Abstract
Understanding the forces driving population dynamics is critical for species conservation and population management. For migratory birds, factors that regulate population abundance could come from effects experienced on breeding areas, wintering grounds, or during migration. We compiled survey data for Pacific and Atlantic subspecies of dunlins (Calidris alpina pacifica and C. a. hudsonica) from range-wide Christmas bird counts (1975–2010), and investigated the influences on this population index of density-dependence, falcon numbers, a set of seasonal environmental conditions during breeding, migration and non-breeding periods, and large-scale meteorological measures. For both sub-species, numbers fluctuated irregularly, varying threefold over the survey period, with no long-term upward or downward trend. Based on Royama’s general model framework, the change in numbers between successive years for both sub-species was negatively affected by the total count in the previous year (i.e., negative density-dependence) and by the eastward component of storm movement during fall migration, with slower motion associated with higher population growth. The remaining environmental factors differed between the sub-species (snowmelt date on the Pacific, temperature on the Atlantic) or acted in opposite directions (soil moisture). The directional effects of each of these factors are consistent with the biology of dunlin, and together they explain 67.4 (72.9 %) of the variation in the rate of change of Pacific (Atlantic) dunlin annual counts. Falcon numbers were not predictive, despite a tenfold increase in abundance, suggesting compensatory mortality. This study highlights directions for future studies, and provides a model for the analysis of other migratory species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
One of the central topics in population ecology is understanding temporal changes in population abundance and how these are related to biotic (e.g., population density, predation, food/resource competition) and abiotic (e.g., climate, weather, and disturbance) factors. Such knowledge is essential to understanding the forces driving population dynamics, and is critical for species conservation and population management (Thomas 1990; Bjørnstad and Grenfell 2001; Fromentin and Powers 2005; Wiegand et al. 2005). There is a long history of debate over the relative roles of biotic and abiotic factors in regulating and limiting population abundance (Nicholson 1933; Andrewartha and Birch 1954; Hunter and Price 1998). Recent studies have suggested that both types of forces are important in shaping population dynamics (Dennis and Otten 2000; Fromentin et al. 2001; Turchin 2003; Xu and Boyce 2009).
Changes in population abundance can result from changes in population growth rate, which in turn is influenced by demographical rates (Ozgul et al. 2004). Variation in demographic rates, namely reproduction, recruitment, or survival, is often associated with variation in biotic and abiotic factors through density-dependent or density-independent processes (Tyler et al. 2008; Brown 2011). Negative feedback between demographic rates and population density may lead to density-dependent changes in population growth (Reed and Slade 2008). Among density-independent processes, local climate and environmental conditions can drive population fluctuations through seasonal variation in food availability and habitat conditions (Karell et al. 2009). In addition, broad climatic phenomena such as the Pacific Decadal Oscillation (PDO), the North Pacific Index (NPI), and the North Atlantic Oscillation (NAO), can have far-reaching impacts on ecological systems (Stenseth et al. 2002; Hallett et al. 2004). These sources of climate variation can work in concert with density-dependent processes to shape the trajectories of fluctuating populations (Goswami et al. 2011).
Although the joint effects of biotic and abiotic factors on population dynamics have been widely studied for fish and mammals (Dennis and Otten 2000; Fromentin et al. 2001), few studies have been done for migratory birds to integrate factors across their distribution and migration range (Sæther et al. 2002, 2006). Migratory birds have distinct breeding, wintering, and migration periods, and conditions encountered across the geographic range during the annual cycle may influence their abundance (Sutherland 1996a; Newton 2004, 2006). Demographic rates in one period may be influenced not only by conditions in the same period, but by conditions in earlier periods of geographical separation (Norris 2005; Harrison et al. 2011). For instance, events that cause individual variation in physiological condition during the non-breeding period can have substantial consequences on the timing and success of reproduction the following breeding period (Norris et al. 2004; Kennedy et al. 2008). Such effects may interact with density-dependent mechanisms at one or multiple periods of the annual cycle to influence population dynamics (Ratikainen et al. 2008). In this study, we choose two dunlin sub-species (Calidris alpina pacifica and C. a. hudsonia) as models to investigate the effects on population dynamics of density-dependence, predation danger, and seasonal environmental conditions across their distribution range.
Dunlins are small shorebirds of the family Scolopacidae. Five—nine ‘races’ or ‘sub-species’ are recognized, all breeding in Arctic or subarctic regions spread around the northern hemisphere. We consider the population dynamics of the two North American subspecies (Warnock and Gill 1996). The Pacific dunlin breeds in coastal western Alaska (Gabrielson and Lincoln 1959) and winters at coastal sites from southern British Columbia to California (MacLean and Holmes 1971; DeSante and Pyle 1986) and western Mexico. Fall migration, peaking in October, is transoceanic over the Gulf of Alaska and northeastern Pacific to southern British Columbia and south through central California (Gill et al. 2013; Warnock et al. 2013). Spring migration, however, appears to be largely coastal. The birds gain fuel at several major stopover sites along the Pacific coast (Senner et al. 1989; Warnock et al. 2004) before arriving on their breeding grounds in early May. The second sub-species is the Atlantic dunlin (C. a. hudsonia), which breeds in western and northern Hudson Bay and west of James Bay, Canada. This population spends the winter along the eastern seaboard of the United States and the Gulf of Mexico. Spring migration of this population starts in mid-March along the Gulf coast up to New England, passing through the Great Lakes region in May and arriving on its breeding grounds in late May/early June. A small portion of the population migrates through North Dakota and Manitoba in May. Fall migration is more easterly than spring migration. In fall, dunlins move along Atlantic coastal mudflats, peak in the Maritime Provinces in October, move down to New England in late October, and peak in New Jersey in December (Warnock and Gill 1996).
Major predators of dunlins are falcons, including the peregrine (Falco peregrinus) and merlin (F. columbarius) (Page and Whitacre 1975), which both are experiencing ongoing population recovery following the 1973 ban on dichlorodiphenyltrichloroethane (DDT) and other conservation measures (Cade et al. 1988). These large increases in predator populations are a plausible factor contributing to declining populations of small shorebirds that have been reported worldwide (Stroud et al. 2006) and in North America (Brown et al. 2001; Morrison et al. 2001; Bart et al. 2007). Population trends for eastern dunlin, based on counts on migration, are negative between 1974 and 2009 (Bart et al. 2007); no data are available for western populations. Since substantial direct mortality of dunlin and other small shorebirds to falcons and other raptors (e.g., Page and Whitacre 1975; Cresswell and Whitefield 1994; Dekker et al. 2012), and raptor induced changes in habitat usage have been well documented (e.g., Ydenberg et al. 2002; van den Hout et al. 2008), it is reasonable to consider the potential relationship between numbers of raptors and their prey (Cresswell 2011).
In this paper, we compile survey data for Pacific and Atlantic dunlins from Christmas bird counts (National Audubon Society 2010). We conduct time series analyses based on Royama’s general model framework (Royama 1992). Time series analyses of population dynamics has proven to be a useful approach in demonstrating the importance of density-dependent and density-independent processes in governing population dynamics (Berryman and Turchin 2001). We test the time-series for density dependence and investigate the effects on the annual changes in dunlin number of density dependence, predation danger, and climate and environmental conditions within the annual cycle. Our objective is to identify the driving forces of population dynamics of dunlin for the purpose of ecological understanding and potential conservation action.
Materials and methods
Population data
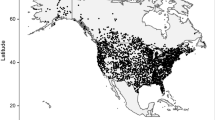
We compiled survey data for dunlins from the North American Christmas bird count (CBC, National Audubon Society 2010). CBC surveys are conducted annually between 14 December and 5 January, on ‘count circles’ with a 12-km radius circles. To provide broad and consistent sampling, we included in our compilation any circle (i) registering five or more dunlins in a single survey from 1975 to 2010, (ii) that had at least 30 years of counts, and (iii) that had no gaps between consecutive surveys >5 years. This filter captured 93 circles for Pacific dunlin and 151 circles for Atlantic dunlin, which cover most of their winter ranges and, we believe, the majority of both populations (Fig. 1). The most significant gaps are underrepresentation of inland sites in the Pacific region, particularly the Willamette and Central Valleys, and a lack of sites from Baja California, Mexico. We also compiled the counts of peregrine falcons and merlins, the most important avian predators, from each selected CBC circle over the same years.
Our compilation included 8784 site-year combinations, 3348 (=36 years × 93 sites) for Pacific dunlin and 5436 (=36 years × 151 sites) for Atlantic dunlin. Of these, 6.80 % (228) surveys were missing for Pacific dunlin and 8.20 % (446) for Atlantic dunlins. We imputed missing surveys using population marginal means (Searle et al. 1980). The percentage of the total number of dunlin imputed was 3.40 % for Pacific dunlin and 1.50 % for Atlantic dunlin. Total annual counts are sums over all sites per population.
Dunlins would seem to be a good species to count using CBC methodology. They occur in a specific habitat, with most individuals in large flocks whose size can be reasonably estimated by observers with some experience. The tally reached for a specific survey would thus seem to be largely independent of survey effort, at least above some minimum. Falcons, on the other hand, occur in low numbers, are highly dispersed, and range widely. The tally reached for a specific survey would thus seem to depend strongly on the survey effort. We followed standard practise and scaled each survey for both dunlins and falcons by the effort invested (measured as ‘party hours’). We found that scaling made only very minor differences to the results we report here, which are based on the scaled data.
Nonlinear relationships between survey effort and the number of birds counted may create bias in analyses based on CBC data, as has been reported for some species (Preston 1979; Johnson 1981; but see McCrimmon et al. 1997). Using the methods of Butcher and McCulloch (1990), we found no evidence for nonlinearity between counts and survey effort for either dunlins or falcons on either Pacific or Atlantic coasts (all P values >0.45).
Environmental and climate data
Environmental factors such as snowmelt, soil moisture, air temperature, and precipitation may affect the annual changes in dunlin number through influences on the availability of breeding or wintering habitats and food resources (Niehaus and Ydenberg 2006; Verhulst et al. 2007; Clark 2009). Storms have been reported as an important factor that causes mortality of migrating birds (Newton 2007). Here we consider snowmelt date (day of year) on the breeding grounds; storm motion (m/s) (eastward & southward) during fall migration; overwinter precipitation (mm), soil moisture (mm), and surface air temperature (°C) on the wintering grounds; and storm motion (m/s) (eastward & southward), soil moisture (mm), and precipitation (mm) at major stopover sites during spring migration. The geographical regions and times of year (see “Introduction” section) relevant to each phase of the annual cycle were derived from Warnock and Gill (1996) and later literature cited above. Time-series data for these variables were derived from modeled climate data extracted from the gridded climate data sets available at Physical Sciences Division (PSD), Earth System Research Laboratory, NOAA (http://www.esrl.noaa.gov/psd/cgi-bin/db_search/SearchMenus.pl), with a spatial resolution of 0.10°–2.50° latitude and longitude, depending on variables. The data extracted were monthly averages except for snow depth. Snow depth was a daily average, which we used to derive snowmelt date, calculated as the last day when snow depth was greater than 2.50 cm. The storm motion index was only available starting in 1979, thus our analyses of relationships between changes in dunlin counts and environmental variables start in that year. For the analysis period, we imputed values for 4.30 % (144) missing surveys for Pacific dunlin and 5.60 % (304) for Atlantic dunlins, representing 2.80 % of the total Pacific dunlin and 0.50 % of Atlantic dunlin counts.
For each of the variables, we used the open-source R software (R Core Team 2012) to access, organize, and extract time-series data from the original NetCDF (.nc) files downloaded from PSD, NOAA. Spatial data are highly correlated. In our analysis, the time-series data we used for each of the variables were centrally located within the region of interest, unless otherwise specified. Using averages over the region did not qualitatively change the results. As dunlins are widely distributed along the Pacific and Atlantic coast in winter, time-series data of the overwinter variables were averaged over the major CBC sites where they are concentrated. Because winter conditions can be more severe in the northeast, for Atlantic dunlin, we also specifically examined whether the colder weather in the coastal northeastern US had any effect on annual counts.
Besides the above environmental variables, we also tested whether large scale climate pattern might predict the year-to-year changes in number of dunlin. Specifically, we looked into annual indices of the PDO (http://jisao.washington.edu/pdo/PDO.latest) and NPI (Trenberth and Hurrell 1994; Hurrell and NCAR staff 2012) for Pacific dunlin, and NAO (Hurrell 2003; Hurrell and NCAR staff 2013) for Atlantic dunlin.
Data analysis
The year-to-year changes in number of dunlin can be modelled according to Royama’s general model framework (Royama 1992) in the following manner. Assume \(N_{t} ,\) \(P_{t} ,\) and \(Z_{t}\) is the number of dunlin, the number of predators, and environmental conditions at year \(t\), respectively, let \(X_{t} = { \log }_{e}({{N_{t} }})\), then the rate of change in number of dunlin \(r_{t} = X_{t} - X_{t - 1}\) can be written in the form:
where \(f\left( \cdot \right)\) is a function of \(X_{t - 1} , P_{t - 1} , Z_{t - 1}\), and \(\varepsilon_{t}\) is other density-independent random effects beyond \(Z_{t - 1}\) or could be considered as an error term.
We were interested in whether density-dependence, predators, and multiple environmental variables had any additive or interactive effects on \(r_{t}\), so model (1) could be written in the following general form
where \(\alpha\), \(\beta\), \(\gamma\), \(\theta_{1}\), \(\varphi_{1i}\), \(\varphi_{2i}\), \(\omega_{ij}\), and \(\delta_{i}\) \(\left( {i = 1, \ldots , n;\;i \ne j} \right)\) are model parameters, which can be estimated through multiple regression analysis.
We investigated the effects of density-dependence, predators, and environmental variables on \(r_{t}\) by developing a set of candidate models of the general form (2). To limit the number of the candidate models, we used correlation tests to facilitate the process. Correlations between environmental variables were checked to avoid multicollinearity. We tested for correlation between \(r_{t}\) and environmental variables and incorporated only those variables as covariates when the correlation was significant at P < 0.15. Our analysis (not shown here) indicated that incorporating the environmental variables when the correlation was not significant did not improve model fit, nor were these variable terms significant in the regression models.
Correlation tests and standardized regression analysis were conducted using the R software (R Core Team 2012). Model selection was based on Akaike’s information criterion adjusted for small sample size (AICc, Hurvitch and Tsai 1989; Burnham and Anderson 1998) and Bayesian information criterion (BIC, Schwarz 1978). AICc and BIC are both penalized-likelihood criteria. BIC penalizes model complexity more heavily than AICc. The AICc for the model of interest minus the smallest AICc for the set of models being considered is denoted by ΔAICc. By definition, the most-supported model has a ΔAICc of zero, which gives the most parsimonious description of the data. In general, ΔAICc < 2 suggests no strong evidence for a difference between candidate models, and models with ΔAICc < 2 are generally considered worthy of consideration. In model selection, we included interactive terms in models only when they were significant.
Besides model selection, time-series of dunlin data were also independently tested for density-dependence using a bootstrap test (Dennis and Taper 1994). This test is based on a randomization procedure that reduces the Type I error. Here, the bootstrap test was used to test null hypothesis β = 0 in equation:
against alternative hypothesis β < 0. We present a plot of this regression model along with the data points (\(X_{t - 1} ,r_{t}\)).
To assess how much of the variation in the rate of change in number of dunlin was explained by each covariate in the top model of form (2) from model selection, we calculated their contribution to the model’s coefficient of multiple determination (Sokal and Rohlf 1981; Vucetich and Peterson 2004). The contribution of each covariate x (denoted \(R_{x}^{2}\)) was calculated as its standardized partial regression coefficient multiplied by the correlation coefficient between x and the response variable (Schumacker and Lomax 1996; Previtali et al. 2009). As storm motion data are available from 1979, data analysis was conducted using data from 1979 to 2010.
Measurement error and model evaluation
Measurement error may influence the reliability in detecting density-dependence in ecological time series, resulting in false detection of density dependence that would otherwise not exist (Shenk et al. 1998; Freckleton et al. 2006; Kölzsch et al. 2007). The bootstrap method that we used to test for density dependence is robust against measurement error as suggested by Dennis and Taper (1994). We also ran a simulation suggested by Freckleton et al. (2006) to quantify the effects of possible count data error on our results. First, we fitted a density-independent model (i.e., the top model from model selection with the density-dependent term dropped) to our data obtaining a density-independent time series. Then we simulated 1000 data series by adding random errors to the density-independent time series. We examined three levels of random perturbation, which are ±10, ±20, and ±30 % of the density-independent time series, to address the uncertainty in possible CBC error, although we have used a number of methods to minimize the potential bias in counts. P values from bootstrap test, AICc and BIC values were calculated for each data series, and we examined the probability that measurement errors could generate the levels of density-dependence observed in our data.
Linear regressions assume that the error term \(\varepsilon_{t}\) in Eq. 2 is independent and normally distributed. To account for the appropriateness of the general model assumptions, we made residuals plot to examine if the distribution of the model residuals was random, and used Shapiro–Wilk normality test to check whether model residuals fit a normal distribution.
Results
Annual variability in dunlin and falcon numbers
There were substantial year-to-year changes in the number of dunlins counted between 1975 and 2010. The total count for Pacific dunlin varied more than threefold among years, ranging between 70,000 and 270,000 in three strong cycles of about 10 years each (Fig. 2a). The number of Atlantic dunlin also varied threefold among years (40,000–120,000; Fig. 2a), though the total counts are smaller. The rate of change in number of dunlins among years (r t ) varied from −0.64 to 0.52 for Pacific dunlin and from −0.47 to 0.58 for Atlantic dunlin (Fig. 2b, c). There was no long-term trend in dunlin counts in either region (least-squares linear regressions: Pacific: R 2 = 0.03, year = −713.90 ± 0.71 SE, 1 df, F = 1.02, P = 0.32; Atlantic: R 2 = 0.02, year = +240.70 ± 0.29 SE, 1 df, F = 0.70, P = 0.41).
The annual counts of Pacific (Calidris alpina pacifica) and Atlantic dunlins (C. a. hudsonia) (a) and the annual counts of falcon (Falco peregrinus and F. columbarius) on the Pacific and Atlantic coasts (b) between 1975 and 2010, based on Christmas bird counts. Also shown are the rates of change in the number of Pacific (c) and Atlantic dunlins (d) r t , from 1976 to 2010. Counts were adjusted by survey effort
In strong contrast to dunlins, the counts of falcons increased steadily in both regions (Fig. 2d) (least-squares linear regressions: Pacific: R 2 = 0.96, year = +20.90 ± 0.70 SE, 1 df, F = 927.00, P < 0.0001; Atlantic: R 2 = 0.93, year = +11.10 ± 0.60 SE, 1 df, F = 463.90, P < 0.0001), resulting in sevenfold increase from 1975 to 2010 in the west, and a threefold increase in the east.
Annual variability in environmental variables
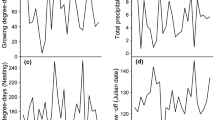
The values of environmental variables vary substantially from year to year in both regions (Fig. 3). Snowmelt date varied between 134 and 181 days in Alaska, and from 170 to 195 days in Arctic Canada (on average, 15 days later than in Alaska) (Fig. 3a). Eastward storm motion during fall migration varied from 8.35 to 15.57 m/s over the Gulf of Alaska and from 4.80 to 14.43 m/s over the Maritime Provinces (on average, 1.68 m/s weaker than over the Gulf of Alaska) (Fig. 3b). Overwinter soil moisture on the Pacific coast varied from 387.32 to 663.14 mm; soil moisture in the Great Lakes region during spring migration of Atlantic dunlin varied from 182.92 to 516.31 mm, which, on average was 195 mm less than on the Pacific coast (Fig. 3c). Overwinter temperature on the Pacific coast varied from 6.16 to 10.31 °C; early winter temperature in the coastal northeastern US varied from −4.02 to 6.43 °C (about 7 °C lower than on the Pacific coast) (Fig. 3d).
Time series plots (1975–2010) of environmental variables a snowmelt date (days) on breeding grounds; b eastward storm motion (m/s) during fall migration; c soil moisture (mm) (overwinter averages on the Pacific coast or monthly averages over the Great Lakes region during spring migration); d winter temperature (°C) (overwinter averages on the Pacific coast or December averages in the coastal northeast US)
Density dependence
The AICc (and BIC) for the density-dependent models were distinctly lower than for the density-independent models [ΔAICc (ΔBIC) = −8.51 (−7.36) for Pacific dunlin, and −6.26 (−5.12) for Atlantic dunlin], supporting the presence of strong density-dependence in both populations. Bootstrap test results were significant for both populations (Ps < 0.05, Fig. 4a, b), suggesting density-dependence was negative. This is further illustrated by the regression plots (Fig. 4c) and the negative estimates of the slope parameter \(\beta\) in Eq. 3 (\(\widehat{\beta } = - 0. 5 4 \pm 0. 1 6\) for Pacific dunlin; −0.48 ± 0.16 for Atlantic dunlin).
Bootstrap test for density dependence in time-series data of a Pacific dunlin (n = 10,000, P = 0.05) and b Atlantic dunlin (n = 10,000, P = 0.02). Histograms are the frequency distributions of test statistic under null hypothesis (β = 0 in Eq. 3; alternative is β < 0), and the black dots are values of test statistic calculated from the dunlin data. A plot of r t versus X t−1 = log e (N t−1) is shown in c. The black circles represent yearly data points for Pacific dunlin and white circles for Atlantic dunlin. The straight lines are the regression models (Pacific dunlin, r t = 6.46 − 0.54 × X t−1), R 2 = 0.29, P = 0.00; Atlantic dunlin, r t = 5.33 − 0.48 × X t−1), R 2 = 0.24, P = 0.01) of Eq. 3
Correlation between r t and environmental variables
A number of factors within the annual cycle were correlated with r t , but the number of predators had no correlation with r t in either region (correlation coefficients <0.02, Ps > 0.90).
For Pacific dunlin, snowmelt date in Alaska, eastward storm motion over Gulf of Alaska during fall migration, overwinter soil moisture, and eastward storm motion around San Francisco Bay during spring migration all had a significant negative correlations with r t (Ps < 0.05) (Table 1). The negative correlation between r t and overwinter precipitation, soil moisture around San Francisco Bay during spring migration, or NPI was relatively weak (Ps < 0.15) (Table 1). Among these environmental variables, overwinter soil moisture, precipitation, and soil moisture around San Francisco Bay during spring migration were highly correlated (correlation coefficients >0.70, Ps < 0.001) (Table 2).
For Atlantic dunlin, eastward storm motion in Maritime Provinces during fall migration, early winter air temperature and soil moisture in New Jersey, and soil moisture in May in Great Lakes region had a significant correlations with r t (Ps varied from 0.003 to 0.12 <0.15) (Table 3). None of these variables were highly intercorrelated (absolute values of correlation coefficients <0.34, Ps > 0.05) (Table 4).
Top models for dunlin populations
The top model for Pacific dunlin consisted of density-dependence, snowmelt date in Alaska, the rate of eastward storm motion over Gulf of Alaska during fall migration, and overwinter soil moisture (Table 5). As with Pacific dunlin, the top model for Atlantic dunlin included density-dependence and eastward storm motion during fall migration. In addition, the model also included early winter air temperature in New Jersey, soil moisture in the Great Lakes region during spring migration and interaction terms between density-dependence and soil moisture and between temperature and soil moisture (Table 6). The rest of the candidate models ΔAICc’s and ΔBIC’s were all >2, thus we only considered the top models for further description.
In the top model of Pacific dunlin, parameter estimates were significantly negative for density-dependence, storm motion, overwinter soil moisture, and marginally significant for snowmelt date (Table 7). This suggested that Pacific dunlin counts between successive years were negatively affected by the tally in the previous year (negative density-dependence), by snowmelt date in Alaska in the previous spring (earlier snowmelt associated with higher population growth), by the eastward component of fall storm movement over the Gulf of Alaska during fall migration (slower motion associated with higher population growth), and by overwinter soil moisture on Pacific coast (lower level of soil moisture associated with higher population growth). Among these factors, density-dependence had the highest contribution to the variation in the rate of change in Pacific dunlin number (\(R_{DD}^{2} = 2 9. 90 \;\%\)), followed by eastward storm motion (\(R_{Ustm10}^{2} = 1 6. 10 \;\%\)), snowmelt date (\(R_{Snowm}^{2} = 1 1. 10\;\%\), and overwinter soil moisture (\(R_{soilM}^{2} = 10. 30\;\%\)). Together these factors captured many features of the population trajectory 1979–2010 (Fig. 5a), and explained 67.40 % of the variation in the rate of change of Pacific dunlin counts (Table 5).
In the top model of Atlantic dunlin, parameter estimates were significantly negative for density-dependence and storm motion and significantly positive for early winter temperature and soil moisture during migration (Table 8). This suggested that Atlantic dunlins, as with Pacific dunlins, were also negatively affected by density-dependence and by the eastward component of fall storm movement during fall migration. In contrast to Pacific dunlin, arctic snowmelt date did not have significant effects on Atlantic dunlin; soil moisture, which was in Great Lakes region during spring migration, positively affected Atlantic dunlin (i.e., higher level of soil moisture associated with higher population growth) (Table 8). In addition, Atlantic dunlins were also positively affected by early winter air temperature in New Jersey and by two interactions (between density-dependence and soil moisture during spring migration, and between early winter air temperature and soil moisture during spring migration) (Table 8). As in Pacific dunlin, density-dependence had the highest contribution to the variation in the rate of change (\(R_{DD}^{2} = 3 2. 40 \;\%\)), followed by eastward storm motion (\(R_{Ustm10}^{2} = 1 5. 50 \;\%\)), early winter air temperature (\(R_{Temp12}^{2} = 1 4. 10 \;\%\)), soil moisture during spring migration (\(R_{SoilM5.GL}^{2} = 1 1 \;\%\)). Together these factors explained 72.90 % of the variation in the rate of change of Atlantic dunlin counts (Table 6, Fig. 5b).
Measurement error and model evaluation
Our analysis using simulated data series with various levels of census errors suggested that measurement error is unlikely to account for the density-dependence. Only 1.60–2.90 % of the 1000 simulated error-perturbed data series showed bootstrap P values smaller than 0.05 for low to high levels of measurement error. Similarly, of the simulated BIC values from low to high levels of measurement error, only 1.50–3.50 % showed indications of spurious density-dependence [BIC(density-dependence) < BIC(density-independence)]; of the simulated AICc values 2.10–4.30 % [AICc(density-dependence) < AICc(density-independence)].
The residuals from the top models are random, almost uniformly scattered along the axis of fitted values (Fig. 6), and did not significantly deviate from a normal distribution (Shapiro–Wilk tests: W = 0.97, P = 0.58 for Pacific dunlin; W = 0.97, P = 0.54 for Atlantic dunlin).
Discussion
Our 36 year compilation of range-wide winter censuses is one of the first for any shorebird, and among only a very few for any long distance migrant. Our data reveal several previously-unknown features, including strong year-to-year fluctuations of up to 50 % or more around long term means of about 170,000 (Pacific) and 80,000 (Atlantic), which do not run in parallel in the two sub-species. In contrast to repeated assertions in the literature that have assigned North American dunlin populations ‘declining’ status (Donaldson et al. 2000; Bart et al. 2007), these data reveal no long-term down- or upward trend in either sub-species. Since our counts cover most of the migratory range, we doubt that these changes generally reflect spurious correlated annual redistribution of wintering birds (e.g., Bart et al. 2007), and consider explanations relating to real population change.
For both Pacific and Atlantic sub-species, the fluctuations in total regional winter counts of dunlin were irregular, and could be accounted for by the combined effects of negative density-dependence and a set of seasonal environmental conditions during breeding, migration and non-breeding periods. As measured by the proportion of variation explained, density-dependence acted with similar magnitude on both sub-species, as did eastward storm motion during southward migration. The remaining environmental factors differed between the sub-species (snowmelt date on the Pacific, temperature on the Atlantic) or acted in opposite directions (soil moisture). Models incorporating these features outcompeted other models (as assessed by AICc) in their ability to replicate the dynamics of both populations. Finally, in both populations the number of falcons recorded in the same CBC count circles rose steadily throughout the 36-year period, but in neither population did the number of falcons enter any of the explanatory models. Novel among these findings are the apparently strong role of storm motion during migration, the apparent lack of any effect due to predators, and the additive effects of environmental factors from all parts of the annual cycle.
The applicability of tests of density dependence to ecological time series in the presence of measurement error, have recently been debated (Shenk et al. 1998; Freckleton et al. 2006; Kölzsch et al. 2007). Studies suggest that measurement error could contribute to a large amount of bias in the estimation of density dependence (Dennis et al. 2006; Knape 2008; Knape and de Valpine 2012; Hosack et al. 2012). The approach we used to test for density dependence is the bootstrapping method of Dennis and Taper (1994), which has been suggested to be robust against measurement error (Dennis and Taper 1994). By adding random errors to density-independent time series our simulation on effects of measurement error further indicated that measurement error alone could not account for the density-dependence in dunlin counts.
Figure 4 shows that the magnitude of density-dependence (i.e., the rate of change in year t in relation to the population size in year t—the slope of the lines) is almost identical in the two sub-species, though the Pacific sub-species is about twice as numerous as is the Atlantic sub-species. Density-dependence could operate by influencing survival (Goss-Custard et al. 1995) through a variety of mechanisms, including intraspecific competition for food resources (Goss-Custard 1980), habitat heterogeneity (Gill et al. 2001; Sutherland and Norris 2002), interference among individuals at high densities (Fernandez et al. 1998; Newton 1998), or altered mortality risk due to predation danger (Cresswell 2011). Fecundity could also be affected (Török and Tóth 1988; Dhondt et al. 1992). Holmes (1966) claimed that dunlin breeding habitats are limited by snow cover and food availability, so at high population densities poorer quality territories may be used disproportionately, thus reducing breeding success (Sutherland 1996b; Rodenhouse et al. 1997; Balbontín and Ferrer 2008) and shrinking population growth (Soutullo et al. 2006). The data we present here are based on annual counts made at mid-winter, and we thus have no way to evaluate which if any of these potential mechanisms is involved. Measures of survival over parts of the annual cycle would help enormously.
In each sub-species, this basic density-dependence is augmented or attenuated by a set of environmental factors. Storm motion during fall migration appears to be a major factor influencing annual changes in dunlin counts, and accounts for 16.10 and 15.50 % of the variation in the annual abundance of Pacific and Atlantic sub-species, respectively. Faster storm motion may decrease migrant’s probabilities of safely completing migration. Storm kills of migrating birds have been recorded for many species (Newton 2006, 2007), including North American songbirds (Butler 2000), the chimney swift (Dionne et al. 2008), ducks (Roth 1976), and warblers (Case et al. 1965). Fall migration of Pacific dunlin is transoceanic (Gill et al. 2013), but this is not true of Atlantic dunlins and so the mortality agent would not seem to involve a long over-ocean flight. Heavy rain associated with strong storms may wet the plumage, increase wing loading and reduce body heat. These stresses, if coupled with disorientation, could cause migrants be killed by drowning or chilling (Frazar 1881; Saunders 1907; Newton 2007). Flying birds could also be killed by electrocution in lightning storms (Glasrud 1976). Storms or more general conditions during southward migration may also cause indirect mortality, by affecting the condition or slowing migratory progress, increasing the risks of starvation or predation, either during the migration itself or in the subsequent nonbreeding period (Dionne et al. 2008). These potential mechanisms require further investigation.
Snowmelt date, soil moisture, and winter temperature affected Pacific dunlin differently from Atlantic dunlin. Early snowmelt, which was associated with high population growth rates of Pacific dunlin, had no significant effects on Atlantic dunlin. This may be due to the different timing of two populations arriving on the breeding grounds. Pacific dunlin arrive on the breeding ground in late April and early May, whereas the arrival of Atlantic dunlin is later by a month (Warnock and Gill 1996). Snowmelt timing is highly variable in Alaska (e.g., Niehaus and Ydenberg 2006), perhaps more so than in the eastern Arctic. Late snow limits the availability of nest sites and food resources of migrant birds and consequently delays breeding (Young et al. 1995; Babcock et al. 2002; Niehaus and Ydenberg 2006), but early snowmelt years lengthens the breeding period and increases the amount of reproduction for Pacific dunlins (Jamieson 2009). This would seem to explain the influence of snowmelt timing on population change in this sub-species.
Soil moisture affected the two dunlin counts in opposite ways, and at different stages of the annual cycle. Pacific dunlin population growth is negatively affected by overwinter soil moisture and Atlantic dunlin is positively affected by soil moisture during spring migration. Positive effects of soil moisture on shorebird populations have been documented in a number of studies (Green 1988; Beintema et al. 1997; Milsom et al. 2000; Verhulst et al. 2007). High soil moisture, which generally reduces the soil penetration resistance, is known to be beneficial to soil-probing birds such as dunlins by increasing the accessibility to food resources (Green 1988). In contrast, the negative effects of soil moisture on Pacific dunlin growth rate is likely a sampling artefact related to underrepresentation of interior sites in our dataset. In wet years, dunlin move from the coast to interior sites in the Central Valley of California and elsewhere (Warnock et al. 1995), thus the numbers on California coast negatively correlated with levels of fall and winter rainfall. However, a direct effect on survivorship is also possible; studies of winter energetics of wild and captive dunlin with respect to rainfall show changes in activity budgets and fat storage strategies that suggest negative fitness consequences under rainier regimes (Kelly and Weathers 2002; Kelly et al. 2002).
Winter temperature had no effect on Pacific dunlin population growth, but low temperatures suppress the population growth of Atlantic dunlins, presumably because the weather on the northeast coast of US is more severe in winter than on the west coast. The average temperature (1979–2010) in December in New Jersey was around 2 °C, roughly 7 °C lower than on the west coast. Low winter temperature that reduces the availability of food resources could lead to starvation, poor body conditions, and a consequent increased mortality of wintering dunlins (Davidson and Evans 1982; Clark 2004, 2009).
A notable feature of the situation examined here is that falcon numbers rise steadily throughout our 36 year record, increasing about sevenfold in the Pacific and threefold in the Atlantic region (Fig. 2). It is well-documented that peregrines and merlins have increased strongly in North America during the past 30 years, and they have substantial distributional overlap with wintering dunlins (White et al. 2002). Several reports verify that falcons kill significant fractions of the dunlin wintering at certain sites (Page and Whitacre 1975; Dekker et al. 2012). For example, Dekker et al. (2012) estimate that 16 % of the approximately 30,000 dunlin wintering at Boundary Bay, British Columbia, are killed by peregrines in the course of an average winter. However, this systematic increase in avian predator counts (peregrine falcon and merlin) has no detectable effect in our models on the year-to-year changes in dunlin numbers. Though initially surprising, this is consistent with other studies in which changes in predator numbers had little numerical effect on bird populations (Newton 1998; Holt et al. 2008). The lack of effect could arise from any of several mechanisms, including complete avoidance of predators by prey, compensatory predation (Errington 1945), or by predators switching to alternative prey as dunlin become less abundant (Cresswell 2011).
It seems implausible that wintering dunlins would be able to avoid falcons completely. Quite aside from the reports documenting high depredation by falcons of dunlins during winter (see above), the data reported here show that the majority of the CBC count circles we analyzed were occupied by both dunlins and falcons. A slightly more tenable possibility is that falcons switch to feeding on other prey species as dunlins decline in abundance. Peregrines and merlins both feed on a wide range of species, including pigeons and doves, waterfowl, songbirds, and shorebirds (e.g., Castellanos et al. 2006). We have no way to evaluate this hypothesis with our data.
In our view the most likely explanation for the lack of a population effect on dunlins of increased predator abundance is that the mortality due to falcons is compensatory, in that the prey killed by predators during winter would have died due to other agents of mortality (Perrins and Geer 1980; Newton 1998). A variety of mechanisms are possible, but we feel that the evidence currently available is most consistent with the hypothesis that the wintering sites occupied by dunlins are sufficiently crowded such that the removal of dunlins by predators lessens the intensity of competition for the resources necessary to avoid starvation. Predation danger has substantial effects on the behavior of migrating and wintering shorebirds, including selection of less food rich habitats by vulnerable birds (Ydenberg et al. 2002; Taylor et al. 2007; Pomeroy et al. 2008), and shorter lengths of stay at stopover sites (Ydenberg et al. 2004). These effects have intensified as falcon populations have increased (Ydenberg et al. 2004), including a well-documented case for wintering dunlins, who reduced their midwinter fat stores and took up energetically expensive over-ocean flight in place of roosting during high tides (Ydenberg et al. 2010). Apparently, these birds have reduced their investment in defense against starvation (lowered fat reserves) to gain defense against predators, which is entirely consistent with the increasing danger posed by falcons. However, without further information concerning the relationship between population density and demographical rates and the interactions between predators and this relationship, we are unsure exactly how compensatory mortality operates.
References
Andrewartha HG, Birch LC (1954) The distribution and abundance of animals. University of Chicago Press, Chicago
Babcock CA, Fowler AC, Ely CR (2002) Nesting ecology of tundra swans on the coastal Yukon-Kuskokwim Delta, Alaska. Waterbirds 25(1):236–240
Balbontín J, Ferrer M (2008) Density-dependence by habitat heterogeneity: individual quality versus territory quality. Oikos 117:1111–1114
Bart J, Brown S, Harrington B, Morrison RIG (2007) Survey trends of North American shorebirds: population declines or shifting distributions? J Avian Biol 38:73–82
Beintema AJ, Dunn E, Stroud DA (1997) Birds and wet grasslands. In: Pain DJ, Pienkowski MW (eds) Farming and birds in Europe. Academic Press, San Diego, pp 269–296
Berryman A, Turchin P (2001) Identifying the density-dependent structure underlying ecological time series. Oikos 92:265–270
Bjørnstad ON, Grenfell BT (2001) Noisy clockwork: time series analysis of population fluctuations in animals. Science 293:638–643
Brown GS (2011) Patterns and causes of demographic variation in a harvested moose population: evidence for the effects of climate and density-dependent drivers. J Anim Ecol 80:1288–1298
Brown S, Hickey C, Harrington B, Gill R (2001) The United States shorebird conservation plan, 2nd edn. Manomet Center for Conservation Studies, Manomet
Burnham KP, Anderson DR (1998) Model selection and inference: a practical information-theoretic approach. Springer, New York
Butcher GS, McCulloch CE (1990) Influence of observer effort on the number of individual birds recorded on Christmas bird counts. Biol Rep 90:120–129
Butler RW (2000) Stormy seas for some North American songbirds: are declines related to severe storms during migration? Auk 117:518–522
Cade TJ, Enderson JH, Thelander CG, White CM (1988) Peregrine falcon populations: their management and recovery. Peregrine Fund Inc., Boise
Case LD, Cruickshank H, Ellis AE, White WF (1965) Weather causes heavy bird mortality. Fla Nat 38:29–30
Castellanos A, Argüelles C, Salinas F, RodrÍguez A, Ortega-Rubio A (2006) Diet of breeding peregrine falcon at a coastal lagoon, Baja California Sur, Mexico. J Raptor Res 40:241–244
Clark JA (2004) Ringing recoveries confirm higher wader mortality in severe winters. Ringing Migr 22:43–50
Clark JA (2009) Selective mortality of waders during severe weather. Bird Study 56:96–102
Cresswell W (2011) Predation in bird population. J Ornithol 152(1):S251–S263
Cresswell W, Whitefield DP (1994) The effects of raptor predation on wintering wader populations at the Tyninghame estuary, Southeast Scotland. Ibis 136:223–232
Davidson NC, Evans PR (1982) Mortality of Redshanks and Oystercatchers from starvation during severe weather. Bird Study 29:183–188
Dekker D, Out M, Tabak M, Ydenberg R (2012) The effect of kleptoparasitic bald eagles and gyrfalcons on the kill rate of peregrine falcons hunting dunlins wintering in British Columbia. Condor 114:290–294
Dennis B, Otten MRM (2000) Joint effects of density dependence and rainfall on abundance of San Joaquin kit fox. J Wildl Manage 64:388–400
Dennis B, Taper ML (1994) Density dependence in time series observations of natural populations: estimation and testing. Ecol Monogr 64:205–224
Dennis B, Ponciano JM, Lele SR, Taper ML, Staples DF (2006) Estimating density dependence, process noise and observation error. Ecol Monogr 76:323–341
DeSante D, Pyle P (1986) Distributional checklist of North American birds. Artemesia Press, Lee Vining
Dhondt AA, Kempenaers B, Adriaensen F (1992) Density-dependent clutch size caused by habitat heterogeneity. J Anim Ecol 61:643–648
Dionne M, Maurice C, Gauthier J, Shaffer F (2008) Impact of hurricane Wilma on migrating birds: the case of the chimney swift. Wilson J Ornithol 120:784–792
Donaldson G, Hyslop C, Morrison G, Dickson L, Davidson I (2000) Canadian shorebirds conservation plan. Authority of the Minister of Environment, Canadian Wildlife Service, Environment Canada, Ottawa
Errington PL (1945) Some contributions of a fifteen-year study of the northern bobwhite to a knowledge of population phenomena. Ecol Monogr 15:1–34
Fernandez C, Azkona P, Donazar JA (1998) Density-dependent effects on productivity in the Griffon vulture Gyps fulvus: the role of interference and habitat heterogeneity. Ibis 140:64–69
Frazar AM (1881) Destruction of birds by a storm while migrating. Bull Nuttall Orn Club 6:250–252
Freckleton RP, Watkinson AR, Green RE, Sutherland WJ (2006) Census error and the detection of density dependence. J Anim Ecol 75:837–851
Fromentin JM, Powers JE (2005) Atlantic bluefin tuna: population dynamics, ecology, fisheries and management. Fish Fish 6:281–306
Fromentin JM, Myers RA, Bjornstad ON, Stenseth NC, Gjosaeter J, Christie H (2001) Effects of density dependent and stochastic processes on the regulation of cod populations. Ecology 82:567–579
Gabrielson IN, Lincoln FC (1959) The birds of Alaska. The Stackpole Company, Harrisburg
Gill JA, Norris K, Potts PM, Gunnarsson TG, Atkinson PW, Sutherland WJ (2001) The buffer effect and large-scale population regulation in migratory birds. Nature 412:436–438
Gill RE Jr, Handel CM, Ruthrauff DR (2013) Intercontinental migratory connectivity and population structuring of dunlins from western Alaska. Condor 115:525–534
Glasrud RD (1976) Canada Geese killed during lightning storm. Can Field Nat 90:503
Goss-Custard JD (1980) Competition for food and interference among waders. Ardea 68:31–52
Goss-Custard JD, Clarke RT, Dit Durell SEA, Le V, Caldow RWG, Ens BJ (1995) Population consequences of winter habitat loss in migratory shorebird. II. Model predictions. J Appl Ecol 32:337–351
Goswami VR, Getz LL, Hostetler JA, Ozgul A, Oli MK (2011) Synergistic influences of phase, density, and climatic variation on the dynamics of fluctuating populations. Ecology 92:1680–1690
Green RE (1988) Effects of environmental factors on the timing and success of breeding of common snipe Gallinago gallinago (Aves: Scolopacidae). J Appl Ecol 25:79–93
Hallett TB, Coulson T, Pilkington JG, Clutton-Brock TH, Pemberton JM, Grenfell BT (2004) Why large-scale climate indices seem to predict ecological processes better than local weather. Nature 430:71–75
Harrison XA, Blount JD, Inger R, Norris DR, Bearhop S (2011) Carry-over effects as drivers of fitness differences in animals. J Anim Ecol 80:4–18
Holmes RT (1966) Breeding ecology and annual cycle adaptations of the red-backed sandpiper (Calidris alpina) in northern Alaska. Condor 68:3–46
Holt RH, Davies ZG, Staddon S (2008) Meta-analysis of the effects of predation on animal prey abundance: evidence from UK vertebrates. PLoS One 3:e2400
Hosack GR, Peters GW, Hayes KR (2012) Estimating density dependence and latent population trajectories with unknown observation error. Methods Ecol Evol 3:1028–1038
Hunter MD, Price PW (1998) Cycles in insect populations: delayed density dependence or exogenous driving variables? Ecol Entomol 23:216–222
Hurrell J (2003) NAO index data provided by the climate analysis section. NCAR, Boulder
Hurrell J, National Center for Atmospheric Research (NCAR) Staff (eds) (2012) The climate data guide: North Pacific (NP) index by Trenberth and Hurrell; monthly and winter. http://climatedataguide.ucar.edu/guidance/north-pacific-index-npi-trenberth-and-hurrell-monthly-and-winter. Accessed 20 Feb 2013
Hurrell J, National Center for Atmospheric Research (NCAR) Staff (eds) (2013) The climate data guide: Hurrell North Atlantic Oscillation (NAO) index (PC-based). https://climatedataguide.ucar.edu/climate-data/hurrell-north-atlantic-oscillation-nao-index-pc-based. Accessed 17 Oct 2013
Hurvitch CM, Tsai CL (1989) Regression and time series model selection in small samples. Biometrika 76:297–307
Jamieson SE (2009) Cross-seasonal factors affecting breeding investment by female Pacific Dunlins. PhD dissertation, Simon Fraser University, Burnaby
Johnson DH (1981) Summarizing remarks: estimating relative abundance, Part 1. In: Ralph CJ, Scott JM (eds) Estimating numbers of terrestrial birds. Studies in Avian Biology 6. Allen Press, Inc., Lawrence, pp 58–59
Karell P, Ahola K, Karstinen T, Zolei A, Brommer JE (2009) Population dynamics in a cyclic environment: consequences of cyclic food abundance on tawny owl reproduction and survival. J Anim Ecol 78:1050–1062
Kelly JP, Weathers WW (2002) Effects of feeding time constraints on body mass regulation and energy expenditure in wintering dunlin (Calidris alpina). Behav Ecol 13:766–775
Kelly JP, Warnock N, Page GW, Weathers WW (2002) Effects of weather on daily body mass regulation in wintering dunlin. J Exp Biol 205:109–120
Kennedy J, Witthames PR, Nash RDM, Fox CJ (2008) Is fecundity in plaice (Pleuronectes platessa L.) down-regulated in response to reduced food intake during autumn? J Fish Biol 72:78–92
Knape J (2008) Estimability of density dependence in models of time series data. Ecology 89:2994–3000
Knape J, de Valpine P (2012) Are patterns of density dependence in the global population dynamics database driven by uncertainty about population abundance? Ecol Lett 15:17–23
Kölzsch A, Sæther SA, Gustafsson H, Fiske P, Höglund J, Kålås JA (2007) Population fluctuations and regulation in great snipe: a time-series analysis. J Anim Ecol 76:740–749
MacLean SF Jr, Holmes RT (1971) Bill lengths, wintering areas, and taxonomy of North American Dunlins, Calidris alpina. Auk 88:893–901
McCrimmon DA Jr, Fryska ST, Ogden JC, Butcher GS (1997) Nonlinear population dynamics of six species of Florida ciconiiformes assessed by Christmas bird counts. Ecol Appl 7:581–592
Milsom TP, Langton SD, Parkin WK, Peel S, Bishop JD, Hart JD, Moore NP (2000) Habitat models of bird species’ distribution: an aid to the management of coastal grazing marshes. J Appl Ecol 37:706–727
Morrison RIG, Aubry Y, Butler RW, Beyersbergen GW, Donaldson GM, Gratto-Trevor CL, Hicklin PW, Johnston VH, Ross RK (2001) Declines in North American shorebird populations. Wader Study Group Bull 94:34–38
National Audubon Society (2010) The Christmas bird count historical results [online]. http://www.christmasbirdcount.org. Accessed 14 Nov 2011
Newton I (1998) Population limitation in birds. Academic Press, London
Newton I (2004) Population limitation in migrants. Ibis 146:197–226
Newton I (2006) Can conditions experienced during migration limit the population levels of birds? J Ornithol 147:146–166
Newton I (2007) Weather-related mass-mortality events in migrants. Ibis 149:453–467
Nicholson AJ (1933) The balance of animal populations. J Anim Ecol 2:132–148
Niehaus AC, Ydenberg RC (2006) Ecological factors associated with the breeding and migratory phenology of high-latitude breeding western sandpipers. Polar Biol 30:11–17
Norris DR (2005) Carry-over effects and habitat quality in migratory populations. Oikos 109:178–186
Norris DR, Marra PP, Montgomerie R, Kyser TK, Ratcliffe LM (2004) Reproductive effort molting latitude, and feather color in a migratory songbird. Science 306:2249–2250
Ozgul A, Getz LL, Oli MK (2004) Demography of fluctuating populations: temporal and phase-related changes in vital rates of Microtus ochrogaster. J Anim Ecol 73:201–215
Page G, Whitacre DF (1975) Raptor predation on wintering shorebirds. Condor 77:73–83
Perrins CM, Geer TA (1980) The effect of sparrowhawks on tit populations. Ardea 68:133–142
Pomeroy AC, Seaman DAA, Butler RW, Elner RW, Williams TD, Ydenberg RC (2008) Feeding-danger trade-offs underlie stopover site selection by migrants. Avian Conserv Ecol 3:7
Preston FW (1979) The invisible birds. Ecology 60:451–454
Previtali MA, Lima M, Meserve PL, Kelt DA, Gutiėrrez JR (2009) Population dynamics of two sympatric rodents in a variable environment: rainfall, resource availability, and predation. Ecology 90:1996–2006
R Core Team (2012) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. ISBN 3-900051-07-0. http://www.R-project.org/. Accessed 6 Feb 2013
Ratikainen I, Gill J, Gunnarsson T, Sutherland W, Kokko H (2008) When density dependence is not instantaneous: theoretical developments and management implications. Ecol Lett 11:184–198
Reed AW, Slade NA (2008) Density-dependent recruitment in grassland small mammals. J Anim Ecol 77:57–65
Rodenhouse NL, Sherry TW, Holmes RT (1997) Site-dependent regulation of population size: a new synthesis. Ecology 78:2025–2042
Roth RR (1976) Effects of a severe thunderstorm on airborne ducks. Wilson Bull 88:654–656
Royama T (1992) Analytical population dynamics. Chapman & Hall, London
Sæther BE, Engen S, Lande R, Both C, Visser ME (2002) Density dependence and stochastic variation in a newly established population of a small songbird. Oikos 99:331–337
Sæther BE, Grøtan V, Tryjanowski P, Barbraud C, Engen S, Fulin M (2006) Climate and spatio-temporal variation in the population dynamics of a long distance migrant, the white stork. J Anim Ecol 75:80–90
Saunders WE (1907) A migration disaster in Western Ontario. Auk 24:108–111
Schumacker RE, Lomax RG (1996) A beginner’s guide to structural equation modeling. Lawrence Erlbaum Associates, Mahwah
Schwarz GE (1978) Estimating the dimension of a model. Ann Stat 6:461–464
Searle SR, Speed FM, Milliken GA (1980) Population marginal means in the linear model—an alternative to least-squares means. Am Stat 34:216–221
Senner SE, Norton DW, West GC (1989) Feeding ecology of Western Sandpipers, Calidris mauri, and Dunlins, C. alpina, during spring migration at Hartney Bay, Alaska. Can Field Nat 103:372–379
Shenk TM, White GC, Burnham KP (1998) Sampling variance effects on detecting density dependence from temporal trends in natural populations. Ecol Monogr 68:445–463
Sokal RR, Rohlf FJ (1981) Biometry, 2nd edn. Freeman, San Francisco
Soutullo A, Liminana R, Urios V, Surroca M, Gill JA (2006) Density dependent regulation of population size in colonial breeders: Allee and buffer effects in the migratory montagu’s harrier. Oecologia 149:543–552
Stenseth NC, Mysterud A, Ottersen G, Hurrell JW, Chan KS, Lima M (2002) Ecological effects of climate fluctuations. Science 297:1292–1296
Stroud DA, Baker A, DE Blanco, Davidson NC, Delany S, Ganter B, Gill R, González P, Haanstra L, Morrison RIG, Piersma T, Scott DA, Thorup O, West R, Wilson J, Zöckler C, On behalf of the International Wader Study Group (2006) The conservation and population status of the world’s waders at the turn of the millennium. In: Boere GC, Galbraith CA, Stroud DA (eds) Waterbirds around the world. The Stationery Office, Edinburgh, pp 643–648
Sutherland WJ (1996a) Predicting the consequences of habitat loss for migratory populations. Proc R Soc B 263:1325–1327
Sutherland WJ (1996b) From individual behaviour to population ecology. Oxford University Press, Oxford
Sutherland WJ, Norris K (2002) Behavioural models of population growth rates: implications for conservation and prediction. Philos Trans R Soc B 357:1273–1284
Taylor CM, Lank DB, Pomeroy AC, Ydenberg RC (2007) Relationship between stopover site choice of migrating sandpipers, their population status, and environmental stressors. Israel J Ecol Evol 53:245–261
Thomas CD (1990) What do real population dynamics tell us about minimum viable population sizes. Conserv Biol 4:324–327
Török J, Tóth L (1988) Density dependence in reproduction in the collared flycatcher (Ficedula albicollis) at high population levels. J Anim Ecol 57:251–258
Trenberth KE, Hurrell J (1994) NP index data provided by the climate analysis section, NCAR, Boulder. https://climatedataguide.ucar.edu/climate-data/north-pacificnp-index-trenberth-and-hurrell-monthly-and-winter. Accessed 15 March 2013
Turchin P (2003) Complex population dynamics: a theoretical/empirical synthesis. Princeton University Press, Princeton
Tyler NJC, Forchhammer MC, Oritsland NA (2008) Nonlinear effects of climate and density in the dynamics of a fluctuating population of reindeer. Ecology 89:1675–1686
van den Hout PJ, Spaans B, Piersma T (2008) Differential mortality of wintering shorebirds on the Banc d’Arguin, Mauritania, due to predation by large falcons. Ibis 150:219–230
Verhulst J, Kleijn D, Berendse F (2007) Direct and indirect effects of the most widely implemented Dutch agri-environment schemes on breeding waders. J Appl Ecol 44:70–80
Vucetich JA, Peterson RO (2004) The influence of top-down, bottom-up and abiotic factors on the moose (Alces alces) population of Isle Royale. Proc R Soc B 271:183–189
Warnock N, Gill RE (1996) Dunlin (Calidris alpina). In: Poole A (ed) The birds of North America online. Cornell Lab of Ornithology, Ithaca. Retrieved from the birds of North America online. http://bna.birds.cornell.edu/bna/species/203. Accessed 28 Feb 2013
Warnock N, Page GW, Stenzel LE (1995) Non-migratory movements of dunlins on their California wintering grounds. Wilson Bull 107:131–139
Warnock N, Takekawa JY, Bishop MA (2004) Migration and stopover strategies of individual dunlin along the Pacific coast of North America. Can J Zool 82:1687–1697
Warnock N, Handel CM, Gill RE Jr, McCaffery BJ (2013) Residency times and patterns of movement of post breeding dunlin on a subarctic staging area in Alaska. Arctic 66:407–416
White CM, Nancy JC, Tom JC, Hunt WG (2002) Peregrine Falcon (Falco peregrinus). In: Poole A (ed) The birds of North America online. Cornell Lab of Ornithology, Ithaca. Retrieved from the birds of North America online: http://bna.birds.cornell.edu/bna/species/660. Accessed 28 Feb 2013
Wiegand T, Revilla E, Moloney KA (2005) Effects of habitat loss and fragmentation on population dynamics. Conserv Biol 19:108–121
Xu CL, Boyce MS (2009) Oil sardine (Sardinella longiceps) off the Malabar Coast: density dependence and environmental effects. Fish Oceanogr 18:359–370
Ydenberg RC, Butler RW, Lank DB, Guglielmo CG, Lemon M, Wolf N (2002) Trade-offs, condition dependence and stopover site selection by migrating sandpipers. J Avian Biol 33:47–55
Ydenberg RC, Butler RW, Lank DB, Smith BD, Ireland J (2004) Western sandpipers have altered migration tactics as peregrine falcon populations have recovered. Proc R Soc B 271:1263–1269
Ydenberg RC, Dekker D, Kaiser G, Shepherd PCF, Evans Ogden LJ, Rickards K, Lank DB (2010) Winter body mass and over-ocean flocking as components of danger management by Pacific dunlins. BMC Ecol 10:1–11
Young DD Jr, McIntyre CL, Bente PJ, McCabe TR, Ambrose RE (1995) Nesting by golden eagles on the north slope of the Brooks Range in northeastern Alaska. J Field Ornithol 66:373–379
Acknowledgments
This study was funded by Environment Canada. We acknowledge the contribution of the Christmas bird count volunteers and the National Audubon Society, acknowledge National Oceanic & Atmospheric Administration (NOAA)—Earth System Research Laboratory for environmental data and National Centre for Atmospheric Research (NCAR) for large-scale climate data. We thank Connie Smith for research assistance, thank Dr. Dick Dekker and Dr. David C. Schneider for discussion, and thank the anonymous reviewers for comments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xu, C., Barrett, J., Lank, D.B. et al. Large and irregular population fluctuations in migratory Pacific (Calidris alpina pacifica) and Atlantic (C. a. hudsonica) dunlins are driven by density-dependence and climatic factors. Popul Ecol 57, 551–567 (2015). https://doi.org/10.1007/s10144-015-0502-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10144-015-0502-5