Abstract
Enlargement and dysfunction of the right atrium might be an early sign for pulmonary hypertension in systemic sclerosis (SSc). This is the first study to analyse right atrial morphology and function in SSc patients compared to healthy controls by speckle-tracking two-dimensional strain echocardiography (2DSE) at rest and during exercise. Furthermore, right atrial function was correlated with further clinical findings. Adult patients with SSc for >3 years (n = 90) and 55 age- and gender-matched healthy controls underwent a panel of non-invasive assessments including transthoracic echocardiography, pulsed Doppler myocardial imaging and 2DSE at rest and during exercise. Furthermore, serological tests and high-resolution chest computed tomography were performed. SSc patients showed significant impairment of right atrial function and the right atrial enlargement, measured by 2DSE at rest and during exercise compared to controls (both p < 0.001). These findings were more evident in SSc patients with pulmonary fibrosis (p < 0.001) and in patients with high pulmonary artery systolic pressures (PAPs) during exercise. In the SSC patients, right atrial lateral strain was significantly associated with PAPs during effort, right atrial area, left ventricle stroke volume and inferior vena cava diameter using multivariable analysis. The findings of this study suggest that a high proportion of SSc patients reveal right atrial dysfunction even without manifest pulmonary hypertension. Impaired right atrial function occurred mostly in patients with pulmonary fibrosis and/or elevated PAPs during exercise, was independently associated with prognostic factors and may therefore be useful for risk stratification. Further studies are needed to analyse if right atrial dysfunction assessed by 2DSE may help to improve early diagnosis of pulmonary hypertension.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Systemic sclerosis (SSc) is a multisystem disorder characterised by widespread vascular lesions and fibrosis of skin and distinct internal organs [1–3]. Cardiac involvement and pulmonary hypertension (PH) are common findings that are often diagnosed at a late stage leading to a markedly reduced survival. In many cases, PH is due to pulmonary arterial hypertension (PAH) associated with SSc. In fact, clinical evidence of myocardial disease may be found in 20–25 % of patients with SSc, whilst at post-mortem examination, the heart is affected in up to 80 % of patients [4, 5]. Therefore, an early diagnosis of PH and cardiac impairment may be essential for SSc patients.
Right atrial size and function reflects right ventricular (RV) function and is strongly associated with clinical outcomes in many conditions as in PH [6–8].
The normal right atrium is acting as reservoir for systemic venous return and by filling the RV in an active contraction during the late diastole. Right atrial contraction is responsible for up to 30 % of normal RV output [9] and can help to compensate short-term right ventricular overload [10].
Enlarged atrial size or dysfunction of right atrial contraction may be an early indicator for pulmonary hypertension and/or primary diseases of the right heart. Therefore, it might be helpful to establish non-invasive echocardiographic techniques to assess right atrial function. Doppler myocardial imaging (DMI) and Doppler strain rate imaging (SRI) can assess important parameters of cardiac function as myocardial timing, contractility and cardiac deformation and are highly feasible and easily-repeatable [11–13]. However, both DMI and SRI are limited by Doppler angle of incidence. A novel approach to quantify regional function from routine grey-scale 2D echocardiographic images is speckle-tracking two-dimensional strain echocardiography (2DSE). This technique calculates myocardial strain independently of angle of incidence. It has been developed to study ventricular function and has also been used to evaluate atrial function [14]. However, the 2DSE at rest and during exercise has not yet been used to detect right atrial dysfunction in SSc patients.
Therefore, the aims of the present study were (1) to analyse right atrial morphology and function in SSc-patients and healthy controls at rest and during exercise using 2DSE, (2) to correlate 2DSE findings with further non-invasive clinical parameters which are usually used as screening for PH or myocardial dysfunction and (3) to investigate the differences in right atrial morphology and function between SSc patients with and without pulmonary fibrosis.
Methods
Study population and design
The study was designed as a cross-sectional study in which SSc patients without known PH or myocardial dysfunction have been systematically screened using echocardiography at rest, 2DSE and high-resolution chest computed tomography (HR-CT). Furthermore, we consecutively included age- and gender-matched healthy non-athletic controls at Monaldi Hospital, Naples, Italy as previously described [15]. The investigators who performed echocardiography and 2DSE were blinded to the results of clinical evaluation and of HR-CT, to minimise bias, as the confirmatory diagnostic test was performed in each patient. Patients with diffuse cutaneous systemic sclerosis (dcSSc) and limited cutaneous systemic sclerosis (lcSSc, CREST-Syndrome), aged ≥18 years, were included. Diagnosis of SSc was confirmed by experienced rheumatologists, according to the standard criteria of the American College of Rheumatology/European League Against Rheumatism [16]. Exclusion criteria were manifest PH confirmed by right heart catheterization (RHC) prior to enrolment, receiving PH therapy, forced vital capacity <40 % of predicted, renal insufficiency, systemic arterial hypertension with pressure values >160/90 mmHg at rest or >220/120 mmHg during exercise despite optimised medical treatment, previous evidence of clinically relevant left heart disease or pregnancy, significant lung disease other than SSc-related interstitial lung disease, evidence of left ventricle (LV) diastolic dysfunction at echocardiography (E/E’ ratio >15) and inadequate echocardiograms.
All patients underwent a detailed clinical work-up including medical history, physical examination, electrocardiogram, 2-dimensional-echocardiography, pulsed DMI and 2DSE at rest and during exercise, lung function test, arterial blood gases, chest Xray, HR-CT, laboratory testing including NTpro-BNP levels. Twelve-lead ECG was performed in all patients.
Pulmonary fibrosis diagnosis was made if HR-CT showed compatible changes in bibasilar reticular or air-space opacities. The extent of the individual HR-CT scan patterns was estimated in each of five lobes. Ground-glass opacity was defined as a hazy increase in lung parenchymal attenuation, and fibrosis score included lobular septal thickening and subpleural honeycomb changes.
This study was conducted in accordance with Good Clinical Practice (GCP) and the current version of the revised Declaration of Helsinki (WMA Declaration of Helsinki). The ethics committee of the V. Monaldi Hospital approved the study. A written informed consent was obtained from each patient prior to enrolment.
Standard transthoracic 2-D-echocardiography
Standard Doppler echocardiography, DMI and 2DSE were performed in the subjects in left semirecumbent position according to the standard criteria of the American Society of Echocardiography by standard ultrasound system (Vivid E9, GE Healthcare, Milwaukee, Wisconsin) [15]. Maximal right atrial area was calculated by planimetry in the apical four-chamber view at the point of tricuspid valve opening as previously described [17].
All the measurements were analysed by 2 experienced readers, on the average of >3 cardiac cycles.
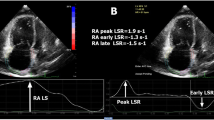
Right atrial non-Doppler two-dimensional strain (2DSE)
Apical 4-chamber view was obtained using the same ultrasound system and probe used for standard echocardiography. Frame rates of 50 to 90 Hz were used for routine grey-scale imaging. A circular region of interest was traced on the endocardial cavity interface of the apical 4-chamber view at right atrial systole (minimum cavity area) by a point-and-click approach. The tracking algorithm followed the endocardium from this one frame throughout the cardiac cycle. The image processing algorithm automatically subdivided the region of interest into blocks of approximately 20 to 40 pixels containing stable patterns of speckles, and a series of regional strain vectors are calculated as change in length/initial length. Accordingly, for atrial longitudinal strain, myocardial thickening was represented with a positive value and myocardial thinning was represented with a negative value. The tracking process and conversion to Lagrangian strains were performed offline with a dedicated software (EchoPAC PC 2D strain - BT 0.5.2 - GE Healthcare).
Analysis was performed for non-Doppler atrial strain from the 4-chamber apical views for the basal segments of right atrial septum and right atrial lateral wall. Continuous care was taken to keep the sample volume out of the caval veins and fossa ovalis. All the measures were performed both at rest and during the peak of effort [18].
Exercise stress echocardiography
All patients enrolled in the study underwent a supine bicycle exercise stress echocardiography after the resting echocardiogram, using a standard protocol (incremental steps of 25 watts for 2 min) as previously reported [19]. The heart rate was continuously monitored, and 12-lead electrocardiogram, echocardiographic images and blood pressure were recorded at every step. Criteria for test interruption were achievement of maximal heart rate, onset of new or worsening wall motion abnormalities, severe dyspnoea or chest pain, horizontal or down-sloping ST-segment depression ≥2 mm, ST-segment elevation ≥1.5 mm, systolic blood pressure ≥220 mmHg, diastolic blood pressure ≥120 mmHg, reduction in systolic blood pressure ≥30 mmHg, supraventricular or ventricular tachyarrhythmias. Two-dimensional images were obtained in 4 standard views (parasternal long-axis, parasternal short-axis, apical 4- and 2-chamber view) using Vivid E9 (GE Healthcare, Milwaukee, Wisconsin) ultrasound system, at baseline, at each exercise step and during recovery, and recorded using a quad-screen cine-loop system.
Cardiologists blinded to the study analysed the test. Functional parameters measured at peak effort included maximal heart rate, maximal systolic blood pressure, maximal workload (number of watts achieved by supine bicycle test) and rate-pressure product, standard echocardiography parameters included ejection fraction (EF), tricuspid annular plane systolic excursion (TAPSE), systolic pulmonary arterial pressure (PAPs), DMI, systolic and diastolic parameters and 2DSE.
Statistical methods
Variables are presented as mean ± SD. Two-tailed T test for unpaired data was used to assess differences between groups. Comparisons were performed between SSc patients and controls and between SSc patients with and without pulmonary fibrosis. Linear regression analyses and partial correlation test by Pearson’s method were done to assess univariate relations between echocardiographic and clinical data.
To identify significant independent determinants of right atrial myocardial strain in patients with systemic sclerosis, its individual association with echocardiographic variables and further clinically relevant parameters were assessed by multivariable linear regression analysis. The variables were selected according to their clinical relevance and potential impact on right atrial function, as shown by earlier studies. The following variables were included into the analysis: clinical and laboratory data (age, sex, heart rate, blood pressure at rest and during effort, BNP), standard echocardiographic parameters (right atrial area, PAPs, IVC diameter, TAPSE, Cardiac Index (CI), stroke volume (SV) at rest and during effort), DMI echocardiographic parameters (RV peak systolic velocity (Sm) and peak early diastolic velocity (Em) at rest and at peak of effort) and strain (right atrial two-dimensional measurements). Variable selection of the multivariable linear regression was performed by stepwise backward elimination, each time excluding the one variable improving the model most by deletion (according to p value of the Wald statistic). The assumption of linearity was checked graphically by studying the smoothed martingale residuals from the null model plotted against the covariate variables. The linearity assumptions were satisfied. The Hosmer-Lemeshow goodness-of-fit test was used to check that the model adequately fits the data. The models underwent also bootstrap validation (200 runs), with the calculation of the c statistic to evaluate discrimination and the shrinkage coefficient to evaluate calibration; in both cases, the closer the value to 1, the better. In order to decrease the inflation of the type 1 error rate due to multiple testing, the statistical significance was defined as two-sided p value <0.01.
Reproducibility of measuring the 2D strain parameters was determined in 50 subjects (30 SSc and 20 controls), according to previously reported methods. Inter- and intra-observer variability was examined using Bland-Altman analysis. Ninety-five percent confidence limits of a single estimate of the measurements were calculated as 2*SD/√l2 and reported as a percent from the mean value.
Receiver operating characteristic (ROC) curve analysis, multiple regression analyses or logistic regression analyses were performed to weigh the independent effects of potential determinants on a dependent variable. Differences were significant at p < 0.05. All analyses were performed using IBM SPSS Statistics for Windows, Version 22.0. IBM Corp. Released 2013, Armonk, NY: IBM Corp.
Results
Characteristics of the study population
During the study, 102 SSc patients were screened, and 12 were excluded due to the following reasons: the diagnosis of systemic sclerosis was not confirmed in 5 patients by an experienced rheumatologist, previous evidence of clinically relevant left heart disease in 4 patients and inadequate echocardiograms in 3 patients. Thus, the final study group consisted of 90 consecutive patients with systemic sclerosis (Table 1). We assessed 55 age- and gender-matched healthy adult non-athletes. Mean age, gender, body surface area, heart rate at rest and systemic blood pressure did not significantly differ between groups (Table 1).
Immunofluorescence analysis detected anti-SCL-70 antibody pattern in 39 patients and anticentromere pattern in 51 patients.
Echocardiographic data are summarised in Tables 2 and 3.
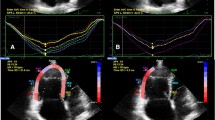
2D echocardiographic analysis (Fig. 1, Table 2)
LV and RV dimensions and standard Doppler indexes were comparable between the two groups. Patients with SSc showed significantly higher IVC diameter, PAPs and PAPm than control subjects. In addition, right atrial area was significantly greater (17.8 ± 4.4 vs. 12.8 ± 3.4), and right atrial 2D strain indexes were significantly impaired in patients with SSc compared with controls (Fig. 2).
Univariable relations
In the SSc group, a close positive association of right atrial lateral 2D strain with increase of LV SV during effort (r = 0.45, p < 0.01; Fig. 3) and powerful negative correlations of the same right atrial 2D strain and right atrial area (r = −0.45, p < 0.001), PAPs during effort (r = −0.7, p < 0.0001; Fig. 4) and IVC diameter were evidenced (r = −0.81, p < 0.0001; Fig. 5).
Multivariable analysis
By stepwise backward, multiple linear regression analysis, after adjusting for potential determinants, the independent direct association of right atrial lateral strain with PAPs during effort (β coefficient = −0.58, p < 0.0005), right atrial area (β coefficient = −0.38, p < 0.01), LV stroke volume (β coefficient = 0.43, p < 0.001) and inferior vena cava (IVC) diameter (β coefficient = −0.71, p < 0.0001) in SSc patients were confirmed.
Analysis of SSc subgroups with or without pulmonary fibrosis
We performed a separate subset analysis in SSc patients with (48 patients) or without (42 patients) SSc-related pulmonary fibrosis detected by HR-CT (Table 3). By standard Doppler echocardiographic analysis LV dimensions, wall thickness, LVEF measured by Simpson modified rule were comparable between the two subgroups. Also, RV dimension and TAPSE were comparable. SSc patients with pulmonary fibrosis showed significantly higher IVC diameter, PAPs, PAPm and right atrial area than SSc patients without pulmonary fibrosis. In addition, right atrial deformation indices were significantly compromised in patients with pulmonary fibrosis.
Finally, we found that a right atrial lateral strain <25 % could identify patients with pulmonary fibrosis with a sensitivity and specificity of 90 and 74 %, respectively (AUC: 0.83, 95 % CI 0.66 to 0.84, p < 0.0001) (Fig. 6).
Exercise stress echocardiography
PAPs was greater in patients with SSc than that in controls at rest as well as during effort. In particular, among SSc patients, those with pulmonary fibrosis had significantly higher PAPs during effort. Conversely, SV was lower both at rest and at peak stress in patients with pulmonary fibrosis. RV and right atrial myocardial function by DMI was similar in the two groups at rest, but was reduced both at rest as well as during exercise by SRI, particularly in the subgroup with pulmonary fibrosis (Tables 2 and 3).
Reproducibility of 2DSE RA strain
Overall, RA strain analysis was feasible in 88 (98 %) of SSc patients and in 54 (98 %) of control subjects at rest and in 85 (93 %) of SSc patients and in 52 (95 %) of control subjects during effort.
Intra-observer variability
Pearson’s correlations: lateral wall: r = 0.87; p < 0.00001—atrial septum: r = 0.88; p < 0.00001.
Bland-Altman analysis: lateral wall (95 % CI ± 1.8; percent error 3.3 %); atrial septum (95 % CI ± 1.2; percent error 3.1 %).
Inter-observer variability
Pearson’s correlations: lateral wall: r = 0.85; p < 0.00001—atrial septum: r = 0.86; p < 0.00001.
Bland-Altman analysis: lateral wall (95 % CI ± 1.9; percent error 3.4 %); atrial septum (95 % CI ± 1.5; percent error 3.5 %).
Discussion
To the best of our knowledge, this is the first study assessing right atrial function in patients with SSc compared to age- and gender-matched healthy controls by 2DSE both at rest and during exercise. In our study cohort, SSc patients without overt PH or RV-LV dysfunction showed already an impaired right atrial function and right atrial dilatation compared to healthy controls. Right atrial dysfunction was significantly associated with pulmonary fibrosis and/or with elevated pulmonary artery pressures or reduced left ventricular stroke volume during exercise in the SSc patients. The results of the present study underline the usefulness of 2DSE in analysing right atrial longitudinal myocardial function in patients with SSc.
Right atrial morphology and function in systemic sclerosis
To date, little is known about right atrial function in SSc patients. Although right atrial enlargement has been described as a negative prognostic factor in patients with primary PAH, to the best of our knowledge, there are only two reports that investigated right atrial area in a small population of SSc patients [20, 21]. Only one of these assessed right atrial functions of SSc patients by using 2DSE, and none had correlated these findings with the response to exercise. Per Lindqvist et al. demonstrated that patients with SSc exhibit an altered RV diastolic function and an increase in RV wall thickness and right atrial area in spite of normal RV end-diastolic diameters. In particular, in patients with SSc, LV function and RV systolic function were normal, but RV abnormal relaxation and filling properties were observed, together with RV hypertrophy and right atrial dilatation in the absence of Doppler indications of pulmonary arterial hypertension estimated from the RV-right atrial drop gradient [22].
Durmus et al. demonstrated that the value of peak early and late diastolic longitudinal strains, determined as right atrial reservoir and conduit functions, were significantly impaired in SSc patients compared to controls [23]. These previous studies are in line with our findings. In our SSc population, despite normal LV and RV function, there was a right atrial enlargement above the cut-off value of 16 cm2 [17] and a significant reduction of right atrial function measured by 2DSE at level of both the analysed segments. 2DSE detected a more impaired right atrial function in the subgroup of SSc patients with pulmonary fibrosis.
Therefore, right atrial area enlargement and even more right atrial function impairment may represent an early marker of RV disturbance, probably in response to intermittent PAH, since LV function was unaffected.
Right atrial function in different diseases
Unlike the left atrium, right atrial functional components are so far poorly investigated, and right atrial strain normal cut-offs have not been assessed yet. Previous studies in patients with heart failure found that diminished right atrial function, as assessed by strain imaging, may reflect maladaptive changes and could also explain why, for a given degree of RV dysfunction, pulmonary artery pressure can vary in its phenotypic right-sided heart failure expressions [24]. Similarly, in a previous report, we found that right atrial area index was increased, and right atrial strain of lateral wall was impaired in patients with dilated cardiomyopathy non-responders to resynchronization therapy [25], whilst Sivak et al. reported that right atrial systolic function accounted for 50 % of total longitudinal RV shortening in patients with pulmonary hypertension [26]. These findings suggest that more emphasis must be placed on investigating right atrial mechanics than previously appreciated, because right atrial function is likely to play a critical role in supporting total right heart function [17].
In the event of increased afterload, diastolic dysfunction may be demonstrated at an earlier stage than systolic dysfunction. Thus, right atrial dysfunction seems a marker that could easily reveal diastolic dysfunction even when RV systolic function is still normal [20].
Therefore, 2DSE could detect early right atrial dysfunction even in asymptomatic SSc patients without clinically evident cardiac disease and in whom no other cause of either diastolic or systolic impairment was detected.
Associations between right atrial myocardial deformation and response to physical effort
In systemic sclerosis, the involvement of the interstitium or vascular system of the lung may lead to PAH. PAH is often asymptomatic or oligosymptomatic in SSc and, when it becomes symptomatic, the pulmonary vascular system is already damaged. Exercise echocardiography, measuring pulmonary artery pressure (PAP) during exercise and allowing differentiating physiologic from altered PAP responses, may identify subclinical PAH. In our previous reports, we demonstrated that exercise echocardiography may identify a subset of SSc patients with an inappropriate exercise-induced increase in PAPs and early signs of right ventricular dysfunction [19]. Furthermore, stress Doppler echocardiography markedly improved sensitivity in detecting manifest PAH [27].
In the present study, PAPs showed significantly higher values in patients with SSc than controls at rest as well as during effort. Among SSc patients, those with pulmonary fibrosis had higher PAPs during effort. SV and CI were reduced both at rest and at peak stress in patients with pulmonary fibrosis. Myocardial function by DMI and SRI, that was similar in the two groups at rest, was reduced during exercise, particularly in the subgroup with pulmonary fibrosis.
Reduced right atrial 2DSE at rest predicted an increased PAPs and a reduced increase of SV during physical effort. These results suggest that SSc patients may present with hypertensive pulmonary arterial pressure during exercise as a likely cause of RV diastolic dysfunction.
Study limitations
Our study has several limitations.
A technical limitation is that speckle-tracking echocardiography is dependent on frame rate as well as image resolution. Low frame rate results in the speckle pattern changing too much from frame to frame, which prevents the precise characterisation of regional myocardial motion and impacts the overall temporal resolution of the regional strain map. In contrast, increasing the frame rate reduces scan line density, which reduces image resolution [28–30]. Frame rate in our setting ranged from 50 to 90 frame/sec; this value is lower than frame rate available with Doppler strain; however, indexes of RA function used in this study didn’t rely on difference in the timing of contraction.
Our analysis was limited by data collection from the apical four-chamber view, as this was the only image plane that provided reliable information on longitudinal right atrial deformation in a sufficient number of patients. However, in the present study, atrial radial strain was not calculated from the parasternal short- and long-axis views because the atrial wall is too thin to be properly analysed in these views, as demonstrated by our previous studies [31, 32].
Conclusions
The findings of this study suggest that a high proportion of SSc-patient reveal right atrial dysfunction even without manifest pulmonary hypertension. Impaired right atrial function that occurred mostly in patients with pulmonary fibrosis and/or elevated PAPs during exercise was independently associated with prognostic factors and may therefore be useful for risk stratification. Further studies are needed to analyse if right atrial dysfunction non-invasively assessed by 2DSE may help to improve an early diagnosis of pulmonary hypertension and risk stratification.
References
(1980) Preliminary criteria for the classification of systemic sclerosis (scleroderma). Subcommittee for scleroderma criteria of the American Rheumatism Association Diagnostic and Therapeutic Criteria Committee. Arthritis Rheum 23:581–90.
LeRoy EC, Black C, Fleischmajer R, Jablonska S, Krieg T, Medsger TA Jr et al (1988) Scleroderma (systemic sclerosis): classification, subsets and pathogenesis. J Rheumatol 15:202–205
Gabrielli A, Avvedimento EV, Krieg T (2009) Scleroderma. N Engl J Med 360(19):1989–2003
Fernandes F, Ramires FJ, Arteaga E, Ianni BM, Bonfá ES, Mady C (2003) Cardiac remodeling in patients with systemic sclerosis with no signs or symptoms of heart failure: an endomyocardial biopsy study. J Card Fail 9:311–317
Plazak W, Zabinska-Plazak E, Wojas-Pelc A, Podolec P, Olszowska M, Tracz W et al (2002) Heart structure and function in systemic sclerosis. Eur J Dermatology 12(3):257–262
Bustamante-Labarta M, Perrone S, De La Fuente RL, Stutzbach P, De La Hoz RP, Torino A et al (2002) Right atrial size and tricuspid regurgitation severity predict mortality or transplantation in primary pulmonary hypertension. J Am Soc Echocardiogr 15(10 Pt 2):1160–1164
Raymond RJ, Hinderliter AL, Willis PW, Ralph D, Caldwell EJ, Williams W et al (2002) Echocardiographic predictors of adverse outcomes in primary pulmonary hypertension. J Am Coll Cardiol 39(7):1214–1219
Grapsa J, Gibbs JS, Cabrita IZ, Watson GF, Pavlopoulos H, Dawson D et al (2012) The association of clinical outcome with right atrial and ventricular remodelling in patients with pulmonary arterial hypertension: study with real-time three-dimensional echocardiography. Eur Heart J Cardiovasc Imaging 13(8):666–672
Gaynor SL, Maniar HS, Prasad SM, Steendijk P, Moon MR (2005) Reservoir and conduit function of right atrium: impact on right ventricular filling and cardiac output. Am J Physiol Heart Circ Physiol 288(5):H2140–H2145
Gaynor SL, Maniar HS, Bloch JB, Steendijk P, Moon MR (2005) Right atrial and ventricular adaptation to chronic right ventricular pressure overload. Circulation 112(9 Suppl):I212–I218
D’Andrea A, Stisi S, Bellissimo S, Vigorito F, Scotto Di Uccio F, Tozzi N et al (2005) Early impairment of myocardial function in systemic sclerosis: non-invasive assessment by Doppler myocardial and strain rate imaging. Eur J Echocardiogr 6(6):407–418
D’Andrea A, Caso P, Cuomo S, Scotto Di Uccio F, Scarafile R, Salerno G et al (2007) Myocardial and vascular dysfunction in systemic sclerosis: the potential role of noninvasive assessment in asymptomatic patients. Int J Cardiol 121(3):298–301
D’Andrea A, Caso P, Galderisi M, Di Maggio D, Cicala S, D’Andrea L et al (2001) Assessment of myocardial response to physical exercise in endurance competitive athletes by pulsed Doppler tissue imaging. Am J Cardiol 87:1226–1230
Ancona R, Comenale Pinto S, Caso P, D’Andrea A, Di Salvo G, Arenga F et al (2014) Left atrium by echocardiography in clinical practice: from conventional methods to new echocardiographic techniques. Sci World J 2014:451042
D’Andrea A, Riegler L, Golia E, Cocchia R, Scarafile R, Salerno G et al (2013) Range of right heart measurements in top-level athletes: the training impact. Int J Cardiol 164(1):48–57
van den Hoogen F, Khanna D, Fransen J, Johnson SR, Baron M, Tyndall A et al (2013) 2013 classification criteria for systemic sclerosis: an American College of Rheumatology/European League against Rheumatism collaborative initiative. Arthritis Rheum 65(11):2737–2747
Grünig E, Henn P, D’Andrea A, Claussen M, Ehlken N, Maier F et al (2013) Reference values for and determinants of right atrial area in healthy adults by 2-dimensional echocardiography. Cardiovasc Imaging 6(1):117–124
D’Andrea A, Caso P, Romano S, Scarafile R, Riegler L, Salerno G et al (2007) Different effects of cardiac resynchronization therapy on left atrial function in patients with either idiopathic or ischaemic dilated cardiomyopathy: a two-dimensional speckle strain study. Eur Heart J 28(22):2738–2748
D’Alto M, Ghio S, D’Andrea A, Pazzano AS, Argiento P, Camporotondo R et al (2011) Inappropriate exercise-induced increase in pulmonary artery pressure in patients with systemic sclerosis. Heart 97(3):112–117
Lindqvist P, Caidahl K, Neuman-Andersen G, Ozolins C, Rantapää-Dahlqvist S, Waldenström A et al (2005) Disturbed right ventricular diastolic function in patients with systemic sclerosis. A Doppler tissue imaging study. Chest 128(2):755–763
Durmus E, Sunbul M, Tigen K, Kivrak T, Ozen G, Sari I et al (2014) Right ventricular and atrial functions in systemic sclerosis patients without pulmonary hypertension: speckle-tracking echocardiographic study. Herz 40(4):709–715
Hirooka K, Naito J, Koretsune Y, Irino H, Abe H, Ichikawa M et al (2003) Analysis of transmural trends in myocardial integrated backscatter in patients with progressive systemic sclerosis. J Am Soc Echocardiogr 16(4):340–346
Sulli A, Ghio M, Bezante GP, Deferrari L, Craviotto C, Sebastiani V et al (2004) Blunted coronary flow reserve in systemic sclerosis. Rheumathology (Oxford) 43(4):505–509
Ojaghi Haghigi Z, Naderi N, Amin A, Taghavi S, Sadeghi M, Moladoust H et al (2011) Quantitative assessment of right atrial function by strain and strain rate imaging in patients with heart failure. Acta Cardiol 66:737–742
D’Andrea A, Scarafilie R, Riegler L, Salerno G, Gravino R, Cocchia R et al (2009) Right atrial size and deformation in patients with dilated cardiomyopathy undergoing cardiac resynchronization therapy. Eur J Heart Fail 11:1169–1177
Sivak J, Raina A, Forfia P (2011) Assessing the physiologic contribution of right atrial function to total right ventricular function in patients with and without pulmonary arterial hypertension. Am J Respir Crit Care Med 183:A4999, Abstract
Nagel C, Henn P, Ehlken N, D’Andrea A, Blank N, Bossone E et al (2015) Stress-Doppler-echocardiography for early detection of systemic sclerosis associated pulmonary arterial hypertension. Arthritis Res Ther 17(1):165
Liang HY, Cauduro S, Pellikka P, Wang J, Urheim S, Yang EH et al (2006) Usefulness of two-dimensional speckle strain for evaluation of left ventricular diastolic deformation in patients with coronary artery disease. Am J Cardiol 98(12):1581–1586
Cho GY, Chan J, Leano R, Strudwick M, Marwick TH (2006) Comparison of two-dimensional speckle and tissue velocity based strain and validation with harmonic phase magnetic resonance imaging. Am J Cardiol 97(11):1661–1666
Ashraf M, Li XK, Young MT, Jensen AJ, Pemberton J, Hui L et al (2006) Delineation of cardiac twist by a sonographically based 2-dimensional strain analysis method: an in vitro validation study. J Ultrasound Med 25:1193–1198
Di Salvo G, Caso P, Lo Piccolo R, Fusco A, Martiniello AR, Russo MG et al (2005) Atrial myocardial deformation properties predict maintenance of sinus rhythm after external cardioversion of recent-onset lone atrial fibrillation: a color Doppler myocardial imaging and transthoracic and transesophageal echocardiographic study. Circulation 112(3):387–395
Di Salvo G, Drago M, Pacileo G, Rea A, Carrozza M, Santoro G et al (2005) Atrial function after surgical and percutaneous closure of atrial septal defect: a strain rate imaging study. J Am Soc Echocardiogr 18(9):930–933
Authors’ contributions
Serena Vettori, Emanuele Romeo, Paola Argiento and Marco Di Maio helped to acquire the clinical data and to perform the interpretation of the data. Rosangela Cocchia, Giovanni Maria De Marco, Michele D’Alto and Antonello D’Andrea helped to acquire the echocardiographic data and to perform the off-line interpretation of the data. Nicola Ehlken and Eduardo Bossone performed the statistical analysis and helped to perform the interpretation of the data. Ekkehard Grunig, Raffaele Calabrò, Maria Giovanna Russo and Gabriele Valentini helped to perform the design of the study and the interpretation of the data. All authors have made substantial contributions to conception and design, have been involved in drafting the manuscript and revised it critically for important intellectual content, have given final approval of the version to be published, and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
None.
Rights and permissions
About this article
Cite this article
D’Andrea, A., D’Alto, M., Di Maio, M. et al. Right atrial morphology and function in patients with systemic sclerosis compared to healthy controls: a two-dimensional strain study. Clin Rheumatol 35, 1733–1742 (2016). https://doi.org/10.1007/s10067-016-3279-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-016-3279-9