Purpose
Abstract
A meta-analysis was performed to asses whether antibiotic prophylaxis is effective in reducing the incidence of surgical site infection (SSI) after open mesh repair of groin hernia.
Methods
A literature search for randomized controlled trials (RCT) evaluating the effectiveness of antibiotic prophylaxis in adult patients undergoing open mesh repair of groin hernia was performed in November 2015. Incidence of overall and deep SSI was considered as primary and secondary outcome measures, respectively. Only studies with a clear definition of SSI and a follow-up of at least 1 month were included. Effect size from each RCT was computed as odds ratio (OR) and 95 % confidence interval (CI) and then data were pooled using a random-effects model.
Results
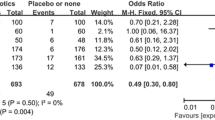
Sixteen RCTs with a total number of 5519 patients were included in the meta-analysis. Considering all the RCTs, antibiotic prophylaxis significantly reduced the overall incidence of SSI from 4.8 % to 3.2 % [OR 0.68, 95 % CI (0.51–0.91)]. However, after removal of two outlier studies, which were identified by evaluating the standardized residual, the result of the meta-analysis became non-significant [OR 0.76, 95 % CI (0.56–1.02)]. The incidence of deep SSI was very low (0–0.7 %) and the effect of antibiotic prophylaxis was not significant [OR 0.80, 95 % CI (0.32–1.99)].
Conclusions
The results of this meta-analysis do not support the routine use of antibiotic prophylaxis for the open mesh repair of groin hernia. In clinical settings with unexpectedly high rates of SSIs, the appropriateness of surgical asepsis should be carefully checked.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Groin hernia repair is traditionally considered a clean wound operation for which antibiotic prophylaxis is not indicated, since the estimated risk of surgical site infection (SSI) is very low (<1 %) [1–3]. However, the true incidence of SSI from groin hernia repair varies widely in the literature, reaching the worrisome rate of 10–13 % in some studies [4–6]. Given the proximity of the groin region to the genitals and perineum, this finding raises the question of whether it would be better to consider this operation as a clean-contaminated procedure, for which antibiotic prophylaxis is mandatory. The majority of the randomised controlled trials (RCT) published on this topic have failed to prove any benefit from the use of antibiotic prophylaxis, although most of them were limited by having small cohorts [4–19]. Conversely, a moderate trend in favour of antibiotic prophylaxis has emerged from the latest meta-analyses, but the data are not sufficiently strong to lead to definitive recommendations [20, 21]. As a result, current guidelines are discordant, some being in favour [22, 23] and others against [24–26] the routine use of antibiotic prophylaxis. Since groin hernia repair is one of the most common surgical procedures worldwide [27], both the inappropriate use of antibiotics and an excessively high rate of SSI are liable to have a major negative impact on health and social costs. Therefore, acquiring stronger evidence on this topic is even more essential. The latest update of the literature dates back to October 2011, and covered 6 and 11 RCTs on patients who underwent, respectively, traditional herniorraphy and mesh repair of groin hernia [21]. The purpose of our study was to perform a systematic review and meta-analysis including further RCTs, in order to gather more evidence on this topic. Since the use of prosthetic devices has become the rule nowadays [26], we focused our study only on mesh repair. An additional objective was to check for the presence of one or more outlier studies (studies whose results differ significantly from the others) and, if found, to verify their influence on the meta-analytic results. Finally, we investigated the extent to which several moderators, such as patient characteristics, surgical skill, duration of surgery, use of drainage and rate of SSI in patients not receiving antibiotics, could influence the correlation, if any, between antibiotic prophylaxis and SSI incidence.
Materials and methods
Eligibility criteria
Characteristics of primary studies
-
Participants Adult patients undergoing elective open inguinal or femoral hernia repair with the use of prosthesis, regardless of the type of anaesthesia and hospital setting (inpatient/outpatient). Studies including patients less than 15 years old were excluded. Studies focused on laparoscopic hernia repair, herniorraphy without the use of prosthesis or emergency hernia repair were also excluded.
-
Intervention any type of antibiotic administrated orally or intravenously before surgery to prevent post-operative wound infections. Studies using topical antibiotic administration were excluded;
-
Comparisons placebo or no treatment;
-
Outcomes (dependent variables) Overall incidence of post-operative SSI detected during a follow-up of at least 30 days. When data were available, incidence of deep post-operative SSI was also evaluated. Only studies with a clear definition of SSI were considered eligible;
-
Study design RCT.
Characteristics of publications
The literature search focused solely on articles published in peer-reviewed journals, to enhance the methodological rigour of the studies examined and the reliability of the conclusions drawn regarding the efficacy of the intervention. No a priori exclusion based on language or year of publication was made.
Search strategy and selection of studies
The literature search was performed in November 2015, using several databases, including PubMed, Scopus, ISI-Web of Science and Google scholar.
For PubMed, the following search strategy was adopted:
#1: antibiotic* OR antimicrob* OR anti-infect* OR anti infect*, #2: prophyla* OR prevent*, #3: #1 AND #2, #4: herni*, #5: #3 AND #4.
For Scopus and ISI-Web of Science, a similar strategy was used by entering the following terms in the field type TITLE-ABS-KEY: (antibiotic* OR antimicrob* OR anti-infect* OR anti infect*) AND (prophyla* OR prevent*) AND herni* AND (repair OR surgery OR tech* OR proced*) AND ((wound infection) OR SSI OR (surgical site infection)).
Google scholar was investigated by searching all the references that included the terms “hernia” AND (“prophyla” OR “prevent” OR “antibiotic” OR “infection”) in the title.
A further search was performed by checking articles in press in the index of journals which published more on this topic, in order to identify relevant studies not yet indexed in literature databases. Finally, a hand-search in the reference lists of the selected articles and of a previous similar review [21] was performed.
This study was performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement [28].
Coding
A coding protocol was prepared and used by the first and the last author to independently extract relevant information from the selected primary studies. Inter-rater reliability was very high (Choen’s kappa = 0.86) and disagreements were resolved through consensus. Six classes of information were coded:
-
1.
Characteristic of the publication First author’s name, year, language and country of publication, number of centres involved, length of the study;
-
2.
Characteristics of the sample Total sample size, male/female ratio, mean age, comorbidities, rate of recurrent hernias;
-
3.
Pre-operative management. Type, dose, mode and timing of antibiotic administration;
-
4.
Characteristics of the operation Type of anaesthesia, type of repair, use of drain, grade of the surgeon (consultant/resident), mean duration of the operation, hospital setting (inpatient/outpatient);
-
5.
Post-operative management Duration and modality of follow-up, criteria used for diagnosis of SSI, management of SSI, rate of mesh removal;
-
6.
Data necessary for effect size computation Number of patients and number of post-operative SSIs in the case and control groups, respectively. The number of deep SSIs was also evaluated when available.
Assessment of the methodological quality of each study was performed according to the criteria published in the Cochrane Handbook for Systematic Reviews of interventions (version 5.1.0) [29].
Statistical analysis
All statistical analyses were performed by means of the meta-analytic software ProMeta 2.0. Initially, we computed effect size as odds ratio (OR) and 95 % confidence interval (CI) from the data reported in each article. Then, data were pooled across studies to obtain an overall effect size using the inverse variance method.
The random-effects model was used as a conservative approach to account for different sources of variation among studies (i.e. within-study variance and between-studies variance). Further, the random-effects model allows for generalization of the meta-analytic findings beyond the studies included in this review [30]. OR was considered statistically significant at the 5 % level if the 95 % CI did not include the value 1. Numbers needed to treat (NNT) with 95 % CI were also calculated for the pooled results according to Altman [31].
To examine heterogeneity across studies, both Q and I 2 statistics were used. A significant Q value indicates the lack of homogeneity of results among studies. I 2 estimates the proportion of observed variance that reflects real differences in effect sizes, with values of 25, 50 and 75 % which might be considered as low, moderate and high, respectively [32].
To further evaluate heterogeneity across studies, an analysis of several moderators was performed. Five continuous moderators, including rate of SSI in the control group, rate of operation performed by residents, duration of surgery, rate of women and mean age of the overall sample, were tested by means of meta-regression analysis, while one categorical moderator, the grade of the surgeon (consultant/resident), was tested through subgroups analysis.
A search for possible outlier studies was performed by evaluating the standardized residual and its statistical significance for each of the selected studies. If two or more outliers were detected, a further meta-analysis was performed after removing those studies, in order to appraise their influence on the overall effect size. Outlier studies were further analysed to detect any qualitative difference from the other studies.
Finally, publication bias analysis was performed. To do so, we initially evaluated the symmetry of the forest plot by ordering the results of primary studies according to sample size. An asymmetrical forest plot, where studies with small samples systematically report significant results, suggests the presence of publication bias. Then, we examined the funnel plot, which is a scatter plot of the effect sizes from selected studies against a measure of their standard error. In the absence of bias, the plot would be shaped as a symmetrical inverted funnel. Egger’s linear regression analysis was employed to statistically test the symmetry of the funnel plot, with non-significant results indicative of absence of publication bias [33]. In addition, the trim and fill method, that is an iterative non-parametric statistical technique, was used to evaluate the effect of potential data censoring on the result of the meta-analysis. The absence of publication bias is indicated by zero trimmed studies or, in the presence of trimmed studies, by trivial differences between the observed and estimated effect sizes [34].
Results
Initial queries identified a total of 1403 articles from all databases and search methods. After duplicates were removed, the subsequent selection process resulted in 16 studies that meet the eligibility criteria of this meta-analysis [4–19] (Fig. 1). The assessment of the methodological quality of each RCT is reported in Fig. 2. As can be seen, only five studies had a low risk of bias in all the seven domains. An unclear risk of bias was found mainly in the domains of selection and performance bias, while high risk of bias was found in the other bias domains in four RCTs.
The main characteristics of the selected studies are reported in Tables 1, 2, 3, 4 and 5. All the articles were published in the period 2000–2015; most of them were written in English with only two published in Spanish [13, 14]. However, the contexts in which the studies had been conducted were heterogeneous, with five studies done in Europe [8, 9, 13, 14, 17] ten in Asia [4–6, 10–12, 15, 16, 18, 19] and one in Africa [7]. None of the articles had been published in North or South America. Most studies were conducted in a single centre and their duration varied from 12 to 55 months. Only three studies demonstrated the effectiveness of antibiotic prophylaxis and were stopped early for ethical reasons [4, 9, 19]. Sample size ranged widely from 98 to 1160, with mean age of participants varying from 37.4 to 70.5 years. The female gender was poorly represented with only 3 studies showing prevalence slightly above 10 % [9, 13, 14]. Patients affected by diabetes or recurrent hernias, which are potential risk factor for SSI, were not excluded in several studies [4, 7, 9, 10, 13, 17] while others did not specify this issue [5, 7, 12–15, 17, 18]. The method of pre-operative groin skin preparation (i.e. shaving and antisepsis) was clearly described in 10 studies [4–7, 9, 10, 15–17, 19], two of which were not in accordance with current recommendations (shaving was performed the day before surgery instead of just before) [6, 16]. No studies specified the methods used for skin shaving (razor/clipper). Antibiotics were always administered intravenously, with first-generation cephalosporin or a combination of beta-lactam/beta-lactamase inhibitors being the most used. Four studies did not clearly specify the timing of antibiotic administration [11, 15, 17, 19] and one did not use a placebo in the control group [14]. Hernia repair was always performed by means of polypropylene mesh, mainly through the Lichtenstein technique, under spinal or general anaesthesia. The use of drains was reported in four studies [7, 8, 17, 19] and was particularly frequent in Yerdel’s study (22.3 %). In eight studies, residents performed a variable number of operations (24–100 %) under the supervision of a consultant surgeon [4, 6, 8–10, 15, 17, 19]. Mean duration of surgery ranged from 34 to 65.7 min. Seven studies reported the length of hospitalization, which varied from less than 24 h (i.e. Day Surgery) to more than 4 days [4, 6, 9, 10, 14, 16, 18]. Only 4 studies had a post-operative follow-up of more than 1 month (3–24 months) [4, 8, 9, 19] and all but one [9] described the follow-up method, which consisted in history-taking and clinical examination.
The overall rate of SSIs was 3.2 % in the case group (range 0–8 %) and 4.8 % in the control group (range 0–12.5 %). This difference was statistically significant, with no evidence of heterogeneity and publication bias (Fig. 3, Table 6). The number needed to treat was 61 (95 % CI 37.2–168.4). The analysis of categorical and continuous moderators showed no significant results (Tables 7, 8). However, meta-regression analysis showed a positive correlation between the incidence rate of SSI in the control group and the effectiveness of antibiotic prophylaxis in the case group, although this finding did not reach statistical significance.
The analysis of standardized residuals (s.r.) allowed the identification of two outlier studies: Mazaki et al. (s.r. = 2.11, p = 0.035) and Yerdel et al. (s.r. = −2.13, p = 0.033). Mazaki et al. considered an elderly population (mean age: 70.5 years) and included patients with diabetes (6.5 %). In this study, residents performed the majority of operations (83.5 %) and the mean length of surgery was the highest recorded among all RCTs (65.7 min). In the study by Yerdel et al., operations were always performed by residents (100 %), drains were frequently used (22.3 %) and mean length of surgery was among the highest reported (63 min). Both studies were discontinued for ethical reasons since the incidence of SSI in the control group was considered to be too high. The meta-analysis performed without these two studies shows no statistical difference between case and control groups in the incidence of post-operative SSI with an NNT of 108 (95 % CI −0.14 to 2.01) (Table 9).
Ten studies reported data about deep SSI [4–6, 8, 10, 11, 14, 15, 18, 19], with an incidence of 0.3 % (range 0–0.7 %) in the case group and 0.5 % in the control group (range 0–0.6 %). This difference was not significant and no heterogeneity or publication bias was found (Fig. 4; Table 6).
Management of SSI was conservative in the majority of cases. Mesh removal was performed in 0.27 % of all hernia repairs, 8.4 % of all SSIs and in 41 % of deep SSIs.
Discussion
The current meta-analysis examined the content of 16 RCTs addressing the use of antibiotic prophylaxis for the open mesh repair of groin hernia. Overall, this study added 5 RCTs and 2286 patients to the latest update published in 2012 [21].
Only 3 of 16 RCTs found significant benefits from the use of antibiotic prophylaxis and were interrupted prematurely due to the very high rate of SSIs in the control group, which varied from 6 % to 13 % [4, 9, 19]. However, the analysis of pooled data showed that prophylaxis significantly reduces the rate of post-operative SSI, from 4.8 % to 3.2 %, with an NNT of 61. These data are apparently stronger and more generalizable than that of the previous meta-analysis because it was obtained with a more conservative method of analysis. Nevertheless, we found several clinical and methodological limitations among the primary studies, which raise some doubts as to the full reliability of this result. Most studies were performed in Asia and Africa, only five in Europe and none in North America. This suggests that the occurrence of SSI after groin hernia repair is not perceived to be a major problem in Western countries. It should be noted that the incidence of SSIs reported in the control group exceeded 5 % in almost all RCTs from Asia and Africa (with 3 studies exceeding 10 %) but only in one study from Europe. Wide heterogeneity was found with regard to several well-recognized risk factors for SSI such as mean age of patients (range 37.4–70.5 years), duration of surgery (34–65.7 min), length of hospital stay (from some hours to 4 days), diabetes (0–18 %), use of drain (0–22.3 %), and repair of recurrent hernias (0–13 %). The description of pre-operative skin preparation was always incomplete and sometimes not in accordance with current recommendations. In some studies, data about the timing of antibiotic administration were missing or imprecise. Finally, the majority of RCTs were affected by one or more methodological bias.
Among all the RCTs included in the study, two were found to have an outlier effect size [4, 19], i.e. a result that differs considerably from the others. This finding could mean that such studies have distinctive characteristics, making them poorly comparable with the other RCTs. In both these studies, the rate of SSI was very high in the control group and several risk factors for post-operative infection, such as diabetes, advanced age, use of drain and long duration of the operation, were present. Although these features were not exclusive of the two studies, their combination might have played a role in determining such results. It is a fact that, when meta-analysis is performed excluding these two RCTs, the effect of antibiotic prophylaxis in reducing SSIs becomes non-significant. The analysis of several moderators did not show any significant correlation with the pooled effect size, although a trend towards positive correlation between effectiveness of antibiotic prophylaxis and the rate of SSIs in the control group was found (Tab.7). This could mean that the higher the rate of SSIs, the more evident is antibiotic effectiveness. Taken together, these findings suggest that antibiotic prophylaxis may be helpful in reducing SSIs, but it seems that this effect becomes evident only in the presence of risk factors or when the incidence of SSIs is high.
The indication for antibiotic prophylaxis depends essentially on two factors: the risk of SSI and the potential severity of the infection, which could have catastrophic outcomes for certain surgical procedures [1, 22, 24]. A survey conducted by the Centers for Disease Control and Prevention (CDC) in more than 1500 US hospitals in the period 2006–2008 found that the risk of SSI in groin hernia procedures is less than 1 %, but may exceed 5 % in the presence of generic risk factors, such as an ASA score greater than 2 and duration of surgery above the 75th percentile [3]. This study does not distinguish between herniorraphy and mesh hernioplasty, although the use of prosthetic devices has become the rule [26] and is a potential risk factor for SSI [1, 22, 24]. However, a meta-analysis of 20 RCTs comparing mesh with non-mesh methods of open groin hernia repair showed no significant difference in the rate of SSI between the groups [35]. In addition, the occurrence of SSI after inguinal hernia repair is not a dramatic event, even when a prosthetic device is used. In fact, antibiotic therapy alone or associated with drainage is adequate to resolve the vast majority of SSIs. We found that the incidence of deep SSI was very low in all the ten studies that provided data on this issue (0–0.7 %) and that the effect of antibiotic prophylaxis was not significant. The occurrence of deep SSI required mesh removal in 41 % of cases, but overall this procedure was performed in only 8.4 % of all SSIs and in 0.27 % of all hernia repairs. The RCTs included in our meta-analysis do not provide data on the incidence of hernia recurrence after mesh removal, but other studies showed that the rate is lower than 5 % and subsequent reoperation is rarely necessary [36, 37]. Based on these considerations, antibiotic prophylaxis should not be indicated for groin hernia repair but, since the incidence of SSI varies widely in the literature, it has been suggested to consider the routine use of antibiotic prophylaxis in those clinical settings that report a high rate of SSIs [20]. However, the effectiveness of this strategy has not been proven and, in the era of clinical governance, it would not seem to be very cost effective. The prevention of SSI requires several pre-, intra- and post-operative steps, among which antibiotic prophylaxis is the only optional one [22, 24]. Any violations while performing these steps can result in contamination and increased risk for SSI. Thus, when the rate of post-operative infections is higher than expected for a clean wound operation, it is best to analyse the appropriateness of all the steps of surgical asepsis before opting for routine antibiotic prophylaxis.
In conclusion, this meta-analysis has shown that antibiotic prophylaxis may be effective in reducing the overall incidence of SSIs after the open mesh hernia repair of groin hernias. However, this result cannot be generalized since it is conditioned by several clinical and methodological limitations of the primary studies and was not confirmed after the exclusion of outlier RCTs. Furthermore, the use of antibiotic prophylaxis is not useful in preventing deep SSI, which is a very uncommon event in groin hernia surgery. The wide heterogeneity in the incidence of SSI reported by the primary studies may be indicative of poor selection of patients or may be the consequence of different quality of surgical asepsis. Hospitals with unexpectedly high rates of SSIs should check for any shortfalls in their aseptic techniques and, where necessary, take corrective measures. Currently, there are no convincing arguments for recommending the routine use of antibiotic prophylaxis for groin hernia repair, especially in clinical settings with low incidence of SSI.
References
Mangram AJ, Horan TC, Pearson ML, Silver LC, Jarvis WR (1999) Guideline for prevention of surgical site infection, 1999. Centers for Disease Control and Prevention (CDC) Hospital Infection Control Practices Advisory Committee. Am J Infect Control 27:97–132
Gaynes RP, Culver DH, Horan TC, Edwards JR, Richards C, Tolson JS (2001) Surgical site infection (SSI) rates in the United States, 1992–1998: the National Nosocomial Infections Surveillance System basic SSI risk index. Clin Infect Dis 33(Suppl 2):S69–S77
Edwards JR, Peterson KD, Mu Y, Banerjee S, Allen-Bridson K, Morrell G, Dudeck MA, Pollock DA, Horan TC (2009) National healthcare safety network (NHSN) report: data summary for 2006 through 2008, issued December 2009. Am J Infect Control 37:783–805
Mazaki T, Mado K, Masuda H, Shiono M, Tochikura N, Kaburagi M (2014) A randomized trial of antibiotic prophylaxis for the prevention of surgical site infection after open mesh-plug hernia repair. Am J Surg 207:476–484
Othman I (2011) Prospective randomized evaluation of prophylactic antibiotic usage in patients undergoing tension free inguinal hernioplasty. Hernia 15:309–313
Shankar VG, Srinivasan K, Sistla SC, Jagdish S (2010) Prophylactic antibiotics in open mesh repair of inguinal hernia: a randomized controlled trial. Int J Surg 8:444–447
Al-Fatah MA (2011) The role of antibiotic prophylaxis in prevention of wound infection after Lichtenstein repair of primary inguinal hernia. http://www.aamj.eg.net/journals/pdf/1355.pdf
Aufenacker TJ, van Geldere D, van Mesdag T, Bossers AN, Dekker B, Scheijde E, van Nieuwenhuizen R, Hiemstra E, Maduro JH, Juttmann JW, Hofstede D, van Der Linden CT, Gouma DJ, Simons MP (2004) The role of antibiotic prophylaxis in prevention of wound infection after Lichtenstein open mesh repair of primary inguinal hernia: a multicenter double-blind randomized controlled trial. Ann Surg 240:955–960
Celdrán A, Frieyro O, de la Pinta JC, Souto JL, Esteban J, Rubio JM, Señarís JF (2004) The role of antibiotic prophylaxis on wound infection after mesh hernia repair under local anesthesia on an ambulatory basis. Hernia 8:20–22
Ergul Z, Akinci M, Ugurlu C, Kulacoglu H, Yilmaz KB (2012) Prophylactic antibiotic use in elective inguinal hernioplasty in a trauma center. Hernia 16:145–151
Jain SK, Jayant M, Norbu C (2008) The role of antibiotic prophylaxis in mesh repair of primary inguinal hernias using prolene hernia system: a randomized prospective double-blind control trial. Trop Doct 38:80–82
Kochhar G, Andley M, Saha S, Kumar A, Pusuluri R, Saurabh G, A Kumar (2014) Randomized controlled trial to evaluate the role of antibiotic prophylaxis in lichtenstein hernia repair. https://www.researchgate.net/publication/268484354
Morales R, Carmona A, Pagán A, García Menéndez C, Bravo R, Hernández MJ, Llobera J (2000) Utilidad de la profilaxis antibiótica en la reducción de la infección de herida en la reparación de la hernia inguinal o crural mediante malla de polipropileno. Cir Esp 67:51–59
Oteiza F, Ciga MA, Ortiz H (2004) Profilaxis antibiótica en la hernioplastia inguinal. Cir Esp 75:69–71
Perez AR, Roxas MF, Hilvano SS (2005) A randomized, double-blind, placebo-controlled trial to determine effectiveness of antibiotic prophylaxis for tension-free mesh herniorraphy. J Am Coll Surg 200:393–397
Razack A, Kapoor KK, Tambat RM (2015) The role of single dose antibiotic prophylaxis in open mesh repair of inguinal hernia: a prospective, double blind randomized trial. J Evol Med Dent Sci 4:6017–6026
Tzovaras G, Delikoukos S, Christodoulides G, Spyridakis M, Mantzos F, Tepetes K, Athanassiou E, Hatzitheofilou C (2007) The role of antibiotic prophylaxis in elective tension-free mesh inguinal hernia repair: results of a single-centre prospective randomised trial. Int J Clin Pract 61:236–239
Wang J, Ji G, Yang Z, Xi M, Wu Y, Zhao P, Wang L, Yu W, Wen A (2013) Prospective randomized, double-blind, placebo controlled trial to evaluate infection prevention in adult patients after tension-free inguinal hernia repair. Int J Clin Pharmacol Ther 51:924–931
Yerdel MA, Akin EB, Dolalan S, Turkcapar AG, Pehlivan M, Gecim IE, Kuterdem E (2001) Effect of single-dose prophylactic ampicillin and sulbactam on wound infection after tension-free inguinal hernia repair with polypropylene mesh: the randomized, double-blind, prospective trial. Ann Surg 233:26–33
Sanabria A, Domínguez LC, Valdivieso E, Gómez G (2007) Prophylactic antibiotics for mesh inguinal hernioplasty: a meta-analysis. Ann Surg 245:392–396
Sanchez-Manuel FJ, Lozano-García J, Seco-Gil JL (2012) Antibiotic prophylaxis for hernia repair. Cochrane Database Syst Rev 2:CD003769
National Collaborating Centre for Women’s and Children’s Health (2008) Surgical site infection: prevention and treatment of surgical site infection. NICE, London. https://www.nice.org.uk/guidance/cg74/evidence/full-guideline-242005933
Bratzler DW, Dellinger EP, Olsen KM, Perl TM, Auwaerter PG, Bolon MK, Fish DN, Napolitano LM, Sawyer RG, Slain D, Steinberg JP, Weinstein RA; American Society of Health-System Pharmacists; Infectious Disease Society of America; Surgical Infection Society; Society for Healthcare Epidemiology of America (2013) Clinical practice guidelines for antimicrobial prophylaxis in surgery. Am J Health Syst Pharm 70:195–283
Scottish Intercollegiate Guidelines Network (SIGN) (2008) Antibiotic prophylaxis in surgery. SIGN, Edinburg (SIGN publication no. 104). http://www.sign.ac.uk/pdf/sign104.pdf
Sistema Nazionale per le Linee Guida (SNLG) (2008) Antibioticoprofilassi perioperatoria nell’adulto. SNLG, Milano (SNLG 17). http://www.snlg-iss.it/cms/files/LG_AntibioticoP_Unico_2008.pdf
Simons MP, Aufenacker T, Bay-Nielsen M, Bouillot JL, Campanelli G, Conze J, de Lange D, Fortelny R, Heikkinen T, Kingsnorth A, Kukleta J, Morales-Conde S, Nordin P, Schumpelick V, Smedberg S, Smietanski M, Weber G, Miserez M (2009) European Hernia Society guidelines on the treatment of inguinal hernia in adult patients. Hernia 13:343–403
Kingsnorth A, LeBlanc K (2003) Hernias: inguinal and incisional. Lancet 362:1561–1571
Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JP, Clarke M, Devereaux PJ, Kleijnen J, Moher D (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ 339:b2700
Higgins JPT, Green S (eds) (2011) Cochrane handbook for systematic reviews of interventions Version 5.1.0 [updated March 2011]. In: The Cochrane Collaboration. http://www.cochrane-handbook.org
Borenstein M, Hedges LV, Higgins JPT, Rothstein HR (2009) Introduction to meta-analysis. Wiley, Chichester
Altman DG (1998) Confidence intervals for the number needed to treat. BMJ 317:1309–1312
Higgins JP, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327:557–560
Egger M, Davey Smith G, Schneider M, Minder C (1997) Bias in meta-analysis detected by a simple, graphical test. BMJ 315:629–634
Duval S, Tweedie R (2000) Trim and fill: a simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics 56:455–463
Scott NW, McCormack K, Graham P, Go PM, Ross SJ, Grant AM (2002) Open mesh versus non-mesh for repair of femoral and inguinal hernia. Cochrane Database Syst Rev 4:CD002197
Fawole AS, Chaparala RP, Ambrose NS (2006) Fate of the inguinal hernia following removal of infected prosthetic mesh. Hernia 10:58–61
Rehman S, Khan S, Pervaiz A, Perry EP (2012) Recurrence of inguinal herniae following removal of infected prosthetic meshes: a review of the literature. Hernia 16:123–126
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
EE, FM, GP, AN, and PGC declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
For this type of study formal consent is not required.
Rights and permissions
About this article
Cite this article
Erdas, E., Medas, F., Pisano, G. et al. Antibiotic prophylaxis for open mesh repair of groin hernia: systematic review and meta-analysis. Hernia 20, 765–776 (2016). https://doi.org/10.1007/s10029-016-1536-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10029-016-1536-0