Abstract
Elderly patients with osteoporotic vertebral fractures often experience severe pain that reduces their quality of life (QOL). Calcitonin, a bone resorption inhibitor, has been reported to alleviate pain in such patients; however, few clinical studies have demonstrated this effect. The objective of this study was to compare changes in pain scores, activities of daily living (ADL), QOL, bone resorption, bone mineral density (BMD), and fracture healing among patients with new vertebral fractures who received different treatment modalities. We conducted an open-label, multicenter, randomized, parallel control group study comprising 107 female patients ≥55 years old with acute back pain from vertebral fracture. All subjects received either intramuscular injections of elcatonin, a derivative of calcitonin, or an oral nonsteroidal antiinflammatory drug (NSAID) combined with an active vitamin D3 (VD3) analogue for 6 months. The pain was assessed using a visual analogue scale, and ADL and QOL were assessed using questionnaires. BMD was measured using dual-energy X-ray absorptiometry. A two-tailed significance level of 5% was used. The elcatonin IM group had significantly higher QOL score at 2 weeks and later, and significantly lower VAS and ADL scores than those in the NSAID + VD3 group at 1 month and later. The elcatonin IM group had significantly reduced TRACP-5b levels compared with those in the NSAID + VD3 group at 3 months and later and significantly higher percent changes in BMD than the NSAID + VD3 group. These results suggest that elcatonin significantly alleviated pain, inhibited bone resorption, and improved ADL, QOL, and BMD compared with NSAID + VD3.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoporosis patients with vertebral fractures often develop severe pain that greatly impairs their ability to perform the activities of daily living (ADL). Recovering from this decline to their ADL status before fracture generally requires about 1 year [1, 2]. Moreover, according to a report from the Ministry of Health, Labour and Welfare of Japan, 11.8% of all elderly patients who require long-term nursing care have a history of “fall and fracture,” indicating the importance of preventing vertebral fractures secondary to osteoporosis in elderly individuals [3].
Patients with existing vertebral fractures have also been shown to be at an increased risk for hip fracture or vertebral fractures at new sites [4–6]. One reason for this is that the patient’s activity level becomes lower for a long time because of pain after a fresh vertebral fracture [1], and immobilization may cause muscle weakness [4–7]. Another reason is that both bone formation markers and bone resorption markers increased, and it is believed that bone density decreased in the non-fracture site as the result of the high turnover [8–10].
Although treatment for osteoporosis should be given following the initial vertebral fracture, many patients do not receive such treatment and, as a result, new fractures occur more frequently [11]. This problem highlights the need for early treatment for osteoporotic vertebral fractures. Bisphosphonate treatment for osteoporosis is known to reduce the incidence of vertebral fractures and help prevent additional fractures if started after the first vertebral fracture [12]. In the early stage of vertebral fractures, treatment for alleviating pain and improving the patient’s ADL and quality of life (QOL) should be started concurrently with treatment for osteoporosis [13].
Although calcitonin is recommended as a bone resorption inhibitor by the American Academy of Orthopaedic Surgeons [14], few studies have reported evidence of its pain alleviation, bone resorption-inhibiting, or bone mineral density-increasing effects after fracture. Therefore, the pain alleviation effect of elcatonin, a synthetic derivative of eel calcitonin [15], will be clarified in comparison with a nonsteroidal antiinflammatory drug (NSAID). The bone resorption-inhibiting and bone mineral density (BMD)-increasing effect of elcatonin will be clarified in comparison with the active vitamin D3 (VD3) analogue used widely in Japan regardless of gender, and it seems to be considered that the active form of 25(OH)D is essential while fracture healing [16].
Therefore, the objective of this study was to compare the changes in pain, ADL, QOL, bone resorption, BMD, and fracture healing for patients with new vertebral fractures who were given intramuscular (IM) injections of elcatonin and those who were given an NSAID in combination with a VD3 analogue.
Materials and methods
Study design
This clinical study was an open-label, multicenter, randomized, parallel control group study conducted in Japan between August 24, 2010, and October 31, 2011. Subjects were 107 women ≥55 years of age (mean, 74.7 years; age range, 62–88 years) who were divided into two groups using a sealed envelope method. One group received intramuscular (IM) injections of elcatonin (Elcitonin; Asahi Kasei Pharma Corporation, Tokyo, Japan) at a dose of 20 units weekly (elcatonin IM group). The control group received an oral NSAID (etodolac; 200 mg, twice daily) in combination with an active VD3 analogue (alfacalcidol; 0.5 μg, once daily) (NSAID + VD3 group). Each group received the treatment for 6 months. This study was conducted in accordance with the ethical principles of the Declaration of Helsinki and was approved by the Ethics Committees of Hokkaido University and all other study sites. Voluntary written informed consent was obtained from all patients.
Patients
This study was carried out at the Yoshida Orthopaedics Clinic, Sapporo Orthopaedic Hospital, and Sapporo Orthopaedics and Cardiovascular Hospital. The inclusion criteria for this study were as follows: female patients who were ≥55 years old at the start of treatment; patients who developed acute back pain in the 2 weeks before the first visit to the clinics if they had a new osteoporotic thoracolumbar vertebral fracture confirmed on both X-ray and magnetic resonance imaging (MRI). Concerning MRI examinations, areas with low signal intensity on T1-weighted images and high signal intensity on T2-weighted images were considered as new fracture sites [17].
The exclusion criteria were as follows: secondary osteoporosis; vertebral fracture including fracture of the posterior wall of the vertebral body; history of surgical treatment for fracture of thoracic or lumbar vertebrae; signs of neurological deficit associated with spinal disorders; severe scoliosis; contraindications for any of the study drugs (calcitonin, etodolac, alfacalcidol); infectious spinal diseases; severe kidney, liver, or heart disease; or currently receiving treatment for malignant tumors. No patients had received drug therapy for osteoporosis before or at the time of injury, and the use of other osteoporosis drugs was restricted during the study. No restrictions were placed on physician visits or consultations that would not contribute to the outcomes of the study.
The use of injected and other non-NSAID analgesics, oral NSAIDs in the elcatonin IM group, and elcatonin in the NSAID + VD3 group was prohibited from the initial visit to the completion of the clinical study. However, if suppository NSAIDs were needed, duration of use, method, and dose was recorded. Only rigid or soft body braces, not body casts, were used for initial immobilization of the trunk in this study because patients often refuse to wear body casts after new vertebral fractures. We recommended rigid braces for patients with a severe fracture (semiquantitative grade 3) [18] and soft braces for patients with less severe fracture; however, the patients’ preferences were also considered.
The use of treatments such as thermal therapy, silver spike point therapy, and transdermal therapeutic patches with NSAIDs, including indomethacin, ketoprofen, felbinac, loxoprofen sodium, and diclofenac sodium, was permitted.
Assessments
The primary endpoint of the study was the mean pain score. We assessed pain as follows using a visual analogue scale (VAS): while in a supine position; while sitting; while getting up; while rolling over; while sitting up in bed; and while walking. We assessed ADL and QOL using the Roland Morris Disability Questionnaire (RDQ) [19] and the EuroQOL 5 Dimension (EQ-5D) [20] before the study (baseline) and at 1, 2, and 4 weeks, and at 2, 3, and 6 months after the study had begun. Patients were asked to complete the questionnaires on their own before meeting the doctor.
The secondary endpoints of this study were changes of the bone resorption marker, tartrate-resistant acid phosphatase 5b (TRACP-5b), from baseline to 4 weeks, and at 3 and 6 months after the study had begun, and changes in BMD of the lumbar vertebrae (mean of L2–L4), total hip, and femoral neck of left femur from baseline to 6 months. All centers used a dual-energy X-ray absorptiometry device (Prodigy Advance; GE Healthcare, Madison, WI, USA). However, if there was a vertebral fracture in L2–L4, the concerned vertebrae were excluded from the calculation of the lumbar vertebral BMD value. We collected samples from both groups before meeting the doctor, and before IM injection in the elcatonin IM group.
Fracture healing was defined as a fracture site showing normal signal intensity on T1- and T2-weighted MRI images. Cases in which an area of low intensity or a buildup of vertebral fluid was evident on T2-weighted images were defined as pseudarthrosis [17, 21].
Statistical analysis
For VAS, ADL, and QOL measured values, we conducted a paired t test after the F test between baseline and each observation time point within the drug groups and an unpaired t test after the F test at each observation time point between drug groups. For TRACP-5b and BMD at each points; we conducted a paired t test between baseline and 6 months within the drug groups after two-way factorial analysis of variance (ANOVA). We also evaluated correlation between percent change from baseline to 1, 3, or 6 months of serum concentration of TRACP-5b and percent change from baseline to 6 months of the values of BMD at each parts. A two-tailed significance level of 5% was used for hypothesis testing. SAS (ver. 9.3; SAS Institute Japan, Tokyo, Japan) was used for all statistical analyses.
Results
Patient characteristics
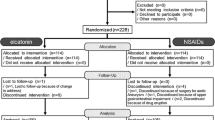
All 107 randomized patients met the criteria for this study (elcatonin IM group, n = 53; NSAID + VD3 group, n = 54) (Fig. 1). The subjects’ characteristics are shown in Table 1. A comparison of new and preexisting vertebral fractures between the groups is shown in Table 2. Rigid and soft body braces were used by 3 and 41 patients, respectively, in the elcatonin IM group, whereas in the NSAID + VD3 group, rigid and soft body braces were used by 5 and 39 patients, respectively. Patients started using braces within 1 week of their initial visit to one of the participating outpatient clinics. However, 9 and 10 patients from the elcatonin IM and NSAID + VD3 groups, respectively, refused to use any body brace. No adverse events were reported during this clinical study.
All patients completed the study. Fracture healing was evident in 51 of 53 patients in the elcatonin IM group and 52 of 54 patients in the NSAID + VD3 group, respectively.
Trends in VAS scores
Single-use NSAID suppositories were prescribed for all patients. Transdermal therapeutic patches containing NSAIDs were also frequently prescribed for patients during the study period. Although the precise usage of the transdermal therapeutic patches was not confirmed, patients reported using them frequently.
Subjects in the elcatonin IM group had significantly higher mean VAS scores for pain in the supine position than subjects in the NSAID + VD3 group at baseline (p = 0.043).
In terms of mean VAS scores for pain in the supine position, sitting, getting up, rolling over, sitting up in bed, and walking, subjects in the elcatonin IM group had significantly lower VAS scores than those in the NSAID + VD3 group at 1 month (supine position, p < 0.01; sitting, rolling over, sitting up in bed, and walking, p < 0.001; getting up, p = 0.001), and later (p < 0.001) (Fig. 2). Significantly lower VAS scores were observed at 1 week (supine position, sitting, getting up, rolling over in elcatonin IM group, and sitting up in bed and walking: p < 0.001; rolling over in NSAID + VD3 group: p = 0.006) and later (p < 0.001) in both groups compared with baseline (Fig. 2).
Regarding mean RDQ scores for ADL, subjects in the elcatonin IM group had significantly lower scores than those in the NSAID + VD3 group at 1 month and later (1 month, p = 0.007; 2 months, p = 0.001; 3 months and later, p < 0.001) (Fig. 3).
Regarding mean EQ-5D scores, subjects in the elcatonin IM group had significantly higher EQ-5D scores than those in the NSAID + VD3 group at 2 weeks and later (2 weeks, p = 0.003; 1 month, p = 0.001; 2 months, p < 0.001; 3 months, p = 0.026; 6 months, p = 0.001) (Fig. 3). Compared to baseline, significant improvements of RDQ and EQ-5D scores were observed at 1 week and later for the elcatonin IM group (1 week, p = 0.001, p < 0.001; 2 weeks and later, p < 0.001, p < 0.001, respectively); and at 2 weeks and later for the NSAID + VD3 group (p < 0.001) (Fig. 3). These results suggest that subjects in the elcatonin IM group experienced significantly less pain at 1 week and later than those in the NSAID + VD3 group based on the RDQ.
Trends in TRACP-5b activity
Regarding mean TRACP-5b levels, subjects in the elcatonin IM group had significantly lower TRACP-5b levels than those in the NSAID + VD3 group at 3 months and later (3 months, p = 0.008; 6 months, p = 0.002) (Fig. 4). Significantly lower TRACP-5b levels were observed at 1 month and later in both groups compared with baseline (p < 0.001) (Fig. 4).
Regarding TRACP-5b percent change, significantly higher absolute values of percent change were observed in the elcatonin IM group compared with the NSAID + VD3 group at 1 month and later (p < 0.001) (Fig. 4).
Based on these results, subjects in the elcatonin IM group had significantly reduced TRACP-5b levels compared with those in the NSAID + VD3 group at 3 months and later.
Trends in BMD
When baseline was compared to 6 months, significant differences were seen in L2–L4 vertebrae, total hip, and femoral neck in the elcatonin IM group (L2–L4 vertebrae, p < 0.001; total hip, p = 0.009; femoral neck, p < 0.001), and only in total hip in the NSAID + VD3 group (L2–L4 vertebrae, p = 0.066; total hip, p = 0.003; femoral neck, p = 0.787) (Table 3).
Percent changes from baseline to 1, 3, or 6 months of serum concentration of TRACP-5b were negatively correlated with percent changes from baseline to 6 months of the values of femoral neck BMD (p = 0.006, r = −0.264; p = 0.0007, r = −0.324; p = 0.0007, r = −0.322, respectively). There were no correlations between percent changes from baseline to all time points of TRACP-5b and percent changes of the values of lumbar or total hip BMD (Table 4).
Discussion
In this study, osteoporosis patients with new vertebral fractures were given IM injections of elcatonin or a combination of NSAID + VD3. Our results demonstrated that elcatonin led to significant improvements in pain alleviation compared with NSAID + VD3 at 1 month and later. Concerning QOL, significant improvements were observed in RDQ scores at 1 month and later, and in EQ-5D scores at 2 weeks and later in the elcatonin IM group. Moreover, TRACP-5b levels were significantly lower at 3 months and later in the elcatonin IM group compared with the NSAID + VD3 group.
Although the incidence of hip fracture in Western European countries is reportedly decreasing [22–24], the number of patients who receive treatment for osteoporosis remains inadequate [11], possibly because of lack of proper knowledge regarding osteoporosis in the general population. Initial fractures, especially highly frequent vertebral fractures, represent good opportunities for starting osteoporosis treatment. During healing of vertebral fractures, whole-body bone metabolism was intensified [10], thereby increasing the risk of hip fracture or vertebral fractures at new sites. The risk of secondary fracture after vertebral fracture is also known to increase at various sites such as the femur, radius, ribs, humerus, and ankle [4]. Therefore, it is essential to start treatment for osteoporosis as soon as possible after a vertebral fracture.
Vertebral fractures secondary to osteoporosis do not exhibit distinct vertebral deformation at the initiation of acute back pain and are therefore frequently overlooked [18]. When an elderly individual presents with acute back pain, doctors should consider the possibility of vertebral or pathological fracture and thus perform multiple imaging sequences, including X-rays and MRI scans. Also, because fractures promote bone metabolism turnover [9, 10, 25], treatment with bone resorption inhibitors should be initiated immediately after fracture to prevent secondary fractures and the progression of osteoporosis. However, using bisphosphonates during fracture healing may induce apoptosis of osteoclasts and suppress bone formation, consequently increasing the risk of prolonged healing [26, 27]. On the other hand, some articles show that bisphosphonates do not influence fracture healing [28, 29]. The effect of bisphosphonates on fracture healing is still controversial. Calcitonin inhibits bone resorption via the disruption of actin rings in osteoclasts, thereby preventing osteoclast apoptosis without significantly suppressing fracture healing [8]. The present study showed that the bone resorption-inhibiting actions of elcatonin are at least equal to or greater than those of VD3, and percent changes of the serum concentration of TRACP-5b showed correlation with the percent change of the value of femoral neck BMD. Thus, elcatonin can effectively suppress systemic turnover after vertebral fracture without prolonging fracture healing. In this 6-month short study that used elcatonin alone, BMD increased by 2.30% in vertebral body, 2.32% in total hip, and 2.48% in femoral neck, and these results exceeded the previous reports from Orimo et al. and Li et al. (1.87% and 1.90% increase in vertebral body, respectively) [30, 31]. In addition, in the study of postmenopausal osteoporotic women with chronic low back pain, combination therapy of elcatonin and risedoronate appeared to be more effective on only QOL but BMD compared with risedronate monotherapy [32]. The reason was considered to be that elcatonin suppressed not the numbers of osteoclasts but their activity, and the number of osteoclasts was thought to not affect bone mineralization [33]. In addition, bone turnover of patients with a new vertebral fracture was higher when compared with women with no fresh fractures, and suppression of only osteoclast activity by elcatonin might effectively increase the BMD of those patients. The maintenance of high activity by analgesics also thought to result in higher gains in the values of BMD. However, even in the NSAID + VD3 group, BMD increased by 0.78% in vertebral body and 2.24% in total hip, and good increases in BMD were observed in all patients of this study. Also, it seemed that as this study was conducted in Hokkaido, a region with plenty of snow, there may be an impact as many of the patients were registered from autumn to winter.
In elderly individuals, being immobilized in bed as the result of severe pain leads to reduced muscle strength, which consequently leads to a greater susceptibility to falls. In this study, reduction of VAS scores was closely related to improving the scores of ADL and QOL. Thus, the results of this study clearly demonstrate that severe pain from vertebral fractures causes decreases in both ADL and QOL. Calcitonin has been reported to inhibit osteoclastic bone resorption and acidification and suppress C-fiber-mediated stimulus-evoked pain [34] and to alleviate pain by increasing blood flow [35]. Although calcitonin has primarily been reported as an effective treatment for alleviating chronic pain [32], our study showed that it alleviated pain on par with NSAIDs both immediately after the start and in the later stages of treatment. Transdermal therapeutic patches were frequently prescribed for and used by patients in both groups during the study period; however, the synergistic effect of elcatonin and transdermal therapeutic patches remains unknown.
Furthermore, considering the ability of elcatonin to inhibit bone resorption and alleviate pain immediately after injury without prolonging fracture healing or causing a decrease in BMD, it should be considered as a suitable candidate for osteoporosis treatment immediately after vertebral fracture, as recommended by the American Academy of Orthopaedic Surgeons [15].
There were some limitations to this study. This study was an open-label trial conducted in general clinical patients, and thus there may have been a bias related to the administration of injections by the medical staff present in the elcatonin IM group. Japanese usually use transdermal therapeutic patches in daily living. Therefore, we could not determine the exact quantity of transdermal therapeutic patches that were used by patients during this study, although we believe that this limitation does not significantly influence the results of this study.
Bone union failure was observed at 6 months in two patients from each group. All four of these patients had a grade 3 severe vertebral fracture as determined by a semiquantitative method [18] and were considered to have severe bone fragility. This result indicated that for such patients, bone anabolic agents such as teriparatide might be more appropriate than elcatonin. Future studies should be conducted with larger study populations to assess the suitability of elcatonin compared to teriparatide after new vertebral fractures.
Conclusion
In patients with new vertebral fractures, elcatonin was shown to alleviate pain as effectively as a combined NSAID + VD3 regimen immediately after starting the treatment, and more effectively in the later stages, thereby improving ADL and QOL. Moreover, even though elcatonin inhibited bone resorption more effectively than NSAID + VD3 and prevented decreases in BMD at other sites, it did not hinder fracture healing.
References
Hagino H, Nakamura T, Fujiwara S, Oeki M, Okano T, Teshima R (2009) Sequential change in the quality of life for patients with incident clinical fracture: a prospective study. Osteoporos Int 20:695–702
Masunari N, Fujiwara S, Nakata Y, Nakashima E, Nakamura T (2007) Historical height loss, vertebral deformity, and health-related quality of life in Hiroshima cohort study. Osteoporos Int 18:1493–1499
Summary of the 2013 National Livelihood Survey by Ministry of Health, Labour and Welfare. http://www.mhlw.go.jp/toukei/saikin/hw/k-tyosa/k-tyosa13/ (2014). Accessed 30 July 2015 (in Japanese)
Klotzbuecher CM, Ross PD, Landsman PB, Abbott TA 3rd, Berger M (2000) Patients with prior fractures have an increased risk of future fractures: a summary of the literature and statistical synthesis. J Bone Miner Res 15:721–739
Hagino H, Sawaguchi T, Endo N, Ito Y, Nakano T, Watanabe Y (2012) The risk of second hip fracture in patients after their first hip fracture. Calcif Tissue Int 90:14–21
Black DM, Arden NK, Palermo L, Pearson J, Cummings SR (1999) Prevalent vertebral deformities predict hip fractures and new vertebral deformities but not wrist fracture. J Bone Miner Res 14:821–828
Matsumoto T, Hoshino M, Tsujio T, Terai H, Namikawa T, Matsumura A, Kato M, Toyoda H, Suzuki A, Takayama K, Takaoka K, Nakamura H (2012) Prognostic factors for reduction of daily living following osteoporotic vertebral fractures. Spine 37:1115–1121
Katae Y, Tanaka S, Sakai A, Nagashima M, Hirasawa H, Nakamura T (2009) Elcatonin injections suppress systemic bone resorption without affecting cortical bone regeneration after drill-hole injuries in mice. J Orthop Res 27:1652–1658
Hedstrom M, Sjoberg K, Svensson J, Brosjo E, Dalen N (2001) Changes in biochemical markers of bone metabolism and BMD during the first year after a hip fracture. Acta Orthop Scand 72:248–251
Ivaska KK, Gerdhem P, Akesson K, Garnero P, Obrant KJ (2007) Effect of fracture on bone turnover markers: a longitudinal study comparing marker levels before and after injury in 113 elderly women. J Bone Miner Res 22:1155–1164
Sakuma M, Endo N, Oinuma T, Endo E, Yazawa T, Watanabe K, Watanabe S (2008) Incidence and outcome of osteoporotic fractures in 2004 in Sado City, Niigata Prefecture, Japan. J Bone Miner Metab 26:373–378
Watts NB, Josse RG, Hamdy RC, Hughes RA, Manhart MD, Barton I, Calligeros D, Felsenberg D (2003) Risedronate prevents new vertebral fractures in postmenopausal women at high risk. J Clin Endocrinol Metab 88:542–549
Knopp-Sihota JA, Newburn-Cook CV, Homik J, Cummings GG, Voaklander D (2012) Calcitonin for treating acute and chronic pain of recent and remote osteoporotic vertebral compression fractures: a systematic review and meta-analysis. Osteoporos Int 23:17–38
McGuire R (2010) Treating spinal compression fractures. AAOS October 2010 issue. http://www.aaos.org/news/aaosnow/oct10/cover1.asp. Accessed 16 Feb 2015
Morikawa T, Munekata E, Sakakibara S, Noda T, Otani M (1976) Synthesis of eel calcitonin and (asu1,7)-eel-calcitonin: contribution of the disulfide bond to the hormonal activity. Experientia (Basel) 32:1104–1106
Meller Y, Kestenbaum RS, Shany S, Galinky D, Zuili I, Yankovitch N, Giat J, Confoti A, Torok G (1985) Parathormone, calcitonin, and vitamin D metabolites during normal fracture healing in geriatric patients. Clin Orthop Relat Res 199:272–279
Baur A, Stabler A, Arbogast S, Duerr HR, Bartl R, Reiser M (2002) Acute osteoporotic and neoplastic vertebral compression fractures: fluid sign at MR imaging 1. Radiology 225:730–735
Genant HK, Wu CY, van Kuijk C, Nevitt MC (1993) Vertebral fracture assessment using a semiquantitative technique. J Bone Miner Res 8:1137–1148
Suzukamo Y, Fukuhara S, Kikuchi S, Konno S, Roland M, Iwamoto Y, Nakamura T, Committee on Science Project, Japanese Orthopaedic Association (2003) Validation of the Japanese version of the Roland–Morris Disability Questionnaire. J Orthop Sci 8:543–548
Brooks R, EuroQoL Group (1990) A new facility for the measurement of health-related quality of life. Health Policy 16:199–208
Hasegawa K, Homma T, Uchiyama S, Takahashi HE (1997) Osteosynthesis without instrumentation for vertebral pseudarthrosis in osteoporotic spine. J Bone Joint Surg Br 79:452–456
Støen RO, Nordsletten L, Meyer HE, Frihagen JF, Falch JA, Lofthus CM (2012) Hip fracture incidence is decreasing in the high incidence area of Oslo, Norway. Osteoporos Int 23:2527–2534
Fisher A, Martin J, Srikusalanukul W, Davis M (2010) Bisphosphonate use and hip fracture epidemiology: ecologic proof from the contrary. Clin Interv Aging 5:355–362
Brauer CA, Coca-Perraillon M, Cutler DM, Rosen AB (2009) Incidence and mortality of hip fractures in the United States. JAMA 302:1573–1579
Hoesel LM, Wehr U, Rambeck WA, Schnettler R, Heiss C (2005) Biochemical bone markers are useful to monitor fracture repair. Clin Orthop Relat Res 440:226–232
Solomon DH, Hochberg MC, Mogun HS, Schneeweiss S (2009) The relation between bisphosphonate use and non-union of fractures of the humerus in older adults. Osteoporos Int 20:895–901
Cao Y, Mori S, Mashiba T, Westmore MS, Westmore MS, Ma L, Sato M, Akiyama T, Shi L, Komatsubara S, Miyamoto K, Norimatsu H (2002) Raloxifene, estrogen, and alendronate affect the processes of fracture repair differently in ovariectomized rats. J Bone Miner Res 17:2237–2246
Kim TY, Ha YC, Kang BJ, Lee YK, Koo KH (2012) Does early administration of bisphosphonate affect fracture healing in patients with intertrochanteric fractures? J Bone Joint Surg Br 94:956–960
Colon-Emeric C, Nordsletten L, Olson S, Major N, Boonen S, Haentjens P, Mesenbrink P, Magaziner J, Adachi J, Lyles KW, Hyldstrup L, Bucci-Rechtweg C, Recknor C, HORIZON Recurrent Fracture Trial (2011) Association between the timing of zoledronic acid infusion and hip fracture healing. Osteoporos Int 22:2329–2336
Orimo H, Morii H, Inoue T, Yamamoto K, Minaguchi H, Ishii Y, Murota K, Fujimaki E, Watsanabe R, Harata S, Honjo H, Fujita T (1996) Effect of elcatonin on involutional osteoporosis. J Bone Miner Metab 14:73–78
Li Y, Xuan M, Wang B, Yang J, Zhang H, Zhang XZ, Li PQ, Tong JC (2013) Comparison of parathyroid hormone (1-34) and elcatonin in postmenopausal women with osteoporosis: an 18-month randomized multicenter controlled trial in China. Chin Med J 126:457–463
Hongo M, Miyakoshi N, Kasukawa Y, Ishikawa Y, Shimada Y (2015) Additive effect of elcatonin to risedronate for chronic back pain and quality of life in postmenopausal women with osteoporosis: a randomized controlled trial. J Bone Miner Metab 33:432–439
Boskey AL, Gelb BD, Pourmand E, Kudrashov V, Doty SB, Spevak L, Schaffler MB (2009) Ablation of cathepsin K activity in the young mouse causes hypermineralization of long bone and growth plates. Calcif Tissue Int 84:229–239
Yoshimura T, Ito A (2010) Calcitonin. J Pharmacol Sci 135:40–41 (in Japanese)
Yoshimura T, Ito A, Saito SY, Takeda M, Kuriyama H, Ishikawa T (2012) Calcitonin ameliorates enhanced arterial contractility after chronic constriction injury of the sciatic nerve in rats. Fundam Clin Pharmacol 26:315–321
Acknowledgements
Asahi Kasei Pharma funded this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Shinya Tanaka has received lecture fees from Asahi Kasei Pharma Corporation, Chugai Pharmaceutical, Daiichi Sankyo, DePuy Synthes Japan, Eisai, Eli Lilly Japan, Taisho Toyama Pharmaceutical, and Teijin Pharma. Akira Yoshida has received lecture fees from Asahi Kasei Pharma, Chugai Pharmaceutical, and Eisai. Manabu Ito has received lecture fees from Asahi Kasei Pharma. Shinjiro Kono, Tadanori Oguma, and Kyoichi Hasegawa have no conflicts of interest to declare.
About this article
Cite this article
Tanaka, S., Yoshida, A., Kono, S. et al. Effectiveness of elcatonin for alleviating pain and inhibiting bone resorption in patients with osteoporotic vertebral fractures. J Bone Miner Metab 35, 544–553 (2017). https://doi.org/10.1007/s00774-016-0791-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-016-0791-6