Abstract
ᅟObjectives
Vitamin-D-dependent rickets type 1A (VDDR1A) is a rare inherited disease caused by defective activation of vitamin D. The aim of the study was to describe the craniofacial characteristics and the dental phenotype of patients with genetically confirmed VDDR1A. The VDDR1A findings were compared to findings in patients with X-linked hypophosphatemia (XLH) and healthy controls.
Material and methods
Ten patients with VDDR1A were identified. The reference group for the comparison of cephalometric findings was 49 adults without chronic disease. The reference group for the comparison of dental findings was 30 adults with XLH. Clinical examination, clinical photos, and radiographs were obtained. Cephalometric analysis was performed. Photos and radiographs were visually evaluated.
Results
The depth of the posterior cranial fossa (d-p and d-s-iop) in VDDR1A adults was reduced compared to the reference group (p < 0.05). Five (83%) of six adults with VDDR1A and one (4%) of 25 adults with XLH had enamel hypoplasia on several incisors and/or canines (p < 0.001). Three (75%) of four adults with VDDR1A and none of 16 adults with XLH had several first molars with enamel hypoplasia (p = 0.004). Five of 7 (71%) adults with VDDR1A and 24 of 30 (80%) adults with XLH had endodontically affected teeth.
Conclusions
The dental aberration of VDDR1A is more in line with the dental aberration of nutritional rickets than with the dental aberrations in XLH, suggesting the combination of low availability of both calcium and phosphate to be critical in periods of enamel formation.
Clinical relevance
Knowledge on craniofacial and dental aberration in patients with rare diseases, e.g., inherited rickets, is of importance to the dental practitioner, especially during diagnostics and treatment in special care units.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
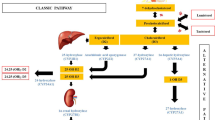
Vitamin-D-dependent rickets type 1A (VDDR1A) is a rare inherited disease with the development of rickets during the first 1–2 years of living, caused by a defective activation of vitamin D. The skeletal symptoms are in line with the symptoms of other inherited or acquired types of rickets, but the dental affection of the disease has only briefly been characterized based on a limited number of patients [1, 2]. The prevalence of VDDR1A has been estimated to be 0.4 per 100,000, which is low compared to the prevalence of X-linked dominant hypophosphatemia (XLH) (4.8 per 100,000) [3]. The lower prevalence is in part due to VDDR1A being an autosomal recessive disorder caused by a mutation of CYP27B1 (OMIM 609506), which encodes for the enzyme 1-alfa-hydroxylase. The gene is located at chromosome 12q, and previous studies have described different locations of mutations in the gene [4,5,6]. The 1-alfa-hydroxylase enzyme is primarily present in the kidneys, where it activates vitamin D (cholecalciferol, 25-hydroxyvitamin D) into the active metabolite calcitriol (1,25-dihydroxyvitamin D). Biochemical characteristics of VDDR1A are low calcitriol, hypocalcemia, and hypophosphatemia with elevated parathyroid hormone and alkaline phosphatase [7, 8]. Replacement treatment with alfacalcidol heals the rickets and normalizes the biochemistry [4, 9, 10]. Apparently, the craniofacial structures in patients with VDDR1A have not previously been characterized. The previously reported dental manifestations of VDDR1A include linear enamel hypoplasia in the permanent dentition, and a link to the hypocalcemia has been suggested [2, 11]. Furthermore, interglobular dentin, large pulp chambers and pulp horns, loss of lamina dura, and premature loss of primary teeth have been reported [2].
Recently, a phenotype study of XLH was performed, including dental and craniofacial characteristics of HR [12]. XLH is caused by a mutation in the phosphate-regulating endopeptidase gene, PHEX [13]. By a still not fully unraveled pathway, this mutation leads to a defect in the bone mineralization and an increase in the phosphaturetic hormone fibroblast growth factor 23 (FGF23), causing renal phosphate wasting and thereby a low serum value of phosphate. Both of the diseases VDDR1A and XLH share the development of rickets within the first 1–2 years of living and of hypophosphatemia. They differ, though, in the disease mechanism and in terms of low calcium levels in VDDR1A as opposed to normal calcium levels in XLH [14]. The dentition in XLH was characterized by a high number of endodontically affected permanent teeth [15,16,17]. The cranium was characterized by a flattened cranial base, a decreased depth of the posterior cranial base, an increased thickness of the skull, and a reduced length of the nasal bone [18].
The primary aim of the present study was to describe the craniofacial characteristics and the dental phenotype of patients with genetically confirmed VDDR1A. Secondly, the aim was to compare craniofacial characteristics between VDDR1A, XLH, and healthy controls. Thirdly, the dental findings in VDDR1A were compared to the dental findings in XLH.
Materials and methods
Study population
The patients were recruited in a large medical study of rickets patients in Denmark, where patients were identified by diagnosis codes of rickets in the Danish National Patient Registry, by contact to treating doctors, and by family screening. The recruitment has previously been described in detail [14]. At that time, seven adults with VDDR1A were identified. One of them was not participating in the dental examination, an edentulous female. In the period from 2008 to 2014, an additional three children and one adult have been included. Thus, a total of ten patients with VDDR1A were included for the present study.
The diagnostic criteria were clinical rickets, in addition to biochemically and genetically confirmed VDDR1A. Biochemical criteria of VDDR1A at the time of diagnosis were the following parameters: serum 1,25-dihydroxyvitamin D below the normal range, 25-hydroxyvitamin D within the normal range, hypocalcemia, hypophosphatemia, elevated alkaline phosphatase, and elevated parathyroid hormone [19]. The genetic criterion was the identification of a mutation in the two alleles of CYP27B1.
The reference group to be used for the comparison of the dental description of the VDDR1A patients is the population of 52 patients with XLH previously reported on [15]. The reference groups for the craniofacial description of the VDDR1A patients are the previously reported population of 53 patients with XLH (36 adults) and 79 healthy controls (49 adults) [18].
Genetic examination
Genomic DNA was extracted from full blood using a DNA purification robot (Maxwell Promega, Ramcon, Denmark). All DNA samples were sequenced in both directions using the BigDye Terminator v3.1 Cycle Sequencing Kit (Applied Biosystems, Foster City, CA, USA) and analyzed on a 3730XL DNA Analyzer (Applied Biosystems, Foster City, CA, USA). Sequence analysis was performed using SeqMan Software (DNA STAR, Madison, WI, USA).
Cephalometric examination
Standardized profile radiographs were obtained as described by Solow [20], using the digital radiographic equipment, Planmeca Promax© (Planmeca Oy, Helsinki, Finland). The sensor-focus distance was 1.50 m and the enlargement factor was 1.13. During exposure, the head of the patient was fixed in a rigid cephalostat, and the patients were instructed to keep the teeth in occlusion. The head posture was adjusted to the best fit of the borders of the sensor.
The analysis of the radiographs was performed using software for cephalometric analysis, Pordios® (Institute of Orthodontic Computer Science, Aarhus, Denmark). Fifteen cephalometric landmarks were digitized (Fig. 1). The digitizing of all radiographs was performed by the first author. Seven angular and six linear variables were calculated by the software Pordios® (Table 1). The variables of the cranial base and the facial skeleton were defined according to the definitions by Bjørk [21] and Solow [20]. The variables of the neurocranium were defined as described by Axelsson and coworkers [22] (thickness of the theca) and by Caspersen and coworkers [23] (the morphology of the posterior cranial fossa). Previously, the intraexaminer reliability of the cephalometric measurements has been performed on 22 randomly selected radiographs, which were digitized twice by the first author. No systematic error was found. The coefficient of reliability was acceptable [18]. Briefly, the coefficient of reliability was estimated according to Houston [24] and ranged from 0.91 to 1.00.
The cephalometric variables in adults with VDDR1A were compared with healthy controls by nonparametric methods (Mann-Whitney rank-sum test). Furthermore, cephalometric variables in adults with XLH were compared with the healthy controls by regression analysis, adjusted for the effect of gender and age.
Dental clinical examination
All patients were given a clinical and a radiographic dental examination at the School of Dentistry, Aarhus University or at the Centre of Oral Health in Rare Diseases at Aarhus University Hospital from 2007 to 2014. Clinical photos by professional photographers and impression for digital study models were taken as part of the clinical examination. Using the clinical photos, the following dental clinical traits were assessed: (1) the number of permanent teeth present; (2) the presence of enamel hypoplasia in a minimum of two teeth in the incisor-canine region (IC region), (3) in the first molar region (M1 region), or (4) in the premolar-second-molar region (PM2 region); (5) the number of extracted first molars; the number of full coverage (FC) prosthetic crowns (6) in the IC region (a maximum of 12 teeth) or (7) in the M1 region (a maximum of 4 teeth); (8) the number of teeth in the IC region with composite filling at the labial or incisal surfaces (a maximum of 12 teeth); (9) the presence of a horizontal overjet exceeding 6 mm; (10) the presence of a vertical overbite exceeding 5 mm; and (11) the presence of lateral crossbite. All patients with VDDR1A and adults from the reference group (XLH) were assessed.
The presence of enamel hypoplasia in the respective dental groups (IC, M1, PM2) in adult patients with VDDR1A were compared with the XLH reference group by Fischer’s exact test. The XLH reference group was adult patients with XLH with a minimum of 20 teeth present in the mouth.
The presence of dental treatment in terms of coverage (FC, CoFi) or extractions of a minimum of three teeth in the respective dental groups was defined as extensive treatment. The occurrences of extensive treatment in the group of adults with VDDR1A were compared with the occurrences in the XLH reference group by Fischer’s exact test.
Radiographic dental examination
The radiographic examination included a digital panoramic radiograph taken in a ProMax unit (Planmeca, Helsinki, Finland). Exposure parameters were adjusted to the individual patient. If the image of the permanent teeth was blurred, supplementary digital intraoral radiographs were taken of these teeth using a GX 1000 dental X-ray unit (Gendex, Des Plaines, IL, USA) with rectangular collimation. We used a sensor (Sidexis, Sirona, Bensheim, Germany) for anterior teeth and a photo-stimulable phosphor plate (Digora, FMX scanner, Soredex, Helsinki, Finland) for posterior teeth. Receptor holders were used for all intraoral radiographs. The panoramic images were assessed in the Dimaxis Pro software (version 4.2.0), whereas the intraoral radiographs were assessed using a general software with facilities to adjust brightness, contrast, gamma curve, and magnification. All image enhancement facilities could be used as wanted.
The radiographs were examined for dental agenesis, taurodontism, and endodontically treated permanent teeth and periapical radiolucency (apical periodontitis). The criteria for the assessment of the endodontic treatment were the presence of radiopaque root filling material in the root channel or radiopaque retrograde root filling material at the apex of the tooth. A diagnosis of apical periodontitis was based on a clear, radiolucent lesion surrounding the apex of the tooth. This will roughly correspond to a periapical index (PAI) score of 3, 4, or 5 [25].
Results
The ten patients with VDDR1A included in the study were characterized according to gender, age, and age at the time of the initiation of the medical treatment (Table 2). In all patients, mutation in both alleles of CYP27B1 was identified.
Cephalometric characteristics
In two patients with VDDR1A, profile radiographs were not obtained (patients 1 and 4). The cephalometric values of the remaining eight patients are presented in Table 3 with the means and confidence intervals (95% CI). In the statistical analyses, the six adults with VDDR1A were compared with the adult controls as reference (Table 3). Five of the six adults with VDDR1A had a reduced mandibular prognathism (s-n-pg), and four of the five adult women tended toward having a shorter nasal-bone-length compared to the means of the control group; however, the differences were not significant. The measurements of the depth of the posterior cranial fossa (d-p and d-s-iop) were recordable in all adults with VDDR1A, and they all had lower values than the means of the gender-matched control group (p < 0.05). According to the statistical analysis, the occipital theca in adults with VDDR1A was thick compared with healthy adults, and the depth of the posterior cranial fossa was decreased (Fig. 2). The comparison between adults with XLH and healthy adults revealed significantly increased thickness of all parts of theca, an increased cranial base angle, and a low posterior cranial fossa.
Dental manifestations
The clinical and radiographic dental pictures of patients with VDDR1A are demonstrated in Figs. 3, 4, and 5. The findings are summarized in Table 4. Dental agenesis was present in one patient (one premolar) and taurodontism (one to two molars) in four patients (two inapplicable). Enamel hypoplasia was present in the incisor-canine and first-molar region of all patients, except for the inapplicable patients having the enamel surfaces covered by veneers or crowns. Oppositely, no premolars or second molars had enamel hypoplasia. Two patients have had extensive prosthetic treatment on incisors and canines, and one of them had additional teeth treated with composite filling on the labial surfaces. Another three patients have had composite filling on the labial surfaces of incisors or canines (three to nine teeth). Four adults (patients 5, 8, 9, and 10) have experienced first molar extraction (one to four teeth). Three of the four individuals with first molar extraction had started alfacalcidol treatment after the enamel formation of both incisors and the first molar were to be completed (at the age of 6, 40, and 33 years). Hence, these patients were not rescued by early treatment. Most likely, enamel hypoplasia was the main reason for the extractions. In exception, patient 5 starting alfacalcidol at the age of 2 years also experienced first molar extraction, and the reason for this is unexplained. One child (patient 3) has had steel crowns cemented on all first molars. Enamel hypoplasia is known to be the reason for this treatment, and the status of the molars before treatment is illustrated in Fig. 5.
In comparison with HR reference group, the adults with VDDR1A were more affected by enamel hypoplasia in the IC region and in the M1 region. In addition, patients with VDDR1A had more teeth with composite coverage in the IC region (Table 5).
An endodontic affection was revealed in five of the seven adult patients with VDDR1 (71%): the two affected teeth in patient 9 and one of the two affected teeth in patient 7 had periapical radiolucency without root filling. Six of the ten endodontically affected teeth in patient 10 and all seven endodontically affected teeth in patient 5 have had previous prosthetic treatment (artificial crowns). In the reference group of adults with XLH and a minimum of 20 teeth, endodontic affection was revealed in 24 of 30 patients (80%): 1–4 endodontically affected teeth in eight patients, 5–8 teeth in eight patients, 9–12 teeth in three patients, and 13–16 teeth in five patients (median number of endodontically affected teeth was five). The number of endodontically affected teeth with an artificial crown ranged from zero (14 patients with 1–15 endodontically affected teeth) to eight (one patient with 13 endodontically affected teeth). In the XLH group, the median number of endodontically affected teeth with an artificial crown was zero.
Discussion
The present study on patients with genetically verified VDDR1A demonstrated dental aberrations in the form of enamel hypoplasia in permanent incisors and first molars, which are aberrations not found in patients with XLH.
According to the present study, the cephalometric characteristics of patients with VDDR1A do not markedly deviate from healthy controls, and the present deviations are in line with the deviations found in patients with XLH [18]. In patients with VDDR1A, the significant deviations from healthy controls were restricted to the bony structures of the posterior cranial fossa. The variation in the flexion of the cranial base varied considerably, and the prognathism of the jaws was close to normal. However, the number of the assessed adults with VDDR1A is limited to six, and definitive conclusions on the cephalometric characteristics of patients with VDDR1A cannot be drawn due to small sample size.
Focusing teeth in patients with VDDR1A, the study demonstrated enamel hypoplasia in the first teeth to develop in the permanent dentition, being incisors, canines, and first molars. In contrast, the premolars and the second molars were unaffected in all patients (Table 4). This difference in enamel affection according to tooth type indicates “the time window” at which the formation of enamel was disrupted by too little available calcium and phosphate from intestinal absorption caused by the defect activation of vitamin D. The affected incisors, canines, and first molars have onset of enamel formation within the first year of postnatal life, and their crown and enamel formation is completed at 5–7 years of age, with canines and maxillary incisors being the latest [26]. The enamel hypoplasia in VDDR1A was mainly present at the incisal part of the dental crown with varying degrees of gingival extension of the affection, and the affection was in some of the patients rather discrete. The enamel formation of the premolars as well as the second molars is initiated approximately at the age of 3 years, where the youngest patients (patients 1 to 7) had started treatment with alfacalcidol. The treatment has most likely normalized the intestinal calcium and phosphate absorption and rescued the enamel formation of the premolars and second molars [26]. Thus, the background of the normalization of the amelogenesis of premolars and second molars supposedly is the initiation of the replacement treatment by alfacalcidol. The assessment of enamel hypoplasia was difficult in the grown-up patients because of dental decay or treatment, which masked the dental surfaces. In three adult patients (patients 5, 7, and 10), all labial surfaces of maxillary incisors and canines were covered by artificial crowns or composite veneers (Table 4, Fig. 3). In addition, two of these patients (patients 5 and 10) had all first molars extracted. This kind of extensive treatment indicates that the first molars developing at the same time period as the incisors might have been affected by severe enamel hypoplasia as well. In general, the enamel hypoplasia in the present VDDR1A population was less severe than previously described in a patient report on one patient with VDDR1A, which was clinically, biochemically, and radiologically, but not genetically verified [1]. The difference might be explained by the time of starting adequate active vitamin D replacement therapy with alfacalcidol, which in the previously reported patient was late (10 years of age). In comparison, the majority of the VDDR1A patients in this study started alfacalcidol treatment early, between the age of 1.0 and 2.2 years: one patient at the age of 6 years, whereas two patients had delayed treatment start at the age of 33 and 40 years, respectively. In Denmark, alfacalcidol was launched in 1978, and before this, patients were treated with high doses of vitamin D (cholecalciferol), thus explaining the delay in alfacalcidol treatment.
In the present study, the group of adults with XLH was less severely affected by enamel hypoplasia in the IC group and M1 group than the adults with VDDR1A. Furthermore and in comparison with VDDR1A, relatively few adults with XLH had experienced extensive first molar extraction and FC treatment in the IC group although the difference did not reach the level of significance (Table 5). Accordingly, enamel hypoplasia does not seem to be a predominant symptom in patients with XLH. A recent microCT study on both deciduous and permanent teeth of patients with XLH focused on the mineralization pattern of the enamel and dentine in comparison with teeth from unaffected family members [27]. The study concluded that there were no deviations in the enamel, in line with the clinical findings in patients with XLH of the present study. In addition, the dentin of the affected patients with XLH revealed a different mineralization pattern [27]. The mineralization pattern was characterized by dentinal abnormalities in terms of porosities and lower mineral density next to the pulp. Previous radiological and histological studies on the dentition of patients with XLH have revealed similar findings in terms of extended dimension of the pulp (large pulp horns), irregular interglobular dentine, and dentinal areas with hypomineralized calcospherites [28,29,30,31].
In the present group of patients with VDDR1A, five adults had endodontically affected teeth of various tooth types (two to ten teeth; Table 4). However, a great number of the endodontically affected teeth had artificial crowns, and the preparation for a dental crown is a well-known risk factor in terms of pulp necrosis. Thus, based on the available data, it is not possible to conclude if the endodontic treatment in the patients with VDDR1A was primary due to, e.g., enamel hypoplasia or dentinal abnormalities in general or a treatment arising secondary to previous dental treatment. The presence of many endodontically affected teeth has previously been described in patients with XLH. Especially in the younger age groups, the percentage of endodontically affected teeth anterior in the mandible was high [15].
Enamel hypoplasia is also seen in nutritional rickets [32, 33], and the extent of the dental aberration is related to the period elapsed with insufficient vitamin D causing low availability of calcium and phosphate during the development of the dentition: The longer period the malnutrition remains, the more teeth are affected. The age of the child, when suffering from nutritional rickets, determines the stage of the development of the teeth, and thus, the teeth undergoing development during the time when suffering from nutritional rickets will show evidence of disturbed formation of the dental structures. Unlike bone, the mineralized dental tissues is not undergoing remodeling, and the distribution of the enamel hypoplasia is thereby a permanent marker of the period with low vitamin D and thereby low levels of calcium and phosphate for mineralization of the dental hard tissues, e.g., enamel [32, 34,35,36]. Like patients with nutritive rickets, patients with VDDR1A are characterized by low calcitriol, hypocalcemia, and hypophosphatemia [7, 8]. In contrast, patients with XLH are hypophosphatemic, but normocalcemic, and enamel hypoplasia was not a dominant symptom in the patients with XLH of the present study. Thus, it is likely that the combination of hypocalcemia and hypophosphatemia has implications for the development of enamel hypoplasias, which was also suggested in the study by Nikiforuk and Fraser [2]. Previously, the role of hypocalcemia in enamel formation has been discussed in relation to hypocalcemia caused by 22q11 deletion syndrome [37, 38], or other medical conditions [39], and in addition in an animal study [40]. However, the causal factors remain nondetermined, and other types of experimental studies are requested.
The strength of the present study was the combined dental and craniofacial assessment of a relatively large group of patients with an extremely rare diagnosis, VDDR1A, which in all patients was genetically verified. We had no access to the previous dental reports. Therefore, the determination of whether the endodontic treatments in the VDDR1A group were primary or secondary to the preparation for the artificial crowns remains unknown. Furthermore, specimens for histological analyses of the dental tissues were not available in the study.
In conclusion, the craniofacial deviations in patients with VDDR1A are discrete (a reduced depth of the posterior cranial fossa) and in line with the deviations found in patients with the predominant inherited types of rickets, XLH. The dental aberration of VDDR1A is more in line with the dental aberration of nutritional rickets than with the dental aberrations of XLH, suggesting low availability of both calcium and phosphate being critical in periods of enamel formation.
References
Zambrano M, Nikitakis NG, Sanchez-Quevedo MC, Sauk JJ, Sedano H, Rivera H (2003) Oral and dental manifestations of vitamin D-dependent rickets type I: report of a pediatric case. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 95(6):705–709. doi:10.1067/moe.2003.116
Nikiforuk G, Fraser D (1981) The etiology of enamel hypoplasia: a unifying concept. J Pediatr 98(6):888–893
Beck-Nielsen SS, Brock-Jacobsen B, Gram J, Brixen K, Jensen TK (2009) Incidence and prevalence of nutritional and hereditary rickets in southern Denmark. Eur J Endocrinol 160:491–497
Kitanaka S, Takeyama K, Murayama A, Sato T, Okumura K, Nogami M, Hasegawa Y, Niimi H, Yanagisawa J, Tanaka T, Kato S (1998) Inactivating mutations in the 25-hydroxyvitamin D3 1alpha-hydroxylase gene in patients with pseudovitamin D-deficiency rickets. N Engl J Med 338(10):653–661. doi:10.1056/NEJM199803053381004
Wang X, Zhang MY, Miller WL, Portale AA (2002) Novel gene mutations in patients with 1alpha-hydroxylase deficiency that confer partial enzyme activity in vitro. J Clin Endocrinol Metab 87(6):2424–2430. doi:10.1210/jcem.87.6.8534
Kim CJ, Kaplan LE, Perwad F, Huang N, Sharma A, Choi Y, Miller WL, Portale AA (2007) Vitamin D 1alpha-hydroxylase gene mutations in patients with 1alpha-hydroxylase deficiency. J Clin Endocrinol Metab 92(8):3177–3182. doi:10.1210/jc.2006-2664
Durmaz E et al (2012) Clinical and genetic analysis of patients with vitamin D-dependent rickets type 1A. Clin Endocrinol (Oxf) 77(3):363–369. doi:10.1111/j.1365-2265.2012.04394.x
Kim CJ (2011) Vitamin D dependent rickets type I. Korean J Pediatr 54(2):51–54. doi:10.3345/kjp.2011.54.2.51
Delvin EE, Glorieux FH (1981) Serum 1,25-dihydroxyvitamin D concentration in hypophosphatemic vitamin D-resistant rickets. Calcif Tissue Int 33(2):173–175
Kato S, Yanagisawa J, Murayama A, Kitanaka S, Takeyama K (1998) The importance of 25-hydroxyvitamin D3 1 alpha-hydroxylase gene in vitamin D-dependent rickets. Curr Opin Nephrol Hypertens 7(4):377–383
Nikiforuk G, Fraser D (1979) Chemical determinants of enamel hypoplasia in children with disorders of calcium and phosphate homeostasis. J Dent Res 58(Spec Issue B):1014–1015
Beck-Nielsen SS, Brusgaard K, Rasmussen LM, Brixen K, Brock-Jacobsen B, Poulsen MR, Vestergaard P, Ralston SH, Albagha OM, Poulsen S, Haubek D, Gjorup H, Hintze H, Andersen MG, Heickendorff L, Hjelmborg J, Gram J (2010) Phenotype presentation of hypophosphatemic rickets in adults. Calcif Tissue Int 87:108–119
The HYP C (1995) A gene (PEX) with homologies to endopeptidases is mutated in patients with X-linked hypophosphatemic rickets. The HYP Consortium. Nat Genet 11(2):130–136
Beck-Nielsen SS (2012) Rickets in Denmark. Dan Med J 59(2):B4384
Andersen MG, Beck-Nielsen SS, Haubek D, Hintze H, Gjorup H, Poulsen S (2012) Periapical and endodontic status of permanent teeth in patients with hypophosphatemic rickets. J Oral Rehabil 39(2):144–150
Chaussain-Miller C, Sinding C, Wolikow M, Lasfargues JJ, Godeau G, Garabedian M (2003) Dental abnormalities in patients with familial hypophosphatemic vitamin D-resistant rickets: prevention by early treatment with 1-hydroxyvitamin D. J Pediatr 142(3):324–331
Opsahl Vital S, Gaucher C, Bardet C, Rowe PS, George A, Linglart A, Chaussain C (2012) Tooth dentin defects reflect genetic disorders affecting bone mineralization. Bone 50(4):989–997. doi:10.1016/j.bone.2012.01.010
Gjørup H, Kjaer I, Sonnesen L, Haubek D, Beck-Nielsen SS, Hintze H, Poulsen S (2011) Craniofacial morphology in patients with hypophosphatemic rickets: a cephalometric study focusing on differences between bone of cartilaginous and intramembranous origin. Am J Med Genet A 155A:2654–2660
Beck-Nielsen SS, Brixen K, Gram J, Brusgaard K (2012) Mutational analysis of PHEX, FGF23, DMP1, SLC34A3 and CLCN5 in patients with hypophosphatemic rickets. J Hum Genet 57(7):453–458
Solow B (1966) The pattern of craniofacial associations. Department of Orthodontics, The Royal Dental College, Copenhagen
Bjørk A (1975) Kæbernes relation til det øvrige kranium. In: Lundström I (ed) Nordisk Lärobok i Ortodonti. Sveriges Tandläkarförbunds Förlagsförening, Stockholm, pp 69–110
Axelsson S, Kjaer I, Bjornland T, Storhaug K (2003) Longitudinal cephalometric standards for the neurocranium in Norwegians from 6 to 21 years of age. Eur J Orthod 25(2):185–198
Caspersen LM, Kjaer I, Sonnesen L (2010) How does occipitalization influence the dimensions of the cranium? Orthod Craniofac Res 13:162–168
Houston WJB (1983) The analysis of errors in orthodontic measurements. Am J Orthod 83:382–390
Orstavik D, Kerekes K, Eriksen HM (1986) The periapical index: a scoring system for radiographic assessment of apical periodontitis. Endod Dent Traumatol 2(1):20–34
AlQahtani SJ, Hector MP, Liversidge HM (2010) Brief communication: The London atlas of human tooth development and eruption. Am J Phys Anthropol 142(3):481–490. doi:10.1002/ajpa.21258
Ribeiro TR, Costa FW, Soares EC, Williams JR Jr, Fonteles CS (2015) Enamel and dentin mineralization in familial hypophosphatemic rickets: a micro-CT study. Dento Maxillo Facial Radiol 44(5):20140347. doi:10.1259/dmfr.20140347
Boukpessi T, Septier D, Bagga S, Garabedian M, Goldberg M, Chaussain-Miller C (2006) Dentin alteration of deciduous teeth in human hypophosphatemic rickets. Calcif Tissue Int 79(5):294–300. doi:10.1007/s00223-006-0182-4
Chaussain-Miller C, Sinding C, Septier D, Wolikow M, Goldberg M, Garabedian M (2007) Dentin structure in familial hypophosphatemic rickets: benefits of vitamin D and phosphate treatment. Oral Dis 13(5):482–489. doi:10.1111/j.1601-0825.2006.01326.x
Murayama T, Iwatsubo R, Akiyama S, Amano A, Morisaki I (2000) Familial hypophosphatemic vitamin D-resistant rickets: dental findings and histologic study of teeth. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 90(3):310–316
Abe K, Masatomi Y, Nakajima Y, Shintani S, Moriwaki Y, Sobue S, Ooshima T (1992) The occurrence of interglobular dentin in incisors of hypophosphatemic mice fed a high-calcium and high-phosphate diet. J Dent Res 71(3):478–483
Pindborg JJ (1970) Disturbances in tooth formation; etiology. In: Pindborg JJ (ed) Pathology of the dental hard tissues. Munksgaard, Copenhagen, pp 77–133
Pinhasi R, Shaw P, White B, Ogden AR (2006) Morbidity, rickets and long-bone growth in post-medieval Britain—a cross-population analysis. Ann Hum Biol 33(3):372–389. doi:10.1080/03014460600707503
Davit-Beal T, Gabay J, Antoniolli P, Masle-Farquhar J, Wolikow M (2014) Dental complications of rickets in early childhood: case report on 2 young girls. Pediatrics 133(4):e1077–e1081. doi:10.1542/peds.2013-0733
Hallas D, Herman NG, Benichou L, Morales EL, Touchette L (2015) Management of a child with nutritional rickets, multiple cavities, enamel hypoplasia, and reactive attachment disorder. J Pediatr Health Care 29(3):283–288. doi:10.1016/j.pedhc.2014.11.010
Grahnen H, Selander P (1954) The effect of rickets and spasmophilia on the permanent dentition. I. The effect on the teeth. Odontol Revy 5(1):7–26
Nordgarden H, Lima K, Skogedal N, Folling I, Storhaug K, Abrahamsen TG (2012) Dental developmental disturbances in 50 individuals with the 22q11.2 deletion syndrome; relation to medical conditions? Acta Odontol Scand 70(3):194–201. doi:10.3109/00016357.2011.629624
Klingberg G, Dietz W, Oskarsdottir S, Odelius H, Gelander L, Noren JG (2005) Morphological appearance and chemical composition of enamel in primary teeth from patients with 22q11 deletion syndrome. Eur J Oral Sci 113(4):303–311. doi:10.1111/j.1600-0722.2005.00223.x
Ranggard L, Ostlund J, Nelson N, Noren JG (1995) Clinical and histologic appearance in enamel of primary teeth from children with neonatal hypocalcemia induced by blood exchange transfusion. Acta Odontol Scand 53(2):123–128
Ranggard L, Noren JG (1994) Effect of hypocalcemic state on enamel formation in rat maxillary incisors. Scand J Dent Res 102(5):249–253
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Signe Sparre Beck-Nielsen received payment from Pharmacosmos for participation in an expert meeting. Hans Gjørup and Dorte Haubek declare that they have no conflict of interest.
Funding
This work was supported by grants from The Danish Dental Association and The Public Health Dentists Association in Denmark.
Ethical approval
All procedures performed in the present study involving human participants were approved by the Ethics Committee of Southern Denmark (record # M-2678-05) and by the Danish Data Protection Agency (record # 2008-41-2187) and in accordance with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study and/or from their parents.
Rights and permissions
About this article
Cite this article
Gjørup, H., Beck-Nielsen, S.S. & Haubek, D. Craniofacial and dental characteristics of patients with vitamin-D-dependent rickets type 1A compared to controls and patients with X-linked hypophosphatemia. Clin Oral Invest 22, 745–755 (2018). https://doi.org/10.1007/s00784-017-2149-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-017-2149-4