Abstract
To compare the safety and efficacy of en bloc resection of non-muscle-invasive bladder cancer (NMIBC) using a 1470-/980-nm dual-wavelength laser (DwLRBT) compared to the gold standard, transurethral resection (TURBT). The study group included 251 patients with a confirmed diagnosis of NMIBC, 97 in the DwLRBT group and 154 in the TURBT group. Clinical characteristics, complications, and recurrence-free survival were compared between the two groups. There were no differences between the two groups with regard to age, sex, mean tumor size, mean tumor number, tumor location, risk, fever, and reoperation. Compared to TURBT, DwLRBT was associated with a shorter hospitalization time (mean±standard deviation: 5.81±1.48 days vs. 4.96±1.32, respectively, p=0.001), shorter catheterization time (4.98±1.47 vs. 4.20±1.48 days, respectively; p=0.035), and smaller volume of intraoperative bleeding (8.43±6.21 ml vs. 6.15±5.08, respectively; p=0.003). Recurrence-free survival (RFS) was better for DwLRBT than TURBT in the overall cohort (hazard ratio [HR], 0.4323; 95% confidence interval [CI], 0.2852–0.6554; p=0.0004) and for the following subgroups and tumor types: intermediate-risk (HR, 0.2654; 95%CI, 0.1020–0.6904; p=0.0245) and high-risk (HR, 0.4461; 95% CI, 0.2778–0.7162; p=0.0027) groups; and for pedunculate bladder tumors (HR, 0.4158; 95%CI, 0.2401–0.7202; p=0.0063), single bladder tumors (HR, 0.4136; 95%CI, 0.2376–0.7293; p=0.0072), and multiple bladder tumors (HR, 0.2727; 95%CI, 0.1408–0.5282; p=0.0014). DwLRBT is associated with better operative and postoperative outcomes, including, importantly, a longer RFS, compared to TURBT.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bladder cancer (BLCA) is the 10th most common cancer worldwide, with a global age-standardized incidence rate of 9.0 per 100,000 men and 2.2 per 100,000 women [1]. Non-muscle-invasive bladder cancer (NMIBC) accounts for 70–75% of all bladder cancers [2]. Transurethral resection of bladder tumors (TURBT) is the gold standard for the treatment of NMIBC [2]. However, conventional TURBT has some significant drawbacks, including intraoperative bleeding, stimulation of the obturator nerve reflex, and bladder perforation [3]. More importantly, as a piecemeal resection process, TURBT is associated with an increased risk of tumor recurrence and difficulty in accurately assessing the pathological stage of tumors due to incomplete resection [4,5,6]. Therefore, en bloc resection of NMIBC, using a laser system, has been recently developed, which provides high-quality specimens, no fragmentation of the tumor, and reduced probability of cauterization and deterioration of the tissue [7]. More importantly, several authors reported better pathological outcomes with en bloc resection of NMIBC than with TURBT [8,9,10]. Various lasers have been developed and used as energy sources to eradicate tumor tissues, including holmium, potassium-titanyl-phosphate, green-light, 1.9 mm Vela, and thulium:yttrium–aluminum–garnet lasers [11]. The first transurethral NMIBC laser surgery was performed in the 1970s and has been widely used in recent years owing to its effectiveness to vaporize tissue, its hemostatic effect, and high safety [12]. The 1470-/980-nm dual-wavelength laser is a newly developed technique which provides the following unique advantages. The 1470 nm laser component provides rapid resection and tissue vaporization, while the 980 nm component provides high hemostasis efficiency and non-contact hemostasis [13]. These characteristics make the 1470-/980-nm dual-wavelength laser very attractive for clinical applications. However, few studies have been reported on the application of this technique for NMIBC. Accordingly, our aim in this study was to evaluate the efficacy and safety of the 1470-/980-nm dual-wavelength laser for en bloc resection of NMIBC (DwLRBT), compared to conventional TURBT.
Methods
Study design and ethics statement
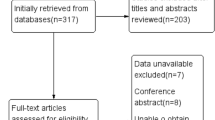
This was a retrospective study of patients with a diagnosis of primary NMIBC who were treated using conventional TURBT or DwLRBT at our institution, between January 2016 and December 2020. Both procedures were performed by the same surgeon. Clinical and surgical outcomes used for analysis were retrieved from our institutional database. Patients provided informed consent for their treatment and for the use of their clinical information for research. Ethics clearance from our institution was waived owing to the retrospective design and use of anonymized data.
Study group
The inclusion criteria were as follows: clinical diagnosis of primary NMIBC based on enhanced magnetic resonance (MR) of the urinary system or cystoscopy and pathological confirmation after surgery; resection using either conventional TURBT or DwLRBT; stratification into low-, intermediate-, or high-risk groups according to the EAU risk stratification criteria [14]; use of standard intravesical epirubicin therapy after surgery; and complete clinical and follow-up data. The exclusion criteria were as follows: locally advanced or metastatic bladder cancer; loss of contact or inadequate clinical information for further analysis; inability to complete intravesical therapy due to patient intolerance or other reasons; and comorbidities of other neoplastic diseases.
After selection, the study group included 251 patients, 97 in the DwLRBT group and 154 in the TURBT group.
Surgical techniques
Under general anesthesia, patients were placed in the lithotomy position and a complete cystoscopic examination of the bladder, including the bladder neck, bladder triangle, posterior wall, lateral wall, anterior wall, and opening of the ureter. TURBT was performed according to standard procedures [15], using a WOLF 26 Fr continuous flow resectoscope (Richard Wolf, Knittlingen, Germany). The electro-resecting and electro-coagulating powers were set to 100 W and 40 W, respectively. A piece-by-piece resection of the muscular layer was performed to remove the body of the tumor, gradually and steadily. For the DwLRBT treatment, a 1470-/980-nm Leonardo® Dual 45 Laser System (Biolitec, Wien, Germany) was used for en bloc resection. A 550 μm fiber with a 26 F continuous laser resectoscope (LISA Laser Products, Lindau-Katlenburg, Germany) was used to enter the bladder through the urethra, with the power of this laser set at 30 W (1470nm 22W and 980nm 8W) during the surgery. First, a circular incision was performed at a relatively safe distance, about 1.0–1.5 cm from the base of the tumor (Figure S1A). The exposed sub-mucosal blood vessels were then precoagulated. Along the circular incision, a fan-shaped incision was created at the base of the tumor to reach the deeper muscle layers along the marked line until the fibrous connective tissue of the outer layer of the bladder was exposed (Figure S1B). Blunt dissection was then performed on the space between the muscle layers. The tumor base was then pushed until the submucosa was exposed by the tip of the resectoscope sheath of the vaporizing laser and pushed the connective tissue layers (Figure S1C). At the visible anatomical level, the muscle fiber was cut from the base of the tumor in different directions until the entire tumor was removed en bloc (Figure S1D).
Postoperative treatment and follow-up
All patients were catheterized using a 20 F three-way catheter and received bladder instillation of epirubicin (50 mg/50 ml, maintained for 60 min) immediately after surgery. Approximately 1-week after the procedure, weekly bladder instillation with epirubicin (50 mg/50 ml, maintained for 60 min) treatment was initiated for all patients for a duration of 8 weeks. This was followed by monthly bladder instillation with epirubicin (50 mg/50 ml, maintained for 60 min) for 11 months or as tolerated by each patient.
The follow-up plan was formulated based on the EAU2 guidelines. Briefly, all patients underwent ultrasound and cystoscopy examination at 1 month post-surgery. These procedures were performed every 3 months, at our hospital, for the first year. Subsequently, all patients underwent ultrasound examination, at approximately every 6 months, at their community hospital. As necessary, a cystoscopy was performed at our hospital. Some routine examinations were simultaneously included in the follow-up plan, such as urine analysis, urine cytology, routine blood tests, and blood biochemistry. The median follow-up period for the TURBT group was 19.5 months and 20 months for the DwLRBT group.
The primary endpoint of our study was the tumor recurrence rate. During the follow-up period, patient information tumor recurrence was obtained by phone or during outpatient consultation.
Statistical analysis
Continous data were reported as the mean±standard deviation and categorical data as a count (percentage). Between-group differences were evaluated using Student’s t-test for continuous data and the chi-squared test or Fisher’s exact test for categorical data. GraphPad Prism 8.4.3 (GraphPad Software, Inc., San Diego, CA, USA) was used to plot the Kaplan-Meier (K-M) curve of RFS and to perform the log-rank test. All other analyses were performed using SPSS (version 20.0; IBM, Armonk, NY, USA) software was used for the statistical analyses.
Results
Baseline characteristics, including age, sex, tumor size, tumor multiplicity, tumor location, and risk groups, were similar between the two groups (Table 1). The intraoperative and postoperative data for both groups are reported in Table 2. While operative time was similar between the two groups (p=0.077), compared to the TURBT group, hospitalization time (p=0.001) and catherization time (p=0.035) were shorter for the DwLRBT group, and the volume of intraoperative bleeding smaller (p=0.003). There were no intraoperative complications with DwLRBT. With TURBT, there were 8 cases of obturator nerve reflex stimulation and 3 cases of bladder perforation. Postoperative complication rates, assessed using the classification of surgical complications [16], included 7 cases of acute rebleeding after TURBT, with reoperation required in 2 of these cases. There were 3 cases of fever, within 48 h post-surgery, in each group. Overall, the rate of surgical complications was not different between the two groups (p=0.787).
The RFS data are shown in Fig. 1 for the group overall and by risk classification (low, intermediate, and high). On K-M analysis, compared to TURBT, DwLRBT was associated with a longer RFS for the overall cohort (hazard ratio [HR], 0.4323; 95% confidence interval [CI], 0.2852–0.6554; p=0.0004), as well as for the intermediate-risk (HR, 0.2654; 95%CI, 0.1020–0.6904; p=0.0245) and high-risk (HR, 0.4461; 95%CI, 0.2778–0.7162; p=0.0027) groups, but not for the low-risk group (p=0.4652). DwLRBT was also associated with a longer RFS, compared to TURBT, for the following tumor groups (Fig. 2): pedunculate bladder tumors (HR, 0.4158; 95%CI, 0.2401–0.7202; p=0.0063); single bladder tumors (HR, 0.4136; 95%CI, 0.2376–0.7293; p=0.0072), and multiple bladder tumors (HR, 0.2727; 95%CI, 0.1408–0.5282; p=0.0014). There was no benefit of DwLRBT for wide-basal bladder tumors (p=0.0716).
Discussion
Currently, TURBT is the gold standard for the treatment of NMIBC despite its many drawbacks which include bleeding, stimulation of the obturator nerve reflex, and bladder perforation [17]. In addition, TURBT is not suitable for patients treated with anticoagulants, which increase the risk of bleeding, and those with artificial pacemakers, as the flow of electricity in tissues during TURBT has the potential to interfere with pacemakers [18, 19]. In addition, the absence of a muscle layer in the resected specimen and, thus, the risk for a positive margin are important shortcomings of TURBT [20]. The advantages of laser-based resection of NMIBC have been reported more recently [21, 22]. In particular, laser surgery for en bloc resection can provide an intact tissue specimen for accurate pathological evaluation, as well as reduce the risk of malignant cell scattering [23]. A meta-analysis of 17 studies, including 2439 participants, showed that, compared to TURBT, laser surgery resulted in a lower incidence of intraoperative complications, lower postoperative tumor recurrence rate, and faster postoperative recovery [24]. The 1470-/980-nm dual-wavelength laser system is a newly employed technology in clinical practice. This system uses a special optical fiber to emit and transmit the laser, which is in turn absorbed by tissues producing vaporization, cutting, and coagulation effects. Fu et al. [25] reported that DwLRBT was as safe and effective as TURBT for the treatment of NMIBC. However, there were no significant differences in terms of RFS or progression-free survival between the two groups. This absence of a survival difference may be due to the small sample size and short follow-up time of that study.
Acute postoperative hemorrhage is a common complication of TURBT. Therefore, continuous postoperative bladder irrigation is important [26]. In our study, there was no incidence of acute rebleeding after DwLRBT and, thus, the postoperative catheterization time was shorter in the DwLRBT than TURBT groups. Although the energy of a laser at a specific wavelength is not absorbed by water during propagation, it can be selectively absorbed by hemoglobin to achieve coagulation and by blood vessels to close leaks, producing a good overall hemostatic effect [27]. Compared to other types of lasers, the combination of the 1470 nm and 980 nm lasers may provide better cutting, vaporization, and hemostasis efficiency as the 1470 nm component is highly absorbed by water, while the 980 nm component provides a high hemoglobin absorption rate and low water absorption rate [28, 29]. Therefore, for patients undergoing long-term anticoagulant therapy, this laser combination provides a novel approach for safe tumor resection.
Stimulation of the obturator nerve reflex is a side effect of TURBT, causing leg jerking during surgery, which may lead to iatrogenic bladder perforation [30]. A systematic review and meta-analysis reported that thulium laser resection of bladder tumors was associated with fewer intraoperative complications, including obturator nerve reflex stimulation and bladder perforation [31]. Similar results were confirmed in our study. With laser resection, the laser probe does not have to be in contact with the tissue for cutting and hemostasis. Furthermore, no electric current is generated during the procedure, thus eliminating the risk of obturator nerve reflex stimulation [32].
Tumor recurrence after TURBT is a very serious problem for patients with NMIBC, especially for patients in the intermediate- and high-risk classification groups, with the possibility of progression to muscle-invasive bladder cancer [33]. Our findings identified a distinct advantage of DwLRBT over conventional TURBT in terms of RFS in the overall cohort, consistent with previous reports on the benefits of laser-based systems for tumor resection [24, 31, 34]. This advantage likely reflects the precise tissue-cutting capability of the laser and superior intraoperative safety, making en bloc resection of NMIBC possible. Complete resection of the tumor, lamina propria, and detrusor can reduce residual tumor and dissemination of tumor cells [35]. In addition, combined with the good tissue vaporization and coagulation effects of the laser, capillaries and lymphatic vessels are effectively closed. Therefore, the possibility of tumor cell dissemination is reduced [36]. Our analysis as a function of tumor type revealed a distinct advantage of DwLRBT over TURBT, in terms of RFS, for pedunculated tumors. This information might assist surgeons and patients in making better choices with regard to procedures. Moreover, compared to TURBT, DwLRBT prolonged RFS regardless of whether patients had single or multiple tumors. Further randomized controlled clinical studies are warranted to validate these results.
The limitations of our study need to be acknowledged for the application of findings to practice. Foremost is the limited number of patients in the DwLRBT group and the absence of multicenter data. As the 1470-/980-nm dual-wavelength laser system was first used in our hospital in June 2015, the median follow-up time was 20 months, which might have also led to bias in the current results.
In conclusion, our study demonstrates that DwLRBT is a safe and effective surgical procedure for NMIBC, which reducedd intraoperative and postoperative bleeding, obturator nerve reflex stimulation, hospitalization time, and catheterization time, compared to conventional TURBT. Importantly, DwLRBT prolonged RFS.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Erratum: global cancer statistics (2018) GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries (2020). CA Cancer J Clin 70(4):313. https://doi.org/10.3322/caac.21609
Babjuk M, Burger M, Capoun O, Cohen D, Compérat EM, Dominguez Escrig JL, Gontero P, Liedberf F, Masson-Leconte A, Mostafid AH, Palou J, van Rhijn BWG, Rouprêt M, Shariat SF, Seisen T, Soukup V, Sylvester RJ (2022) European Association of Urology Guidelines on non-muscle-invasive bladder cancer (Ta, T1, and carcinoma in situ). Eur Urol 81:75–94. https://doi.org/10.1016/j.eururo.2021.08.010
Balci M, Tuncel A, Keten T, Guzel O, Lokman U, Koseoglu E, Aslan Y, Atan A (2018) Comparison of monopolar and bipolar transurethral resection of non-muscle invasive bladder cancer. Urol Int 100:100–104. https://doi.org/10.1159/000467397
Guo CC, Al-Ahmadie HA, Flaig TW, Kamat AM (2021) Contribution of bladder cancer pathology assessment in planning clinical trials. Urol Oncol 39:713–719. https://doi.org/10.1016/j.urolonc.2018.01.001
Koie T, Ohyama C, Hosogoe S, Yamamoto H, Imai A, Hatakeyama S, Yoneyama T, Hashiomoto Y, Yoneyama T, Tobisawa Y, Mori K (2015) Oncological outcomes of a single but extensive transurethral resection followed by appropriate intra-vesical instillation therapy for newly diagnosed non-muscle-invasive bladder cancer. Int Urol Nephrol 47:1509–1514. https://doi.org/10.1007/s11255-015-1048-3
Mariappan P, Zachou A, Grigor KM, Edinburgh Uro-Oncology Group (2010) Detrusor muscle in the first, apparently complete transurethral resection of bladder tumour specimen is a surrogate marker of resection quality, predicts risk of early recurrence, and is dependent on operator experience. Eur Urol 57:843–849. https://doi.org/10.1016/j.eururo.2009.05.047
Kramer MW, Altieri V, Hurle R, Lusuardi L, Merseburger AS, Rassweiler J, Struck JP, Herrmann TRW (2017) Current evidence of transurethral en-bloc resection of nonmuscle invasive bladder cancer. Eur Urol Focus 3:567–576. https://doi.org/10.1016/j.euf.2016.12.004
Kramer MW, Rassweiler JJ, Klein J (2015) En bloc resection of urothelium carcinoma of the bladder (EBRUC): a European multicenter study to compare safety, efficacy, and outcome of laser and electrical en bloc transurethral resection of bladder tumor. World J Urol 33:1937–1943. https://doi.org/10.1007/s00345-015-1568-6
Mori K, D'Andrea D, Enikeev DV, Egawa S, Shariat SF (2020) En bloc resection for nonmuscle invasive bladder cancer: review of the recent literature. Curr Opin Urol 30(1):41–47. https://doi.org/10.1097/MOU.0000000000000697
Hurle R, Lazzeri M, Colombo P et al (2016) “En bloc” resection of nonmuscle invasive bladder cancer: a prospective single-center study. Urology 90:126–130. https://doi.org/10.1016/j.urology.2016.01.004
Kramer MW, Bach T, Wolters M, Imkamp F, Gross AJ, Kuczyk MA, Merseburger AS, Herrmann TRW (2011) Current evidence for transurethral laser therapy of non-muscle invasive bladder cancer. World J Urol 29:433–442. https://doi.org/10.1007/s00345-011-0680-5
Korn SM, Hübner NA, Seitz C, Shariat SF, Fajkovic H (2019) Role of lasers in urology. Photochem Photobiol Sci 18:295–303. https://doi.org/10.1039/c8pp00409a
Aktas AR, Celik O, Ozkan U, Cetin M, Koroglu M, Yilmaz S, Daphan BU, Oguzkurt L (2015) Comparing 1470- and 980-nm diode lasers for endovenous ablation treatments. Lasers Med Sci 30:1583–1587. https://doi.org/10.1007/s10103-015-1768-8
Sylvester RJ, Rodríguez O, Hernández V et al (2021) European Association of Urology (EAU) prognostic factor risk groups for non-muscle-invasive bladder cancer (NMIBC) incorporating the WHO 2004/2016 and WHO 1973 classification systems for grade: an Update from the EAU NMIBC Guidelines Panel. Eur Urol 79:480–488. https://doi.org/10.1016/j.eururo.2020.12.033
Mariappan P (2021) Attention to detail and a permissive set-up: crucial for an effective TURBT. Nat Rev Urol 18:253–254. https://doi.org/10.1038/s41585-021-00441-9
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240:205–213. https://doi.org/10.1097/01.sla.0000133083.54934.ae
Kim LHC, Patel MI (2020) Transurethral resection of bladder tumour (TURBT). Transl Androl Urol 9:3056–3072. https://doi.org/10.21037/tau.2019.09.38
De Nunzio C, Franco G, Cindolo L, Autorino R, Cicione A, Perdonà S, Falsaperla M, Gacci M, Leonardo C, Daminao R, De Sio M, Tubaro A (2014) Transuretral resection of the bladder (TURB): analysis of complications using a modified Clavien system in an Italian real life cohort. Eur J Surg Oncol 40:90–95. https://doi.org/10.1016/j.ejso.2013.11.003
Bolat D, Gunlusoy B, Aydogdu O, Aydin ME, Dincel C (2018) Comparing the short-term outcomes and complications of monopolar and bipolar transurethral resection of bladder tumors in patients with coronary artery disease: a prospective, randomized, controlled study. Int Braz J Urol 44:717–725. https://doi.org/10.1590/S1677-5538.IBJU.2017.0309
Herr HW, Donat SM (2008) Quality control in transurethral resection of bladder tumours. BJU Int 9:1242–1246. https://doi.org/10.1111/j.1464-410X.2008.07966.x
Enikeev D, Shariat SF, Taratkin M, Glybochko P (2020) The changing role of lasers in urologic surgery. Curr Opin Urol 30:24–29. https://doi.org/10.1097/MOU.0000000000000695
Chen GF, Shi TP, Wang BJ, Wang XY, Zang Q (2015) Efficacy of different resections on non-muscle-invasive bladder cancer and analysis of the optimal surgical method. J Biol Regul Homeost Agents 29:465–470
Fried NM (2006) Therapeutic applications of lasers in urology: an update. Expert Rev Med Devices 3(1):81–94. https://doi.org/10.1586/17434440.3.1.81
Xu J, Wang C, Ouyang J, Sun J, Hu C (2020) Efficacy and safety of transurethral laser surgery versus transurethral resection for non-muscle-invasive bladder cancer: a meta-analysis and systematic review. Urol Int 104:810–823. https://doi.org/10.1159/000506655
Fu J, Fu F, Wang Y (2021) 1470 nm/980 nm dual-wavelength laser is safe and efficient for the en-bloc resection of non-muscle invasive bladder cancer: a propensity score-matched analysis. J Int Med Res 49(12):3000605211065388. https://doi.org/10.1177/03000605211065388
Furuse H, Ozono S (2010) Transurethral resection of the bladder tumour (TURBT) for non-muscle invasive bladder cancer: basic skills. Int J Urol 17:698–699. https://doi.org/10.1111/j.1442-2042.2010.02556.x
Bohigian GM (1986) Lasers in medicine and surgery: the other issues. JAMA 256:909–910. https://doi.org/10.1001/jama.1986.03380070115031
Arslan Ü, Çalık E, Tort M, Yildiz Z, Tekin AI, Limandal HK, Kaygin MA, Dağ O, Erkut B (2017) More successful results with less energy in endovenous laser ablation treatment: long-term comparison of bare-tip fiber 980-nm laser and radial-tip fiber 1470-nm laser application. Ann Vasc Surg 45:166–172. https://doi.org/10.1016/j.avsg.2017.06.042
Wollina U, Goldman A (2020) The dual 980-nm and 1470-nm diode laser for vascular lesions. Dermatol Ther 33:e13558. https://doi.org/10.1111/dth.13558
Panagoda PI, Vasdev N, Gowrie-Mohan S (2018) Avoiding the obturator jerk during TURBT. Curr Urol 12:1–5. https://doi.org/10.1159/000447223
Long G, Zhang Y, Sun G, Ouyang W, Liu Z, Li H (2021) Safety and efficacy of thulium laser resection of bladder tumors versus transurethral resection of bladder tumors: a systematic review and meta-analysis. Lasers Med Sci 36:1807–1816. https://doi.org/10.1007/s10103-021-03272-7
Marks AJ, Teichman JM (2007) Lasers in clinical urology: state of the art and new horizons. World J Urol 25:227–233. https://doi.org/10.1007/s00345-007-0163-x
Kassouf W, Traboulsi SL, Schmitz-Dräger B, Palou J, Witjex JA, van Rhijn BWG, Grossman HB, Kiemeney LA, Goebell PJ, Kamat AM (2016) Follow-up in non-muscle-invasive bladder cancer-International Bladder Cancer Network recommendations. Urol Oncol 34:460–468. https://doi.org/10.1016/j.urolonc.2016.05.028
Li C, Gao L, Zhang J, Yang X, Liu C (2020) The effect of holmium laser resection versus standard transurethral resection on non-muscle-invasive bladder cancer: a systematic review and meta-analysis. Lasers Med Sci 35:1025–1034. https://doi.org/10.1007/s10103-020-02972-w
Oswald D, Pallauf M, Herrmann TRW, Netsch C, Becker B, Lehrich K, Meirnik A, Schöb DS, Sievert KD, Gross AJ, Westphal J, Lusuardi L, Deininger S (2022) Transurethral resection of bladder tumors (TURBT). Urologe A 61:71–82. https://doi.org/10.1007/s00120-021-01741-z
Yang H, Wang N, Han S, Male M, Zhao C, Yao D, Chen Z (2017) Comparison of the efficacy and feasibility of laser enucleation of bladder tumor versus transurethral resection of bladder tumor: a meta-analysis. Lasers Med Sci 32:2005–2012. https://doi.org/10.1007/s10103-017-2308-5
Funding
This work is supported by the Natural Science Fund of Guangdong Province (Grant Number: 2021A1515010697). The funding sources had no role in the design and conduct of the study, collection, management, analysis, and interpretation of the data, and preparation, review, or approval of the manuscript.
Author information
Authors and Affiliations
Contributions
Qi-wei Nie and Wen-jun Ni made substantial contributions to the concept of the work and revised the paper. Wen-qiang Zhang, Bin Zhou, and Jian Deng collected, analyzed, interpreted the data, and wrote the manuscript. Geng-yu Han collected clinical data. All authors have read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Ethics approval
Ethics clearance from our institution was waived owing to the retrospective design and use of anonymized data.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Figure S1.
The surgical procedure for DwLRBT. A circular incision is performed at a relatively safe distance, approximately 1.0–1.5 cm proximal to the base of the tumor (A). The fibrous connective tissue of the outer layer of the bladder is exposed (B). The submucosa is exposed (C). En-bloc resection at the (TIF 8405 kb) visible anatomical level (D).
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, W., Zhou, B., Deng, J. et al. Retrospective analysis of 1470-/980-nm dual-wavelength laser en bloc resection versus transurethral resection of bladder tumor for primary non-muscle-invasive bladder cancer. Lasers Med Sci 38, 44 (2023). https://doi.org/10.1007/s10103-023-03708-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10103-023-03708-2