood circulation [18•].
So far, the presence of different types and abundance of microplastics has been found in the human feces, colon, lung, placenta, breast milk, blood, liver, spleen, kidney, hand, and face skin in many places (Fig. 2), among which PP, PE, PVC, and PS are the most abundant, and these plastics are the types that humans are frequently exposed to in daily life. When microplastics in the environment enter the body through different pathways, they may reach peripheral organs through blood transport and other means. The particle size of microplastics detected in the placenta, breast milk, lungs, and other places is relatively small, while the particle size in the colon and feces is larger. We speculate that smaller particles are easily absorbed or transferred through the human body, while larger particles are more likely to accumulate and be expelled through the human body. In summary, the results of this part of the study show that environmental microplastics will accumulate in multiple organ tissues after entering the human body through different ways. This phenomenon not only threatens the body’s health but also causes certain effects on the newborn, which deserves extensive attention.
Potential Health Risks and Mechanisms of Microplastics Exposure
The increasing pollution of microplastics poses huge potential risks to human health. After entering the human body through the respiratory tract and digestive tract, microplastics can damage various barriers, induce oxidative stress, regulate gene expression, and eventually lead to functional impairment of corresponding organs and systems, which seriously endangers health.
Nervous System Function Impairment
Small-size microplastics can penetrate the blood–brain barrier, which leads to increased reactive oxygen species (ROS) and malondialdehyde (MDA) levels and significantly lowered glutathione (GSH) levels. Therefore, microplastics can induce oxidative stress in mouse brain tissue, reduce acetylcholine levels, and affect learning and memory function in mice [94]. Microplastics exposure also reduce the expression of blood–brain barrier (BBB) connexin, stimulates reactive oxygen species production to induce nerve cell apoptosis, and promotes microthrombosis; these phenomena reduce the number of Purkinje cells [95], which leads to neurological dysfunction (Fig. 3A).
A Exposure to microplastics can cause neurological dysfunction. B Microplastics exposure to lung inflammatory response and functional damage. C Microplastics exposure causes liver fibrosis and metabolic disorders. D Microplastics lead to ecological imbalance in the intestinal flora and metabolic disorders. E Microplastics exposure causes serious damage to the kidney function. F Effect of microplastics on the reproductive system
Pulmonary Inflammatory Response and Functional Impairment
Studies have found that microplastics that enter the body through respiration after exposure can enter the deep alveoli. Long-term exposure to microplastics in the air may lead to lung damage, altered lung morphology, protective lung barrier damage, inflammatory response, and functional damage [96]. The results of lung examination of workers exposed to high concentrations of microplastic factories showed that the incidence of chronic interstitial pneumonia among workers was significantly increased [97], and it was accompanied by pulmonary hair glass nodule phenomenon [6, 7]. Microplastics that enter the human body through the respiratory tract will enter the respiratory epithelial cells through various ways. This condition will induce phosphorylation and related protein expression reduction through oxidative stress, which destroys the tight connection between lung cells. Ultimately, lung barrier function damage occurs [98].
Microplastics disruption of protective lung barrier function increases the risk of lung disease and easily induces inflammatory responses and functional impairment. Previous results have shown that cellular inflammatory infiltration induced pneumonia in the lungs of mice exposed to high concentrations of microplastics, macrophage aggregation in bronchi and alveoli, and increased mucus production with asthma symptoms [99]. At the same time, microplastics can induce the formation of reactive oxygen species, cause cytotoxic and inflammatory effects, and trigger the corresponding apoptotic pathway by inducing the expression of pro-inflammatory cytokines and proapoptotic proteins [100]; ultimately, the normal function of the lungs is affected (Fig. 3B).
Hepatic Fibrosis and Metabolic Disorders
The liver, as the largest gland in the human body, has extremely complex functions and plays an important role in maintaining the body’s health. The liver is the most active metabolic organ of the body. It is involved in the synthesis, transformation, and decomposition of proteins, lipids, sugars, and other substances. When the microplastics invade the liver through blood circulation, it will cause impaired liver function, induce the damage and release of DNA in the nucleus and mitochondria, activate the corresponding pathway, promote the expression of pro-inflammatory cytokines, and enhance liver fibrosis [101, 102] (Fig. 3C).
The aggregation of microplastics in liver tissues can inhibit the accumulation of fatty acids, fatty acid methyl esters, and fatty acid ethyl esters in the liver. This condition destroys the normal lipid metabolism, which causes hepatic steatosis [103, 104]. In addition to the abnormal lipid metabolism, some studies have reported that exposure to microplastics in the environment can affect the fatty acid metabolism, amino acid metabolism, and carbon metabolism in the zebrafish liver. Therefore, microplastics exposure can induce hepatic glucose and lipid metabolism disorders [102]. At the same time, microplastics can absorb more toxic and harmful substances than their large surface area, such as heavy metal cadmium [105•]). Microplastics have been reported to induce liver iron death caused by heavy metal poisoning [106].
Ecological Imbalance and Metabolic Disorder of Intestinal Flora
The intestine, including the large and small intestines, is an important part of digestion and absorption, and it is also immune competent (lymph nodes). Microplastics that enter the body through feeding and respiration can gather in the intestine, larger microplastics can be excreted with metabolites, and smaller microplastics can enter the system and tissues through the active uptake and phagocytosis of intestinal epithelial cells. The microplastics in the gut can lead to intestinal microbial community imbalance, intestinal barrier dysfunction, and metabolic disorders.
Gut flora can regulate intestinal peristalsis and digestive juice secretion and participate in the nutrient digestion and absorption of the body and is the health guard of the body [107]. Intake of high concentrations of microplastics can affect the balance of intestinal flora. It can change the species and number of intestinal microorganisms, which results in altered bacterial abundance and flora diversity due to the imbalance of intestinal flora [108, 109]. This condition causes the release of some toxic bacterial products, which causes inflammation. As a result, the immune system is affected, which leads to the body’s increased susceptibility to pathological infections or chronic diseases, as well as intestinal nutrient metabolism disorders [110]. After exposure to microplastics by feeding methods, the abundance of Staphylococcus in the gut increases significantly, while the abundance of Parabacteroides is significantly decreased. This variation changes the special physiological function that the gut is supposed to have, which potentially leads to related diseases. Microplastics exposure also reduce mouse intestinal mucus secretion, induces oxidative stress, causes epithelial cell apoptosis, increases intestinal permeability, and leads to intestinal barrier damage [111, 112] (Fig. 3D).
Significant Impairment of Renal Function
The kidney is one of the important target organs of microplastics aggregation. Its exposure can cause significant damage to kidney function in mice [113, 114]. The main mechanism is through the induced oxidative stress [115], which causes an inflammatory response and tissue damage. When microplastics enter the body and are absorbed by kidney cells, they can lead to mitochondrial ROS production and the expression of associated proteins [116]. Meanwhile, they can increase the expression of genes related to ER oxidative stress and inflammatory response in kidney cells. Regulation of related signaling pathways through gene expression affects renal cell ER stress, cellular inflammation, and autophagy pathways, which leads to kidney injury [51, 117] (Fig. 3E).
Reproductive Capacity Impairment
The accumulation of microplastics in the reproductive organs can lead to reproductive toxicity and affect the reproductive capacity. Microplastics can induce testicular inflammation, destroy the testicular blood barrier [84], activate the NF-kB signaling pathway and the p38 MAPK signaling pathway, and induce inflammation to cause testicular sperm abnormalities. These phenomena lead to a significant decrease in sperm number and motility and a significant increase in sperm malformation rate [118, 119]. At the same time, microplastics can also induce ovarian inflammation, reduce the first pole body extrusion rate and superovulation survival rate [86], and decrease the quality of oocytes in female mice. Microplastics induce apoptosis of ovarian granulosa cell and uterine pyroptosis and fibrosis through oxidative stress [31, 35, 118,119,120], which lead to infertility in female mice (Fig. 3F).
In brief, microplastics exposure is known to cause metabolic disorders in the liver, pulmonary inflammatory response, and significant impairment of renal function and affect the nervous and reproductive systems. At the same time, they will also cause intestinal flora disorders, seriously affect immunity and digestion, and lead to great hidden dangers to the body. All these considerations remind human beings to pay attention to environmental protection, curb microplastics pollution, and protect life and health.
Summary and Outlook
The large number of microplastics detected in the aqueous environment has encouraged most of the current research to focus on the detection of the type and abundance of microplastics in the aqueous environment. However, the results of this study found that the abundance and variety of microplastics absorbed by humans through atmospheric environments may be larger than those in water and soil environments. Microplastics absorbed by the human body through the atmospheric environment also enter the body directly without any processing (e.g., drinking water may be filtered and food may be cooked) and may therefore cause a greater health risk. Microplastics in the atmosphere are smaller in size, are more penetrating when absorbed by humans, have a larger specific surface area, are more likely to adsorb toxic organic compounds and heavy metals, and can be directly and continuously exposed to humans compared with those in marine and soil environments. As a result, airborne microplastics pose a greater potential risk to human health. However, a gap still exists in the research on modeling exposure to microplastics in the “real atmosphere.” Therefore, more attention should be paid to the health risks posed by microplastics in the atmosphere. However, no standardized procedure is available for the collection and analysis of microplastics. Therefore, standardizing the collection of microplastics in the environment is still impossible. Microplastics smaller than 5 µm cannot be detected by current detection methods, but the abundance of microplastics in the air tends to increase with the decrease in particle size. Therefore, new technologies that can detect smaller particle sizes and a wider range of microplastics need to be developed.
To date, microplastics have been detected in more than 10 organs and tissues in the human body. However, a gap still exists in the detection of important parts of the body such as the heart, brain, and spinal cord. Researchers should minimize the use of plastic products during sampling and testing to reduce possible cross-contamination issues. Current studies on the health effect of microplastics exposure are still limited to high concentrations, single species, single particle size, and acute exposure conditions. The uptake and accumulation of microplastics in the real environment are a complex, long-term, chronic process. Therefore, researchers need to pay more attention to the potential health risk of microplastics exposure under real environmental conditions. The long-term lasting effects of microplastics exposure and the impact on their offspring should be comprehensively explored as well. Researchers can also draw from epidemiological and occupational studies to understand the current potential health risks regarding microplastics. The complexity of environmental conditions is that environmental pollutants can often act in concert with other pollutants to expose humans. Thus, research should focus more on the synergistic and antagonistic effects of pollutants.
This study explores the exposure of microplastics in the human body under different routes, investigates the distribution characteristics of microplastics in different parts of the human body, and summarizes the current analysis on microplastics exposure, target effector organs, and potential health risk assessment. The results show that air inhalation is the most common mode of microplastics intake in humans compared with dietary and drinking routes. Reviewing the detection of microplastics in different parts of the human body and analyzing the adverse effects of microplastics exposure on liver fibrosis and metabolic disorders, significant impairment of kidney function, inflammatory response and functional impairment of the lung, ecological imbalance and metabolic disorders of intestinal flora, reproduction, and neurological impairment can arouse public attention and encourage people to reduce the use of single-use plastic products. In conclusion, exposure to microplastics in the environment will cause many adverse effects on body health. Thus, raising people’s awareness of the crisis, urgent discussion, and practical actions are necessary to reduce microplastics pollution in the environment.
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Lebreton L, Andrady A. Future scenarios of global plastic waste generation and disposal. Palgrave Commun. 2019;5(1). https://doi.org/10.1057/s41599-018-0212-7.
Liu K, Wang X, Fang T, Xu P, Zhu L, Li D. Source and potential risk assessment of suspended atmospheric microplastics in Shanghai. Sci Total Environ. 2019;675:462–71. https://doi.org/10.1016/j.scitotenv.2019.04.110.
Liu K, Wu T, Wang X, Song Z, Zong C, Wei N, Li D. Consistent transport of terrestrial microplastics to the ocean through atmosphere. Environ Sci Technol. 2019;53(18):10612–9. https://doi.org/10.1021/acs.est.9b03427.
Thompson RC, Olsen Y, Mitchell RP, Davis A, Rowland SJ, John AWG, McGonigle D, Russell AE. Lost at sea: Where is all the plastic? Science. 2004;304(5672):838–838. https://doi.org/10.1126/science.1094559.
Chen G, Feng Q, Wang J. Mini-review of microplastics in the atmosphere and their risks to humans. Sci Total Environ. 2020;703. https://doi.org/10.1016/j.scitotenv.2019.135504.
Chen Q, Gao J, Yu H, Su H, Yang Y, Cao Y, Zhang Q, Ren Y, Hollert H, Shi H, Chen C, Liu H. An emerging role of microplastics in the etiology of lung ground glass nodules. Environ Sci Eur. 2022;34(1):25. https://doi.org/10.1186/s12302-022-00605-3.
Chen Y, Li X, Zhang X, Zhang Y, Gao W, Wang R, He D. Air conditioner filters become sinks and sources of indoor microplastics fibers. Environ Pollut. 2022;292. https://doi.org/10.1016/j.envpol.2021.118465.
Kole PJ, Löhr AJ, Van Belleghem F, Ragas A. Wear and tear of tyres: A stealthy source of microplastics in the environment. Int J Environ Res Public Health. 2017;14(10). https://doi.org/10.3390/ijerph14101265.
Kang H, Park S, Lee B, Kim I, Kim S. Concentration of microplastics in road dust as a function of the drying period—a case study in G City, Korea. Sustainability. 2022;14(5). https://doi.org/10.3390/su14053006.
Dehghani S, Moore F, Akhbarizadeh R. Microplastic pollution in deposited urban dust, Tehran metropolis. Iran Environ Sci Pollut Res Int. 2017;24(25):20360–71. https://doi.org/10.1007/s11356-017-9674-1.
Habib D, Cannone L. Synthetic fibers as indicators of municipal sewage sludge, sludge products, and sewage treatment plant effluents. Wat Air and Soil Poll. 1998.
Horton AA, Walton A, Spurgeon DJ, Lahive E, Svendsen C. Microplastics in freshwater and terrestrial environments: Evaluating the current understanding to identify the knowledge gaps and future research priorities. Sci Total Environ. 2017;586:127–41. https://doi.org/10.1016/j.scitotenv.2017.01.190.
Zubris KAV, Richards BK. Synthetic fibers as an indicator of land application of sludge. Environ Pollut. 2005;138(2):201–11. https://doi.org/10.1016/j.envpol.2005.04.013.
Kasirajan S, Ngouajio M. Polyethylene and biodegradable mulches for agricultural applications: A review. Agron Sustain Dev. 2012;32(2):501–29. https://doi.org/10.1007/s13593-011-0068-3.
Kyrikou I, Briassoulis D. Biodegradation of agricultural plastic films: A critical review. J Polym Environ. 2007;15(2):125–50. https://doi.org/10.1007/s10924-007-0053-8.
Amato-Lourenço LF, Carvalho-Oliveira R, Júnior GR, dos Santos Galvão L, Ando RA, Mauad T. Presence of airborne microplastics in human lung tissue. J Hazard Mater. 2021;416: 126124.
Jenner LC, Rotchell JM, Bennett RT, Cowen M, Tentzeris V, Sadofsky LR. Detection of microplastics in human lung tissue using μFTIR spectroscopy. Sci Total Environ. 2022;831: 154907.
• Horvatits T, Tamminga M, Liu B, Sebode M, Carambia A, Fischer L, Püschel K, Huber S, Fischer EK. Microplastics detected in cirrhotic liver tissue. EBioMedicine. 2022;82: 104147. These results indicate that chronic liver disease seems to be a key driver in MP accumulation in the human liver.
Ibrahim YS, Tuan Anuar S, Azmi AA, Wan Mohd Khalik WMA, Lehata S, Hamzah SR, Ismail D, Ma ZF, Dzulkarnaen A, Zakaria Z. Detection of microplastics in human colectomy specimens. JGH Open. 2021;5(1):116–21.
Luqman A, Nugrahapraja H, Wahyuono RA, Islami I, Haekal MH, Fardiansyah Y, Putri BQ, Amalludin FI, Rofiqa EA, Götz F. Microplastic contamination in human stools, foods, and drinking water associated with Indonesian coastal population. Environments. 2021;8(12):138.
Schwabl P, Köppel S, Königshofer P, Bucsics T, Trauner M, Reiberger T, Liebmann B. Detection of various microplastics in human stool: A prospective case series. Ann Intern Med. 2019;171(7):453–7.
• Zhang J, Wang L, Trasande L, Kannan K. Occurrence of polyethylene terephthalate and polycarbonate microplastics in infant and adult feces. Environ Sci Technol Lett. 2021;8(11):989–94. The study authors concluded that infants have higher exposure to microplastics than adults and attributed the higher exposure to infants to the widespread use of products such as toys, teether, and bottles.
Zhang N, Li YB, He HR, Zhang JF, Ma GS. You are what you eat: Microplastics in the feces of young men living in Beijing. Sci Total Environ. 2021;767.
Leslie HA, Van Velzen MJ, Brandsma SH, Vethaak AD, Garcia-Vallejo JJ, Lamoree MH. Discovery and quantification of plastic particle pollution in human blood. Environ Int. 2022;163: 107199.
Abbasi S, Turner A. Human exposure to microplastics: A study in Iran. J Hazard Mater. 2021;403.
Amereh F, Amjadi N, Mohseni-Bandpei A, Isazadeh S, Mehrabi Y, Eslami A, Naeiji Z, Rafiee M. Placental plastics in young women from general population correlate with reduced foetal growth in IUGR pregnancies. Environ Pollut. 2022;314: 120174.
Braun T, Ehrlich L, Henrich W, Koeppel S, Lomako I, Schwabl P, Liebmann B. Detection of microplastic in human placenta and meconium in a clinical setting. Pharmaceutics. 2021;13(7):921.
Ragusa A, Svelato A, Santacroce C, Catalano P, Notarstefano V, Carnevali O, Papa F, Rongioletti MCA, Baiocco F, Draghi S. Plasticenta: First evidence of microplastics in human placenta. Environ Int. 2021;146.
Zhu L, Zhu J, Zuo R, Xu Q, Qian Y, Lihui A. Identification of microplastics in human placenta using laser direct infrared spectroscopy. Sci Total Environ. 2022;159060.
Ragusa A, Notarstefano V, Svelato A, Belloni A, Gioacchini G, Blondeel C, Zucchelli E, De Luca C, D’Avino S, Gulotta A. Raman microspectroscopy detection and characterisation of microplastics in human breastmilk. Polymers. 2022;14(13):2700.
Wu D, Feng Y, Wang R, Jiang J, Guan Q, Yang X, Wei H, Xia Y, Luo Y. Pigment microparticles and microplastics found in human thrombi based on Raman spectral evidence. J Adv Res. 2022.
Wu H, Xu T, Chen T, Liu J, Xu S. Oxidative stress mediated by the TLR4/NOX2 signalling axis is involved in polystyrene microplastic-induced uterine fibrosis in mice? Sci Total Environ. 2022;838:155825. https://doi.org/10.1016/j.scitotenv.2022.155825.
Renzi M, Blaskovic A. Litter & microplastics features in table salts from marine origin: Italian versus Croatian brands. Mar Pollut Bull. 2018;135:62–8. https://doi.org/10.1016/j.marpolbul.2018.06.065.
Masia P, Ardura A, Garcia-Vazquez E. Microplastics in seafood: Relative input of Mytilus galloprovincialis and table salt in mussel dishes. Food Res Int (Ottawa, Ont). 2022;153:110973. https://doi.org/10.1016/j.foodres.2022.110973.
Kim JS, Lee HJ, Kim SK, Kim HJ. Global pattern of microplastics (MPs) in commercial food-grade salts: Sea salt as an indicator of seawater MP pollution. Environ Sci Technol. 2018;52(21):12819–28. https://doi.org/10.1021/acs.est.8b04180.
• Yang DQ, Shi HH, Li L, Li JN, Jabeen K, Kolandhasamy P. Microplastic pollution in table salts from China. Environ Sci Technol. 2015;49(22):13622–7. https://doi.org/10.1021/acs.est.5b03163. This study demonstrates that the abundance of microplastics in sea salt is significantly higher than in lake salt and rock/well salt. This suggests that seafood is heavily contaminated with microplastics.
Iniguez ME, Conesa JA, Fullana A. Microplastics in Spanish table salt. Sci Rep. 2017;7:8620. https://doi.org/10.1038/s41598-017-09128-x.
Gundogdu S. Contamination of table salts from Turkey with microplastics. Food Addit Contam Part A-Chem Anal Control Expo Risk Assess. 2018;35(5):1006–14. https://doi.org/10.1080/19440049.2018.1447694.
Seth CK, Shriwastav A. Contamination of Indian sea salts with microplastics and a potential prevention strategy. Environ Sci Pollut Res. 2018;25(30):30122–31. https://doi.org/10.1007/s11356-018-3028-5.
Lee H, Kunz A, Shim WJ, Walther BA. Microplastic contamination of table salts from Taiwan, including a global review. Sci Rep. 2019;9:10145. https://doi.org/10.1038/s41598-019-46417-z.
Zick Y. Uncoupling insulin signalling by serine/threonine phosphorylation: A molecular basis for insulin resistance. Biochem Soc Trans. 2004;32:812–6. https://doi.org/10.1042/bst0320812.
Karami A, Golieskardi A, Choo CK, Larat V, Galloway TS, Salamatinia B. The presence of microplastics in commercial salts from different countries. Sci Rep. 2017;7:46173. https://doi.org/10.1038/srep46838.
Xu XY, Wong CY, Tam NFY, Lo HS, Cheung SG. Microplastics in invertebrates on soft shores in Hong Kong: Influence of habitat, taxa and feeding mode. Sci Total Environ. 2020;715:136999. https://doi.org/10.1016/j.scitotenv.2020.136999.
Phuong NN, Poirier L, Pham QT, Lagarde F, Zalouk-Vergnoux A. Factors influencing the microplastic contamination of bivalves from the French Atlantic coast: Location, season and/or mode of life? Mar Pollut Bull. 2018;129(2):664–74. https://doi.org/10.1016/j.marpolbul.2017.10.054.
Avio CG, Pittura L, d’Errico G, Abel S, Amorello S, Marino G, Gorbi S, Regoli F. Distribution and characterization of microplastic particles and textile microfibers in Adriatic food webs: General insights for biomonitoring strategies. Environ Pollut. 2020;258:113766. https://doi.org/10.1016/j.envpol.2019.113766.
Reguera P, Vinas L, Gago J. Microplastics in wild mussels (Mytilus spp.) from the north coast of Spain. Sci Mar. 2019;83(4):337–47. https://doi.org/10.3989/scimar.04927.05A.
Digka N, Tsangaris C, Torre M, Anastasopoulou A, Zeri C. Microplastics in mussels and fish from the Northern Ionian Sea. Mar Pollut Bull. 2018;135:30–40. https://doi.org/10.1016/j.marpolbul.2018.06.063.
Li JN, Green C, Reynolds A, Shi HH, Rotchell JM. Microplastics in mussels sampled from coastal waters and supermarkets in the United Kingdom. Environ Pollut. 2018;241:35–44. https://doi.org/10.1016/j.envpol.2018.05.038.
Li JN, Qu XY, Su L, Zhang WW, Yang DQ, Kolandhasamy P, Li DJ, Shi HH. Microplastics in mussels along the coastal waters of China. Environ Pollut. 2016;214:177–84. https://doi.org/10.1016/j.envpol.2016.04.012.
Wu FZ, Wang YJ, Leung ONH, Huang W, Zeng JN, Tang YB, Chen JF, Shi AQ, Yu X, Xu XQ, Zhang HG, Cao L. Accumulation of microplastics in typical commercial aquatic species: A case study at a productive aquaculture site in China. Sci Total Environ. 2020;708. https://doi.org/10.1016/j.scitotenv.2019.135432.
Zhang DD, Cui YZ, Zhou HH, Jin C, Yu XW, Xu YJ, Li YH, Zhang CF. Microplastic pollution in water, sediment, and fish from artificial reefs around the Ma’an Archipelago, Shengsi, China. Sci Total Environ. 2020;703:134768. https://doi.org/10.1016/j.scitotenv.2019.134768.
Cho Y, Shim WJ, Jang M, Han GM, Hong SH. Abundance and characteristics of microplastics in market bivalves from South Korea. Environ Pollut. 2019;245:1107–16. https://doi.org/10.1016/j.envpol.2018.11.091.
Park TJ, Lee SH, Lee MS, Lee JK, Lee SH, Zoh KD. Occurrence of microplastics in the Han River and riverine fish in South Korea. Sci Total Environ. 2020;708:134535. https://doi.org/10.1016/j.scitotenv.2019.134535.
Bucol LA, Romano EF, Cabcaban SM, Siplon LMD, Madrid GC, Bucol AA, Polidoro B. Microplastics in marine sediments and rabbitfish (Siganus fuscescens) from selected coastal areas of Negros Oriental, Philippines. Mar Pollut Bull. 2020;150:110685. https://doi.org/10.1016/j.marpolbul.2019.110685.
Vries AN, Govoni D, Arnason SH, Carlsson P. Microplastic ingestion by fish: Body size, condition factor and gut fullness are not related to the amount of plastics consumed. Mar Pollut Bull. 2020;151:110827. https://doi.org/10.1016/j.marpolbul.2019.110827.
Scott N, Porter A, Santillo D, Simpson H, Lloyd-Williams S, Lewis C. Particle characteristics of microplastics contaminating the mussel Mytilus edulis and their surrounding environments. Mar Pollut Bull. 2019;146:125–33. https://doi.org/10.1016/j.marpolbul.2019.05.041.
Zhu JM, Zhang Q, Li YP, Tan SD, Kang ZJ, Yu XY, Lan WL, Cai L, Wang JZ, Shi HH. Microplastic pollution in the Maowei Sea, a typical mariculture bay of China. Sci Total Environ. 2019;658:62–8. https://doi.org/10.1016/j.scitotenv.2018.12.192.
Catarino AI, Macchia V, Sanderson WG, Thompson RC, Henry TB. Low levels of microplastics (MP) in wild mussels indicate that MP ingestion by humans is minimal compared to exposure via household fibres fallout during a meal. Environ Pollut. 2018;237:675–84. https://doi.org/10.1016/j.envpol.2018.02.069.
Martinelli JC, Phan S, Luscombe CK, Padilla-Gamino JL. Low incidence of microplastic contaminants in Pacific oysters (Crassostrea gigas Thunberg) from the Salish Sea, USA. Sci Total Environ. 2020;715:136826. https://doi.org/10.1016/j.scitotenv.2020.136826.
Danopoulos E, Twiddy M, Rotchell JM. Microplastic contamination of drinking water: A systematic review. PloS One. 2020;15(7):e0236838. https://doi.org/10.1371/journal.pone.0236838.
Mukotaka A, Kataoka T, Nihei Y. Rapid analytical method for characterization and quantification of microplastics in tap water using a Fourier-transform infrared microscope. Sci Total Environ. 2021;790:148231. https://doi.org/10.1016/j.scitotenv.2021.148231.
Kosuth M, Mason SA, Wattenberg EV. Anthropogenic contamination of tap water, beer, and sea salt. PloS One. 2018;13(4):e0194970. https://doi.org/10.1371/journal.pone.0194970.
Lachenmeier DW, Kocareva J, Noack D, Kuballa T. Microplastic identification in German beer - an artefact of laboratory contamination? Dtsch Lebensm-Rundsch. 2015;111(10):437–440. Go to ISI http://WOS:000364600300008
Liebezeit G, Liebezeit E. Synthetic particles as contaminants in German beers. Food Addit Contam Part A Chem Anal Control Expo Risk Assess. 2014;31(9):1574–8. https://doi.org/10.1080/19440049.2014.945099.
Mintenig SM, Loder MGJ, Primpke S, Gerdts G. Low numbers of microplastics detected in drinking water from ground water sources. Sci Total Environ. 2019;648:631–5. https://doi.org/10.1016/j.scitotenv.2018.08.178.
Pivokonsky M, Cermakova L, Novotna K, Peer P, Cajthaml T, Janda V. Occurrence of microplastics in raw and treated drinking water. Sci Total Environ. 2018;643:1644–51. https://doi.org/10.1016/j.scitotenv.2018.08.102.
Wang ZF, Lin T, Chen W. Occurrence and removal of microplastics in an advanced drinking water treatment plant (ADWTP). Sci Total Environ. 2020;700:134520. https://doi.org/10.1016/j.scitotenv.2019.134520.
Shruti VC, Perez-Guevara F, Elizalde-Martinez I, Kutralam-Muniasamy G. First study of its kind on the microplastic contamination of soft drinks, cold tea and energy drinks-future research and environmental considerations. Sci Total Environ. 2020;726:138580. https://doi.org/10.1016/j.scitotenv.2020.138580.
Shruti VC, Perez-Guevara F, Elizalde-Martinez I, Kutralam-Muniasamy G. Toward a unified framework for investigating micro(nano)plastics in packaged beverages intended for human consumption. Environ Pollut. 2021;268:115811. https://doi.org/10.1016/j.envpol.2020.115811.
Dris R, Gasperi J, Mirande C, Mandin C, Guerrouache M, Langlois V, Tassin B. A first overview of textile fibers, including microplastics, in indoor and outdoor environments. Environ Pollut. 2017;221:453–8. https://doi.org/10.1016/j.envpol.2016.12.013.
Liao Z, Ji X, Ma Y, Lv B, Huang W, Zhu X, Fang M, Wang Q, Wang X, Dahlgren R, Shang X. Airborne microplastics in indoor and outdoor environments of a coastal city in Eastern China. J Hazard Mater. 2021;417:126007. https://doi.org/10.1016/j.jhazmat.2021.126007.
Abbasi S, Keshavarzi B, Moore F, Turner A, Kelly FJ, Dominguez AO, Jaafarzadeh N. Distribution and potential health impacts of microplastics and microrubbers in air and street dusts from Asaluyeh County. Iran Environ Pollut. 2019;244:153–64. https://doi.org/10.1016/j.envpol.2018.10.039.
Syafei AD, Nurasrin NR, Assomadi AF, Boedisantoso R. Microplastic pollution in the ambient air of Surabaya, Indonesia. Curr World Environ. 2019;14(2):290–8. https://doi.org/10.12944/cwe.14.2.13.
Gaston E, Woo M, Steele C, Sukumaran S, Anderson SJAS. EXPRESS: Microplastics differ between indoor and outdoor air masses: Insights from multiple microscopy methodologies. 2020b;74(6):000370282092065.
•• Zhu X, Huang W, Fang M, Liao Z, Wang Y, Xu L, Mu Q, Shi C, Lu C, Deng H, Dahlgren R, Shang X. Airborne microplastic concentrations in five megacities of Northern and Southeast China. Environ Sci Technol. 2021;55(19):12871–81. https://doi.org/10.1021/acs.est.1c03618. The results of this study show thatmaximum annual exposure of humans to airborne microplastics was estimated in the range of 1–2 million/year in these megacities, highlighting the need for additional research examining the human health risks from the inhalation of airborne microplastics.
Zhu X, Huang W, Fang M, Liao Z, Wang Y, Xu L, Mu Q, Shi C, Lu C, Deng H, Dahlgren R, Shang X. Airborne microplastic concentrations in five megacities of Northern and Southeast China. Environ Sci Technol. 2021;55(19):12871–81. https://doi.org/10.1021/acs.est.1c03618.
Vianello A, Jensen RL, Liu L, Vollertsen J. Simulating human exposure to indoor airborne microplastics using a breathing thermal manikin. Sci Rep. 2019;9(1):8670. https://doi.org/10.1038/s41598-019-45054-w.
Gaston E, Woo M, Steele C, Sukumaran S, Anderson S. Microplastics differ between indoor and outdoor air masses: Insights from multiple microscopy methodologies. Appl Spectrosc. 2020;74(9):1079–98. https://doi.org/10.1177/0003702820920652.
Xumiao L, Prata JC, Alves JR, Duarte AC, Rocha-Santos T, Cerqueira M. Airborne microplastics and fibers in indoor residential environments in Aveiro, Portugal. Environ Adv. 2021b;6:100134. https://doi.org/10.1016/j.envadv.2021.100134.
•• Oliveri Conti G, Ferrante M, Banni M, Favara C, Nicolosi I, Cristaldi A, Fiore M, Zuccarello P. Micro- and nano-plastics in edible fruit and vegetables. The first diet risks assessment for the general population. Environ Res. 2020;187:109677. https://doi.org/10.1016/j.envres.2020.109677. This study is the first to estimate the daily intake of microplastics by adults and children via vegetables and fruits and found that fruits have higher levels of microplastic pollution compared to vegetables.
Fang C, Zheng RH, Chen HZ, Hong FK, Lin LS, Lin H, Guo HG, Bailey C, Segner H, Mu JL, Bo J. Comparison of microplastic contamination in fish and bivalves from two major cities in Fujian province, China and the implications for human health. Aquaculture. 2019;512:734322. https://doi.org/10.1016/j.aquaculture.2019.734322.
Su L, Cai HW, Kolandhasamy P, Wu CX, Rochman CM, Shi HH. Using the Asian clam as an indicator of microplastic pollution in freshwater ecosystems. Environ Pollut. 2018;234:347–55. https://doi.org/10.1016/j.envpol.2017.11.075.
Wootton N, Sarakinis K, Varea R, Reis-Santos P, Gillanders BM. Microplastic in oysters: A review of global trends and comparison to southern Australia. Chemosphere. 2022;307:136065. https://doi.org/10.1016/j.chemosphere.2022.136065.
Jin H, Ma T, Sha X, Liu Z, Zhou Y, Meng X, Chen Y, Han X, Ding J. Polystyrene microplastics induced male reproductive toxicity in mice. J Hazard Mater. 2021;401:123430. https://doi.org/10.1016/j.jhazmat.2020.123430.
Qi Y, Yang X, Pelaez AM, Huerta Lwanga E, Beriot N, Gertsen H, Garbeva P, Geissen V. Macro- and micro- plastics in soil-plant system: Effects of plastic mulch film residues on wheat (Triticum aestivum) growth. Sci Total Environ. 2018;645:1048–56. https://doi.org/10.1016/j.scitotenv.2018.07.229.
Liu Z, Zhuan Q, Zhang L, Meng L, Fu X, Hou Y. Polystyrene microplastics induced female reproductive toxicity in mice. J Hazard Mater. 2022;424:127629. https://doi.org/10.1016/j.jhazmat.2021.127629.
Chang M. Reducing microplastics from facial exfoliating cleansers in wastewater through treatment versus consumer product decisions. Mar Pollut Bull. 2015;101(1):330–3. https://doi.org/10.1016/j.marpolbul.2015.10.074.
• Hartline NL, Bruce NJ, Karba SN, Ruff EO, Sonar SU, Holden PA. Microfiber masses recovered from conventional machine washing of new or aged garments. Environ Sci Technol. 2016;50(21):11532–8. https://doi.org/10.1021/acs.est.6b03045. This study illustrates that washing synthetic jackets or sweaters causes most microfibers to enter the environment, which is one of the important ways that microplastics enter the environment.
Schymanski D, Goldbeck C, Humpf HU, Furst P. Analysis of microplastics in water by micro-Raman spectroscopy: Release of plastic particles from different packaging into mineral water. Water Res. 2018;129:154–62. https://doi.org/10.1016/j.watres.2017.11.011.
Li YA, Peng L, Fu JX, Dai XL, Wang GQ. A microscopic survey on microplastics in beverages: The case of beer, mineral water and tea. Analyst. 2022;147(6):1099–105. https://doi.org/10.1039/d2an00083k.
Baeza-Martínez C, Olmos S, González-Pleiter M, López-Castellanos J, García-Pachón E, Masiá-Canuto M, Hernández-Blasco L, Bayo J. First evidence of microplastics isolated in European citizens’ lower airway. J Hazard Mater. 2022;438: 129439.
• Jiang Y, Han J, Na J, Fang J, Qi C, Lu J, Liu X, Zhou C, Feng J, Zhu W. Exposure to microplastics in the upper respiratory tract of indoor and outdoor workers. Chemosphere. 2022;307:136067. This study compared the abundance of microplastics in the nasal wash and sputum of couriers from office workers and outdoor workers and found that the abundance of microplastics in the nasal wash of office workers was significantly higher than that of couriers.
•• Huang S, Huang X, Bi R, Guo Q, Yu X, Zeng Q, Huang Z, Liu T, Wu H, Chen Y. Detection and analysis of microplastics in human sputum. Environ Sci Technol. 2022;56(4):2476–86. In this study, 21 microplastics were detected in the sputum of 22 patients with respiratory diseases. This suggests that respiratory inhalation is an important way for airborne microplastics to enter the human body.
Wang S, Han Q, Wei Z, Wang Y, Xie J, Chen M. Polystyrene microplastics affect learning and memory in mice by inducing oxidative stress and decreasing the level of acetylcholine. Food Chem Toxicol. 2022;162:112904. https://doi.org/10.1016/j.fct.2022.112904.
Yin K, Wang D, Zhao H, Wang Y, Zhang Y, Liu Y, Li B, Xing M. Polystyrene microplastics up-regulates liver glutamine and glutamate synthesis and promotes autophagy-dependent ferroptosis and apoptosis in the cerebellum through the liver-brain axis. Environ Pollut. 2022;307:119449. https://doi.org/10.1016/j.envpol.2022.119449.
Prata JC. Airborne microplastics: Consequences to human health? Environ Pollut. 2018;234:115–26. https://doi.org/10.1016/j.envpol.2017.11.043.
Kern DG, Kuhn C, Ely EW, Pransky GS, Mello CJ, Fraire AE, Müller J. Flock worker’s lung: Broadening the spectrum of clinicopathology, narrowing the spectrum of suspected etiologies. Chest. 2000;117(1):251–9. https://doi.org/10.1378/chest.117.1.251.
Dong CD, Chen CW, Chen YC, Chen HH, Lee JS, Lin CH. Polystyrene microplastic particles: In vitro pulmonary toxicity assessment. J Hazard Mater. 2020;385:121575. https://doi.org/10.1016/j.jhazmat.2019.121575.
Lu K, Lai KP, Stoeger T, Ji S, Lin Z, Lin X, Chan TF, Fang JKH, Lo M, Gao L, Qiu C, Chen S, Chen G, Li L, Wang L. Detrimental effects of microplastic exposure on normal and asthmatic pulmonary physiology. J Hazard Mater. 2021;416:126069. https://doi.org/10.1016/j.jhazmat.2021.126069.
Xu M, Halimu G, Zhang Q, Song Y, Fu X, Li Y, Zhang H. Internalization and toxicity: A preliminary study of effects of nanoplastic particles on human lung epithelial cell. Sci Total Environ. 2019;694:133794. https://doi.org/10.1016/j.scitotenv.2019.133794.
Shen R, Yang K, Cheng X, Guo C, Xing X, Sun H, Liu D, Liu X, Wang D. Accumulation of polystyrene microplastics induces liver fibrosis by activating cGAS/STING pathway. Environ Pollut. 2022;300:118986. https://doi.org/10.1016/j.envpol.2022.118986.
Zhao Y, Bao Z, Wan Z, Fu Z, Jin Y. Polystyrene microplastic exposure disturbs hepatic glycolipid metabolism at the physiological, biochemical, and transcriptomic levels in adult zebrafish. Sci Total Environ. 2020;710:136279. https://doi.org/10.1016/j.scitotenv.2019.136279.
Cheng W, Li X, Zhou Y, Yu H, Xie Y, Guo H, Wang H, Li Y, Feng Y, Wang Y. Polystyrene microplastics induce hepatotoxicity and disrupt lipid metabolism in the liver organoids. Sci Total Environ. 2022;806:150328. https://doi.org/10.1016/j.scitotenv.2021.150328.
Ye G, Zhang X, Liu X, Liao X, Zhang H, Yan C, Lin Y, Huang Q. Polystyrene microplastics induce metabolic disturbances in marine medaka (Oryzias melastigmas) liver. Sci Total Environ. 2021;782:146885. https://doi.org/10.1016/j.scitotenv.2021.146885.
• Bradney L, Wijesekara H, Palansooriya KN, Obadamudalige N, Bolan NS, Ok YS, Rinklebe J, Kim KH, Kirkham MB. Particulate plastics as a vector for toxic trace-element uptake by aquatic and terrestrial organisms and human health risk. Environ Pollut. 2019;131:104937. https://doi.org/10.1016/j.envint.2019.104937. This study describes particulate plastics made of synthetic polymers, their origin, and their characteristics with emphasis on how particulate plastics and associated toxic trace elements contaminate terrestrial and aquatic ecosystems.
Xie Y, Hou W, Song X, Yu Y, Huang J, Sun X, Kang R, Tang D. Ferroptosis: Process and function. Cell Death Differ. 2016;23(3):369–79. https://doi.org/10.1038/cdd.2015.158.
Montalban-Arques A, Scharl M. Intestinal microbiota and colorectal carcinoma: Implications for pathogenesis, diagnosis, and therapy. EBioMedicine. 2019;48. https://doi.org/10.1016/j.ebiom.2019.09.050.
Li B, Ding Y, Cheng X, Sheng D, Xu Z, Rong Q, Wu Y, Zhao H, Ji X, Zhang Y. Polyethylene microplastics affect the distribution of gut microbiota and inflammation development in mice. Chemosphere. 2020;244:125492. https://doi.org/10.1016/j.chemosphere.2019.125492.
Lu L, Wan Z, Luo T, Fu Z, Jin Y. Polystyrene microplastics induce gut microbiota dysbiosis and hepatic lipid metabolism disorder in mice. Sci Total Environ. 2018;631–632:449–58. https://doi.org/10.1016/j.scitotenv.2018.03.051.
Gallant MJ, Ellis AK. Prenatal and early-life exposure to indoor air-polluting factors and allergic sensitization at 2 years of age. Ann Allergy Asthma Immunol. 2020;124(3):283–7. https://doi.org/10.1016/j.anai.2019.11.019.
Bisanz JE, Upadhyay V, Turnbaugh JA, Ly K, Turnbaugh PJ. Meta-analysis reveals reproducible gut microbiome alterations in response to a high-fat diet. Cell Host Microbe. 2019;26(2):265-272.e264. https://doi.org/10.1016/j.chom.2019.06.013.
Liang B, Zhong Y, Huang Y, Lin X, Liu J, Lin L, Hu M, Jiang J, Dai M, Wang B, Zhang B, Meng H, Lelaka JJJ, Sui H, Yang X, Huang Z. Underestimated health risks: Polystyrene micro- and nanoplastics jointly induce intestinal barrier dysfunction by ROS-mediated epithelial cell apoptosis. Part Fibre Toxicol. 2021;18(1):20–20. https://doi.org/10.1186/s12989-021-00414-1.
Deng Y, Zhang Y, Lemos B, Ren H. Tissue accumulation of microplastics in mice and biomarker responses suggest widespread health risks of exposure. Sci Rep. 2017;7(1):46687. https://doi.org/10.1038/srep46687.
Prata JC, da Costa JP, Lopes I, Duarte AC, Rocha-Santos T. Environmental exposure to microplastics: An overview on possible human health effects. Sci Total Environ. 2020;702:134455. https://doi.org/10.1016/j.scitotenv.2019.134455.
Deng Y, Zhang Y, Qiao R, Bonilla MM, Yang X, Ren H, Lemos B. Evidence that microplastics aggravate the toxicity of organophosphorus flame retardants in mice (Mus musculus). J Hazard Mater. 2018;357:348–54. https://doi.org/10.1016/j.jhazmat.2018.06.017.
Wang YL, Lee YH, Hsu YH, Chiu IJ, Huang CCY, Huang CC, Chia ZC, Lee CP, Lin YF, Chiu HW. The kidney-related effects of polystyrene microplastics on human kidney proximal tubular epithelial cells HK-2 and male C57BL/6 mice. Environ Health Perspect. 2021;129(5):057003. https://doi.org/10.1289/EHP7612.
Tang J, Ni X, Zhou Z, Wang L, Lin S. Acute microplastic exposure raises stress response and suppresses detoxification and immune capacities in the scleractinian coral Pocillopora damicornis. Environ Pollut. 2018;243:66–74. https://doi.org/10.1016/j.envpol.2018.08.045.
Hou B, Wang F, Liu T, Wang Z. Reproductive toxicity of polystyrene microplastics: In vivo experimental study on testicular toxicity in mice. J Hazard Mater. 2021;405:124028. https://doi.org/10.1016/j.jhazmat.2020.124028.
Hou J, Lei Z, Cui L, Hou Y, Yang L, An R, Wang Q, Li S, Zhang H, Zhang L. Polystyrene microplastics lead to pyroptosis and apoptosis of ovarian granulosa cells via NLRP3/caspase-1 signaling pathway in rats. Ecotoxicol Environ Saf. 2021;212:112012. https://doi.org/10.1016/j.ecoenv.2021.112012.
An R, Wang X, Yang L, Zhang J, Wang N, Xu F, Hou Y, Zhang H, Zhang L. Polystyrene microplastics cause granulosa cells apoptosis and fibrosis in ovary through oxidative stress in rats. Toxicology. 2021;449:152665. https://doi.org/10.1016/j.tox.2020.152665.
Acknowledgements
We thank all the members in our lab for their great assistance with this study.
Funding
This work was supported by the National Key Research and Development Program of China (grant numbers: 2022YFF1202900), the National Natural Science Foundation of China (grant numbers: 31771100 and 32000815), and the key technologies R & D program of Tianjin (21JCZDJC00580).
Author information
Authors and Affiliations
Contributions
Under the supervision of Liqun Chen and Can Wang, Ziye Yang, Zhihong Feng, and Meixue Wang conducted article search, information analysis, and article writing. Ziqi Wang sorted out the information and wrote part of the content. Mingxia Lv cooperatively collected information and compiled some table contents. Jinghao Chang supplemented and revised the details of the article. All authors read and contributed to the manuscript.
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, Z., Wang, M., Feng, Z. et al. Human Microplastics Exposure and Potential Health Risks to Target Organs by Different Routes: A Review. Curr Pollution Rep 9, 468–485 (2023). https://doi.org/10.1007/s40726-023-00273-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40726-023-00273-8