Abstract
The aim of this study was to evaluate the effects of diode laser biostimulation on cementoblasts (OCCM.30). A total of 40 root plates were obtained from healthy third molar teeth and assigned to the following two groups: (1) control group and (2) laser-treated group. Root plates were placed into the cell culture inserts, and OCCM.30 cells were seeded onto root plates. Cells were irradiated with a low level of diode laser (power: 0.3 W in continuous wave, 60 s/cm2). Proliferation and mineralized tissue-associated gene’s and BMP’s messenger RNA (mRNA) expressions of cementoblasts were evaluated. Total RNAs were isolated on day 3 and integrin-binding sialoprotein (Ibsp), bone gamma-carboxyglutamate protein (Bglap), Type I collagen (Col1a1), osteoblastic transcription factor, runt-related transcription factor (Runx2), and Bone Morphogenetic Protein (BMP)-2, 3, 4, 6, and 7 mRNA expressions were determined using quantitative RT-PCR. von Kossa staining was performed to evaluate biomineralization of OCCM.30 cells. In the proliferation experiment, while there was no significant difference until 96 h, laser irradiation retarded the decrease in cell proliferation trend after 96 h compared to the untreated control group. Statistically significant increase in Ibsp, Bglap, and BMP-2,3,6,7 mRNA expressions were noted in the laser groups when compared to the untreated control group (p < 0.05). Laser irradiation induced mineralized nodule formation of cementoblasts. The results of this study reveal that the biostimulation setting of diode laser modulates the behavior of cementoblasts inducing mineralized tissue-associated gene’s mRNA expressions and mineralization. Therefore, biostimulation can be used during regenerative periodontal therapies to trigger cells with periodontal attachment apparatus.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Among the different types of lasers, diode lasers (800–980 nm), which are semiconductor lasers, are used for soft tissue operations, such as incision and homeostasis of gingiva and oral mucosa, soft tissue curettage, periodontal pocket decontamination, and wound healing stimulation [1]. Studies have reported that low-level laser therapy regulates the events of wound healing, including cell proliferation, migration, and extracellular matrix synthesis [2,3,4]. Low-level laser light affects the mitochondria of the cell increasing adenosine triphosphate and diminishes inflammation-modulating reactive oxygen metabolites and cytokines (i.e., interleukin 1-β), and thus, wound healing is improved [5, 6] Our previous in vitro study [3] demonstrated that low-level therapy with diode lasers had no effect on the proliferation of gingival fibroblasts, whereas it considerably stimulated IGF, VEGF, and TGF-β messenger RNA (mRNA) expressions of gingival fibroblasts when compared to the untreated control group. Furthermore, increased collagen type I mRNA expression was observed in the biostimulation set-up of diode laser [3].
Cementum is a unique, avascular mineralized tissue formed by cementoblasts [7]. Periodontal tissue repair/regeneration requires the bioactivity of periodontal cells within the periodontium, including cementoblasts, periodontal ligament cells, and osteoblasts [7]. Research on cementoblasts functions under laser light might be helpful to explain the role of light energy on the cells, which is critical in periodontal wound healing/periodontal regeneration. Diode lasers are not used for application on dental hard tissues, including enamel, dentin, cementum, and bone [8], but the cells/stem cells of these tissues are affected by laser light.
However, the positive effects of laser have been reported in the literature when the laser was used as an adjunct to surgical/nonsurgical periodontal therapies [6, 9,10,11,12,13,14,15], and very recently, meta-analyses have revealed that laser therapy has no additional benefit to clinical parameters, including pocket depth reduction and clinical attachment gain compared to mechanical debridement alone [1]. It is very difficult to determine the effects of laser on the periodontal wound healing owing to the variation in laser protocols. Different clinical outcomes of laser applications require establishing optimal laser treatment protocols with better-defined set-up and indications. Although lasers may offer additional benefits over conventional periodontal therapy, detailed information on the enhancement of wound healing/tissue regeneration using lasers should be further investigated at the cellular and molecular level. Molecular evidences in periodontal wound healing/tissue regeneration by laser light are still limited. Thus, in this study, we planned to assess whether biostimulation with a diode laser regulates cementoblasts functions, such as proliferation, gene transcripts associated with mineralized tissue and biomineralization.
Materials and methods
Cell culture
A mouse-derived immortalized cementoblast cell line (OCCM.30) was used for these studies, and methods for isolating these cells have been previously published [16].
Laser application
Cells were treated with the biostimulation setting of diode lasers (Ezlase, Biolase, USA) (InGaAsP, Indium Gallium Arsenide Phosphate, 940 nm). The laser settings were selected according to the manual of the manufacturer. The parameter set-up was as follows:
-
Power: 0.3 W in continuous wave
-
Irradiation time: 60 s/cm2
-
Energy: 18 J/cm2
A laser optical fiber tip with a 90° angle was used, and the lasing procedure was conducted 0.5–1 mm from the cell culture plates. Biostimulation was carried out using a 300-μm-diameter surgical tip homogenously moving on the well plates/root plates according to time that calculated for the surface square (60 s/cm2). Cell proliferation experiments were performed on the 96-well e-plate. Cell survival and RNA isolation experiments were performed on the root plates. Mineralization experiments were performed on the 48-well plates. (root plates = 0,25 cm2; 96-well plate = 0.19 cm2; 24-well plate = 1,75 cm2). The biostimulation application tip was not initialized according to the manufacturer’s recommendations. To avoid absorption of the laser light by the media component, the media was removed from the well and culture plates, and the cells were irradiated. After laser irradiation, the same culture condition was applied to the wells again [3]. The same procedure was performed for both the untreated control and the laser-irradiated group.
Proliferation of the cementoblasts
Proliferation of the cells was evaluated on the plastic cell culture plates and root plates using a real-time cell analyzer (RT-CA) (xCELLigence, ACEA Biosciences, Inc., CA, USA) and WST** assay, respectively.
For the RT-CA experiment, OCCM.30 cells were plated as 200 μL of the cell suspensions into the wells (5000 cells/well) of the E-plate 96 (xCELLigence, ACEA Biosciences, Inc., CA, USA), and the cells (12 well per group) were monitored every 15 min for 140 h. Cementoblasts on the E-plate 96, which has 96 wells, were treated with DMEM (InVitrogen, Camarillo, CA, USA) containing 10% FBS‡‡. To avoid laser beam scattering effects, the control and laser treatment groups were located far from each other. The values of the electrode impedance were represented as the “cell index” [17, 18].
Preparation of root plates
To mimic clinical conditions, cementoblasts were seeded onto the root plates. For this purpose, a total of 40 root plates were obtained from healthy third molar teeth. Root plates were prepared by removing the crown and apex of each tooth. The middle third of the root was sectioned longitudinally in a bucco-lingual plane, and the rectangular root plates approximately 5 × 5 × 2 mm in size were obtained (Fig. 1). Periodontal ligament tissue from the root surface was scaled. These root plates were assigned to two groups: (1) control group and (2) laser-treated group. The root plates were placed in the cell culture inserts (Millicell, Millipore, Billerica, MA, USA) and OCCM.30 cells were seeded onto the root plates.
Cell survival evaluation on the root plates
Cell survival was evaluated using water-soluble tetrazolium salt (WST)-based assay on days 2 and 5. Briefly, cells (8 × 104/25 μl) were seeded on the root plates in the cell culture insert. After attachment of cells to the root plates, the root specimens were transferred to 48-well plates, and after 24 h, laser light was applied. Then, WST-1 assay was carried out according to the instruction of the supplier. To determine OCCM.30 cell survival, 20 μl of cell proliferation reagent (final dilution 1:10) WST-1(Roche Diagnostics GmbH, Germany) was added to each well and incubated at 37 °C under 5% CO2 in a humidified incubator for 2 h. The absorbance was measured using a microplate reader (μ-Quant) at 480 nm.
Scanning electron microscopy
For scanning electron microscopy (SEM) evaluation, cells (8 × 104/25 μl) were seeded on the root plates in the tissue culture insert. After attachment of cells to the root plates, root specimens were transferred to 48-well plates after 24 h. The morphology and attachment of the cementoblasts on the root surfaces was evaluated on day 3. For SEM evaluation, cells were fixed on root surfaces for 15 min with 2.5% gluteraldehyde in 0.01-M PBS. The specimens were dried and sputter-coated with gold. Cementoblasts on the root surface were photographed with SEM (Zeiss EVO ® LS 10, Brock & Michelsen, Denmark) (at Selcuk University.
RNA isolation
Total RNAs were isolated from cells attached to the root surfaces (as described for cell survival and SEM evaluation experiments) using a monophasic solution of phenol and guanidine isothiocyanate (InVitrogen, Camarillo, CA, USA)on day 3 after laser irradiation. RNA concentration and quality were quantified at 260 nm and 260/280 nm ratio, respectively, by using a spectrophotometer (NanoDrop, Wilmington, DE, USA), and the RNA samples were stored at −80 °C. Integrin-binding sialoprotein (Ibsp), bone gamma-carboxyglutamate protein (Bglap), Type I collagen (Col1a1), osteoblastic transcription factor; runt-related transcription factor (Runx2), and Bone Morphogenetic Protein (BMP)-2, 3, 4, 6, and 7 mRNA expressions were determined using quantitative RT-PCR.
Complementary DNA synthesis and real-time quantitative reverse transcription polymerase chain reaction
Complementary DNAs (cDNAs) were synthesized from 1.0 μg total RNA according to the complementary DNA (cDNA) synthesis kit (Thermo Fisher Scientific, RevertAid First Strand cDNA Synthesis Kit Waltham, MA USA) protocol. PCR reactions were carried out with master mix (Thermo Fisher Scientific, Fermentas Maxima SYBR Green qPCR Master Mix (2X), Waltham, MA USA) in a total volume of 25 μL with three replicates. From the resulting cDNA product, 1.0 μL was used per 25 μL final reaction volume in a thermal cycler (Stratagene, La Jolla, CA, USA). The primers used are listed in Table 1. From a number of different candidate housekeeping genes tested, GAPDH served as the most stable reference gene under our experimental conditions. The amplification profile for Ibsp, Bglap, RunX2, Col1a1, BMP-2,-3,-4,-6,-7, and GAPDH was 95/600; 95/15; 60/60 and melting curve analyses for 95/60; 55/30; 95/30 (temperature in °C/time in seconds) and 35 to 40 cycles. Quantitative RT-PCR experiments were repeated three times.
Mineralization assay
Mineralization experiments were performed on plastic petri dishes instead of root plates. In the first mineralization experiment, root plates were used for von Kossa staining, but after 30 min of fluorescent light incubation, visually apparent mineralized nodules became invisible since the root plates were totally stained to dark brown. Thus, cells were seeded at 5 × 104 cells/cm2 into 48-well plates. After 24 h, the cells were exposed to laser light for the mineralization experiment and were maintained with mineralization media (50 μg/mL ascorbic acid and 10 mM β-glycerophosphate) for 8 days to induce biomineralized nodules in both the positive control and laser irradiation groups. The negative control group was treated without mineralization media. Briefly, wells were washed with phosphate-buffered saline twice and fixed with 100% ethanol at 37 °C for 1 h, and washing was performed with descending alcohol series (90, 80, 70, and 50%) to deionized water. Cells attached to the root were treated with 5% AgNO3 and incubated at 37 °C in the dark for 15 min and washed with deionized water. Plates were exposed to fluorescent light for 30 min [18].
Statistical analysis
The WST and RT-CA experiments were performed twice to ensure reproducibility. For the WST assay, one-way analysis of variance (ANOVA) and Tukey HSD multiple comparison tests were used to compare the groups.
The gene expression data normalization process was performed via the \( {2}^{-\Delta {C}_T^{\prime }} \) method where \( \Delta {C}_T^{\prime }={C}_{T,\mathrm{target}}-{C}_{T,\mathrm{reference}} \) (here, C T , target and C T , reference are the threshold cycles for the target and reference gene amplification, respectively. The averages of the technical replicates of the normalized data of the groups were compared with a Mann–Whitney U test.
Results
Real-time cell proliferation analysis (Fig. 2)
In the proliferation experiment, while there was no significant difference until 96 h, laser irradiation retarded the decrease in cell proliferation trend after 96 h compared to the untreated control group. This effect lasted until the end of the experiment (140 h).
Cell index graph of biostimulation induced OCCM.30 cells (n 12) using real-time cell analyzer. Although there was no significant difference until 96 h, significantly higher proliferation rate was observed after this time point until the end of the experiment. Proliferation experiments were repeated twice. Asterisk represents significant differences (p < 0.05). Cell indexes were given as mean ± standard deviation (M ± SD)
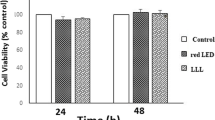
Cell survival assay (Fig. 3)
Although real-time analysis showed higher cell index values after 96 h in the laser-irradiated group, no significant difference was noted in the survival of the cementoblasts on days 2 (48 h) and 5 (120 h). However, the day 5 result of the WST experiment was not exactly the same as that with RT-CA; a similar trend was observed, but the difference was not statistically significant.
Cementoblast attachment to the root surface (Fig. 4)
OCCM.30 cells were attached to the root surface, and the cells were very well organized. However, no quantitative evaluation was performed, and cells exhibited a fibroblastic pattern in the control group, while they were more cuboidal in the laser treated group.
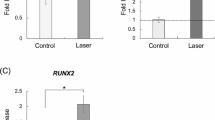
Gene expressions (Fig. 5)
Statistically significant increases in Ibsp and Bglap, and BMP-2,3,6,7 mRNA expressions were noted in the laser groups when compared to the untreated control group (p < 0.05). There was no significant difference between the groups for Col1a1, Runx2, and BMP-4 mRNA expressions. Laser irradiation positively regulated genes related to proteins associated with mineralized tissue, including Ibsp and Bglap, and genes related to mineralized tissue morphogenesis, such as BMP-2, BMP-3, BMP-6, and BMP-7.
mRNA expression of mineralized tissue-associated genes and BMPs in OCCM.30 by real-time PCR (n 3). a Bglap, b, Ibsp, c Col1a1, d Runx2, e BMP-2, f BMP-3, g BMP-4, h BMP-6 and i BMP-7 mRNA expressions of cementoblasts irradiated by diode laser and untreated control. Except for Runx2, COL I, and BMP4, the expressions of all analyzed genes were higher in the laser-treated OCCM.30 cells (target genes/GAPDH). Asterisk represents significant differences (p < 0.05)
Mineralization (Fig. 6)
Pronounced induction in mineralized nodules occurred in the cells treated with laser irradiation when compared to the control group.
Discussion
During the wound healing process, hemostasis, inflammation, proliferation, and remodeling phases occur [19]. For the periodontium, this process is complex since four different tissues, namely gingiva, periodontal ligament, cementum, and bone, play major roles during periodontal wound healing. The proliferation and migration profiles of the cells within the periodontium determine the type of healing as repair or regeneration (re-attachment or new attachment). Low-level laser therapy affects cells in a lineage specific manner [20]. It has been reported that oral fibroblast cell lines may behave different from keratinocytes in terms of the cell proliferation pattern, catalase activity [20], and human β-defensin-2 (HBD-2) [21] expression. Although laser treatments increased reactive oxygen species (ROS) levels in keratinocytes compared to fibroblasts (inversely correlate with higher basal catalase expression), it induced HBD-2 expression in an oral fibroblast cell line compared to a keratinocyte cell line.
Recent systematic review concluded that the laser therapy positively influences in vitro proliferation of stem cells [22]. In our previous study, biostimulation of gingival fibroblasts by laser was ineffective on the proliferation pattern of the cells [3] while laser light positively affected cementoblast proliferation pattern after 96 h in the present study with the same laser system and the same methodology. However, in general, the literature suggests the induction of laser on cell proliferation [2, 4, 22]; in this study, cementoblast proliferation was not changed at the beginning, but at a later phase, cells survived longer than the untreated control group. Considering cementoblasts remain longer within the periodontal area, this may lead to a better attachment gain.
Biological activity of the cells in response to laser application within the periodontal compartment should be investigated. Since new cementum is very critical for the formation of new attachment apparatus [7, 18], determining the response of the cementoblasts exposed to low-level laser irradiation is valuable in order to clarify laser effects at the cellular and molecular level. To the best of our knowledge, there is no study examining the effects of low-level laser irradiation on cementoblasts.
Low-level laser irradiation as an adjunct to surgical periodontal therapy showed better clinical attachment gain with coronally advanced flap or with regenerative materials such as enamel matrix derivatives [13, 14]. This improved clinical attachment gain can be explained by the induction of the cells actively playing a role in healing for both soft and mineralized tissues. PDL fibroblasts, cementoblasts, and osteoblasts play a critical role in reconstructing new attachments. In vitro studies demonstrated that the laser promoted wound healing by stimulating cell migration, proliferation, and differentiation in different types of osteoblasts (Saos-2 cells, MC3T3-E1 cells, MG-63 cells, human fetal osteoblasts) [23–27]. However, although tooth cementum possess different characteristics when compared to bone, the cells of these mineralized tissues may act similarly when exposed to low-level laser irradiation.
An animal study revealed that low-level laser irradiation improved bone healing in tibial defects of rats [28]. Another animal study related to orthodontics showed that low-level laser irradiation during orthodontic force load increased both the reparative and resorptive processes, but laser therapy has significant reparative effects on orthodontically induced inflammatory root resorption in rats [29]. We may speculate that during orthodontic force application, irradiated cementoblasts play an active role in the protection of root resorption. Further, cementoblast co-culture studies with periodontal ligament cells/osteoblasts/osteclasts can be performed in order to clarify the exact mechanism of the effects of laser on the cementoblasts during root resorption process.
In this study, cementoblasts attached to the root surfaces were very well-oriented. The appearance of the cells exposed to low-level laser light was more cuboidal. In our previous studies with cementoblasts, when the cells were treated with BMP, Dex, etc. look more cuboidal [18, 30] and formed more mineralized nodules in vitro. Cells appeared fibroblastic when exposed with basic fibroblast growth factor, which completely downregulated biomineralization of cementoblasts [7]. For von Kossa staining experiment for mineralization, we observed more mineralized nodules in the laser-treated groups, which confirms this finding. The traditional cell survival assay and the real-time cell analyzer experiment results were not exactly the same, but this may be explained by the methodological differences, i.e., plastic surface vs. root surfaces used. As an advantage of RT-CA, we may observe cells over a longer period under constant conditions. Thus, in the present study, we were able to observe the influence of laser at the late phase of cell proliferation. However, the limitations of our studies, which may affect the results, should also be taken into account; e.g., variations in the laser used for this purpose, in the set-up of lasers, and in the methodology of the evaluation of cell function., In this study, cells may not receive homogenous stable energy due to experimental conditions. Biostimulation handpiece was not appropriate for the well size of culture plates (24-, 48-, 96-well plates) used proliferation and mineralization. The lasing procedure was conducted 0.5–1.0 mm from the cell culture plates, using a moving handpiece. Reproducibility of the methods and the energy that the cells received on culture plate, which might not output energy of the laser device during irradiation, should be considered, It should not be also underestimated that results of the studies reported in the literature were obtained from different cell types (human vs. animal; primary or immortalized cells). In our study, we used mouse cementoblasts that may not completely mimic human primary cells and clinical human conditions. This might also be a limitation for a certain conclusion. Further studies should focus on the use of human cementoblasts.
Ibsp and Bglap transcripts required for cementum formation were strongly expressed in the cementum of the tooth. Although there was no change in Col1a1 and Runx2 transcripts, low-level laser therapy induced Ibsp and Bglap mRNA expressions in the cementoblasts. However, although increased Col1a1 expression for gingival fibroblasts [3] and osteoblasts [23] was noted in the literature, laser light may affect cell differently in a cell lineage specific manner. In order to induce new cementum formation, laser might be a valuable tool as an adjunct to the surgical or nonsurgical therapies in periodontics. Further animal studies are required in order to establish appropriate protocols.
During the tooth morphogenesis in the formation of cementum, PDL and alveolar bone is regulated by the signaling of BMP/OP family members [31,31,33] Regulatory functions of BMPs at all stages of tooth morphogenesis were investigated, and the results revealed that BMP-2, BMP−4, and BMP−7 were important for tooth initiation and shape development, whereas BMP-3 was important for root development [34] Although BMP-3 was a suppressor of osteoblastic differentiation [35], laser irradiation up-regulated BMP-3 transcript as well. This is probably because there was a strong balance among the bone morphogenetic proteins, which were the inducers or suppressors of morphogenesis in mineralized tissues.
Variations in the wavelength, power density, and radiation time of the laser, the differences in the protocols used (single or repetitive applications), and the differences in the handpieces used (surgical or biostimulation) may explain the controversial literature reports in studies on laser applications. The results of this study indicated that biostimulation of cementoblasts might positively affect attachment gain since cementoblasts play an important role in establishing the new periodontal attachment apparatus after regenerative periodontal therapies. Further studies are required to determine the effects of low-level laser irradiation on different cell types and to generate established treatment protocols in periodontology/peri-implantology.
References
Mizutani K, Aoki A, Coluzzi D et al (2016) Lasers in minimally invasive periodontal and peri-implant therapy. Periodontol 71(1):185–212
Saygun I, Karacay S, Serdar M et al (2008) Effects of laser irradiation on the release of basic fibroblast growth factor (bFGF), insulin like growth factor-1 (IGF-1), and receptor of IGF-1 (IGFBP3) from gingival fibroblasts. Lasers Med Sci 23(2):211–215
Hakki SS, Bozkurt SB (2012) Effects of different setting of diode laser on the mRNA expression of growth factors and type I collagen of human gingival fibroblasts. Lasers Med Sci 27(2):325–331
Basso FG, Soares DG, de Souza Costa CA et al (2016) Low-level laser therapy in 3D cell culture model using gingival fibroblasts. Lasers Med Sci 31(5):973–978
Karu T (2010) Mitochondrial mechanisms of photobiomodulation in context of new data about multiple roles of ATP. Photomed Laser Surg 28(2):159–160
Lui J, Corbet EF, Jin L (2011) Combined photodynamic and low-level laser therapies as an adjunct to nonsurgical treatment of chronic periodontitis. J Periodontal Res 46:89–96
Saygin NE, Giannobile WV, Somerman MJ (2000) Molecular and cell biology of cementum. Periodontol 24:73–98
Aoki A, Mizutani K, Schwarz F et al (2015) Periodontal and peri-implant wound healing following laser therapy. Periodontol 68(1):217–269
Low SB, Mott A (2014) Laser technology to manage periodontal disease: a valid concept? J Evid Based Dent Pract 14(Suppl):154–159
Aykol G, Baser U, Maden I et al (2011) The effect of low-level laser therapy as an adjunct to non-surgical periodontal treatment. J Periodontol 82(3):481–488
Saglam M, Kantarci A, Dundar N et al (2014) Clinical and biochemical effects of diode laser as an adjunct to nonsurgical treatment of chronic periodontitis: a randomized, controlled clinical trial. Lasers Med Sci 29(1):37–46
Koçak E, Sağlam M, Kayış SA et al (2016) Nonsurgical periodontal therapy with/without diode laser modulates metabolic control of type 2 diabetics with periodontitis: a randomized clinical trial. Lasers Med Sci 31(2):343–353
Ozturan S, Durukan SA, Ozcelik O et al (2011) Coronally advanced flap adjunct with low intensity laser therapy: a randomized controlled clinical pilot study. J Clin Periodontol 38(11):1055–1062
Ozcelik O, Cenk Haytac M, Seydaoglu G (2008) Enamel matrix derivative and low-level laser therapy in the treatment of intra-bony defects: a randomized placebo-controlled clinical trial. J Clin Periodontol 35(2):147–156
Sanz-Moliner JD, Nart J, Cohen R et al (2013) The effect of an 810-nm diode laser on postoperative pain and tissue response after modified Widman flap surgery: a pilot study in humans. J Periodontol 84(2):152–158
D’Errico JA, Ouyang H, Berry JE et al (1999) Immortalized cementoblasts and periodontal ligament cells in culture. Bone 25:39–47
Hakki SS, Bozkurt B, Hakki EE et al (2014) Bone morphogenetic protein-2, -6, and -7 differently regulate osteogenic differentiation of human periodontal ligament stem cells. J Biomed Mater Res B Appl Biomater 102(1):119–130
Hakki SS, Foster BL, Nagatomo KJ et al (2010) Bone morphogenetic protein-7 enhances cementoblast function in vitro. J Periodontol 81(11):1663–1674
Solmaz H, Dervisoglu S, Gulsoy M et al (2016) Laser biostimulation of wound healing: bioimpedance measurements support histology. Lasers Med Sci 31(8):1547–1554
Engel KW, Khan I, Arany PR (2016) Cell lineage responses to photobiomodulation therapy. J Biophotonics 9(11–12):1148–1156
Tang E, Khan I, Andreana S et al (2016) Laser-activated transforming growth factor-β1 induces human β-defensin 2: implications for laser therapies for periodontitis and peri-implantitis. J Periodontal Res. doi:10.1111/jre.12399
Ginani F, Soares DM, Barreto MP et al (2015) Effect of low-level laser therapy on mesenchymal stem cell proliferation: a systematic review. Lasers Med Sci 30(8):2189–2194
Tschon M, Incerti-Parenti S, Cepollaro S et al (2015) Photobiomodulation with low-level diode laser promotes osteoblast migration in an in vitro micro wound model. J Biomed Opt 20(7):78002
Migliario M, Pittarella P, Fanuli M et al (2014) Laser-induced osteoblast proliferation is mediated by ROS production. Lasers Med Sci 29(4):1463–1467
Medina-Huertas R, Manzano-Moreno FJ, De Luna-Bertos E et al (2014) The effects of low-level diode laser irradiation on differentiation, antigenic profile, and phagocytic capacity of osteoblast-like cells (MG-63). Lasers Med Sci 29(4):1479–1484
Jawad MM, Husein A, Azlina A et al (2013) Effect of 940 nm low-level laser therapy on osteogenesis in vitro. J Biomed Opt 18(12):128001
Bloise N, Ceccarelli G, Minzioni P et al (2013) Investigation of low-level laser therapy potentiality on proliferation and differentiation of human osteoblast-like cells in the absence/presence of osteogenic factors. J Biomed Opt 18(12):128006
Tim CR, Pinto KN, Rossi BR et al (2014) Low-level laser therapy enhances the expression of osteogenic factors during bone repair in rats. Lasers Med Sci 29(1):147–156
Altan AB, Bicakci AA, Mutaf HI et al (2015) The effects of low-level laser therapy on orthodontically induced root resorption. Lasers Med Sci 30(8):2067–2076
Hakki SS, Nohutcu RM, Hakki EE et al (2005) Dexamethasone and basic-fibroblast growth factor regulate markers of mineralization in cementoblasts in vitro. J Periodontol 76(9):1550–1558
Ripamonti U, Renton L (2006) Bone morphogenetic proteins and the induction of periodontal tissue regeneration. Periodontol 41:73–87
Kishigami S, Mishina Y (2005) BMP signaling and early embryonic patterning. Cytokine Growth Factor Rev 16:265–278
Thesleff I, Sharpe P (1997) Signalling networks regulating dental development. Mech Dev 67:111–123
Aberg T, Wozney J, Thesleff I (1997) Expression patterns of bone morphogenetic proteins (Bmps) in the developing mouse tooth suggest roles in morphogenesis and cell differentiation. Dev Dyn 210:383–396
Kokabu S, Gamer L, Cox K et al (2012) BMP3 suppresses osteoblast differentiation of bone marrow stromal cells via interaction with Acvr2b. Mol Endocrinol 26(1):87–94
Acknowledgements
A mouse-derived immortalized cementoblast cell line (OCCM.30) was kindly provided by Prof Martha J. Somerman, from NIH. This study was funded from Selcuk University Research Foundation, Konya, Turkey (SSH). This work was performed at Selcuk University, Research Center of Dental Faculty, Konya, Turkey.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Cells used in this study were a mouse-derived immortalized cementoblast cell line (OCCM.30) and were kindly provided by Prof Martha J. Somerman, from NIH. Ethical approval is not needed.
Conflict of interest
The authors declare that they have no conflict of interest.
Funding
This study was supported by Selcuk University Research Foundation. Grant was used for cell culture studies and SEM.
Rights and permissions
About this article
Cite this article
Bozkurt, S.B., Hakki, E.E., Kayis, S.A. et al. Biostimulation with diode laser positively regulates cementoblast functions, in vitro. Lasers Med Sci 32, 911–919 (2017). https://doi.org/10.1007/s10103-017-2192-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-017-2192-z