Abstract
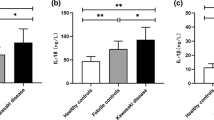
Kawasaki disease is a pressing acute self-limiting inflammatory disorder disease which lack of specific biomarkers. Our research aims to investigate the serum expression of a novel immune regulator PK2 in children with Kawasaki disease and to evaluate the ability of PK2 to predict Kawasaki disease. A total of 70 children with Kawasaki disease in the Children’s Hospital of Chongqing Medical University who were first diagnosed, 20 children with common fever admitted to hospital due to bacterial infection during the same period, and 31 children underwent physical examination were included in this study. Venous blood was collected for complete blood count, CRP, ESR, PCT, and PK2 before clinical intervention. The predictive potential of PK2 as a biomarker for the diagnosis of Kawasaki disease was judged by correlation analysis, the receiver operating characteristic (ROC) and combined score. Compared with healthy children and children with common fever, children diagnosed with Kawasaki disease had significantly lower serum PK2 concentrations (median 28,503.7208 ng/ml, 26,242.5484 ng/ml, and 16,890.2452 ng/ml, respectively, Kruskal–Wallis test: p < 0.0001). Analysis of the existing indicators in other laboratories showed that WBC (Kruskal–Wallis test: p < 0.0001), PLT (Kruskal–Wallis test: p = 0.0018), CRP (Mann–Whitney U: p < 0.0001), ESR (Mann–Whitney U: p = 0.0092), NLR (Kruskal–Wallis test: p < 0.0001), and other indicators were significantly increased compared with healthy children and children with common fever, RBC (Kruskal–Wallis test: p < 0.0001), and Hg (Kruskal–Wallis test: p < 0.0001) were significantly decreased in children with Kawasaki disease conversely. In the analysis of the Spearman correlation, it was found that serum PK2 concentration and NLR ratio were significantly negatively correlated in children with Kawasaki disease (rs = -0.2613, p = 0.0301). In the analysis of the ROC curves, it was found that the area under the PK2 curve was 0.782 (95% confidence interval 0.683–0.862; p < 0.0001), the ESR was 0.697 (95% confidence interval 0.582–0.796; p = 0.0120), the CRP was 0.601 (95% confidence interval 0.683–0.862; p = 0.1805), and the NLR was 0.735 (95% confidence interval 0.631–0.823; p = 0.0026). PK2 can significantly predict Kawasaki disease independently of CRP and ESR (p < 0.0001). The combined score of PK2 and ESR can significantly improve the diagnostic performance of PK2 (AUC = 0.827, 95% CI 0.724–0.903, p < 0.0001). The sensitivity was 87.50%, the sensitivity was 75.81%, the positive likelihood ratio was 6.0648, and the Youden index was 0.6331. PK2 has the potential to be a biomarker for early diagnosis of Kawasaki disease, and the combined use of ESR can further improve its diagnostic performance. Our study identifies PK2 as an important biomarker for Kawasaki disease and provides a potential new diagnostic strategy for Kawasaki disease.





Similar content being viewed by others
Data availability
Not applicable.
References
Tang B, Lo HH, Lei C, et al. Adjuvant herbal therapy for targeting susceptibility genes to Kawasaki disease: an overview of epidemiology, pathogenesis, diagnosis and pharmacological treatment of Kawasaki disease. Phytomedicine. 2020;70:153208. https://doi.org/10.1016/j.phymed.2020.153208.
Uehara R, Belay ED. Epidemiology of Kawasaki disease in Asia, Europe, and the United States. J Epidemiol. 2012;22(2):79–85. https://doi.org/10.2188/jea.je20110131.
Rife E, Gedalia A. Kawasaki disease: an update. Curr Rheumatol Rep. 2020;22(10):75. https://doi.org/10.1007/s11926-020-00941-4.
Makino N, Nakamura Y, Yashiro M, et al. Nationwide epidemiologic survey of Kawasaki disease in Japan, 2015–2016. Pediatr Int. 2019;61(4):397–403. https://doi.org/10.1111/ped.13809.
Candelino M, Tagi VM, Chiarelli F. Cardiovascular risk in children: a burden for future generations. Ital J Pediatr. 2022;48(1):57. https://doi.org/10.1186/s13052-022-01250-5.
Franco A, Shimizu C, Tremoulet AH, et al. Memory T-cells and characterization of peripheral T-cell clones in acute Kawasaki disease. Autoimmunity. 2010;43(4):317–24. https://doi.org/10.3109/08916930903405891.
Peng Q, Deng Y, Yang X, et al. Genetic variants of ADAM17 are implicated in the pathological process of Kawasaki disease and secondary coronary artery lesions via the TGF-beta/SMAD3 signaling pathway. Eur J Pediatr. 2016;175(5):705–13. https://doi.org/10.1007/s00431-016-2696-8.
Eleftheriou D, Levin M, Shingadia D, et al. Management of Kawasaki disease. Arch Dis Child. 2014;99(1):74–83. https://doi.org/10.1136/archdischild-2012-302841.
Shulman ST, Rowley AH. Kawasaki disease: insights into pathogenesis and approaches to treatment. Nat Rev Rheumatol. 2015;11(8):475–82. https://doi.org/10.1038/nrrheum.2015.54.
Kobayashi T, Ayusawa M, Suzuki H, et al. Revision of diagnostic guidelines for Kawasaki disease (6th revised edition). Pediatr Int. 2020;62(10):1135–8. https://doi.org/10.1111/ped.14326.
Messina AF, Namtu K, Guild M, et al. Trimethoprim-sulfamethoxazole therapy for children with acute osteomyelitis. Pediatr Infect Dis J. 2011;30(12):1019–21. https://doi.org/10.1097/INF.0b013e31822db658.
Ogata S, Tremoulet AH, Sato Y, et al. Coronary artery outcomes among children with Kawasaki disease in the United States and Japan. Int J Cardiol. 2013;168(4):3825–8. https://doi.org/10.1016/j.ijcard.2013.06.027.
Ng KL, Li JD, Cheng MY, et al. Dependence of olfactory bulb neurogenesis on prokineticin 2 signaling. Science. 2005;308(5730):1923–7. https://doi.org/10.1126/science.1112103.
Shojaei F, Wu X, Zhong C, et al. Bv8 regulates myeloid-cell-dependent tumour angiogenesis. Nature. 2007;450(7171):825–31. https://doi.org/10.1038/nature06348.
Monnier J, Samson M. Cytokine properties of prokineticins. FEBS J. 2008;275(16):4014–21. https://doi.org/10.1111/j.1742-4658.2008.06559.x.
Noda K, Dufner B, Ito H, et al. Differential inflammation-mediated function of prokineticin 2 in the synovial fibroblasts of patients with rheumatoid arthritis compared with osteoarthritis. Sci Rep. 2021;11(1):18399. https://doi.org/10.1038/s41598-021-97809-z.
Martucci C, Franchi S, Giannini E, et al. Bv8, the amphibian homologue of the mammalian prokineticins, induces a proinflammatory phenotype of mouse macrophages. Br J Pharmacol. 2006;147(2):225–34. https://doi.org/10.1038/sj.bjp.0706467.
LeCouter J, Zlot C, Tejada M, et al. Bv8 and endocrine gland-derived vascular endothelial growth factor stimulate hematopoiesis and hematopoietic cell mobilization. Proc Natl Acad Sci U S A. 2004;101(48):16813–8. https://doi.org/10.1073/pnas.0407697101.
Franchi S, Giannini E, Lattuada D, et al. The prokineticin receptor agonist Bv8 decreases IL-10 and IL-4 production in mice splenocytes by activating prokineticin receptor-1. BMC Immunol. 2008;9:60. https://doi.org/10.1186/1471-2172-9-60.
Abou-Hamdan M, Costanza M, Fontana E, et al. Critical role for prokineticin 2 in CNS autoimmunity. Neurol Neuroimmunol Neuroinflamm. 2015;2(3):e95. https://doi.org/10.1212/NXI.0000000000000095.
He X, Shen C, Lu Q, et al. Prokineticin 2 plays a pivotal role in psoriasis. EBioMedicine. 2016;13:248–61. https://doi.org/10.1016/j.ebiom.2016.10.022.
Wang H, Jia Y, Yu X, et al. Circulating Prokineticin 2 levels are increased in children with obesity and correlated with insulin resistance. Int J Endocrinol. 2021;2021:6630102. https://doi.org/10.1155/2021/6630102.
Yu X, Chen J, Tang H, et al. Identifying Prokineticin2 as a novel immunomodulatory factor in diagnosis and treatment of sepsis. Crit Care Med. 2022;50(4):674–84. https://doi.org/10.1097/CCM.0000000000005335.
Si F, Wu Y, Gao F, et al. Relationship between IL-27 and coronary arterial lesions in children with Kawasaki disease. Clin Exp Med. 2017;17(4):451–7. https://doi.org/10.1007/s10238-017-0451-8.
Su Y, Feng S, Luo L, et al. Association between IL-35 and coronary arterial lesions in children with Kawasaki disease. Clin Exp Med. 2019;19(1):87–92. https://doi.org/10.1007/s10238-018-0513-6.
McCrindle BW, Rowley AH, Newburger JW, et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a scientific statement for health professionals from the American heart association. Circulation. 2017;135(17):e927–99. https://doi.org/10.1161/CIR.0000000000000484.
Ito H, Noda K, Yoshida K, et al. Prokineticin 2 antagonist, PKRA7 suppresses arthritis in mice with collagen-induced arthritis. BMC Musculoskelet Disord. 2016;17:387. https://doi.org/10.1186/s12891-016-1243-0.
Tsang MS, Hou T, Chan BC, et al. Immunological roles of NLR in allergic diseases and its underlying mechanisms. Int J Mol Sci. 2021. https://doi.org/10.3390/ijms22041507.
Dragoescu AN, Padureanu V, Stanculescu AD, et al. Neutrophil to Lymphocyte Ratio (NLR)-A useful tool for the prognosis of sepsis in the ICU. Biomedicines. 2021. https://doi.org/10.3390/biomedicines10010075.
Liu J, Ao W, Zhou J, et al. The correlation between PLR-NLR and prognosis in acute myocardial infarction. Am J Transl Res. 2021;13(5):4892–9.
Nebigil CG. Prokineticin Is a new linker between obesity and cardiovascular diseases. Front Cardiovasc Med. 2017;4:20. https://doi.org/10.3389/fcvm.2017.00020.
Funding
This study was supported by Chongqing Municipal Health Commission under Grant Number: 2020MSXM050.
Author information
Authors and Affiliations
Contributions
We thank CW for clinical samples collection. ZS for the guide of statistical analysis. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
All authors have no actual or potential competing interests with other people or organizations to this work.
Ethical approval
The study protocol was approved by the Ethics Committee of Children’s Hospital of Chongqing Medicine University. No informed consent required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zeng, L., Wang, C., Song, Z. et al. Prokineticin 2 as a potential biomarker for the diagnosis of Kawasaki disease. Clin Exp Med 23, 3443–3451 (2023). https://doi.org/10.1007/s10238-023-01078-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10238-023-01078-1